A Carbamoylase-Based Bioassay for the Detection of Paralytic Shellfish Poisoning Toxins
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Extraction and Purification of PST Transforming Enzymes
2.3. Enzyme Characterization
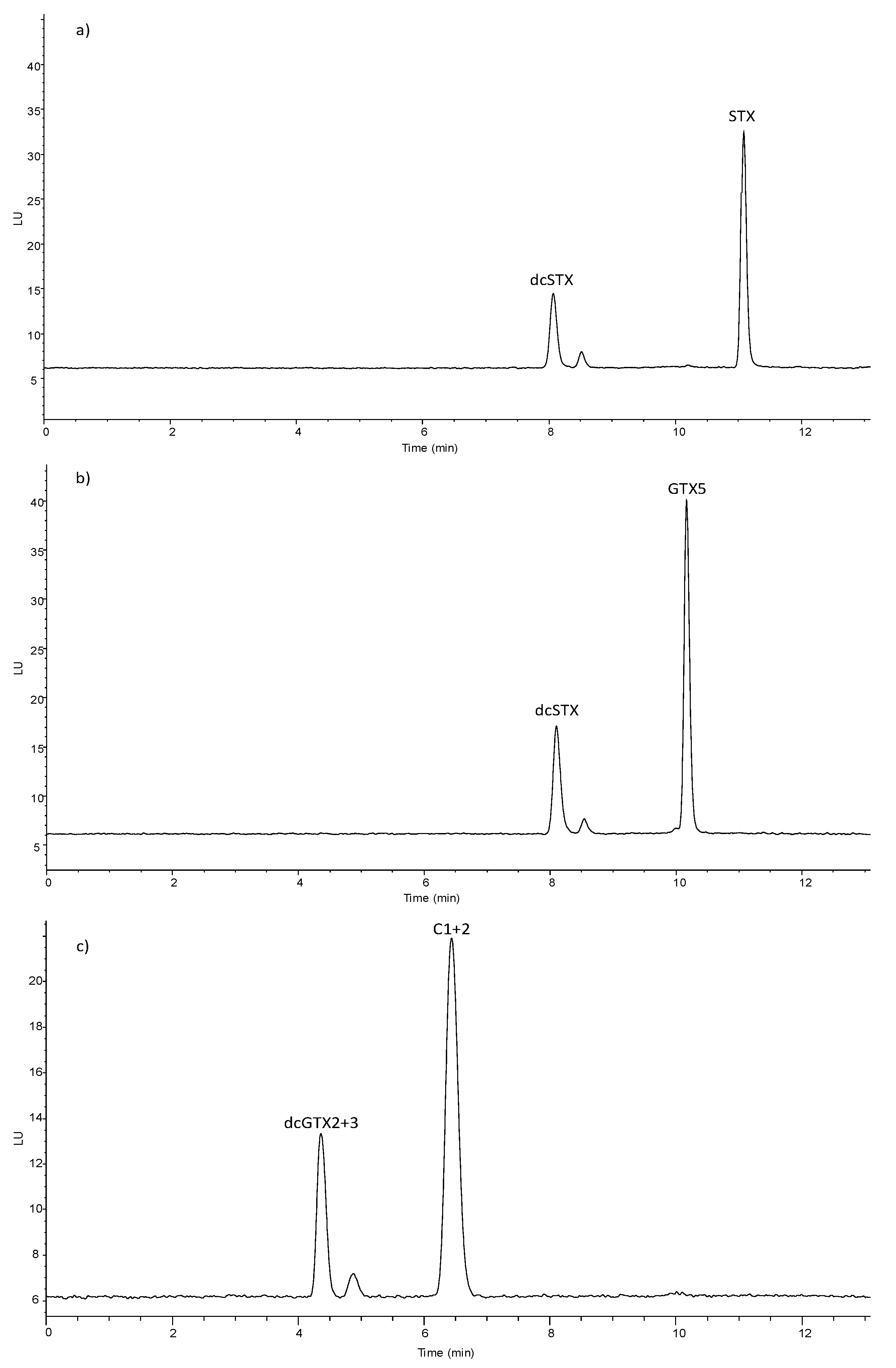
2.4. PSTs’ Quantification
2.5. Sensor Fabrication and Potentiometric Measurements
3. Results and Discussion
3.1. Carbamoylase Characterization
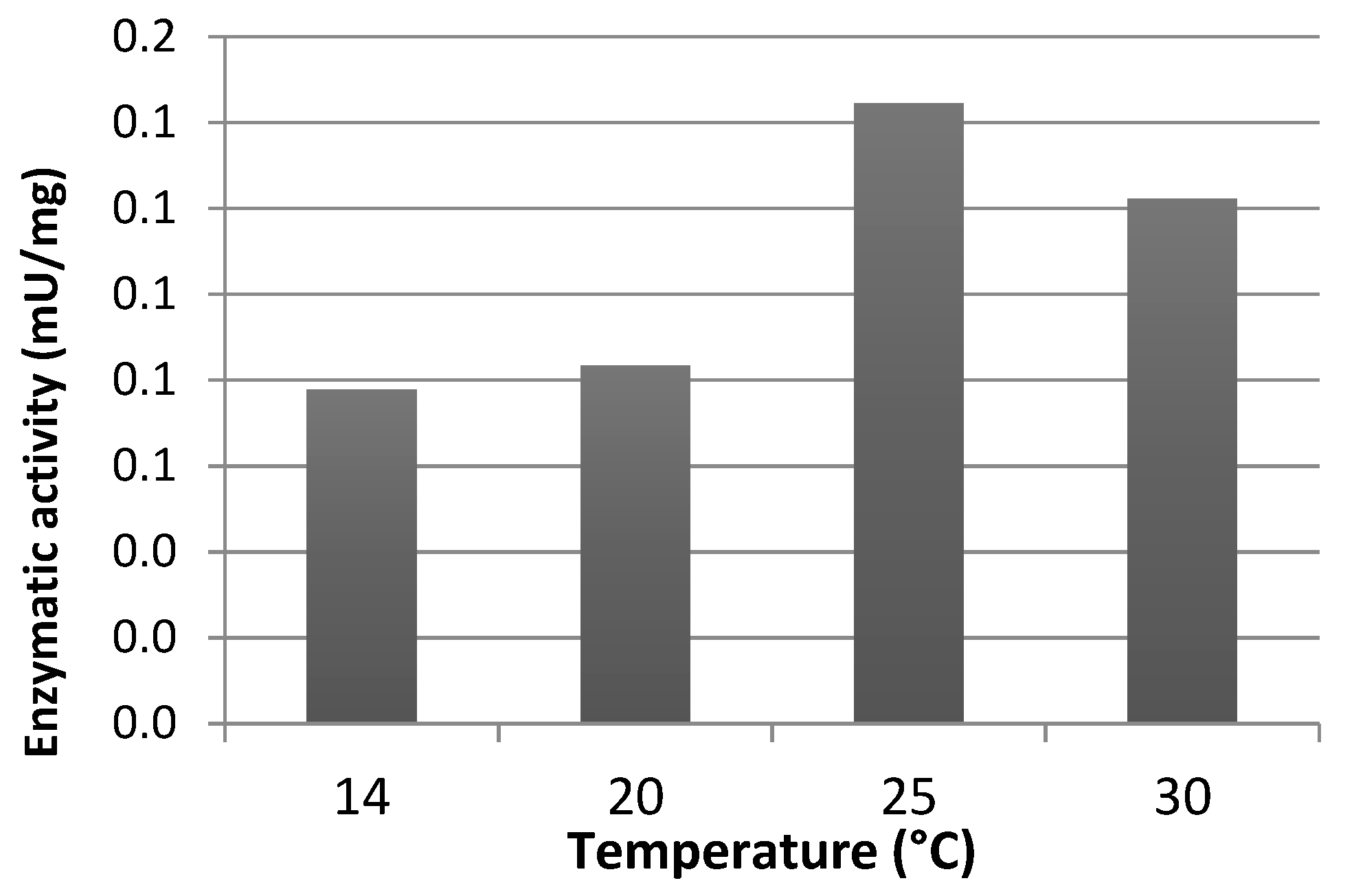
3.2. Effect of Experimental Conditions on Enzymatic Activity
3.3. Enzymatic Assay with Potentiometric Detection
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gerssen, A.; Pol-Hofstad, I.E.; Poelman, M.; Mulder, P.P.J.; Van Den Top, H.J.; Dde Boer, J. Marine Toxins: Chemistry, Toxicity, Occurrence and Detection, with Special Reference to the Dutch Situation. Toxins 2010, 2, 878–904. [Google Scholar] [CrossRef]
- Zingone, A.; Oksfeldt Enevoldsen, H. The Diversity of Harmful Algal Blooms: A Challenge for Science and Management. Ocean Coast. Manag. 2000, 43, 725–748. [Google Scholar] [CrossRef]
- Anderson, D.M. Approaches to Monitoring, Control and Management of Harmful Algal Blooms (HABs). Ocean Coast. Manag. 2009, 52, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Anon. Regulation (EC) no. 854/2004 of the European Parliament and of the Council of 29 April 2004. Off. J. Eur. Communities 2004, L139, 88–102. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32004R0854&from=EN (accessed on 15 January 2020).
