Smartphone Sensors for Health Monitoring and Diagnosis
Abstract
1. Introduction
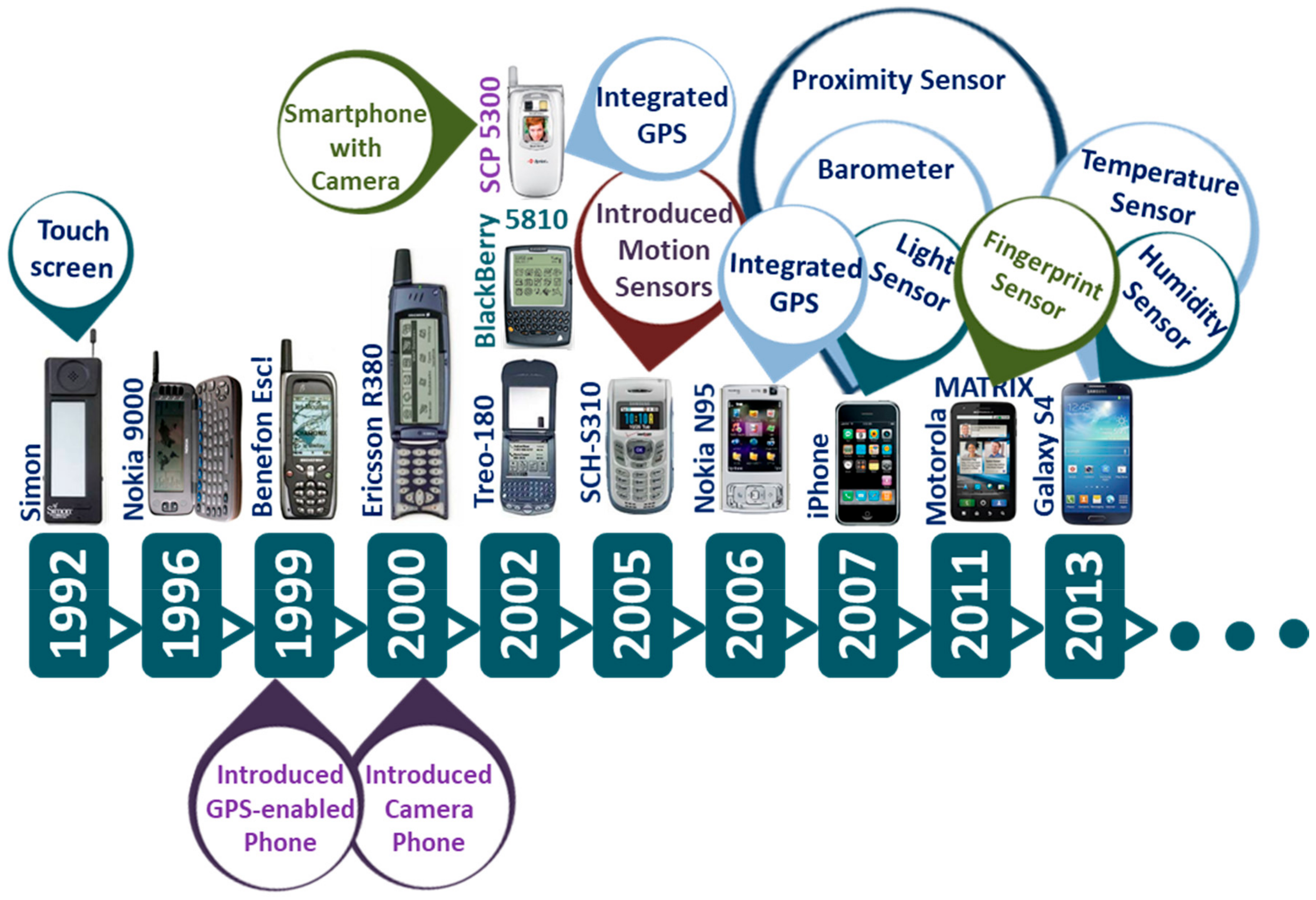
2. Evolution of Smartphone
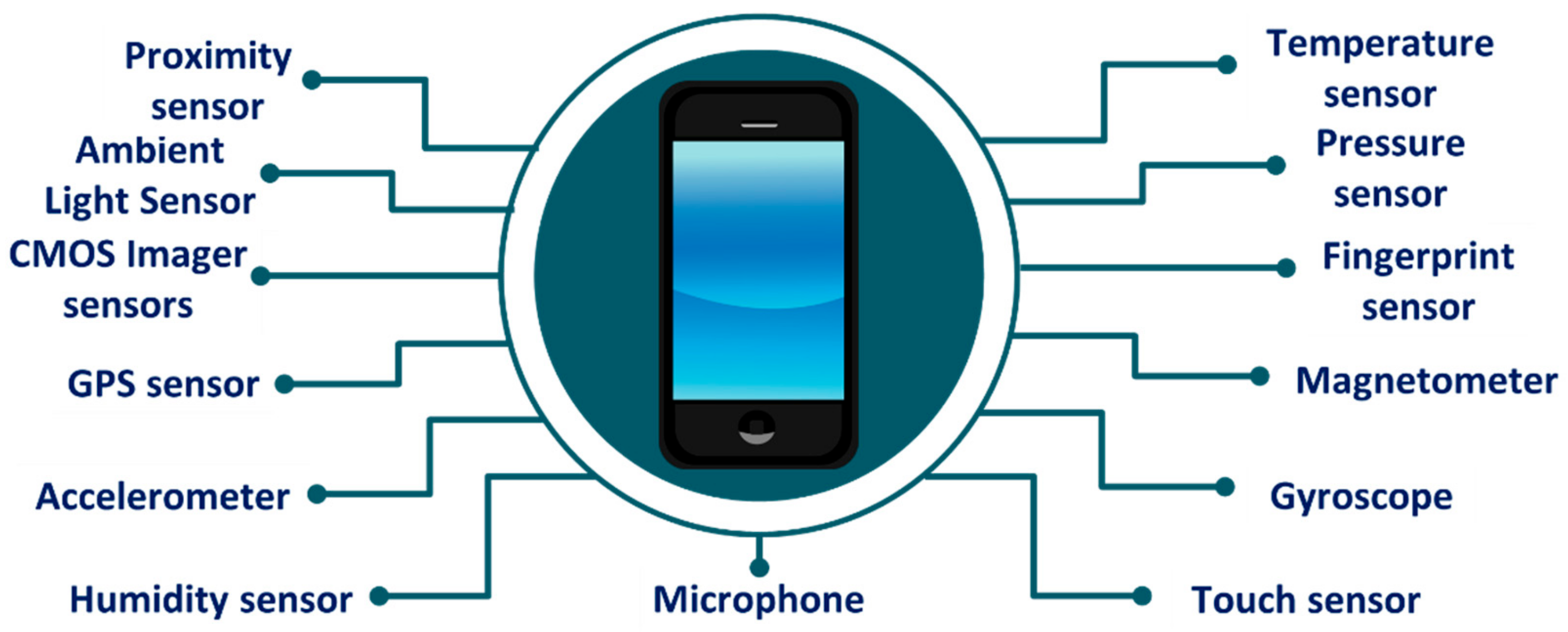
3. Smartphone Sensors for Health Monitoring
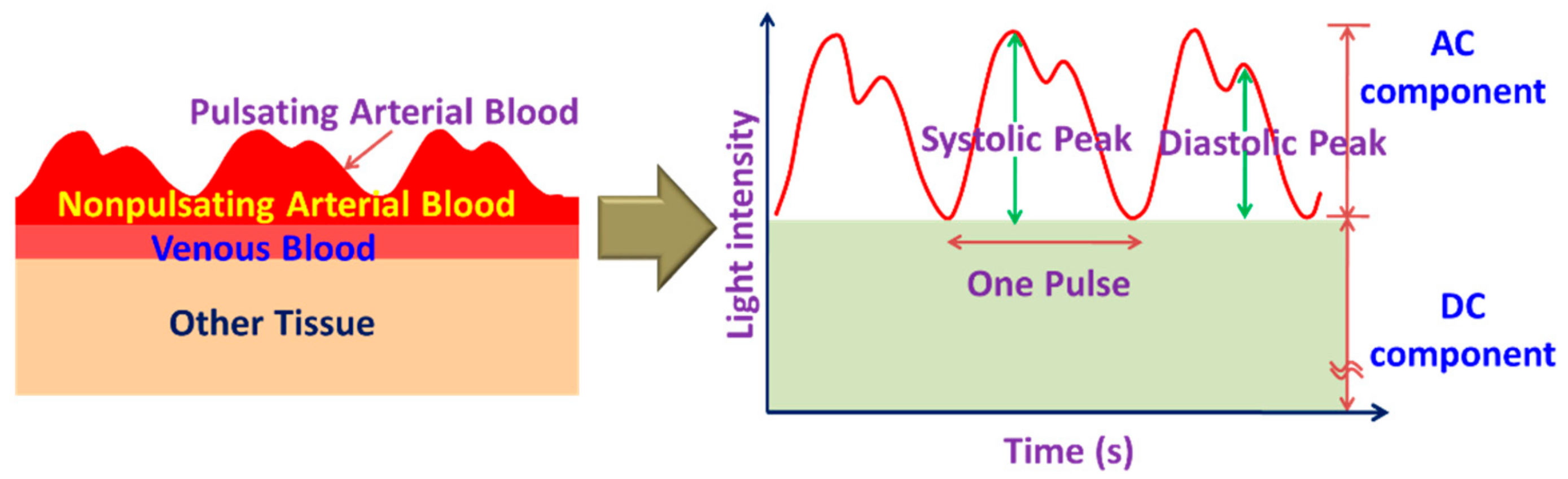
3.1. Cardiovascular Health Monitoring
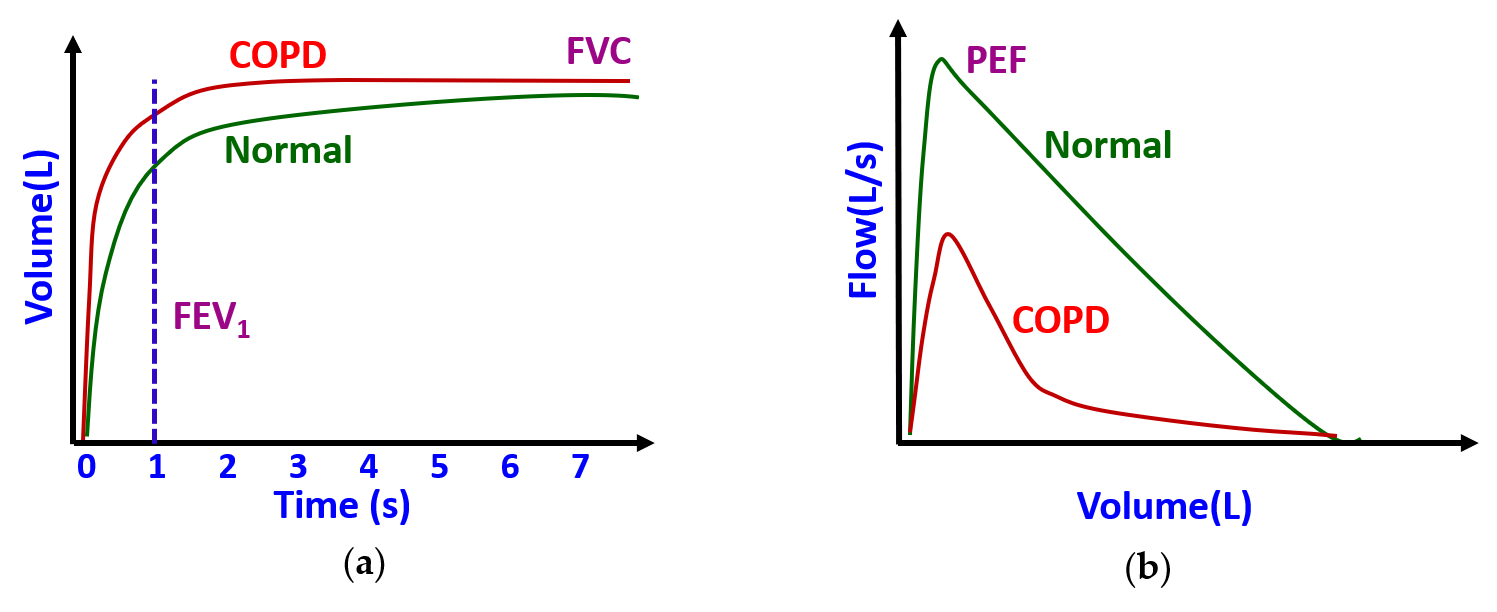
3.2. Pulmonary Health Monitoring
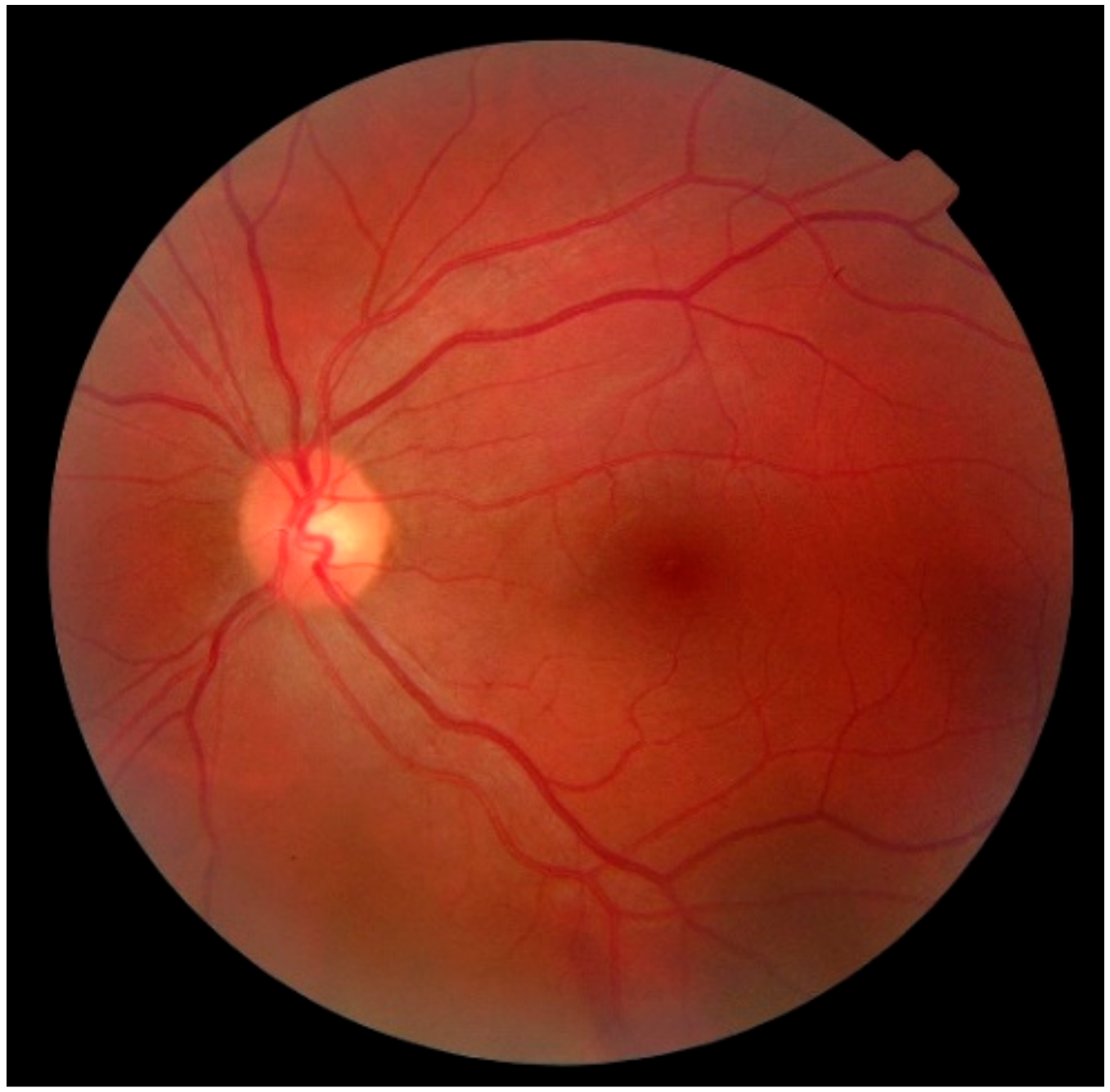
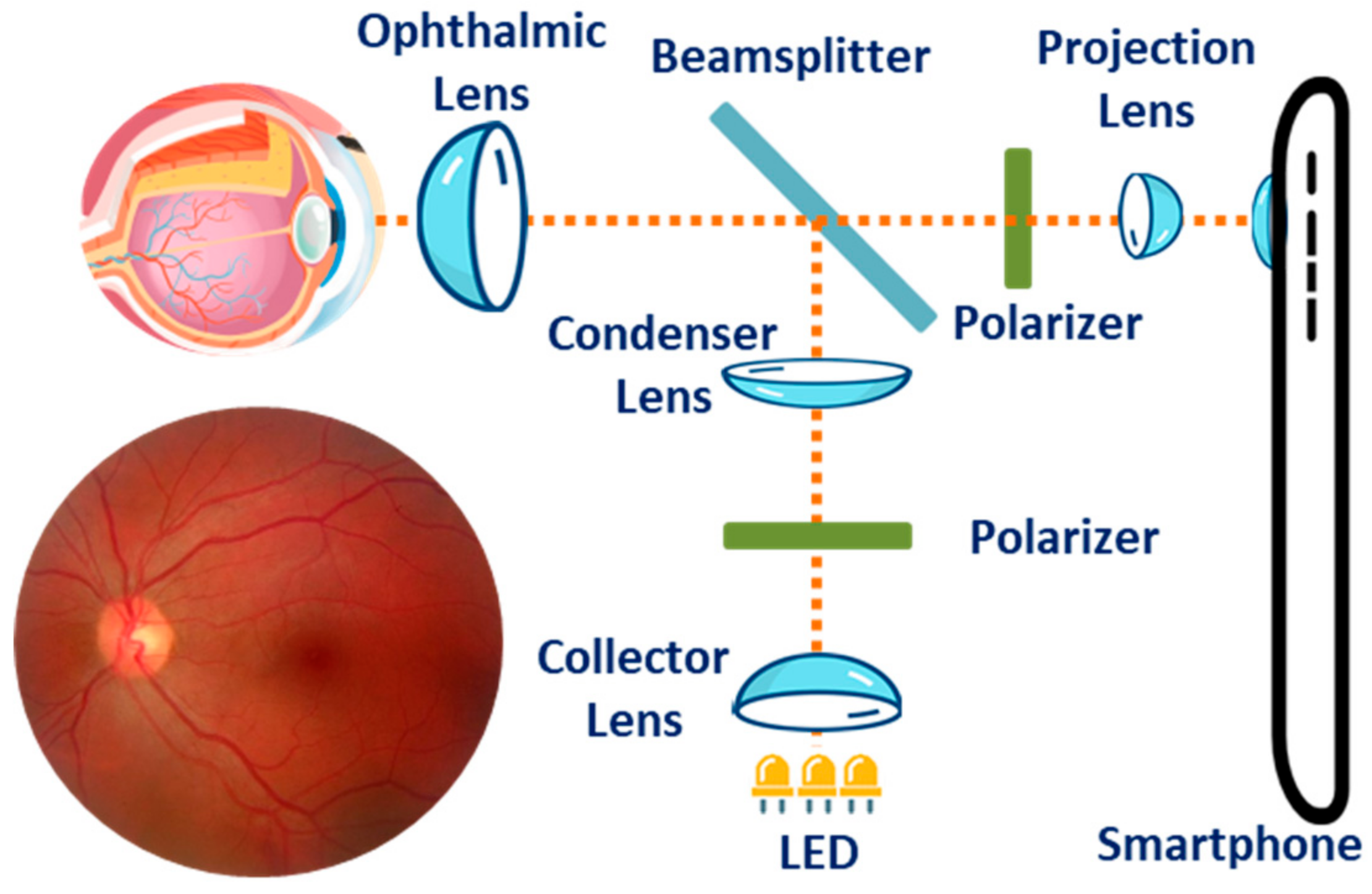
3.3. Ophthalmic Health Monitoring
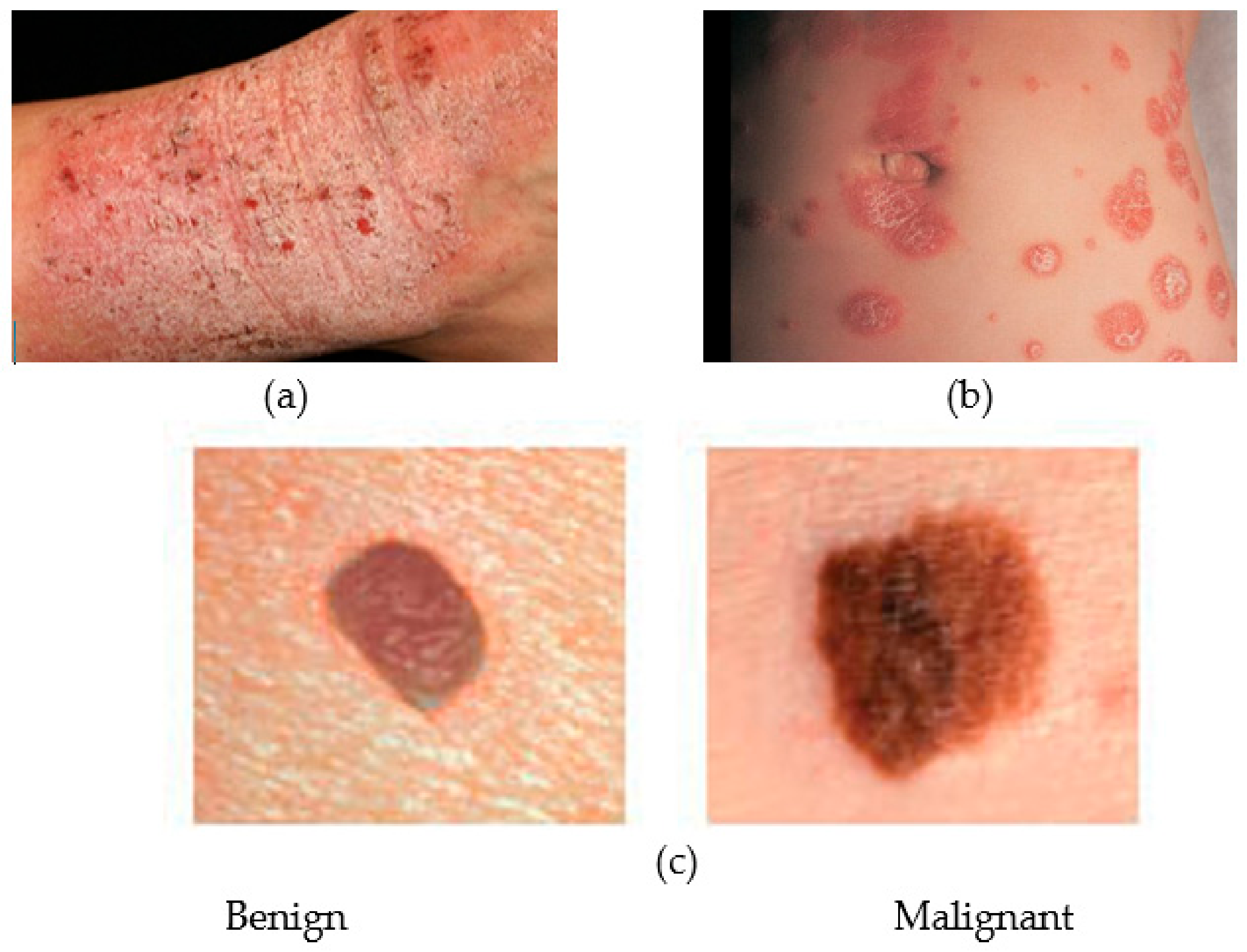
3.4. Skin Health Monitoring
3.5. Mental Health Assessment
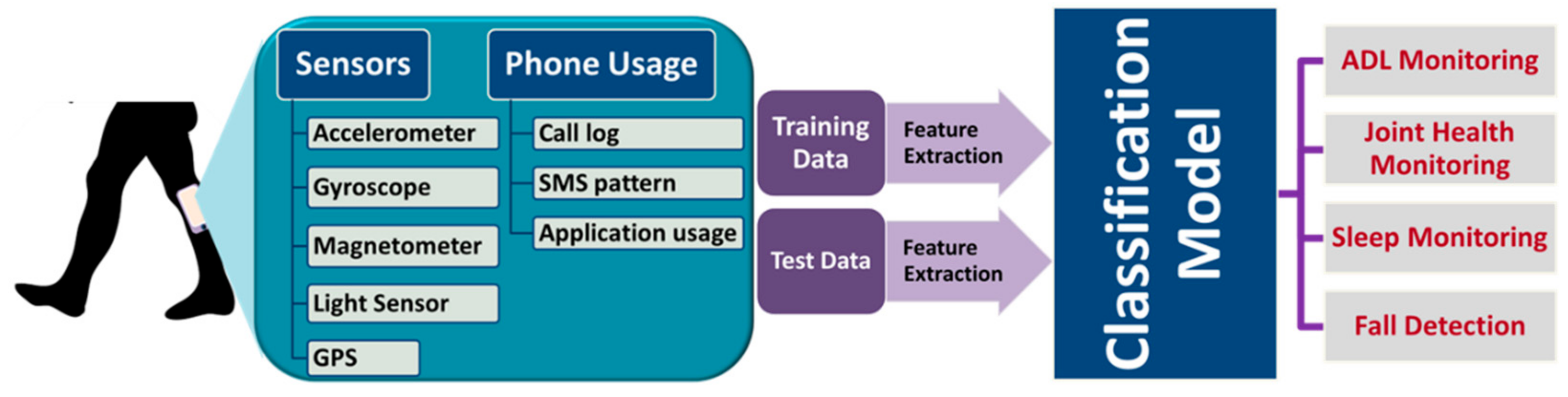
3.6. Activity and Sleep Monitoring Systems
3.7. Hearing Impairment Monitoring Systems
4. Regulatory Policies
5. Conclusions and Research Challenges
Author Contributions
Funding
Conflicts of Interest
References
- Thomas, V.S.; Darvesh, S.; Macknight, C.; Rockwood, K. Estimating the Prevalence of Dementia in Elderly People: A Comparison of the Canadian Study of Health and Aging and National Population Health Survey Approaches. Int. Psychogeriatr. 2001, 13, 169–175. [Google Scholar] [CrossRef]
- Kalache, A.; Gatti, A. Active Ageing: A Policy Framework. Adv. Gerontol. Uspekhi Gerontol. Akad. Nauk. Gerontol. Obs. 2002, 11, 7–18. [Google Scholar]
