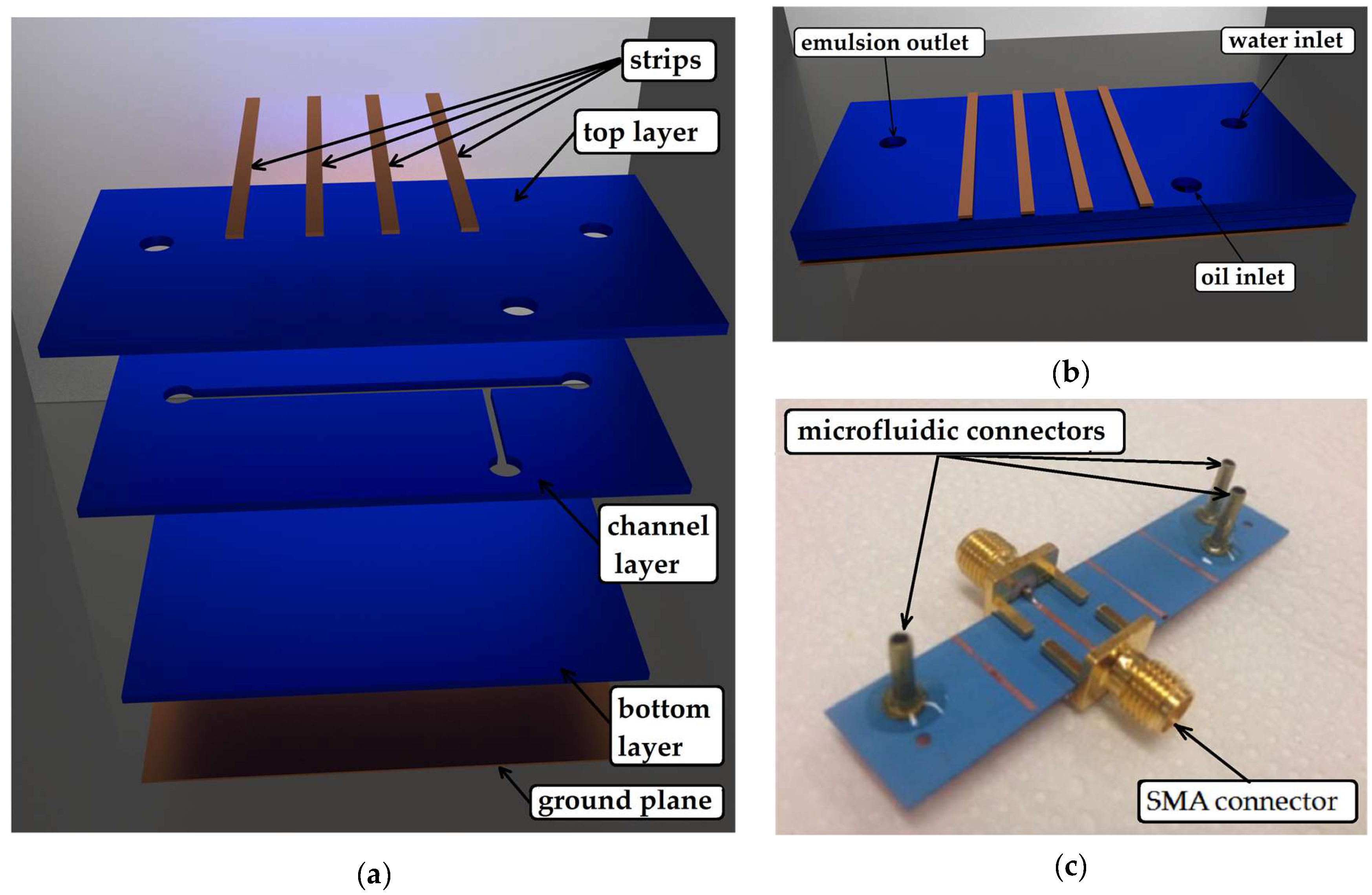
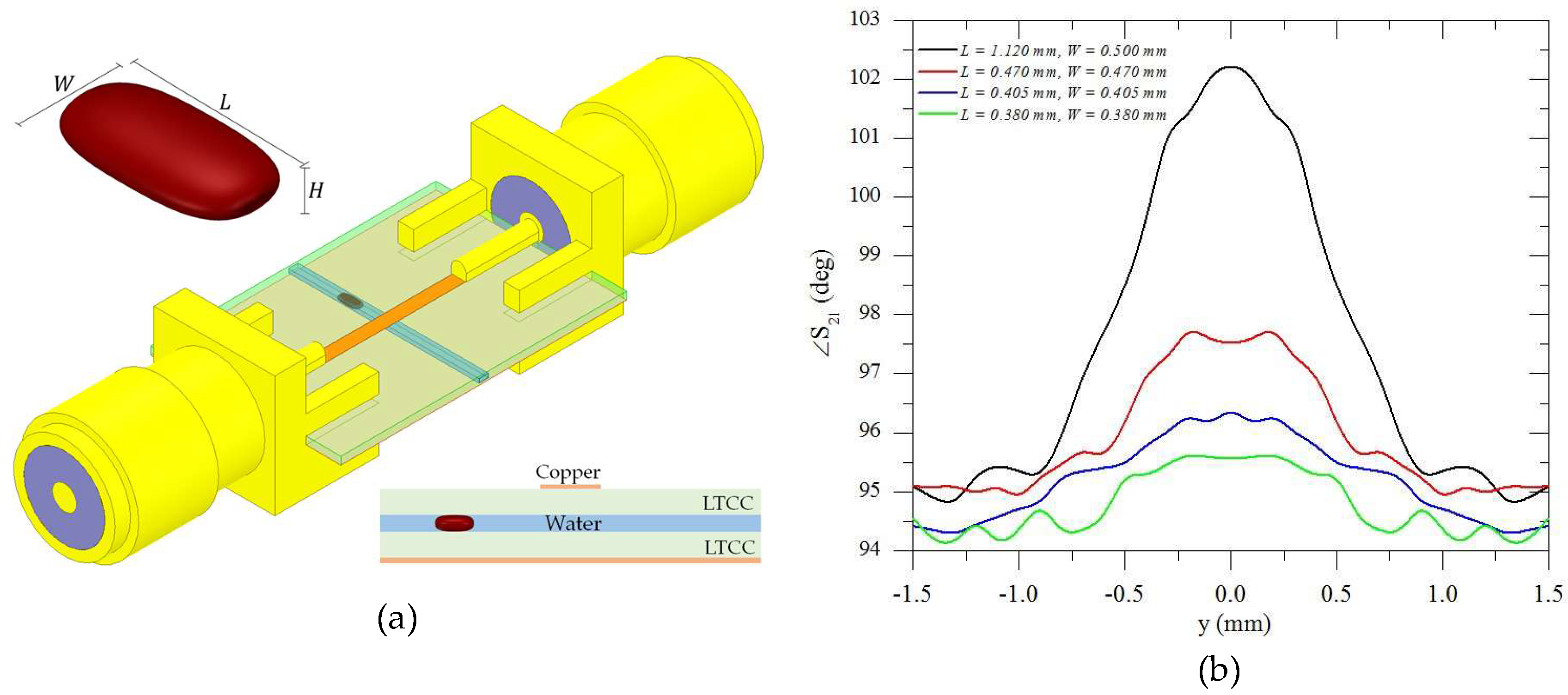
1. Introduction
An emulsion is a liquid dispersion composed of single or multiple droplets suspended in an immiscible continuous phase. These droplets are frequently used as carriers for different types of liquid or solids, including drugs and cells for a breadth of applications. In this way, emulsions have been widely used in the pharmaceutical, chemical and food industries, as well as in material science and biotechnological areas, in order to improve the quality and bioavailability of chemicals, as well as to guarantee protection and encapsulation of drugs providing controlled release [
1]. Microfluidic devices have shown to be a promising means for producing emulsion essentially due to the high degree of control over the droplet uniformity in size. By using these devices in parallel, the throughput can be increased significantly, making it comparable to batch production in chemical reactors.
Micro-channels fabricated using different techniques have been successfully used to generate, in a very controlled manner, droplets aimed towards different applications, such as encapsulation of flavors and fragrances [
2], vitamins [
3] and nanoparticles [
4] in the pharmaceutical and food industries. In biotechnology, cells [
5,
6] and even microorganisms such as algae [
7], bacteria and viruses [
8] are encapsulated inside droplets for studying these organisms in a controlled environment.
With microfluidic devices, emulsions are produced in a continuous way and with very good control over droplet sizes. These parameters lead to emulsions with a high degree of homogeneity, as well as controlled physical and chemical characteristics. This is combined with other advantages of microfluidic processing, such as low reagent and energy consumption. Droplet characterization is a crucial step for the success of any application concerning emulsions. Information like average size, size homogeneity, composition and concentration, droplet velocity and frequency of droplets formation are key parameters. However, this characterization requires expensive equipment and is conducted sometime after production, usually by using high-speed cameras [
9,
10,
11].
