The Potential Role of Sensors, Wearables and Telehealth in the Remote Management of Diabetes-Related Foot Disease
Abstract
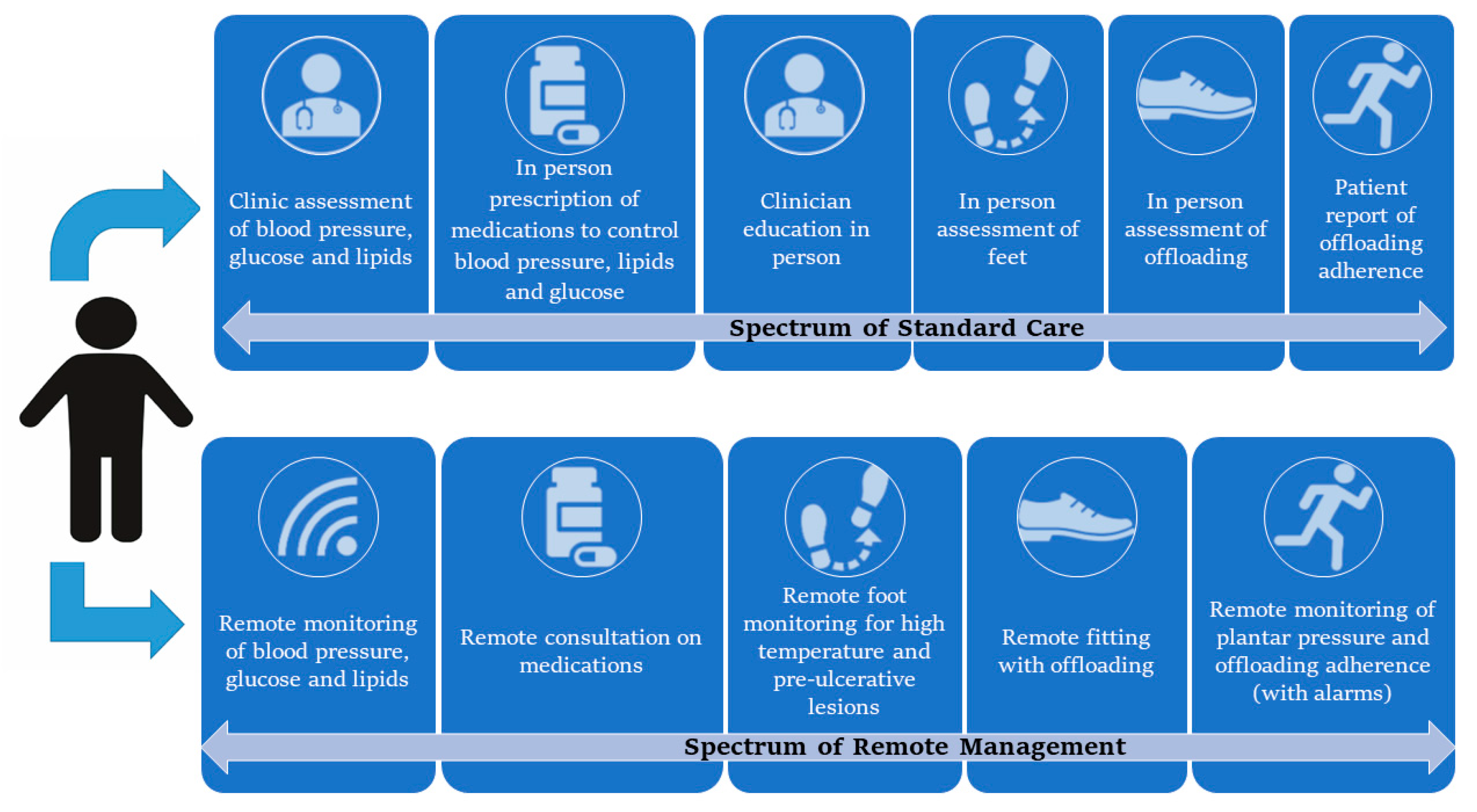
1. Introduction
2. Monitoring Foot Temperature
3. Monitoring Plantar Pressures
4. Offloading Footwear Adherence Monitoring
5. Remotely Monitoring Medical Management
6. Wearables for Assessing Sensation, Peripheral Perfusion and Gait
7. Telehealth
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, Y.; Lazzarini, P.A.; McPhail, S.M.; Van Netten, J.J.; Armstrong, D.G.; Pacella, R.E. Global Disability Burdens of Diabetes-Related Lower-Extremity Complications in 1990 and 2016. Diabetes Care 2020, 43, 964–974. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.G.; Boulton, A.J.M.; Bus, S.A. Diabetic Foot Ulcers and Their Recurrence. N. Engl. J. Med. 2017, 376, 2367–2375. [Google Scholar] [CrossRef] [PubMed]
- Heart Protection Study Collaborative Group. Randomized trial of the effects of cholesterol-lowering with simvastatin on peripheral vascular and other major vascular outcomes in 20,536 people with peripheral arterial disease and other high-risk conditions. J. Vasc. Surg. 2007, 45, 645–654. [Google Scholar] [CrossRef]
- Golledge, J.; Ward, N.C.; Watts, G.F. Lipid management in people with peripheral artery disease. Curr. Opin. Lipidol. 2019, 30, 470–476. [Google Scholar] [CrossRef]
- van Netten, J.; Raspovic, A.; Lavery, L.A.; Monteiro-Soares, M.; Rasmussen, A.; Sacco, I.C.N.; Bus, S.A. Prevention of foot ulcers in the at-risk patient with diabetes: A systematic review. Diabetes Metab. Res. Rev. 2020, 36, e3270. [Google Scholar] [CrossRef]
- Anand, S.S.; Caron, F.; Eikelboom, J.W.; Bosch, J.; Dyal, L.; Aboyans, V.; Abola, M.T.; Branch, K.R.; Keltai, K.; Bhatt, D.L.; et al. Major Adverse Limb Events and Mortality in Patients With Peripheral Artery Disease: The COMPASS Trial. J. Am. Coll. Cardiol. 2018, 71, 2306–2315. [Google Scholar] [CrossRef]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Rogers, L.C.; Lavery, L.A.; Joseph, W.S.; Armstrong, D.G. All Feet On Deck-The Role of Podiatry During the COVID-19 Pandemic: Preventing hospitalizations in an overburdened healthcare system, reducing amputation and death in people with diabetes. J. Am. Podiatr. Med. Assoc. 2020. [Google Scholar] [CrossRef]
- de Stegge, W.B.A.; Mejaiti, N.; Van Netten, J.J.; Dijkgraaf, M.G.W.; Van Baal, J.G.; Busch-Westbroek, T.E.; Bus, S.A. The cost-effectiveness and cost-utility of at-home infrared temperature monitoring in reducing the incidence of foot ulcer recurrence in patients with diabetes (DIATEMP): Study protocol for a randomized controlled trial. Trials 2018, 19, 520. [Google Scholar] [CrossRef] [PubMed]
- Ming, A.; Walter, I.; Alhajjar, A.; Leuckert, M.; Mertens, P.R. Study protocol for a randomized controlled trial to test for preventive effects of diabetic foot ulceration by telemedicine that includes sensor-equipped insoles combined with photo documentation. Trials 2019, 20, 521. [Google Scholar] [CrossRef]
- Manji, K. Pressure-Sensing Insoles in the Neuropathic Ulcer Treatment Pathway (PINUP). Available online: https://clinicaltrials.gov/ct2/show/NCT02586519?term=insoles&cond=Diabetic+Foot&draw=2&rank=8 (accessed on 12 August 2020).
- Furler, J.; O’Neal, D.; Speight, J.; Blackberry, I.; Manski-Nankervis, J.-A.; Thuraisingam, S.; De La Rue, K.; Ginnivan, L.; Doyle, R.; Holmes-Truscott, E.; et al. Use of professional-mode flash glucose monitoring, at 3-month intervals, in adults with type 2 diabetes in general practice (GP-OSMOTIC): A pragmatic, open-label, 12-month, randomised controlled trial. Lancet Diabetes Endocrinol. 2020, 8, 17–26. [Google Scholar] [CrossRef]