- World Health Organization. Are You Ready? What You Need to Know About Ageing. 27 March 2012. Available online: http://www.who.int/world-health-day/2012/toolkit/background/en/ (accessed on 3 January 2019).
- Kulik, C.T.; Ryan, S.; Harper, S.; George, G. Aging populations and management. Acad. Manag. J. 2014, 57, 929–935. [Google Scholar] [CrossRef]
- World Health Organization. Disability and Health. Available online: https://www.who.int/en/news-room/fact-sheets/detail/disability-and-health (accessed on 3 January 2019).
- Centers for Disease Control and Prevention. About Chronic Diseases. 19 November 2018. Available online: https://www.cdc.gov/chronicdisease/about/index.htm (accessed on 3 January 2019).
- Majumder, S.; Aghayi, E.; Noferesti, M.; Memarzadeh-Tehran, H.; Mondal, T.; Pang, Z.; Deen, M.J. Smart Homes for Elderly Healthcare—Recent Advances and Research Challenges. Sensors 2017, 17, 2496. [Google Scholar] [CrossRef] [PubMed]
- “National Center for Health Statistics,” Centers for Disease Control and Prevention. 3 May 2017. Available online: https://www.cdc.gov/nchs/fastats/deaths.htm (accessed on 3 January 2019).
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef]
- World Health Organization. Priority Eye Diseases. 18 April 2018. Available online: https://www.who.int/blindness/causes/priority/en/index5.html (accessed on 3 January 2019).