Alternative techniques have been proposed in order to have a real time characterization, allowing faster and more precise decisions during the production of droplets for different applications [
12,
13]. In this respect, many microfluidic devices have been proposed, where micro-channels are integrated with some tool for detection, in configurations that can be capacitive- [
14], impedance- [
15] or optical-based [
16], among others. Methods relying on the electrochemical properties of droplets are a common way to detect many properties. However, not all applications involve materials that have electroactive properties [
17,
18]. Optical detection has also been applied to characterize droplets. Usually, high-speed camera images are used, and optical fibers combined with lasers and photo-detectors are applied to acquire information [
12,
13]. These techniques usually require a transparent substrate or the cumbersome integration of optical fibers, which could lead to leakage among other problems.
Recently, techniques that use electromagnetic (EM) waves in the Radiofrequency (RF) and microwave region of the frequency spectrum have been used to perform a label-free detection and measurement of droplet dimensions [
19,
20]. In these works, microwave resonators [
21] with fundamental resonant frequency at 3 GHz were fabricated using microstrip transmission lines on glass substrates. Microfluidic channels were then fabricated on top of the resonators using polydimethylsiloxane (PDMS) and were used to generate droplets. The difference in permittivity of the liquid inside the droplet with respect to the surrounding fluid changes the resonant frequency of the resonator allowing the detection and measurement of the droplets.
In this work we proposed an alternative EM wave-based method where microstrip transmission lines were used in conjunction with microfluidic devices in order to detect as well as measure droplet dimensions directly from the phase shift induced by the local variation of the relative permittivity (εr) due to the presence of such droplets. Being originally designed for RF and microwave applications, low temperature co-fired ceramic (LTCC) substrate (εr ≃ 7.8) was the platform we chose to fabricate and integrate the microfluidic channels and the microstrip transmission lines used for droplet detection.
