Atomic Force Microscopy Imaging in Turbid Liquids: A Promising Tool in Nanomedicine
Abstract
1. Introduction
2. Materials and Methods
2.1. Optical Beam Deflection Measurements
2.2. Self-Sensing Cantilevers
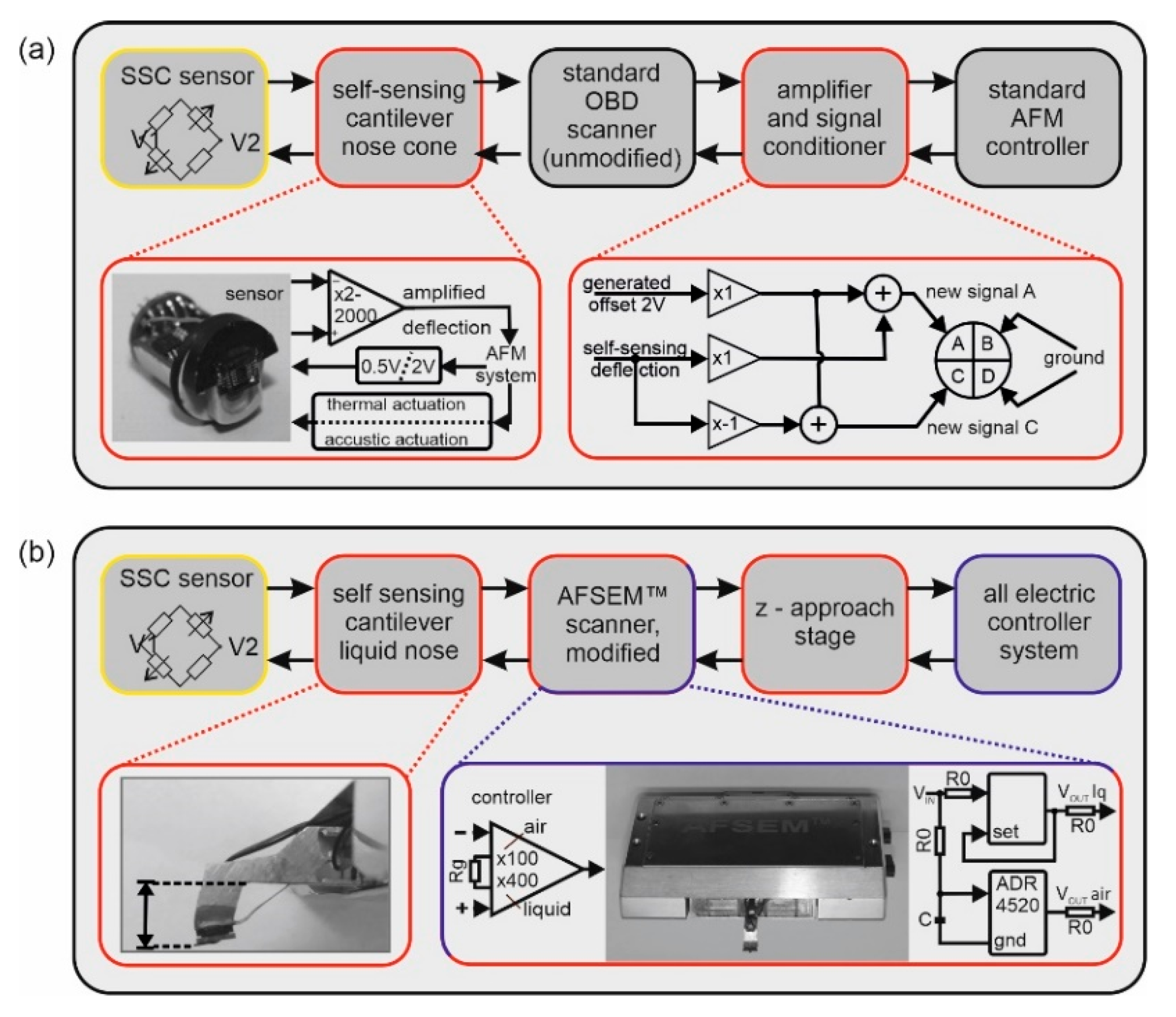
2.3. Implementation of the SSC in a Commercial AFM with Optical Beam Deflection
2.4. Software Implementation
2.5. Adaptation of the AFSEM® for Measurements in Liquids
2.6. Preparation of the Cell Samples
2.7. Sample Preparations for Measurements in Blood, Blood Serum and Ink
2.8. Speed and Noise Measurements
3. Results
3.1. System Integration and Adaptation
3.2. Imaging of Biological Samples in Dry State
3.3. Combined AFM–SEM Investigations and Measurements in Different Environments
3.4. Detailed System Characterization
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ando, T. High-speed atomic force microscopy and its future prospects. Biophys. Rev. 2018, 10, 285–292. [Google Scholar] [CrossRef]
- Li, M.; Xi, N.; Wang, Y.; Liu, L. Atomic Force Microscopy in Probing Tumor Physics for Nanomedicine. IEEE Trans. Nanotechnol. 2019, 18, 83–113. [Google Scholar] [CrossRef]
- Santos, N.C.; Carvalho, F.A. Atomic Force Microscopy; Springer: Berlin, Germany, 2019. [Google Scholar]
- Lamprecht, C.; Hinterdorfer, P.; Ebner, A. Applications of biosensing atomic force microscopy in monitoring drug and nanoparticle delivery. Expert Opin. Drug Deliv. 2014, 11, 1237–1253. [Google Scholar] [CrossRef]
- Dufrêne, Y.F.; Ando, T.; Garcia, R.; Alsteens, D.; Martinez-Martin, D.; Engel, A.; Gerber, C.; Müller, D.J. Imaging modes of atomic force microscopy for application in molecular and cell biology. Nat. Nanotechnol. 2017, 12, 295–307. [Google Scholar] [CrossRef]
- Tortonese, M.; Yamada, H.; Barrett, R.C.; Quate, C.F. Atomic force microscopy using a piezoresistive cantilever. In Proceedings of the TRANSDUCERS ’91: 1991 International Conference on Solid-State Sensors and Actuators. Digest of Technical Papers, San Francisco, CA, USA, 24–27 June 1991. [Google Scholar]
- Tortonese, M. Atomic resolution with an atomic force microscope using piezoresistive detection. Appl. Phys. Lett. 1993, 62, 834–836. [Google Scholar] [CrossRef]
- Linnemann, R.; Gotszalk, T.; Hadjiiski, L.; Rangelow, I.W. Characterization of a cantilever with an integrated deflection sensor. Thin Solid Films 1995, 264, 159–164. [Google Scholar] [CrossRef]
- Jumpertz, R.; Hart, A.V.D.; Ohlsson, O.; Saurenbach, F.; Schelten, J. Piezoresistive sensors on AFM cantilevers with atomic resolution. Microelectr. Eng. 1998, 41–42, 441–444. [Google Scholar] [CrossRef]
