Noise-Reducing Fabric Electrode for ECG Measurement
Abstract
:1. Introduction
- We have discovered that elements made of fabric can play the same role as general passive elements;
- We show that the impedance of the human body and passive devices made of conductive fabric can work as a filter;
- We show that changing the internal structure of the conductive fabric is effective in noise reduction.
2. Related Works
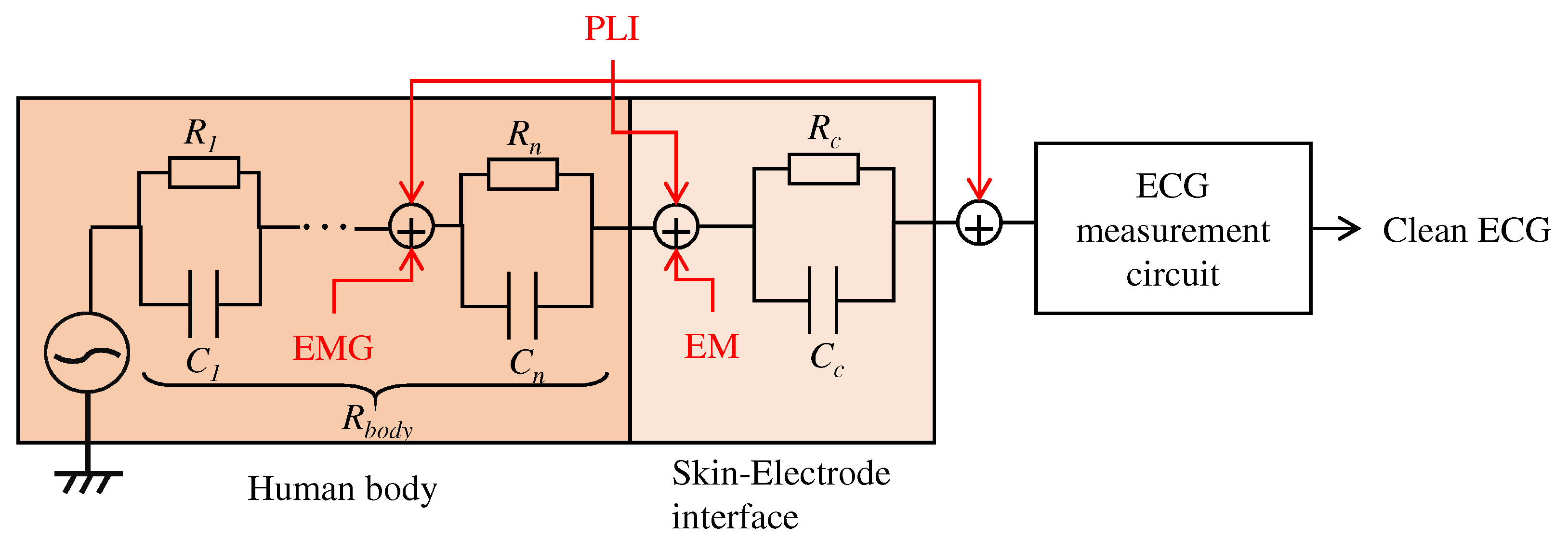
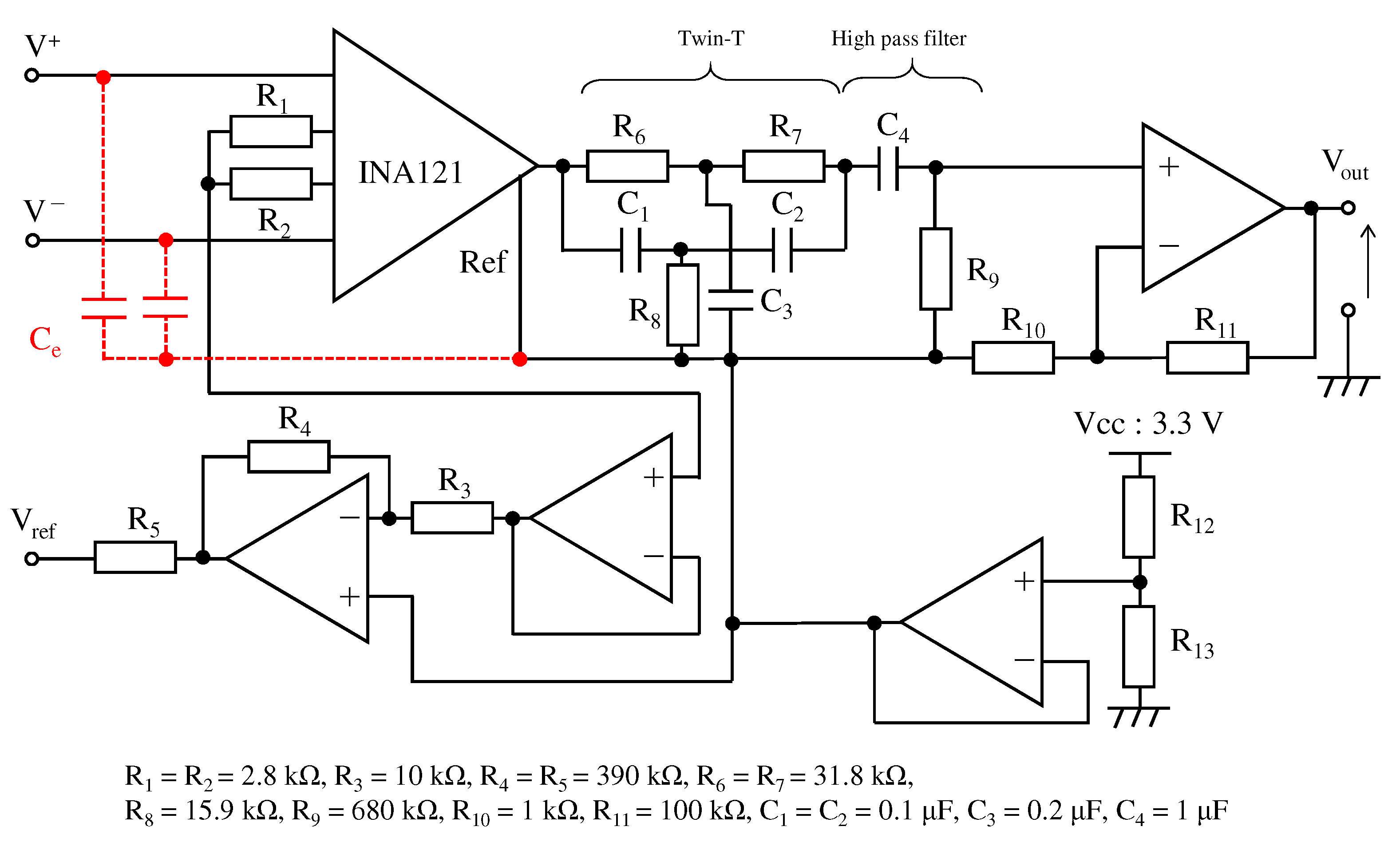
3. An Analog Filter by the Conductive Fabric
3.1. Requirements for Analog Filter
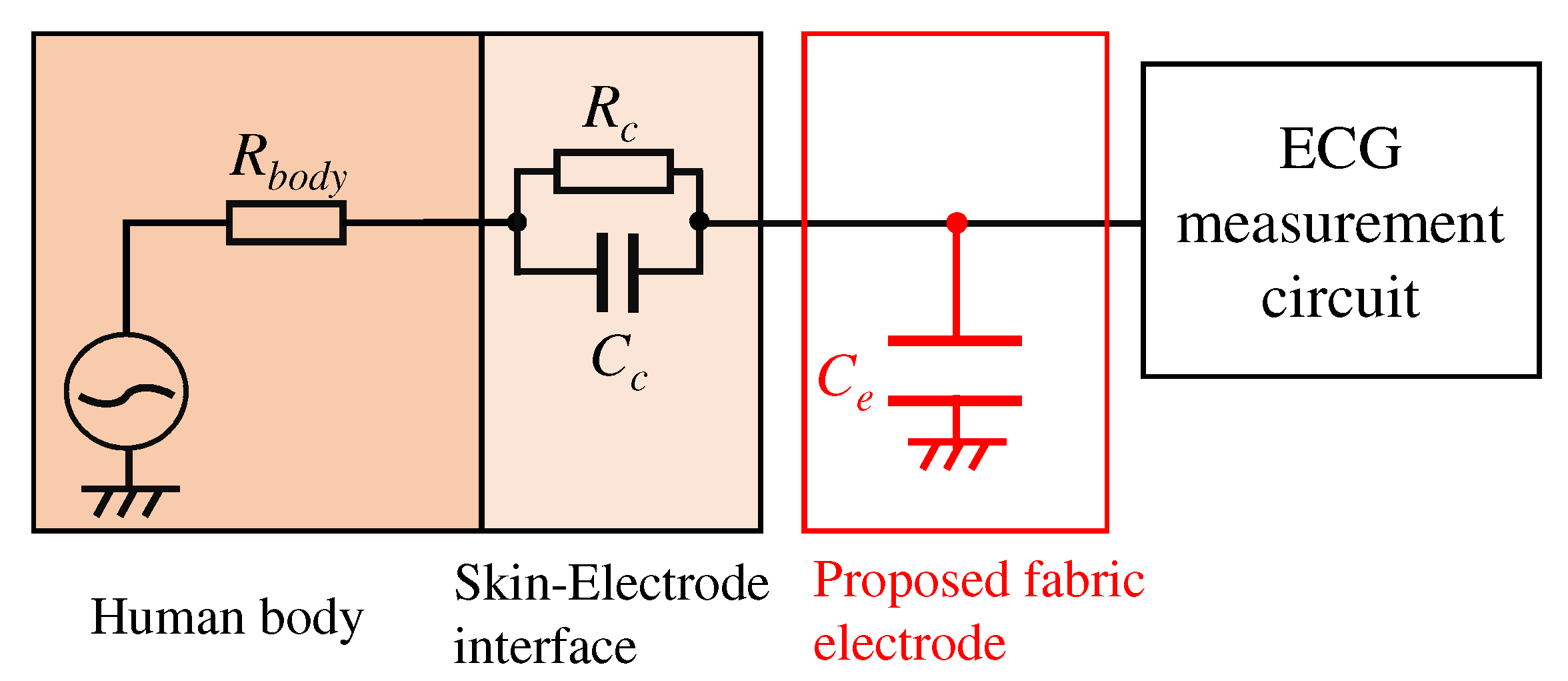
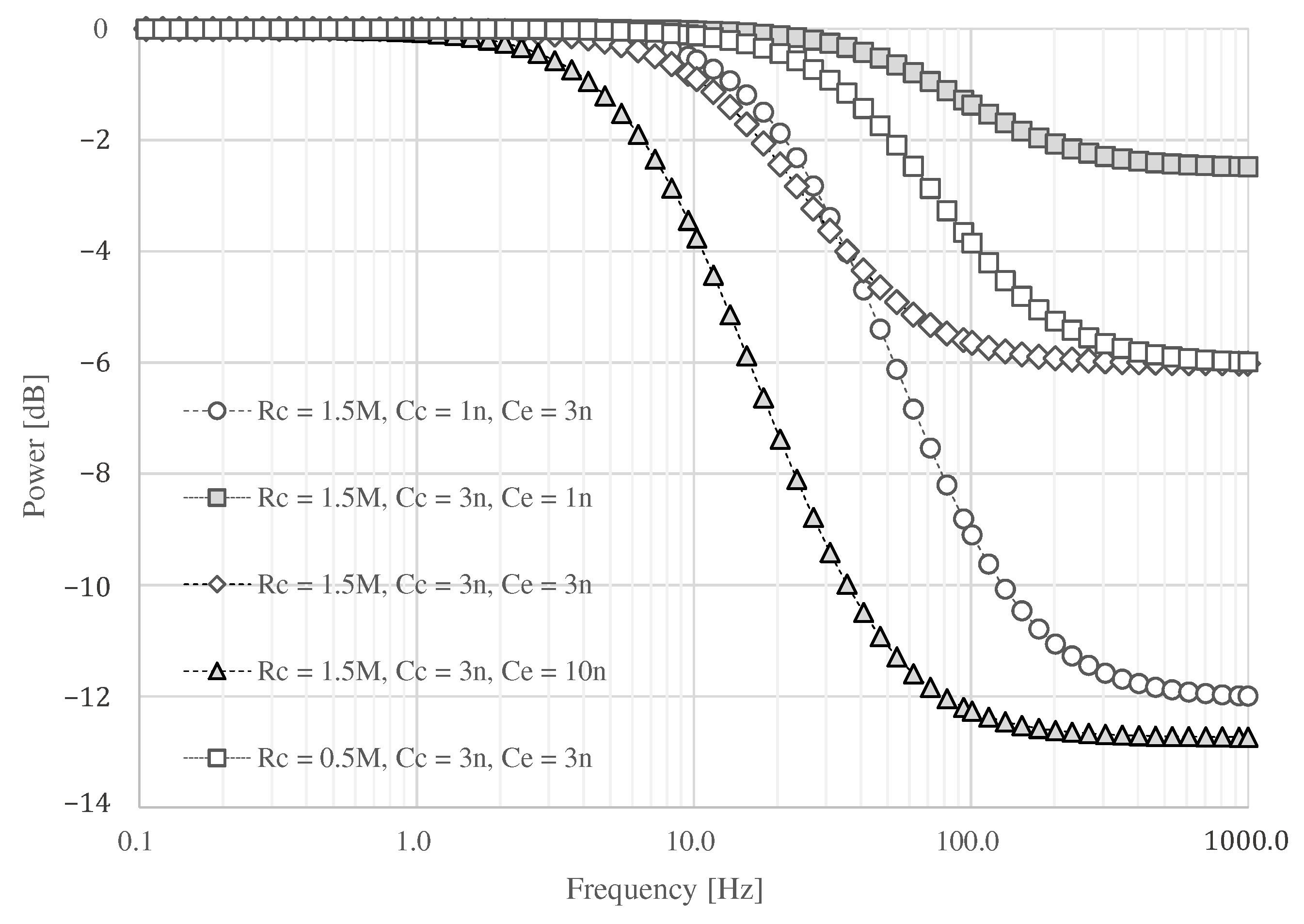
3.2. Impedance Constituting the Textile Filter
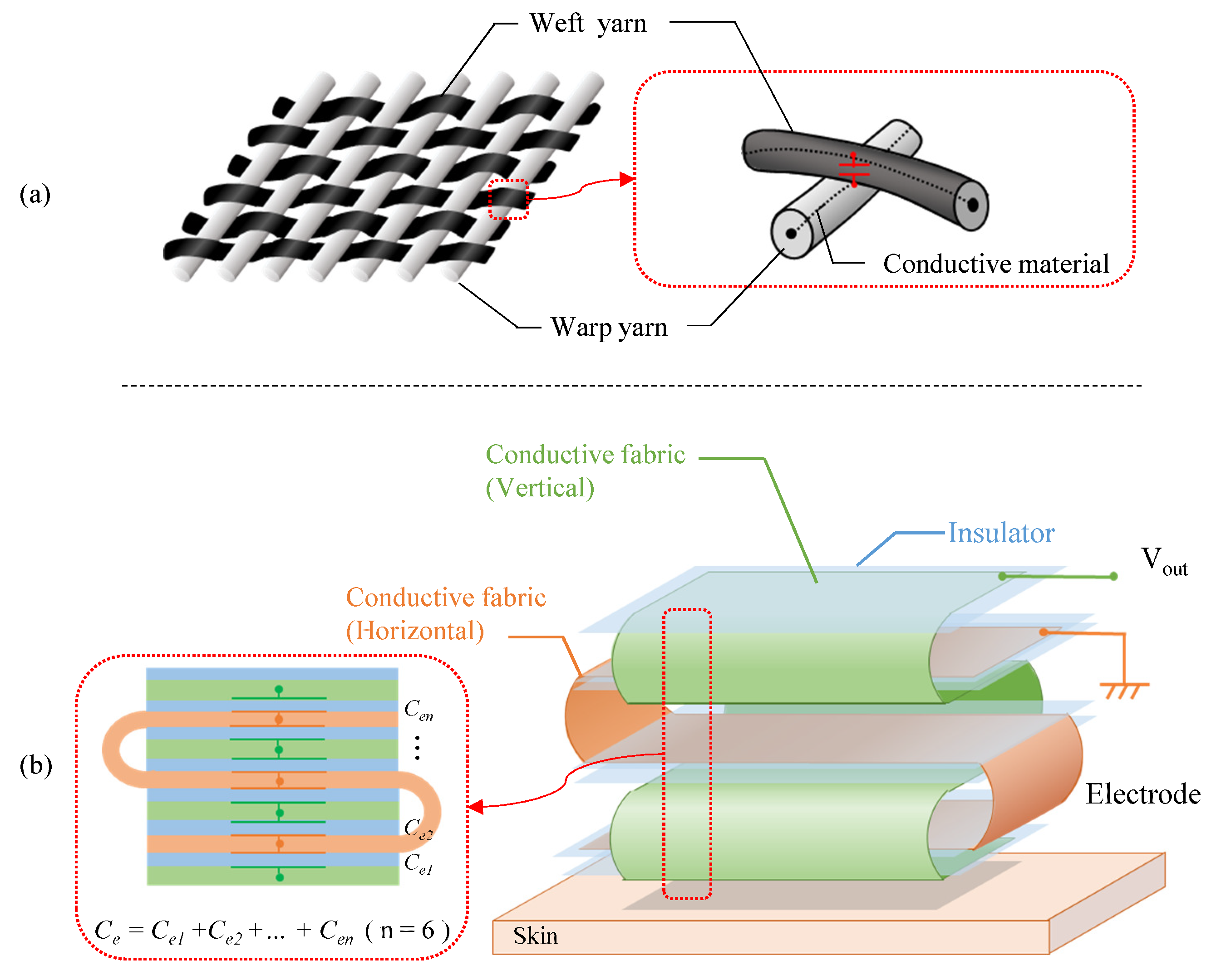
3.3. Fabric Structure
4. Evaluation of Textile Filter
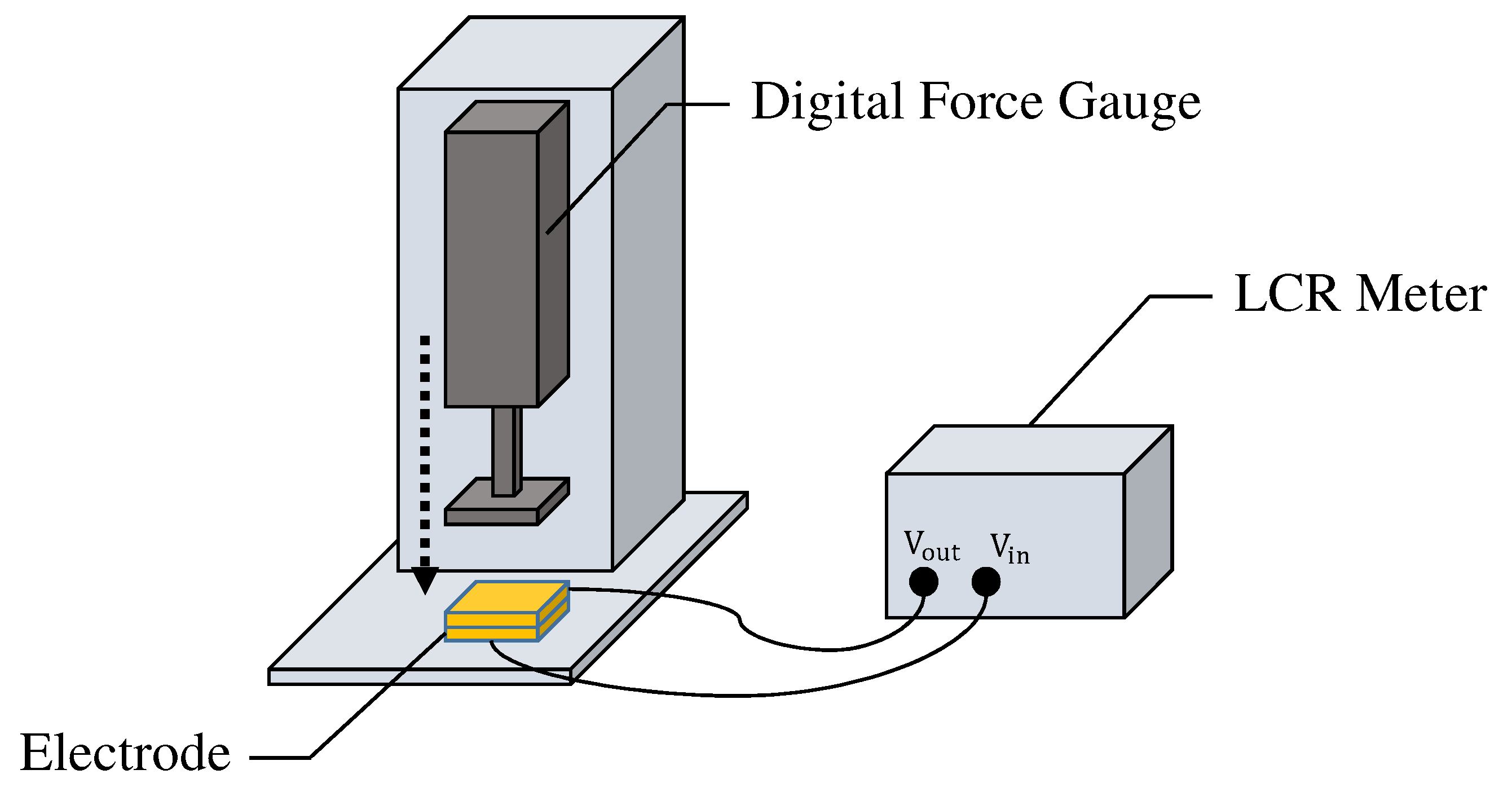
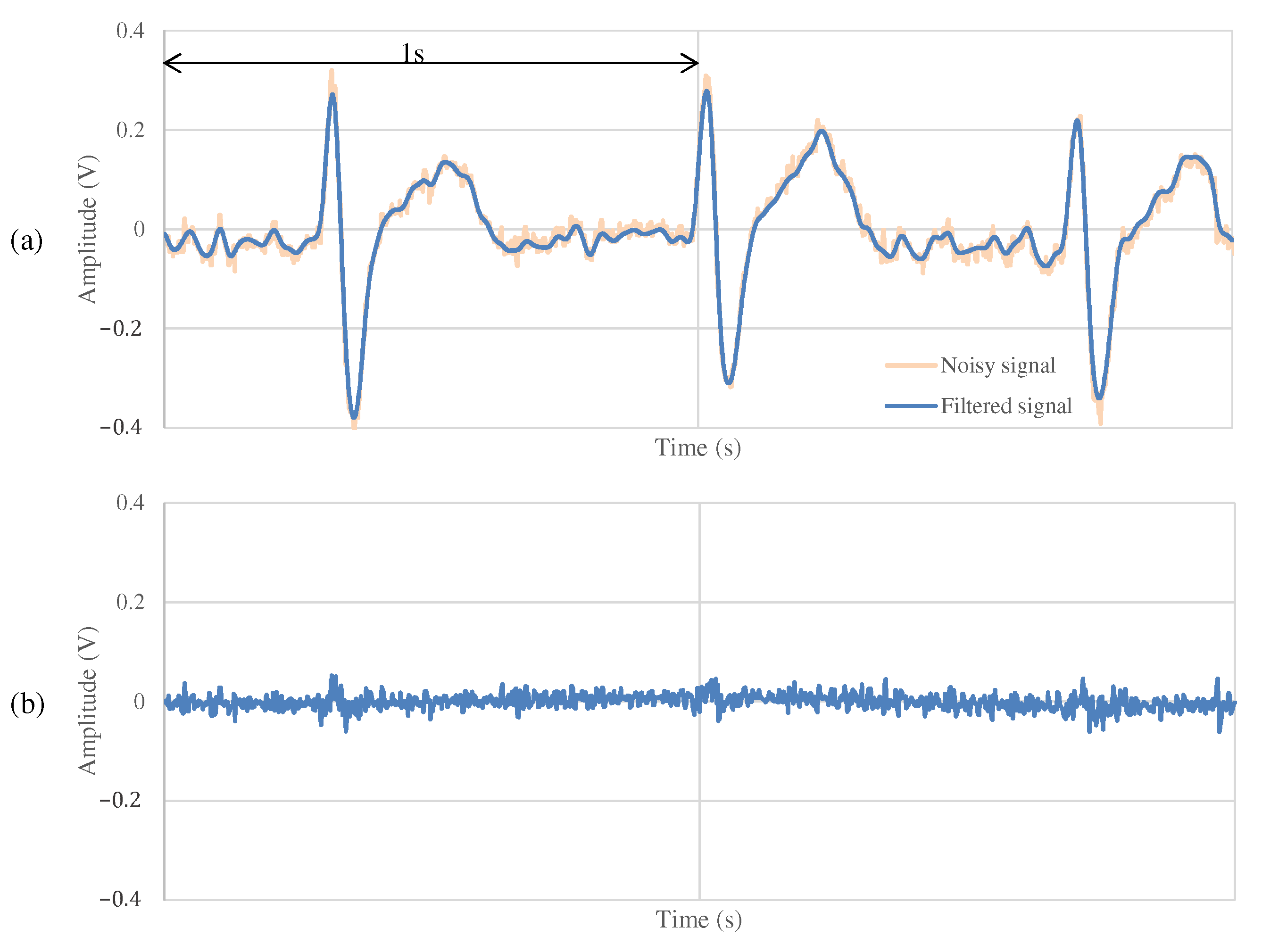
4.1. Settings
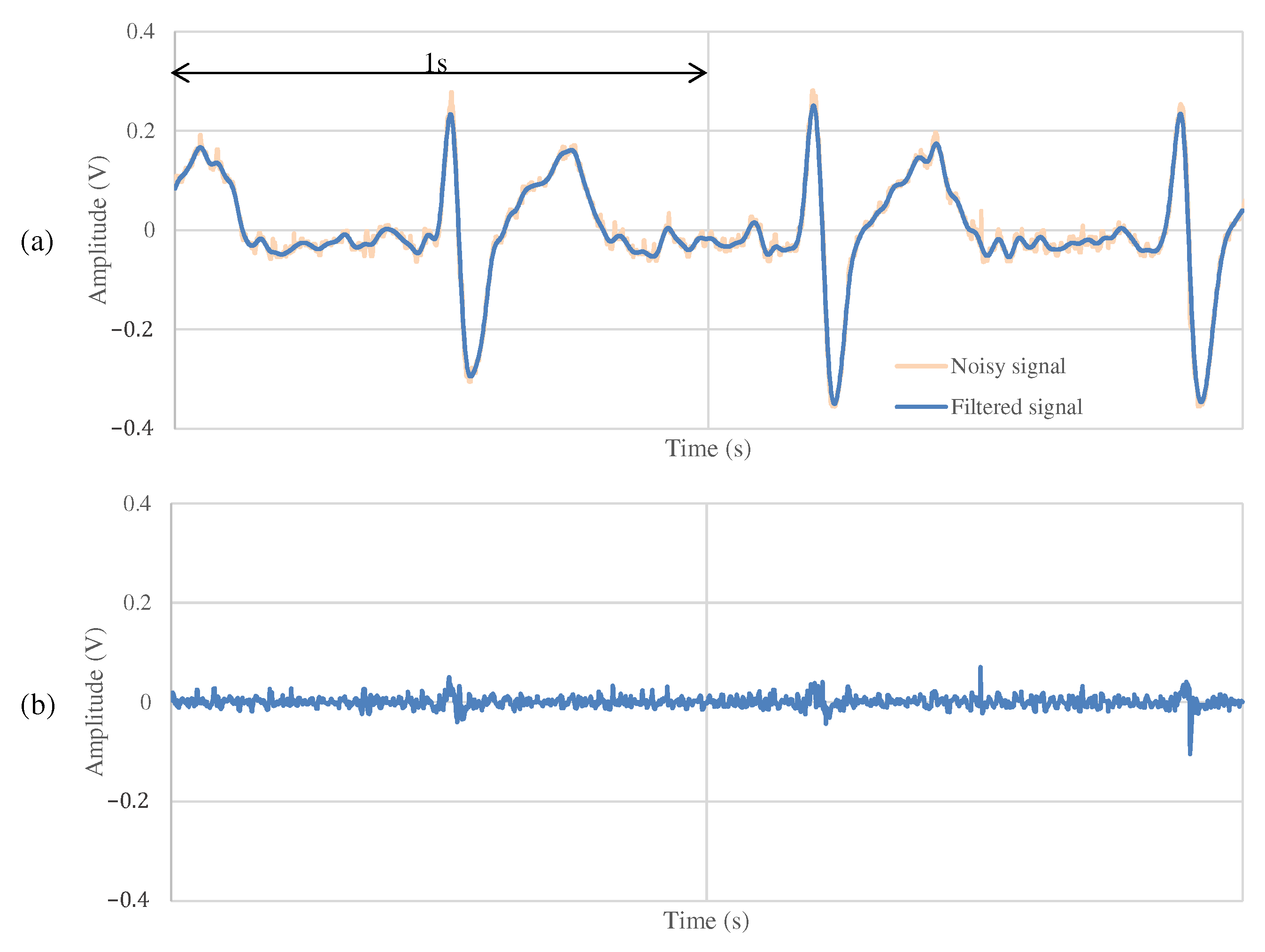
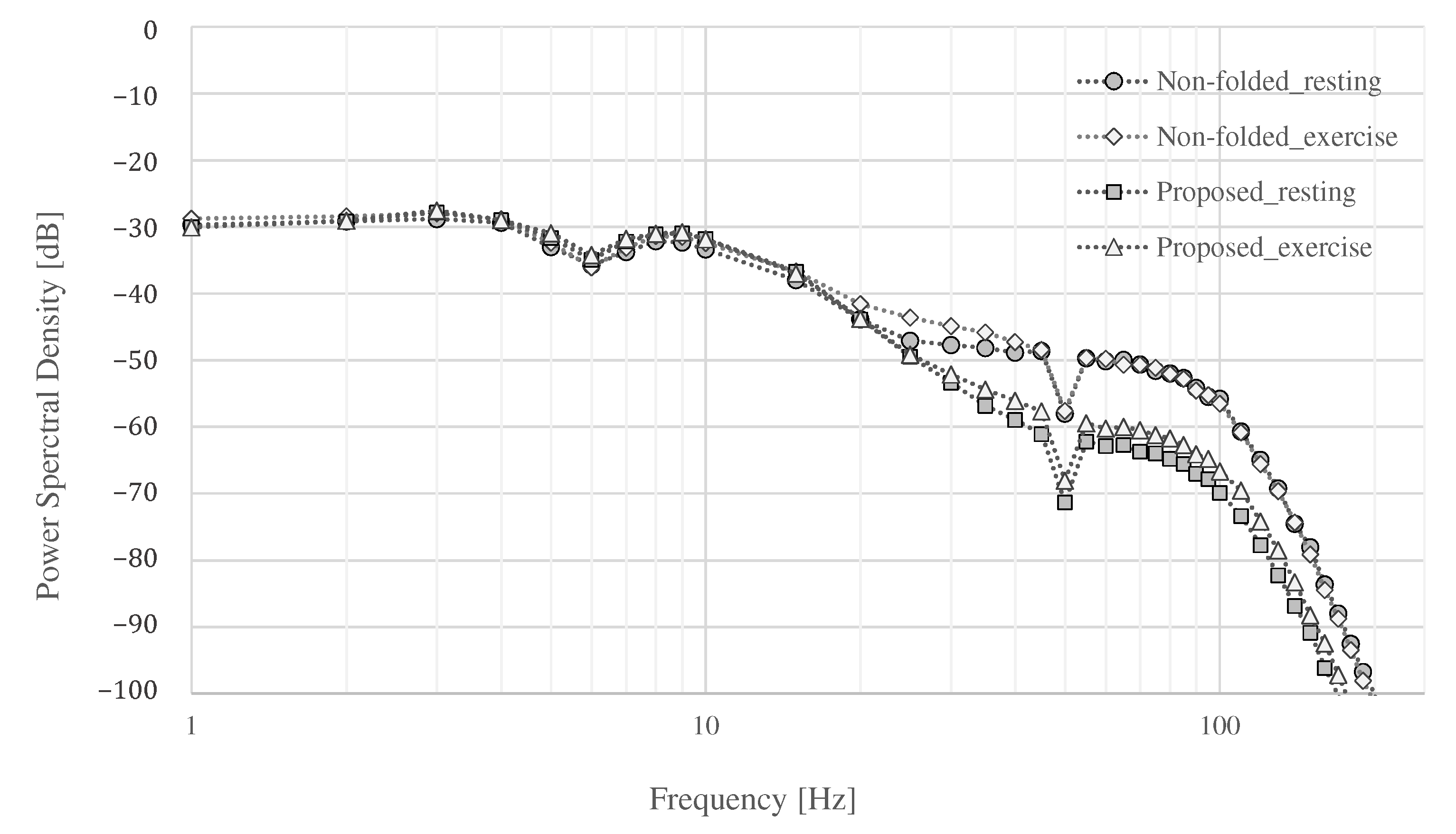
4.2. The Measurement Result during Resting
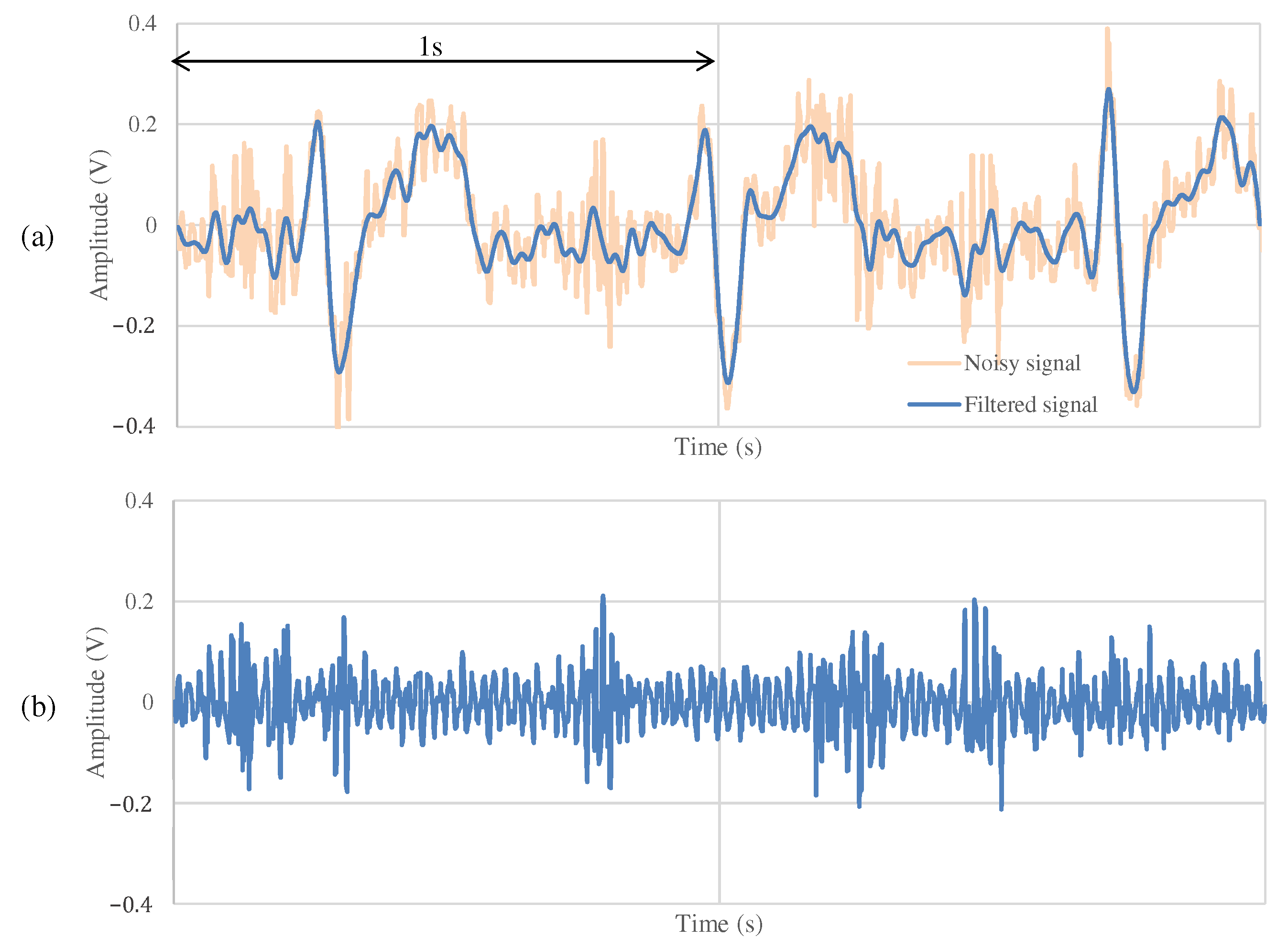
4.3. The Measurement Results during Exercise
4.4. Signal Quality Comparison with Ag/AgCl Electrode