3. Results
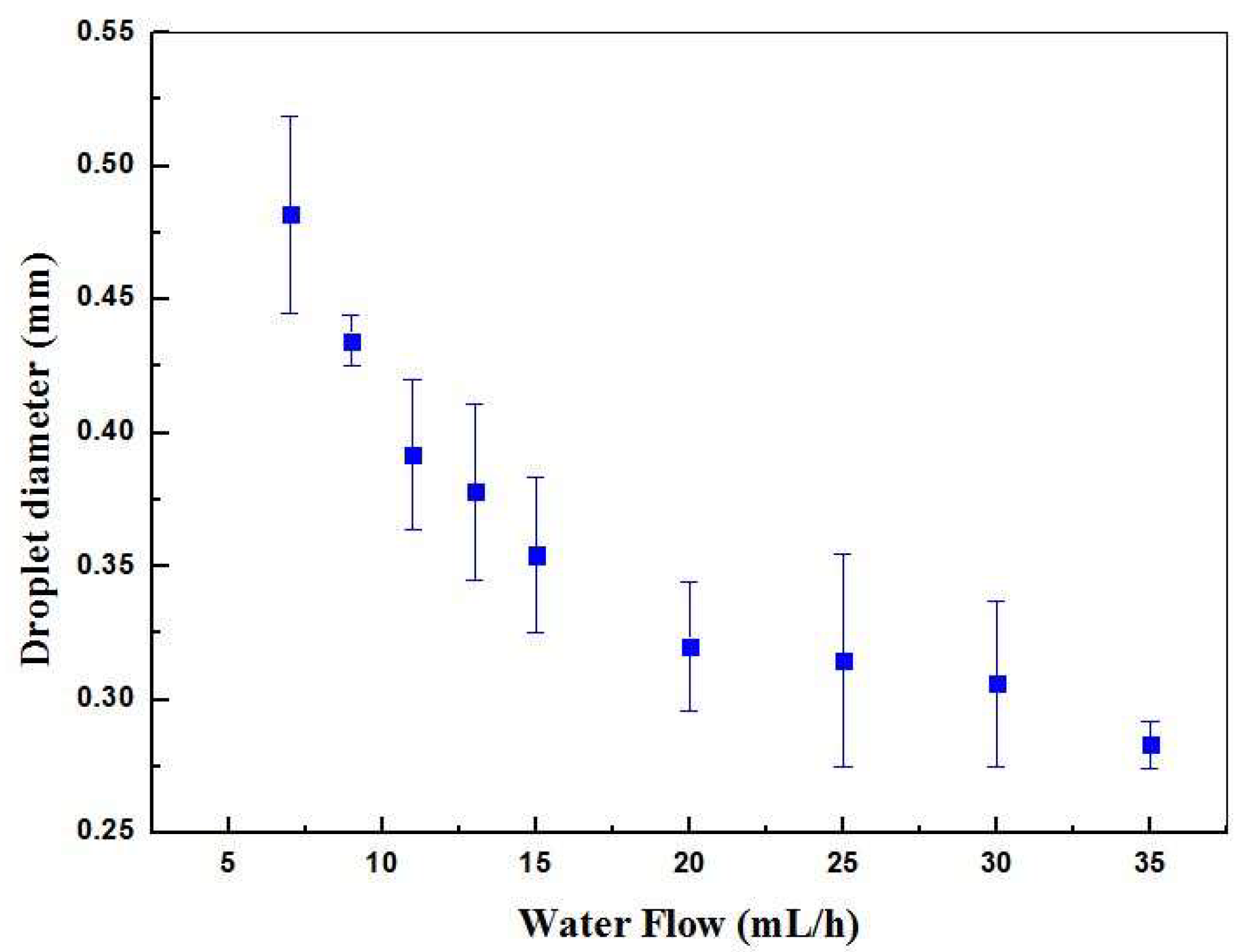
The optical method was applied to measure the oil droplets diameter using a microscope and image analysis software as a function of the water flow rate, shown in
Figure 4. As expected, the droplet size decreased with the increase of the water flow. Droplets with diameter as large as 480 μm were obtained for the lowest water flow value of 7 mL/h. The diameter was largely dependent on the dimensions of the channels that formed the T-junction of the microfluidic device. This could be seen from the fact that the largest droplet diameter (480 μm) corresponded approximately to the width of the microchannel that contains the continuous phase (500 μm). This was expected and rather common for single emulsions produced using microchannels.
As the water flow rate increased to 35 mL/h, the droplets diameter decreased to 310 µm. The maximum flow ratio between phases, i.e., the ratio between water phase and oil phase flows (Q
water/Q
oil), was set to 70. It was observed that any flow ratios larger than this value induced the oil phase to be blocked, preventing the formation of droplets in the device. Below this ratio, by increasing the water flow rate, it was also possible to observe an increase in the number of droplets produced per second.
