Optical Interferometric Fringe Pattern-Incorporated Spectrum Calibration Technique for Enhanced Sensitivity of Spectral Domain Optical Coherence Tomography
Abstract
1. Introduction
2. Materials and Methods
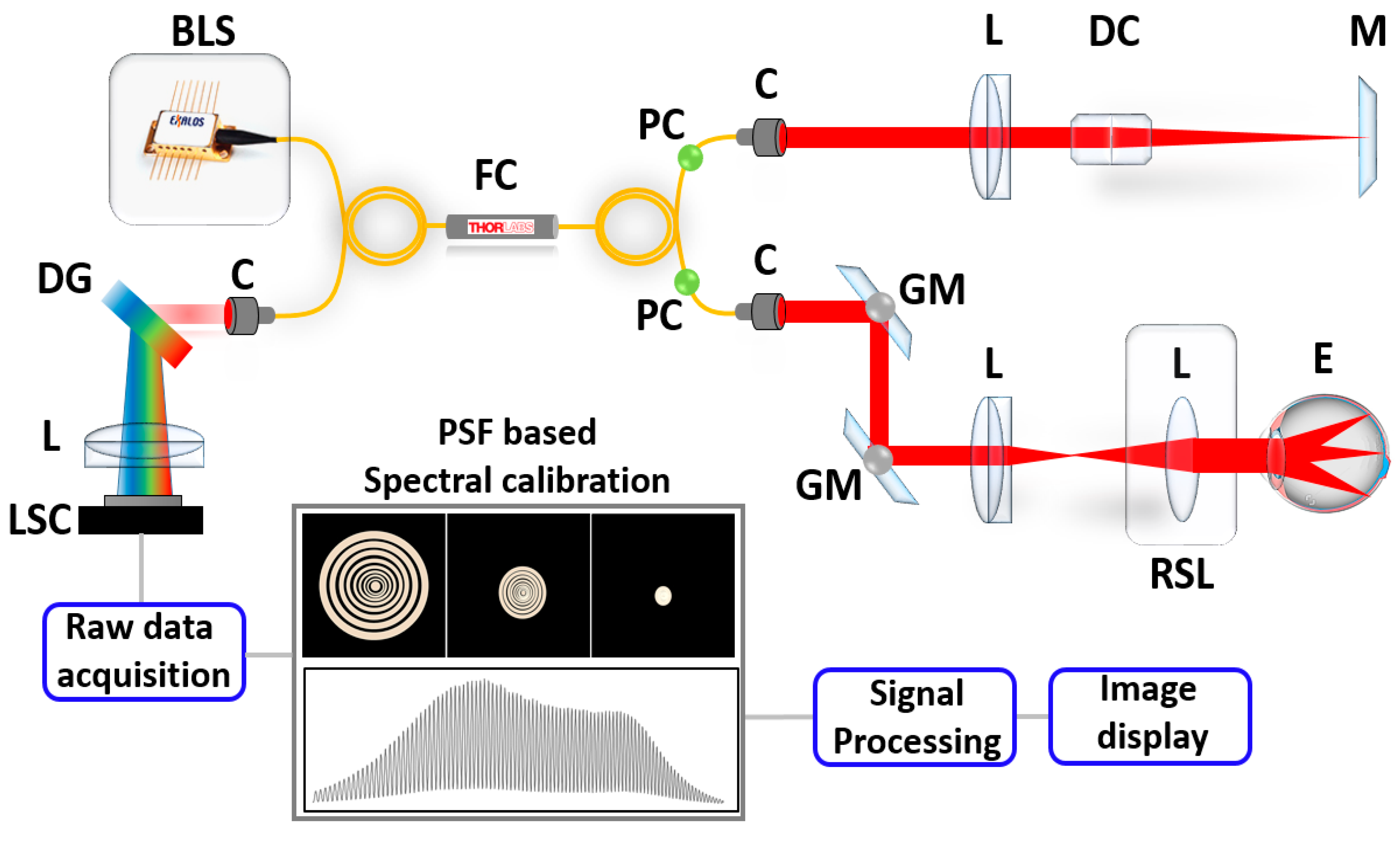
2.1. Description of Optical Coherence Tomography (OCT) System Configuration
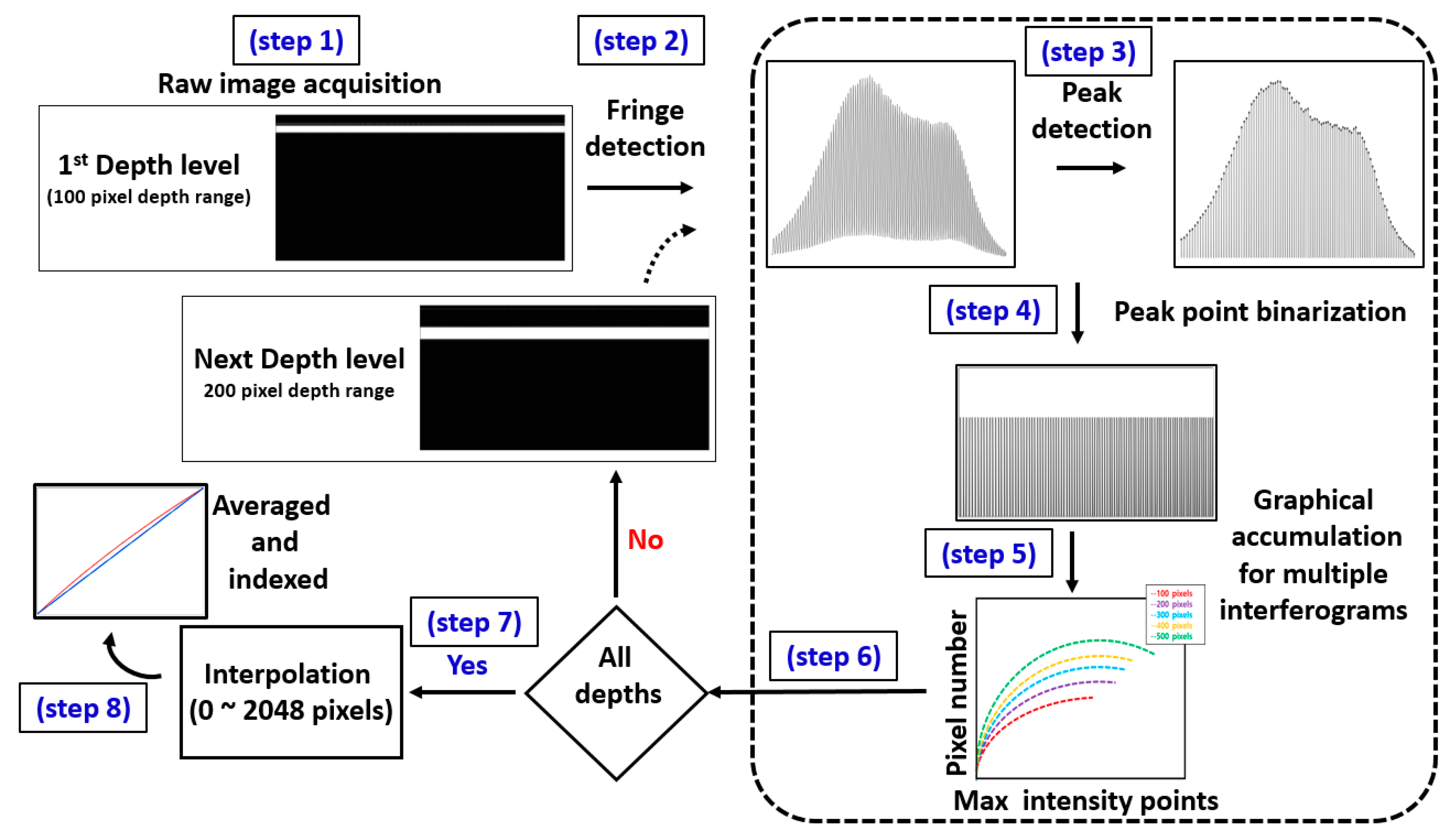
2.2. Spectral Calibration Algorithm
2.3. In Vivo Experimental Procedure
3. Results and Discussion
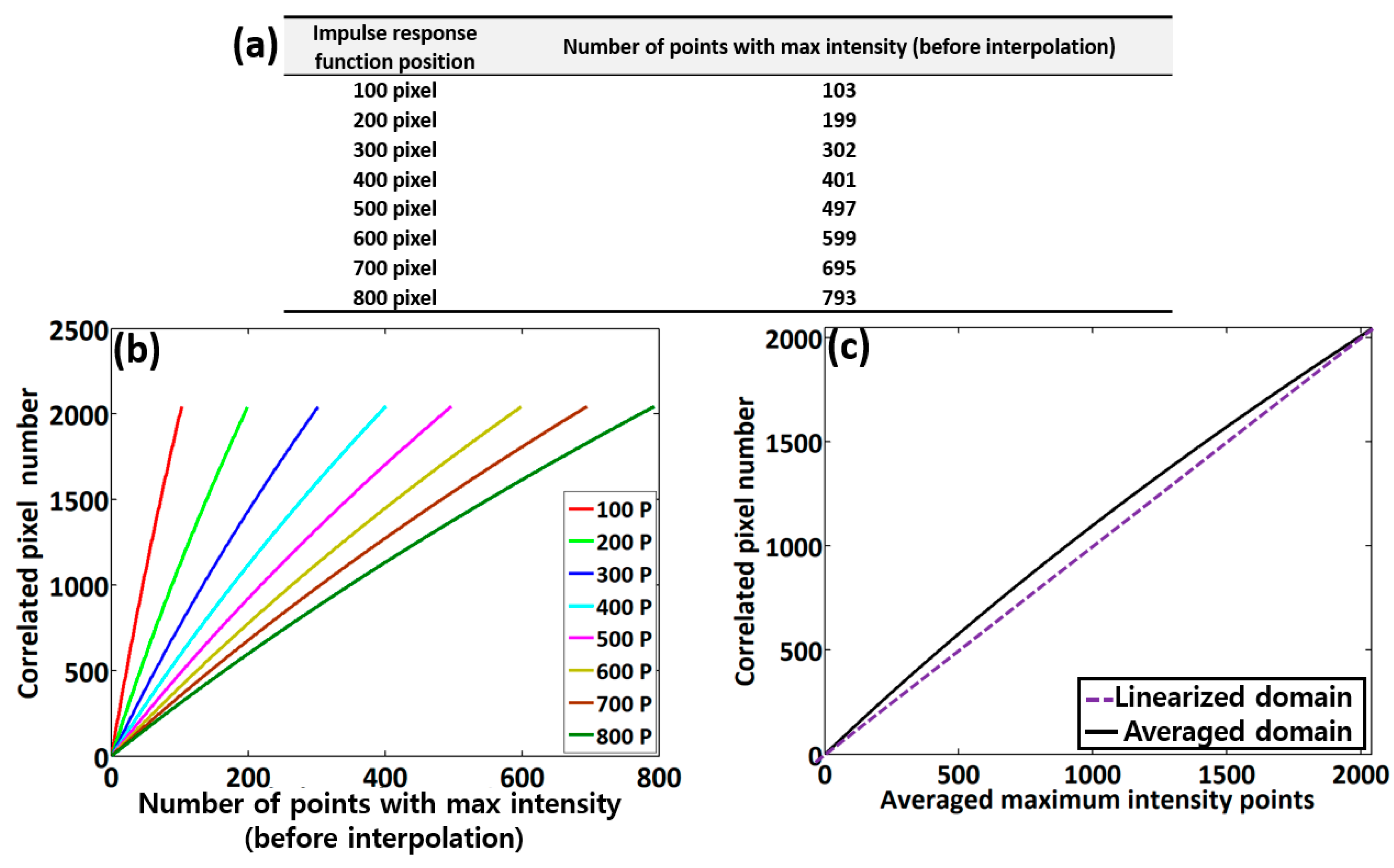
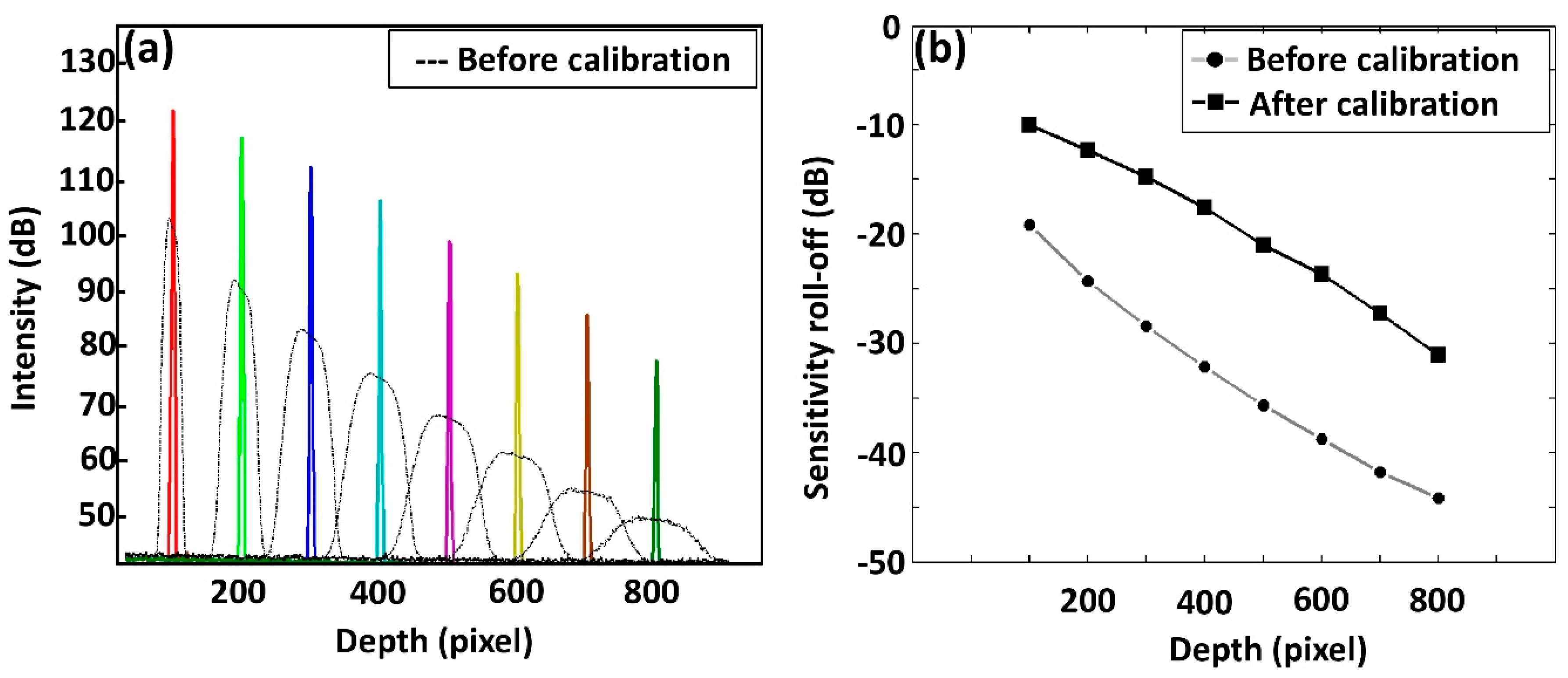
3.1. Analysis of Point Spread Function (PSF) Incorporated Spectral Calibration
3.2. Depth-Dependent Sensitivity Assessments after Spectral Calibration
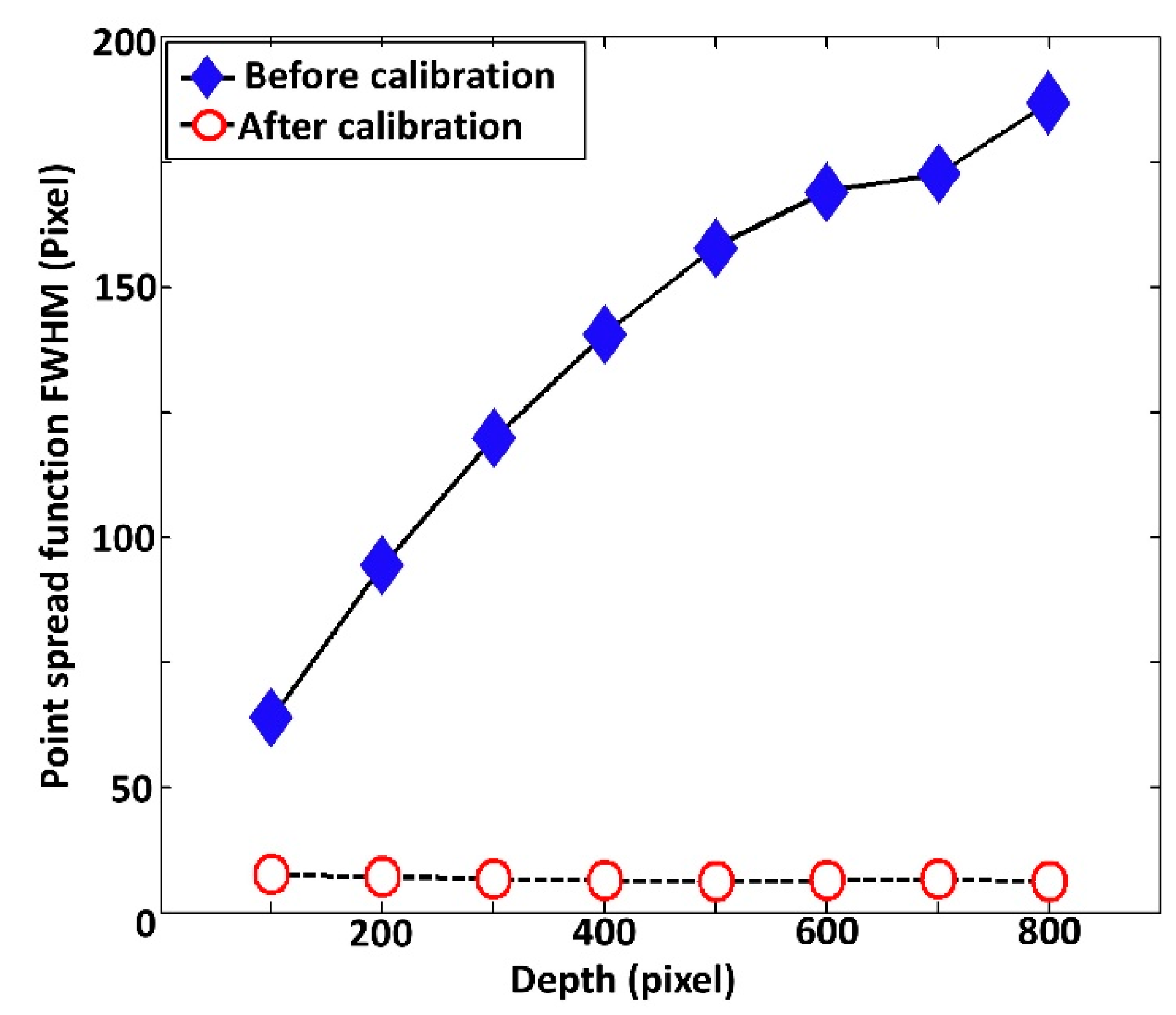
3.3. Full Width at Half Maximum (FWHM) Evaluation of Depth-Dependent PSF
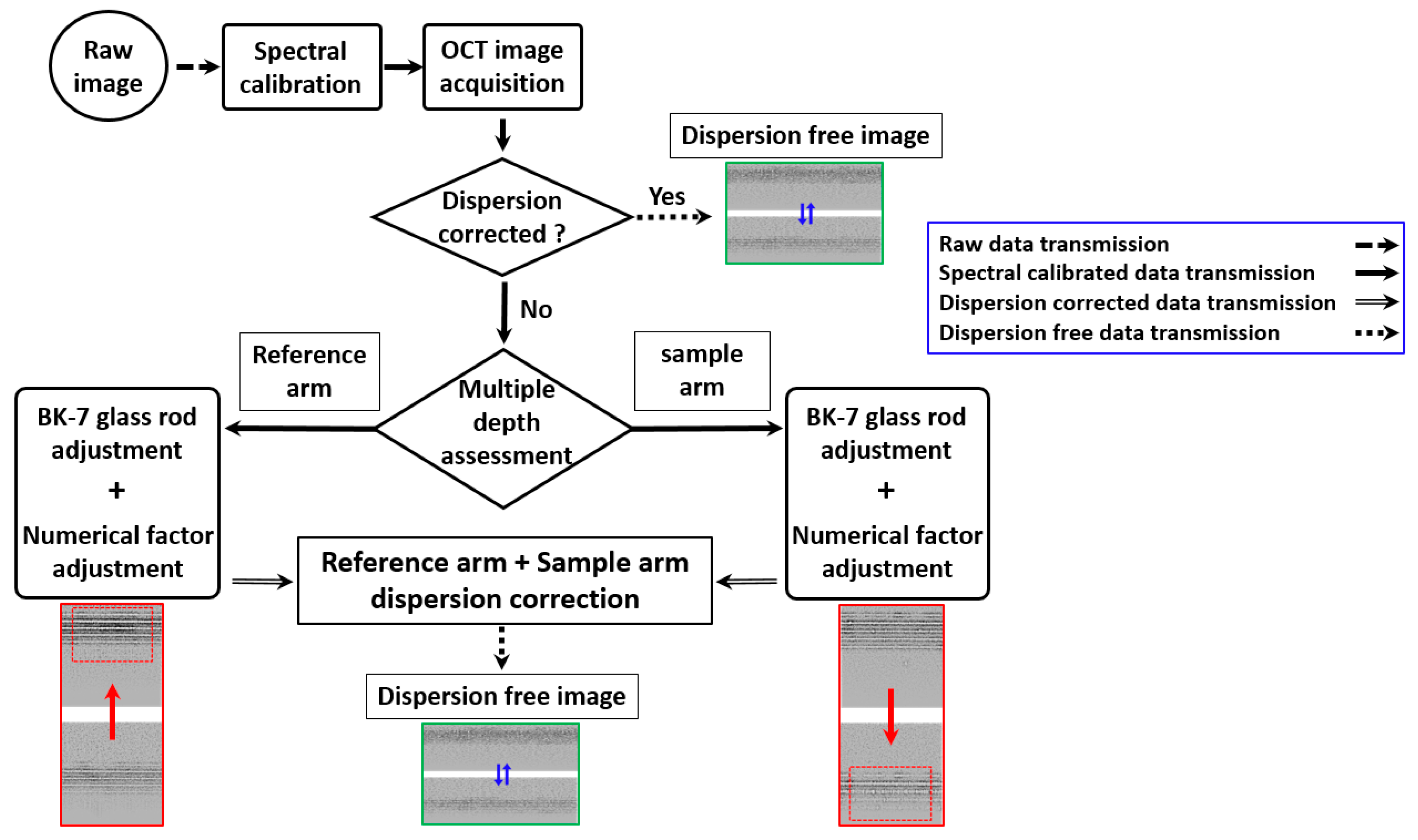
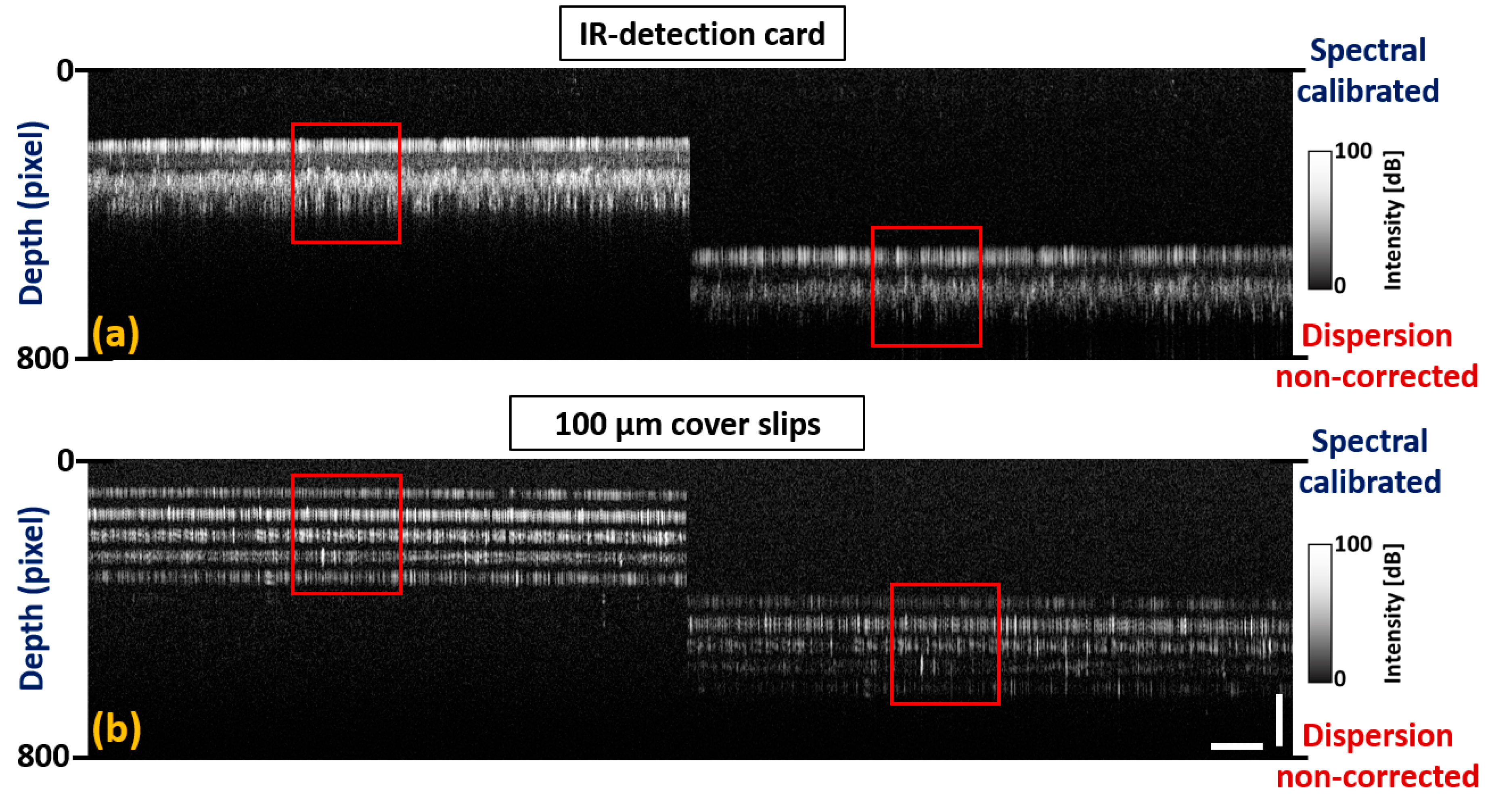
3.4. Implementation of Dispersion Correction
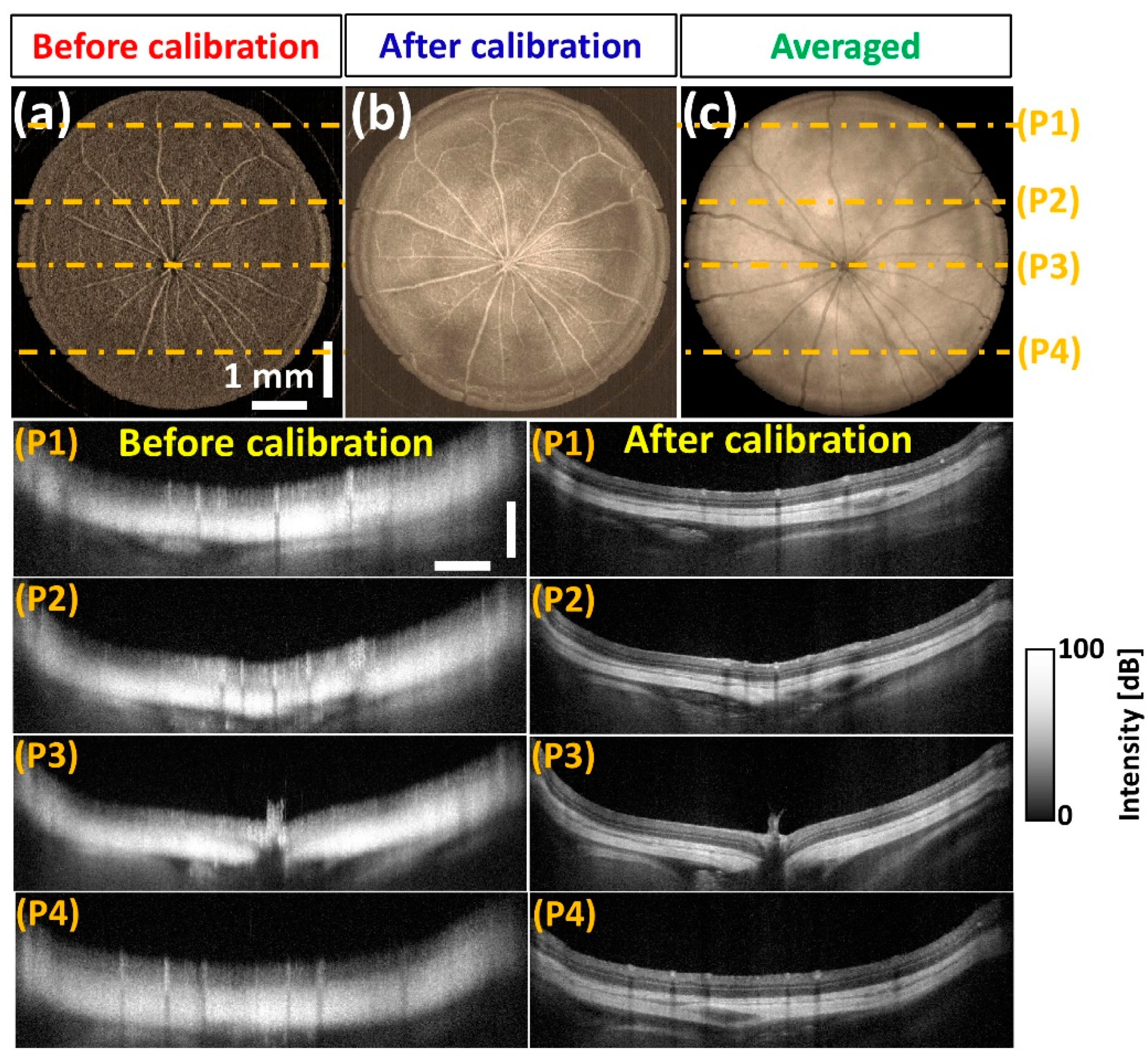
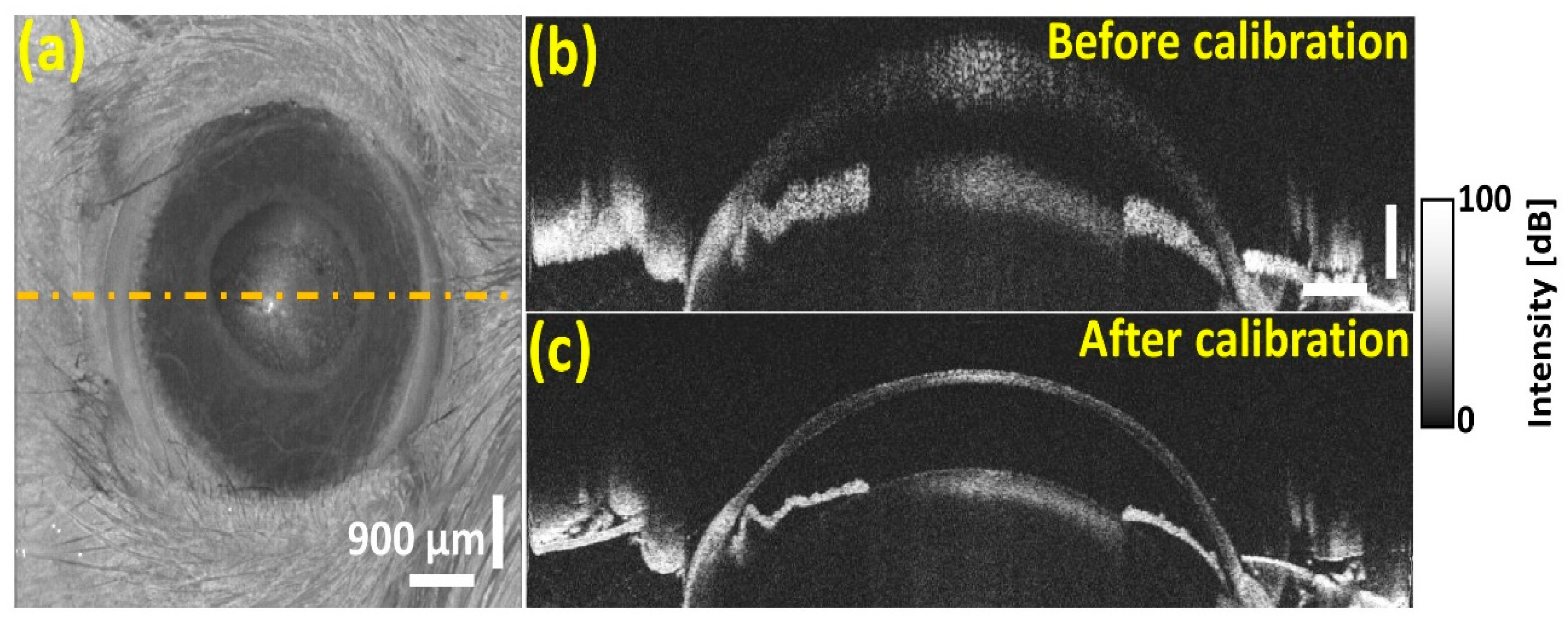
3.5. In Vivo OCT Image Verification of the Developed Spectral Calibration Method
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wojtkowski, M.; Srinivasan, V.J.; Ko, T.H.; Fujimoto, J.G.; Kowalczyk, A.; Duker, J.S. Ultrahigh-resolution, high-speed, fourier domain optical coherence tomography and methods for dispersion compensation. Opt. Express 2004, 12, 2404–2422. [Google Scholar] [CrossRef] [PubMed]
- Wojtkowski, M.; Srinivasan, V.; Fujimoto, J.G.; Ko, T.; Schuman, J.S.; Kowalczyk, A.; Duker, J.S. Three-dimensional retinal imaging with high-speed ultrahigh-resolution optical coherence tomography. Ophthalmology 2005, 112, 1734–1746. [Google Scholar] [CrossRef] [PubMed]
