Dimensionally Stable Anode Based Sensor for Urea Determination via Linear Sweep Voltammetry
Abstract
1. Introduction
2. Materials and Methods
2.1. Electrode Preparation
2.2. Sample Preparation
2.3. Physicochemical Characterizations
2.4. Electrochemical Characterizations
3. Results and Discussion
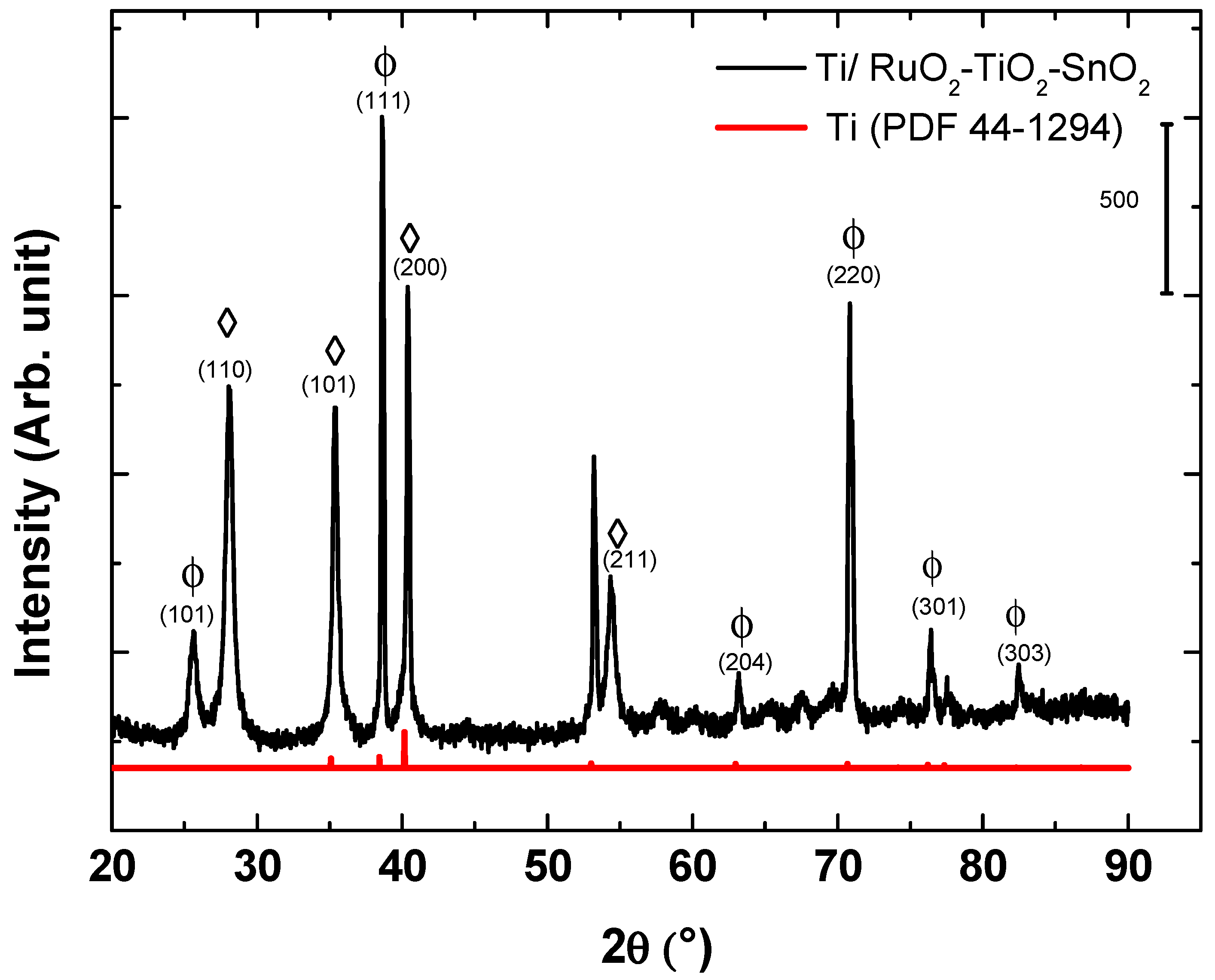
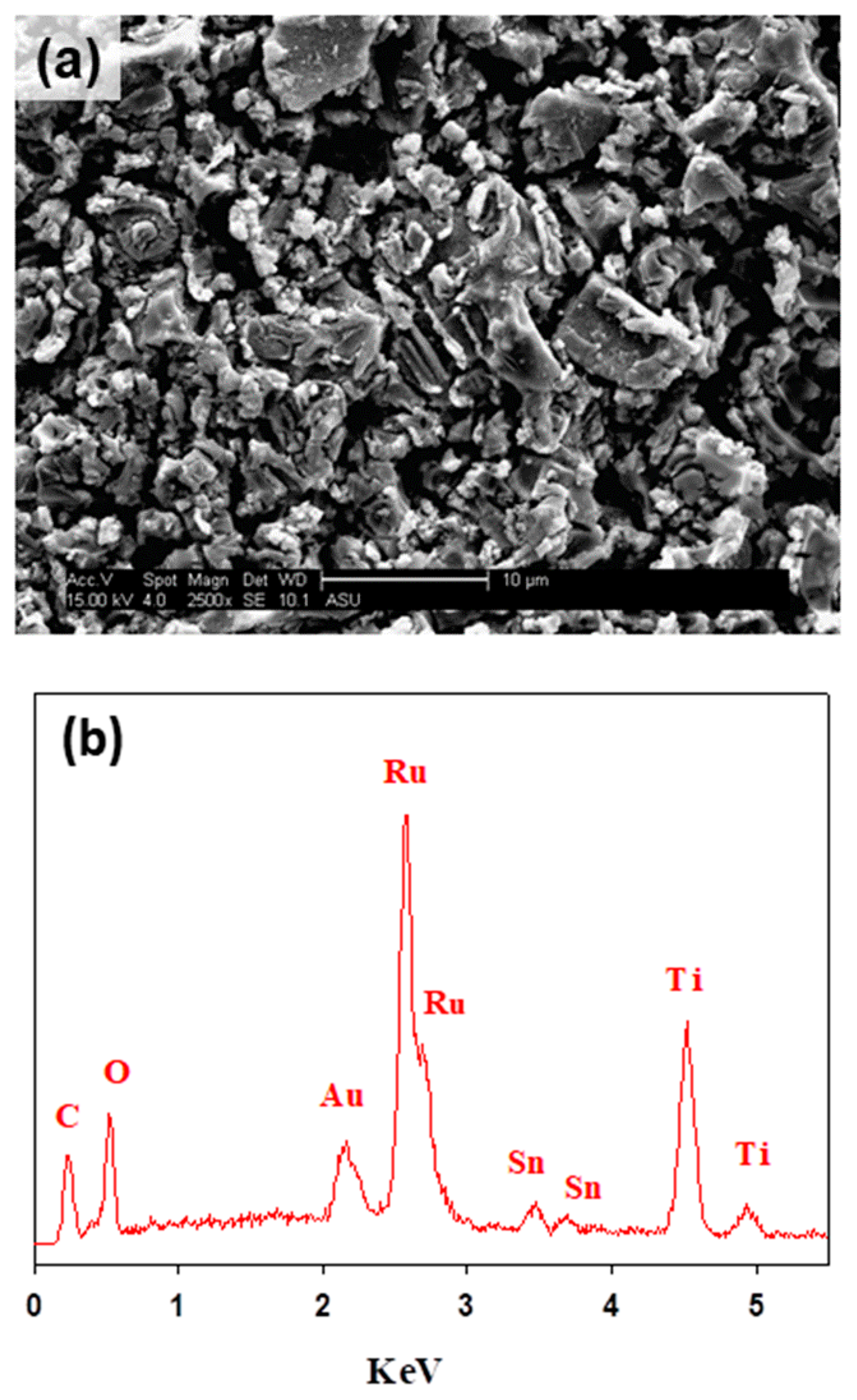
3.1. Physicochemical Characterizations of Ti/RuO2-TiO2-SnO2
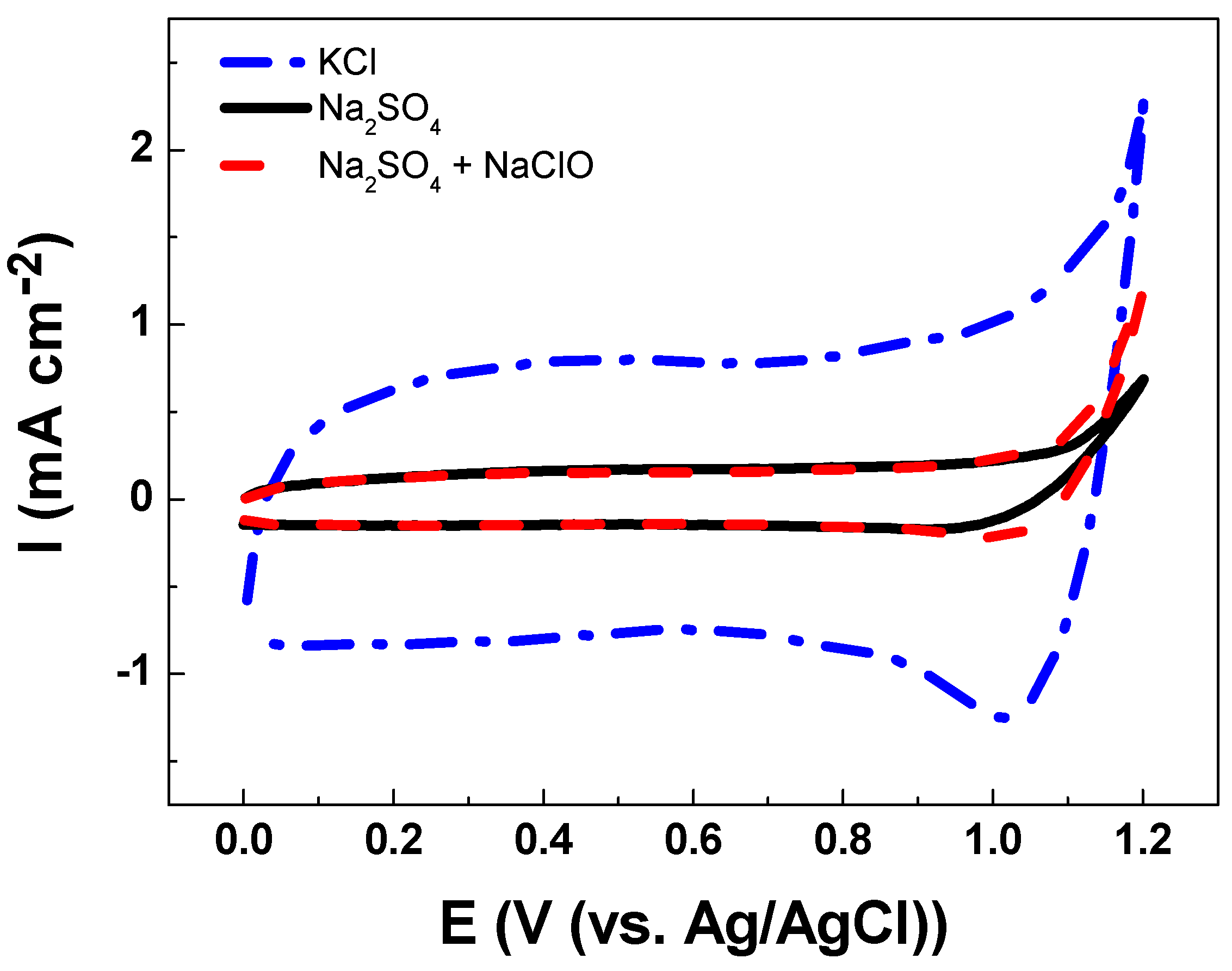
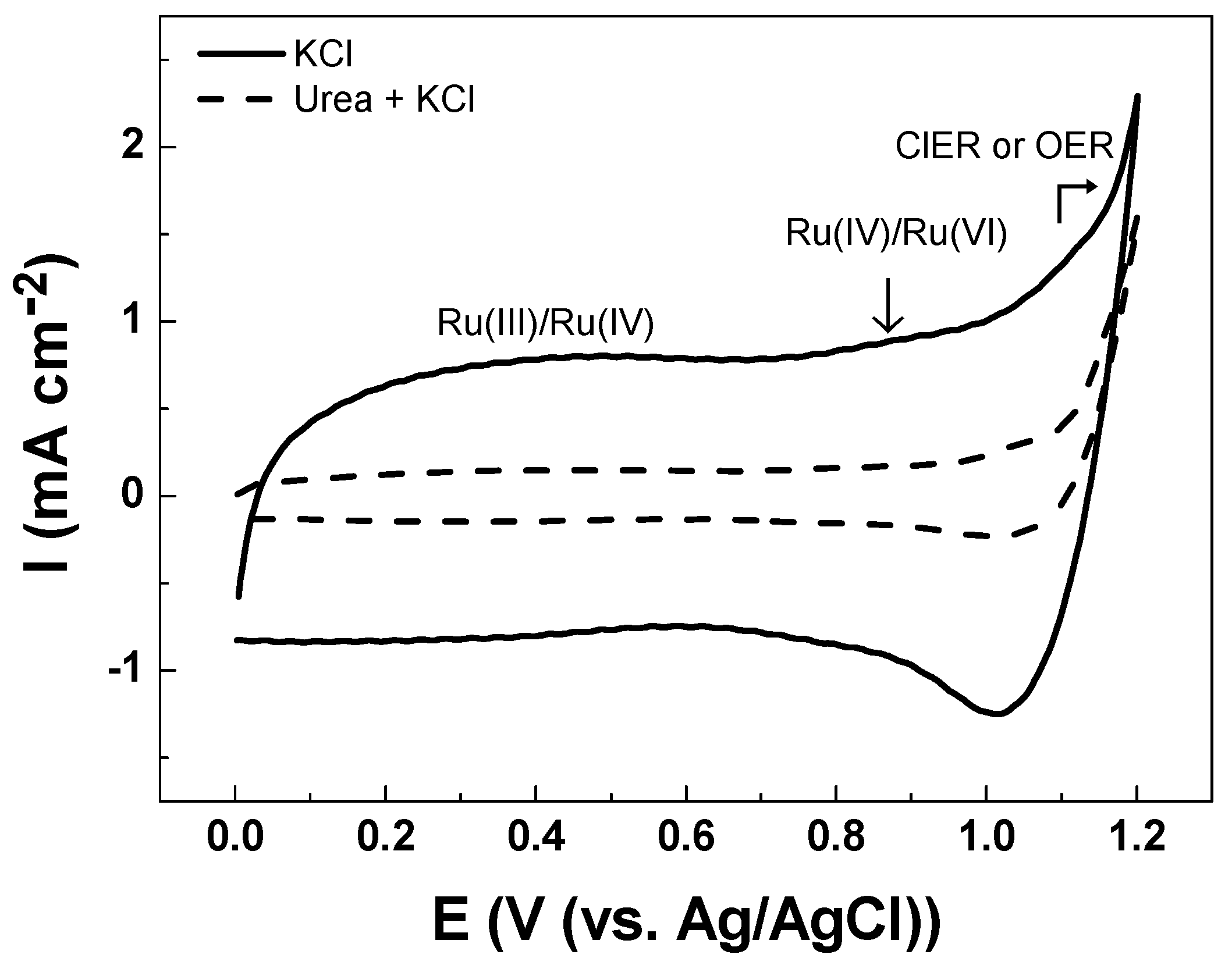
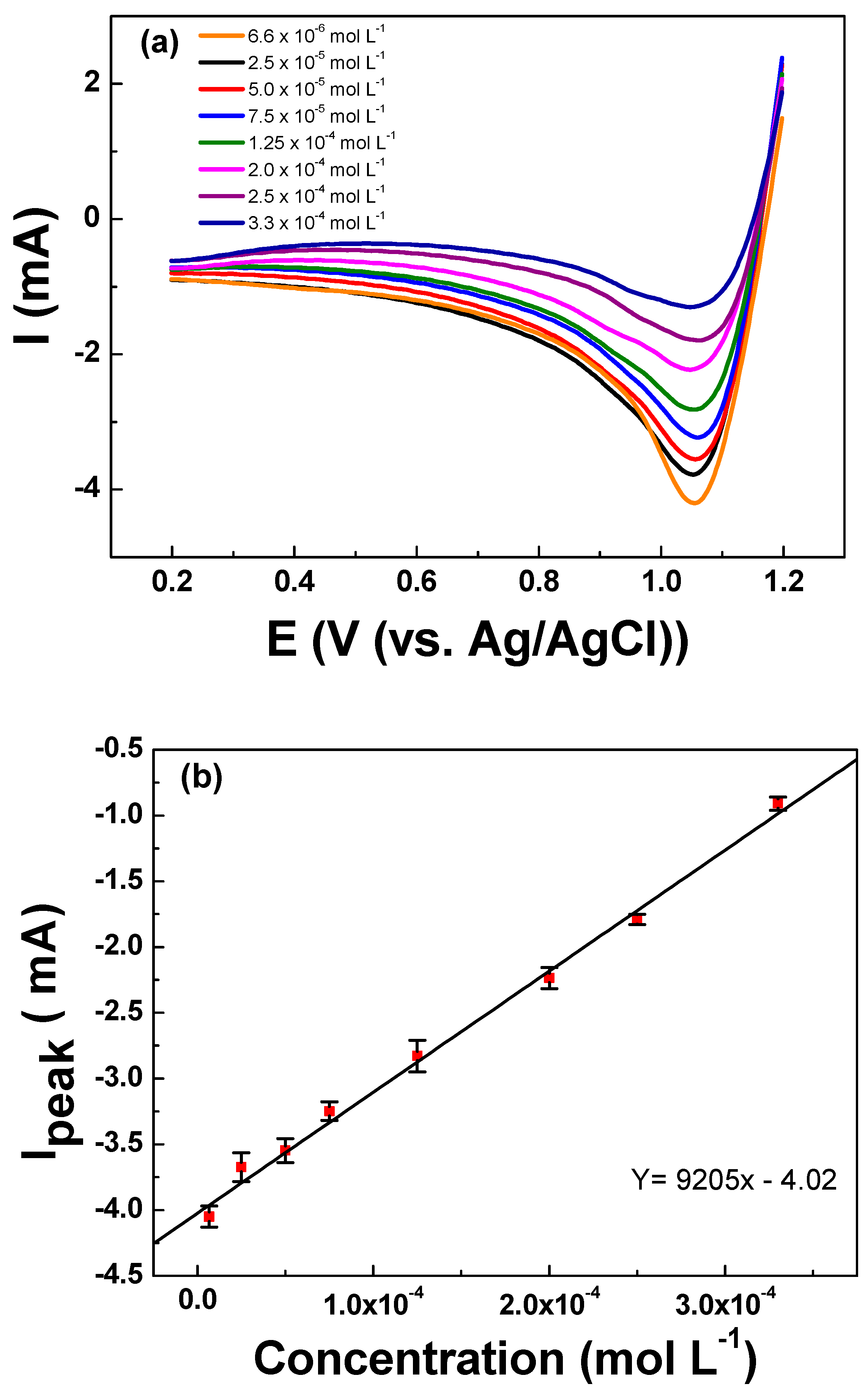
3.2. Electrochemical Characterizations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Erfkamp, J.; Guenther, M.; Gerlach, G. Enzyme-Functionalized piezoresistive hydrogel biosensors for the detection of urea. Sensors 2019, 19, 2858. [Google Scholar] [CrossRef]
- Li, L.; Long, Y.; Gao, J.-M.; Song, K.; Yang, G. Label-free and pH-sensitive colorimetric materials for the sensing of urea. Nanoscale 2016, 8, 4458–4462. [Google Scholar] [CrossRef]
- Fiorillo, A.S.; Tiriolo, R.; Pullano, S.A. Absorption of Urea Into Zeolite Layer Integrated With Microelectronic Circuits. IEEE Trans. Nanotechnol. 2014, 14, 214–217. [Google Scholar] [CrossRef]
- Pundir, C.; Jakhar, S.; Narwal, V. Determination of urea with special emphasis on biosensors: A review. Biosens. Bioelectron. 2018, 123, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Rahmanian, R.; Mozaffari, S.A.; Abedi, M. Disposable urea biosensor based on nanoporous ZnO film fabricated from omissible polymeric substrate. Mater. Sci. Eng. C 2015, 57, 387–396. [Google Scholar] [CrossRef]
- Hilding-Ohlsson, A.; Fauerbach, J.A.; Sacco, N.J.; Bonetto, M.C.; Corton, E. Voltamperometric Discrimination of Urea and Melamine Adulterated Skimmed Milk Powder. Sensors 2012, 12, 12220–12234. [Google Scholar] [CrossRef] [PubMed]
