A Low-Cost Micro-Volume Nephelometric System for Quantitative Immunoagglutination Assays
Abstract
1. Introduction
2. Materials and Methods
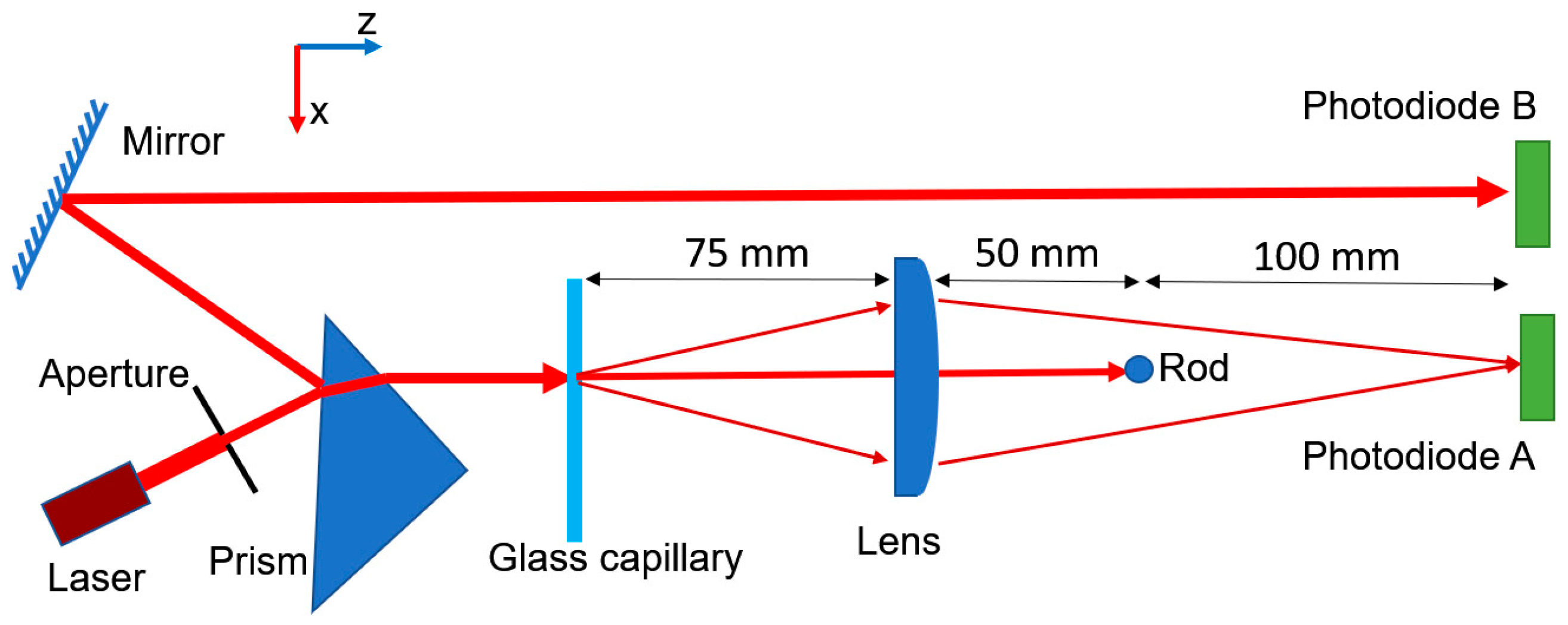
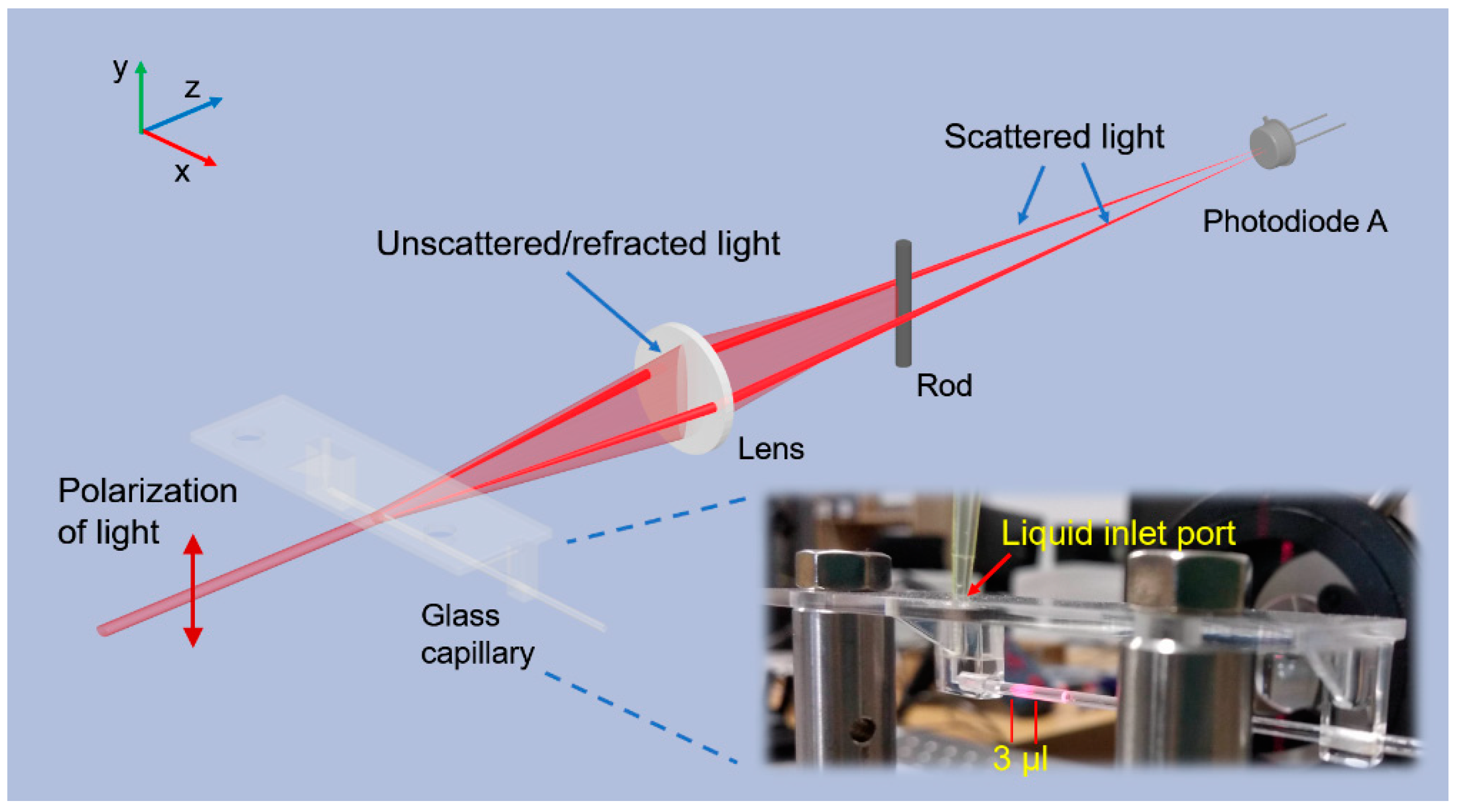
2.1. Nephelometric System Setup
2.2. Immunoagglutination Assay for CRP Detection
3. Results and Discussion
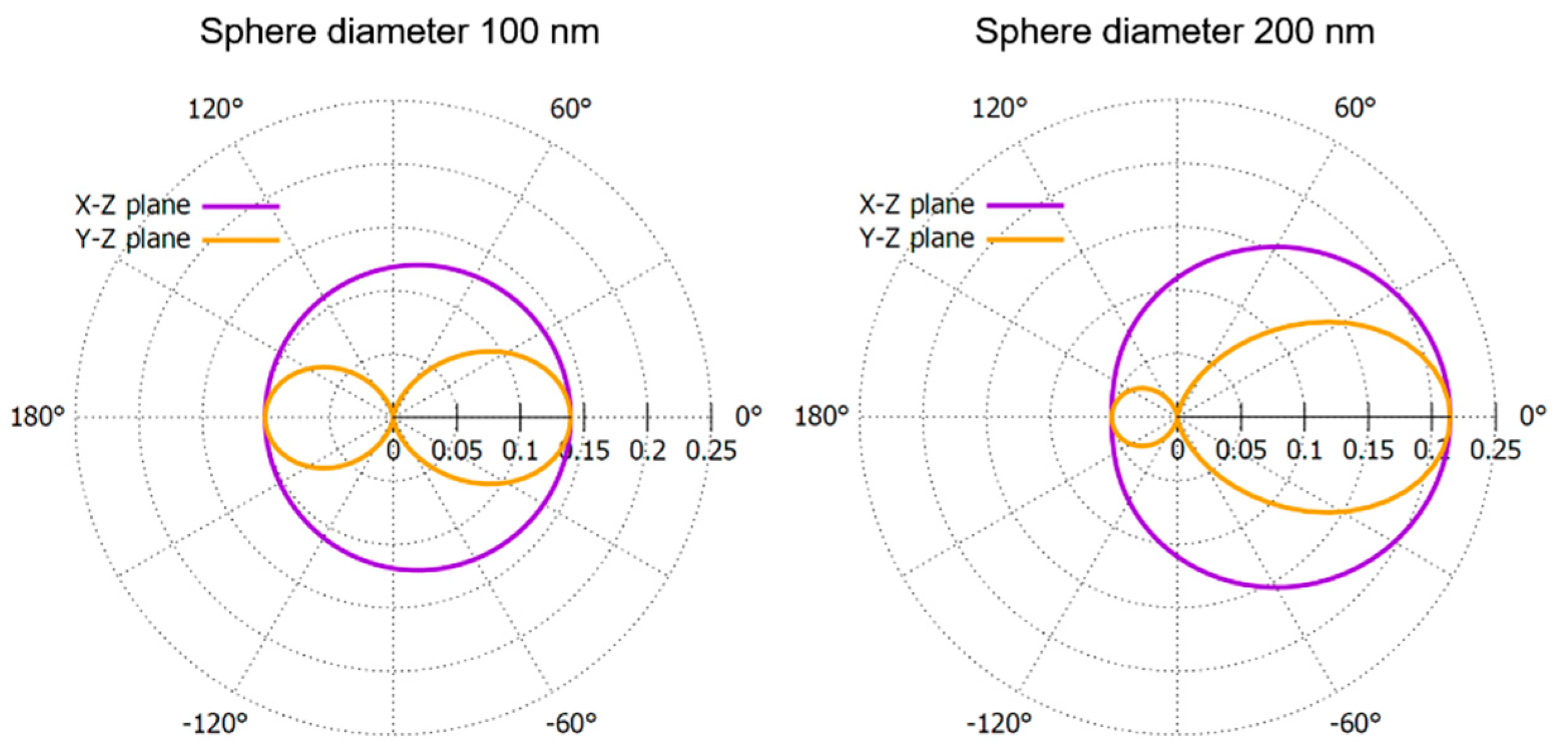
3.1. System Design
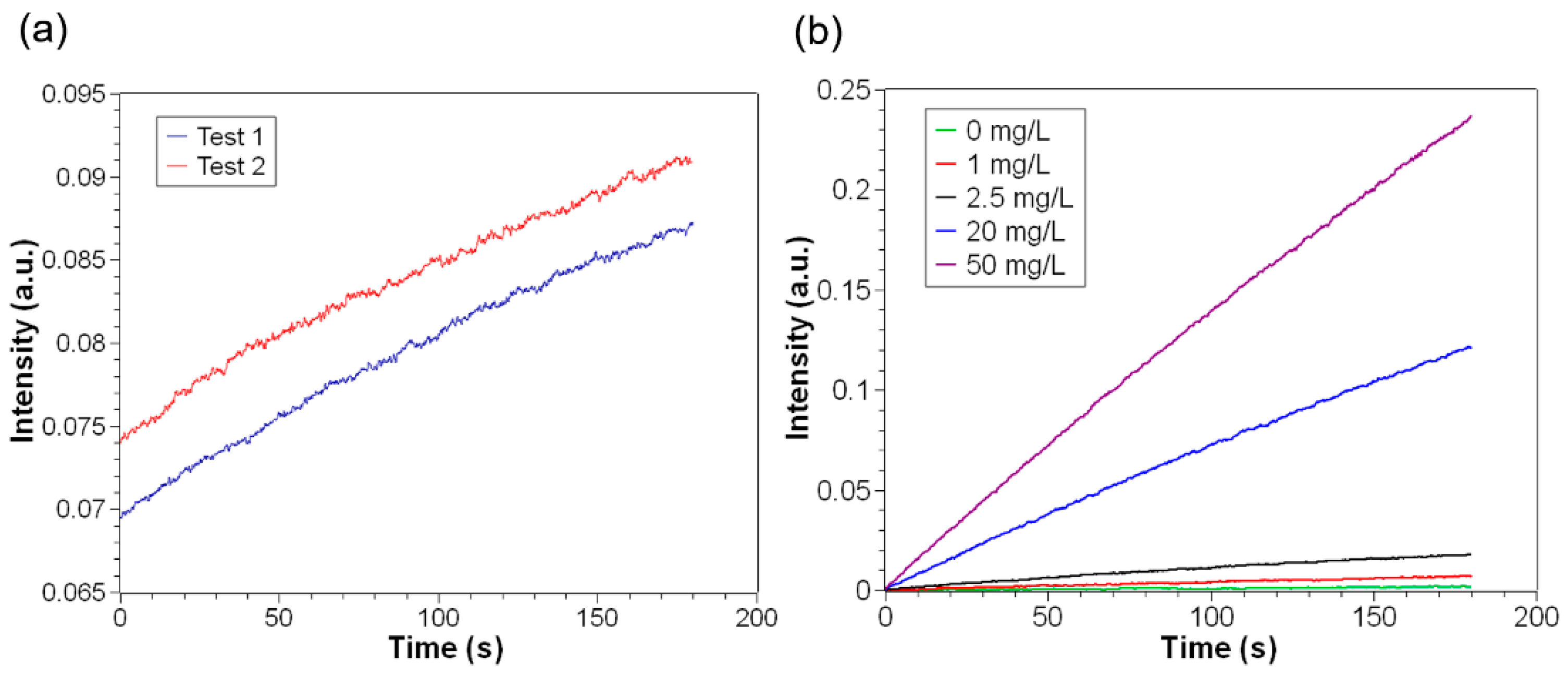
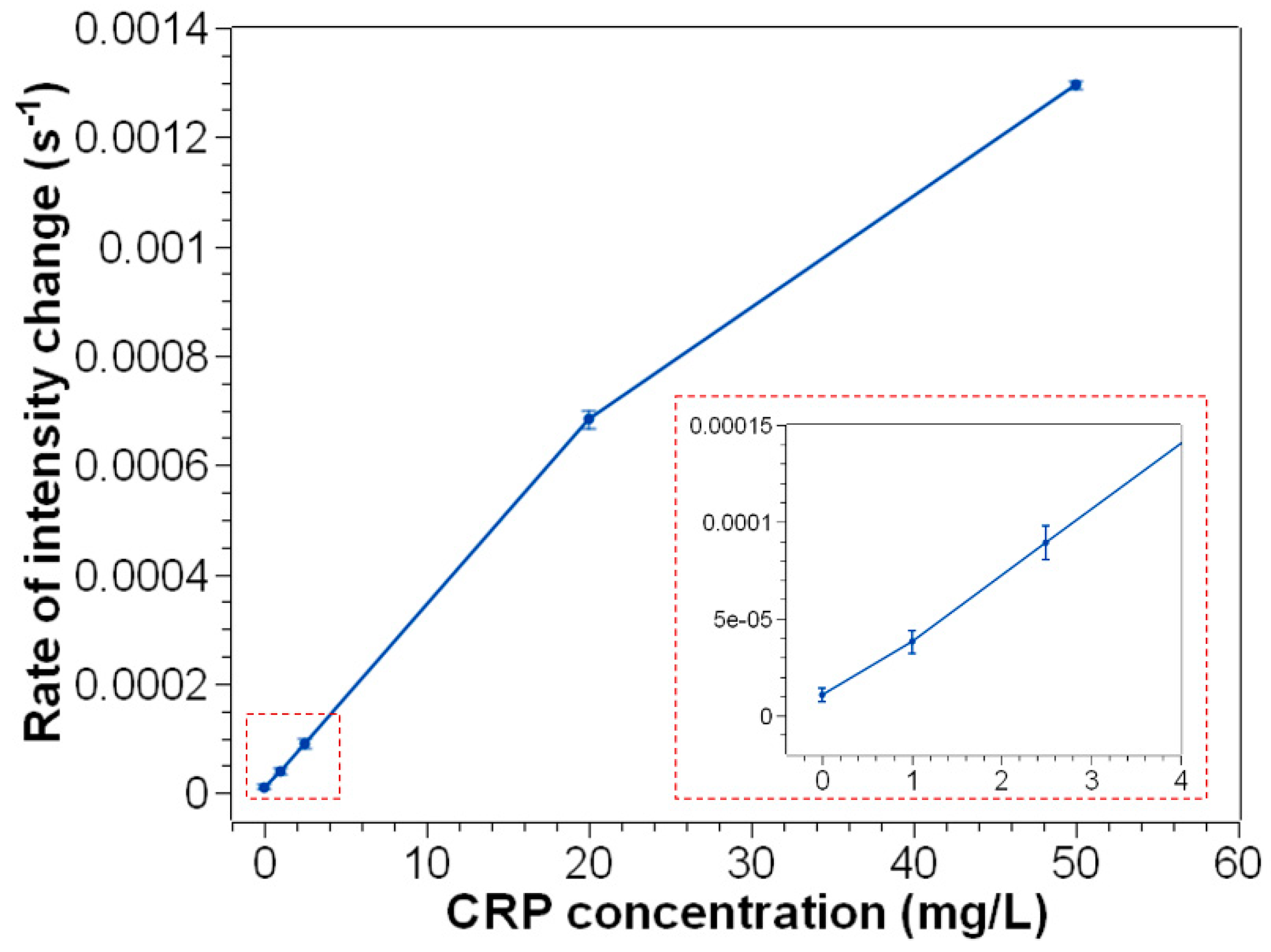
3.2. Quantitative CRP Detection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Darwish, I.A. Immunoassay methods and their applications in pharmaceutical analysis: Basic methodology and recent advances. Int. J. Biomed. Sci. 2006, 2, 217. [Google Scholar] [PubMed]
- Vashist, S.K.; Luong, J.H.T. Immunoassays: An Overview. In Handbook of Immunoassay Technologies; Vashist, S.K., Luong, J.H.T., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 1–18. [Google Scholar]
