A Microservices e-Health System for Ecological Frailty Assessment Using Wearables †
Abstract
1. Introduction
2. Related Work
3. Materials and Methods
3.1. Sample Description
3.2. Fried Test and Frailty Status Variable
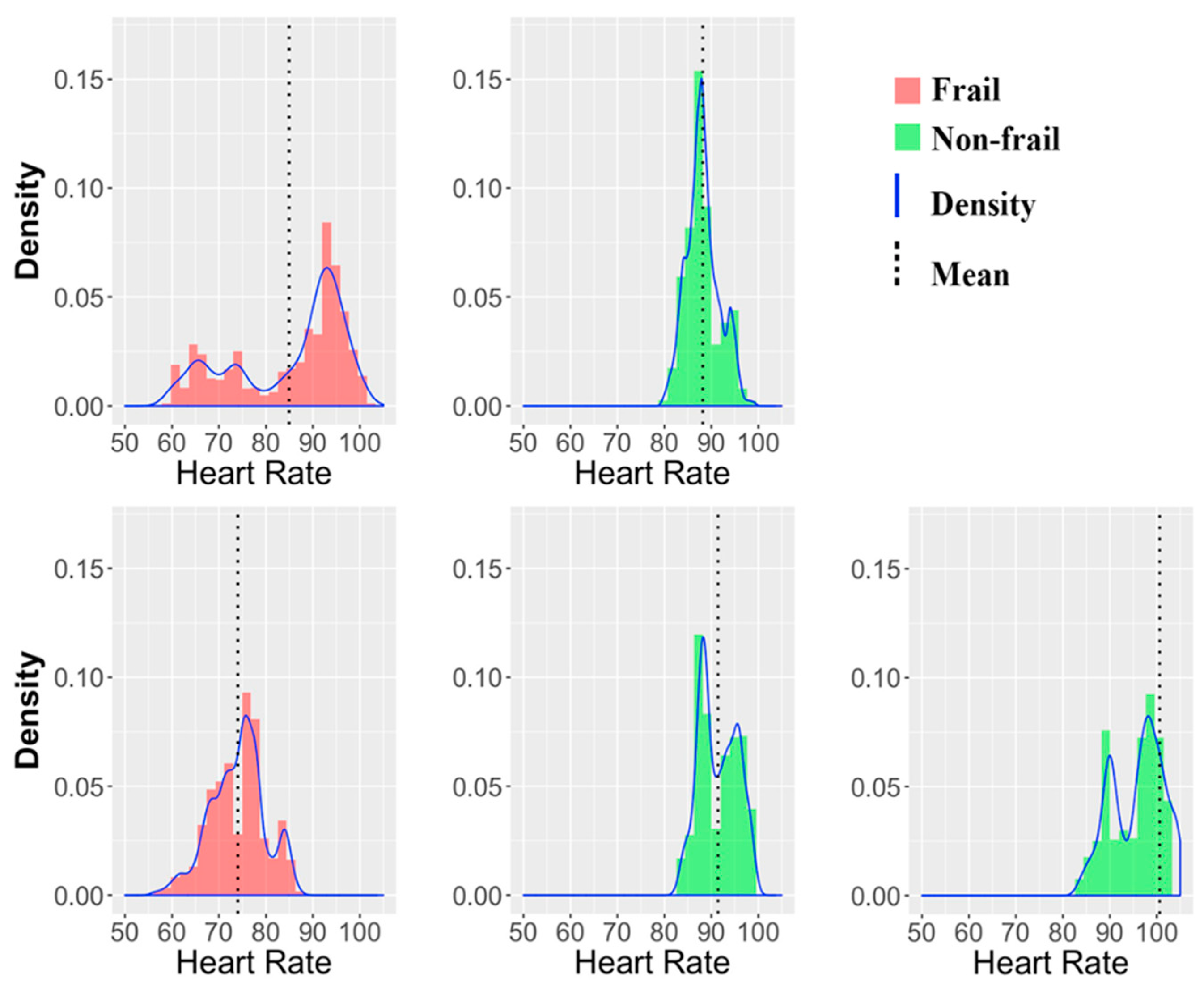
3.3. Wearable Sensors Variables
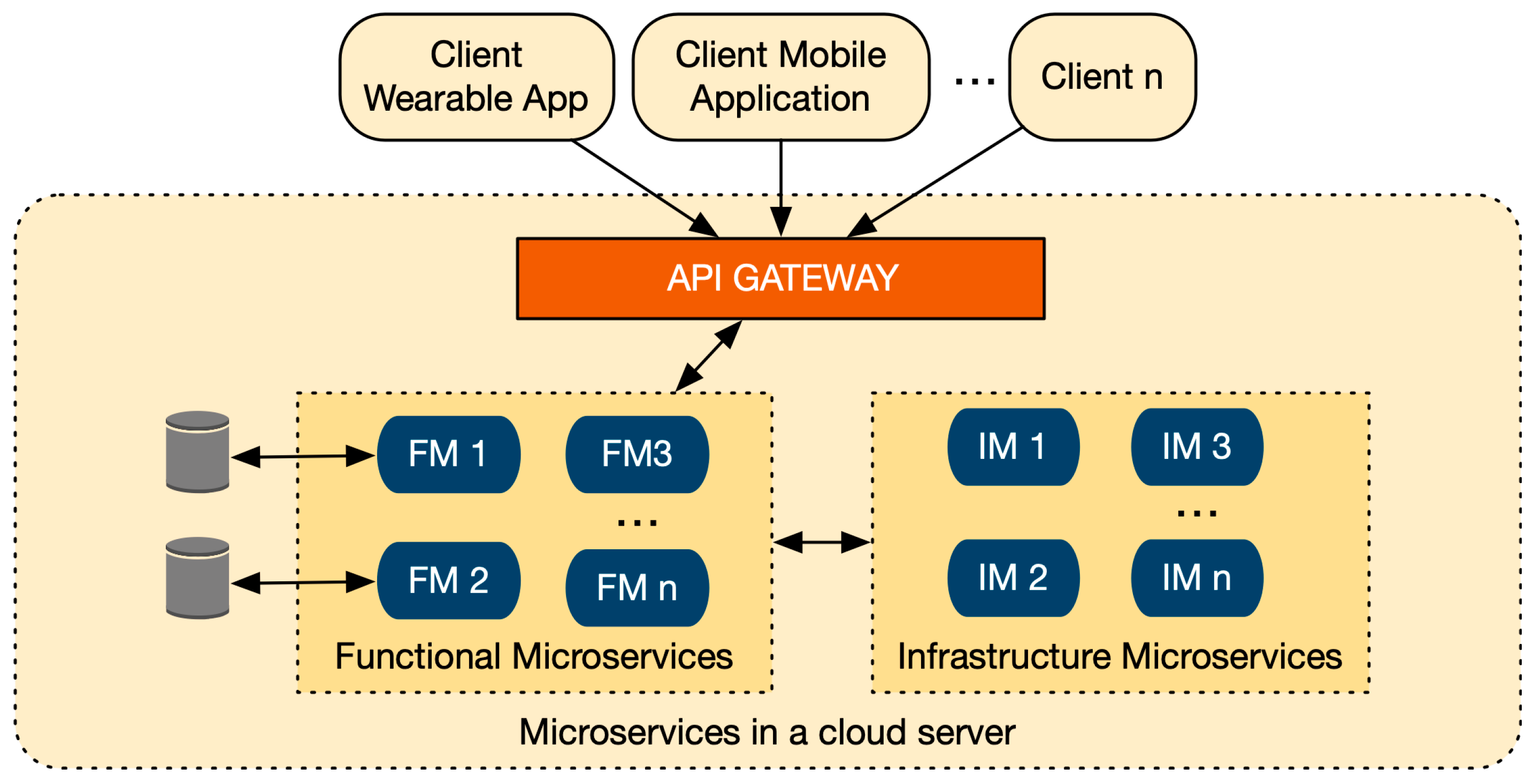
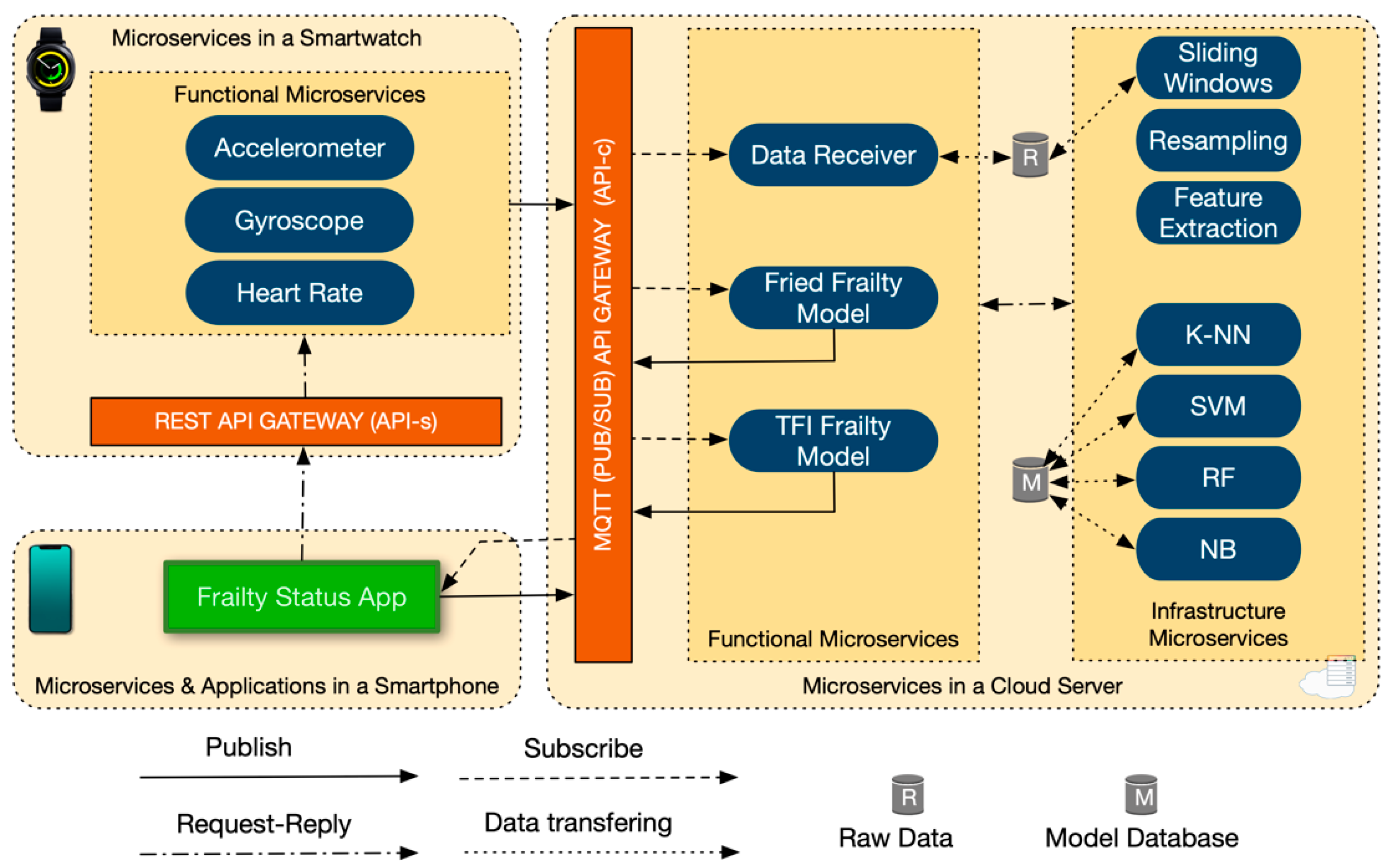
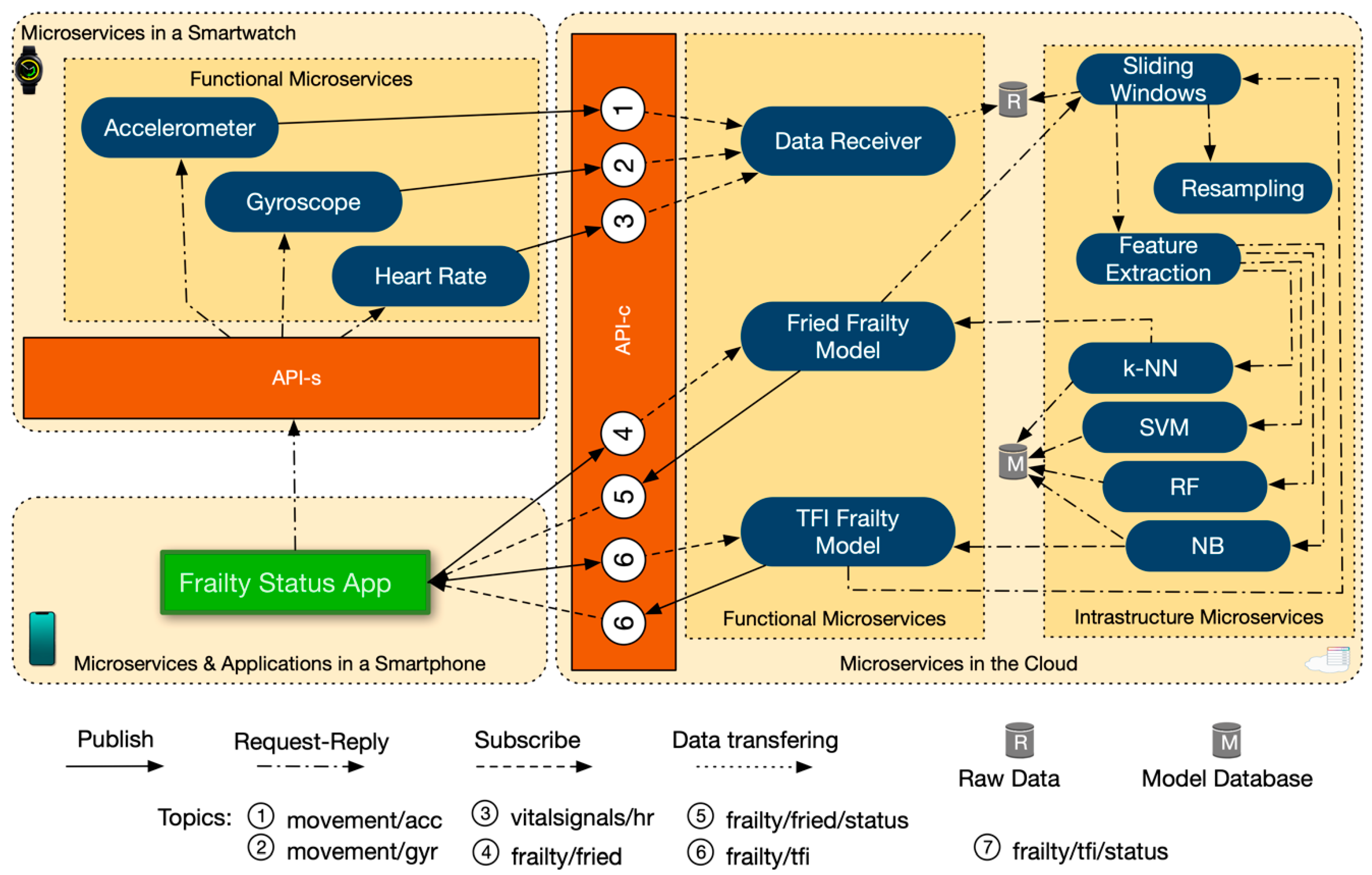
3.4. Microservices System Architecture
3.4.1. Microservices Deployed in Wearable Devices