- Manjakkal, L.; Szwagierczak, D.; Dahiya, R. Metal oxides based electrochemical pH sensors: Current progress and future perspectives. Prog. Mater. Sci. 2020, 109, 100635. [Google Scholar] [CrossRef]
- Li, S.; Simonian, A.; Chin, B.A. Sensors for Agriculture and the Food Industry. Electrochem. Soc. Interface 2010, 19, 41–46. [Google Scholar] [CrossRef]
- Zhuiykov, S. Solid-state sensors monitoring parameters of water quality for the next generation of wireless sensor networks. Sens. Actuators B Chem. 2012, 161, 1–20. [Google Scholar] [CrossRef]
- Grgur, B.N. Electrochemical Oxidation of Bromides on DSA/RuO2Anode in the Semi-Industrial Batch Reactor for On-Site Water Disinfection. J. Electrochem. Soc. 2019, 166, E50–E61. [Google Scholar] [CrossRef]
- Wang, L.; Morison, K. Implementation of online security assessment. IEEE Power Energy Mag. 2006, 4, 46–59. [Google Scholar] [CrossRef]
- Nakamura, K.C.; Guimarães, L.S.; Magdalena, A.G.; Angelo, A.C.D.; De Andrade, A.R.; Garcia-Segura, S.; Pipi, A.R. Electrochemically-driven mineralization of Reactive Blue 4 cotton dye: On the role of in situ generated oxidants. J. Electroanal. Chem. 2019, 840, 415–422. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Nienhauser, A.B.; Fajardo, A.S.; Bansal, R.; Coonrod, C.L.; Fortner, J.D.; Marcos-Hernández, M.; Rogers, T.; Villagran, D.; Wong, M.S.; et al. Disparities between experimental and environmental conditions: Research steps toward making electrochemical water treatment a reality. Curr. Opin. Electrochem. 2020, 22, 9–16. [Google Scholar] [CrossRef]
- Trasatti, S. Electrocatalysis in the anodic evolution of oxygen and chlorine. Electrochimica Acta 1984, 29, 1503–1512. [Google Scholar] [CrossRef]