- Ju, H.; Lai, G.; Yan, F. Immunosensing for Detection of Protein Biomarkers; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Hasan, M.M.; Alam, M.W.; Wahid, K.A.; Miah, S.; Lukong, K.E. A Low-Cost Digital Microscope with Real-Time Fluorescent Imaging Capability. PLoS ONE 2016, 11, e0167863. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, S.; Boehm, S.A.; Chau, K.J. Automated, portable, low-cost bright-field and fluorescence microscope with autofocus and autoscanning capabilities. Appl. Opt. 2012, 51, 2581–2588. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, I.; Matute, T.; Herrera, R.; Keymer, J.; Marzullo, T.; Rudge, T.; Federici, F. Low cost and open source multi-fluorescence imaging system for teaching and research in biology and bioengineering. PLoS ONE 2017, 12, e0187163. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Nordman, E.; Johnson, M.; Oldham, M. A low-cost, high-performance system for fluorescence lateral flow assays. Biosensors 2013, 3, 360–373. [Google Scholar] [CrossRef] [PubMed]
- Walczak, R.; Kubicki, W.; Dziuban, J. Low cost fluorescence detection using a CCD array and image processing for on-chip gel electrophoresis. Sens. Actuators B Chem. 2017, 240, 46–54. [Google Scholar] [CrossRef]