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Puurtinen, M.M.; Komulainen, S.M.; Kauppinen, P.K.; Malmivuo, J.A.; Hyttinen, J.A. Measurement of noise and impedance of dry and wet textile electrodes, and textile electrodes with hydrogel. In Proceedings of the International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 30 August–3 September 2006; pp. 6012–6015. [Google Scholar]
- Apandi, Z.F.M.; Ikeura, R.; Hayakawa, S.; Tsutsumi, S. An Analysis of the Effects of Noisy Electrocardiogram Signal on Heartbeat Detection Performance. Bioengineering 2020, 7, 53. [Google Scholar] [CrossRef] [PubMed]
- Terada, T.; Toyoura, M.; Sato, T.; Mao, X. Functional Fabric Pattern—Examining the Case of Pressure Detection and Localization. IEEE Trans. Ind. Electron. 2018, 66, 8224–8234. [Google Scholar] [CrossRef]
- Muzammal, M.; Talat, R.; Sodhro, A.H.; Pirbhulal, S. A multi-sensor data fusion enabled ensemble approach for medical data from body sensor networks. Inf. Fusion 2020, 53, 155–164. [Google Scholar] [CrossRef]
- Komensky, T.; Jurcisin, M.; Ruman, K.; Kovac, O.; Laqua, D.; Husar, P. Ultra-wearable capacitive coupled and common electrode-free ECG monitoring system. In Proceedings of the IEEE International Conference of the Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 1594–1597. [Google Scholar]
