Design of a Fully Integrated Inductive Coupling System: A Discrete Approach Towards Sensing Ventricular Pressure
Abstract
1. Introduction
2. Theoretical Inductive Power Transfer Design
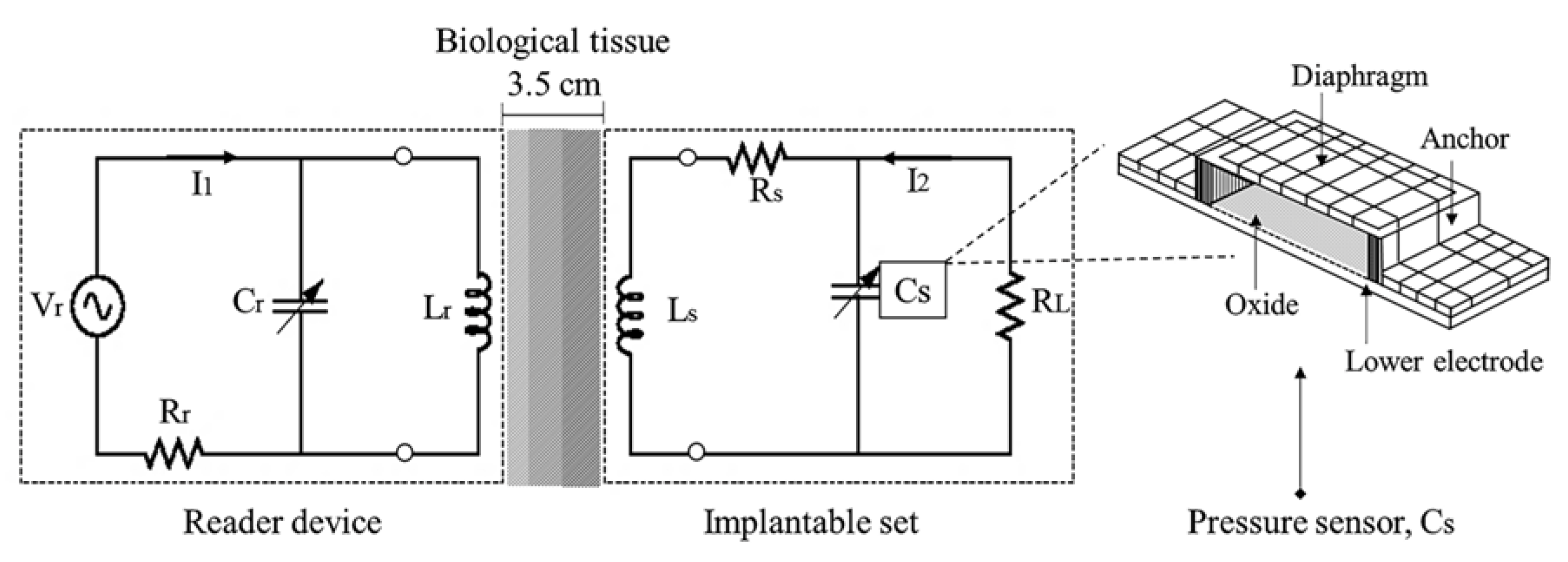
2.1. Mechanical Sensing Action
2.2. Inductive Power Calculation
2.3. Electromagnetic Flux Coupling
2.4. Electromagnetic Flux Calculation
2.4.1. RCL Implantable Set
2.4.2. RCL External Device
2.5. Results
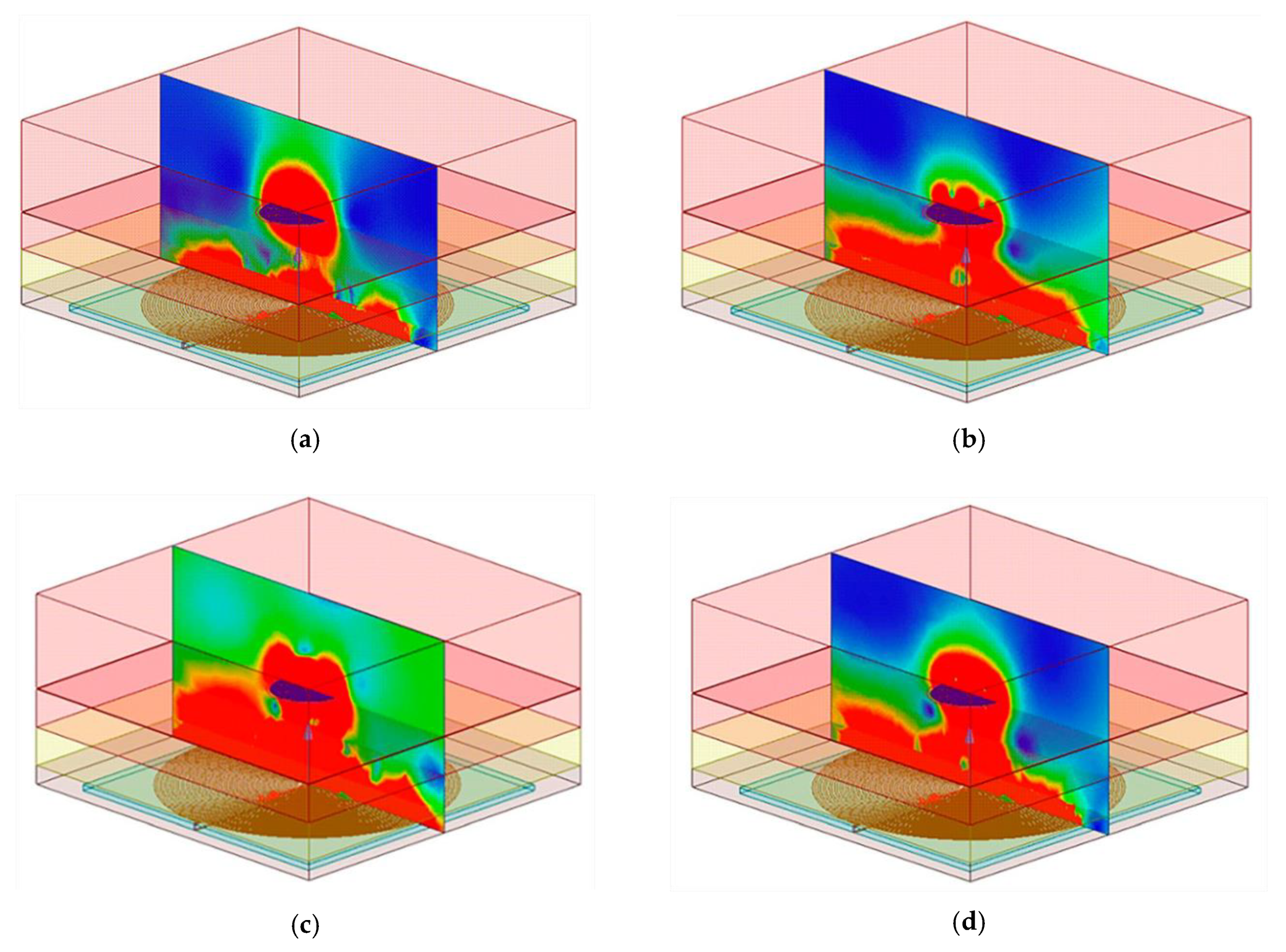
3. Electromagnetic Flux Simulation
4. Experimental Coupling Evaluation
4.1. Experimental Methodology
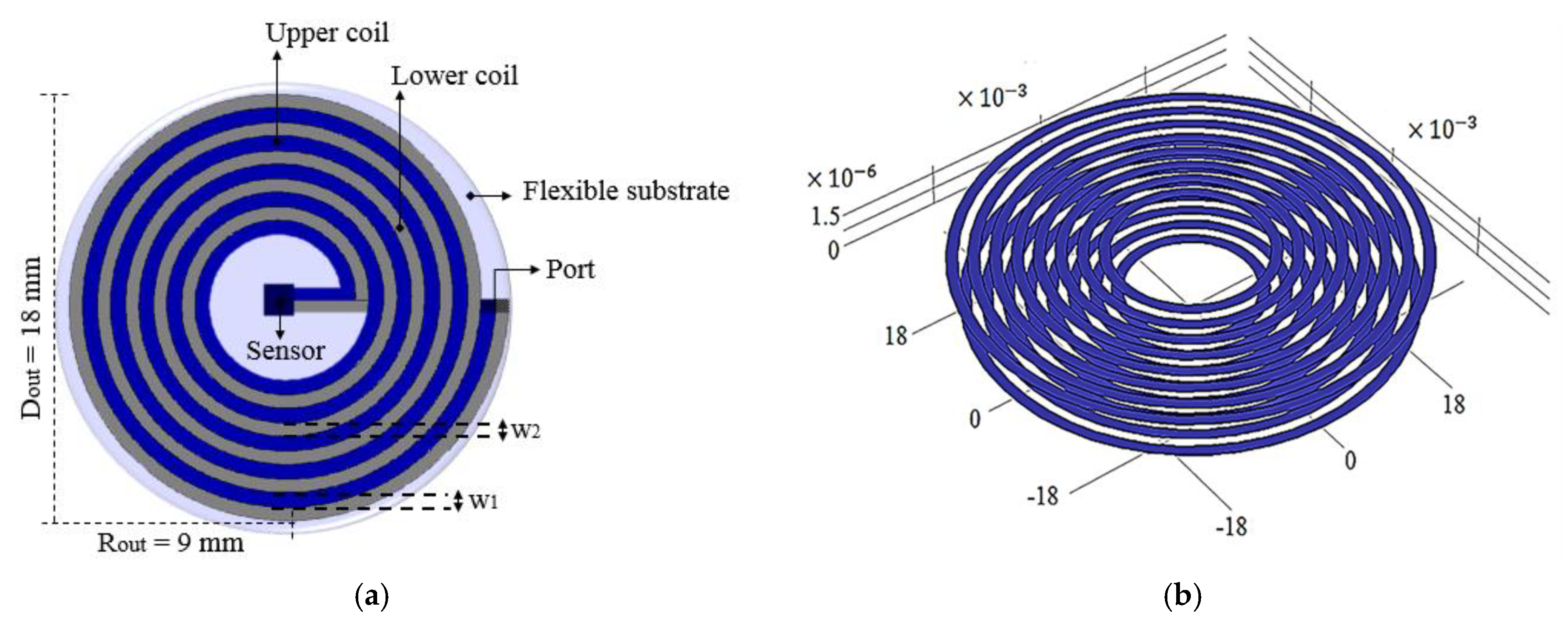
4.1.1. Implantable Coil Over PCB
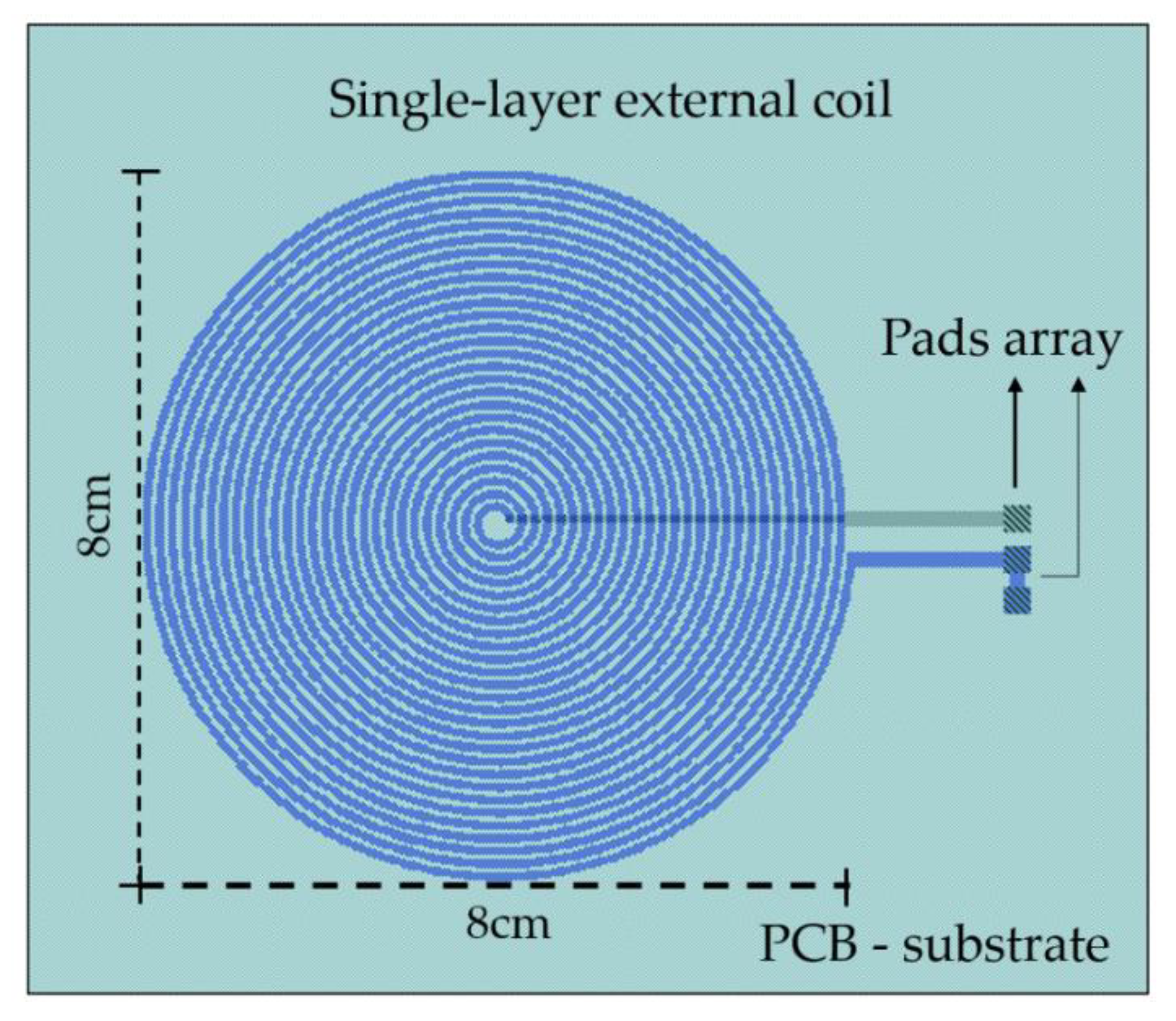
4.1.2. The External Module
4.2. Experimental Results
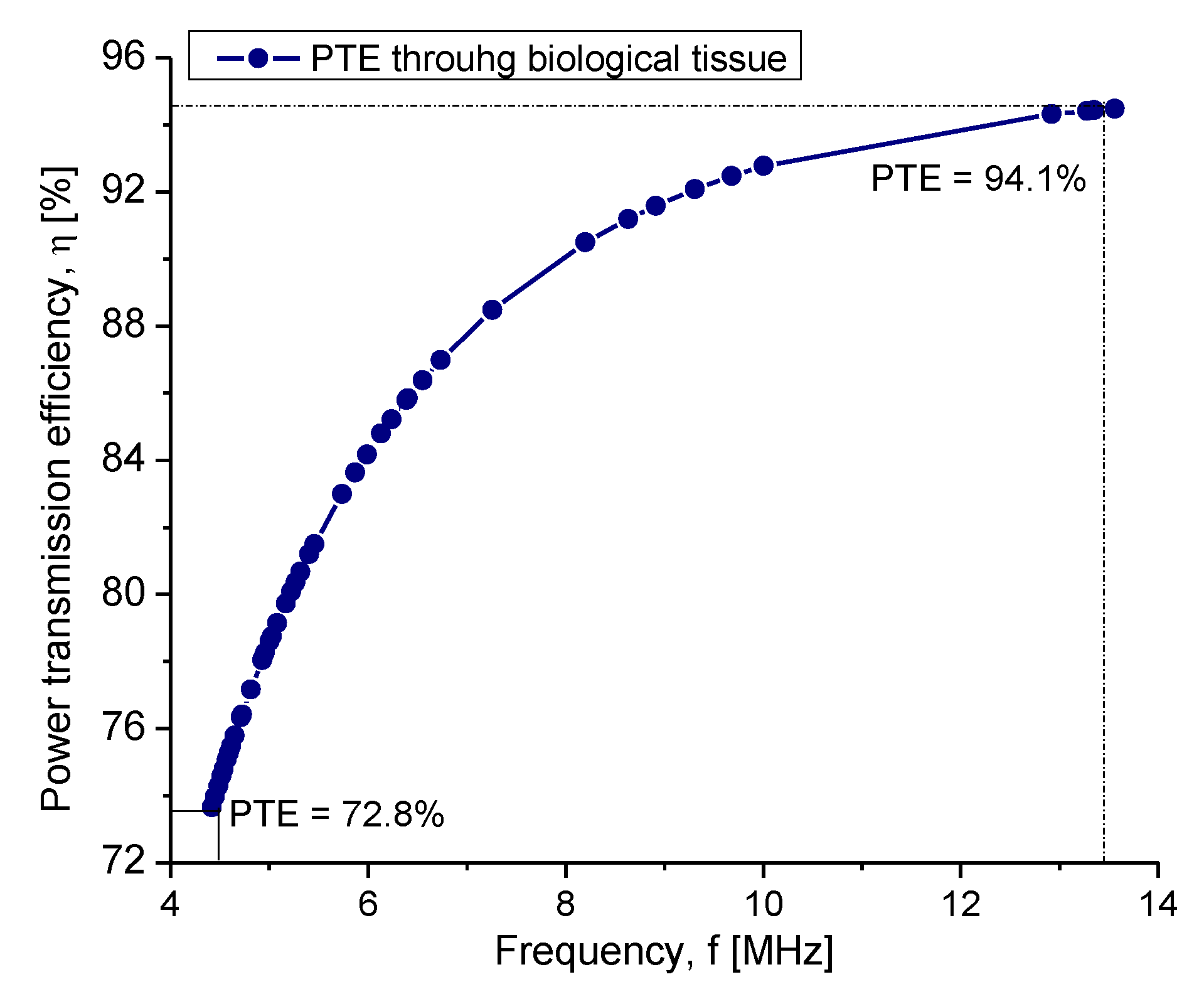
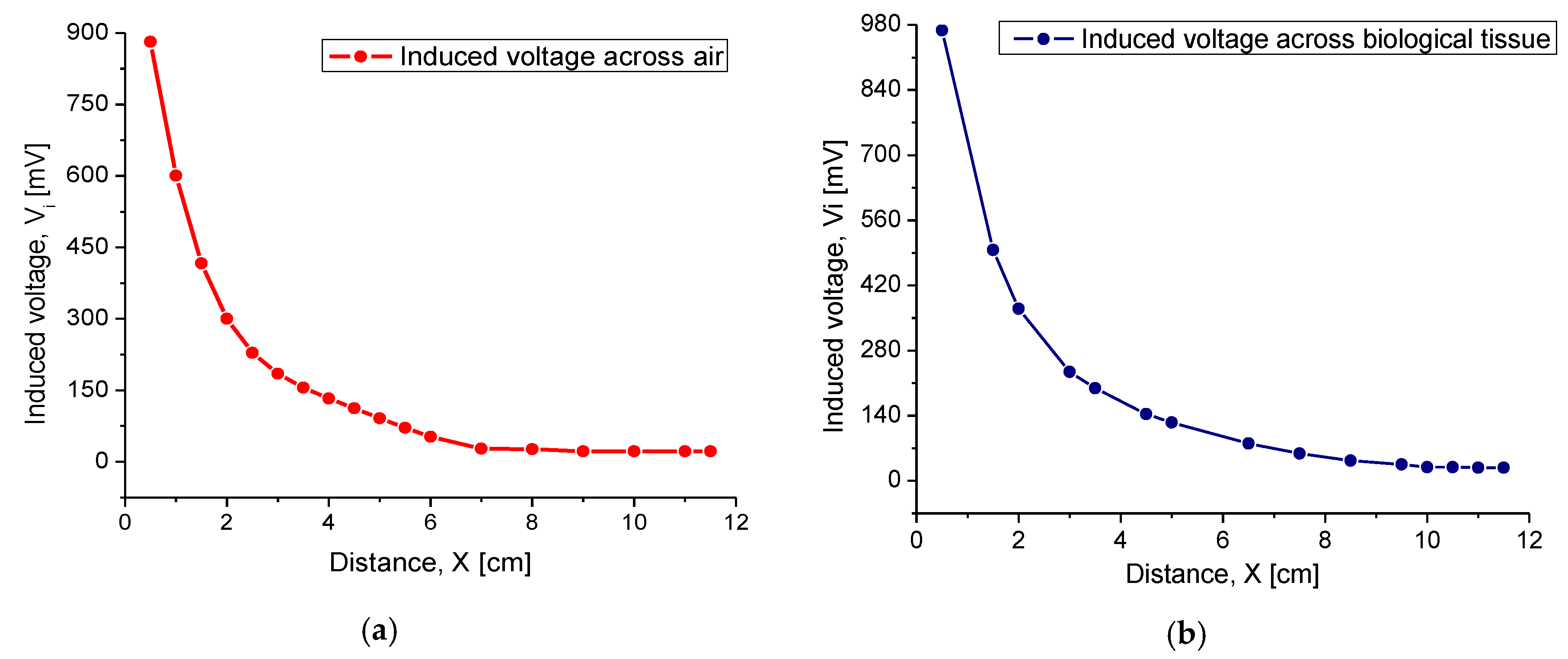
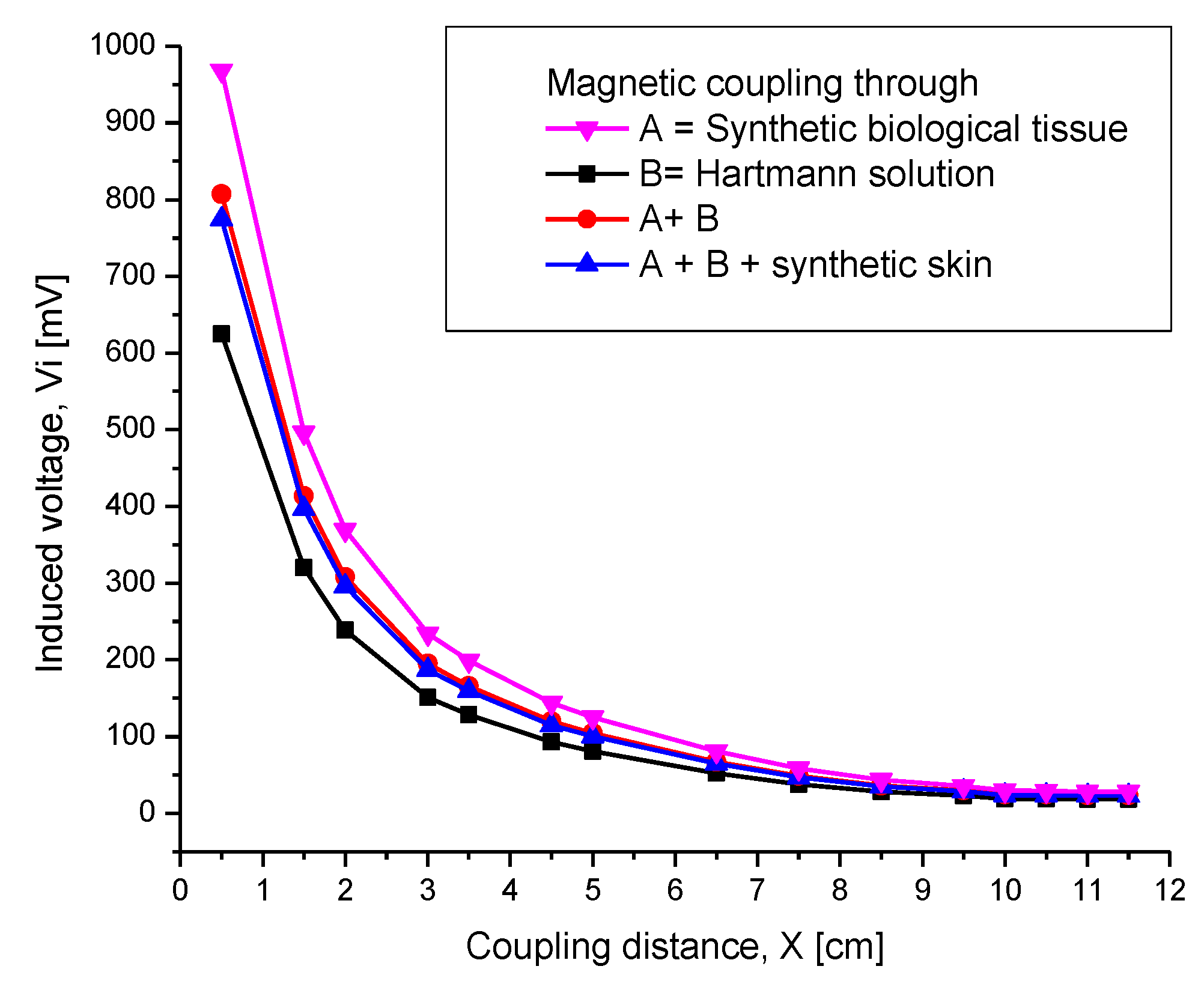
4.2.1. Inductive Coupling
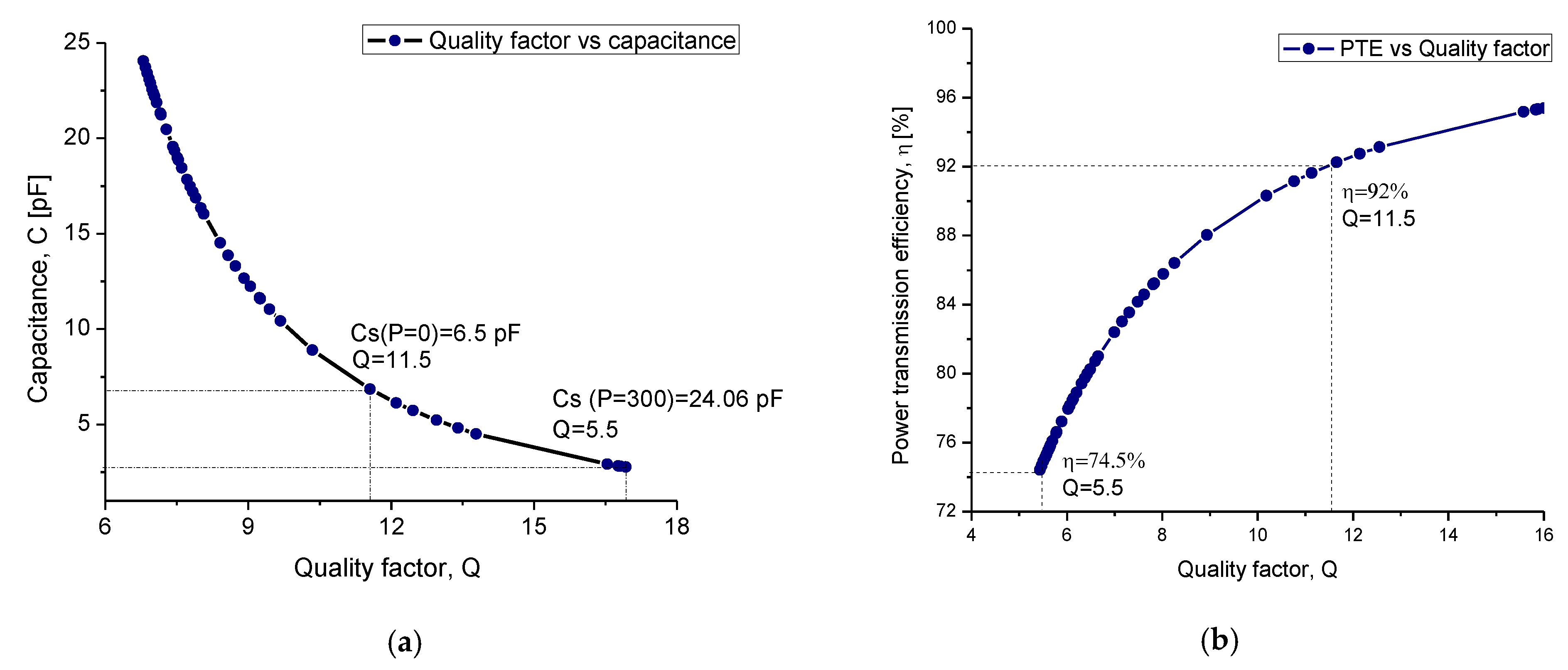
4.2.2. Capacitance Factor
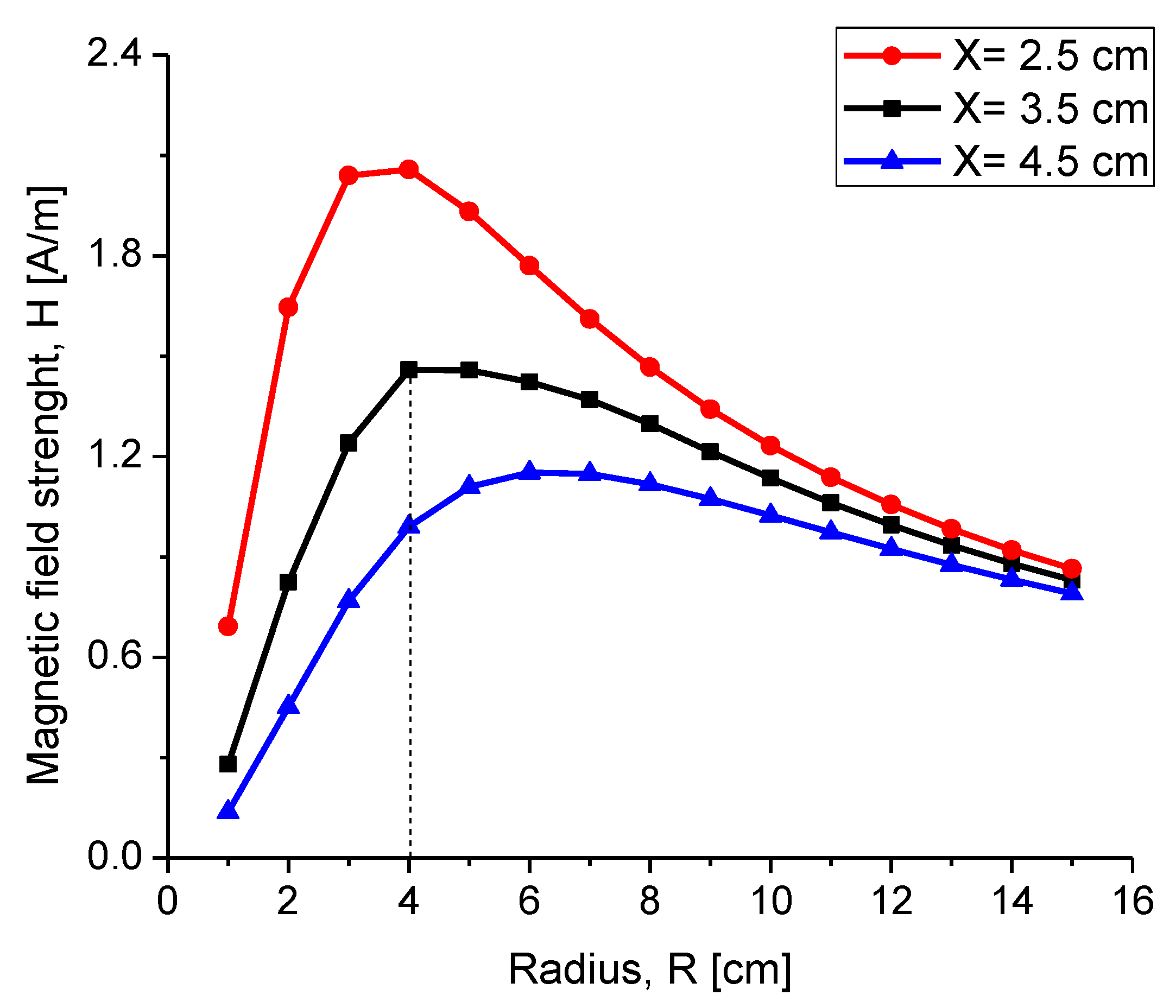
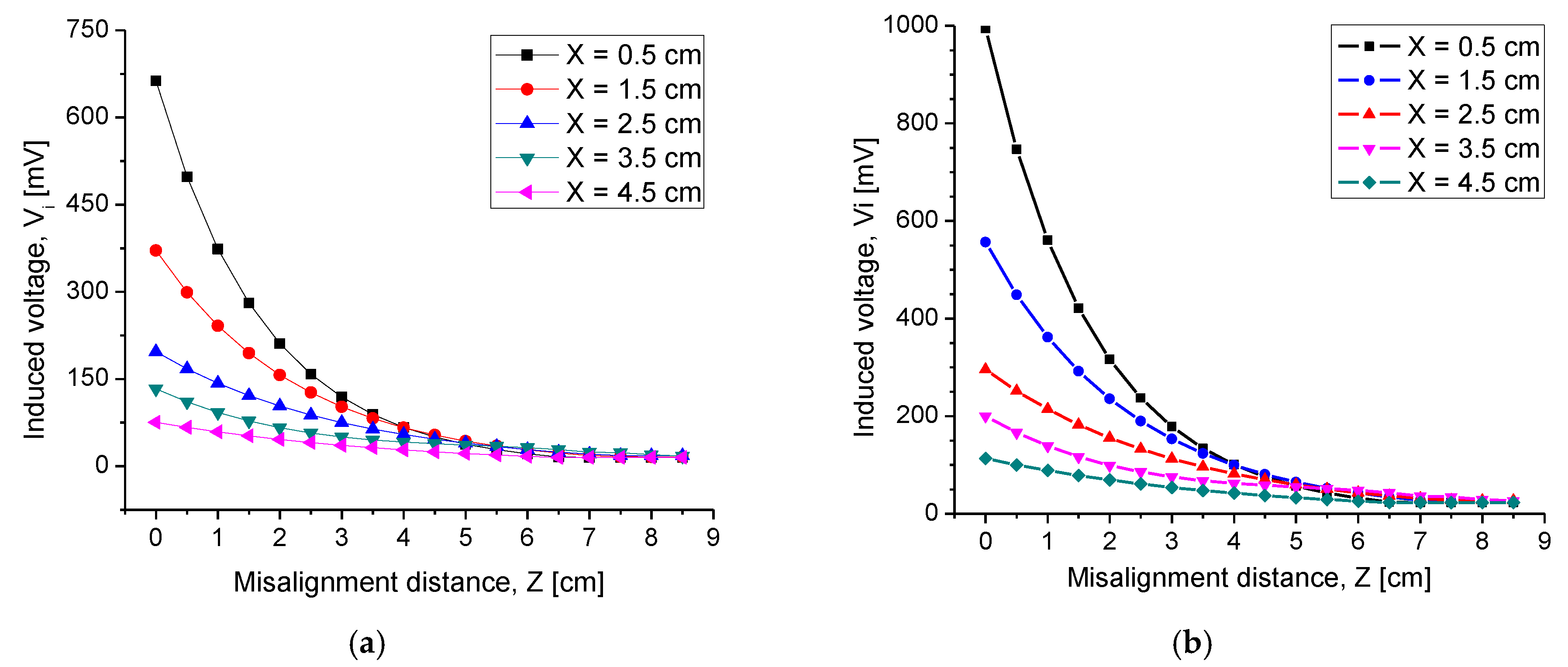
4.2.3. Misalignment Response
4.2.4. Combined Electromagnetic Core