- Cense, B.; Nassif, N.A.; Chen, T.C.; Pierce, M.C.; Yun, S.-H.; Park, B.H.; Bouma, B.E.; Tearney, G.J.; de Boer, J.F. Ultrahigh-resolution high-speed retinal imaging using spectral-domain optical coherence tomography. Opt. Express 2004, 12, 2435–2447. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, J.G.; Brezinski, M.E.; Tearney, G.J.; Boppart, S.A.; Bouma, B.; Hee, M.R.; Southern, J.F.; Swanson, E.A. Optical biopsy and imaging using optical coherence tomography. Nat. Med. 1995, 1, 970–972. [Google Scholar] [CrossRef] [PubMed]
- Zysk, A.M.; Nguyen, F.T.; Oldenburg, A.L.; Marks, D.L.; Boppart, S.A. Optical coherence tomography: A review of clinical development from bench to bedside. J. Biomed. Opthalmol. 2007, 12, 051403–051424. [Google Scholar] [CrossRef] [PubMed]
- Spaide, R.F.; Klancnik, J.M.; Cooney, M.J. Retinal vascular layers imaged by fluorescein angiography and optical coherence tomography angiography. JAMA Ophthalmol. 2015, 133, 45–50. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, R.; Vasa-Nicotera, M.; Leistner, D.M.; Reis, S.M.; Thome, C.E.; Boeckel, J.-N.; Fichtlscherer, S.; Zeiher, A.M. Coronary atherosclerotic plaque characteristics and cardiovascular risk factors―insights from an optical coherence tomography study. Circ. J. 2017, 81, 1165–1173. [Google Scholar] [CrossRef]
- Hariri, L.P.; Adams, D.C.; Wain, J.C.; Lanuti, M.; Muniappan, A.; Sharma, A.; Colby, T.V.; Mino-Kenudson, M.; Mark, E.J.; Kradin, R.L. Endobronchial optical coherence tomography for low-risk microscopic assessment and diagnosis of idiopathic pulmonary fibrosis in vivo. Am. J. Respir. Crit. Care Med. 2018, 197, 949–952. [Google Scholar] [CrossRef]