- Persell, S.D.; Peprah, Y.A.; Lipiszko, D.; Lee, J.Y.; Li, J.J.; Ciolino, J.D.; Karmali, K.N.; Sato, H. Effect of Home Blood Pressure Monitoring via a Smartphone Hypertension Coaching Application or Tracking Application on Adults With Uncontrolled Hypertension: A Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e200255. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.S.S.P.; Júnior, R.H.C.; Silva, E.Q.; Veríssimo, J.L.; Monteiro, R.L.; Pereira, D.S.; Suda, E.Y.; Sartor, C.; Sacco, I. Study protocol for a randomized controlled trial on the effect of the Diabetic Foot Guidance System (SOPeD) for the prevention and treatment of foot musculoskeletal dysfunctions in people with diabetic neuropathy: The FOotCAre (FOCA) trial I. Trials 2020, 21, 73. [Google Scholar] [CrossRef] [PubMed]
- Saunders, D.J.; Bleasdale, L.; Summerton, L.; Hancock, A.; Homer-Vanniasinkam, S.; Russell, D.A. Assessment of the Utility of a Vascular Early Warning System Device in the Assessment of Peripheral Arterial Disease in Patients with Diabetes and Incompressible Vessels. Ann. Vasc. Surg. 2019, 58, 160–165. [Google Scholar] [CrossRef]
- Armstrong, D.G. Infrared dermal thermometry: The foot and ankle stethoscope. J. Foot Ankle Surg. 1998, 37, 75–76. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Holtz-Neiderer, K.; Wendel, C.; Mohler, M.J.; Kimbriel, H.R.; Lavery, L.A. Skin Temperature Monitoring Reduces the Risk for Diabetic Foot Ulceration in High-risk Patients. Am. J. Med. 2007, 120, 1042–1046. [Google Scholar] [CrossRef]
- Lavery, L.A.; Higgins, K.R.; Lanctot, D.R.; Constantinides, G.P.; Zamorano, R.G.; Armstrong, D.G.; Athanasiou, K.A.; Agrawal, C.M. Home monitoring of foot skin temperatures to prevent ulceration. Diabetes Care 2004, 27, 2642–2647. [Google Scholar] [CrossRef]
- Lavery, L.A.; Higgins, K.R.; Lanctot, D.R.; Constantinides, G.P.; Zamorano, R.G.; Athanasiou, K.A.; Armstrong, D.G.; Agrawal, C.M. Preventing diabetic foot ulcer recurrence in high-risk patients: Use of temperature monitoring as a self-assessment tool. Diabetes Care 2007, 30, 14–20. [Google Scholar] [CrossRef]
- Skafjeld, A.; Iversen, M.M.; Holme, I.; Ribu, L.; Hvaal, K.; Kilhovd, B.K. A pilot study testing the feasibility of skin temperature monitoring to reduce recurrent foot ulcers in patients with diabetes--a randomized controlled trial. BMC Endocr. Disord. 2015, 15, 55. [Google Scholar] [CrossRef]
- Armstrong, D.G.; Lavery, L.A. Monitoring neuropathic ulcer healing with infrared dermal thermometry. J. Foot Ankle Surg. 1996, 35, 335–338. [Google Scholar] [CrossRef]
- Armstrong, D.; Lavery, L. Predicting neuropathic ulceration with infrared dermal thermometry. J. Am. Podiatr. Med. Assoc. 1997, 87, 336–337. [Google Scholar] [CrossRef]
- Lavery, L.A.; Petersen, B.J.; Linders, D.R.; Bloom, J.D.; Rothenberg, G.M.; Armstrong, D.G. Unilateral remote temperature monitoring to predict future ulceration for the diabetic foot in remission. BMJ Open Diabetes Res. Care 2019, 7, e000696. [Google Scholar] [CrossRef]
- Petrova, N.L.; Donaldson, N.K.; Tang, W.; Macdonald, A.; Allen, J.; Lomas, C.; Leech, N.; Ainarkar, S.; Bevans, J.; Plassmann, P.; et al. Infrared thermography and ulcer prevention in the high-risk diabetic foot: Data from a single-blind multicentre controlled clinical trial. Diabet. Med. 2019, 37, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Bus, S.A.; Lavery, L.A.; Monteiro-Soares, M.; Rasmussen, A.; Raspovic, A.; Sacco, I.C.N.; van Netten, J.J.; on behalf of the International Working Group on the Diabetic Foot. IWGDF Guideline on the Prevention of Foot Ulcers in Persons with Diabetes. Available online: www.iwgdfguidelines.org (accessed on 12 August 2020).
- Carabott, M.; Formosa, C.; Mizzi, A.; Papanas, N.; Gatt, A. Thermographic Characteristics of the Diabetic Foot With Peripheral Arterial Disease Using the Angiosome Concept. Exp. Clin. Endocrinol. Diabetes 2019. [Google Scholar] [CrossRef] [PubMed]