5. Discussion
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Xue, R.; Cheng, K.; Je, M. High-Efficiency Wireless Power Transfer for Biomedical Implants by Optimal Resonant Load Transformation. IEEE Trans. Circuits Syst. I Regul. Pap. 2013, 60, 867–874. [Google Scholar] [CrossRef]
- Zeng, Y.; Qiu, D.; Meng, X.; Zhang, B.; Tang, S. Optimized Design of Coils for Wireless Power Transfer in Implanted Medical Devices. IEEE J. Electromagn. RF Microw. Med. Biol. 2018, 2, 277–285. [Google Scholar] [CrossRef]
- Lu, Y.; Ki, W.H. Introduction of Wireless Power Transfer. In CMOS Integrated Circuit Design for Wireless Power Transfer; Springer: Singapore, 2018. [Google Scholar]
- Senjuti, S.; Fricke, K.; Dounavis, A.; Sobot, R. Misalignment analysis of resonance-based wireless power transfer to biomedical implants. In Proceedings of the 25th IEEE Canadian Conference on Electrical Computer Engineering (CCECE), Montreal, QC, Canada, 29 April–2 May 2012; pp. 1–5. [Google Scholar]
- Harrison, R.; Kier, R.; Chestek, C.; Gilja, V.; Nuyujukian, P.; Ryu, S.; Greger, B.; Solzbacher, F.; Shenoy, K. Wireless neural recording with single low-power integrated circuit. IEEE Trans. Neural Syst. Rehab. Eng. 2009, 17, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Liu, M.; Tang, Z.; Ma, C. Optimal design of megahertz wireless power transfer systems for biomedical implants. In Proceedings of the 26th International Symposium on Industrial Electronics (ISIE), Edinburgh, Scotland, UK, 19–21 June 2017; pp. 805–810. [Google Scholar]
- Sauer, C.; Stanacevic, M.; Cauwenberghs, G.; Thakor, N. Power harvesting and telemetry in CMOS for implanted devices. IEEE Trans. Circuits Syst. I Regul. Pap. 2005, 52, 2605–2613. [Google Scholar] [CrossRef]
- Mehri, S.; Ammari, A.; Slama, J.; Sawan, M. Design Optimization of Multiple-Layer PSCs with Minimal Losses for Efficient and Robust Inductive Wireless Power Transfer. IEEE Access 2018, 6, 31924–31934. [Google Scholar] [CrossRef]
- Feenaghty, M.; Dahle, R. A compact and high-quality factor archimedean coil geometry for wireless power transfer. In Proceedings of the 2016 IEEE Wireless Power Transfer Conference (WPTC), Aveiro, Portugal, 5–6 May 2016; pp. 1–3. [Google Scholar]