- Wang, K.C.; Astell, C.J.; Wijesinghe, P.; Larcombe, A.N.; Pinniger, G.J.; Zosky, G.R.; Kennedy, B.F.; Berry, L.J.; Sampson, D.D.; James, A.L. Optical coherence tomography-based contact indentation for diaphragm mechanics in a mouse model of transforming growth factor alpha induced lung disease. Sci. Rep. 2017, 7, 1517. [Google Scholar] [CrossRef]
- Schuh, S.; Holmes, J.; Ulrich, M.; Themstrup, L.; Jemec, G.B.; De Carvalho, N.; Pellacani, G.; Welzel, J. Imaging blood vessel morphology in skin: Dynamic optical coherence tomography as a novel potential diagnostic tool in dermatology. Dermatol. Ther. 2017, 7, 187–202. [Google Scholar] [CrossRef]
- Wijesinghe, R.E.; Park, K.; Jung, Y.; Kim, P.; Jeon, M.; Kim, J. Industrial resin inspection for display production using automated fluid-inspection based on multimodal optical detection techniques. Opt. Las. Eng. 2017, 96, 75–82. [Google Scholar] [CrossRef]
- Cho, N.H.; Park, K.; Kim, J.-Y.; Jung, Y.; Kim, J. Quantitative assessment of touch-screen panel by nondestructive inspection with three-dimensional real-time display optical coherence tomography. Opt. Las. Eng. 2015, 68, 50–57. [Google Scholar] [CrossRef]
- Shirazi, M.F.; Wijesinghe, R.E.; Ravichandran, N.K.; Kim, P.; Jeon, M.; Kim, J. Quality assessment of the optical thin films using line field spectral domain optical coherence tomography. Opt. Las. Eng. 2018, 110, 47–53. [Google Scholar] [CrossRef]
- Wijesinghe, R.E.; Lee, S.-Y.; Ravichandran, N.K.; Han, S.; Jeong, H.; Han, Y.; Jung, H.-Y.; Kim, P.; Jeon, M.; Kim, J. Optical coherence tomography-integrated, wearable (backpack-type), compact diagnostic imaging modality for in situ leaf quality assessment. Appl. Opt. 2017, 56, D108–D114. [Google Scholar] [CrossRef]
