Machine Learning to Quantify Physical Activity in Children with Cerebral Palsy: Comparison of Group, Group-Personalized, and Fully-Personalized Activity Classification Models
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Individual Activity Trials
2.3. Instrumentation
2.4. Machine Learning Activity Classification Models
2.4.1. Data Pre-Processing and Feature Extraction
2.4.2. Model Training and Cross-Validation
2.5. Simulated Free-Living Evaluation
2.6. Statistical Evaluation
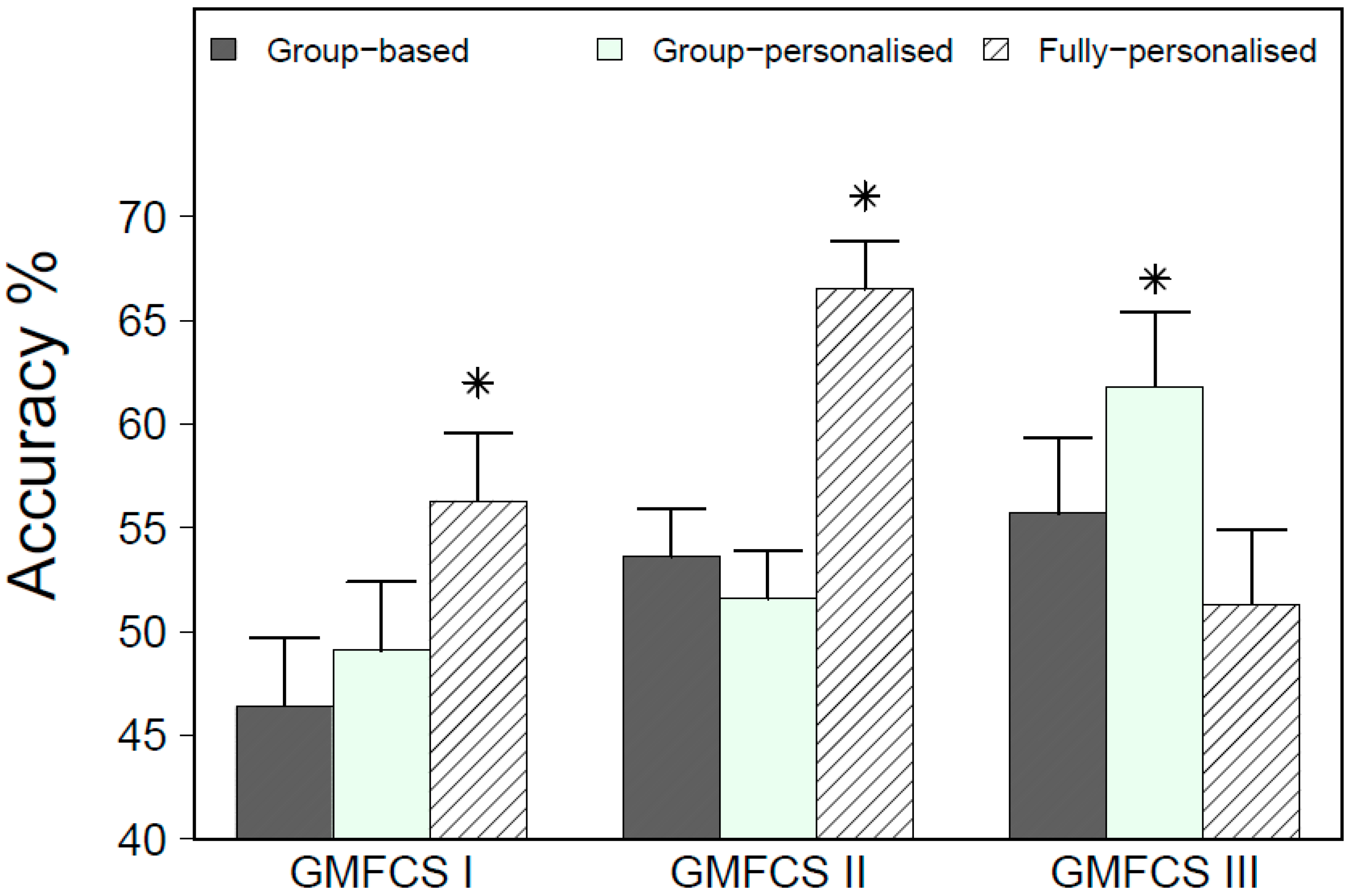
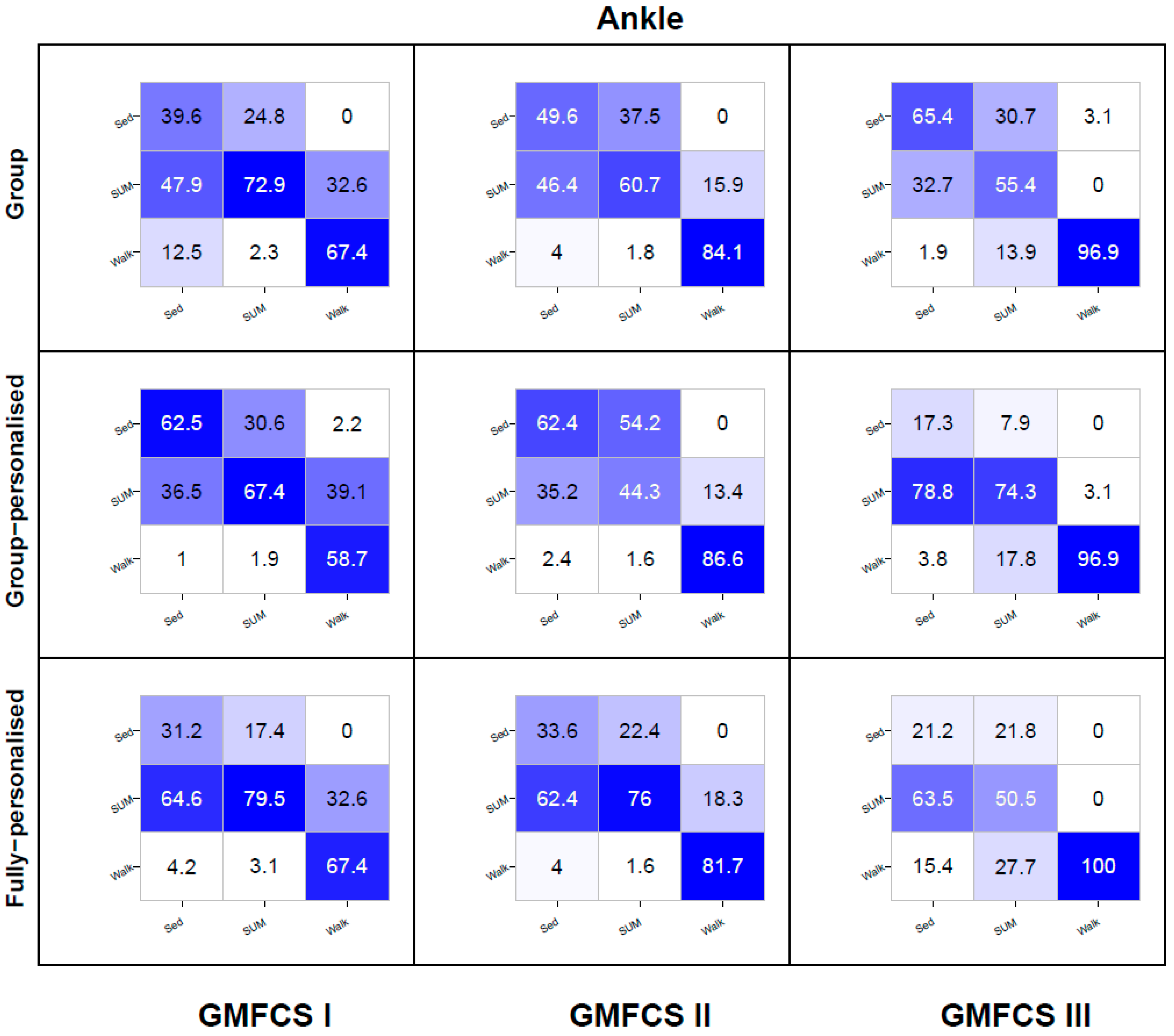
3. Results
3.1. Leave-One-Subject-Out Cross-Validation
3.2. Simulated Free-Living Trial
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Novak, I.; Morgan, C.; Adde, L.; Blackman, J.; Boyd, R.N.; Brunstrom-Hernandez, J.; Cioni, G.; Damiano, D.; Darrah, J.; Eliasson, A.C.; et al. Early, accurate diagnosis and early intervention in cerebral palsy: Advances in diagnosis and treatment. JAMA Pediatr. 2017, 171, 897–907. [Google Scholar] [CrossRef]
- Report of the Australian Cerebral Palsy Register Birth Years 1995–2012. 2018. Available online: https://cpregister.com/wp-content/uploads/2019/02/Report-of-the-Australian-Cerebral-Palsy-Register-Birth-Years-1995-2012.pdf (accessed on 18 June 2020).