- Gatt, A.; Falzon, O.; Cassar, K.; Ellul, C.; Camilleri, K.P.; Gauci, J.; Mizzi, S.; Mizzi, A.; Sturgeon, C.; Camilleri, L.; et al. Establishing Differences in Thermographic Patterns between the Various Complications in Diabetic Foot Disease. Int. J. Endocrinol. 2018, 2018, 9808295. [Google Scholar] [CrossRef] [PubMed]
- Wallace, G.A.; Singh, N.; Quiroga, E.; Tran, N.T. The Use of Smart Phone Thermal Imaging for Assessment of Peripheral Perfusion in Vascular Patients. Ann. Vasc. Surg. 2018, 47, 157–161. [Google Scholar] [CrossRef]
- Staffa, E.; Bernard, V.; Kubicek, L.; Vlachovsky, R.; Vlk, D.; Mornstein, V.; Bourek, A.; Staffa, R. Infrared thermography as option for evaluating the treatment effect of percutaneous transluminal angioplasty by patients with peripheral arterial disease. Vascular 2016, 25, 42–49. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Gordon, I.L.; Reyzelman, A.M.; Cazzell, S.; Fitzgerald, R.H.; Rothenberg, G.M.; Bloom, J.D.; Petersen, B.J.; Linders, D.R.; Nouvong, A.; et al. Feasibility and Efficacy of a Smart Mat Technology to Predict Development of Diabetic Plantar Ulcers. Diabetes Care 2017, 40, 973–980. [Google Scholar] [CrossRef]
- Najafi, B.; Mohseni, H.; Grewal, G.S.; Talal, T.K.; Menzies, R.A.; Armstrong, D.G. An Optical-Fiber-Based Smart Textile (Smart Socks) to Manage Biomechanical Risk Factors Associated With Diabetic Foot Amputation. J. Diabetes Sci. Technol. 2017, 11, 668–677. [Google Scholar] [CrossRef]
- Reyzelman, A.M.; Koelewyn, K.; Murphy, M.; Shen, X.; Yu, E.; Pillai, R.; Fu, J.; Scholten, H.J.; Ma, R.; Dini, M.; et al. Continuous Temperature-Monitoring Socks for Home Use in Patients With Diabetes: Observational Study. J. Med. Internet Res. 2018, 20, e12460. [Google Scholar] [CrossRef]
- van Doremalen, R.; Van Netten, J.; Van Baal, J.; Vollenbroek-Hutten, M.; Van Der Heijden, F. Validation of low-cost smartphone-based thermal camera for diabetic foot assessment. Diabetes Res. Clin. Pract. 2019, 149, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Fernando, M.E.; Crowther, R.G.; Pappas, E.; Lazzarini, P.A.; Cunningham, M.; Sangla, K.S.; Buttner, P.; Golledge, J. Plantar Pressure in Diabetic Peripheral Neuropathy Patients with Active Foot Ulceration, Previous Ulceration and No History of Ulceration: A Meta-Analysis of Observational Studies. PLoS ONE 2014, 9, e99050. [Google Scholar] [CrossRef] [PubMed]
- Bus, S.A.; Maas, J.C.; Otterman, N.M. Lower-extremity dynamics of walking in neuropathic diabetic patients who wear a forefoot-offloading shoe. Clin. Biomech. 2017, 50, 21–26. [Google Scholar] [CrossRef]
- Lazzarini, P.A.; Crews, R.T.; Van Netten, J.J.; Bus, S.A.; Fernando, M.E.; Chadwick, P.J.; Najafi, B. Measuring Plantar Tissue Stress in People With Diabetic Peripheral Neuropathy: A Critical Concept in Diabetic Foot Management. J. Diabetes Sci. Technol. 2019, 13, 869–880. [Google Scholar] [CrossRef] [PubMed]
- van Netten, J.J.; Woodburn, J.; Bus, S.A. The future of prevention of diabetic foot ulcer: A paradigm shift from stratified healthcare towards personalized medicine. Diabetes Metab. Res. Rev. 2020, 36, e3234. [Google Scholar] [CrossRef] [PubMed]
- Fernando, M.E.; Crowther, R.; Wearing, S. The Importance of Foot Pressure in Diabetes. In Handbook of Human Motion; Springer International Publishing: Cham, Germany, 2018; pp. 759–787. [Google Scholar]
- Armstrong, D.G.; Najafi, B.; Shahinpoor, M. Potential Applications of Smart Multifunctional Wearable Materials to Gerontology. Gerontology 2017, 63, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Najafi, B.; Ron, E.; Enriquez, A.; Marin, I.; Razjouyan, J.; Armstrong, D.G. Smarter Sole Survival: Will Neuropathic Patients at High Risk for Ulceration Use a Smart Insole-Based Foot Protection System? J. Diabetes Sci. Technol. 2017, 11, 702–713. [Google Scholar] [CrossRef]