- Trigui, A.; Hached, S.; Mounaim, F.; Ammari, A.; Sawan, M. Inductive power transfer system with self-calibrated primary resonant frequency. IEEE Trans. Power Electron. 2015, 30, 6078–6087. [Google Scholar] [CrossRef]
- Tang, S.; Lun, T.; Guo, Z.; Kwok, K.; McDannold, N. Intermediate Range Wireless Power Transfer with Segmented Coil Transmitters for Implantable Heart Pumps. IEEE Trans. Power Electron. 2017, 32, 3844–3857. [Google Scholar] [CrossRef]
- DeHennis, A.; Wise, K.D. A fully-integrated multisite pressure sensor for wireless arterial flow characterization. J. Microelectromech. Syst. 2004, 15, 168–171. [Google Scholar] [CrossRef]
- Zargham, M.; Gulak, P. Maximum Achievable Efficiency in Near-Field Coupled Power-Transfer Systems. IEEE Trans. Biomed. Circuits Syst. 2012, 6, 228–245. [Google Scholar] [CrossRef] [PubMed]
- Mark, M.; Björninen, T.; Ukkonen, L.; Sydänheimo, L.; Rabaey, J. SAR reduction and link optimization for mm-size remotely powered wireless implants using segmented loop antennas. In Proceedings of the IEEE Topical Conference on Biomedical Wireless Technologies, Networks, and Sensing Systems, Phoenix, AZ, USA, 16–20 January 2011; pp. 7–10. [Google Scholar]