- Wijesinghe, R.E.; Lee, S.-Y.; Ravichandran, N.K.; Shirazi, M.F.; Kim, P.; Jung, H.-Y.; Jeon, M.; Kim, J. Biophotonic approach for the characterization of initial bitter-rot progression on apple specimens using optical coherence tomography assessments. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, R.E.H.; Lee, S.-Y.; Kim, P.; Jung, H.-Y.; Jeon, M.; Kim, J. Optical sensing method to analyze germination rate of capsicum annum seeds treated with growth-promoting chemical compounds using optical coherence tomography. J. Biomed. Opt. 2017, 22, 091502–091510. [Google Scholar] [CrossRef]
- Lan, G.; Li, G. Design of a k-space spectrometer for ultra-broad waveband spectral domain optical coherence tomography. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Zhang, N.; Huo, T.; Wang, C.; Chen, T.; Zheng, J.-g.; Xue, P. Compressed sensing with linear-in-wavenumber sampling in spectral-domain optical coherence tomography. Opt. Lett. 2012, 37, 3075–3077. [Google Scholar] [CrossRef]
- Yun, S.; Tearney, G.; Bouma, B.; Park, B.; de Boer, J.F. High-speed spectral-domain optical coherence tomography at 1.3 µm wavelength. Opt. Express 2003, 11, 3598–3604. [Google Scholar] [CrossRef]
- Van der Jeught, S.; Bradu, A.; Podoleanu, A.G. Real-time resampling in fourier domain optical coherence tomography using a graphics processing unit. J. Biomed. Opt. 2010, 15, 030511–030514. [Google Scholar] [CrossRef]
- Agrawal, A.; Pfefer, T.J.; Gilani, N.; Drezek, R. Three-dimensional characterization of optical coherence tomography point spread functions with a nanoparticle-embedded phantom. Opt. Lett. 2010, 35, 2269–2271. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Kang, J.U. Real-time 4d signal processing and visualization using graphics processing unit on a regular nonlinear-k fourier-domain oct system. Opt. Express 2010, 18, 11772–11784. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Rollins, A.M. Fourier domain optical coherence tomography with a linear-in-wavenumber spectrometer. Opt. Lett. 2007, 32, 3525–3527. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Ding, Z. Spectral calibration in spectral domain optical coherence tomography. Chin. Opt. Lett. 2008, 6, 902–904. [Google Scholar] [CrossRef]
- Gelikonov, V.; Gelikonov, G.; Shilyagin, P. Linear-wavenumber spectrometer for high-speed spectral-domain optical coherence tomography. Opt. Spectrosc. 2009, 106, 459–465. [Google Scholar] [CrossRef]
- Xu, D.; Huang, Y.; Kang, J.U. Compressive sensing with dispersion compensation on non-linear wavenumber sampled spectral domain optical coherence tomography. Biomed. Opt. Express 2013, 4, 1519–1532. [Google Scholar] [CrossRef]
- Sugita, M.; Brown, R.A.; Popov, I.; Vitkin, A. K-distribution three-dimensional mapping of biological tissues in optical coherence tomography. J. Biomed. 2018, 11, 1–18. [Google Scholar] [CrossRef]
- Uribe-Patarroyo, N.; Kassani, S.H.; Villiger, M.; Bouma, B.E. Robust wavenumber and dispersion calibration for fourier-domain optical coherence tomography. Opt. Express 2018, 26, 9081–9094. [Google Scholar] [CrossRef]
- Yuan, Q.; Wang, Z.; Song, L.; Ge, C.; Lu, Z.; Yang, T. Ultrafast wavenumber linear-step-swept source based on synchronous lightwave synthesized frequency sweeper. IEEE Photonics J. 2019, 11, 1–9. [Google Scholar] [CrossRef]