- Abbott, C.A.; Chatwin, K.E.; Foden, P.; Hasan, A.N.; Sange, C.; Rajbhandari, S.M.; Reddy, P.N.; Vileikyte, L.; Bowling, F.L.; Boulton, A.J.M.; et al. Innovative intelligent insole system reduces diabetic foot ulcer recurrence at plantar sites: A prospective, randomised, proof-of-concept study. Lancet Digit. Health 2019, 1, e308–e318. [Google Scholar] [CrossRef]
- Bus, S.A. Preventing foot ulcers in diabetes using plantar pressure feedback. Lancet Digit. Health 2019, 1, e250–e251. [Google Scholar] [CrossRef]
- Uccioli, L.; Faglia, E.; Monticone, G.; Favales, F.; Durola, L.; Aldeghi, A.; Quarantiello, A.; Calia, P.; Menzinger, G. Manufactured Shoes in the Prevention of Diabetic Foot Ulcers. Diabetes Care 1995, 18, 1376–1378. [Google Scholar] [CrossRef]
- Reiber, G.E.; Smith, D.G.; Wallace, C.; Sullivan, K.; Hayes, S.; Vath, C.; Maciejewski, M.L.; Yu, O.; Heagerty, P.J.; LeMaster, J. Effect of Therapeutic Footwear on Foot Reulceration in Patients With Diabetes. JAMA 2002, 287, 2552–2558. [Google Scholar] [CrossRef] [PubMed]
- Lavery, L.A.; Lafontaine, J.; Higgins, K.R.; Lanctot, D.R.; Constantinides, G. Shear-Reducing Insoles to Prevent Foot Ulceration in High-Risk Diabetic Patients. Adv. Ski. Wound Care 2012, 25, 519–526. [Google Scholar] [CrossRef]
- Rizzo, L.; Tedeschi, A.; Fallani, E.; Coppelli, A.; Vallini, V.; Iacopi, E.; Piaggesi, A. Custom-Made Orthesis and Shoes in a Structured Follow-Up Program Reduces the Incidence of Neuropathic Ulcers in High-Risk Diabetic Foot Patients. Int. J. Low. Extrem. Wounds 2012, 11, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Westphal, C.; Neame, I.M.; Harrison, J.C.; Bower, V.M.; Gurr, J.M. A diabetic foot ulcer pilot study: Does silicone gel sheeting reduce the incidence of reulceration? J. Am. Podiatr. Med. Assoc. 2011, 101, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Ulbrecht, J.S.; Hurley, T.; Mauger, D.T.; Cavanagh, P. Prevention of Recurrent Foot Ulcers With Plantar Pressure–Based In-Shoe Orthoses: The CareFUL Prevention Multicenter Randomized Controlled Trial. Diabetes Care 2014, 37, 1982–1989. [Google Scholar] [CrossRef] [PubMed]
- Bus, S.A.; Waaijman, R.; Arts, M.; De Haart, M.; Busch-Westbroek, T.; Van Baal, J.; Nollet, F. Effect of Custom-Made Footwear on Foot Ulcer Recurrence in Diabetes: A multicenter randomized controlled trial. Diabetes Care 2013, 36, 4109–4116. [Google Scholar] [CrossRef]
- López-Moral, M.; Lázaro-Martínez, J.L.; García-Morales, E.; García-Álvarez, Y.; Álvaro-Afonso, F.; Molines-Barroso, R.J. Clinical efficacy of therapeutic footwear with a rigid rocker sole in the prevention of recurrence in patients with diabetes mellitus and diabetic polineuropathy: A randomized clinical trial. PLoS ONE 2019, 14, e0219537. [Google Scholar] [CrossRef]
- Ehrmann, D.; Spengler, M.; Jahn, M.; Niebuhr, D.; Haak, T.; Kulzer, B.; Hermanns, N. Adherence Over Time: The Course of Adherence to Customized Diabetic Insoles as Objectively Assessed by a Temperature Sensor. J. Diabetes Sci. Technol. 2017, 12, 695–700. [Google Scholar] [CrossRef]
- Lutjeboer, T.; Netten, J.; Postema, K.; Hijmans, J. Validity and feasibility of a temperature sensor for measuring use and non-use of orthopaedic footwear. J. Rehabil. Med. 2018, 50, 920–926. [Google Scholar] [CrossRef]
- Bus, S.A.; Waaijman, R.; Nollet, F. New Monitoring Technology to Objectively Assess Adherence to Prescribed Footwear and Assistive Devices During Ambulatory Activity. Arch. Phys. Med. Rehabil. 2012, 93, 2075–2079. [Google Scholar] [CrossRef]
- Jarl, G.; Lundqvist, L.-O. Adherence to wearing therapeutic shoes among people with diabetes: A systematic review and reflections. Patient Prefer. Adherence 2016, 10, 1521–1528. [Google Scholar] [CrossRef]
- Najafi, B.; Reeves, N.D.; Armstrong, D.G. Leveraging smart technologies to improve the management of diabetic foot ulcers and extend ulcer-free days in remission. Diabetes/Metabolism Res. Rev. 2020, 36, e3239. [Google Scholar] [CrossRef] [PubMed]