- Trieu, V.; Schley, B.; Natter, H.; Kintrup, J.; Bulan, A.; Hempelmann, R. RuO2-based anodes with tailored surface morphology for improved chlorine electro-activity. Electrochimica Acta 2012, 78, 188–194. [Google Scholar] [CrossRef]
- Papulova, G.N.; Kvasnikov, M.Y. Water-Soluble Urea-Formaldehyde Oligomers in Paint for Single-Firing Decoration of Ceramic. Glas. Ceram. 2020, 77, 116–118. [Google Scholar] [CrossRef]
- Kapałka, A.; Katsaounis, A.; Michels, N.-L.; Leonidova, A.; Souentie, S.; Comninellis, C.; Udert, K.M. Ammonia oxidation to nitrogen mediated by electrogenerated active chlorine on Ti/PtOx-IrO2. Electrochem. Commun. 2010, 12, 1203–1205. [Google Scholar] [CrossRef]
- Carneiro, J.F.; Silva, J.R.; Rocha, R.S.; Ribeiro, J.; Lanza, M.R.V. Morphological and Electrochemical Characterization of Ti/MxTiySnzO2 (M = Ir or Ru) Electrodes Prepared by the Polymeric Precursor Method. Adv. Chem. Eng. Sci. 2016, 6, 364–378. [Google Scholar] [CrossRef]
- Ribeiro, J.; Alves, P.D.P.; De Andrade, A.R. Effect of the preparation methodology on some physical and electrochemical properties of Ti/IrxSn(1−x)O2 materials. J. Mater. Sci. 2007, 42, 9293–9299. [Google Scholar] [CrossRef]
- Manjakkal, L.; Cvejin, K.; Kulawik, J.; Zaraska, K.; Szwagierczak, D.; Stojanovic, G. Sensing mechanism of RuO2–SnO2 thick film pH sensors studied by potentiometric method and electrochemical impedance spectroscopy. J. Electroanal. Chem. 2015, 759, 82–90. [Google Scholar] [CrossRef]
- Shin, Y.-U.; Yoo, H.-Y.; Kim, S.; Chung, K.-M.; Park, Y.-G.; Hwang, K.-H.; Hong, S.W.; Park, H.; Cho, K.; Lee, J. Sequential Combination of Electro-Fenton and Electrochemical Chlorination Processes for the Treatment of Anaerobically-Digested Food Wastewater. Environ. Sci. Technol. 2017, 51, 10700–10710. [Google Scholar] [CrossRef] [PubMed]