- Thaysen, J.; Boisen, A.; Hansen, O.; Bouwstra, S. Atomic force microscopy probe with piezoresistive read-out and a highly symmetrical Wheatstone bridge arrangement. Sens. Actuators A Phys. 2000, 83, 47–53. [Google Scholar] [CrossRef]
- Yu, X.; Thaysen, J.; Hansen, O.; Boisen, A. Optimization of sensitivity and noise in piezoresistive cantilevers. J. Appl. Phys. 2002, 92, 6296–6301. [Google Scholar] [CrossRef]
- Doll, J.C.; Pruitt, B.L. High-bandwidth piezoresistive force probes with integrated thermal actuation. J. Micromech. Microeng. 2012, 22, 095012. [Google Scholar] [CrossRef]
- Tosolini, G.; Scarponi, F.; Cannistraro, S.; Bausells, J. Biomolecule recognition using piezoresistive nanomechanical force probes. Appl. Phys. Lett. 2013, 102, 253701. [Google Scholar] [CrossRef]
- Bausells, J. Piezoresistive cantilevers for nanomechanical sensing. Microelectr. Eng. 2015, 145, 9–20. [Google Scholar] [CrossRef]
- Dukic, M.; Adams, J.D.; Fantner, G.E. Piezoresistive AFM cantilevers surpassing standard optical beam deflection in low noise topography imaging. Sci. Rep. 2015, 5, 16393. [Google Scholar] [CrossRef]
- Kunicki, P.; Angelov, T.; Ivanov, T.; Gotszalk, T.; Rangelow, I. Sensitivity Improvement to Active Piezoresistive AFM Probes Using Focused Ion Beam Processing. Sensors 2019, 19, 4429. [Google Scholar] [CrossRef]
- Brugger, J.; Buser, R.; de Rooij, N. Micromachined atomic force microprobe with integrated capacitive read-out. J. Micromech. Microeng. 1992, 2, 218. [Google Scholar] [CrossRef]
- Blanc, N.; Brugger, J.; De Rooij, N.F.; Dürig, U. Scanning force microscopy in the dynamic mode using microfabricated capacitive sensors. J. Vac. Sci. Technol. B Microelectr. Nanometer Struct. Process. Meas. Phenom. 1996, 14, 901–905. [Google Scholar] [CrossRef][Green Version]
- Forsen, E.; Abadal, G.; Ghatnekar-Nilsson, S.; Teva, J.; Verd, J.; Sandberg, R.; Svendsen, W.; Perez-Murano, F.; Esteve, J.; Figueras, E.; et al. Ultrasensitive mass sensor fully integrated with complementary metal-oxide-semiconductor circuitry. Appl. Phys. Lett. 2005, 87, 043507. [Google Scholar] [CrossRef]
- Kim, S.-J.; Ono, T.; Esashi, M. Capacitive resonant mass sensor with frequency demodulation detection based on resonant circuit. Appl. Phys. Lett. 2006, 88, 053116. [Google Scholar] [CrossRef]
- Verd, J.; Uranga, A.; Abadal, G.; Teva, J.; Torres, F.; Pérez-Murano, F.; Fraxedas, J.; Esteve, J.; Barniol, N. Monolithic mass sensor fabricated using a conventional technology with attogram resolution in air conditions. Appl. Phys. Lett. 2007, 91, 013501. [Google Scholar] [CrossRef]
- Itoh, T.; Suga, T. Development of a force sensor for atomic force microscopy using piezoelectric thin films. Nanotechnology 1993, 4, 218. [Google Scholar] [CrossRef]