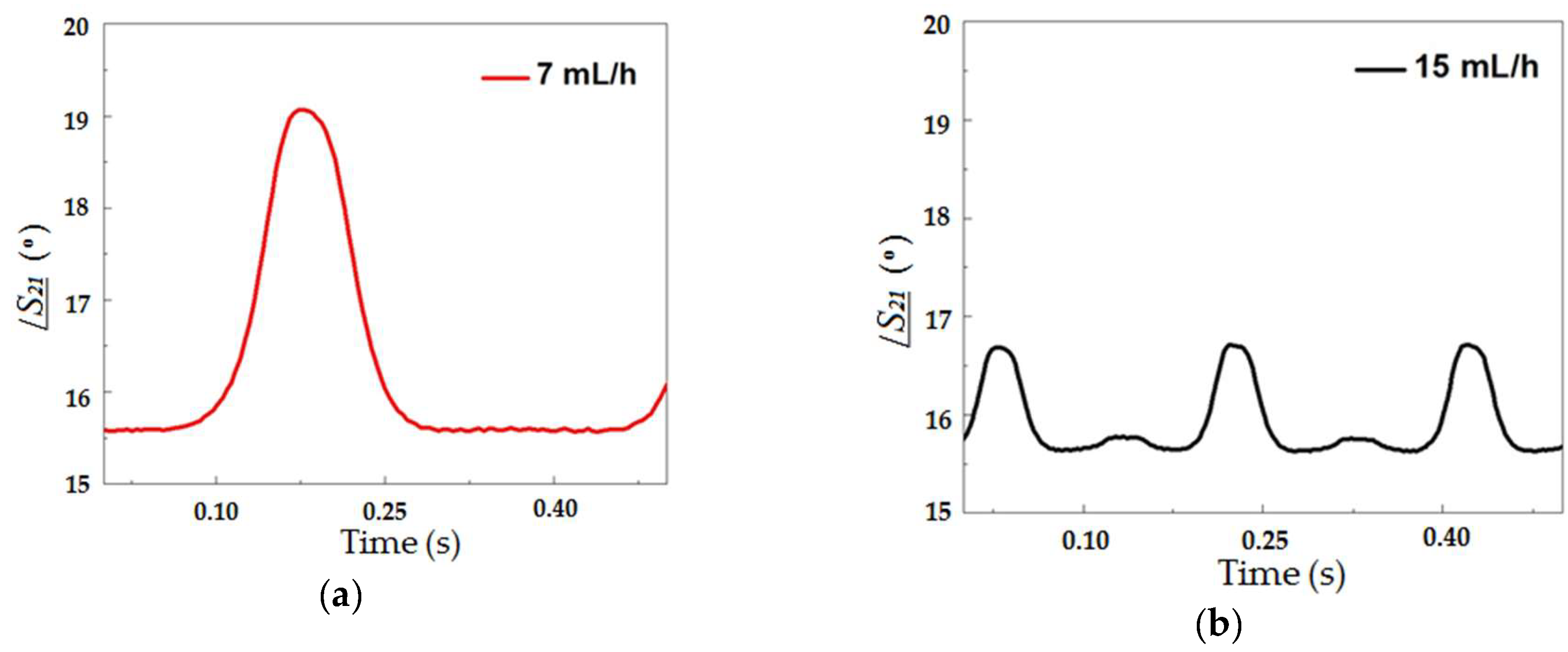
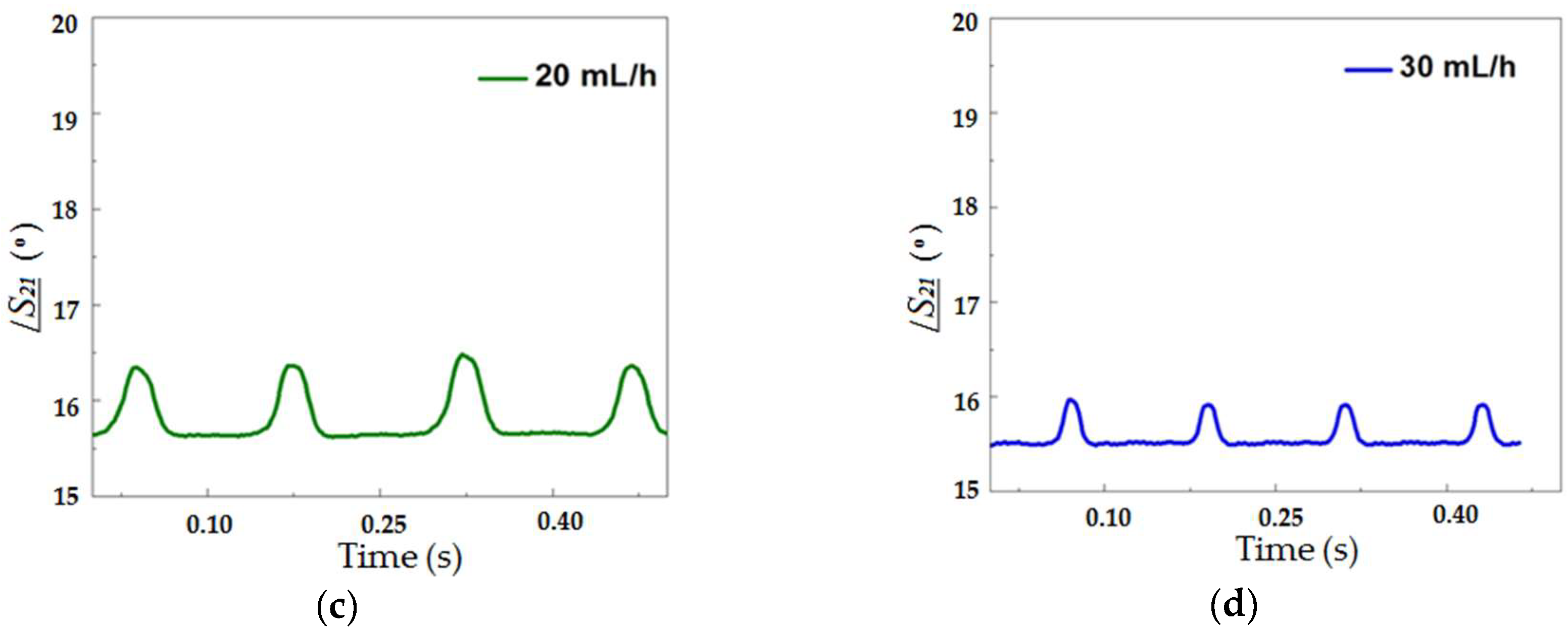
Figure 5 illustrates this tendency through images of sequences of droplets with different diameters obtained for four distinct water flow rates (7, 15, 20 and 30 mL/h). The droplet generation rates were 2.6, 5.1, 7.1 and 8.0 droplets/s for continuous phase flow rates of 7, 15, 20 and 30 mL/h, respectively.