- Neto, S.A.; de Andrade, A. Electrooxidation of glyphosate herbicide at different DSA® compositions: pH, concentration and supporting electrolyte effect. Electrochim. Acta 2009, 54, 2039–2045. [Google Scholar] [CrossRef]
- Lopez-Ojeda, G.C.; Gutierrez-lara, M.R.; Duran-Moreno, A. Efecto del pH sobre la oxidación electroquímica de fenol empleando un ánodo dimensionalmente estable de SnO2-Sb2O5-RuO2. Rev. Mex. Ing. Quím. 2015, 14, 437–452. [Google Scholar]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. Water Treatment Principles and Design, 3rd ed.; Wiley: Hoboken, NJ, USA, 2012; pp. 532–542. [Google Scholar]
- Garcia-Segura, S.; Mostafa, E.; Baltruschat, H. Electrogeneration of inorganic chloramines on boron-doped diamond anodes during electrochemical oxidation of ammonium chloride, urea and synthetic urine matrix. Water Res. 2019, 160, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Deroco, P.B.; Vicentini, F.C.; Oliveira, G.G.; Rocha-Filho, R.C.; Fatibello-Filho, O. Square-wave voltammetric determination of hydroxychloroquine in pharmaceutical and synthetic urine samples using a cathodically pretreated boron-doped diamond electrode. J. Electroanal. Chem. 2014, 719, 19–23. [Google Scholar] [CrossRef]
- Laube, N.; Mohr, B.; Hesse, A. Laser-probe-based investigation of the evolution of particle size distributions of calcium oxalate particles formed in artificial urines. J. Cryst. Growth 2001, 233, 367–374. [Google Scholar] [CrossRef]
- Dbira, S.; Bensalah, N.; Cañizares, P.; Rodrigo, M.A.; Bedoui, A. The electrolytic treatment of synthetic urine using DSA electrodes. J. Electroanal. Chem. 2015, 744, 62–68. [Google Scholar] [CrossRef]