- Islam, A.; Islam, B. Design of a Multi-Spiral Solenoidal Inductor for Inductive Power Transfer in Biomedical Applications. In Proceedings of the 27th Annual Review of Progress in Applied Computational Electromagnetics, Williamsburg, VA, USA, 27–31 March 2011; pp. 27–31. [Google Scholar]
- Hernández-Sebastián, N.; Diaz-Alonso, D.; Renero-Carrilo, F.J.; Villa-Villaseñor, N.; Calleja-Arriaga, W. Design and simulation and integrated wireless capacitive Sensors array for measuring Ventricular Pressure. Sensors 2018, 18, 2781. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Kim, B.J.; Meng, E. Chronically Implanted Pressure Sensors: Challenges and State of the Field. Sensors 2014, 14, 20620–20644. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, M.; English, J.; Von Arx, M.; Allen, M. Wireless micromachined ceramic pressure sensor for high-temperature applications. J. Microelectromech. Syst. 2002, 11, 337–343. [Google Scholar] [CrossRef]
- Ho, J.; Kim, S.; Poon, A. Midfield Wireless Powering for Implantable Systems. Proc. IEEE 2013, 101, 1369–1378. [Google Scholar] [CrossRef]
- Hannan, M.A.; Mutashar, S.; Abdul, S.; Hussain, A. Energy harvesting for the implantable biomedical devices: Issues and challenges. Biomed. Eng. 2014, 13, 79. [Google Scholar] [CrossRef] [PubMed]
- Adrián, E.; Rendon, N.; Alejandro, J.; Rivera, L.; Calleja, W.; Gil, F.; Díaz, D. Study of the Effect of Distance and Misalignment between Magnetically Coupled Coils for Wireless Power Transfer in Intraocular Pressure Measurement. Sci. World J. 2014, 2014, 692434. [Google Scholar]
- Mutashar, S.; Hannan, M.A.; Samad, S.A.; Hussain, A. Analysis and Optimization of Spiral Circular Inductive Coupling Link for Bio-Implanted Applications on Air and within Human Tissue. Sensors 2014, 14, 11522–11541. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J. A new calculation for designing multilayer planar spiral inductors. EDN 2010, 29, 37–40. [Google Scholar]
- Cinematica. Available online: http://www.sc.ehu.es/sbweb/fisica_/cinematica/circular/casete/casete2.html (accessed on 13 December 2019).