- World Health Organization. Global Data on Visual Impairments 2010. Available online: https://www.who.int/blindness/GLOBALDATAFINALforweb.pdf (accessed on 3 January 2019).
- World Health Organization. Asthma. Available online: http://www.who.int/mediacentre/factsheets/fs307/en/ (accessed on 3 January 2019).
- 2017 Budget in Brief: Strengthening Health Care. ontario.ca. Available online: https://www.ontario.ca/page/2017-budget-brief-strengthening-health-care (accessed on 3 January 2019).
- CIHI. National Health Expenditure Trends, 1975 to 2018. 20 December 2018. Available online: https://www.cihi.ca/en/health-spending/2018/national-health-expenditure-trends (accessed on 12 March 2019).
- Venkat, R. Global Outlook of the Healthcare Industry. In Advances in Information and Communication; Frost & Sullivan: San Antonio, TX, USA, 2015. [Google Scholar]
- Population Ageing Projections. Helping Older People Live Full and Secure Lives. Available online: http://www.helpage.org/global-agewatch/populationageing-data/population-ageing-projections/ (accessed on 3 January 2019).
- The Globe and Mail. Canada Ranks Fifth in Well-Being of Elderly: Study. 11 May 2018. Available online: https://www.theglobeandmail.com/life/health-and-fitness/health/canada-ranks-fifth-in-well-being-of-elders-study/article14621721/ (accessed on 3 January 2019).
- Canadian Institute for Health Information. National Health Expenditure Trends, 1975 to 2014. Available online: https://www.cihi.ca/en/nhex_2014_report_en.pdf (accessed on 3 January 2019).
- Working-Age Shift. The Economist. 26 January 2013. Available online: https://www.economist.com/finance-and-economics/2013/01/26/working-age-shift (accessed on 3 January 2019).
- Anderson, G.; Knickman, J.R. Changing the Chronic Care System To Meet People’s Needs. Heal. Aff. 2001, 20, 146–160. [Google Scholar] [CrossRef]
- Advantages & Disadvantages of Nursing Homes. AmeriGlide Stair lifts and Vertical Platform lifts. Available online: http://www.ameriglide.com/advantages-disadvantages-nursing-homes.htm (accessed on 3 January 2019).
- Majumder, S.; Mondal, T.; Deen, M.J. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Deen, M.J. Information and communications technologies for elderly ubiquitous healthcare in a smart home. Pers. Ubiquitous Comput. 2015, 19, 573–599. [Google Scholar] [CrossRef]
- Agoulmine, N.; Deen, M.J.; Lee, J.S.; Meyyappan, M. U-Health Smart Home: Innovative solutions for the management of the elderly and chronic diseases. IEEE Nanotechnol. Mag. 2011, 5, 6–11. [Google Scholar] [CrossRef]
- National Center for Chronic Disease Prevention and Health Promotion | CDC. Centers for Disease Control and Prevention. 19 December 2018. Available online: https://www.cdc.gov/chronicdisease/ (accessed on 3 January 2019).
- Takei, K.; Honda, W.; Harada, S.; Arie, T.; Akita, S. Toward flexible and wearable human-interactive health-monitoring devices. Adv. Healthc. Mater. 2015, 4, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Pantelopoulos, A.; Bourbakis, N. A Survey on Wearable Sensor-Based Systems for Health Monitoring and Prognosis. IEEE Trans. Syst. Man Cybern. Part C Appl. Rev. 2010, 40, 1–12. [Google Scholar] [CrossRef]
- Kooistra, J. Newzoo’s 2018 Global Mobile Market Report: Insights into the World’s 3 Billion Smartphone Users. Newzoo. 11 September 2018. Available online: https://newzoo.com/insights/articles/newzoos-2018-global-mobile-market-report-insights-into-the-worlds-3-billion-smartphone-users/ (accessed on 3 January 2019).
- Tweedie, S. The World’s First Smartphone, Simon, Was Created 15 Years Before the iPhone. Business Insider. 14 June 2015. Available online: https://www.businessinsider.com/worlds-first-smartphone-simon-launched-before-iphone-2015-6 (accessed on 4 January 2019).
- The Evolution of the Mobile Phone, from the Motorola DynaTAC to the Samsung Galaxy S9. The Telegraph. 23 February 2018. Available online: https://www.telegraph.co.uk/technology/0/evolution-mobile-phone-pictures/ (accessed on 4 January 2019).
- Woyke, E. The Smartphone: Anatomy of an Industry. The New Press: New York, NY, USA, 2015. [Google Scholar]
- Sager, I. Before IPhone and Android Came Simon, the First Smartphone. Bloomberg.com. 29 June 2012. Available online: https://www.bloomberg.com/news/articles/2012-06-29/before-iphone-and-android-came-simon-the-first-smartphone (accessed on 4 January 2019).
- Vaishnav, S.; Stevenson, R.; Marchant, B.; Lagi, K.; Ranjadayalan, K.; Timmis, A.D. Relation between heart rate variability early after acute myocardial infarction and long-term mortality. Am. J. Cardiol. 1994, 73, 653–657. [Google Scholar] [CrossRef]
- Nemati, E.; Deen, M.; Mondal, T. A wireless wearable ECG sensor for long-term applications. IEEE Commun. Mag. 2012, 50, 36–43. [Google Scholar] [CrossRef]
- Rolfe, P. In Vivo Near-Infrared Spectroscopy. Annu. Rev. Biomed. Eng. 2000, 2, 715–754. [Google Scholar] [CrossRef]
- Siddiqui, S.A.; Zhang, Y.; Feng, Z.; Kos, A. A Pulse Rate Estimation Algorithm Using PPG and Smartphone Camera. J. Med Syst. 2016, 40, 126. [Google Scholar] [CrossRef]
- Bánhalmi, A.; Borbás, J.; Fidrich, M.; Bilicki, V.; Gingl, Z.; Rudas, L. Analysis of a Pulse Rate Variability Measurement Using a Smartphone Camera. J. Healthc. Eng. 2018, 2018, 4038034. [Google Scholar] [CrossRef]
- Nam, Y.; Kong, Y.; Reyes, B.; Reljin, N.; Chon, K.H. Monitoring of Heart and Breathing Rates Using Dual Cameras on a Smartphone. PLoS One 2016, 11, 0151013. [Google Scholar] [CrossRef]
- Koenig, N.; Seeck, A.; Eckstein, J.; Mainka, A.; Huebner, T.; Voss, A.; Weber, S. Validation of a New Heart Rate Measurement Algorithm for Fingertip Recording of Video Signals with Smartphones. Telemed. e-Health 2016, 22, 631–636. [Google Scholar] [CrossRef]
- Matsumura, K.; Rolfe, P.; Lee, J.; Yamakoshi, T. iPhone 4s Photoplethysmography: Which Light Color Yields the Most Accurate Heart Rate and Normalized Pulse Volume Using the iPhysioMeter Application in the Presence of Motion Artifact? PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Lavanya, M.P. Real Time Motion Detection Using Background Subtraction Method and Frame Difference. Int. J. Sci. Res. 2014, 3, 1857–1861. [Google Scholar]
- Singla, N. Motion Detection Based on Frame Difference Method. Int. J. Inf. Comput. Technol. 2014, 4, 1559–1565. [Google Scholar]
- Warren, S.; Krishnan, R.; Natarajan, B. Two-Stage Approach for Detection and Reduction of Motion Artifacts in Photoplethysmographic Data. IEEE Trans. Biomed. Eng. 2010, 57, 1867–1876. [Google Scholar]
- Lamonaca, F.; Kurylyak, Y.; Grimaldi, D.; Spagnuolo, V. Reliable pulse rate evaluation by smartphone. In Proceedings of the 2012 IEEE International Symposium on Medical Measurements and Applications, Budapest, Hungary, 18–19 May 2012. [Google Scholar]
- Kwon, S.; Kim, H.; Park, K.S. Validation of heart rate extraction using video imaging on a built-in camera system of a smartphone. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012. [Google Scholar]
- Poh, M.-Z.; McDuff, D.J.; Picard, R.W. Non-contact, automated cardiac pulse measurements using video imaging and blind source separation. Opt. Express 2010, 18, 10762. [Google Scholar] [CrossRef]
- Verkruysse, W.; Svaasand, L.O.; Nelson, J.S. Remote plethysmographic imaging using ambient light. Opt. Express 2008, 16, 21434. [Google Scholar] [CrossRef]
- Papin, C.; Greenwald, S.; Hu, S.; Sun, Y.; Azorin-Peris, V.; Kalawsky, R. Use of ambient light in remote photoplethysmographic systems: Comparison between a high-performance camera and a low-cost webcam. J. Biomed. Opt. 2012, 17, 37005. [Google Scholar]
- Sanyal, S.; Nundy, K.K. Algorithms for Monitoring Heart Rate and Respiratory Rate from the Video of a User’s Face. IEEE J. Transl. Eng. Heal. Med. 2018, 6, 1–11. [Google Scholar] [CrossRef]
- Guede-Fernandez, F.; Ferrer-Mileo, V.; Ramos-Castro, J.; Fernandez-Chimeno, M.; Garcia-Gonzalez, M. Real time heart rate variability assessment from Android smartphone camera photoplethysmography: Postural and device influences. In Proceedings of the 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Milan, Italy, 25–29 August 2015; pp. 7332–7335. [Google Scholar]
- Lagido, R.; Lobo, J.; Leite, S.; Sousa, C.; Ferreira, L.; Silva-Cardoso, J. Using the smartphone camera to monitor heart rate and rhythm in heart failure patients. In Proceedings of the IEEE-EMBS International Conference on Biomedical and Health Informatics (BHI), Valencia, Spain, 1–4 June 2014; pp. 556–559. [Google Scholar]
- Bolkhovsky, J.B.; Scully, C.G.; Chon, K.H. Statistical analysis of heart rate and heart rate variability monitoring through the use of smart phone cameras. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 1610–1613. [Google Scholar]
- Gregoski, M.J.; Mueller, M.; Vertegel, A.; Shaporev, A.; Jackson, B.B.; Frenzel, R.M.; Sprehn, S.M.; Treiber, F.A. Development and Validation of a Smartphone Heart Rate Acquisition Application for Health Promotion and Wellness Telehealth Applications. Int. J. Telemed. Appl. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vidal, J. Air Pollution Rising at an ‘Alarming Rate’ in World’s Cities. The Guardian. 12 May 2016. Available online: https://www.theguardian.com/environment/2016/may/12/air-pollution-rising-at-an-alarming-rate-in-worlds-cities (accessed on 21 March 2019).
- World Health Organization. Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 21 March 2019).