- Koczula, K.M.; Gallotta, A. Lateral flow assays. Essays Biochem. 2016, 60, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Burtis, C.A.; Bruns, D.E. Tietz Fundamentals of Clinical Chemistry and Molecular Diagnostics; Elsevier Health Sciences: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Molina-Bolívar, J.A.; Galisteo-González, F. Latex immunoagglutination assays. J. Macromol. Sci. Polym. Rev. 2005, 45, 59–98. [Google Scholar] [CrossRef]
- Price, C.P. Agglutination Techniques for Detecting Antigen–Antibody Reactions. In eLS; American Cancer Society: Atlanta, GA, USA, 2001. [Google Scholar]
- Xia, Y.; Shen, H.; Zhu, Y.; Xu, H.; Li, Z.; Si, J. A sensitive three monoclonal antibodies based automatic latex particle-enhanced turbidimetric immunoassay for Golgi protein 73 detection. Sci. Rep. 2017, 7, 40090. [Google Scholar] [CrossRef] [PubMed]
- Guaita, S.; Simo, J.M.; Ferré, N.; Joven, J.; Camps, J. Evaluation of a particle-enhanced turbidimetric immunoassay for the measurement of immunoglobulin E in an ILab 900 Analyzer. Clin. Chem. 1999, 45, 1557–1561. [Google Scholar] [PubMed]