- Cullity, B.D. Elements of X-Ray Diffraction, 3rd ed.; Addison-Wesley: San Francisco, CA, USA, 2001; pp. 99–106. [Google Scholar]
- Comninellis, C.; Vercesi, G.P. Characterization of DSA|oxygen evolving electrodes: Choice of a coating. J. Appl. Electrochem. 1991, 21, 335–345. [Google Scholar] [CrossRef]
- Coteiro, R.D.; Teruel, F.S.; Ribeiro, J.; De Andrade, A.R. Effect of solvent on the preparation and characterization of DSA®-type anodes containing RuO2-TiO2-SnO2. J. Braz. Chem. Soc. 2006, 17, 771–779. [Google Scholar] [CrossRef][Green Version]
- Hutchings, R.; Stucki, S. A structural investigation of stabilized oxygen evolution catalysts. J. Mater. Sci. 1984, 19, 3987–3994. [Google Scholar] [CrossRef]
- Srinivasan, N.; Kiruthika, G. Conductivity studies on the substituted stannate pyrochlore system Gd2Sn2-x-yMxAyO7 (M= Ti and A = Ru; x = 0.5, 1.0 and 1.5; y = 0.2) . Solid State Sci. 2019, 96, 105957. [Google Scholar] [CrossRef]
- Kolesnikov, V.A.; Novikov, V.T.; Isaev, M.K.; Alekseeva, T.V.; Kolesnikov, A.V. Investigation of Electrodes with an Active Layer of a Mixture of the Oxides TiO2, RuO2, SnO2. Glas. Ceram. 2018, 75, 148–153. [Google Scholar] [CrossRef]
- Ribeiro, J.; De Andrade, A.R. Characterization of RuO[sub 2]-Ta[sub 2]O[sub 5] Coated Titanium Electrode. J. Electrochem. Soc. 2004, 151, D106–D112. [Google Scholar] [CrossRef]
- Zeradjanin, A.R.; Schilling, T.; Seisel, S.; Bron, M.; Schuhmann, W. Visualization of Chlorine Evolution at Dimensionally Stable Anodes by Means of Scanning Electrochemical Microscopy. Anal. Chem. 2011, 83, 7645–7650. [Google Scholar] [CrossRef] [PubMed]
- Bulska, E. Metrology in Chemistry, 1st ed.; Springer: Cham, Switzerland, 2018; pp. 109–110. [Google Scholar]