- Chan, K.K.; Tang, S. High-speed spectral domain optical coherence tomography using non-uniform fast fourier transform. Biomed. Opt. Express 2010, 1, 1309–1319. [Google Scholar] [CrossRef]
- Eigenwillig, C.M.; Biedermann, B.R.; Palte, G.; Huber, R. K-space linear fourier domain mode locked laser and applications for optical coherence tomography. Opt. Express 2008, 16, 8916–8937. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yuan, Z.; Wang, H.; Pan, Y. Increasing the imaging depth of spectral-domain oct by using interpixel shift technique. Opt. Express 2006, 14, 7014–7023. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Gao, W.; Bian, H.; Chen, C.; Liao, J. Self-spectral calibration for spectral domain optical coherence tomography. Opt. Eng. 2013, 52, 063603–063611. [Google Scholar] [CrossRef][Green Version]
- Jeon, M.; Kim, J.; Jung, U.; Lee, C.; Jung, W.; Boppart, S.A. Full-range k-domain linearization in spectral-domain optical coherence tomography. Appl. Opt. 2011, 50, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Kwon, O.; Wijesinghe, R.; Kim, P.; Jung, U.; Song, J.; Lee, C.; Jeon, M.; Kim, J. Numerical-sampling-functionalized real-time index regulation for direct k-domain calibration in spectral domain optical coherence tomography. Electronics 2018, 7, 182. [Google Scholar] [CrossRef]
- Hagen, N.; Tkaczyk, T.S. Compound prism design principles, iii: Linear-in-wavenumber and optical coherence tomography prisms. Appl. Opt. 2011, 50, 5023–5030. [Google Scholar] [CrossRef]
- Attendu, X.; Ruis, R.M.; Boudoux, C.; van Leeuwen, T.G.; Faber, D.J. Simple and robust calibration procedure for k-linearization and dispersion compensation in optical coherence tomography. J. Biomed. Opt. 2019, 24, 056001–056013. [Google Scholar] [CrossRef]