- Stafford, M.; Lin, F.; Xu, W. Flappy Breath: A Smartphone-Based Breath Exergame. In Proceedings of the 2016 IEEE First International Conference on Connected Health: Applications, Systems and Engineering Technologies (CHASE), Washington, DC, USA, 27–29 June 2016; pp. 332–333. [Google Scholar]
- Larson, E.C.; Lee, T.; Liu, S.; Rosenfeld, M.; Patel, S.N. Accurate and privacy preserving cough sensing using a low-cost microphone. In Proceedings of the 13th International Conference on Ubiquitous Computing, Beijing, China, 17–21 September 2011; p. 375. [Google Scholar]
- Chen, N.-C.; Wang, K.-C.; Chu, H.-H. Listen-to-nose: A low-cost system to record nasal symptoms in daily life. In Proceedings of the 2012 ACM Conference on Ubiquitous Computing, Pittsburgh, PA, USA, 5–8 September 2012; pp. 590–591. [Google Scholar]
- Larson, E.C.; Goel, M.; Boriello, G.; Heltshe, S.; Rosenfeld, M.; Patel, S.N. SpiroSmart: Using a microphone to measure lung function on a mobile phone. In Proceedings of the 2012 ACM Conference Ubiquitous Comput.-UbiComp ’12, Pittsburgh, PA, USA, 5–8 September 2012; pp. 280–289. [Google Scholar]
- Larson, E.C.; Goel, M.; Redfield, M.; Boriello, G.; Rosenfeld, M.; Patel, S.N. Tracking lung function on any phone. In Proceedings of the 3rd ACM Symposium on Computing for Development, Bangalore, India, 11–12 January 2013; p. 29. [Google Scholar]
- Goel, M.; Saba, E.; Stiber, M.; Whitemire, E.; Fromm, J.; Larson, E.C.; Borriello, G.; Patel, S.N. SpiroCall: Measuring Lung Function over a Phone Call. In Proceedings of the 2016 CHI Conference on Human Factors in Computing Systems, San Jose, CA, USA, 7–12 May 2016; pp. 5675–5685. [Google Scholar]
- Thap, T.; Chung, H.; Jeong, C.; Hwang, K.-E.; Kim, H.-R.; Yoon, K.-H.; Lee, J.; Chon, K.H. High-Resolution Time-Frequency Spectrum-Based Lung Function Test from a Smartphone Microphone. Sensors 2016, 16, 1305. [Google Scholar] [CrossRef]
- Szanto, Z.; Benko, I.; Jakab, L.; Szalai, G.; Vereczkei, A. The use of a smartphone application for fast lung cancer risk assessment†. Eur. J. Cardio-Thoracic Surg. 2017, 51, 1171–1176. [Google Scholar] [CrossRef]
- Szanto, Z.; Szalai, G.; Jakab, L.; Vereczkei, A. P1.03-034 Implementing Smartphone Application in Early Lung Cancer Detection and Screening. J. Thorac. Oncol. 2017, 12, S562–S563. [Google Scholar] [CrossRef][Green Version]
- Early Treatment Diabetic Retinopathy Study Research Group. Grading Diabetic Retinopathy from Stereoscopic Color Fundus Photographs—An Extension of the Modified Airlie House Classification: ETDRS Report Number 10. Ophthalmology 1991, 98, 786–806. [Google Scholar] [CrossRef]
- Lawrence, M.G. The accuracy of digital-video retinal imaging to screen for diabetic retinopathy: an analysis of two digital-video retinal imaging systems using standard stereoscopic seven-field photography and dilated clinical examination as reference standards. Trans. Am. Ophthalmol. Soc. 2004, 102, 321–340. [Google Scholar]
- Williams, G.A.; Scott, I.U.; Haller, J.A.; Maguire, A.M.; Marcus, D.; McDonald, H.R. Single-field fundus photography for diabetic retinopathy screening: A report by the American Academy of Ophthalmology. Ophthalmology 2004, 111, 1055–1062. [Google Scholar] [CrossRef]
- Toy, B.C.; Myung, D.J.; He, L.; Pan, C.K.; Chang, R.T.; Polkinhorne, A.; Merrell, D.; Foster, D.; Blumenkranz, M.S. Smartphone-Based Dilated Fundus Photography and Near Visual Acuity Testing as Inexpensive Screening Tools to Detect Referral Warranted Diabetic Eye Disease. Retina 2016, 36, 1000–1008. [Google Scholar] [CrossRef]
- Lord, R.K.; Shah, V.A.; Filippo, A.N.S.; Krishna, R. Novel Uses of Smartphones in Ophthalmology. Ophthalmology 2010, 117, 1274–1274.e3. [Google Scholar] [CrossRef]
- Strauss, R.W.; Krieglstein, T.R.; Priglinger, S.G.; Reiß, W.; Ulbig, M.W.; Kampik, A.; Neubauer, A.S. Image quality characteristics of a novel colour scanning digital ophthalmoscope (SDO) compared with fundus photography. Ophthalmic Physiol. Opt. 2007, 27, 611–618. [Google Scholar] [CrossRef]
- Bastawrous, A. Smartphone fundoscopy. Ophthalmology 2012, 119, 432–433. [Google Scholar] [CrossRef] [PubMed]
- Haddock, L.J.; Kim, D.Y.; Mukai, S. Simple, Inexpensive Technique for High-Quality Smartphone Fundus Photography in Human and Animal Eyes. J. Ophthalmol. 2013, 2013, 1–5. [Google Scholar] [CrossRef]
- Myung, D.; Jais, A.; He, L.; Blumenkranz, M.S.; Chang, R.T. 3D Printed Smartphone Indirect Lens Adapter for Rapid, High Quality Retinal Imaging. J. Mob. Technol. Med. 2014, 3, 9–15. [Google Scholar] [CrossRef]
- Sharma, A.; Subramaniam, S.D.; Ramachandran, K.I.; Lakshmikanthan, C.; Krishna, S.; Sundaramoorthy, S.K. Smartphone-based fundus camera device (MII Ret Cam) and technique with ability to image peripheral retina. Eur. J. Ophthalmol. 2015, 26, 142–144. [Google Scholar] [CrossRef]
- Maamari, R.N.; Keenan, J.D.; Fletcher, D.A.; Margolis, T.P. A mobile phone-based retinal camera for portable wide field imaging. Br. J. Ophthalmol. 2014, 98, 438–441. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Morescalchi, F.; Costagliola, C.; Delcassi, L.; Semeraro, F. A Novel Device to Exploit the Smartphone Camera for Fundus Photography. J. Ophthalmol. 2015, 2015, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Morescalchi, F.; Costagliola, C.; Delcassi, L.; Semeraro, F. Comparison of Smartphone Ophthalmoscopy with Slit-Lamp Biomicroscopy for Grading Diabetic Retinopathy. Am. J. Ophthalmol. 2015, 159, 360–364.e1. [Google Scholar] [CrossRef] [PubMed]
- Giardini, M.; Livingstone, I.; Bolster, N.; Jordan, S.; Bastawrous, A. Phone-based ophthalmoscopy for Peek, the Portable Eye Examination Kit. Eng. Med. Biol. Soc. (EMBC) 2014, 96, 2014. [Google Scholar]
- Bastawrous, A. Increasing access to eye care … there’s an app for that. Peek: Smartphone technology for eye health. Int. J. Epidemiol. 2016, 45, 1040–1043. [Google Scholar] [CrossRef]
- Eysenbach, G.; Sawesi, S.; Jones, J.; Sheets, L.; Lodhia, V.; Karanja, S.; Lees, S.; Bastawrous, A. Acceptability, Usability, and Views on Deployment of Peek, a Mobile Phone mHealth Intervention for Eye Care in Kenya: Qualitative Study. JMIR mHealth uHealth 2016, 4, e30. [Google Scholar]
- Rono, H.K.; Bastawrous, A.; MacLeod, D.; Wanjala, E.; Di Tanna, G.L.; A Weiss, H.; Burton, M.J. Smartphone-based screening for visual impairment in Kenyan school children: A cluster randomised controlled trial. Lancet Glob. Health 2018, 6, e924–e932. [Google Scholar] [CrossRef]
- Pasolini, A. Smartphone-Based Kit Makes Eye Tests Cheap and Portable. Newatlas.com. 2013. Available online: https://newatlas.com/peek-eye-testing-smartphone/28764/ (accessed on 9 May 2019).
- American Cancer Society. Key Statistics for Melanoma Skin Cancer. Available online: https://www.cancer.org/cancer/melanoma-skin-cancer/about/key-statistics.html (accessed on 6 January 2019).
- Employment and Benefits|American Academy of Dermatology. New Study Shows Significant Economic Burden of Skin Disease in the United States. 1 March 2017. Available online: https://www.aad.org/media/news-releases/burden-of-skin-disease (accessed on 6 January 2019).
- Hubiche, T.; Valério, L.; Boralevi, F.; Mahe, E.; Skandalis, C.B.; Phan, A.; del Giudice, P. for the Research Group of the French Society of Pediatric Dermatology (Groupe de Recherche de la Société Française de Dermatologie Pédiatrique) Visualization of patients’ skin lesions on their smartphones: A newstep during dermatology visits. JAMA Dermatol. 2016, 152, 95–97. [Google Scholar] [CrossRef]
- Börve, A.; Gyllencreutz, J.; Terstappen, K.; Backman, E.; Aldenbratt, A.; Danielsson, M.; Gillstedt, M.; Sandberg, C.; Paoli, J. Smartphone Teledermoscopy Referrals: A Novel Process for Improved Triage of Skin Cancer Patients. Acta Derm. Venereol. 2015, 95, 186–190. [Google Scholar] [CrossRef]
- Friedman, R.J.; Rigel, D.S.; Kopf, A.W. Early Detection of Malignant Melanoma: The Role of Physician Examination and Self-Examination of the Skin. CA Cancer. J. Clin. 1985, 35, 130–151. [Google Scholar] [CrossRef]
- Wadhawan, T.; Situ, N.; Lancaster, K.; Yuan, X.; Zouridakis, G. SkinScan©: A Portable Library for Melanoma Detection on Handheld Devices. In Proceedings of the 2011 IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Chicago, IL, USA, 30 March–2 April 2011; pp. 133–136. [Google Scholar]
- Vo-Dinh, T.; Cullum, B.; Kasili, P. Development of a multi-spectral imaging system for medical applications. J. Phys. D Appl. Phys. 2003, 36, 1663–1668. [Google Scholar] [CrossRef]
- Kim, S.; Cho, D.; Kim, J.; Kim, M.; Youn, S.; Jang, J.E.; Je, M.; Lee, D.H.; Lee, B.; Farkas, D.L.; Hwang, J.Y. Smartphone-based multispectral imaging: system development and potential for mobile skin diagnosis. Biomed. Opt. Express 2016, 7, 5294–5307. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; He, X.; Wang, Y.; Liu, H.; Xu, D.; Guo, F. Review of spectral imaging technology in biomedical engineering: Achievements and challenges. J. Biomed. Opt. 2013, 18, 100901. [Google Scholar] [CrossRef]
- Kapsokalyvas, D.; Cicchi, R.; Bruscino, N.; Alfieri, D.; Prignano, F.; Massi, D.; Lotti, T.; Pavone, F.S. In-vivo imaging of psoriatic lesions with polarization multispectral dermoscopy and multiphoton microscopy. Biomed. Opt. Express 2014, 5, 2405–2419. [Google Scholar] [CrossRef] [PubMed]
- Elbaum, M.; Kopf, A.W.; Rabinovitz, H.S.; Langley, R.G.; Kamino, H.; Mihm, M.C.; Sober, A.J.; Peck, G.L.; Bogdan, A.; Gutkowicz-Krusin, D.; et al. Automatic differentiation of melanoma from melanocytic nevi with multispectral digital dermoscopy: A feasibility study. J. Am. Acad. Dermatol. 2001, 44, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Yanagisawa, T.; Mitsui, M.; Murakami, Y.; Yamaguchi, M.; Ohyama, N.; Abe, T.; Yokoi, I.; Matsuoka, Y.; Kubota, Y. Extraction of acne lesion in acne patients from multispectral images. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 4078–4081. [Google Scholar]
- Kuzmina, I.; Lacis, M.; Spigulis, J.; Berzina, A.; Valeine, L. Study of smartphone suitability for mapping of skin chromophores. J. Biomed. Opt. 2015, 20, 90503. [Google Scholar] [CrossRef] [PubMed]
- Groner, W.; Winkelman, J.W.; Harris, A.G.; Ince, C.; Bouma, G.J.; Messmer, K.; Nadeau, R.G. Orthogonal polarization spectral imaging: A new method for study of the microcirculation. Nat. Med. 1999, 5, 1209–1212. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.E.; Semeere, A.; Osman, H.; Peterson, G.; Rajadhyaksha, M.; González, S.; Martin, J.N.; Anderson, R.R.; Tearney, G.J.; Kang, D. Smartphone confocal microscopy for imaging cellular structures in human skin in vivo. Biomed. Opt. Express 2018, 9, 1906–1915. [Google Scholar] [CrossRef]
- Kim, S.; Crose, M.; Eldridge, W.J.; Cox, B.; Brown, W.J.; Wax, A. Design and implementation of a low-cost, portable OCT system. Biomed. Opt. Express 2018, 9, 1232–1243. [Google Scholar] [CrossRef] [PubMed]
- Karargyris, A.; Karargyris, O.; Pantelopoulos, A. DERMA/Care: An Advanced image-Processing Mobile Application for Monitoring Skin Cancer. In Proceedings of the 2012 IEEE 24th International Conference on Tools with Artificial Intelligence (ICTAI 2012), Athens, Greece, 7–9 November 2012; pp. 1–7. [Google Scholar]
- Bourouis, A.; Zerdazi, A.; Feham, M.; Bouchachia, A. M-health: Skin disease analysis system using smartphone’s camera. Procedia Comput. Sci. 2013, 19, 1116–1120. [Google Scholar] [CrossRef]
- Abuzaghleh, O.; Barkana, B.D.; Faezipour, M. Noninvasive Real-Time Automated Skin Lesion Analysis System for Melanoma Early Detection and Prevention. IEEE J. Transl. Eng. Heal. Med. 2015, 3, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Cornet, V.P.; Holden, R.J. Systematic review of smartphone-based passive sensing for health and wellbeing. J. Biomed. Inform. 2018, 77, 120–132. [Google Scholar] [CrossRef]
- Lu, H.; Frauendorfer, D.; Rabbi, M.; Mast, M.S.; Chittaranjan, G.T.; Campbell, A.T.; Gatica-Perez, D.; Choudhury, T. StressSense: Detecting stress in unconstrained acoustic environments using smartphones. In Proceedings of the 2012 ACM Conference on Ubiquitous Computing—UbiComp ’12, Pittsburgh, PA, USA, 5–8 September 2012; p. 351. [Google Scholar]
- Estevão, M.S.; Duarte, F.J.; Fernandes, E.; Gil Santos, A.; Marques, M.M.B. Unexpected reactivity of trifluoromethylated olefins with indole: A mechanistic investigation. Tetrahedron Lett. 2012, 53, 2132–2136. [Google Scholar] [CrossRef]
- Huang, Y.; Xiong, H.; Leach, K.; Zhang, Y.; Chow, P.; Fua, K.; Teachman, B.A.; Barnes, L.E. Assessing social anxiety using gps trajectories and point-of-interest data. In Proceedings of the 2016 ACM International Joint Conference on Pervasive and Ubiquitous Computing, Heidelberg, Germany, 12–16 September 2016; pp. 898–903. [Google Scholar]
- Eysenbach, G.; Saeb, S.; Mohr, D.; Reis, D.; Asselbergs, J.; Ruwaard, J.; Ejdys, M.; Schrader, N.; Sijbrandij, M.; Riper, H. Mobile Phone-Based Unobtrusive Ecological Momentary Assessment of Day-to-Day Mood: An Explorative Study. J. Med Internet Res. 2016, 18, e72. [Google Scholar]
- Ma, Y.; Xu, B.; Bai, Y.; Sun, G.; Zhu, R. Infer Daily Mood using Mobile Phone Sensing. Ad Hoc Sens. Wirel. Netw. 2014, 20, 133–152. [Google Scholar]
- Ben-Zeev, D.; Scherer, E.A.; Wang, R.; Xie, H.; Campbell, A.T. Next-Generation Psychiatric Assessment: Using Smartphone Sensors to Monitor Behavior and Mental Health. Psychiatr. Rehabil. J. 2015, 38, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Eysenbach, G.; Proudfoot, J.; Dear, B.; Burns, M.N.; Begale, M.; Duffecy, J.; Gergle, D.; Karr, C.J.; Giangrande, E.; Mohr, D.C. Harnessing Context Sensing to Develop a Mobile Intervention for Depression. J. Med. Internet Res. 2011, 13, e55. [Google Scholar]
- Canzian, L.; Musolesi, M. Trajectories of depression. In Proceedings of the 2015 ACM International Joint Conference on Pervasive and Ubiquitous Computing—UbiComp ’15, New York, NY, USA, 07–11 September 2015; pp. 1163–1304. [Google Scholar]
- Saeb, S.; Lattie, E.G.; Schueller, S.M.; Kording, K.P.; Mohr, D.C.; Jorm, A. The relationship between mobile phone location sensor data and depressive symptom severity. PeerJ 2016, 4, 2137. [Google Scholar] [CrossRef]
- Eysenbach, G.; Ruwaard, J.; Bardram, J.; Saeb, S.; Zhang, M.; Karr, C.J.; Schueller, S.M.; E Corden, M.; Körding, K.P.; Mohr, D.C. Mobile Phone Sensor Correlates of Depressive Symptom Severity in Daily-Life Behavior: An Exploratory Study. J. Med. Internet Res. 2015, 17, e175. [Google Scholar]
- Wahle, F.; Kowatsch, T.; Fleisch, E.; Rufer, M.; Weidt, S. Mobile Sensing and Support for People with Depression: A Pilot Trial in the Wild. JMIR mHealth uHealth 2016, 4, e111. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Chen, F.; Chen, Z.; Li, T.; Harari, G.; Tignor, S.; Zhou, X.; Ben-Zeev, D.; Campbell, A.T. StudentLife: Assessing mental health, academic performance and behavioral trends of college students using smartphones. In Proceedings of the 2014 ACM International Joint Conference on Pervasive and Ubiquitous Computing, Seattle, WA, USA, 13–17 September 2014; pp. 3–14. [Google Scholar]
- Osmani, V.; Maxhuni, A.; Grünerbl, A.; Lukowicz, P.; Haring, C.; Mayora, O. Monitoring activity of patients with bipolar disorder using smart phones. In Proceedings of the International Conference on Advances in Mobile Computing & Multimedia, Vienna, Austria, 2–4 December 2013; pp. 85–92. [Google Scholar]
- Gruenerbl, A.; Osmani, V.; Bahle, G.; Carrasco-Jimenez, J.C.; Oehler, S.; Mayora, O.; Haring, C.; Lukowicz, P. Using smart phone mobility traces for the diagnosis of depressive and manic episodes in bipolar patients. In Proceedings of the 5th Augmented Human International Conference, Kobe, Japan, 7–8 March 2014. [Google Scholar]
- Grüunerbl, A.; Muaremi, A.; Osmani, V.; Bahle, G.; Ohler, S.; Troster, G.; Mayora, O.; Haring, C.; Lukowicz, P. Smartphone-based recognition of states and state changes in bipolar disorder patients. IEEE J. Biomed. Heal. Informatics 2015, 19, 140–148. [Google Scholar] [CrossRef]
- Abdullah, S.; Matthews, M.; Frank, E.; Doherty, G.; Gay, G.; Choudhury, T. Automatic detection of social rhythms in bipolar disorder. J. Am. Med Informatics Assoc. 2016, 23, 538–543. [Google Scholar] [CrossRef]
- Eysenbach, G.; Faurholt-Jepsen, M.; Mayora, O.; Buntrock, C.; Beiwinkel, T.; Kindermann, S.; Maier, A.; Kerl, C.; Moock, J.; Barbian, G.; et al. Using Smartphones to Monitor Bipolar Disorder Symptoms: A Pilot Study. JMIR Heal. 2016, 3, e2. [Google Scholar]
- Ben-Zeev, D.; Wang, R.; Abdullah, S.; Brianv, R.; Scherer, E.A.; Mistler, L.A.; Hauser, M.; Kane, J.M.; Campbell, A.; Choudhury, T. Mobile Behavioral Sensing for Outpatients and Inpatients with Schizophrenia. Psychiatr. Serv. 2016, 67, 558–591. [Google Scholar] [CrossRef]
- DiFrancesco, S.; Fraccaro, P.; Van Der Veer, S.N.; Alshoumr, B.; Ainsworth, J.; Bellazzi, R.; Peek, N. Out-of-Home Activity Recognition from GPS Data in Schizophrenic Patients. In Proceedings of the 2016 IEEE 29th International Symposium on Computer-Based Medical Systems (CBMS), Dublin, Ireland, 20–24 June 2016; pp. 324–328. [Google Scholar]
- Wang, R.; Aung, M.S.H.; Abdullah, S.; Brian, R.; Campbell, A.T.; Choudhury, T.; Hauser, M.; Kane, J.; Scherer, E.A.; Tseng, V.W.S.; Ben-Zeev, D. CrossCheck: Toward passive sensing and detection of mental health changes in people with schizophrenia. In Proceedings of the 2016 ACM International Joint Conference on Pervasive and Ubiquitous Computing—UbiComp ’16, Heidelberg, Germany, 12–16 September 2016. [Google Scholar]
- About the Study. Resources for Parents|Autism & Beyond. Available online: https://autismandbeyond.researchkit.duke.edu/study (accessed on 6 January 2019).