- Iacopi, E.; Coppelli, A.; Riitano, N.; Abbruzzese, L.; Pieruzzi, L.; Goretti, C.; Piaggesi, A. Adherence to guideline recommended medical therapies in type 2 diabetic patients with chronic critical limb ischemia. Diabetes Res. Clin. Pract. 2019, 158, 107898. [Google Scholar] [CrossRef] [PubMed]
- Brownrigg, J.R.W.; Davey, J.; Holt, P.J.E.; Davis, W.A.; Thompson, M.M.; Ray, K.K.; Hinchliffe, R.J. The association of ulceration of the foot with cardiovascular and all-cause mortality in patients with diabetes: A meta-analysis. Diabetologia 2012, 55, 2906–2912. [Google Scholar] [CrossRef] [PubMed]
- Mader, J.K.; Haas, W.; Aberer, F.; Boulgaropoulos, B.; Baumann, P.; Pandis, M.; Horvath, K.; Aziz, F.; Köhler, G.; Pieber, T.R.; et al. Patients with healed diabetic foot ulcer represent a cohort at highest risk for future fatal events. Sci. Rep. 2019, 9, 10325. [Google Scholar] [CrossRef] [PubMed]
- Hasan, R.; Firwana, B.; Elraiyah, T.; Domecq, J.P.; Prutsky, G.; Nabhan, M.; Prokop, L.J.; Henke, P.; Tsapas, A.; Montori, V.M.; et al. A systematic review and meta-analysis of glycemic control for the prevention of diabetic foot syndrome. J. Vasc. Surg. 2016, 63, 22S–28S. [Google Scholar] [CrossRef]
- Colagiuri, S.; Dickinson, S.; Girgis, S.; Colagiuri, R. National Evidence Based Guideline for BloodGlucose Control in Type 2 Diabetes. Diabetes Aust. NHMRC 2009, 1–39. [Google Scholar]
- Klonoff, D.C.; Ahn, D.; Drincic, A. Continuous glucose monitoring: A review of the technology and clinical use. Diabetes Res. Clin. Pract. 2017, 133, 178–192. [Google Scholar] [CrossRef]
- Brown, S.A.; Kovatchev, B.P.; Raghinaru, D.; Lum, J.W.; Buckingham, B.A.; Kudva, Y.C.; Laffel, L.M.; Levy, C.J.; Pinsker, J.E.; Wadwa, R.P.; et al. Six-Month Randomized, Multicenter Trial of Closed-Loop Control in Type 1 Diabetes. NEJM 2019, 381, 1707–1717. [Google Scholar] [CrossRef]
- Janapala, R.N.; Jayaraj, J.S.; Fathima, N.; Kashif, T.; Usman, N.; Dasari, A.; Jahan, N.; Sachmechi, I. Continuous Glucose Monitoring Versus Self-monitoring of Blood Glucose in Type 2 Diabetes Mellitus: A Systematic Review with Meta-analysis. Cureus 2019, 11, e5634. [Google Scholar] [CrossRef]
- Tweden, K.S.; Deiss, D.; Rastogi, R.; Addaguduru, S.; Kaufman, F. Longitudinal Analysis of Real-World Performance of an Implantable Continuous Glucose Sensor over Multiple Sensor Insertion and Removal Cycles. Diabetes Technol. Ther. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Soupal, J.; Petruželková, L.; Grunberger, G.; Hásková, A.; Flekač, M.; Matoulek, M.; Mikeš, O.; Pelcl, T.; Horová, E.; Škrha, J.; et al. Glycemic Outcomes in Adults With T1D Are Impacted More by Continuous Glucose Monitoring Than by Insulin Delivery Method: 3 Years of Follow-Up From the COMISAIR Study. Diabetes Care 2019, 43, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, V.; Grunberger, G.; Anhalt, H.; Bailey, T.S.; Blevins, T.; Garg, S.K.; Handelsman, Y.; Hirsch, I.B.; Orzeck, E.A.; Roberts, V.L.; et al. Continuous glucose monitoring: A consensus conference of the american association of clinical endocrinologists and american college of endocrinology. Endocr. Pract. 2016, 22, 1008–1021. [Google Scholar] [CrossRef] [PubMed]
- Östergren, J.; Sleight, P.; Dagenais, G.; Danisa, K.; Bosch, J.; Qilong, Y.; Yusuf, S. Impact of ramipril in patients with evidence of clinical or subclinical peripheral arterial disease. Eur. Heart J. 2004, 25, 17–24. [Google Scholar] [CrossRef]
- Manapurathe, D.T.; Moxon, J.V.; Krishna, S.M.; Rowbotham, S.; Quigley, F.; Jenkins, J.; Bourke, M.; Bourke, B.; Jones, R.E.; Golledge, J. Cohort Study Examining the Association Between Blood Pressure and Cardiovascular Events in Patients With Peripheral Artery Disease. J. Am. Heart Assoc. 2019, 8, e010748. [Google Scholar] [CrossRef]