- Bjornson, K.; Fiss, A.; Avery, L.; Wentz, E.; Kerfeld, C.; Cicirello, N.; Hanna, S.E. Longitudinal trajectories of physical activity and walking performance by gross motor function classification system level for children with cerebral palsy. Disabil Rehabil. 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nooijen, C.F.J.; Slaman, J.; Stam, H.J.; Roebroeck, M.E.; Van Den Berg-Emons, R.J. Inactive and sedentary lifestyles amongst ambulatory adolescents and young adults with cerebral palsy. J. Neuroeng. Rehabil. 2014, 11, 49. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.M.; Smit, E.; Yun, J.; Bogart, K.; Hatfield, B.; Logan, S.W. Updated national estimates of disparities in physical activity and sports participation experienced by children and adolescents with disabilities: NSCH 2016–2017. J. Phys. Act. Health 2020, 17, 443–455. [Google Scholar] [CrossRef]
- Van Wely, L.; Becher, J.G.; Balemans, A.C.J.; Dallmeijer, A.J. Ambulatory activity of children with cerebral palsy: Which characteristics are important? Dev. Med. Child Neurol. 2012, 54, 436–442. [Google Scholar] [CrossRef]
- Gorter, J.W.; Noorduyn, S.G.; Obeid, J.; Timmons, B.W. Accelerometry: A Feasible Method to Quantify Physical Activity in Ambulatory and Nonambulatory Adolescents with Cerebral Palsy. Int. J. Pediatr.s 2012, 2012, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bjornson, K.F.; Belza, B.; Kartin, D.; Logsdon, R.; McLaughlin, J.F. Ambulatory physical activity performance in youth with cerebral palsy and youth who are developing typically. Phys. Ther. 2007, 87, 248–257. [Google Scholar] [CrossRef]
- Reedman, S.E.; Boyd, R.N.; Trost, S.G.; Elliott, C.; Sakzewski, L. Efficacy of participation-focused therapy on performance of physical activity participation goals and habitual physical activity in children with cerebral palsy: A randomized controlled trial. Arch. Phys. Med. Rehabil. 2019, 100, 676–686. [Google Scholar] [CrossRef]
- Maher, C.A.; Williams, M.T.; Olds, T.; Lane, A.E. An internet-based physical activity intervention for adolescents with cerebral palsy: A randomized controlled trial. Dev. Med. Child Neurol. 2010, 52, 448–455. [Google Scholar] [CrossRef]
- Boyd, R.N.; Davies, P.S.; Ziviani, J.; Trost, S.; Barber, L.; Ware, R.; Rose, S.; Whittingham, K.; Sakzewski, L.; Bell, K.; et al. PREDICT-CP: Study protocol of implementation of comprehensive surveillance to predict outcomes for school-aged children with cerebral palsy. BMJ Open 2017, 7, 1–19. [Google Scholar] [CrossRef]
- Trost, S.G.; O’Neil, M. Clinical use of objective measures of physical activity. Br. J. Sports Med. 2014, 48, 178–181. [Google Scholar] [CrossRef]
- Van Wely, L.; Balemans, A.C.J.; Becher, J.G.; Dallmeijer, A.J. Physical activity stimulation program for children with cerebral palsy did not improve physical activity: A randomised trial. J. Physiother. 2014, 60, 40–49. [Google Scholar] [CrossRef]