- Lee, J.H.; Hwang, K.S.; Park, J.; Yoon, K.H.; Yoon, D.S.; Kim, T.S. Immunoassay of prostate-specific antigen (PSA) using resonant frequency shift of piezoelectric nanomechanical microcantilever. Biosens. Bioelectr. 2005, 20, 2157–2162. [Google Scholar] [CrossRef] [PubMed]
- Karabalin, R.B.; Matheny, M.H.; Feng, X.L.; Defaÿ, E.; Le Rhun, G.; Marcoux, C.; Hentz, S.; Andreucci, P.; Roukes, M.L. Piezoelectric nanoelectromechanical resonators based on aluminum nitride thin films. Appl. Phys. Lett. 2009, 95, 103111. [Google Scholar] [CrossRef]
- Ivaldi, P.; Abergel, J.; Matheny, M.H.; Villanueva, L.G.; Karabalin, R.B.; Roukes, M.L.; Andreucci, P.; Hentz, S.; Defaÿ, E. 50 nm thick AlN film-based piezoelectric cantilevers for gravimetric detection. J. Micromech. Microeng. 2011, 21, 085023. [Google Scholar] [CrossRef]
- Moore, S.I.; Ruppert, M.G.; Yong, Y.K. An optimization framework for the design of piezoelectric AFM cantilevers. Precis. Eng. 2019, 60, 130–142. [Google Scholar] [CrossRef]
- Choi, Y.-S.; Gwak, M.-J.; Lee, D.-W. Polymeric cantilever integrated with PDMS/graphene composite strain sensor. Rev. Sci. Instrum. 2016, 87, 105004. [Google Scholar] [CrossRef]
- Akiyama, T.; Staufer, U.; De Rooij, N.F.; Frederix, P.L.T.M.; Engel, A. Symmetrically arranged quartz tuning fork with soft cantilever for intermittent contact mode atomic force microscopy. Rev. Sci. Instrum. 2003, 74, 112–117. [Google Scholar] [CrossRef]
- Gonzalez, L.; Martínez-Martín, D.; Otero, J.; De Pablo, P.J.; Puig-Vidal, M.; Gómez-Herrero, J. Improving the lateral resolution of quartz tuning fork-based sensors in liquid by integrating commercial AFM tips into the fiber end. Sensors 2015, 15, 1601–1610. [Google Scholar] [CrossRef]
- Saitoh, K.; Taguchi, A.; Kawata, S. Tip-Enhanced Raman Scattering Microscope Using Quartz-Tuning-Fork AFM Probe. In JSAP-OSA Joint Symposia; Optical Society of America: Washington, DC, USA, 2017. [Google Scholar]
- Voigtländer, B. Quartz Sensors in Atomic Force Microscopy. In Atomic Force Microscopy; Springer: Berlin, Germany, 2019; pp. 301–307. [Google Scholar]
- Li, M.; Tang, H.X.; Roukes, M.L. Ultra-sensitive NEMS-based cantilevers for sensing, scanned probe and very high-frequency applications. Nat. Nanotechnol. 2007, 2, 114. [Google Scholar] [CrossRef]
- Dukic, M.; Winhold, M.; Schwalb, C.H.; Adams, J.D.; Stavrov, V.; Huth, M.; Fantner, G.E. Direct-write nanoscale printing of nanogranular tunnelling strain sensors for sub-micrometre cantilevers. Nat. Commun. 2016, 7, 12487. [Google Scholar] [CrossRef]