- Islam, S.M.S.; Cartledge, S.; Karmakar, C.; Rawstorn, J.C.; Fraser, S.F.; Chow, C.; Maddison, R. Validation and Acceptability of a Cuffless Wrist-Worn Wearable Blood Pressure Monitoring Device Among Users and Health Care Professionals: Mixed Methods Study. JMIR mHealth uHealth 2019, 7, e14706. [Google Scholar] [CrossRef]
- Pellaton, C.; Vybornova, A.; Fallet, S.; Marques, L.; Grossenbacher, O.; De Marco, B.; Chapuis, V.; Bertschi, M.; Alpert, B.S.; Solà, J. Accuracy testing of a new optical device for noninvasive estimation of systolic and diastolic blood pressure compared to intra-arterial measurements. Blood Press. Monit. 2020, in press. [Google Scholar] [CrossRef]
- Krisai, P.; Vischer, A.S.; Kilian, L.; Meienberg, A.; Mayr, M.; Burkard, T. Accuracy of 24-hour ambulatory blood pressure monitoring by a novel cuffless device in clinical practice. Hear. 2018, 105, 399–405. [Google Scholar] [CrossRef]
- Bonaca, M.P.; Nault, P.; Giugliano, R.; Keech, A.C.; Pineda, A.L.; Kanevsky, E.; Kuder, J.; Murphy, S.A.; Jukema, J.W.; Lewis, B.S.; et al. Low-Density Lipoprotein Cholesterol Lowering With Evolocumab and Outcomes in Patients With Peripheral Artery Disease: Insights From the FOURIER Trial (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk). Circulation 2018, 2018, 4. [Google Scholar] [CrossRef]
- Chunta, S.; Suedee, R.; Lieberzeit, P.A. Low-Density Lipoprotein Sensor Based on Molecularly Imprinted Polymer. Anal. Chem. 2015, 88, 1419–1425. [Google Scholar] [CrossRef]
- Delamater, A.M. Improving Patient Adherence. Clin. Diabetes 2006, 24, 71. [Google Scholar] [CrossRef]
- Winkler, A.; Teuscher, A.U.; Mueller, B.; Diem, P. Monotoring adherence to prescribed medication in type 2 diabetic patients treated with sulfonylureas. Swiss. Med. Wkly. 2002, 132, 379–385. [Google Scholar] [PubMed]
- Paes, A.H.P.; Bakker, A.; Soe-Agnie, C.J. Impact of dosage frequency on patient compliance. Diabetes Care 1997, 20, 1512. [Google Scholar] [CrossRef] [PubMed]
- Frias, J.P.; Virdi, N.; Raja, P.; Kim, Y.; Savage, G.; Osterberg, L.; Exeter, C.; Da Silva, E.; Naik, A. Effectiveness of Digital Medicines to Improve Clinical Outcomes in Patients with Uncontrolled Hypertension and Type 2 Diabetes: Prospective, Open-Label, Cluster-Randomized Pilot Clinical Trial. J. Med. Internet Res. 2017, 19, e246. [Google Scholar] [CrossRef]
- Fernando, M.E.; Crowther, R.G.; Lazzarini, P.A.; Sangla, K.S.; Wearing, S.; Buttner, P.; Golledge, J. Gait in People With Nonhealing Diabetes-Related Plantar Ulcers. Phys. Ther. 2019, 99, 1602–1615. [Google Scholar] [CrossRef]
- Brodie, M.A.; Okubo, Y.; Annegarn, J.; Wieching, R.; Lord, S.R.; Delbaere, K. Disentangling the health benefits of walking from increased exposure to falls in older people using remote gait monitoring and multi-dimensional analysis. Physiol. Meas. 2016, 38, 45–62. [Google Scholar] [CrossRef]
- Lee, S.-H.; Lee, H.-J.; Chang, W.H.; Choi, B.-O.; Lee, J.; Kim, J.; Ryu, G.H.; Kim, Y.-H. Gait performance and foot pressure distribution during wearable robot-assisted gait in elderly adults. J. Neuroeng. Rehabil. 2017, 14, 123. [Google Scholar] [CrossRef]
- Ferreira, J.S.S.P.; Sacco, I.C.N.; Siqueira, A.A.; Almeida, M.H.M.; Sartor, C.D. Rehabilitation technology for self-care: Customised foot and ankle exercise software for people with diabetes. PLoS ONE 2019, 14, e0218560. [Google Scholar] [CrossRef]
- Wilasrusmee, C.; Suthakorn, J.; Guerineau, C.; Itsarachaiyot, Y.; Sa-Ing, V.; Proprom, N.; Lertsithichai, P.; Jirasisrithum, S.; Kittur, D. A Novel Robotic Monofilament Test for Diabetic Neuropathy. Asian J. Surg. 2010, 33, 193–198. [Google Scholar] [CrossRef][Green Version]
- Yang, Z.; Zhang, Y.; Chen, R.; Huang, Y.; Ji, L.; Sun, F.; Hong, T.; Zhan, S. Simple tests to screen for diabetic peripheral neuropathy. Cochrane Database Syst. Rev. 2018, 2018, CD010975. [Google Scholar] [CrossRef]