- Merritt, C.R.; Nagle, H.T.; Grant, E. Fabric-based active electrode design and fabrication for health monitoring clothing. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 274–280. [Google Scholar] [CrossRef] [Green Version]
- Spanò, E.; Di Pascoli, S.; Iannaccone, G. Low-power wearable ECG monitoring system for multiple-patient remote monitoring. IEEE Sens. J. 2016, 16, 5452–5462. [Google Scholar] [CrossRef]
- Paul, G.; Torah, R.; Beeby, S.; Tudor, J. Novel active electrodes for ECG monitoring on woven textiles fabricated by screen and stencil printing. Sens. Actuators Phys. 2015, 221, 60–66. [Google Scholar] [CrossRef]
- Celik, N.; Manivannan, N.; Strudwick, A.; Balachandran, W. Graphene-enabled electrodes for electrocardiogram monitoring. Nanomaterials 2016, 6, 156. [Google Scholar] [CrossRef] [Green Version]
- Yapici, M.K.; Alkhidir, T.E. Intelligent Medical Garments with Graphene-Functionalized Smart-Cloth ECG Sensors. Sensors 2017, 17, 875. [Google Scholar] [CrossRef] [Green Version]
- Lam, C.L.; Saleh, S.M.; Yudin, M.B.M.; Harun, F.K.; Sriprachuabwong, C.; Tuantranont, A.; Wicaksono, D.H. Graphene Ink-Coated Cotton Fabric-Based Flexible Electrode for Electrocardiography. In Proceedings of the International Conference on Instrumentation, Communications, Information Technology, and Biomedical Engineering, Bandung, Indonesia, 6–7 November 2017; pp. 73–75. [Google Scholar]
- Reyes, B.A.; Posada-Quintero, H.F.; Bales, J.R.; Clement, A.L.; Pins, G.D.; Swiston, A.; Riistama, J.; Florian, J.P.; Shykoff, B.; Qin, M.; et al. Novel electrodes for underwater ECG monitoring. IEEE Trans. Biomed. Eng. 2014, 61, 1863–1876. [Google Scholar] [CrossRef]
- Weder, M.; Hegemann, D.; Amberg, M.; Hess, M.; Boesel, L.F.; Abächerli, R.; Meyer, V.R.; Rossi, R.M. Embroidered electrode with silver/titanium coating for long-term ECG monitoring. Sensors 2015, 15, 1750–1759. [Google Scholar] [CrossRef] [Green Version]
- An, X.; Stylios, G. A Hybrid Textile Electrode for Electrocardiogram (ECG) Measurement and Motion Tracking. Materials 2018, 11, 1887. [Google Scholar] [CrossRef] [Green Version]
- Taji, B.; Shirmohammadi, S.; Groza, V.; Bolic, M. An ECG monitoring system using conductive fabric. In Proceedings of the IEEE International Symposium on Medical Measurements and Applications, Gatineau, QC, Canada, 4–5 May 2013; pp. 309–314. [Google Scholar] [CrossRef]