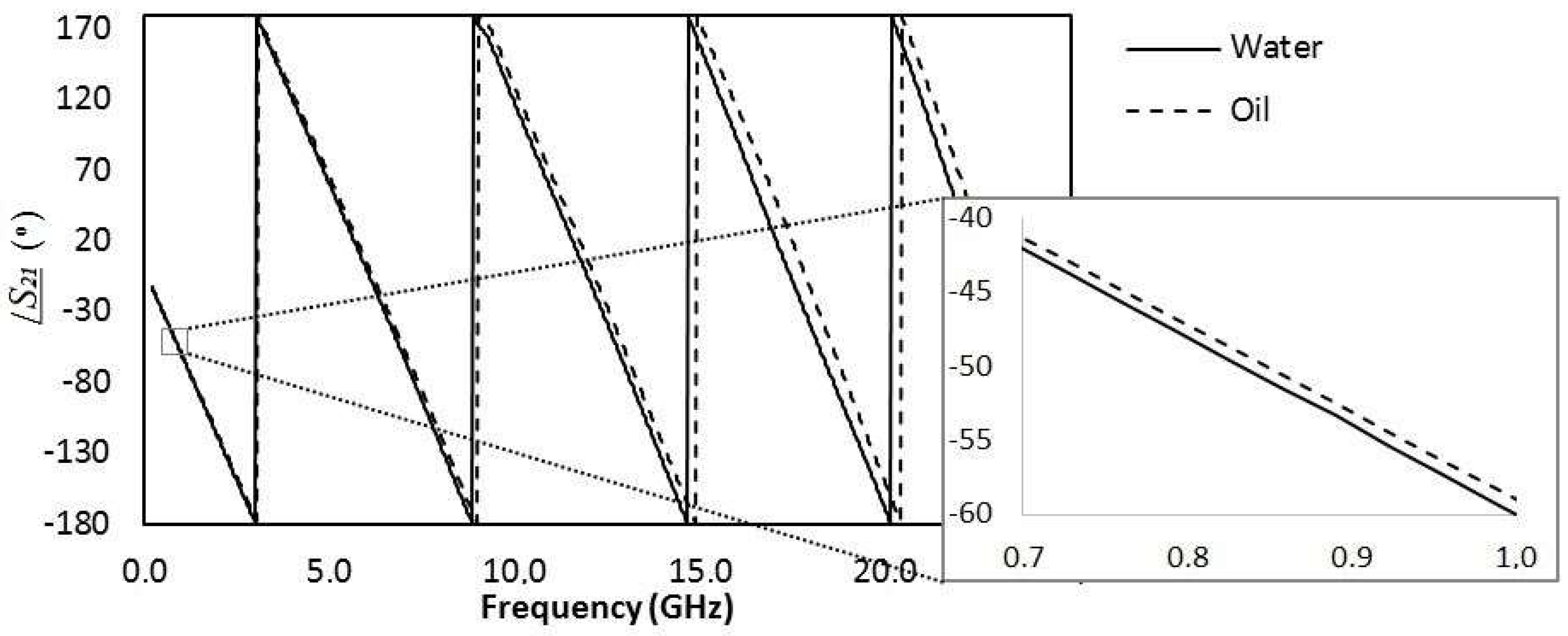
Considering the method using microwave frequencies, first the transmission line was measured with only water, then only oil flowing through the microchannel. The phase response of the transmitted signal (S
21) for each case is shown in
Figure 6, showing that indeed there was a phase difference when oil or water flows. This figure corroborates the statement explained in
Section 2.1 that the higher the frequency, the larger the phase shift between two liquids with different relative permittivities passing through the microchannel. Therefore, we used the higher measured frequency (22 GHz) to provide the information about the droplets, although the phase shift could be detected in lower frequencies such as 1 GHz (inset of
Figure 6). Using another frequency would change the “calibration” of the method that correlated the measured phase shift to the dimension of the droplet. Lower frequencies provide smaller phase shift, thus smaller droplets would be harder to measure. Similarly, higher frequencies provide larger phase shift, allowing one to detect and measure even smaller droplets than the ones detected in this article.
Then, the phase of the transmitted signal was measured as a function of time as droplets flowed through the microchannel across the microstrip line.