- Coutinho, E.S.F.; Bloch, K.V.; Coeli, C.M. One-year mortality among elderly people after hospitalization due to fall-related fractures: comparison with a control group of matched elderly. Cad. Saúde Pública 2012, 28, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.; Fawcett, G.; Brisebois, L.; Hughes, J. A Demographic, Employment and Income Profile of Canadians with Disabilities Aged 15 Years and Over, 2017. Statistics Canada: Canada’s National Statistical Agency. 28 November 2018. Available online: https://www150.statcan.gc.ca/n1/pub/89-654-x/89-654-x2018002-eng.htm (accessed on 5 February 2019).
- Edjoc, R.; Gal, J. Sleep Apnea in Canada, 2016 and 2017. Statistics Canada: Canada’s National Statistical Agency. 24 October 2018. Available online: https://www150.statcan.gc.ca/n1/pub/82-625-x/2018001/article/54979-eng.htm (accessed on 5 February 2019).
- Nandakumar, R.; Gollakota, S.; Watson, N. Contactless Sleep Apnea Detection on Smartphones. GetMobile: Mob. Comput. Commun. 2015, 19, 22–24. [Google Scholar] [CrossRef]
- Sin, D.D.; Fitzgerald, F.; Parker, J.D.; Newton, G.; Floras, J.S.; Bradley, T.D. Risk Factors for Central and Obstructive Sleep Apnea in 450 Men And Women with Congestive Heart Failure. Am. J. Respir. Crit. Care Med. 1999, 160, 1101–1106. [Google Scholar] [CrossRef] [PubMed]
- Ni, B.; Wang, G.; Moulin, P. RGBD-HuDaAct: A color-depth video database for human daily activity recognition. In Proceedings of the 2011 IEEE International Conference on Computer Vision Workshops (ICCV Workshops), Barcelona, Spain, 6–13 November 2011; pp. 1147–1153. [Google Scholar]
- Derawi, M.; Bours, P. Gait and activity recognition using commercial phones. Comput. Secur. 2013, 39, 137–144. [Google Scholar] [CrossRef]
- Ronao, C.A.; Cho, S.-B. Human activity recognition using smartphone sensors with two-stage continuous hidden Markov models. In Proceedings of the 2014 10th International Conference on Natural Computation (ICNC), Xiamen, China, 19–21 August 2014; pp. 681–686. [Google Scholar]
- Seera, M.; Loo, C.K.; Lim, C.P. A hybrid FMM-CART model for human activity recognition. In Proceedings of the 2014 IEEE International Conference on Systems, Man and Cybernetics—SMC, San Diego, CA, USA, 5–8 October 2014; pp. 182–187. [Google Scholar]
- Eastwood, M.; Jayne, C. Evaluation of hyperbox neural network learning for classification. Neurocomputing 2014, 133, 249–257. [Google Scholar] [CrossRef]
- Catal, C.; Tufekci, S.; Pirmit, E.; Kocabag, G. On the use of ensemble of classifiers for accelerometer-based activity recognition. Appl. Soft Comput. 2015, 37, 1018–1022. [Google Scholar] [CrossRef]
- De, D.; Bharti, P.; Das, S.; Chellappan, S. Multi-modal Wearable Sensing for Fine-grained Activity Recognition in Healthcare. IEEE Comput. Soc. 2015, 19, 1. [Google Scholar]
- Wang, A.; Chen, G.; Yang, J.; Zhao, S.; Chang, C.-Y. A Comparative Study on Human Activity Recognition Using Inertial Sensors in a Smartphone. IEEE Sensors J. 2016, 16, 4566–4578. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, Q.; Soh, Y.C.; Zhang, L. Robust Human Activity Recognition Using Smartphone Sensors via CT-PCA and Online SVM. IEEE Trans. Ind. Informatics 2017, 13, 3070–3080. [Google Scholar] [CrossRef]
- Anguita, D.; Ghio, A.; Oneto, L.; Parra, X.; Reyes-Ortiz, J.L. Human Activity Recognition on Smartphones Using a Multiclass Hardware-Friendly Support Vector Machine. Adv. Nonlinear Speech Process. 2012, 7657, 216–223. [Google Scholar]
- Kwon, Y.; Kang, K.; Bae, C. Unsupervised learning for human activity recognition using smartphone sensors. Expert Syst. Appl. 2014, 41, 6067–6074. [Google Scholar] [CrossRef]
- Ronao, C.A.; Cho, S.-B. Human activity recognition with smartphone sensors using deep learning neural networks. Expert Syst. Appl. 2016, 59, 235–244. [Google Scholar] [CrossRef]
- Ronao, C.A.; Cho, S.-B. Deep Convolutional Neural Networks for Human Activity Recognition with Smartphone Sensors. Adv. Nonlinear Speech Process. 2015, 9492, 46–53. [Google Scholar]
- Tao, D.; Wen, Y.; Hong, R. Multicolumn Bidirectional Long Short-Term Memory for Mobile Devices-Based Human Activity Recognition. IEEE Internet Things J. 2016, 3, 1124–1134. [Google Scholar] [CrossRef]
- Ordóñez, F.J.; Roggen, D. Deep Convolutional and LSTM Recurrent Neural Networks for Multimodal Wearable Activity Recognition. Sensors 2016, 16, 115. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Li, Y.; Bao, S.-O. Fall detection by built-in tri-accelerometer of smartphone. In Proceedings of the 2012 IEEE-EMBS International Conference on Biomedical and Health Informatics (BHI), Hong Kong, China, 5–7 January 2012; pp. 184–187. [Google Scholar]
- Thammasat, E.; Chaicharn, J. A simply fall-detection algorithm using accelerometers on a smartphone. In Proceedings of the 2012 5th Biomedical Engineering International Conference (BMEiCON), Ubon Ratchathani, Thailand, 5–7 December 2012; pp. 1–4. [Google Scholar]
- Lee, H.S.; Choi, Y.S.Y.S.; Seo, Y.Y.; Shim, E.E.; Hosub, L.; Sunjae, L.; Sang, C.Y. A new posture monitoring system for preventing physical illness of smartphone users. In Proceedings of the 2013 IEEE 10th Consumer Communications and Networking Conference (CCNC), Las Vegas, NV, USA, 11–14 January 2013; pp. 821–825. [Google Scholar]
- Fleury, A.; Mourcou, Q.; Franco, C.; Diot, B.; Demongeot, J.; Vuillerme, N. Evaluation of a Smartphone-based audio-biofeedback system for improving balance in older adults—A pilot study. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 1198–1201. [Google Scholar]
- Chen, Z.; Jiang, C.; Xie, L. A Novel Ensemble ELM for Human Activity Recognition Using Smartphone Sensors. IEEE Trans. Ind. Informatics 2018, 1. [Google Scholar] [CrossRef]
- Vaughn, A.; Biocco, P.; Liu, Y.; Anwar, M. Activity Detection and Analysis Using Smartphone Sensors. In Proceedings of the 2018 IEEE International Conference on Information Reuse and Integration (IRI), Salt Lake City, UT, USA, 6–9 July 2018. [Google Scholar]
- Hassan, M.M.; Uddin, M.Z.; Mohamed, A.; Almogren, A. A robust human activity recognition system using smartphone sensors and deep learning. Futur. Gener. Comput. Syst. 2018, 81, 307–313. [Google Scholar] [CrossRef]
- Li, S.; Li, C.; Li, W.; Hou, Y.; Cook, C. Smartphone-sensors Based Activity Recognition Using IndRNN. In Proceedings of the 2018 ACM International Joint Conference and 2018 International Symposium on Pervasive and Ubiquitous Computing and Wearable Computers—UbiComp ’18, Singapore, Singapore, 08–12 October 2018; pp. 1541–1547. [Google Scholar]
- Li, H.; Trocan, M. Deep learning of smartphone sensor data for personal health assistance. Microelectron. J. 2018. [Google Scholar] [CrossRef]
- Bort-Roig, J.; Puig-Ribera, A.; Contreras, R.S.; Chirveches-Pérez, E.; Martori, J.C.; Gilson, N.D.; McKenna, J. Monitoring sedentary patterns in office employees: validity of an m-health tool (Walk@Work-App) for occupational health. Gac. Sanit. 2018, 32, 563–566. [Google Scholar] [CrossRef]
- Wan, N.; Lin, G. Classifying Human Activity Patterns from Smartphone Collected GPS data: a Fuzzy Classification and Aggregation Approach. Trans. GIS 2016, 20, 869–886. [Google Scholar] [CrossRef]
- Shoaib, M.; Scholten, H.; Havinga, P. Towards Physical Activity Recognition Using Smartphone Sensors. In Proceedings of the 2013 IEEE 10th International Conference on Ubiquitous Intelligence & Computing and 2013 IEEE 10th International Conference on Autonomic & Trusted Computing (UIC/ATC), Vietri sul Mere, Italy, 18–21 December 2013; pp. 80–87. [Google Scholar]
- Eysenbach, G.; Dunton, G.; Hekler, E.; Gay, V.; Wu, W.; Dasgupta, S.; Ramirez, E.E.; Peterson, C.; Norman, G.J. Classification Accuracies of Physical Activities Using Smartphone Motion Sensors. J. Med. Internet Res. 2012, 14, e130. [Google Scholar]
- Lee, Y.; Yeh, H.; Kim, K.-H.; Choi, O. A Real-time Fall Detection System Based on the Acceleration Sensor of Smartphone. Int. J. Autom. 2017, 10, 315–326. [Google Scholar] [CrossRef]
- Hakim, A.; Huq, M.S.; Shanta, S.; Ibrahim, B. Smartphone Based Data Mining for Fall Detection: Analysis and Design. Procedia Comput. Sci. 2017, 105, 46–51. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Chen, K.H.; Yang, J.J.; Jaw, F.S. Smartphone-based fall detection algorithm using feature extraction. In Proceedings of the 2016 9th International Congress on Image and Signal Processing, BioMedical Engineering and Informatics, CISP-BMEI 2016, Datong, China, 15–17 October 2016; pp. 1535–1540. [Google Scholar]
- Shen, V.R.; Lai, H.-Y.; Lai, A.-F. The implementation of a smartphone-based fall detection system using a high-level fuzzy Petri net. Appl. Soft Comput. 2015, 26, 390–400. [Google Scholar] [CrossRef]
- Ashfak Habib, M.; Mohktar, M.S.; Bahyah Kamaruzzaman, S.; Seang Lim, K.; Maw Pin, T.; Ibrahim, F. Smartphone-based solutions for fall detection and prevention: Challenges and open issues. Sensors (Switzerland) 2014, 14, 7181–7208. [Google Scholar]
- Luque, R.; Casilari, E.; Morón, M.-J.; Redondo, G. Comparison and Characterization of Android-Based Fall Detection Systems. Sensors 2014, 14, 18543–18574. [Google Scholar] [CrossRef]
- Støve, M.P.; Palsson, T.S.; Hirata, R.P. Smartphone-based accelerometry is a valid tool for measuring dynamic changes in knee extension range of motion. Knee 2018, 25, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Jenny, J.Y.; Bureggah, A.; Diesinger, Y. Measurement of the knee flexion angle with smartphone applications: Which technology is better? Knee Surg. Sport. Traumatol. Arthrosc. 2016, 24, 2874–2877. [Google Scholar] [CrossRef] [PubMed]
- Min, J.-K.; Doryab, A.; Wiese, J.; Amini, S.; Zimmerman, J.; Hong, J.I. Toss ‘N’ Turn: Smartphone as Sleep and Sleep Quality Detector. In Proceedings of the 32nd Annual ACM Conference on Human Factors in Computing Systems—CHI ’14, Toronto, ON, Canada, 26 April–1 May 2014; pp. 477–486. [Google Scholar]
- Chen, Z.; Lin, M.; Chen, F.; Lane, N.; Cardone, G.; Wang, R.; Li, T.; Chen, Y.; Choudhury, T.; Cambell, A. Unobtrusive Sleep Monitoring using Smartphones. In Proceedings of the ICTs for Improving Patients Rehabilitation Research Techniques, Venice, Italy, 5–8 May 2013; pp. 145–152. [Google Scholar]
- Natale, V.; Drejak, M.; Erbacci, A.; Tonetti, L.; Fabbri, M.; Martoni, M. Monitoring sleep with a smartphone accelerometer. Sleep Boil. Rhythm. 2012, 10, 287–292. [Google Scholar] [CrossRef]
- Prevention of Blindness and Deafness World Health Organization. 26 July 2018. Available online: https://www.who.int/pbd/deafness/estimates/en/ (accessed on 25 April 2019).