- Mitchell, L.E.; Ziviani, J.; Boyd, R.N. A randomized controlled trial of web-based training to increase activity in children with cerebral palsy. Dev. Med. Child Neurol. 2016, 58, 767–773. [Google Scholar] [CrossRef]
- Trost, S.G.; Mciver, K.L.; Pate, R.R. Conducting accelerometer-based activity assessments in field-based research. Med. Sci. Sports Exerc. 2005, 37 (Suppl. 11), S531–S543. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, M.E.; Fragala-Pinkham, M.; Lennon, N.; George, A.; Forman, J.; Trost, S.G. Reliability and validity of objective measures of physical activity in youth with cerebral palsy who are ambulatory. Phys. Ther. 2016, 96, 37–45. [Google Scholar] [CrossRef] [PubMed]
- O′Neil, M.E.; Fragala-Pinkham, M.; Forman, J.L.; Trost, S.G. Measuring reliability and validity of the ActiGraph GT3X accelerometer for children with cerebral palsy: A feasibility study. J. Pediatr. Rehabil. Med. 2014, 7, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Clanchy, K.M.; Tweedy, S.M.; Boyd, R.N.; Trost, S.G. Validity of accelerometry in ambulatory children and adolescents with cerebral palsy. Eur. J. Appl. Physiol. 2011, 111, 2951–2959. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, L.E.; Ziviani, J.; Boyd, R.N. Variability in measuring physical activity in children with cerebral palsy. Med. Sci. Sports Exerc. 2015, 47, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.; Walsh, M.; Gormley, J. Ability of RT3 accelerometer cut points to detect physical activity intensity in ambulatory children with cerebral palsy. Adapt. Phys. Act. Q. 2014, 31, 310–324. [Google Scholar] [CrossRef]
- Trost, S.G.; Fragala-Pinkham, M.; Lennon, N.; O’Neil, M.E. Decision trees for detection of activity intensity in youth with cerebral palsy. Med. Sci. Sports Exerc. 2016, 48, 958–966. [Google Scholar] [CrossRef]
- Ahmadi, M.N.; O’Neil, M.E.; Fragala-pinkham, M.; Lennon, N.; Trost, S.G. Machine learning algorithms for activity recognition in ambulant children and adolescents with cerebral palsy. J. Neuroeng. Rehabil. 2018, 15, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.K.; Tjondronegoro, D.; Chandran, V.; Trost, S.G. Physical activity recognition using posterior-adapted class-based fusion of multiaccelerometer data. IEEE J. Biomed. Health Inform. 2018, 22, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Mannini, A.; Intille, S.S.; Rosenberger, M.; Sabatini, A.M.; Haskell, W. Activity recognition using a single accelerometer placed at the wrist or ankle. Med. Sci. Sports Exerc. 2013, 45, 2193–2203. [Google Scholar] [CrossRef] [PubMed]
- Lockhart, J.W.; Weiss, G.M. The benefits of personalized smartphone-based activity recognition models. In Proceedings of the 2014 SIAM International Conference on Data Mining, Philadelphia, PA, USA, 24–26 April 2014; p. 9. [Google Scholar]
- Carcreff, L.; Paraschiv-Ionescu, A.; Gerber, C.N.; Newman, C.J.; Armand, S.; Aminian, K. A personalized approach to improve walking detection in real-life settings: Application to children with cerebral palsy. Sensors 2019, 19, 5316. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, J.E.; Hickey, A.M.; Staudenmayer, J.W.; John, D.; Kent, J.A.; Freedson, P.S. Performance of activity classification algorithms in free-living older adults. Med. Sci. Sports Exerc. 2016, 48, 941–949. [Google Scholar] [CrossRef]