- Llewellyn, L.E. Saxitoxin, a Toxic Marine Natural Product That Targets a Multitude of Receptors. Nat. Prod. Rep. 2006, 23, 200. [Google Scholar] [CrossRef]
- Vale, P.; Botelho, M.J.; Rodrigues, S.M.; Gomes, S.S.; Sampayo, M.A.D.M. Two Decades of Marine Biotoxin Monitoring in Bivalves from Portugal (1986–2006): A Review of Exposure Assessment. Harmful Algae 2008, 7, 11–25. [Google Scholar] [CrossRef]
- Visciano, P.; Schirone, M.; Berti, M.; Milandri, A.; Tofalo, R.; Suzzi, G. Marine Biotoxins: Occurrence, Toxicity, Regulatory Limits and Reference Methods. Front. Microbiol. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Stewart, I.; McLeod, C. The Laboratory Mouse in Routine Food Safety Testing for Marine Algal Biotoxins and Harmful Algal Bloom Toxin Research: Past, Present and Future. J. AOAC Int. 2014, 97, 356–372. [Google Scholar] [CrossRef]
- Lawrence, J.F.; Niedzwiadek, B.; Menard, C. Quantitative Determination of Paralytic Shellfish Poisoning. J. AOAC Int. 2005, 88, 1714–1732. [Google Scholar]
- Humpage, A.R.; Magalhaes, V.F.; Froscio, S.M. Comparison of Analytical Tools and Biological Assays for Detection of Paralytic Shellfish Poisoning Toxins. Anal. Bioanal. Chem. 2010, 397, 1655–1671. [Google Scholar] [CrossRef]
- Campbell, K.; Vilariño, N.; Botana, L.M.; Elliott, C.T. A European Perspective on Progress in Moving Away from the Mouse Bioassay for Marine-Toxin Analysis. TrAC Trends Anal. Chem. 2011, 30, 239–253. [Google Scholar] [CrossRef]
- Fonfría, E.S.; Vilariño, N.; Campbell, K.; Elliott, C.; Haughey, S.A.; Ben-Gigirey, B.; Vieites, J.M.; Kawatsu, K.; Botana, L.M. Paralytic Shellfish Poisoning Detection by Surface Plasmon Resonance-Based Biosensors in Shellfish Matrixes. Anal. Chem. 2007, 79, 6303–6311. [Google Scholar] [CrossRef]
- Campbell, K.; Huet, A.C.; Charlier, C.; Higgins, C.; Delahaut, P.; Elliott, C.T. Comparison of ELISA and SPR Biosensor Technology for the Detection of Paralytic Shellfish Poisoning Toxins. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2009, 877, 4079–4089. [Google Scholar] [CrossRef]
- Yakes, B.J.; Prezioso, S.; Haughey, S.A.; Campbell, K.; Elliott, C.T.; Degrasse, S.L. An Improved Immunoassay for Detection of Saxitoxin by Surface Plasmon Resonance Biosensors. Sens. Actuators B Chem. 2011, 156, 805–811. [Google Scholar] [CrossRef]
- McNamee, S.E.; Elliott, C.T.; Delahaut, P.; Campbell, K. Multiplex Biotoxin Surface Plasmon Resonance Method for Marine Biotoxins in Algal and Seawater Samples. Environ. Sci. Pollut. Res. 2013, 20, 6794–6807. [Google Scholar] [CrossRef]
- Haughey, S.A.; Campbell, K.; Yakes, B.J.; Prezioso, S.M.; Degrasse, S.L.; Kawatsu, K.; Elliott, C.T. Comparison of Biosensor Platforms for Surface Plasmon Resonance Based Detection of Paralytic Shellfish Toxins. Talanta 2011, 85, 519–526. [Google Scholar] [CrossRef]
- Dubois, M.; Demoulin, L.; Charlier, C.; Singh, G.; Godefroy, S.B.; Campbell, K.; Elliott, C.T.; Delahaut, P. Development of ELISAs for Detecting Domoic Acid, Okadaic Acid, and Saxitoxin and Their Applicability for the Detection of Marine Toxins in Samples Collected in Belgium. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2010, 27, 859–868. [Google Scholar] [CrossRef] [PubMed]
- Eangoor, P.; Indapurkar, A.S.; Vakkalanka, M.D.; Knaack, J.S. Multiplexed ELISA Screening Assay for Nine Paralytic Shellfish Toxins in Human Plasma. Analyst 2019, 144, 4702–4707. [Google Scholar] [CrossRef] [PubMed]
- Fraga, M.; Vilariño, N.; Louzao, M.C.; Campbell, K.; Elliott, C.T.; Kawatsu, K.; Vieytes, M.R.; Botana, L.M. Detection of Paralytic Shellfish Toxins by a Solid-Phase Inhibition Immunoassay Using a Microsphere-Flow Cytometry System. Anal. Chem. 2012, 84, 4350–4356. [Google Scholar] [CrossRef] [PubMed]