Figure 7 shows this response for a continuous phase flow of 7 (a), 15 (b), 20 (c) and 30 (d) mL/h at 22 GHz. This frequency was chosen for all the results for being the highest valid measured frequency that was limited by the operating range of the connector used. The higher the frequency, the larger the phase shift, although the phase shift could be detected in lower frequencies such as 1 GHz (inset of
Figure 6).
As it can be seen in
Figure 7, as the droplet crossed the transmission line, the larger the fraction of the droplet underneath the microstrip line, the larger the phase shift measured. This type of curve, aside from allowing one to detect the presence of the droplet at the given position, i.e., the crossing between the fluidic channel and the microstrip, allows for the measurement of the droplet size. The size of the droplets in the emulsion was obtained from the peak value of the curves presented in
Figure 7.
Each peak in
/S21 shown in
Figure 7 corresponds to the presence of a single droplet under the microstrip line. We believe that the difference observed when comparing the maximum phase shifts seen in
Figure 3b, obtained with numerical modelling, and those of
Figure 7 was due to the fact that the relative permittivity of the liquids are strongly frequency-dependent, and the values used in the simulation
and
may not correspond to the actual ones at 22GHz, since the permittivities found in the literature are measured at lower frequencies. The main point that can be observed when comparing the curves in
Figure 7a–d is that the peak value of
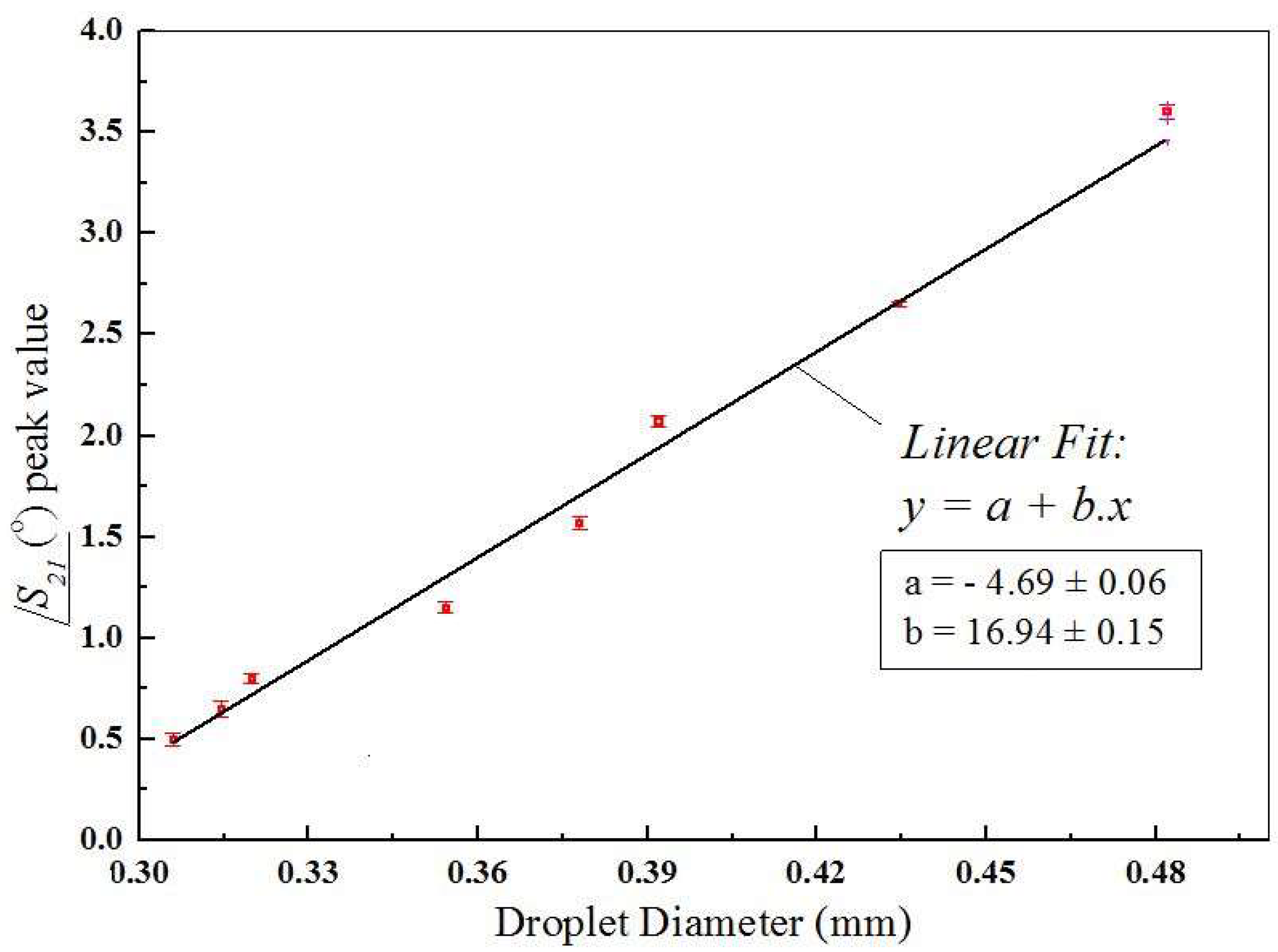
/S21 depends on the continuous phase flow rate and is directly proportional to the droplet diameter. Several curves similar to these were measured and considering the dimensions obtained from the optical method, it resulted in the graph presented in
Figure 8, which shows the peaks in
/S21 as a function of the droplet diameter. At the frequency of 22GHz, the peak value of
/S21 induced for droplets with 310 µm in diameter was 0.5°. For droplets with 480 µm diameter this peak was 3.7°. In this graph the linear relation between
/S21 and droplet diameter is clearly observed. This linear relation was observed because the droplet diameters were smaller than the width of the microchannel (500 µm). As the droplets become larger than this value, they will tend to conform to the cross-section dimensions of the microchannel and get elongated in the direction of fluid flow. Though we are not able to generate such larger droplets with our device, these droplets would also be measurable by this device because the width of the curves under the peaks in
Figure 7 would become larger as the size of the droplets increase. In that case, a relationship between the peak width and droplet diameter could be found, even if it were not linear as the one seen in
Figure 8. In experiments where overlapping droplets are generated, which was not the case in the present work, additional studies would have to be performed in order to find out how the phase and width of curves similar to the ones shown in
Figure 7 could be used to distinguish these overlapping droplets.