- Yoshikawa, G.; Lang, H.P.; Akiyama, T.; Aeschimann, L.; Staufer, U.; Vettiger, P.; Aono, M.; Sakurai, T.; Gerber, C. Sub-ppm detection of vapors using piezoresistive microcantilever array sensors. Nanotechnology 2008, 20, 015501. [Google Scholar] [CrossRef]
- Holz, M.; Reuter, C.; Ahmad, A.; Reum, A.; Ivanov, T.; Guliyev, E.; Rangelow, I.W.; Lee, H.S. Parallel Active Cantilever AFM Tool for High-Throughput Inspection and Metrology; SPIE Advanced Lithography; SPIE: Washington, DC, USA, 2019; Volume 10959. [Google Scholar]
- Somnath, S.; Kim, H.J.; Hu, H.; King, W.P. Parallel nanoimaging and nanolithography using a heated microcantilever array. Nanotechnology 2013, 25, 014001. [Google Scholar] [CrossRef] [PubMed]
- Kreith, J.; Strunz, T.; Fantner, E.J.; Fantner, G.E.; Cordill, M.J. A versatile atomic force microscope integrated with a scanning electron microscope. Rev. Sci. Instrum. 2017, 88, 053704. [Google Scholar] [CrossRef] [PubMed]
- Winhold, M.; Leitner, M.; Lieb, A.; Frederix, P.; Hofbauer, F.; Strunz, T.; Sattelkov, J.; Plank, H.; Schwalb, C.H. Correlative In-Situ AFM & SEM & EDX Analysis of Nanostructured Materials. Microsc. Microanal. 2017, 23, 26–27. [Google Scholar]
- Angelov, T.; Ahmad, A.; Guliyev, E.; Reum, A.; Atanasov, I.; Ivanov, T.; Ishchuk, V.; Kaestner, M.; Krivoshapkina, Y.; Lenk, S.; et al. Six-axis AFM in SEM with self-sensing and self-transduced cantilever for high speed analysis and nanolithography. J. Vac. Sci. Technol. B Microelectr. Nanometer Struct. Process. Meas. Phenom. 2016, 34, 06KB01. [Google Scholar] [CrossRef]
- Rangelow, I.W.; Kaestner, M.; Ivanov, T.; Ahmad, A.; Lenk, S.; Lenk, C.; Guliyev, E.; Reum, A.; Hofmann, M.; Reuter, C.; et al. Atomic force microscope integrated with a scanning electron microscope for correlative nanofabrication and microscopy. J. Vac. Sci. Technol. B 2018, 36, 06J102. [Google Scholar] [CrossRef]
- Rogers, B.; York, D.; Whisman, N.; Jones, M.; Murray, K.; Adams, J.D.; Sulchek, T.; Minne, S.C. Tapping mode atomic force microscopy in liquid with an insulated piezoelectric microactuator. Rev. Sci. Instrum. 2002, 73, 3242–3244. [Google Scholar] [CrossRef]
- Buguin, A.; Roure, O.D.; Silberzan, P. Active atomic force microscopy cantilevers for imaging in liquids. Appl. Phys. Lett. 2001, 78, 2982–2984. [Google Scholar] [CrossRef]
- Florin, E.L.; Radmacher, M.; Fleck, B.; Gaub, H.E. Atomic force microscope with magnetic force modulation. Rev. Sci. Instrum. 1994, 65, 639–643. [Google Scholar] [CrossRef]
- Han, W.; Lindsay, S.; Jing, T. A magnetically driven oscillating probe microscope for operation in liquids. Appl. Phys. Lett. 1996, 69, 4111–4113. [Google Scholar] [CrossRef]
- Lantz, M.; O’shea, S.; Welland, M. Force microscopy imaging in liquids using ac techniques. Appl. Phys. Lett. 1994, 65, 409–411. [Google Scholar] [CrossRef]