3.4.2. App Deployed in the Smartphone
3.4.3. Microservices Deployed in the Cloud Server
3.4.4. Workflow
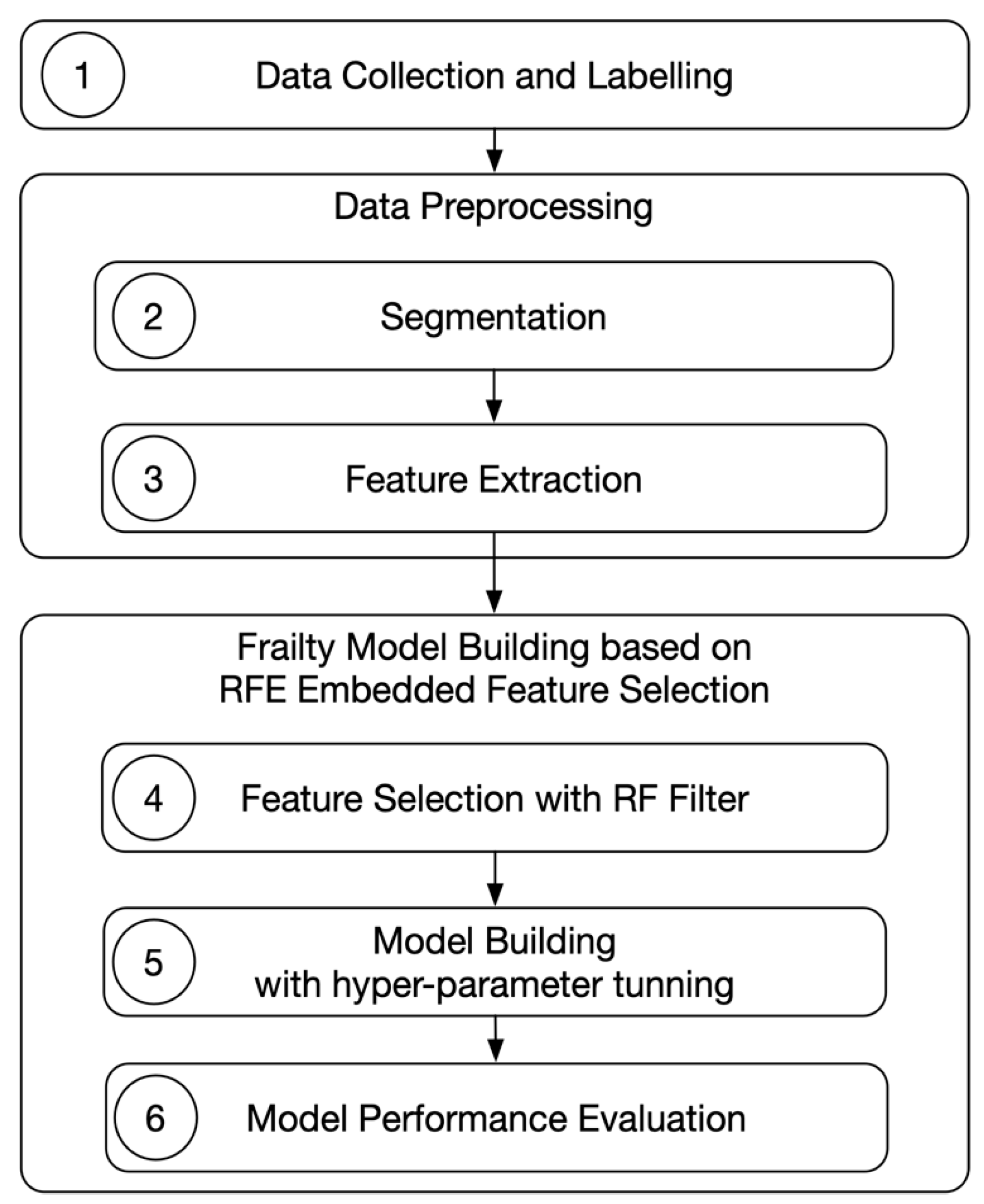
3.5. The Data Analysis Pipeline to Build a Predictive Model for Frailty Assessment
3.5.1. Data Collection and Labelling Process
3.5.2. Data Preprocessing
Segmentation
Feature Extraction
3.5.3. Frailty Model
Feature Selection
Frailty Model Building
- k-NN: (1) k: {1, 3, 5, ..., square of number of rows} (only odd numbers).
- SVM: (1) cost function: {0.1, 1, 10, 100}; (2) gamma value: {0.5, 1, 2}; (3) and, kernel type: {“radial”, “polynomial”, “linear”, “sigmoid”}.
- RF: (1) number of trees: {10, 100, 200, 500, 1000}; (2) number of variables randomly sampled: {10, 25, 50}.
- NB: (1) use of kernel: {True, False}; (2) use of poisson: {True, False}.
- k-NN: O(nm)
- SVM: O(n2m + n3)
- RF: O(n2mntrees)
- NB: O(nm)
- k-NN: O(nm)
- SVM: O(nsvm), where nsv is the number of support vectors, which is the resulting points of the SVM model, close to the decision boundary.