- Ahmadi, M.N.; Brookes, D.; Chowdhury, A.; Pavey, T.; Trost, S.G. Free-living evaluation of laboratory-based activity classifiers in preschoolers. Med. Sci. Sports Exerc. 2020, 52, 1227–1234. [Google Scholar] [CrossRef]
- Bastian, T.; Maire, A.; Dugas, J.; Ataya, A.; Villars, C.; Gris, F.; Perrin, E.; Caritu, Y.; Doron, M.; Blanc, S.; et al. Automatic identification of physical activity types and sedentary behaviors from triaxial accelerometer: Laboratory-based calibrations are not enough. J. Appl. Physiol. 2015, 118, 716–722. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Chowdhury, A.K.; Tjondronegoro, D.; Chandran, V.; Trost, S.G. Ensemble methods for classification of physical activities from wrist accelerometry. Med. Sci. Sports Exerc. 2017, 49, 1965–1973. [Google Scholar] [CrossRef]
- Peng, H.; Long, F.; Ding, C. Feature selection based on mutual information: Criteria of max-dependency, max-relevance, and min-redundancy. IEEE Trans. Pattern Anal. Mach. Intell. 2005, 27, 1226–1238. [Google Scholar] [CrossRef] [PubMed]
- Report of the Australian Cerebral Palsy Register, Birth Years 1993–2009. 2016. Available online: https://cpregister.com/wp-content/uploads/2018/05/ACPR-Report_Web_2016.pdf (accessed on 18 June 2020).
- Kerr, J.; Patterson, R.E.; Ellis, K.; Godbole, S.; Johnson, E.; Lanckriet, G.; Staudenmayer, J. Objective assessment of physical activity: Classifiers for public health. Med. Sci. Sports Exerc. 2016, 48, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, D.; Godbole, S.; Ellis, K.; Di, C.; LaCroix, A.Z.; Natarajan, L.; Kerr, J. Classifiers for accelerometer-measured behaviors in older women. Med. Sci. Sports Exerc. 2017, 49, 610–616. [Google Scholar] [CrossRef] [PubMed]
- Strath, S.J.; Kate, R.J.; Keenan, K.G.; Welch, W.A.; Swartz, A.M. Ngram time series model to predict activity type and energy cost from wrist, hip and ankle accelerometers: Implications of age. Physiol. Meas. 2015, 36, 2335–2351. [Google Scholar] [CrossRef]
- Gjoreski, M.; Gjoreski, H.; Lustrek, M.; Gams, M. How accurately can your wrist device recognize daily activities and detect falls? Sensors 2016, 16, 800. [Google Scholar] [CrossRef]
- Preece, S.J.; Goulermas, J.Y.; Kenney, L.P.J.; Howard, D. A comparison of feature extraction methods for the classification of dynamic activities from accelerometer data. IEEE Trans. Biomed. Eng. 2009, 56, 871–879. [Google Scholar] [CrossRef]
- Wingstrand, M.; Hägglund, G.; Rodby-Bousquet, E. Ankle-foot orthoses in children with cerebral palsy: A cross sectional population based study of 2200 children. BMC Musculoskelet. Disord. 2014, 15, 327. [Google Scholar] [CrossRef] [PubMed]