- Bernstein, J.G.W.; Grant, K.W. Auditory and auditory-visual intelligibility of speech in fluctuating maskers for normal-hearing and hearing-impaired listeners. J. Acoust. Soc. 2009, 125, 3358. [Google Scholar] [CrossRef] [PubMed]
- Darwin, C.J. Listening to speech in the presence of other sounds. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 1011–1021. [Google Scholar] [CrossRef] [PubMed]
- Arlinger, S. Negative consequences of uncorrected hearing loss—A review. Int. J. Audiol. 2003, 42, 17–20. [Google Scholar] [CrossRef]
- Pichora-Fuller, M.K.; Dupuis, K.; Reed, M.; Lemke, U. Helping Older People with Cognitive Decline Communicate: Hearing Aids as Part of a Broader Rehabilitation Approach. Semin. Hear. 2013, 34, 308–330. [Google Scholar]
- American Academy of Audiology. Childhood Hearing Screening Guidelines. 2011. Available online: https://www.cdc.gov/ncbddd/hearingloss/documents/aaa_childhood-hearing-guidelines_2011.pdf (accessed on 25 April 2019).
- American Speech-Language-Hearing Association. Guidelines for Audiologic Screening. 1 January 1997. Available online: https://www.asha.org/policy/GL1997-00199.htm (accessed on 25 April 2019).
- Maclennan-Smith, F.; Swanepoel, D.W.; Hall, J.W. Validity of diagnostic pure-tone audiometry without a sound-treated environment in older adults. Int. J. Audiol. 2013, 52, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Abu-Ghanem, S.; Handzel, O.; Ness, L.; Ben-Artzi-Blima, M.; Fait-Ghelbendorf, K.; Himmelfarb, M. Smartphone-based audiometric test for screening hearing loss in the elderly. Eur. Arch. Oto Rhino Laryngol. 2016, 273, 333–339. [Google Scholar] [CrossRef]
- Hussein, S.Y.; Swanepoel, D.W.; De Jager, L.B.; Myburgh, H.C.; Hugo, J.; Eikelboom, R.H. Smartphone hearing screening in mHealth assisted community-based primary care. J. Telemed. Telecare 2016, 22, 405–412. [Google Scholar] [CrossRef]
- Louw, C.; Swanepoel, D.W.; Eikelboom, R.H.; Myburgh, H.C. Smartphone-Based Hearing Screening at Primary Health Care Clinics. Ear Hear. 2017, 38, 1. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wang, S.; Li, J.; Tan, H.; Jia, W.; Wang, Z. Smartphone-Based Hearing Self-Assessment System Using Hearing Aids with Fast Audiometry Method. IEEE Trans. Biomed. Circuits Syst. 2019, 13, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Eysenbach, G.; Paglialonga, A.; Handzel, O.; Mahomed-Asmail, F.; Moodie, S.; Bright, T.; Pallawela, D. Validated Smartphone-Based Apps for Ear and Hearing Assessments: A Review. JMIR Rehabilitation Assist. Technol. 2016, 3, e13. [Google Scholar]
- Amlani, A.M.; Taylor, B.; Levy, C.; Robbins, R. Utility of smartphone-based hearing aid applications as a substitute to traditional hearing aids. Hear. Rev. 2013, 20, 16–18. [Google Scholar]
- Lin, Y.C.; Lai, Y.H.; Chang, H.W.; Tsao, Y.; Chang, Y.P.; Chang, R.Y. SmartHear: A Smartphone-Based Remote Microphone Hearing Assistive System Using Wireless Technologies. IEEE Syst. J. 2018, 12, 20–29. [Google Scholar] [CrossRef]
- Boulos, M.N.K.; Brewer, A.C.; Karimkhani, C.; Buller, D.B.; Dellavalle, R.P. Mobile medical and health apps: state of the art, concerns, regulatory control and certification. Online J. Public Health Inform. 2014, 5, 229. [Google Scholar]
- Van Norman, G.A. Drugs and Devices: Comparison of European and U.S. Approval Processes. JACC Basic Transl. Sci. 2016, 1, 399–412. [Google Scholar] [CrossRef]
- Buijink, A.W.G.; Visser, B.J.; Marshall, L. Medical apps for smartphones: Lack of evidence undermines quality and safety. Evid. Based Med. 2013, 18, 90–92. [Google Scholar] [CrossRef]
- Cummings, E.; Borycki, E.M.; Roehrer, E. Issues and considerations for healthcare consumers using mobile applications. Stud. Heal. Technol. Inform. 2013, 183, 227–231. [Google Scholar]
- O’Neill, S.; Brady, R.R.W. Colorectal smartphone apps: Opportunities and risks. Color. Dis. 2012, 14. [Google Scholar] [CrossRef]
- Demidowich, A.P.; Bloomgarden, Z.; Lu, K.; Tamler, R. An evaluation of diabetes self-management applications for Android smartphones. J. Telemed. Telecare 2012, 18, 235–238. [Google Scholar] [CrossRef]
- Ferrero, N.A.; Morrell, D.S.; Burkhart, C.N. Skin scan: A demonstration of the need for FDA regulation of medical apps on iPhone. J. Am. Acad. Dermatol. 2013, 68, 515–516. [Google Scholar] [CrossRef]
- Wolf, J.A.; Ferris, L.K. Diagnostic Inaccuracy of Smartphone Applications for Melanoma Detection—Reply. JAMA Dermatol. 2013, 149, 885. [Google Scholar] [CrossRef] [PubMed]
- Visvanathan, A.; Hamilton, A.; Brady, R. Smartphone apps in microbiology—is better regulation required? Clin. Microbiol. Infect. 2012, 18, E218–E220. [Google Scholar] [CrossRef]
- Robson, Y.; Blackford, S.; Roberts, D. Caution in melanoma risk analysis with smartphone application technology. Br. J. Dermatol. 2012, 167, 703–704. [Google Scholar] [CrossRef]
- Huckvale, K.; Car, M.; Morrison, C.; Car, J. Apps for asthma self-management: a systematic assessment of content and tools. BMC Med. 2012, 10, 144. [Google Scholar] [CrossRef]
- McKinstry, B. Currently available smartphone apps for asthma have worrying deficiencies. Evidence-Based Med. 2013, 18, 45. [Google Scholar] [CrossRef]
- O’neill, S.; Brady, R.R. Clinical involvement and transparency in medical apps; not all apps are equal. Colorectal Dis. 2013, 15, 122. [Google Scholar] [CrossRef] [PubMed]
- Sucala, M.; Cuijpers, P.; Muench, F.; Cardoș, R.; Soflau, R.; Dobrean, A.; Achimas-Cadariu, P.; David, D.; Achimas-Cadariu, P. Anxiety: There is an app for that. A systematic review of anxiety apps. Depress. Anxiety 2017, 34, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Strickland, E. The FDA Takes on Mobile Health Apps. IEEE Spectrum: Technology, Engineering, and Science News. 12 September 2012. Available online: https://spectrum.ieee.org/biomedical/devices/the-fda-takes-on-mobile-health-apps (accessed on 6 January 2019).
- U.S. Department of Health and Human Services Food and Drug Administration. Mobile Medical Applications: Guidance for Industry and Food and Drug Administration Staff. 2015; Available online: https://www.fda.gov/media/80958/download (accessed on 6 January 2019).
- Premarket Notification 510(k). U S Food and Drug Administration Home Page. Available online: https://www.fda.gov/MedicalDevices/DeviceRegulationandGuidance/HowtoMarketYourDevice/PremarketSubmissions/PremarketNotification510k/default.htm (accessed on 6 January 2019).
- Institute of Medicine (US); Committee on the Public Health Effectiveness of the FDA 510 (k) Clearance Process. Medical Devices and the Public’s Health: The FDA 510(k) Clearance Process at 35 Years; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Waxman, H.A. H.R.3095—101st Congress (1989–1990): Safe Medical Devices Act of 1990. Congress.gov, 28 November 1990. Available online: https://www.congress.gov/bill/101st-congress/house-bill/3095 (accessed on 6 January 2019).
- Cain, M. One company’s experience: blazing the trail with the first FDA-approved medical imaging app. Biomed. Instrum. Technol. 2012, 46, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Dolan, B. FDA Clears First Diagnostic Radiology App, Mobile MIM. MobiHealthNews. 4 February 2011. Available online: https://www.mobihealthnews.com/10173/fda-clears-first-diagnostic-radiology-app-mobile-mim (accessed on 6 January 2019).
- AliveCor, Inc. KardiaMobile. Available online: https://store.alivecor.com/products/kardiamobile (accessed on 6 January 2019).
- IExaminer. iEXAMINER. Available online: http://www.welchallyn.ca/en/microsites/iexaminer.html (accessed on 6 January 2019).
- Welldoc Inc. Product. Available online: https://www.welldoc.com/product/ (accessed on 6 January 2019).
- PureWeb. Healthcare Solutions|PureWeb|ResolutionMD. Available online: https://www.pureweb.com/healthcare (accessed on 6 January 2019).
- The European Parliament and the Council of the European Union. Medical Device Directive 93/42/EEC. Off. J. Eur. Union 1993, 169, 1–60. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CONSLEG:1993L0042:20071011:en:PDF (accessed on 6 January 2019).
- The European Parliament and the Council of the European Union. Medical Device Directive 2007/47/EC. Off. J. Eur. Union 2007, 1–35. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2007:247:0021:0055:en:PDF (accessed on 6 January 2019).
- CE Marking. Together Against Trafficking in Human Beings. 30 August 2017. Available online: http://ec.europa.eu/growth/single-market/ce-marking/ (accessed on 6 January 2019).
- ONCOassist. Adjuvant Calculator—Oncology Apps—ONCOassist healthcare professional. Available online: https://oncoassist.com/ (accessed on 6 January 2019).
- European Parliament and Council of the European Union. Regulation (EU) 2017/746 of the European Parliament and of the Council of 5 April 2017 on medical devices. Off. J. Eur. Union 2017, 60, 1–175. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32017R0745 (accessed on 6 January 2019).
- Edwards, C. EU Medical Device Regulation Changes: What do They Mean? Verdict Medical Devices. 16 October 2018. Available online: https://www.medicaldevice-network.com/features/eu-medical-device-regulation-changes/ (accessed on 6 January 2019).
- Making a Success of Brexit. GOV.UK. Available online: https://www.gov.uk/government/news/medicines-and-healthcare-products-regulatory-agency-statement-on-the-outcome-of-the-eu-referendum (accessed on 6 January 2019).
- How Medicines, Medical Devices and Clinical Trials Would Be Regulated If There’s No Brexit deal. GOV.UK. Available online: https://www.gov.uk/government/publications/how-medicines-medical-devices-and-clinical-trials-would-be-regulated-if-theres-no-brexit-deal/how-medicines-medical-devices-and-clinical-trials-would-be-regulated-if-theres-no-brexit-deal (accessed on 6 January 2019).
- Medicines & Healthcare products Regulatory Agency. Medical Device Stand—Alone Software Including Apps. GOV.UK. 2014. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/.uploads/attachment_data/file/548313/Software_flow_chart_Master.pdf (accessed on 6 January 2019).
- Lightley, D. When Is A Mobile App Classed as a Medical Device | MHRA Compliant Apps. Genetic Digital. 9 December 2013. Available online: https://www.geneticdigital.co.uk/2013/03/when-should-an-app-be-classed-as-a-device/ (accessed on 6 January 2019).
- Heather, B. Explainer: When Is An App Not An App (But A Medical Device)? Digital Health. 7 March 2017. Available online: https://www.digitalhealth.net/2016/09/explainer-when-is-an-app-not-an-app-but-a-medical-device/ (accessed on 6 January 2019).
- Medical Device Technology Forum on the Use of Software as a Medical Device—12 May 2010. Ofcom|Statutory Duties and Regulatory Principles. 11 January 2012. Available online: https://webarchive.nationalarchives.gov.uk/.20150113200213/http://www.mhra.gov.uk/Howweregulate/NewTechnologiesForums/DevicesNewTechnologyForum/Forums/CON084987 (accessed on 6 January 2019).