- Meneely, J.P.; Campbell, K.; Greef, C.; Lochhead, M.J.; Elliott, C.T. Development and Validation of an Ultrasensitive Fluorescence Planar Waveguide Biosensor for the Detection of Paralytic Shellfish Toxins in Marine Algae. Biosens. Bioelectron. 2013, 41, 691–697. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Su, K.; Qiu, X.; Fang, J.; Zou, Q.; Wang, P. An Improved Efficient Biochemical Detection Method to Marine Toxins with a Smartphone-Based Portable System—Bionic e-Eye. Sens. Actuators B Chem. 2017, 238, 1165–1172. [Google Scholar] [CrossRef]
- Schulz, K.; Pöhlmann, C.; Dietrich, R.; Märtlbauer, E.; Elßner, T. Electrochemical Biochip Assays Based on Anti-Idiotypic Antibodies for Rapid and Automated on-Site Detection of Low Molecular Weight Toxins. Front. Chem. 2019, 7, 1–12. [Google Scholar] [CrossRef]
- Okumura, M.; Tsuzuki, H.; Tomita, B.-I. A Rapid Detection Method for Paralytic Shellfish Poisoning Toxins by Cell Bioassay. Toxicon 2005, 46, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, R.; Saito, H.; Okumura, M.; Kondo, F. Cell Bioassay for Paralytic Shellfish Poisoning (PSP): Comparison with Postcolumn Derivatization Liquid Chromatographic Analysis and Application to the Monitoring of PSP in Shellfish. J. Agric. Food Chem. 2006, 54, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Zou, L.; Wu, C.; Wang, Q.; Zhou, J.; Su, K.; Li, H.; Hu, N.; Wang, P. An Improved Sensitive Assay for the Detection of PSP Toxins with Neuroblastoma Cell-Based Impedance Biosensor. Biosens. Bioelectron. 2015, 67, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zheng, H.; Yi, X.; Shao, C.; Xiang, B.; Wang, S.; Zhao, Z.; Zhang, X.; Hui, G. Paralytic Shellfish Poisoning Toxin Detection Based on Cell-Based Sensor and Non-Linear Signal Processing Model. Int. J. Food Prop. 2019, 22, 890–897. [Google Scholar] [CrossRef]
- Van Dolah, F.M.; Fire, S.E.; Leighfield, T.A.; Mikulski, C.M.; Doucette, G.J. Determination of Paralytic Shellfish Toxins in Shellfish by Receptor Binding Assay: Collaborative Study. J. AOAC Int. 2012, 95, 795–812. [Google Scholar] [CrossRef]
- Turner, A.D.; Broadwater, M.; Van Dolah, F. Use of the Receptor Binding Assay for Determination of Paralytic Shellfish Poisoning Toxins in Bivalve Molluscs from Great Britain and the Assessment of Method Performance in Oysters. Toxicon 2018, 148, 155–164. [Google Scholar] [CrossRef]
- Bergantin, J.H.; Sevilla, F. Quartz Crystal Microbalance Biosensor for Saxitoxin Based on Immobilized Sodium Channel Receptors. Anal. Lett. 2010, 43, 476–486. [Google Scholar] [CrossRef]
- Cheun, B.S.; Loughran, M.; Hayashi, T.; Nagashima, Y.; Watanabe, E. Use of a Channel Biosensor for the Assay of Paralytic Shellfish Toxins. Toxicon 1998, 36, 1371–1381. [Google Scholar] [CrossRef]
- Llewellyn, L.E.; Doyle, J.; Jellett, J.; Barrett, R.; Alison, C.; Bentz, C.; Quilliam, M. Measurement of Paralytic Shellfish Toxins in Molluscan Extracts: Comparison of the Microtitre Plate Saxiphilin and Sodium Channel Radioreceptor Assays with Mouse Bioassay, HPLC Analysis and a Commercially Available Cell Culture Assay. Food Addit. Contam. 2001, 18, 970–980. [Google Scholar] [CrossRef]
- Campàs, M.; Prieto-Simón, B.; Marty, J.L. Biosensors to Detect Marine Toxins: Assessing Seafood Safety. Talanta 2007, 72, 884–895. [Google Scholar] [CrossRef] [PubMed]
- Saxitoxin Plate Kit, Beacon Analytical Systems Inc. Available online: https://d6a597b0-184e-4af7-b02d-98845136bc9b.filesusr.com/ugd/c8f857_36cebe2121c54f27ba0d6f70d5d3f0d8.pdf (accessed on 15 January 2020).