- Lobo, M.A.; Hall, M.L.; Greenspan, B.; Rohloff, P.; Prosser, L.A.; Smith, B.A. Wearables for pediatric rehabilitation: How to optimally design and use products to meet the needs of users. Phys. Ther. 2019, 99, 647–657. [Google Scholar] [CrossRef]
- McDonald, C.M.; Widman, L.; Abresch, R.T.; Walsh, S.A.; Walsh, D.D. Utility of a step activity monitor for the measurement of daily ambulatory activity in children. Arch. Phys. Med. Rehabil. 2005, 86, 793–801. [Google Scholar] [CrossRef]
- Clanchy, K.M.; Tweedy, S.M.; Boyd, R. Measurement of habitual physical activity performance in adolescents with cerebral palsy: A systematic review. Dev. Med. Child Neurol. 2011, 53, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Bjornson, K.F.; Zhou, C.; Stevenson, R.D.; Christakis, D. Relation of stride activity and participation in mobility-based life habits among children with cerebral palsy. Arch. Phys. Med. Rehabil. 2014, 95, 360–368. [Google Scholar] [CrossRef]
- Skotte, J.; Korshøj, M.; Kristiansen, J.; Hanisch, C.; Holtermann, A. Detection of physical activity types using triaxial accelerometers. J. Phys. Act. Health 2014, 11, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Edwardson, C.L.; Rowlands, A.V.; Bunnewell, S.; Sanders, J.P.; Esliger, D.W.; Gorely, T.; O’Connell, S.; Davies, M.J.; Khunti, K.; Yates, T.E. Accuracy of posture allocation algorithms for thigh- and waist-worn accelerometers. Med. Sci. Sports Exerc. 2016, 48, 1085–1090. [Google Scholar] [CrossRef] [PubMed]
- Gjoreski, H.; Luštrek, M.; Gams, M. Accelerometer placement for posture recognition and fall detection. In Proceedings of the 2011 7th International Conference on Intelligent Environments, Nottingham, UK, 25–28 July 2011; pp. 47–54. [Google Scholar]
- Tang, Q.; John, D.; Thapa-Chhetry, B.; Arguello, D.J.; Intille, S. Posture and physical activity detection. Med. Sci. Sports Exerc. 2020. [Google Scholar] [CrossRef]
- Narayanan, A.; Stewart, T.; Mackay, L. A Dual-accelerometer system for detecting human movement in a free-living environment. Med. Sci. Sports Exerc. 2020, 52, 252–258. [Google Scholar] [CrossRef]
- O’Donoghue, D.; Kennedy, N. Validity of an activity monitor in young people with cerebral palsy gross motor function classification system level I. Physiol. Meas. 2014, 35, 2307–2318. [Google Scholar] [CrossRef]
- Pirpiris, M.; Graham, H.K. Uptime in children with cerebral palsy. J. Pediatr. Orthop. 2004, 24, 521–528. [Google Scholar] [CrossRef]
- Grant, P.M.; Ryan, C.G.; Tigbe, W.W.; Granat, M.H. The validation of a novel activity monitor in the measurement of posture and motion during everyday activities. Br. J. Sports Med. 2006, 40, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Lyden, K.; Kozey Keadle, S.L.; Staudenmayer, J.W.; Freedson, P.S. Validity of two wearable monitors to estimate breaks from sedentary time. Med. Sci. Sports Exerc. 2012, 44, 2243–2252. [Google Scholar] [CrossRef] [PubMed]
- Steeves, J.A.; Bowles, H.R.; Mcclain, J.J.; Dodd, K.W.; Brychta, R.J.; Wang, J.; Chen, K.Y. Ability of thigh-worn actigraph and activpal monitors to classify posture and motion. Med. Sci. Sports Exerc. 2015, 47, 952–959. [Google Scholar] [CrossRef]