- Han, Y.; Wu, H.; Liu, F.; Cheng, G.; Zhe, J. A multiplexed immunoaggregation biomarker assay using a two-stage micro resistive pulse sensor. Biomicrofluidics 2016, 10, 024109. [Google Scholar] [CrossRef] [PubMed]
- Rajan, N.K.; Rajauria, S.; Ray, T.; Pennathur, S.; Cleland, A.N. A simple microfluidic aggregation analyzer for the specific, sensitive and multiplexed quantification of proteins in a serum environment. Biosens. Bioelectron. 2016, 77, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Kilpatrick, P.K.; Melvin, E.; Velev, O.D. On-chip latex agglutination immunoassay readout by electrochemical impedance spectroscopy. Lab. Chip 2012, 12, 4279–4286. [Google Scholar] [CrossRef] [PubMed]
- Fronczek, C.F.; You, D.J.; Yoon, J.-Y. Single-pipetting microfluidic assay device for rapid detection of Salmonella from poultry package. Biosens. Bioelectron. 2013, 40, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Laiwattanapaisal, W.; Songjaroen, T.; Maturos, T.; Lomas, T.; Sappat, A.; Tuantranont, A. On-chip immunoassay for determination of urinary albumin. Sensors 2009, 9, 10066–10079. [Google Scholar] [CrossRef] [PubMed]