- Saxitoxin (PSP) ELISA, Microtiter plate, Abraxis Inc. Available online: https://www.abraxiskits.com/wp-content/uploads/2017/03/Saxitoxin-Users-Guide.pdf (accessed on 15 January 2020).
- Botelho, M.J.; Vale, C.; Grilo, R.V.; Ferreira, J.G. Uptake and Release of Paralytic Shellfish Toxins by the Clam Ruditapes Decussatus Exposed to Gymnodinium Catenatum and Subsequent Depuration. Mar. Environ. Res. 2012, 77, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Graham, H.W. Gymnodinium Catenatum, a New Dinoflagellate from the Gulf of California. Trans. Am. Microsc. Soc. 1943, 62, 259. [Google Scholar] [CrossRef]
- Hallegraeff, G.M. Harmful Algal Blooms: A Global Overview. In Manual on Harmful Marine Microalgae; Hallegraeff, G.M., Anderson, D.M., Cembella, A.D., Eds.; UNESCO Publishing: Paris, France, 2003; pp. 25–50. [Google Scholar]
- Cruz, M.G.N.; Ferreira, N.S.; Gomes, M.T.S.R.; Botelho, M.J.; Costa, S.T.; Vale, C.; Rudnitskaya, A. Determination of Paralytic Shellfish Toxins Using Potentiometric Electronic Tongue. Sens. Actuators B Chem. 2018, 263, 550–556. [Google Scholar] [CrossRef]
- Oshima, Y. Chemical and Enzymatic Transformation of Paralytic Shellfish Toxins in Marine Organisms. In Harmful Marine Algal Blooms; Lassus, P., Arzul, G., Erard, E., Gentien, C., Marcaillou, C., Eds.; Lavoisier, Intercept Ltd.: Paris, France, 1995; pp. 475–480. [Google Scholar]
- Fast, M.D.; Cembella, A.D.; Ross, N.W. In Vitro Transformation of Paralytic Shellfish Toxins in the Clams Mya Arenaria and Protothaca Staminea. Harmful Algae 2006, 5, 79–90. [Google Scholar] [CrossRef]
- Turner, A.D.; Lewis, A.M.; O’Neil, A.; Hatfield, R.G. Transformation of Paralytic Shellfish Poisoning Toxins in UK Surf Clams (Spisula Solida) for Targeted Production of Reference Materials. Toxicon 2013, 65, 41–58. [Google Scholar] [CrossRef]
- Lin, H.P.; Cho, Y.; Yashiro, H.; Yamada, T.; Oshima, Y. Purification and Characterization of Paralytic Shellfish Toxin Transforming Enzyme from Mactra Chinensis. Toxicon 2004, 44, 657–668. [Google Scholar] [CrossRef]
- Cho, Y.; Ogawa, N.; Takahashi, M.; Lin, H.; Oshima, Y. Purification and Characterization of Paralytic Shellfish Toxin-Transforming Enzyme, Sulfocarbamoylase I, from the Japanese Bivalve Peronidia Venulosa. Biochim. Biophys. Acta Proteins Proteom. 2008, 1784, 1277–1285. [Google Scholar] [CrossRef]
- Botelho, M.J.; Vale, C.; Ferreira, J.G. Profiles of Paralytic Shellfish Toxins in Bivalves of Low and Elevated Toxicities Following Exposure to Gymnodinium Catenatum Blooms in Portuguese Estuarine and Coastal Waters. Chemosphere 2015, 138, 1028–1036. [Google Scholar] [CrossRef]
- Artigas, M.L.; Vale, P.J.V.; Gomes, S.S.; Botelho, M.J.; Rodrigues, S.M.; Amorim, A. Profiles of Paralytic Shellfish Poisoning Toxins in Shellfish from Portugal Explained by Carbamoylase Activity. J. Chromatogr. A 2007, 1160, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of Structural Proteins during the Assembly of the Head of Bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, J.F.; Niedzwiadek, B.; Menard, C. Quantitative determination of paralytic shellfish poisoning toxins in shellfish using prechromatographic oxidation and liquid chromatography with fluorescence detection: Collaborative study. J. AOAC Int. 2005, 88, 1714–1732. [Google Scholar] [PubMed]