- Revenko, I.; Proksch, R. Magnetic and acoustic tapping mode microscopy of liquid phase phospholipid bilayers and DNA molecules. J. Appl. Phys. 2000, 87, 526–533. [Google Scholar] [CrossRef]
- Pedrak, R.; Ivanov, T.; Ivanova, K.; Gotszalk, T.; Abedinov, N.; Rangelow, I.W.; Edinger, K.; Tomerov, E.; Schenkel, T.; Hudek, P. Micromachined atomic force microscopy sensor with integrated piezoresistive sensor and thermal bimorph actuator for high-speed tapping-mode atomic force microscopy phase-imaging in higher eigenmodes. J. Vac. Sci. Technol. B Microelectr. Nanometer Struct. Process. Meas. Phenom. 2003, 21, 3102–3107. [Google Scholar] [CrossRef]
- Fantner, G.E.; Burns, D.J.; Belcher, A.M.; Rangelow, I.W.; Youcef-Toumi, K. DMCMN: In depth characterization and control of AFM cantilevers with integrated sensing and actuation. J. Dyn. Syst. Meas. Control 2009, 131, 061104. [Google Scholar] [CrossRef]
- Herruzo, E.T.; Garcia, R. Frequency response of an atomic force microscope in liquids and air: Magnetic versus acoustic excitation. Appl. Phys. Lett. 2007, 91, 143113. [Google Scholar] [CrossRef]
- Winhold, M.; Leitner, M.; Frank, P.; Hosseini, N.; Sattelkov, J.; Fantner, G.E.; Plank, H.; Schwalb, C.H. Correlative In-Situ Analysis on the Nanoscale by combination of AFM and SEM. Microsc. Microanal. 2018, 24, 1922–1923. [Google Scholar] [CrossRef][Green Version]
- Frank, P.; Leitner, M.; Hummel, S.; Hosseini, N.; Wang, Y.; Winkler, R.; Fantner, G.E.; Plank, H.; Zeng, Y.; Schwalb, C.H. Crystallographic and Nanomechanical Analysis by Correlative In-situ AFM & SEM. Microsc. Microanal. 2018, 24, 2280–2281. [Google Scholar]
- Sattelkow, J.; Fröch, J.E.; Winkler, R.; Hummel, S.; Schwalb, C.; Plank, H. Three-Dimensional Nanothermistors for Thermal Probing. ACS Appl. Mater. Interfaces 2019, 11, 22655–22667. [Google Scholar] [CrossRef]
- Semenenko, B.; Esquinazi, P. Diamagnetism of bulk graphite revised. Magnetochemistry 2018, 4, 52. [Google Scholar] [CrossRef]
- Huber, C.; Reith, P.; Badarlis, A. Gas Density and Viscosity Measurement with a Microcantilever for Online Combustion Gas Monitoring. In Proceedings of the Sensors and Measuring Systems, 19th ITG/GMA-Symposium, Nuremberg, Germany, 26–27 June 2018; VDE: Berlin, Germany. [Google Scholar]
- Xia, F.; Yang, C.; Wang, Y.; Youcef-Toumi, K.; Reuter, C.; Ivanov, T.; Holz, M.; Rangelow, I.W. Lights Out! Nano-Scale Topography Imaging of Sample Surface in Opaque Liquid Environments with Coated Active Cantilever Probes. Nanomaterials 2019, 9, 1013. [Google Scholar]