- Goodlich, B.I.; Armstrong, E.L.; Horan, S.A.; Baque, E.; Carty, C.P.; Ahmadi, M.N.; Trost, S.G. Machine learning to quantify habitual phyisical activity in children with cerebral palsy. Dev. Med. Child Neurol. 2020. [Google Scholar] [CrossRef]
- Trost, S.G.; Cliff, D.P.; Ahmadi, M.N.; Van Tuc, N.; Hagenbuchner, M. Sensor-enabled activity class recognition in preschoolers: Hip versus wrist data. Med. Sci. Sports Exerc. 2018, 50, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, M.N.; Pfeiffer, K.A.; Trost, S.G. Physical activity classification in youth using raw accelerometer data from the hip. Meas. Phys. Educ. Exerc. Sci. 2020, 24, 129–136. [Google Scholar] [CrossRef]





| GMFCS I (N = 10) | GMFCS II (N = 20) | GMFCS III (N = 8) | |
|---|---|---|---|
| Sex | |||
| Male N (%) | 4 (40%) | 7 (35%) | 2 (25%) |
| Female N (%) | 6 (60%) | 13 (65%) | 6 (75%) |
| Motor Distribution | |||
| Hemiplegia N (%) | 8 (80%) | 13 (65%) | 0 (0%) |
| Diplegia N (%) | 2 (20%) | 7 (35%) | 5 (62.5%) |
| Quadriplegia N (%) | 0 (0%) | 0 (0%) | 3 (37.5%) |
| Age (y) | 10.8 ± 1.8 | 12.0 ± 3.1 | 11.8 ± 4.5 |
| Height (cm) | 145.1 ± 9.9 | 147.2 ± 16.7 | 129.7 ± 19.2 |
| Weight (kg) | 40.8 ± 12.6 | 42.3 ± 15.6 | 34.9 ± 17.8 |
| GMFCS I | GMFCS II | GMFCS III | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G | GP | FP | G | GP | FP | G | GP | FP | |||||||||||
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | |||||||||||
| Wrist | SED | 92.7 | (13.5) | 94.1 | (15.0) | 99.3 | (0.4) | 94.7 | (9.5) | 93.2 | (12.2) | 99.0 | (0.3) | 85.0 | (27.6) | 57.7 | (38.4) | 99.1 | (1.2) |
| SUM | 89.3 | (10.0) | 87.0 | (9.5) | 97.0 | (2.3) | 82.6 | (14.6) | 79.6 | (20.1) | 96.2 | (1.2) | 76.7 | (15.0) | 71.4 | (25.5) | 96.3 | (1.1) | |
| WALK | 96.2 | (6.6) | 91.9 | (12.4) | 98.4 | (0.9) | 90.4 | (16.4) | 92.6 | (13.7) | 97.2 | (0.6) | 84.2 | (14.1) | 75.5 | (32.4) | 96.2 | (1.2) | |
| Hip | SED | 94.4 | (12.5) | 89.1 | (16.0) | 100.0 | (0.1) | 92.1 | (13.2) | 94.4 | (10.2) | 100.0 | (0.3) | 89.3 | (22.7) | 76.9 | (34.2) | 100.0 | (0.4) |
| SUM | 83.8 | (22.2) | 76.8 | (28.7) | 98.5 | (0.8) | 87.6 | (18.6) | 85.3 | (21.8) | 98.4 | (0.4) | 75.1 | (21.3) | 73.3 | (17.6) | 98.2 | (0.2) | |
| WALK | 99.6 | (1.1) | 88.3 | (11.2) | 99.2 | (0.3) | 97.3 | (3.9) | 94.4 | (10.1) | 98.8 | (0.9) | 53.5 | (34.7) | 65.5 | (38.5) | 98.9 | (0.2) | |
| Ankle | SED | 95.8 | (5.3) | 97.6 | (2.9) | 100.0 | (0.1) | 94.7 | (12.9) | 96.6 | (11.6) | 100.0 | (0.3) | 89.8 | (26.9) | 78.5 | (37.8) | 100.0 | (0.1) |
| SUM | 91.9 | (16.2) | 91.1 | (10.0) | 98.8 | (1.3) | 90.4 | (15.3) | 93.2 | (7.9) | 99.2 | (0.6) | 71.6 | (23.1) | 74.5 | (23.6) | 99.1 | (0.8) | |
| WALK | 99.6 | (1.1) | 99.0 | (2.3) | 98.7 | (1.5) | 98.0 | (2.4) | 96.8 | (5.5) | 99.4 | (0.5) | 84.0 | (16.8) | 96.3 | (4.4) | 99.0 | (1.1) | |
| W + H | SED | 89.2 | (22.6) | 94.0 | (14.0) | 100.0 | (0.2) | 94.4 | (11.9) | 94.9 | (11.2) | 100.0 | (0.2) | 99.6 | (1.1) | 75.0 | (30.2) | 100.0 | (0.4) |