- Makita, S.; Fabritius, T.; Miura, M.; Yasuno, Y. 1st Canterbury Workshop on Optical Coherence Tomography and Adaptive Optics. In Full-Range, High-Speed, High-Resolution 1 μm Spectral-Domain Optical Coherence Tomography with bm-Scan Method for the Human Posterior Eye Imaging; International Society for Optics and Photonics: Canterbury, UK, 2008; p. 713914. [Google Scholar]
- Podoleanu, A.G.; Bradu, A. Master–slave interferometry for parallel spectral domain interferometry sensing and versatile 3d optical coherence tomography. Opt. Express 2013, 21, 19324–19338. [Google Scholar] [CrossRef]
- Bradu, A.; Israelsen, N.M.; Maria, M.; Marques, M.J.; Rivet, S.; Feuchter, T.; Bang, O.; Podoleanu, A. Recovering distance information in spectral domain interferometry. Sci. Rep. 2018, 8, 1–16. [Google Scholar] [CrossRef]
- Cui, D.; Liu, X.; Zhang, J.; Yu, X.; Ding, S.; Luo, Y.; Gu, J.; Shum, P.; Liu, L. Dual spectrometer system with spectral compounding for 1-μm optical coherence tomography in vivo. Opt. Lett. 2014, 39, 6727–6730. [Google Scholar] [CrossRef]
- Choi, W.; Baumann, B.; Swanson, E.A.; Fujimoto, J.G. Extracting and compensating dispersion mismatch in ultrahigh-resolution fourier domain oct imaging of the retina. Opt. Express 2012, 20, 25357–25368. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, R.E.; Park, K.; Kim, P.; Oh, J.; Kim, S.-W.; Kim, K.; Kim, B.-M.; Jeon, M.; Kim, J. Optically deviated focusing method based high-speed sd-oct for in vivo retinal clinical applications. Opt. Rev. 2015, 23, 1–9. [Google Scholar] [CrossRef]
- Jian, Y.; Wong, K.; Sarunic, M.V. Graphics processing unit accelerated optical coherence tomography processing at megahertz axial scan rate and high resolution video rate volumetric rendering. J. Biomed. Opt. 2013, 18, 026002–026007. [Google Scholar] [CrossRef] [PubMed]