- Finkenzeller, K. RFID Handbook: Fundamentals and Applications in Contactless Smart Cards and Identification, 2nd ed.; Wiley: New York, NY, USA, 2003. [Google Scholar]


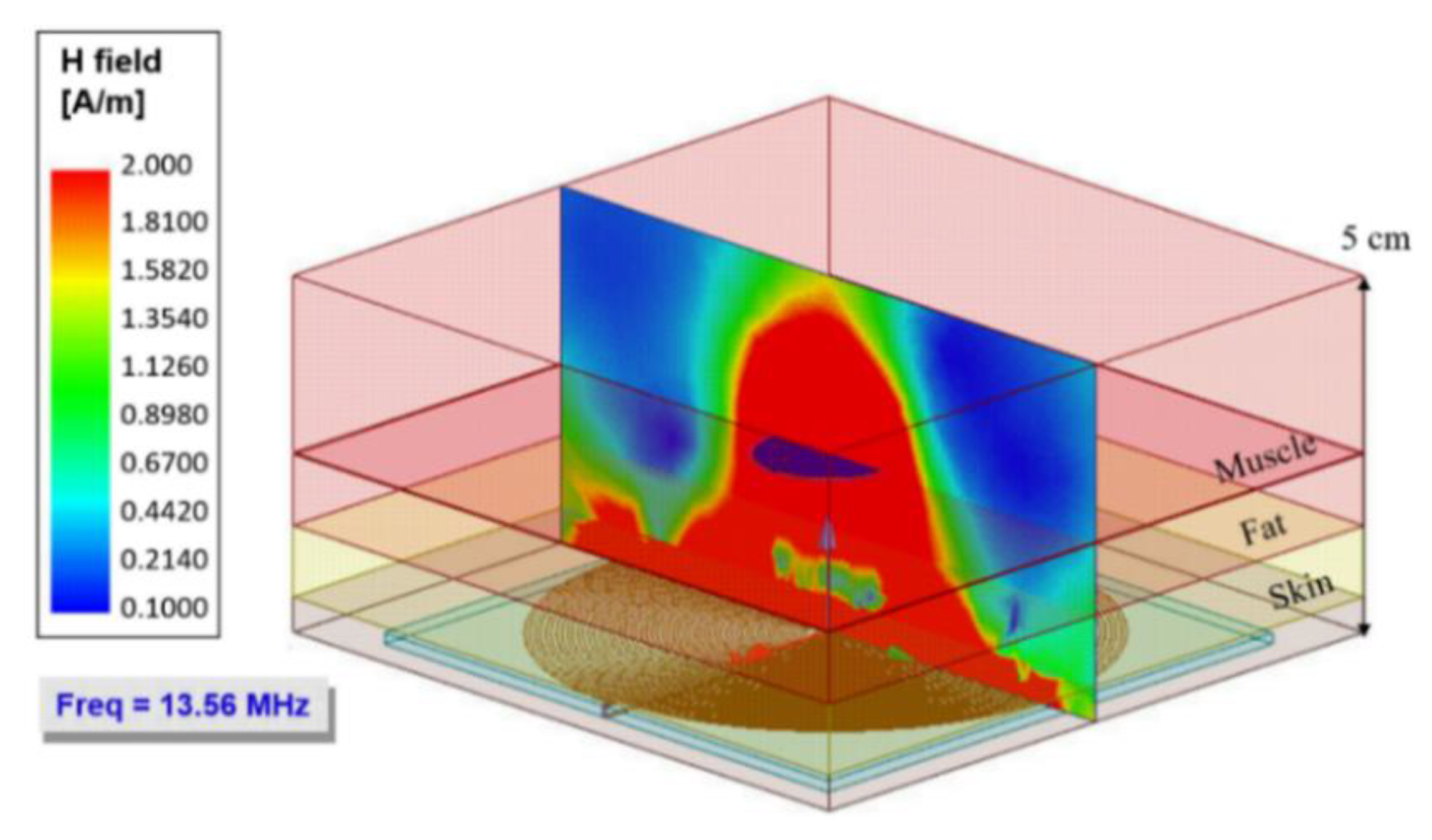
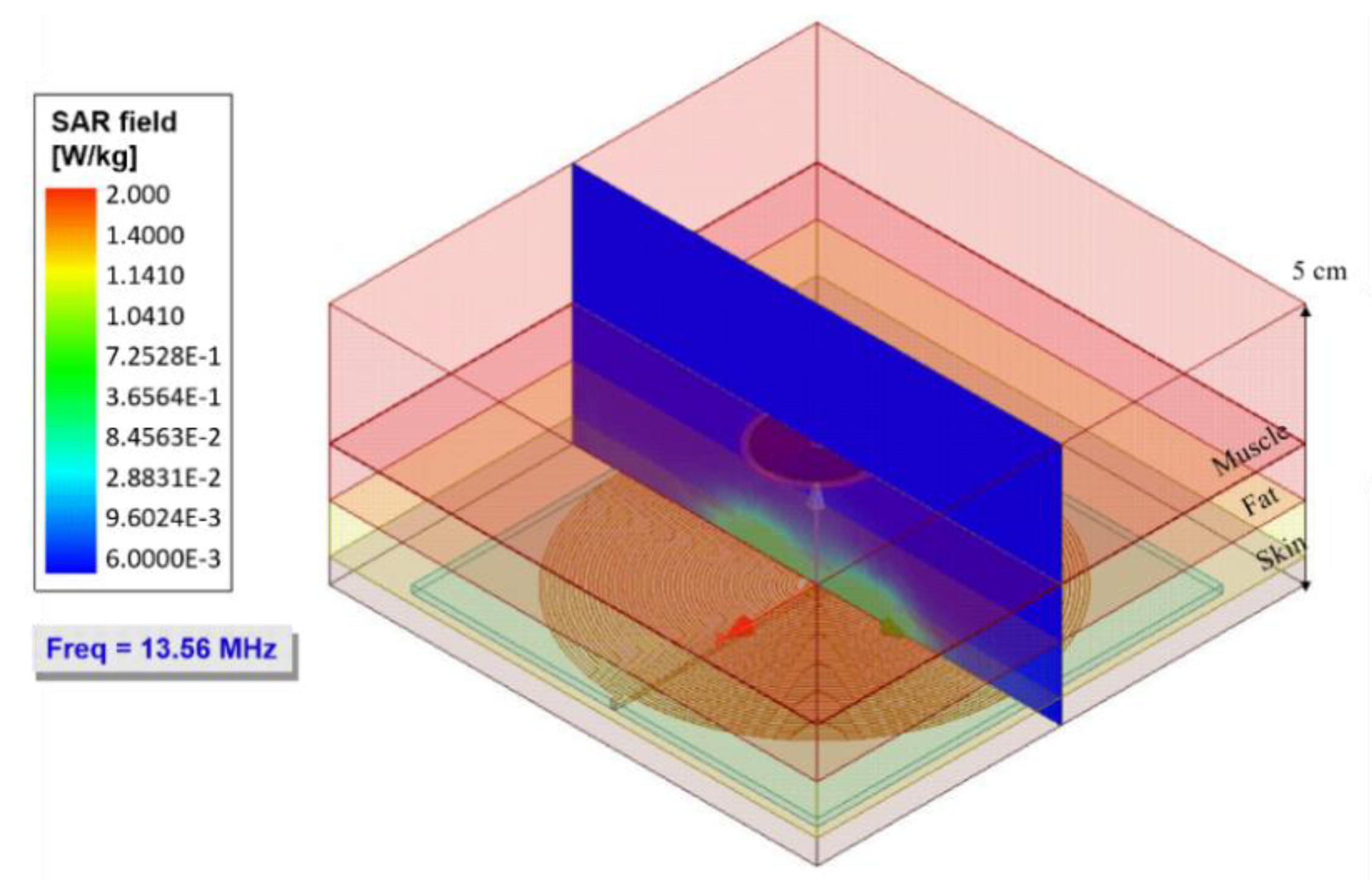


| Model | Thickness [cm] | Conductivity [Sm−1] | Relative Permittivity | Wavelength [m] |
|---|---|---|---|---|
| Skin | 0.5 | 0.38421 | 177.13 | 2.87 |
| Fat | 1 | 0.030354 | 11.827 | 11.11 |
| Muscle | 2 | 0.62818 | 138.44 | 3.24 |
| Quantity | Symbol | Internal Coil | External Coil |
|---|---|---|---|
| Internal diameter | Din | 2 mm | 2 mm |
| External diameter | Dout | 18 mm | 8 cm |
| Width | w | 160 μm | 700 μm |
| Thickness | h | 1.5 μm and 1 μm | 35 μm |
| Number of turns | N | 28 each loop | 27 |
| length | l | 1.14 m | 1.77 m |
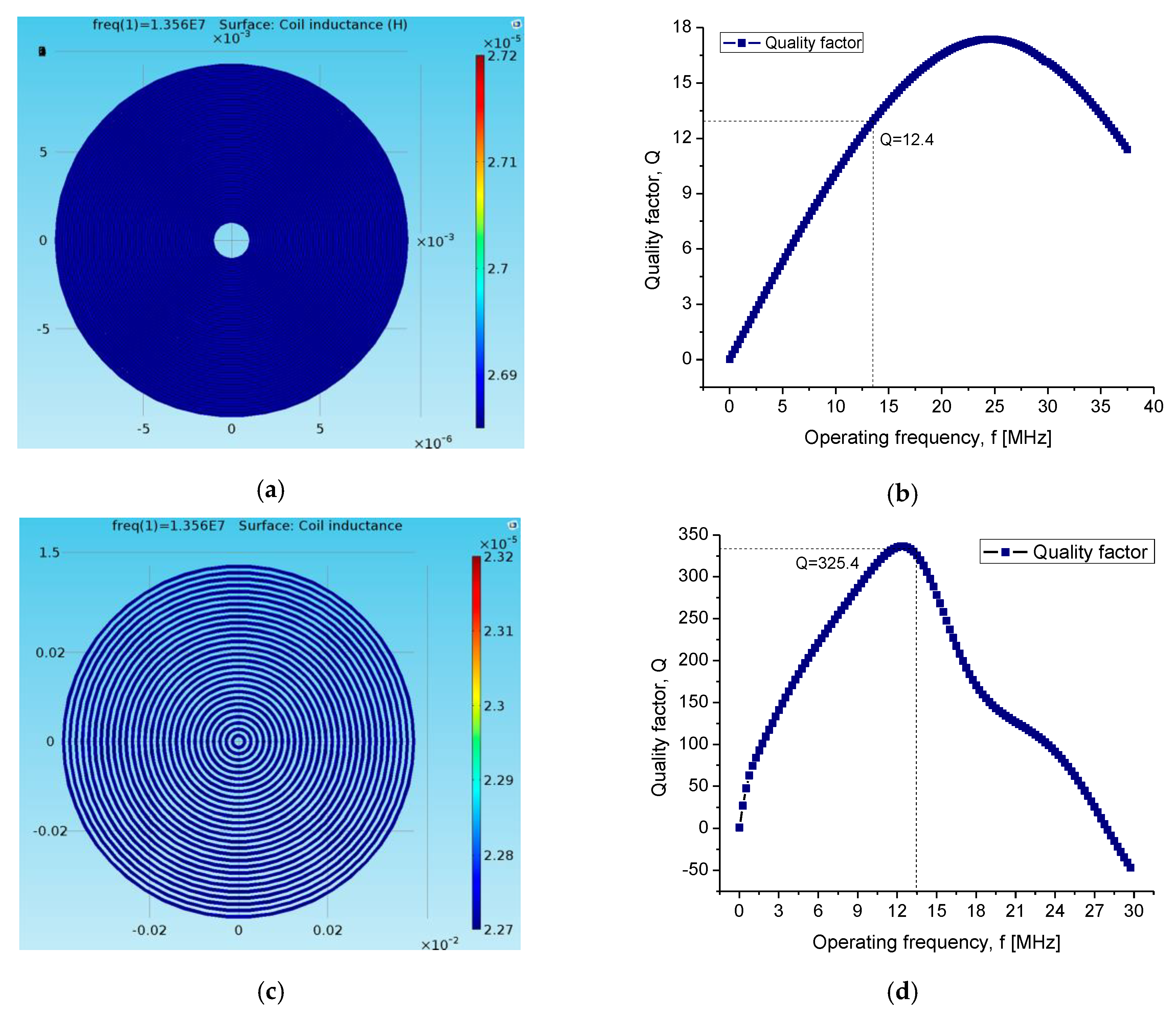
| Self-inductance | L | 20.98 μH | 21.24 μH |
| Electrical resistance | R | 171.86 Ω | 5.6 Ω |
| Quality factor | Q | 11.5 | 354 |
| Operating frequency | fs | 13.56 MHz | |
| Radiation distance | X | 3.5 cm | |
| Coupling coefficient | k | 0.16 | |
| Mutual inductance | M | 3.79 μH | |
| Power transmission efficiency | η | 92.%–74.5% | |
| Parameter | Symbol | Magnitude |
|---|---|---|
| Internal diameter | Din | 2 mm |
| External diameter | Dout | 3.8 cm |
| Width | w | 250 µm |
| Separation | S | 250 µm |
| Thickness | h | 35 µm |
| Number turns | N | 37 |
| Length | l | 1.3 m |