- Piaggesi, A.; Läuchli, S.; Bassetto, F.; Biedermann, T.; Marques, A.; Najafi, B.; Palla, I.; Scarpa, C.; Seimetz, D.; Triulzi, I.; et al. Advanced therapies in wound management: Cell and tissue based therapies, physical and bio-physical therapies smart and IT based technologies. J. Wound Care 2018, 27, S1–S137. [Google Scholar] [CrossRef] [PubMed]
- Ploderer, B.; Brown, R.; Seng, L.; Lazzarini, P.A.; Van Netten, J.; He, Q.; Strong, D.; Margolis, D.; Mamillapalli, C. Promoting Self-Care of Diabetic Foot Ulcers Through a Mobile Phone App: User-Centered Design and Evaluation. JMIR Diabetes 2018, 3, e10105. [Google Scholar] [CrossRef] [PubMed]
- Boodoo, C.; Perry, J.A.; Hunter, P.J.; Duta, D.I.; Newhook, S.C.; Leung, G.; Cross, K.; Fatehi, F.; Yderstraede, K. Views of Patients on Using mHealth to Monitor and Prevent Diabetic Foot Ulcers: Qualitative Study. JMIR Diabetes 2017, 2, e22. [Google Scholar] [CrossRef] [PubMed]
- Basatneh, R.; Najafi, B.; Armstrong, D.G. Health Sensors, Smart Home Devices, and the Internet of Medical Things: An Opportunity for Dramatic Improvement in Care for the Lower Extremity Complications of Diabetes. J. Diabetes Sci. Technol. 2018, 12, 577–586. [Google Scholar] [CrossRef]
- van Netten, J.; Clark, D.; Lazzarini, P.A.; Janda, M.; Reed, L.F. The validity and reliability of remote diabetic foot ulcer assessment using mobile phone images. Sci. Rep. 2017, 7, 9480. [Google Scholar] [CrossRef]
- Perednia, D.A.; Allen, A. Telemedicine technology and clinical applications. JAMA 1995, 273, 483–488. [Google Scholar] [CrossRef]
- Hazenberg, C.E.V.B.; Aan de Stegge, W.B.; Van Baal, S.G.; Moll, F.L.; Bus, S.A. Telehealth and telemedicine applications for the diabetic foot: A systematic review. Diabetes/Metab. Res. Rev. 2020, 36, e3247. [Google Scholar] [CrossRef]
- Singh, T.P.; Vangaveti, V.N.; Kennedy, R.L.; Malabu, U.H. Role of telehealth in diabetic foot ulcer management—A systematic review. Aust. J. Rural Health 2016, 24, 224–229. [Google Scholar] [CrossRef]
- Tchero, H.; Noubou, L.; Becsangele, B.; Mukisi-Mukaza, M.; Retali, G.-R.; Rusch, E. Telemedicine in Diabetic Foot Care: A Systematic Literature Review of Interventions and Meta-analysis of Controlled Trials. Int. J. Low. Extrem. Wounds 2017, 16, 274–283. [Google Scholar] [CrossRef]
- Rasmussen, B.S.B.; Froekjaer, J.; Bjerregaard, M.R.; Lauritsen, J.M.; Hangaard, J.; Halekoh, U.; Henriksen, C.W.; Yderstraede, K.B. A Randomized Controlled Trial Comparing Telemedical and Standard Outpatient Monitoring of Diabetic Foot Ulcers. Diabetes Care 2015, 38, 1723–1729. [Google Scholar] [CrossRef] [PubMed]
- Smith-Strom, H.; Igland, J.; Ostbye, T.; Tell, G.S.; Hausken, M.F.; Graue, M.; Skeie, S.; Cooper, J.G.; Iversen, M.M. The Effect of Telemedicine Follow-up Care on Diabetes-Related Foot Ulcers: A Cluster-Randomized Controlled Noninferiority Trial. Diabetes Care 2018, 41, 96–103. [Google Scholar] [CrossRef] [PubMed]
| Risk Factor | Current Management Approach | Sensors or Wearable Devices | References | Potential Value of Sensor/Wearable | Potential Impact on Prevention |
|---|---|---|---|---|---|
| Pre-ulcerative lesions | Visits to podiatrist | Home foot temperature monitor and mobile phone applications | [9,10] | Offloading of “hot spots” following confirmed persistent temperature differences | Reduced progression of at-risk sites prone to develop foot ulcers |
| Elevated plantar pressures | Offloading footwear | Plantar pressure monitor | [11] | Warning systems to stimulate offloading and better design and modification of footwear | Improved offloading with reduced ulcer development |
| Elevated plantar pressures and tissue stress | Patient education | Footwear adherence monitor | [11] | Behaviour change support counselling informed by objective data | Improved offloading adherence |