- RF: O(ntreesm)
- NB: O(m)
Model Performance Evaluation
4. Results
4.1. System Validation Results
4.2. Frailty Model Results
5. Discussion
6. Conclusions and Future Work
Author Contributions
Funding
Conflicts of Interest
References
- Carretero, L.; Navarro-Pardo, E.; Cano, A. Progression in healthy ageing: Frailty, cognitive decline and gender in the European Innovation Partnership for Active and Healthy Ageing. Eur. J. Psychiatry 2015, 29, 231–237. [Google Scholar] [CrossRef]
- Xue, Q.-L. The Frailty Syndrome: Definition and Natural History. Clin. Geriatr. Med. 2011, 27, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Toosizadeh, N.; Wendel, C.; Hsu, C.-H.; Zamrini, E.; Mohler, J. Frailty assessment in older adults using upper-extremity function: Index development. BMC Geriatr. 2017, 17, 117. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older Adults: Evidence for a Phenotype. J. Gerontol. Med. Sci. Am. 2001, 56, 146–156. [Google Scholar] [CrossRef]
- Van Velsen, L.; Illario, M.; Jansen-Kosterink, S.; Crola, C.; Di Somma, C.; Colao, A.; Vollenbroek-Hutten, M. A Community-Based, Technology-Supported Health Service for Detecting and Preventing Frailty among Older Adults: A Participatory Design Development Process. J. Aging Res. 2015, 2015, 216084. [Google Scholar] [CrossRef]
- Gobbens, R.J.; Luijkx, K.G.; Wijnen-Sponselee, M.T.; Schols, J.M. Toward a conceptual definition of frail community dwelling older people. Nurs. Outlook 2010, 58, 76–86. [Google Scholar] [CrossRef]
- Gobbens, R.J.J.; van Assen, M.A.L.M.; Luijkx, K.G.; Wijnen-Sponselee, M.T.; Schols, J.M.G.A. The Tilburg Frailty Indicator: Psychometric Properties. J. Am. Med. Dir. Assoc. 2010, 11, 344–355. [Google Scholar] [CrossRef]
- De Witte, N.; Gobbens, R.; De Donder, L.; Dury, S.; Buffel, T.; Schols, J.; Verté, D. The comprehensive frailty assessment instrument: Development, validity and reliability. Geriatr. Nurs. 2013, 34, 274–281. [Google Scholar] [CrossRef]
- Pérez Mármol, J.M.; Flores Antigüedad, M.L.; Castro Sánchez, A.M.; Tapia Haro, R.M.; García Ríos, M.; Aguilar Ferrándiz, M.E. Inpatient dependency in activities of daily living predicts informal caregiver strain: A cross-sectional study. J. Clin. Nurs. 2018, 27, e177–e185. [Google Scholar] [CrossRef]
- Kekade, S.; Hseieh, C.-H.; Islam, M.M.; Atique, S.; Mohammed Khalfan, A.; Li, Y.-C.; Abdul, S.S. The usefulness and actual use of wearable devices among the elderly population. Comput. Methods Programs Biomed. 2018, 153, 137–159. [Google Scholar] [CrossRef]
- Harari, Y.N. Yuval Noah Harari: The World after Coronavirus. Available online: https://www.ft.com/content/19d90308-6858-11ea-a3c9-1fe6fedcca75 (accessed on 1 April 2020).
- Majumder, S.; Mondal, T.; Deen, M. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Wang Member, F.; Stone Student Member, E.; Skubic Member, M.; Keller Fellow, J.M.; Abbott, C.; Rantz Member, M. Towards a Passive Low-Cost In-Home Gait Assessment System for Older Adults. IEEE J. Biomed. Health Inf. 2013, 17, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Tegou, T.; Kalamaras, I.; Tsipouras, M.; Giannakeas, N.; Votis, K.; Tzovaras, D. A Low-Cost Indoor Activity Monitoring System for Detecting Frailty in Older Adults. Sensors 2019, 19, 452. [Google Scholar] [CrossRef] [PubMed]
- Schwenk, M.; Mohler, J.; Wendel, C.; D’Huyvetter, K.; Fain, M.; Taylor-Piliae, R.; Najafi, B. Wearable Sensor-Based In-Home Assessment of Gait, Balance, and Physical Activity for Discrimination of Frailty Status: Baseline Results of the Arizona Frailty Cohort Study. Gerontology 2015, 61, 258–267. [Google Scholar] [CrossRef]
- Kumar, D.P.; Toosizadeh, N.; Mohler, J.; Laksari, K. Frailty Assessment Based on the Quality of Daily Walking. Innov. Aging 2019, 3, S85. [Google Scholar] [CrossRef]
- Greene, B.R.; McManus, K.; Redmond, S.J.; Caulfield, B.; Quinn, C.C. Digital assessment of falls risk, frailty, and mobility impairment using wearable sensors. NPJ Digit. Med. 2019, 2, 125. [Google Scholar] [CrossRef]
- Razjouyan, J.; Naik, A.; Horstman, M.; Kunik, M.; Amirmazaheri, M.; Zhou, H.; Sharafkhaneh, A.; Najafi, B. Wearable Sensors and the Assessment of Frailty among Vulnerable Older Adults: An Observational Cohort Study. Sensors 2018, 18, 1336. [Google Scholar] [CrossRef]
- Fontecha, J.; Navarro, F.J.; Hervás, R.; Bravo, J. Elderly frailty detection by using accelerometer-enabled smartphones and clinical information records. Pers. Ubiquitous Comput. 2013, 17, 1073–1083. [Google Scholar] [CrossRef]
- González, I.; Fontecha, J.; Hervás, R.; Bravo, J. Estimation of Temporal Gait Events from a Single Accelerometer Through the Scale-Space Filtering Idea. J. Med. Syst. 2016, 40, 251. [Google Scholar] [CrossRef]
- Zhu, L.; Speechley, M.S. Measuring Community Mobility in Older Adults with Parkinson’s Disease Using A Wearable GPS Sensor and Self-Report Assessment Tools; Graduate Program in Epidemiology and Biostatistics; University of Western Ontario: London, ON, Canada, 2017. [Google Scholar]
- DeMasi, O.; Feygin, S.; Dembo, A.; Aguilera, A.; Recht, B. Well-Being Tracking via Smartphone-Measured Activity and Sleep: Cohort Study. JMIR mHealth uHealth 2017, 5, e137. [Google Scholar] [CrossRef]
- Do, T.M.T.; Gatica-Perez, D. Human interaction discovery in smartphone proximity networks. Pers. Ubiquitous Comput. 2013, 17, 413–431. [Google Scholar] [CrossRef]
- García-Moreno, F.M.; Rodríguez-García, E.; Rodríguez-Fórtiz, M.J.; Garrido, J.L.; Bermúdez-Edo, M.; Villaverde-Gutiérrez, C.; Pérez-Mármol, J.M. Designing a Smart Mobile Health System for Ecological Frailty Assessment in Elderly. Proceedings 2019, 31, 41. [Google Scholar] [CrossRef]
- Esposito, M.; Minutolo, A.; Megna, R.; Forastiere, M.; Magliulo, M.; De Pietro, G. A smart mobile, self-configuring, context-aware architecture for personal health monitoring. Eng. Appl. Artif. Intell. 2018, 67, 136–156. [Google Scholar] [CrossRef]
- Schmidt, M.; Obermaisser, R. Adaptive and technology-independent architecture for fault-tolerant distributed AAL solutions. Comput. Biol. Med. 2018, 95, 236–247. [Google Scholar] [CrossRef]
- Harous, S.; El Menshawy, M.; Serhani, M.A.; Benharref, A. Mobile health architecture for obesity management using sensory and social data. Inform. Med. Unlocked 2018, 10, 27–44. [Google Scholar] [CrossRef]
- Qi, J.; Yang, P.; Min, G.; Amft, O.; Dong, F.; Xu, L. Advanced internet of things for personalised healthcare systems: A survey. Pervasive Mob. Comput. 2017, 41, 132–149. [Google Scholar] [CrossRef]
- Guerrero-Contreras, G.; Navarro-Galindo, J.L.; Samos, J.; Garrido, J.L. A Collaborative Semantic Annotation System in Health: Towards a SOA Design for Knowledge Sharing in Ambient Intelligence. Mob. Inf. Syst. 2017, 2017, 4759572. [Google Scholar] [CrossRef]
- Richards, M. Microservices vs. Service-Oriented Architecture; O’Reilly Media: Sevastopol, CA, USA, 2016; ISBN 978-1-491-95242-9. [Google Scholar]
- Lu, D.; Huang, D.; Walenstein, A.; Medhi, D. A Secure Microservice Framework for IoT. In Proceedings of the 2017 IEEE Symposium on Service-Oriented System Engineering (SOSE), San Francisco, CA, USA, 6−9 April 2017; pp. 9–18. [Google Scholar]