- Approval of Medical Devices. Planning D-Day (April 2003)—Library of Congress Information Bulletin. 1 September 2014. Available online: http://www.loc.gov/law/help/medical-devices/ (accessed on 6 January 2019).
- Kramer, D.B.; Xu, S.; Kesselheim, A.S. Regulation of Medical Devices in the United States and European Union. New Engl. J. Med. 2012, 366, 848–855. [Google Scholar] [CrossRef]
- Sorenson, C.; Drummond, M. Improving Medical Device Regulation: The United States and Europe in Perspective. Milbank Q. 2014, 92, 114–150. [Google Scholar] [CrossRef]
- Cohen, D. How a fake hip showed up failings in European device regulation. BMJ 2012, 345, e7090. [Google Scholar] [CrossRef]
- Cohen, D.; Billingsley, M. Europeans are left to their own devices. BMJ 2011, 342, d2748. [Google Scholar] [CrossRef]
- Curfman, G.D.; Redberg, R.F. Medical Devices—Balancing Regulation and Innovation. New Engl. J. Med. 2011, 365, 975–977. [Google Scholar] [CrossRef]
- Emergo. EUROPE—Overview of medical device industry and healthcare statistics. 23 October 2018. Available online: https://www.emergobyul.com/resources/market-europe (accessed on 27 April 2019).
- Legislative Services Branch, “Consolidated federal laws of Canada, Medical Devices Regulations. Medical Devices Regulations. Available online: https://laws-lois.justice.gc.ca/eng/regulations/sor-98-282/fulltext.html (accessed on 27 April 2019).
- Emergo. Health Canada Medical Device License (MDL) and MDEL Registration. 12 November 2018. Available online: https://www.emergobyul.com/services/canada/canada-device-license (accessed on 27 April 2019).
- Australian Government Department of Health; Therapeutic Goods Administration (TGA). Consultation: Designation of Australian conformity assessment bodies for medical devices—Implementation. 14 June 2017. Available online: https://www.tga.gov.au/consultation/consultation-designation-australian-conformity-assessment-bodies-medical-devices-implementation (accessed on 27 April 2019).
- Australian Government Department of Health; Therapeutic Goods Administration (TGA). Use of market authorisation evidence from comparable overseas regulators/assessment bodies for medical devices (including IVDs). 28 November 2018. Available online: https://www.tga.gov.au/publication/use-market-authorisation-evidence-comparable-overseas-regulators-assessment-bodies-medical-devices-including-ivds (accessed on 27 April 2019).
- Australian Government Department of Health; Therapeutic Goods Administration (TGA). Medical Device Single Audit Program (MDSAP). 6 December 2016. Available online: https://www.tga.gov.au/medical-device-single-audit-program-mdsap (accessed on 27 April 2019).
- Notice: Medical Device Single Audit Program (MDSAP) Transition Plan—Frequently Asked Questions (FAQ). Canada.ca. 22 April 2016. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/medical-devices/activities/international/notice-transition-plan-medical-device-audit-program.html (accessed on 27 April 2019).

| Monitored Health Issues | Typically Used Smartphone Sensors |
|---|---|
| Cardiovascular activity e.g., heart rate (HR) and HR variability (HRV) | Image sensor (camera), microphone |
| Eye health | Image sensor (camera) |
| Respiratory and lung health | Image sensor (camera), microphone |
| Skin health | Image sensor camera) |
| Daily activity and fall | Motion sensors (accelerometer, gyroscope, proximity sensor), Global positioning system (GPS) |
| Sleep | Motion sensors (accelerometer, gyroscope) |
| Ear health | Microphone |
| Cognitive function and mental health | Motion sensors (accelerometer, gyroscope), camera, light sensor, GPS |
| Ref. | Year | Measured Signs | Type | Smartphone Model | Sensor Used | Video Resolution | Frame Rate (fps) | Video Length | Method | Performance wrt Standard Monitors | # of Subjects |
|---|---|---|---|---|---|---|---|---|---|---|---|
| [37] | 2018 | HR, HRV | Contact-based (index finger) | iPhone 6, Apple Inc., Cupertino CA | Front camera | 1280 × 720 | 240 | 5 min | • Reflection of light from the finger is measured. | Pearson Correlation coefficient (PC) for most parameters between PPG and ECG: >0.99 | 50 (11 F, 39 M) |
| [39] | 2016 | HR, HRV | Contact-based (index finger) | iPhone 4S, Apple Inc., Cupertino CA | Rear camera | 30 | 5 min | • Combination of the steepest slope detection of pulse wave derived from the green channel of the reflected light and its correlation to an optimized pulse wave pattern. | PC: >0.99 (HR), ≥0.90 (HRV) | 68 (28 F, 40 M) | |
| [38] | 2016 | HR, RR | Contact-based (HR) and contactless (RR) | HTC One M8, HTC Corporation, New Taipei City, Taiwan | Front (for RR) and rear (for HR) camera | RR: 320 × 240 (ROI: 49 × 90 abdomen) HR: 176 × 144 (ROI:176 × 72) | 30 (down-sampled to 20 (RR), 25 (HR)) | -- | • Frequency domain analysis of the noncontact video recordings of chest and abdominal motion. | Average of median errors for RR: 1.43%–1.62% between 6 and 60 breaths per minute | 11 (2 F, 9 M) |
| [45] | 2012 | HR | Contactless (face) | iPhone 4, Apple Inc., Cupertino CA | Front camera | 640 × 480 | 30 | 20 s | • Analysis of the raw video signal (green channel) and ICA-decomposed signals of the face in the frequency domain. | Error rate: 1.1% (raw signal), 1.5% (ICA-decomposed signals) | 10 (2 F, 8 M) |
| [49] | 2018 | HR, RR | Contactless (face) | LG G2, LG Electronics Inc., Korea | Rear camera | -- | 30 (down-sampled to 10) | 20 s | • Frequency domain analysis of the color variations in the reflected light (hue) from the face. | PC: 0.9201 (HR) and 0.6575 (RR) | 25 (10 F, 15 M) |
| [36] | 2016 | HR | Contact-based (index finger) | -- | Rear camera | 1920 × 1080 | -- | -- | • Frame-difference based motion detection for improving data quality. • Uses all 3 channels (R, G, B) for PPG extraction. | 20 | |
| • Blood volume flow was observed clearly in the Red channel. | Average accuracy: 98% | ||||||||||
| [50] | 2015 | Pulse, HR, HRV | Contact-based (index finger) | Motorola Moto X, Motorola, Libertyville, IL and Samsung S 5 | Rear camera | 640 × 480 | 30 | 100 s | • Extracts PPG by averaging the Green channel data of the video. • HR is calculated by detecting the consecutive PPG peaks. | PC of pulse and R-R interval from two phone models > 0.95 | 11 |
| [40] | 2014 | HR, NPV | Contact-based (index finger) | iPhone 4S, Apple Inc., Cupertino CA | Rear camera | ROI: 192 × 144 | 30 | 20 s | • HR and NPV were measured in the presence of a controlled motion (6 Hz) of the left hand. • Evaluated the effect of motion artifact (MA) on the PPG in all three color (R, G, B) channels. | Higher SNR for B and G channel PPG in presence of 6Hz MA. PC: HR>0.996 (R, B, G), NPV = 0.79 (G) | 12 (M) |
| [51] | 2014 | HR, HRV | Contact-based (index finger) | Sony Xperia S, Sony Corporation, Tokyo, Japan. | Rear camera | -- | -- | 60 s | • HR was estimated by detecting the consecutive PPG peaks and also the dominant frequency. • Combines several parameters (HR, HRV, Shannon entropy) to detect Atrial fibrillation (AF). | HR error rate: 4.8% AF detection: 97% specificity, 75% sensitivity | |
| [52] | 2012 | HR, HRV | Contact-based (index finger) | iPhone 4s and Motorola Droid, Motorola, Libertyville, IL | Rear camera | ROI: 50 × 50 | 30 (iPhone), 20 (Droid) | 2, 5 min (iPhone, Droid) | • Several ECG parameters were extracted with two different models of smartphone both in supine and tilt position and performed comparative analysis with the data obtained from a standard five lead ECG. | PC: ~ 1.0 (HR), PC for Other ECG parameters: 0.72-1 (Droid), 0.8-1 (iPhone) | 9 (iPhone) 13 (Droid) |
| [44] | 2012 | HR | Contact-based (index finger) | HTC HD2 and Samsung Galaxy S | Rear camera | ROI: 288 × 352 (HTC) 480 × 720 (Samsung) | 25 (HTC) 30 (Samsung) | 6 s | • HR is calculated by detecting the consecutive PPG peaks. | Error: ± 2 bpm | 10 |
| [53] | 2012 | HR | Contact-based (index finger) | Motorola Droid, Motorola, Libertyville, IL | Rear camera | ROI: 176 × 144 | 20 | 5 min | • HR from the PPG signals was obtained at sitting, reading and video gaming by using an Android-based software. | PC: ≥ 0.99 Error: ± 2.1 bpm | 14 (11 F, 3 M) |
| Spatial Domain | Temporal Domain | Frequency Domain | Statistical Domain |
|---|---|---|---|
| Step length | Double support time | Spectral power | Correlation |
| Stride length | Stance time | Peak frequency | Mean |
| Step width | Swing time | Maximum spectral amplitude | standard deviation |
| RMS acceleration | Step time | Covariance | |
| Walking speed | Stride time | energy | |
| Signal vector magnitude (SMV) | Cadence (steps/min) | Kurtosis |
| Ref. | Proposition | Phone | Sensors | Experiment Protocol | n | Method | Performance/Comment |
|---|---|---|---|---|---|---|---|
| [130] | Human activity and gait recognition | Samsung Nexus S | , ω | • Subjects walked ~30 m for each of three different walking speeds • Smartphone in the trouser pocket • Sampling rate: 150 sample/s | 25 | • Each gait cycle was detected and normalized in length. • Several distance metrics between the test and template cycle were calculated as features. • Statistical analysis and machine learning used for recognition. | • Gait recognition accuracy 89.3% with dynamic time warping (DTW) distance metric. • Activity recognition accuracy >99%. |
| [131] | Human activity recognition | Samsung Galaxy S II | , ω | • University of California Irvine (UCI) Human activity recognition (HAR) dataset • Subjects performed an activity twice, with the phone (1) mounted on the belt at the left side (2) placed according to the user’s preference. | 30 | • Feature selection using random forests variable importance measures. • Two-stage continuous HMM for activity recognition. • First and 2nd level for coarse classification and fine classification, respectively. | • Activity (walking, ascending and descending stairs, sitting, standing, and laying) recognition accuracy 91.76%. |
| [132] | Human activity recognition | Samsung Galaxy S II | , ω | • UCI HAR dataset • Activities are: walking, ascending and descending stairs, sitting, standing, and laying | 30 | • A hybrid model based on the fuzzy min-max (FMM) neural network and the classification and regression tree (CART). | • Activity (walking, ascending and descending stairs, sitting, standing, and laying) recognition accuracy 96.52%. |
| [133] | Evaluation of hyperbox (HB) NN for classifying activities | Samsung Galaxy S II | , ω | • UCI HAR dataset • Five subsets of varying sizes (5%, 10%, 20%, 50% and 100% of the dataset) were created for training purpose | 30 | • One HB is assigned for all attributes of a class and has one or more associated neurons for class distribution. • Points falling into (1) only one HB are immediately classified (2) overlapping regions of HBs use the neural outputs for prediction. | • Performance was comparable to SVM, decision tree, KNN and MLP classifier. • Activity (walking, ascending and descending stairs, sitting, standing, and laying) recognition accuracy 75%–87.4% |
| [134] | Human activity recognition | Nexus One, HTC Hero, Motorola Backflip | • Wireless sensor data mining (WISDM) dataset from http://www.cis.fordham.edu/wisdm/dataset.php • Sampling rate: 20 samples/s | 36 | • Extracted 43 features from the mean and standard deviation of acceleration, mean absolute difference, mean resultant acceleration, time between peaks and binned distribution. • A Voting scheme to combine the results from the J48 decision tree, logistic regression and MLP. | • Accuracy > ~97% (walking, jogging, sitting and standing), ~86% (ascending stairs), and ~73% (descending stairs) | |
| [156] | Human activity recognition | iPod Touch | , ω | • Measured activities: sitting, walking, jogging, and ascending and descending stairs at different paces | 16 | • Evaluated different classification models (decision tree, multilayer perception, Naive Bayes, logistic regression, KNN and meta-algorithms such as boosting and bagging) in terms of recognition accuracy. | • Accuracy for sitting, walking, and jogging at different paces: 90.1%–94.1% • Accuracy for ascending and descending stairs: 52.3%–79.4% |
| [135] | Complex activity recognition system | Samsung Galaxy S IV | , ω, P, T, H ( and Gimbal beacons) | • Four smartphones worn on the waist lower back, thigh, and wrist. • Participants performed 19 activities in 45 minutes according to their own order of choice and repetition. | • Conditional random field (CRF) based classification was performed on each device separately. | • Activity recognition accuracy > 80% | |
| • Final recognition was based on the result from the most relevant device to that particular activity. • 19 activities are: walk and run indoors, clean utensil, cook, sit and eat, use - bathroom sink and refrigerator, move from/to indoor to/from outdoor, ascending and descending stairs, stand, lie on the - bed, floor, and, sofa, sit on the bed, floor, sofa, and, toilet. | |||||||
| [136] | A feature selection approach for faster recognition | Samsung Galaxy S II | , ω | • UCI HAR dataset • Activities are: walking, ascending and descending stairs, sitting, standing, and laying. | 30 | • Data segmentation by sliding window and extraction of time and frequency domain features • A hybrid of the filter and the wrapper (FW) methods for feature selection • Performance verified by naïve Bayes and KNN. | • Activity recognition Accuracy, precision and F1-score to 87.8%, 88.0% and 87.7% (with , ω data) • Significant reduction in recognition time. |
| [137] | Algorithm for Human activity recognition | Google NEXUS 4 | , ω | • Subjects performed each activity twice for 30 s each, keeping the device at five different orientations. | 5 | • Employed coordinate transformation and principal component analysis (CT-PCA) on the data to eliminate the effect of orientation variation. • Used several classification models for evaluation. | • Activity (static, walking, running, going upstairs, and going downstairs) recognition accuracy 88.74% with online-independent SVM (OISVM) |
| [138] | A hardware friendly SVM for HAR | Samsung Galaxy S II | , ω | • UCI HAR dataset • Activities are: walking, ascending and descending stairs, sitting, standing, and laying. | 30 | • Standard support vector machine (SVM) with fixed-point arithmetic for computational cost reduction. | Activity recognition accuracy ~89% (similar to standard SVM) |