- Liu, J.; Moakhar, R.S.; Perumal, A.S.; Roman, H.N.; Mahshid, S.; Wachsmann-Hogiu, S. An AgNP-deposited commercial electrochemistry test strip as a platform for urea detection. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Irzalinda, A.D.; Gunlazuardi, J.; Wibowo, R. Development of a non-enzymatic urea sensor based on a Ni/Au electrode. J. Phys. Conf. Ser. 2020, 1442, 012054. [Google Scholar] [CrossRef]
- Naik, T.S.K.; Saravanan, S.; Saravana, K.N.S.; Pratiush, U.; Ramamurthy, P.C. A non-enzymatic urea sensor based on the nickel sulfide / graphene oxide modified glassy carbon electrode. Mater. Chem. Phys. 2020, 245, 122798. [Google Scholar] [CrossRef]
- Nguyen, N.S.; Das, G.; Yoon, H.H. Nickel/cobalt oxide-decorated 3D graphene nanocomposite electrode for enhanced electrochemical detection of urea. Biosens. Bioelectron. 2016, 77, 372–377. [Google Scholar] [CrossRef]
- Nguyen, N.S.; Yoon, H.H. Nickel oxide-deposited cellulose/CNT composite electrode for non-enzymatic urea detection. Sens. Actuators B Chem. 2016, 236, 304–310. [Google Scholar] [CrossRef]
- Gimeno-García, E.; Andreu, V.; Boluda, R. Heavy metals incidence in the application of inorganic fertilizers and pesticides to rice farming soils. Environ. Pollut. 1996, 92, 19–25. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Textile dye wastewater characteristics and constituents of synthetic effluents: A critical review. Int. J. Environ. Sci. Technol. 2018, 16, 1193–1226. [Google Scholar] [CrossRef]
- Squissato, A.L.; Richter, E.M.; Munoz, R.A. Voltammetric determination of copper and tert-butylhydroquinone in biodiesel: A rapid quality control protocol. Talanta 2019, 201, 433–440. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.E.; Stoianov, I.; O’Hare, D. Continuous Chlorine Detection in Drinking Water and a Review of New Detection Methods. Johns. Matthey Technol. Rev. 2019, 63, 103–118. [Google Scholar] [CrossRef]