- Dragoni, N.; Giallorenzo, S.; Lafuente, A.L.; Mazzara, M.; Montesi, F.; Mustafin, R.; Safina, L. Microservices: Yesterday, Today, and Tomorrow BT—Present and Ulterior Software Engineering; Mazzara, M., Meyer, B., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 195–216. ISBN 978-3-319-67425-4. [Google Scholar]
- Thönes, J. Microservices. IEEE Softw. 2015, 32, 116. [Google Scholar] [CrossRef]
- Kolozali, S.; Kuemper, D.; Tonjes, R.; Bermudez-Edo, M.; Farajidavar, N.; Barnaghi, P.; Gao, F.; Intizar Ali, M.; Mileo, A.; Fischer, M.; et al. Observing the Pulse of a City: A Smart City Framework for Real-Time Discovery, Federation, and Aggregation of Data Streams. IEEE Internet Things J. 2019, 6, 2651–2668. [Google Scholar] [CrossRef]
- Guth, J.; Breitenbucher, U.; Falkenthal, M.; Leymann, F.; Reinfurt, L. Comparison of IoT platform architectures: A field study based on a reference architecture. In Proceedings of the 2016 Cloudification of the Internet of Things (CIoT), Paris, France, 23–25 November 2016; pp. 1–6. [Google Scholar]
- Carranza-García, F.; García-Moreno, F.M.; Rodriguez-Dominguez, C.; Garrido, J.L.; Bermúdez-Edo, M.; Rodriguez-Fortiz, M.J.; Pérez-Mármol, J.M. Supporting Active Ageing Interventions with Web and Mobile/Wearable Technologies and Using Microservice Oriented Architectures. In Proceedings of the First International Workshop, IWoG 2018, Cáceres, Spain, and Évora, Portugal, 14 and 17 December, 2018; García-Alonso, J., Fonseca, C., Eds.; Springer: Cáceres, Spain; Évora, Portugal, 2019; pp. 1–9. ISBN 978-3-030-16028-9. [Google Scholar]
- Srinivasa, K.G.; Sowmya, B.J.; Shikhar, A.; Utkarsha, R.; Singh, A. Data Analytics Assisted Internet of Things Towards Building Intelligent Healthcare Monitoring Systems. J. Organ. End User Comput. 2018, 30, 83–103. [Google Scholar] [CrossRef]
- Elsaleh, T.; Enshaeifar, S.; Rezvani, R.; Acton, S.T.; Janeiko, V.; Bermudez-Edo, M. IoT-Stream: A Lightweight Ontology for Internet of Things Data Streams and Its Use with Data Analytics and Event Detection Services. Sensors 2020, 20, 953. [Google Scholar] [CrossRef] [PubMed]
- Abril-Jiménez, P.; Javier, R.L.; De Los Ríos Pérez, S.; Páramo, M.; Colomer, J.B.M.; Arredondo Waldmeyer, M.T. Ageing-friendly cities for assessing older adults’ decline: IoT-based system for continuous monitoring of frailty risks using smart city infrastructure. Aging Clin. Exp. Res 2020, 32, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Esposito, C.; Castiglione, A.; Tudorica, C.-A.; Pop, F. Security and privacy for cloud-based data management in the health network service chain: A microservice approach. IEEE Commun. Mag. 2017, 55, 102–108. [Google Scholar] [CrossRef]
- Jita, H.; Pieterse, V. A Framework to Apply the Internet of Things for Medical Care in a Home Environment. In Proceedings of the 2018 International Conference on Cloud Computing and Internet of Things—CCIOT 2018, Singapore, 29–31 October 2018; ACM Press: New York, NY, USA, 2018; pp. 45–54. [Google Scholar]
- Jarwar, M.A.; Ali, S.; Chong, I. Exploring Web Objects enabled Data-Driven Microservices for E-Health Service Provision in IoT Environment. In Proceedings of the 2018 International Conference on Information and Communication Technology Convergence (ICTC), Jeju Island, Korea, 17–19 October 2018; pp. 112–117. [Google Scholar]
- Avci, A.; Bosch, S.; Marin-Perianu, M.; Marin-Perianu, R.; Havinga, P. Activity Recognition Using Inertial Sensing for Healthcare, Wellbeing and Sports Applications: A Survey. In Proceedings of the 23th International Conference on Architecture of Computing Systems 2010, Hannover, Germany, 22–23 February 2010; pp. 1–10. [Google Scholar]
- Mannini, A.; Intille, S.S. Classifier Personalization for Activity Recognition Using Wrist Accelerometers. IEEE J. Biomed. Heal. Inform. 2019, 23, 1585–1594. [Google Scholar] [CrossRef]
- Twomey, N.; Diethe, T.; Fafoutis, X.; Elsts, A.; McConville, R.; Flach, P.; Craddock, I. A Comprehensive Study of Activity Recognition Using Accelerometers. Informatics 2018, 5, 27. [Google Scholar] [CrossRef]
- Debes, C.; Merentitis, A.; Sukhanov, S.; Niessen, M.; Frangiadakis, N.; Bauer, A. Monitoring Activities of Daily Living in Smart Homes: Understanding human behavior. IEEE Signal Process. Mag. 2016, 33, 81–94. [Google Scholar] [CrossRef]
- Watanabe, Y.; Sara, S. Toward an Immunity-based Gait Recognition on Smart Phone: A Study of Feature Selection and Walking State Classification. Procedia Comput. Sci. 2016, 96, 1790–1800. [Google Scholar] [CrossRef][Green Version]
- Gjoreski, M.; Gjoreski, H.; Luštrek, M.; Gams, M. How Accurately Can Your Wrist Device Recognize Daily Activities and Detect Falls? Sensors 2016, 16, 800. [Google Scholar] [CrossRef]
- Attal, F.; Mohammed, S.; Dedabrishvili, M.; Chamroukhi, F.; Oukhellou, L.; Amirat, Y. Physical Human Activity Recognition Using Wearable Sensors. Sensors 2015, 15, 31314–31338. [Google Scholar] [CrossRef]
- Fida, B.; Bernabucci, I.; Bibbo, D.; Conforto, S.; Schmid, M. Varying behavior of different window sizes on the classification of static and dynamic physical activities from a single accelerometer. Med. Eng. Phys. 2015, 37, 705–711. [Google Scholar] [CrossRef]
- Garcia-Ceja, E.; Brena, R.; Carrasco-Jimenez, J.; Garrido, L. Long-Term Activity Recognition from Wristwatch Accelerometer Data. Sensors 2014, 14, 22500–22524. [Google Scholar] [CrossRef] [PubMed]
- Greene, B.R.; Doheny, E.P.; O’Halloran, A.; Anne Kenny, R. Frailty status can be accurately assessed using inertial sensors and the TUG test. Age Ageing 2014, 43, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Lobo, A.; Ezquerra, J.; Gómez Burgada, F.; Sala, J.M.; Seva Díaz, A. Cognocitive mini-test (a simple practical test to detect intellectual changes in medical patients). Actas Luso. Esp. Neurol. Psiquiatr. Cienc. Afines 1979, 7, 189–202. [Google Scholar] [PubMed]
- Patel, S.; Park, H.; Bonato, P.; Chan, L.; Rodgers, M. A review of wearable sensors and systems with application in rehabilitation. J. Neuroeng. Rehabil. 2012, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- Genovese, V.; Mannini, A.; Sabatini, A.M. A Smartwatch Step Counter for Slow and Intermittent Ambulation. IEEE Access 2017, 5, 13028–13037. [Google Scholar] [CrossRef]
- Schwenk, M.; Hauer, K.; Zieschang, T.; Englert, S.; Mohler, J.; Najafi, B. Sensor-Derived Physical Activity Parameters Can Predict Future Falls in People with Dementia. Gerontology 2014, 60, 483–492. [Google Scholar] [CrossRef]
- Hernandez, J.; Riobo, I.; Rozga, A.; Abowd, G.D.; Picard, R.W. Using electrodermal activity to recognize ease of engagement in children during social interactions. In Proceedings of the 2014 ACM International Joint Conference on Pervasive and Ubiquitous Computing—UbiComp’14 Adjunct, Seattle, WA, USA, 13–17 September 2014; ACM Press: New York, NY, USA, 2014; pp. 307–317. [Google Scholar]
- Tizen Device Sensors. Available online: https://developer.tizen.org/development/guides/.net-application/location-and-sensors/device-sensors (accessed on 3 July 2019).