- Rattfält, L.; Lindén, M.; Hult, P.; Berglin, L.; Ask, P. Electrical characteristics of conductive yarns and textile electrodes for medical applications. Med. Biol. Eng. Comput. 2007, 45, 1251–1257. [Google Scholar] [CrossRef]
- Sinha, S.; Alamer, F.A.; Woltornist, S.J.; Noh, Y.; Chen, F.; McDannald, A.; Allen, C.; Daniels, R.; Deshmukh, A.A.; Jain, M.; et al. Graphene and Poly(3,4-ethylene dioxythiophene): Poly(4-styrene sulfonate) on Nonwoven Fabric as a Room Temperature Metal and Its Application as Dry Electrodes for Electrocardiography. ACS Appl. Mater. Interfaces 2019, 11, 32339–32345. [Google Scholar] [CrossRef]
- Wu, W.; Pirbhulal, S.; Sangaiah, A.K.; Mukhopadhyay, S.C.; Li, G. Optimization of signal quality over comfortability of textile electrodes for ECG monitoring in fog computing based medical applications. Future Gener. Comput. Syst. 2018, 86, 515–526. [Google Scholar] [CrossRef]
- Xiao, X.; Pirbhulal, S.; Dong, K.; Wu, W.; Mei, X. Performance Evaluation of Plain Weave and Honeycomb Weave Electrodes for Human ECG Monitoring. J. Sensors 2017, 2017, 7539840:1–7539840:13. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Chen, X.; Cao, L.; Zhang, C.; Tang, C.; Li, E.; Feng, X.; Liang, H. Textile-based ECG acquisition system with capacitively coupled electrodes. Trans. Inst. Meas. Control. 2017, 39, 141–148. [Google Scholar] [CrossRef]