- Syshchyk, O.; Skryshevsky, V.A.; Soldatkin, O.O.; Soldatkin, A.P. Enzyme biosensor systems based on porous silicon photoluminescence for detection of glucose, urea and heavy metals. Biosens. Bioelectron. 2015, 66, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, W. Determination of Heavy Metal Inos Using Conductometric Biosensor Based on Sol-Gel Immobilized Urease. Bull. Korean Chem. Soc. 2002, 23, 1169–1172. [Google Scholar] [CrossRef][Green Version]
| D (nm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| (101) | (110) | (200) | (211) | (111) | (204) | (220) | (301) | (303) | |
| Solid solution | 20 | 15 | 38 | 17. | - | - | - | - | - |
| TiO2 phase | 16 | - | - | - | 44 | 29 | 28 | 26 | 31 |
| Nominal Composition (atom. %) | Experimental Composition (atom. %) | ||
|---|---|---|---|
| Ru | Ti | Sn | |
| Ti/RuO2-TiO2-SnO2 (50:40:10 atom. %) | 45 | 50 | 4.7 |
| Solution | Cathodic Charge Density, qc (mC cm−2) |
|---|---|
| Na2SO4 | 0.0 |
| KCl | 9.4 |
| Na2SO4 with NaClO | 1.0 |
| Urea in KCl | 1.1 |
| Performance Characteristics * | Urea |
|---|---|
| Linear range (mol L−1) | 6.66 × 10−6 to 3.33 × 10−4 |
| Intercept | −4.02 ± 0.004 |
| Sensitivity (mA mol L−1) | 9205 ± 0.004 |
| LOQ (mol L−1) | 7.66 × 10−6 |
| LOD (mol L−1) | 1.83 × 10−6 |
| R2 | 0.997 |
| Repeatability (RSD for n = 32) | 5.10% |
| Reproducibility (RSD for n = 7) | 1.81% |
| Electrode | Technique * | Linear Range (mol L−1) | LOD (mol L−1) | Ref. |
|---|---|---|---|---|
| AgNP-deposited commercial Au-Pd electrode | CV | 1.66 × 10−4 to 1.67 × 10−3 | 0.141 | [38] |