| Self-inductance | L | 21.29 µH |
| Resistance | R | 6.9 Ω |
| Component | Magnitude | Units | |
|---|---|---|---|
| Resistances | |||
| Capacitances | |||
| Inductances | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández Sebastián, N.; Villa Villaseñor, N.; Renero-Carrillo, F.-J.; Díaz Alonso, D.; Calleja Arriaga, W. Design of a Fully Integrated Inductive Coupling System: A Discrete Approach Towards Sensing Ventricular Pressure. Sensors 2020, 20, 1525. https://doi.org/10.3390/s20051525
Hernández Sebastián N, Villa Villaseñor N, Renero-Carrillo F-J, Díaz Alonso D, Calleja Arriaga W. Design of a Fully Integrated Inductive Coupling System: A Discrete Approach Towards Sensing Ventricular Pressure. Sensors. 2020; 20(5):1525. https://doi.org/10.3390/s20051525
Chicago/Turabian StyleHernández Sebastián, Natiely, Noé Villa Villaseñor, Francisco-Javier Renero-Carrillo, Daniela Díaz Alonso, and Wilfrido Calleja Arriaga. 2020. "Design of a Fully Integrated Inductive Coupling System: A Discrete Approach Towards Sensing Ventricular Pressure" Sensors 20, no. 5: 1525. https://doi.org/10.3390/s20051525
APA StyleHernández Sebastián, N., Villa Villaseñor, N., Renero-Carrillo, F.-J., Díaz Alonso, D., & Calleja Arriaga, W. (2020). Design of a Fully Integrated Inductive Coupling System: A Discrete Approach Towards Sensing Ventricular Pressure. Sensors, 20(5), 1525. https://doi.org/10.3390/s20051525