- Lee, J.; Heo, J.; Lee, W.; Lim, Y.; Kim, Y.; Park, K. Flexible capacitive electrodes for minimizing motion artifacts in ambulatory electrocardiograms. Sensors 2014, 14, 14732–14743. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.M.; Sim, K.S.; Kim, K.K.; Lim, Y.G.; Park, K.S. Thin and flexible active electrodes with shield for capacitive electrocardiogram measurement. Med. Biol. Eng. Comput. 2010, 48, 447–457. [Google Scholar] [CrossRef]
- Takano, M.; Yamagishi, S.; Ohmuta, T.; Fukuoka, Y.; Ueno, A. Non-contact simultaneous measurements of electrocardiogram and respiratory movements using capacitive sheet electrodes. Adv. Biomed. Eng. 2017, 6, 28–36. [Google Scholar] [CrossRef] [Green Version]
- Ozkan, H.; Ozhan, O.; Karadana, Y.; Gulcu, M.; Macit, S.; Husain, F. A Portable Wearable Tele-ECG Monitoring System. IEEE Trans. Instrum. Meas. 2019, 69, 173–182. [Google Scholar] [CrossRef]
- Chi, Y.M.; Jung, T.P.; Cauwenberghs, G. Dry-contact and noncontact biopotential electrodes: Methodological review. IEEE Rev. Biomed. Eng. 2010, 3, 106–119. [Google Scholar] [CrossRef] [Green Version]