- Botelho, M.J.; Vale, C.; Mota, A.M.; Rodrigues, S.M.; Costa, P.R.; Gonçalves, M.L.S.S. Matrix Effect on Paralytic Shellfish Toxins Quantification and Toxicity Estimation in Mussels Exposed to Gymnodinium Catenatum. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2010, 27, 1724–1732. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.T.; Vale, C.; Raimundo, J.; Matias, D.; Botelho, M.J. Changes of Paralytic Shellfish Toxins in Gills and Digestive Glands of the Cockle Cerastoderma Edule under Post-Bloom Natural Conditions. Chemosphere 2016, 149, 351–357. [Google Scholar] [CrossRef]
- Ferreira, N.S.; Cruz, M.G.N.; Gomes, M.T.S.R.; Rudnitskaya, A. Potentiometric Chemical Sensors for the Detection of Paralytic Shellfish Toxins. Talanta 2018, 181, 380–384. [Google Scholar] [CrossRef]
- Paralytic Shellfish Poisoning and other Polar Toxins, National Research Council, Canada. Available online: https://www.nrc-cnrc.gc.ca/eng/solutions/advisory/crm/list_product.html#B-PSP (accessed on 15 January 2020).
- Martínez-Rodríguez, S.; Martínez-Gómez, A.I.; Rodríguez-Vico, F.; Clemente-Jiménez, J.M.; Las Heras-Vázquez, F.J. Carbamoylases: Characteristics and Applications in Biotechnological Processes. Appl. Microbiol. Biotechnol. 2010, 85, 441–458. [Google Scholar] [CrossRef]
- Trojanowicz, M.; Krawczyński Vel Krawczyk, T.; Zmorzyńska, M.; Campanella, L. Amperometric Sensing of Ammonia in Aqueous Solutions Using a Polyaniline-Modified Electrode in Flow Injection Systems. Electroanalysis 1997, 9, 1062–1066. [Google Scholar] [CrossRef]



| Extract | Activity, mU/mg |
|---|---|
| Digestive gland, crude | 1.1 ± 2.0 |
| Digestive gland, purified | 162.7 ± 1.1 |
| Crystalline style, crude | 33.5 ± 0.3 |
| Crystalline style, purified | 478.5 ± 0.5 |
| Toxin | Specific Activity, mU/mg | Substrate Consumption, % |
|---|---|---|
| STX | 38 ± 3 | 78 |
| GTX5 | 37 ± 1 | 60 |
| C1+2 | 35 ± 2 | 81 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raposo, M.; Botelho, M.J.; Costa, S.T.; Gomes, M.T.S.R.; Rudnitskaya, A. A Carbamoylase-Based Bioassay for the Detection of Paralytic Shellfish Poisoning Toxins. Sensors 2020, 20, 507. https://doi.org/10.3390/s20020507
Raposo M, Botelho MJ, Costa ST, Gomes MTSR, Rudnitskaya A. A Carbamoylase-Based Bioassay for the Detection of Paralytic Shellfish Poisoning Toxins. Sensors. 2020; 20(2):507. https://doi.org/10.3390/s20020507
Chicago/Turabian StyleRaposo, Mariana, Maria João Botelho, Sara T. Costa, Maria Teresa S. R. Gomes, and Alisa Rudnitskaya. 2020. "A Carbamoylase-Based Bioassay for the Detection of Paralytic Shellfish Poisoning Toxins" Sensors 20, no. 2: 507. https://doi.org/10.3390/s20020507
APA StyleRaposo, M., Botelho, M. J., Costa, S. T., Gomes, M. T. S. R., & Rudnitskaya, A. (2020). A Carbamoylase-Based Bioassay for the Detection of Paralytic Shellfish Poisoning Toxins. Sensors, 20(2), 507. https://doi.org/10.3390/s20020507