- Newman, S. Building Microservices, 1st ed.; Loukides, M., MacDonald, B., Eds.; O’Reilly Media: Sebastopol, CA, USA, 2015; Volume 52, ISBN 978-1-491-95035-7. [Google Scholar]
- Nadareishvili, I.; Mitra, R.; McLarty, M.; Amundsen, M. Microservice Architecture: Aligning Principles, Practices, and Culture; O’Reilly Media, Inc.: Sebastopol, CA, USA, 2016; ISBN 9781491956328. [Google Scholar]
- Fielding, R.T. REST: Architectural Styles and the Design of Network-Based Software Architectures; University of California: Oakland, CA, USA, 2000. [Google Scholar]
- Shelby, Z.; Hartke, K.; Bormann, C. Constrained Application Protocol (CoAP); IETF Internet-Draft Draft-Ietf-Core-Coap. Available online: http://tools.ietf.org/id/draft-ietf-core-coap. (accessed on 26 May 2013).
- AMQP. Available online: https://www.amqp.org/ (accessed on 4 April 2019).
- MQTT.org MQ Telemetry Transport. Available online: http://mqtt.org/ (accessed on 4 April 2019).
- Eugster, P.T.; Felber, P.A.; Guerraoui, R.; Kermarrec, A.-M. The many faces of publish/subscribe. ACM Comput. Surv. 2003, 35, 114–131. [Google Scholar] [CrossRef]
- Amini, D.A.; Kannenberg, K.; Bodison, S.; Chang, P.F.; Colaianni, D.; Goodrich, B.; Mahaffey, L.; Painter, M.; Urban, M.; Handley-More, D.; et al. Occupational Therapy Practice Framework: Domain and Process (3rd Edition). Am. J. Occup. Ther. 2017, 68, S1. [Google Scholar]
- Cleland, I.; Kikhia, B.; Nugent, C.; Boytsov, A.; Hallberg, J.; Synnes, K.; McClean, S.; Finlay, D. Optimal Placement of Accelerometers for the Detection of Everyday Activities. Sensors 2013, 13, 9183–9200. [Google Scholar] [CrossRef]
- Ogliari, G.; Mahinrad, S.; Stott, D.J.; Jukema, J.W.; Mooijaart, S.P.; Macfarlane, P.W.; Clark, E.N.; Kearney, P.M.; Westendorp, R.G.J.; de Craen, A.J.M.; et al. Resting heart rate, heart rate variability and functional decline in old age. Can. Med. Assoc. J. 2015, 187, E442–E449. [Google Scholar] [CrossRef]
- Hamedinia, M.R.; Sardorodian, M.; Haghighi, A.H.; Vahdat, S. The Effects of Moderate Swimming Training on Blood Pressure Risk Factors in Hypertensive Postmenopausal Women. Iran. J. Heal. Phys. Act. 2010, 1, 24–28. [Google Scholar]
- Hassler, A.P.; Menasalvas, E.; García-García, F.J.; Rodríguez-Mañas, L.; Holzinger, A. Importance of medical data preprocessing in predictive modeling and risk factor discovery for the frailty syndrome. BMC Med. Inform. Decis. Mak. 2019, 19, 33. [Google Scholar] [CrossRef] [PubMed]
- Bermudez-Edo, M.; Barnaghi, P.; Moessner, K. Analysing real world data streams with spatio-temporal correlations: Entropy vs. Pearson correlation. Autom. Constr. 2018, 88, 87–100. [Google Scholar] [CrossRef]
- Godfrey, A. Wearables for independent living in older adults: Gait and falls. Maturitas 2017, 100, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Zak, I.; Klein, I.; Katz, R. A Feasibility Study of Machine Learning Based Coarse Alignment. Proceedings 2018, 4, 50. [Google Scholar] [CrossRef]
- Ravi, N.; Dandekar, N.; Mysore, P.; Littman, M.L. Activity Recognition from Accelerometer Data. In Proceedings of the 20th National Conference on Artificial Intelligence, AAAI Press, Pittsburgh, PA, USA, 9–13 July 2005; pp. 1541–1546. [Google Scholar]
- Preece, S.J.; Goulermas, J.Y.; Kenney, L.P.J.; Howard, D. A Comparison of Feature Extraction Methods for the Classification of Dynamic Activities from Accelerometer Data. IEEE Trans. Biomed. Eng. 2009, 56, 871–879. [Google Scholar] [CrossRef]