| [139] | Unsupervised learning for activity recognition | Samsung Galaxy Nexus | , ω | • Smartphone was kept in a pants pocket for measurements • n = 5 activities: walking, running, sitting, standing, and lying down • Each activity was performed for 10 min. | -- | • Experiment 1: known n. k-means, Gaussian mixer model, and average-linkage hierarchical agglomerative clustering (HIER) were used for recognition. • Experiment 2: unknown n. Density-based spatial clustering of applications with noise (DBSCAN) along with three other models used for classification. | • GMM achieved 100% recognition accuracy when n is known • HIER and DBSCAN achieved over 90% recognition accuracy when n is unknown. |
| • DBSCAN requires setting two parameters (eps and minPts) and for other models, n was chosen based on local maxima of the Calin´ ski–Harabasz index (CH). | |||||||
| [140,141] | DNN for Human activity recognition | Samsung Galaxy S II | , ω | • UCI HAR dataset • Activities are: walking, ascending and descending stairs, sitting, standing, and laying. | 30 | • DNN was formed by stacking several convolutional and pooling layers to extract discriminative features. | |
| • Number of layers, number of feature maps, pooling and convolutional filter size were adjusted to maximize test-accuracy by ‘softmax’ classifier. | |||||||
| • Multilayer perceptron for final recognition. | • Accuracy: 94.79%–95.75% | ||||||
| [148] | Human activity recognition | Samsung Galaxy S II and Huawei P20 Pro | , ω | • Smartphone was attached to the waist. • Sampling frequency = 50 Hz • 10,299 samples with Samsung Galaxy SII and 4752 samples with Huawei P20 | 30 | • An Ensemble Extreme learning machine with Gaussian random projection (GRP). • GRP was used for the initialization of input weights of base ELMs. | Activity (sitting, standing, laying, walking, walking upstairs and downstairs) recognition accuracies: 97.35% (Samsung), 98.88% (Huawei) |
| [154] | Human activity recognition | Samsung Galaxy Note I, Motorola Droid, | • Collected 2 weeks of GPS data continuously • Subjects prepared a journal of real-time information about their everyday activities. | 3 | • A fuzzy logic -based approach for classification. | Classification accuracy: ~96% | |
| • Location uncertainty improved by calculating the probabilities of different activities at a single location. | |||||||
| Nokia N900 | GPS | • Recognized activities by a segment aggregation method while adjusting for location uncertainties. | |||||
| [149] | Human activity recognition | Samsung Galaxy S 4 | , ω | • Free walk at a natural pace and run in a straight path, maintain a standing position and minimize additional bodily movement (25 s each). | 1 | • Feature set consisted of linear acceleration, normal acceleration and angular velocity. • Naive Bayes and k-means clustering for classification | Classification accuracy: 85% |
| [150] | Human activity recognition | , ω | • A database of 12 activities (standing, sitting, lying down, walking, ascending and descending stairs, stand-to/from-sit, sit-to/from-lie, stand-to/from-lie, and lie-to/from-stand). | -- | • Extracted features were processed by a kernel principal component analysis (KPCA) and linear discriminant analysis (LDA). | ||
| • Deep belief network (DBN) for classification. | Mean recognition rate: 89.61% and overall accuracy: 95.85% | ||||||
| [151] | Human activity recognition | Huawei Mate 9 | , ω | • Activities were logged approximately 5–8 hours a day for 4 months | 1 | • A six-layer independently recurrent neural network (IndRNN) processed data of different lengths and captured the temporal patterns at different time intervals. | Classification accuracy: ~96% |
| [152] | Human activity recognition | Samsung Galaxy S II | , ω, ф, and P | • UCI HAR dataset • Activities are: walking, ascending and descending stairs, sitting, standing, and laying | 30 | • DNN-based subassembly divides sensor data into various motion states. The transformation subassembly derives the intrinsic correlation between the sensor data and personal health. | • Accuracy: 95.9% with unsupervised feature extraction • 96.5% with manual feature extraction |
| [153] | Walk@Work(W@W)-App for HAR | -- | , ω | • 1 h laboratory protocol and two continuous hours of occupational free-living activities | 17 (10F 7 M) | • Calculated agreement, intra-class correlation coefficients (ICC) and mean differences of sitting time against the inclinometer ActivPAL3TM, and step counts against the SW200 Yamax Digi-Walker pedometer for performance comparison. | • ICC: 0.85 for self-paced walking, 0.80 for active working tasks. • ICC (free-living): 0.99, 0.92 with a difference of 0.5 min and 18 steps for sitting time and stepping, respectively. |
| [155] | Human activity recognition | Samsung Galaxy S II | , ω, ф, | • Four smartphones attached to four body position: right pocket, belt, right arm, and right wrist • Measured activities: walking, running, sitting, standing, walking upstairs and downstairs | 4 | • Data from three types of sensors were evaluated in terms of recognition accuracy using seven classifiers (naïve Bayes, SVM, neural networks, logistic regression, KNN, rule-based classifiers and decision trees). | • Best performance was achieved using both gyroscope and accelerometer data together. • Magnetometer data played little role. |
| [147] | Balance analysis and Audio Bio-Feedback (ABF) system | iPhone 4 | , ω, ф, mic | • Smartphone was mounted on a belt. • Subjects wore the belt on the posterior low back at the level of the L5 vertebra and a pair of earphones, placed arms close to the trunk, stood barefoot, with their eyes closed. | 20 (11F and 9 M) | • Tilt angles and heading were calculated from accelerometer and gyroscope, respectively as well as from the magnetometer. | -- |
| • Kalman filter was used to correctly estimate the rotation angles from the difference between the two previous estimates. • Audio feedback sent through the mic when trunk orientation is above a threshold. | |||||||
| • Subjects kept sway minimum in parallel feet (10 cm apart), tandem stance-positions, and 2 experimental conditions with and without ABF. • Each experimental condition was performed in random order six times, each for 30 s. | |||||||
| [144] | Fall detection and notification system | Lenovo Le-phone | • Smartphone mounted on the waist | -- | • Extracted signal magnitude area (SMA), signal magnitude vector (SMV) and tilt angle from the median filtered accelerometer data. | ||
| • Fall detection with a decision tree-based algorithm. • In case of a fall, a multimedia messaging service (MMS) was sent with time and location info. | • Performance comparison not reported. | ||||||
| [145] | Fall detection | Samsung Galaxy S III | • Collected acceleration data | • Detected a fall if the acceleration along a direction changed at a faster rate than that in normal daily activities. | • Performance comparison not reported. | ||
| [157] | Fall detection, tracking and notification system | -- | • Evaluated the tracking error range at two outdoors and one indoor fall location. • Tests conducted near a school and a subway station at three periods of the day: 7:00–12:00, 12:00–18:00, and 18:00–24:00 to evaluate the accuracy of tracking with mobile obstacles. | 10 | • Calculated accelerometer SMV. • Rapid change in the SMV to a large value indicated a fall. • In case of a fall detected, the GPS location of the smartphone is communicated. • The real-time location tracking system used Google’s 3D mapping services. | • Overall accuracy of the location tracking system: < 9 m. • Larger error range observed between 12:00 and 18:00. • High density of Wi-Fi installations improves location accuracy. | |
| [158] | Fall detection and daily activity recognition | Sony C6002 Xperia Z, Apple iPhone 4s | , ω, ф | • Subjects kept phones in the right, left and front-pockets and fall onto a 15 cm thick cushion. • Activities: four types of fall (forward, backward, toward the left and right) and ADL. | 8 | • Activities were classified using supervised machine learning (SVM, Decision tree, KNN and discriminant analysis) algorithms. • A fall is detected when SMV goes above a threshold value (24.2 ms-2). | • ADL (sitting, standing, walking, laying, walking upstairs and walking downstairs) recognition accuracy 99% with the SVM. |
| [159] | Fall detection algorithm | Sony Z3 | • Smartphone was placed in the front pocket • Subjects performed six activities of daily living and six fall activities | 10 (7 M and 3 F | • Six features (SMV, sum vector excluding gravity magnitude, max and min value of acceleration in gravity vector direction, mean of the absolute derivation of acceleration in gravity vector direction, and gravity vector changing angle) were derived from the accelerometer data. | ||
| • SVM was used to classify fall and non-fall events. | • 96.67% sensitivity, 95% specificity | ||||||
| [160] | Fall detection based on high-level fuzzy petri net (HLFPN) | HTC Desire S | • Smartphone was placed in the thigh pocket • Activities: Falls (forward, backward, vertical, and sideways) and ADLs (walking, jogging, jumping, sitting, and squatting). | 12 (7 F and 5 M) | • Calculated accelerometer SMV and frequency of occurrences from the accelerometer data. | • Fall detection accuracy 90% with HLFPN | |
| • Fuzzy degree was generated by substituting the calculated values into the membership function formulated by the experiment. • Final classification with HLFPN. | |||||||
| [163] | Knee Joint ROM | iPhone 6 | • Dynamic knee extension ROM was measured three times with an interval of 5 min. • Phone was attached to the tibia • An isokinetic dynamometer used to generate and measure the knee motion for validation. | 21 (M) | • A MATLAB program automatically detected the min/max values of knee extension angles from the accelerometer data. • The difference between the min and max values was calculated as the dynamic knee extension ROM. | • Highly correlated (rs = 0.899) and low error (~0.62) wrt the commercial system (Biodex System 4 Pro) • Limits of agreement: −9.1 to 8.8 deg. • ICCE between two methods >0.862 | |
| [164] | Assessment of smartphone apps for measuring knee range of motion | -- | Camera, inclinometer (, ω, ф) | • Five measurements of knee range of motion from each subject by a commercial system, two apps - Goniometer Pro and Dr. Goniometer • Goniometer Pro (by 5fuf5) and Dr. Goniometer (by CDM S.R.L.) were based on smartphone inclinometer and camera, respectively. | 10 (5 F and 5 M) | • Goniometer Pro: attached to the anterior of the thigh proximal to the skin incision, and on the anterior of the distal tibia distal to the skin incision and knee flexion angle () was derived by adding the two measured angles. | • by Dr. Goniometer was clinically identical to from the commercial system, with a mean difference of <1° and 1/50 difference >3° |
| • Dr. Goniometer: calculated by taking pictures from the lateral side of the operated knee with markers virtually placed at the level of the greater trochanter, the knee joint and the ankle joint. | |||||||
| [165] | An app Toss ‘N’ Turn (TNT) for sleep quality monitoring | Any Android phone (version 4.0 +) | , Mic, light sensor, screen proximity sensor | • Subjects installed TNT in the phones and kept it in the bedroom while sleeping and entered a daily sleep diary every morning. • TNT stores sensor data, data about running processes, battery and display screen state in a protected database on the phone. | 27 (19 F and 8 M) | • The time-series sensor data were divided into a series of non-overlapped 10 min windows for data analysis and feature extraction. • Extracted 32, 122 and 198 features associated with sleep detection, daily sleep quality inference, and global sleep quality inference, respectively. | • Classification accuracy: 93.06% (Sleep state), 83.97% (daily sleep quality), 81.48% (overall sleep quality) |
| [166] | Best effort sleep (BES) model for sleep duration monitoring | Light sensor and Mic (+ Phone usage) | • BES tracked six phone usage features (total duration of phone-lock, phone-off, phone charging, phone in darkness, phone in a stationary state and phone in a silent environment) on a daily basis for one week. | 8 | • Model assumption: Sleep duration is a weighted linear combination of six features. • Weights are estimated using a non-negative least-squares regression. | • Sleep duration estimation error range: ± 42 min • Estimated duration is close to commercial wearable systems. | |
| [167] | Sleep monitoring system | iPhone | • Subjects recorded data for at least four consecutive nights using both the ActiGraph, attached to the non-dominant wrist and the smartphone, placed close to the pillow. | 13 (4 F and 9 M) | • Four sleep measures (sleep onset latency (SOL), total sleep time (TST), wake after sleep onset (WASO), and sleep efficiency (SE%)) are extracted from both systems. | • Satisfactory agreement with the ActiGraph for all sleep parameters except for the SOL. | |
| [127] | Contactless Sleep Apnea Detection | Samsung Galaxy S4 | Phone speaker and micro-phone | • The speaker transmits 18–20 kHz sound waves and the microphone senses the reflections. • Total of 296 hours of measurements • Polysomnography (simultaneously done EEG, EMG, airflow, SpO2, electrooculogram) was conducted for performance comparison. | 37 (17 F and 20 M) | • Employed FMCW (frequency modulated continuous wave) transmissions to isolate reflections arriving at different times. • Mapped the human body-specific arrival times of the reflected signals to carrier frequency shift, allowing for extracting the amplitude changes due to breathing. • Reflection patterns of non-breathing movements are different than breathing movements. | • Highly correlated (correlation coefficient of 0.9957, 0.9860, and 0.9533 for central apnea, obstructive apnea and hypopnea, respectively) with the ground truth • Average error (rate of apnea and hypopnea events) < 1.9 events/h. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majumder, S.; Deen, M.J. Smartphone Sensors for Health Monitoring and Diagnosis. Sensors 2019, 19, 2164. https://doi.org/10.3390/s19092164
Majumder S, Deen MJ. Smartphone Sensors for Health Monitoring and Diagnosis. Sensors. 2019; 19(9):2164. https://doi.org/10.3390/s19092164
Chicago/Turabian StyleMajumder, Sumit, and M. Jamal Deen. 2019. "Smartphone Sensors for Health Monitoring and Diagnosis" Sensors 19, no. 9: 2164. https://doi.org/10.3390/s19092164
APA StyleMajumder, S., & Deen, M. J. (2019). Smartphone Sensors for Health Monitoring and Diagnosis. Sensors, 19(9), 2164. https://doi.org/10.3390/s19092164