4. Discussion
The microfluidic platform proposed here for droplet detection and size measurement has some key advantages over other platforms used for a similar purpose. A first point that is readily seen in
Figure 8 is the linear relationship between the phase of the
S21 parameter and the droplet diameter. This was applicable for the range of droplet dimensions studied in this work only. More important than the linear behavior is the fact that the platform proposed here could provide a higher sensibility in terms of phase variation induced by changes in permittivity, when compared to similar platforms [
19,
20]. The main reason for this is the unique feature, which has to do with the relative position of the microchannel with respect to the microstrip line. Specifically, the microchannel was placed inside the transmission line, between the top metal track and the ground plane.
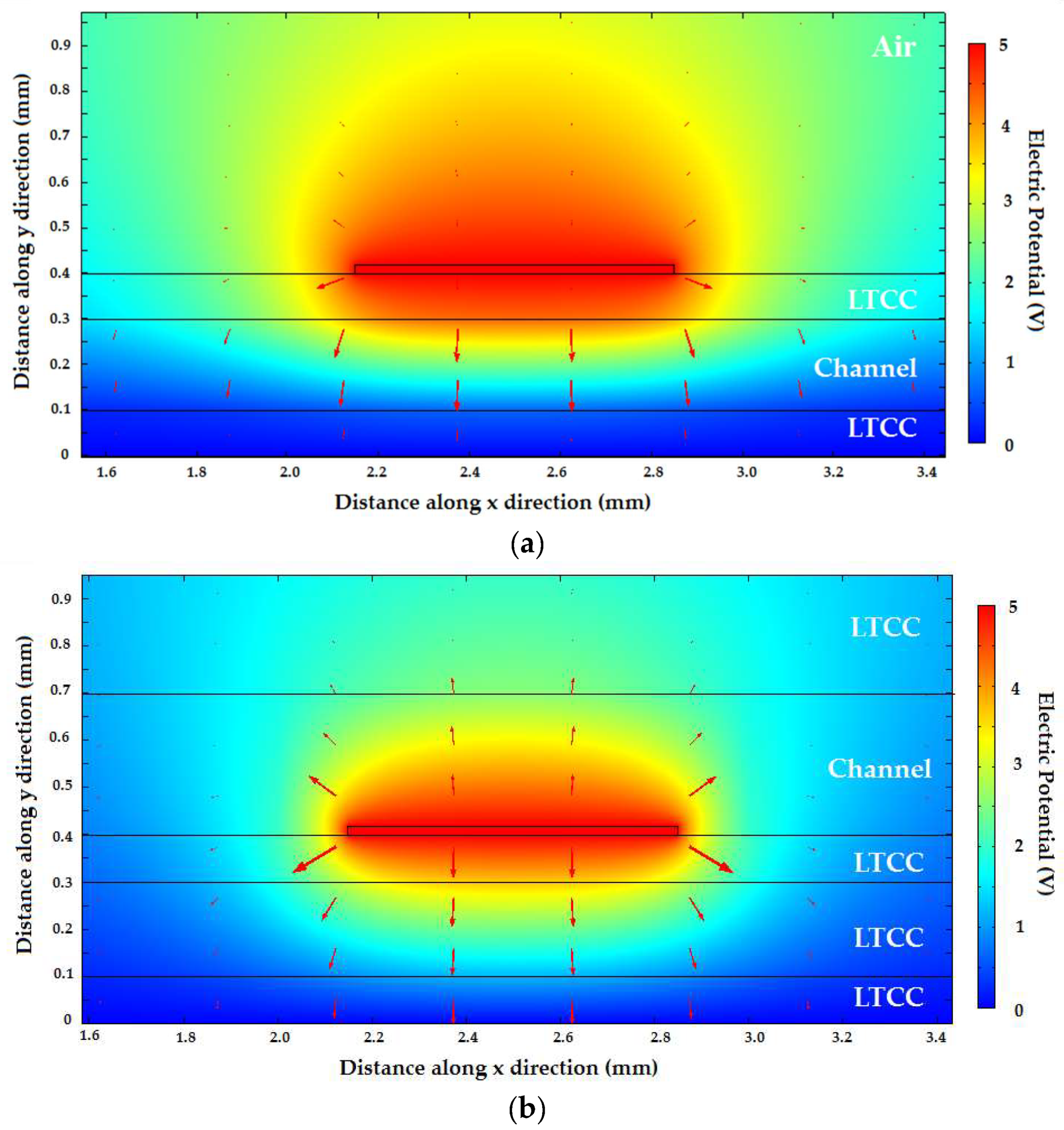
To obtain a better grasp of how much the phase was affected by variation in permittivity of the liquids surrounding the transmission line,
Figure 9a,b shows plots of the electric potential (V) and the electric field (red arrows) of the fundamental mode supported by the transmission lines with two different configurations. These quantities were numerically calculated using Mode Analysis based on the Finite Element Method (FEM). The dimensions and materials used for each layer in the simulation are shown in the Figure. In
Figure 9a, the microchannel was placed between the strip and the ground plane. The height of the microchannel was 200 µm, and it was placed in between two 100 µm-thick LTCC sheets. The material above the microstrip was also green ceramic. In
Figure 9b, on the other hand, the dielectric substrate between the strip and the ground plane was solid LTCC, and the microchannel was placed on top of the microstrip line. In these figures, the microchannel was filled with oil (ε
r = 2.2), but we also analyzed these same structures filled with water, obtaining qualitatively similar results.