- Zhao, Z.; Morstatter, F.; Sharma, S.; Alelyani, S.; Liu, H. Advancing Feature Selection Research; ASU Feature Selection Repository: Tempe, AZ, USA, 2010; pp. 1–28. [Google Scholar]
- Zhang, M.; Sawchuk, A.A. USC-HAD: A daily activity dataset for ubiquitous activity recognition using wearable sensors. In Proceedings of the 2012 ACM Conference on Ubiquitous Computing—UbiComp’12, Pittsburgh, PA, USA, 5–8 September 2012; ACM Press: New York, NY, USA, 2012; pp. 1036–1043. [Google Scholar]
- Lee, H.; Joseph, B.; Enriquez, A.; Najafi, B. Toward Using a Smartwatch to Monitor Frailty in a Hospital Setting: Using a Single Wrist-Wearable Sensor to Assess Frailty in Bedbound Inpatients. Gerontology 2018, 64, 389–400. [Google Scholar] [CrossRef]
- Guyon, I.; Elisseeff, A. An Introduction to Variable and Feature Selection. J. Mach. Learn. Res. 2003, 3, 1157–1182. [Google Scholar]
- Patel, S.; Hughes, R.; Huggins, N.; Standaert, D.; Growdon, J.; Dy, J.; Bonato, P. Using wearable sensors to predict the severity of symptoms and motor complications in late stage Parkinson’s Disease. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 21–24 August 2008; pp. 3686–3689. [Google Scholar]
- Ehmen, H.; Haesner, M.; Steinke, I.; Dorn, M.; Gövercin, M.; Steinhagen-Thiessen, E. Comparison of four different mobile devices for measuring heart rate and ECG with respect to aspects of usability and acceptance by older people. Appl. Ergon. 2012, 43, 582–587. [Google Scholar] [CrossRef]
| Work | Aim | Eco | Data Sources | System | Frailty Status | Best ML |
|---|---|---|---|---|---|---|
| [14] | To assess frailty by a system based on Bluetooth RSSI fingerprints using beacons, collecting data derived from transitions among rooms | Yes. Transitions between rooms. | Smartphone Beacons (low-cost) | RSS | Three 2 and two 3 | RF 2: Accuracy: 82.33% Sensibilty: 83.83% RF 3: Accuracy: 97.92% Sensibilty: 94.2% |
| [15] | To discriminate between frailty status with gait, balance or during a physical activity. | No | LEGSys 1 ($10,000) BalanSens 1 ($4450) | None | Three 2 | MLR: AUC: 85.7% |
| [16] | To implement a wearable to characterize the quantity and quality of everyday walking, and to establish associations between gait impairment and frailty. | Yes. Walking ADL during 2 days | PAMSys 1 Demographic Clinical | None | Two 4 | MLR: Accuracy: 77.7% Sensibilty: 76.8% Specificity: 80% |
| [3] | To assess frailty by a wearable during the flexibility of upper-extremity movements. | No | Gyroscope 1 | None | Three 2 | OLR: Accuracy: 69% |
| [17,52] | To design a digital assessment protocol and algorithm for prediction of falls, frailty and mobility impairment. | No | Shimmer ($495) Demographic Clinical | None | Two 4 | LR: Accuracy: 72.8% Sensibilty: 72.99% |
| [18] | To remotely monitor the frailty status using an accelerometer. | Yes. Walking & Sleeping ADLs during 2 days | PAMSys 1 Demographic Clinical | None | Two 5 | EFS: Accuracy: 84.7% Sensibilty: 91.8% Specificity: 81.4% |
| Variable Description | Type |
|---|---|
| Accelerometer X-axis value | Float |
| Accelerometer Y-axis value | Float |
| Accelerometer Z-axis value | Float |
| Gyroscope X-axis value | Float |
| Gyroscope Y-axis value | Float |
| Gyroscope Z-axis value | Float |