- Lucas, L.J.; Han, J.-H.; Chesler, J.; Yoon, J.-Y. Latex immunoagglutination assay for a vasculitis marker in a microfluidic device using static light scattering detection. Biosens. Bioelectron. 2007, 22, 2216–2222. [Google Scholar] [CrossRef] [PubMed]
- Montagne, P.; Varcin, P.; Cuilliere, M.L.; Duheille, J. Microparticle-enhanced nephelometric immunoassay with microsphere-antigen conjugates. Bioconjugate Chem. 1992, 3, 187–193. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Review Criteria for Assessment of C-reactive Protein (CRP), High Sensitivity C-reactive Protein, and Cardiac C-reactive Protein (cCRP) Assays. Available online: https://www.fda.gov/media/71337/download (accessed on 1 September 2019).
- Von Schulthess, G.K.; Giglio, M.; Cannell, D.S.; Benedek, G.B. Detection of agglutination reactions using anisotropic light scattering: An immunoassay of high sensitivity. Mol. Immunol. 1980, 17, 81–92. [Google Scholar] [CrossRef]
- Shrinivasan, S.; Norris, P.M.; Landers, J.P.; Ferrance, J.P. A Low-Cost, Low-Power Consumption, Miniature Laser-Induced Fluorescence System for DNA Detection on a Microfluidic Device. Clin. Lab. Med. 2007, 27, 173–181. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Q.; Zheng, W.; Lin, C.; Shen, D. A Low-Cost Micro-Volume Nephelometric System for Quantitative Immunoagglutination Assays. Sensors 2019, 19, 4359. https://doi.org/10.3390/s19204359
Sun Q, Zheng W, Lin C, Shen D. A Low-Cost Micro-Volume Nephelometric System for Quantitative Immunoagglutination Assays. Sensors. 2019; 19(20):4359. https://doi.org/10.3390/s19204359
Chicago/Turabian StyleSun, Qiqi, Wei Zheng, Chao Lin, and Dongxuan Shen. 2019. "A Low-Cost Micro-Volume Nephelometric System for Quantitative Immunoagglutination Assays" Sensors 19, no. 20: 4359. https://doi.org/10.3390/s19204359
APA StyleSun, Q., Zheng, W., Lin, C., & Shen, D. (2019). A Low-Cost Micro-Volume Nephelometric System for Quantitative Immunoagglutination Assays. Sensors, 19(20), 4359. https://doi.org/10.3390/s19204359