| Au electrode deposited with Ni | CV | - | 0.033 | [39] |
| Glassy carbon modified with nickel sulfide/graphene oxide | DPV | 9.99 × 10−3 to 0.049 | 3.80 × 10−3 | [40] |
| 3D graphene/NiCo2O4 | CA | 0.049 to 0.249 | 2.66 × 10−3 | [41] |
| NiO/celulose/CNT | CA | 9.99 × 10−3 to 1.40 | 3.78 × 10−3 | [42] |
| Ti/RuO2-TiO2-SnO2 | LSV | 6.66 × 10−6 to 3.33 × 10−4 | 1.83 × 10−6 | This work |
| Current Signal Variation (%) | |
|---|---|
| Interferents | Interferent: Analyte Ratio 1:1 |
| Ni(II) | −8.2 |
| Zn(II) | +9.7 |
| S(II) | −13.2 |
| Cd(II) | −20.0 |
| Fe(III) | −20.4 |
| Pb(II) | −22.4 |
| Cu(II) | −25.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasconcellos, M.d.L.S.; Silva, L.R.G.; Lee, C.-S.; Fajardo, A.S.; Garcia-Segura, S.; Ribeiro, J. Dimensionally Stable Anode Based Sensor for Urea Determination via Linear Sweep Voltammetry. Sensors 2021, 21, 3450. https://doi.org/10.3390/s21103450
Vasconcellos MdLS, Silva LRG, Lee C-S, Fajardo AS, Garcia-Segura S, Ribeiro J. Dimensionally Stable Anode Based Sensor for Urea Determination via Linear Sweep Voltammetry. Sensors. 2021; 21(10):3450. https://doi.org/10.3390/s21103450
Chicago/Turabian StyleVasconcellos, Maria de Lourdes S., Luiz Ricardo G. Silva, Chung-Seop Lee, Ana Sofia Fajardo, Sergi Garcia-Segura, and Josimar Ribeiro. 2021. "Dimensionally Stable Anode Based Sensor for Urea Determination via Linear Sweep Voltammetry" Sensors 21, no. 10: 3450. https://doi.org/10.3390/s21103450
APA StyleVasconcellos, M. d. L. S., Silva, L. R. G., Lee, C.-S., Fajardo, A. S., Garcia-Segura, S., & Ribeiro, J. (2021). Dimensionally Stable Anode Based Sensor for Urea Determination via Linear Sweep Voltammetry. Sensors, 21(10), 3450. https://doi.org/10.3390/s21103450