- Choi, D.; Lee, J.; Jeon, M.; Kim, J. In vivo fascicle bifurcation imaging of rat sciatic nerve using swept-source optical coherence tomography. IEEE Access 2018, 6, 7713–7718. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, S.; Wijesinghe, R.E.; Jeon, D.; Han, Y.; Lee, J.; Lee, J.; Jo, H.; Lee, D.-E.; Jeon, M.; Kim, J. Optical Interferometric Fringe Pattern-Incorporated Spectrum Calibration Technique for Enhanced Sensitivity of Spectral Domain Optical Coherence Tomography. Sensors 2020, 20, 2067. https://doi.org/10.3390/s20072067
Han S, Wijesinghe RE, Jeon D, Han Y, Lee J, Lee J, Jo H, Lee D-E, Jeon M, Kim J. Optical Interferometric Fringe Pattern-Incorporated Spectrum Calibration Technique for Enhanced Sensitivity of Spectral Domain Optical Coherence Tomography. Sensors. 2020; 20(7):2067. https://doi.org/10.3390/s20072067
Chicago/Turabian StyleHan, Sangyeob, Ruchire Eranga Wijesinghe, Deokmin Jeon, Youngmin Han, Jaeyul Lee, Junsoo Lee, Hosung Jo, Dong-Eun Lee, Mansik Jeon, and Jeehyun Kim. 2020. "Optical Interferometric Fringe Pattern-Incorporated Spectrum Calibration Technique for Enhanced Sensitivity of Spectral Domain Optical Coherence Tomography" Sensors 20, no. 7: 2067. https://doi.org/10.3390/s20072067
APA StyleHan, S., Wijesinghe, R. E., Jeon, D., Han, Y., Lee, J., Lee, J., Jo, H., Lee, D.-E., Jeon, M., & Kim, J. (2020). Optical Interferometric Fringe Pattern-Incorporated Spectrum Calibration Technique for Enhanced Sensitivity of Spectral Domain Optical Coherence Tomography. Sensors, 20(7), 2067. https://doi.org/10.3390/s20072067