| Heart Rate value | Integer |
| Algorithm | Features | Accuracy | F1-Score | Sensitivity | Specificity |
|---|---|---|---|---|---|
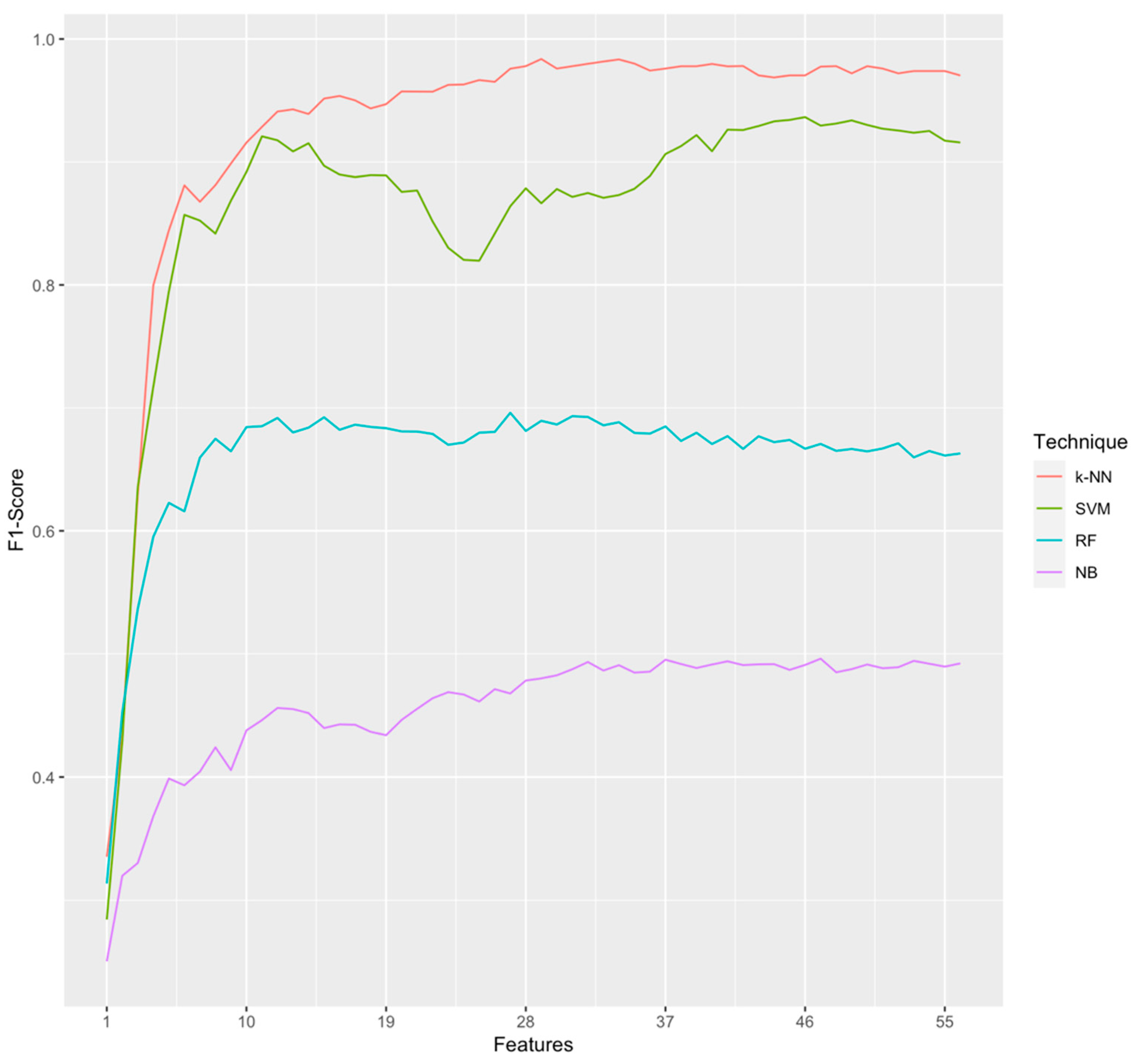
| k-NN 1 | 29 | 0.9917641 | 0.9837171 | 0.9764216 | 0.9947197 |
| SVM | 46 | 0.9670102 | 0.9364576 | 0.9108271 | 0.9779242 |
| RF | 27 | 0.8461648 | 0.6960141 | 0.6244533 | 0.8733734 |
| NB | 47 | 0.6621256 | 0.4960688 | 0.4353061 | 0.7659894 |
| Frailty Status | Sensitivity | Specificity |
|---|---|---|
| Frail | 0.9375 | 0.9946237 |
| Pre-frail | 0.9851852 | 0.9879518 |
| Non-frail | 0.962963 | 0.9939024 |
| Experiment (Phases) | Tasks or Sub-activities |
|---|---|
| Walking | (1) Walking to the supermarket (2) Coming back |
| Sitting/Standing | (1) Sitting (2) Standing (3) Standing at start point (4) Sitting back. |
| Shopping | (1) Participant is in the supermarket (2) Looking for the product to purchase (3) Picking the product (4) Going to the checkout (5) In the checkout (6) Paying (7) Go to the exit (8) In the outside |
| Packed Shopping | (1) Same phases as the shopping experiment but considered as a unique phase by computing the arithmetic mean of the values. |
| Algorithm | Features | Accuracy | F1-Score | Sensitivity | Specificity |
|---|---|---|---|---|---|
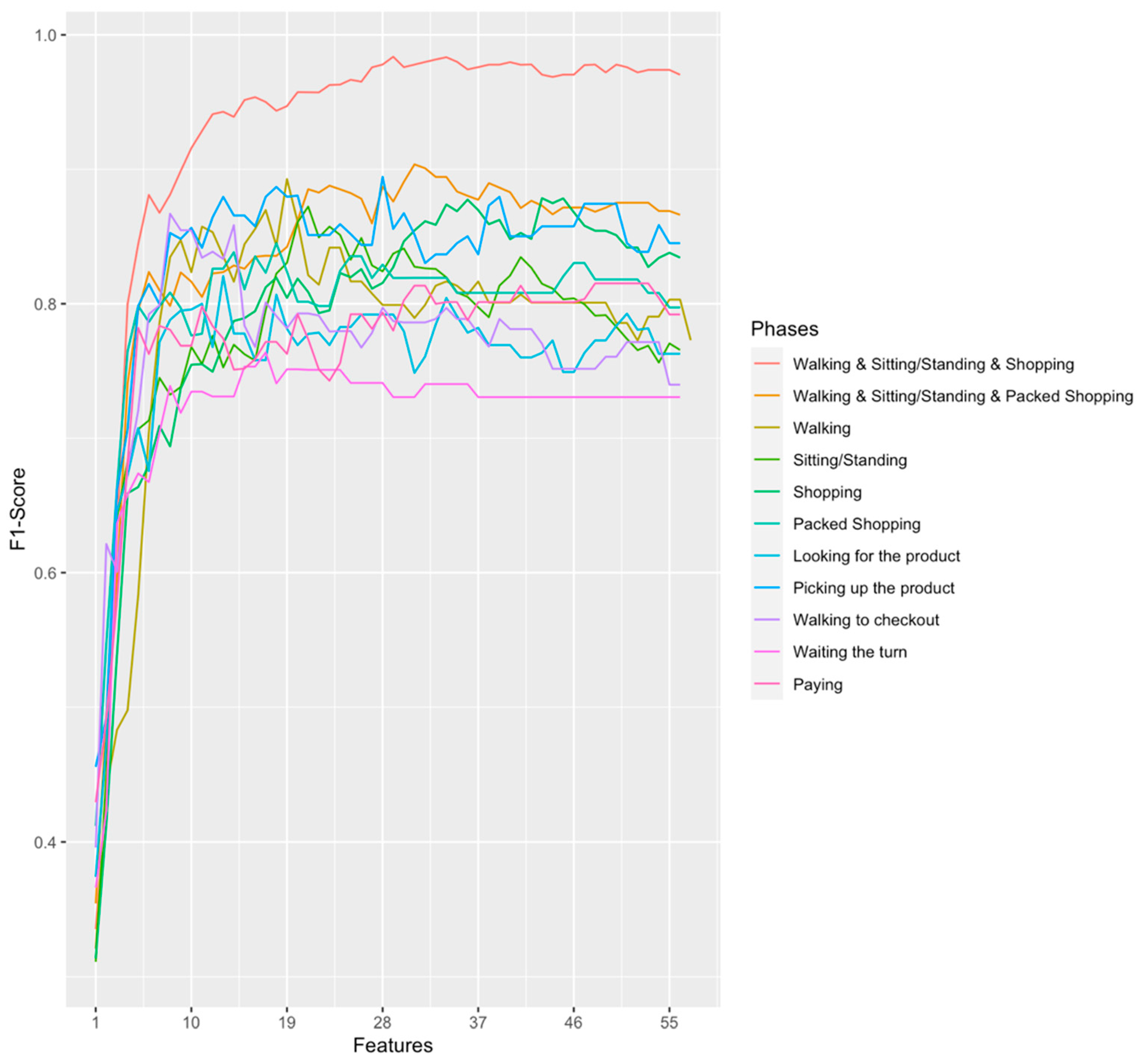
| Walking 1 & Sitting/Standing 2 & Shopping 3 | 29 | 0.9917641 | 0.9837171 | 0.9764216 | 0.9947197 |
| Walking 1 & Sitting/Standing 2 Packed Shopping 4 | 31 | 0.9503722 | 0.9036798 | 0.8792540 | 0.9705433 |
| Walking 1 | 19 | 0.9425269 | 0.8927655 | 0.8656145 | 0.9650742 |
| Sitting/Standing 2 | 21 | 0.9325653 | 0.8722884 | 0.8430592 | 0.9594234 |
| Shopping 3 | 42 | 0.9359852 | 0.8785180 | 0.8397436 | 0.9588485 |
| Packed Shopping 4 | 18 | 0.9091168 | 0.8450966 | 0.8034157 | 0.9488836 |
| Looking for the product | 28 | 0.9322537 | 0.8944003 | 0.8754377 | 0.9671364 |
| Picking up the product | 15 | 0.8940171 | 0.8218771 | 0.7855828 | 0.9441125 |
| Walking to checkout | 8 | 0.9245014 | 0.8669296 | 0.8295743 | 0.9552347 |
| Waiting for their turn | 17 | 0.851007 | 0.7638345 | 0.7256926 | 0.9190001 |
| Paying | 48 | 0.8863248 | 0.8151564 | 0.7741049 | 0.9367538 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Moreno, F.M.; Bermudez-Edo, M.; Garrido, J.L.; Rodríguez-García, E.; Pérez-Mármol, J.M.; Rodríguez-Fórtiz, M.J. A Microservices e-Health System for Ecological Frailty Assessment Using Wearables. Sensors 2020, 20, 3427. https://doi.org/10.3390/s20123427
Garcia-Moreno FM, Bermudez-Edo M, Garrido JL, Rodríguez-García E, Pérez-Mármol JM, Rodríguez-Fórtiz MJ. A Microservices e-Health System for Ecological Frailty Assessment Using Wearables. Sensors. 2020; 20(12):3427. https://doi.org/10.3390/s20123427
Chicago/Turabian StyleGarcia-Moreno, Francisco M., Maria Bermudez-Edo, José Luis Garrido, Estefanía Rodríguez-García, José Manuel Pérez-Mármol, and María José Rodríguez-Fórtiz. 2020. "A Microservices e-Health System for Ecological Frailty Assessment Using Wearables" Sensors 20, no. 12: 3427. https://doi.org/10.3390/s20123427
APA StyleGarcia-Moreno, F. M., Bermudez-Edo, M., Garrido, J. L., Rodríguez-García, E., Pérez-Mármol, J. M., & Rodríguez-Fórtiz, M. J. (2020). A Microservices e-Health System for Ecological Frailty Assessment Using Wearables. Sensors, 20(12), 3427. https://doi.org/10.3390/s20123427