It can readily be seen that the intensity of the electric field vector inside the microchannel in
Figure 9a is greater than its intensity on top of the transmission line. This is because the channel is in between the metals in this transmission line. By integrating the electric field intensity in the different regions, we calculated the percentage of total Electric Field E-Field of the Transverse Electromagnetic TEM mode which permeated the microchannel in this configuration, which turned out to be 36.7% for the described dimensions. For the configuration of
Figure 9b, which is similar to the one used in [
19] and [
20], the electric field inside the microchannel clearly had a smaller intensity than that of the field underneath the top metal track. For this configuration, only 17% of the field was inside the microchannel in the best case, i.e., using a microchannel with overestimated dimensions of 300 µm in height.
We also estimated the amount of change in phase constant for two scenarios: when the microchannel was filled with water and when it was filled with oil. For the structure proposed in this work (
Figure 9a) the phase constant variation was Δβ = β
water − β
oil = 378 rad/m. For the structure with the microchannel on top (
Figure 9b) the phase constant variation was Δβ = β
water − β
oil = 259 rad/m. This meant that for the same interaction length of the transmission line and liquid phase, a phase variation 46% higher could be obtained with the microchannel between the strip and the ground plane of the microstrip line.
The intent here was not to directly compare our platform with that used in the aforementioned works, since those works do not describe the dimensions of their devices in detail and also, the substrate material is glass with a top layer which consists of PDMS polymer, as opposed to LTCC. The intent here was to compare the effect of placing the microchannel inside or on top of transmission lines made with the same material platform. A larger fraction of the modal field interacting with the material inside the microchannels translated into a larger induced phase variation caused by change in the permittivity in this region. The key parameter here was the phase constant β. Variations in the permittivity of some portion of the transmission line led to variations in β. The magnitude of the latter can be greater depending on the fraction of the mode field inside the region where the change in permittivity takes place. This is similar to what happens in optical evanescent field-based sensors, where the extent of the evanescent field determines how much β will vary in response to a given change in the refractive index.
The main reason for this phase difference is the fact that, when the electromagnetic mode propagates in a given medium, its phase constant is given by:
where ω is the angular frequency, μ is the magnetic permeability, which is equal to the magnetic permeability of vacuum (
) since all media are non-magnetic, and ε is the effective electric permittivity, which depends on the medium surrounding the mode and is very different for water
) and for oil
. The permittivity of free space is
.
The phase shift when the electromagnetic mode propagates through a given medium is given by:
where
l is the length of interaction of the electromagnetic wave with a given medium in which the mode propagates with a given phase constant β. As seen in Equations (1,2), if an electromagnetic mode, such as the TEM mode of the microstrip line, propagates through media with different ε, such as oil and water, the phase shift is different, which is the reason for the results observed in
Figure 3b and in
Figure 7.
Aside from the higher sensibility of the structure proposed here, the microchannels were also fabricated on the LTCC substrate, which has many advantages over other materials, such as PDMS soft polymer, which is prone to being attacked by different chemicals and cannot be used for applications which required temperatures higher than 150 °C.
Although the proposed design allowed us to measure droplet size directly from the phase of the microwave signal without the need for a resonator, such a device could lead to a dramatic improvement. This is because resonators allow for small phase variation to be translated into significant amplitude variation. As shown in Reference [
20], this could lead to detectors with much larger throughput and even to be able to distinguish the content inside droplets used to encapsulate different substances. Therefore, the next logical step is to use the microstrip line/microchannel configuration proposed here to make devices such as resonators and interferometers. It is very likely that the phase sensibility could translate into a larger variation of the resonant frequencies, although this is beyond the scope of the present work. Finally, by using different microstrip lines to detect the time in which a given droplet crosses these different detectors, it would be possible to determine the speed of the droplets. This could work even for self-propelled droplets such as the metal droplets described in Reference [
24]. Although this needs to be studied carefully in future works, the scheme proposed here could also be applied to the detection and measurement of liquid metal droplets. Lastly, since we were able to detect and measure the size of droplets in an emulsion environment, these detectors could be applied in conjunction with switching arrays devised to open specific outlets to enable the separation of droplets with different sizes. This application can be pursued in future works as well.