| Hyperglycaemia | Capillary glucose monitoring | Continuous glucose monitor | [12] | Intensive glycaemic control | Better informed management of hyper and hypoglycaemia and reduced progression of macro and microvascular disease |
| Hypertension | Outpatient blood pressure measurement | Cuff-less blood pressure monitor | [13] | Better implementation of anti-hypertensive medications and more frequent monitoring | Better informed management of blood pressure and reduced progression of macro and microvascular disease and mortality |
| Abnormal gait | Not routinely managed | Gait and activity monitor | [14] | Gait retraining and encouraging remote physical activity | Reduce gait abnormalities potentially reducing plantar pressures and ulcer incidence |
| Peripheral artery disease | Vascular laboratory assessment using ultrasound or Doppler | Foot blood supply sensor | [15] | Earlier identification of complications and prompt medical management | Reduced progression of macro and microvascular disease |
| Remote Monitoring | Available Evidence | Current Limitations of Available Evidence | Relevant Studies |
|---|---|---|---|
| Home foot temperature monitor | A number of small RCTs show a decreased incidence of foot ulcers in people performing home-based temperature monitoring | Lack of a widely tested and user-friendly way of identifying “hot spots” Generalizability from prior smaller studies in select populations | [9,10] |
| Plantar pressure monitor | Possible to monitor plantar pressure remotely and use patient alarms to warn patients of impending sites of tissue breakdown as reported in one small RCT | Unclear if technology can be further developed to be more user-friendly and whether the findings are applicable and would be effective on a widespread basis | [11] |
| Footwear adherence monitor | Technology has been developed to accurately measure footwear adherence | Need for widespread testing of value of using devices Patients’ views on use of adherence monitoring is still unclear | [11] |
| Continuous glucose monitor | Highly developed area of monitoring and tested in multiple RCTs with proven benefit in improving glycaemic control (HbA1c) | Whether this remote monitoring improves outcomes in people at risk of developing DFD remains unclear | [12] |
| Cuff-less blood pressure monitor | Technology developed to assess this reported to be accurate in a small number of studies | Currently unclear whether these devices can be used on a widespread scale | [13] |
| Foot blood supply and sensation assessment | Technology still in the early developmental stages for monitoring | The benefit of these devices in improving clinical outcomes need to be further evaluated in RCTs | [15,82] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golledge, J.; Fernando, M.; Lazzarini, P.; Najafi, B.; G. Armstrong, D. The Potential Role of Sensors, Wearables and Telehealth in the Remote Management of Diabetes-Related Foot Disease. Sensors 2020, 20, 4527. https://doi.org/10.3390/s20164527
Golledge J, Fernando M, Lazzarini P, Najafi B, G. Armstrong D. The Potential Role of Sensors, Wearables and Telehealth in the Remote Management of Diabetes-Related Foot Disease. Sensors. 2020; 20(16):4527. https://doi.org/10.3390/s20164527
Chicago/Turabian StyleGolledge, Jonathan, Malindu Fernando, Peter Lazzarini, Bijan Najafi, and David G. Armstrong. 2020. "The Potential Role of Sensors, Wearables and Telehealth in the Remote Management of Diabetes-Related Foot Disease" Sensors 20, no. 16: 4527. https://doi.org/10.3390/s20164527
APA StyleGolledge, J., Fernando, M., Lazzarini, P., Najafi, B., & G. Armstrong, D. (2020). The Potential Role of Sensors, Wearables and Telehealth in the Remote Management of Diabetes-Related Foot Disease. Sensors, 20(16), 4527. https://doi.org/10.3390/s20164527