- Fantner, G.E.; Schumann, W.; Barbero, R.J.; Deutschinger, A.; Todorov, V.; Gray, D.S.; Belcher, A.M.; Rangelow, I.W.; Youcef-Toumi, K. Use of self-actuating and self-sensing cantilevers for imaging biological samples in fluid. Nanotechnology 2009, 20, 434003. [Google Scholar] [CrossRef]
- Karagkiozaki, V.; Pappa, F.; Arvaniti, D.; Moumkas, A.; Konstantinou, D.; Logothetidis, S. The melding of nanomedicine in thrombosis imaging and treatment: A review. Future Sci. 2016, 2, FSO113. [Google Scholar] [CrossRef] [PubMed]
- Posch, S.; Neundlinger, I.; Leitner, M.; Siostrzonek, P.; Panzer, S.; Hinterdorfer, P.; Ebner, A. Activation induced morphological changes and integrin αIIbβ3 activity of living platelets. Methods 2013, 60, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Butt, H.J.; Siedle, P.; Seifert, K.; Fendler, K.; Seeger, T.; Bamberg, E.; Weisenhorn, A.L.; Goldie, K.; Engel, A. Scan speed limit in Atomic force microscopy. J. Microsc. 1993, 169, 75–84. [Google Scholar] [CrossRef]
- Rosenson, R.S.; Mccormick, A.; Uretz, E.F. Distribution of blood viscosity values and biochemical correlates in healthy adults. Clin. Chem. 1996, 42, 1189–1195. [Google Scholar] [CrossRef] [PubMed]



| Noise in Different Media | Dry State [Å] | Deionized Water [Å] | Diluted Ink [Å] | Blood Sera [Å] | Diluted Blood [Å] | Pure Blood [Å] | |
|---|---|---|---|---|---|---|---|
| Topography | RMS * | 2.91 | 3.64 | 3.36 | 3.62 | 3.03 | 3.52 |
| SD # | 0.024 | 0.02 | 0.043 | 0.15 | 0.077 | 0.069 | |
| Deflection | RMS * | 2.63 | 3.00 | 2.67 | 2.50 | 2.31 | 3.10 |
| SD # | 0.026 | 0.042 | 0.023 | 0.07 | 0.026 | 0.15 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leitner, M.; Seferovic, H.; Stainer, S.; Buchroithner, B.; Schwalb, C.H.; Deutschinger, A.; Ebner, A. Atomic Force Microscopy Imaging in Turbid Liquids: A Promising Tool in Nanomedicine. Sensors 2020, 20, 3715. https://doi.org/10.3390/s20133715
Leitner M, Seferovic H, Stainer S, Buchroithner B, Schwalb CH, Deutschinger A, Ebner A. Atomic Force Microscopy Imaging in Turbid Liquids: A Promising Tool in Nanomedicine. Sensors. 2020; 20(13):3715. https://doi.org/10.3390/s20133715
Chicago/Turabian StyleLeitner, Michael, Hannah Seferovic, Sarah Stainer, Boris Buchroithner, Christian H. Schwalb, Alexander Deutschinger, and Andreas Ebner. 2020. "Atomic Force Microscopy Imaging in Turbid Liquids: A Promising Tool in Nanomedicine" Sensors 20, no. 13: 3715. https://doi.org/10.3390/s20133715
APA StyleLeitner, M., Seferovic, H., Stainer, S., Buchroithner, B., Schwalb, C. H., Deutschinger, A., & Ebner, A. (2020). Atomic Force Microscopy Imaging in Turbid Liquids: A Promising Tool in Nanomedicine. Sensors, 20(13), 3715. https://doi.org/10.3390/s20133715