- Kannaian, T.; Neelaveni, R.; Thilagavathi, G. Design and development of embroidered textile electrodes for continuous measurement of electrocardiogram signals. J. Ind. Text. 2013, 42, 303–318. [Google Scholar] [CrossRef]
- Welch, P. The use of fast Fourier transform for the estimation of power spectra: a method based on time averaging over short, modified periodograms. IEEE Trans. Audio Electroacoust. 1967, 15, 70–73. [Google Scholar] [CrossRef] [Green Version]






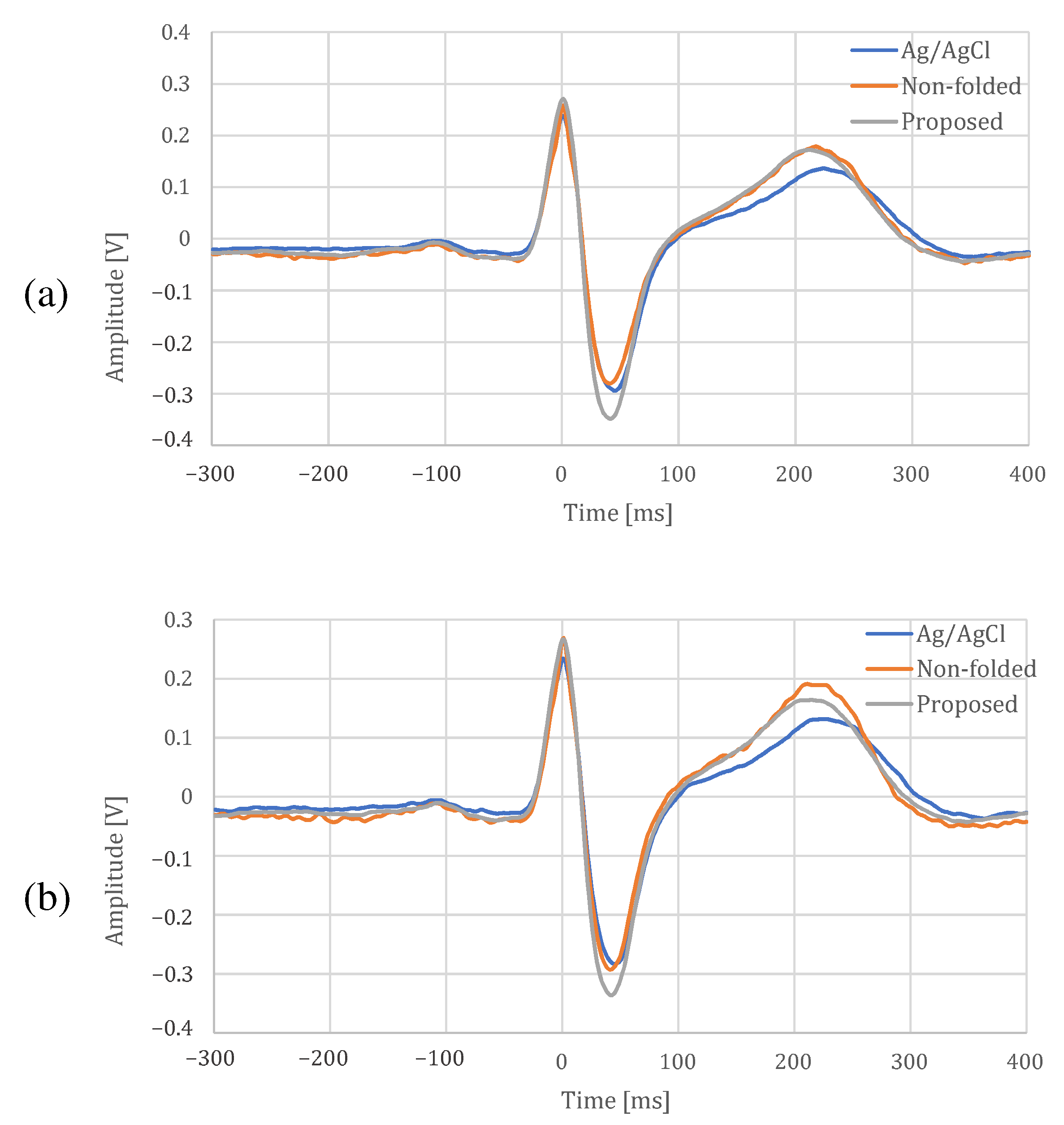
| Parameter | Ag/AgCl | Non-Folded | Proposed | |
|---|---|---|---|---|
| Cross-correlation index | (unitless) | — | 0.980 | 0.988 |
| Temporal measures of HRV | meanNN(ms) | 729.47 | 740.29 | 701.88 |
| SDNN(ms) | 50.352 | 33.434 | 27.941 | |
| RMSSD(ms) | 31.712 | 30.151 | 20.256 | |
| NN50(ms) | 25 | 20 | 2 | |
| Spectral measures of HRV | LF () | 367.27 | 259.33 | 291.41 |
| HF () | 527.88 | 188.06 | 124.63 | |
| Total () | 1151.20 | 628.23 | 498.12 | |
| LF/HF (unitless) | 0.696 | 1.379 | 2.338 | |
| Peak-to-peak amplitude | (V) | 0.545 ± 0.040 | 0.587 ± 0.050 | 0.626 ± 0.042 |
| Parameter | Ag/AgCl | Non-Folded | Proposed | |
|---|---|---|---|---|
| Cross-correlation index | (unitless) | — | 0.974 | 0.988 |