| SUM | 90.8 | (16.9) | 87.0 | (25.4) | 99.1 | (1.3) | 85.2 | (17.6) | 88.1 | (20.2) | 99.3 | (1.2) | 92.1 | (10.4) | 80.4 | (15.2) | 99.8 | (0.7) | |
| WALK | 90.9 | (14.9) | 98.3 | (2.5) | 99.3 | (1.1) | 94.9 | (11.9) | 96.2 | (5.3) | 99.1 | (1.3) | 95.8 | (4.4) | 94.1 | (8.1) | 99.7 | (0.4) | |
| W + A | SED | 94.2 | (12.2) | 98.4 | (3.2) | 100.0 | (0.2) | 93.9 | (12.1) | 97.3 | (10.0) | 100.0 | (0.1) | 92.0 | (18.6) | 80.1 | (33.4) | 100.0 | (0.2) |
| SUM | 91.6 | (11.4) | 91.9 | (12.3) | 98.1 | (2.3) | 93.8 | (10.4) | 93.7 | (6.5) | 99.2 | (1.7) | 68.6 | (23.2) | 81.1 | (14.9) | 99.5 | (1.0) | |
| WALK | 99.6 | (1.1) | 99.0 | (2.3) | 98.4 | (1.4) | 97.6 | (3.7) | 97.9 | (2.8) | 99.4 | (1.9) | 90.5 | (9.9) | 96.8 | (3.7) | 98.2 | (1.7) | |
| H + A | SED | 94.6 | (10.5) | 96.9 | (7.0) | 100.0 | (0.1) | 96.3 | (7.9) | 96.5 | (11.7) | 100.0 | (0.2) | 86.9 | (27.1) | 76.3 | (35.2) | 100.0 | (0.2) |
| SUM | 87.3 | (21.7) | 89.4 | (14.1) | 98.6 | (1.6) | 93.8 | (12.9) | 92.1 | (11.1) | 98.2 | (1.3) | 70.5 | (20.6) | 73.6 | (24.1) | 99.4 | (0.7) | |
| WALK | 99.6 | (1.1) | 98.6 | (3.3) | 99.3 | (0.8) | 98.3 | (2.3) | 97.4 | (2.4) | 98.9 | (1.4) | 87.8 | (13.6) | 96.3 | (4.5) | 99.2 | (0.4) | |
| W + H + A | SED | 95.3 | (10.4) | 96.4 | (7.9) | 100.0 | (0.2) | 96.2 | (9.6) | 97.0 | (7.2) | 100.0 | (0.2) | 87.5 | (27.6) | 81.3 | (32.7) | 100.0 | (0.2) |
| SUM | 91.9 | (12.9) | 91.0 | (12.4) | 99.3 | (1.2) | 93.8 | (8.5) | 91.4 | (10.7) | 99.1 | (0.7) | 77.2 | (19.8) | 82.1 | (14.7) | 98.8 | (1.4) | |
| WALK | 99.6 | (1.1) | 98.8 | (2.8) | 99.5 | (0.4) | 98.1 | (2.8) | 98.2 | (2.6) | 99.2 | (0.9) | 89.4 | (11.1) | 96.8 | (3.7) | 99.0 | (1.1) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmadi, M.N.; O’Neil, M.E.; Baque, E.; Boyd, R.N.; Trost, S.G. Machine Learning to Quantify Physical Activity in Children with Cerebral Palsy: Comparison of Group, Group-Personalized, and Fully-Personalized Activity Classification Models. Sensors 2020, 20, 3976. https://doi.org/10.3390/s20143976
Ahmadi MN, O’Neil ME, Baque E, Boyd RN, Trost SG. Machine Learning to Quantify Physical Activity in Children with Cerebral Palsy: Comparison of Group, Group-Personalized, and Fully-Personalized Activity Classification Models. Sensors. 2020; 20(14):3976. https://doi.org/10.3390/s20143976
Chicago/Turabian StyleAhmadi, Matthew N., Margaret E. O’Neil, Emmah Baque, Roslyn N. Boyd, and Stewart G. Trost. 2020. "Machine Learning to Quantify Physical Activity in Children with Cerebral Palsy: Comparison of Group, Group-Personalized, and Fully-Personalized Activity Classification Models" Sensors 20, no. 14: 3976. https://doi.org/10.3390/s20143976
APA StyleAhmadi, M. N., O’Neil, M. E., Baque, E., Boyd, R. N., & Trost, S. G. (2020). Machine Learning to Quantify Physical Activity in Children with Cerebral Palsy: Comparison of Group, Group-Personalized, and Fully-Personalized Activity Classification Models. Sensors, 20(14), 3976. https://doi.org/10.3390/s20143976