| Temporal measures of HRV | meanNN(ms) | 716.47 | 722.22 | 684.02 |
| SDNN(ms) | 42.514 | 33.083 | 27.719 | |
| RMSSD(ms) | 32.563 | 29.722 | 26.176 | |
| NN50(ms) | 29 | 24 | 17 | |
| Spectral measures of HRV | LF () | 384.62 | 193.73 | 123.57 |
| HF () | 324.55 | 212.31 | 126.47 | |
| Total () | 1010.30 | 518.33 | 298.14 | |
| LF/HF (unitless) | 1.185 | 0.913 | 0.977 | |
| Peak-to-peak amplitude | (V) | 0.541 ± 0.043 | 0.625 ± 0.068 | 0.627 ± 0.047 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terada, T.; Toyoura, M.; Sato, T.; Mao, X. Noise-Reducing Fabric Electrode for ECG Measurement. Sensors 2021, 21, 4305. https://doi.org/10.3390/s21134305
Terada T, Toyoura M, Sato T, Mao X. Noise-Reducing Fabric Electrode for ECG Measurement. Sensors. 2021; 21(13):4305. https://doi.org/10.3390/s21134305
Chicago/Turabian StyleTerada, Takamasa, Masahiro Toyoura, Takahide Sato, and Xiaoyang Mao. 2021. "Noise-Reducing Fabric Electrode for ECG Measurement" Sensors 21, no. 13: 4305. https://doi.org/10.3390/s21134305
APA StyleTerada, T., Toyoura, M., Sato, T., & Mao, X. (2021). Noise-Reducing Fabric Electrode for ECG Measurement. Sensors, 21(13), 4305. https://doi.org/10.3390/s21134305






