A Mobile Crowd Sensing Application for Hypertensive Patients
Abstract
1. Introduction
2. Materials and Methods
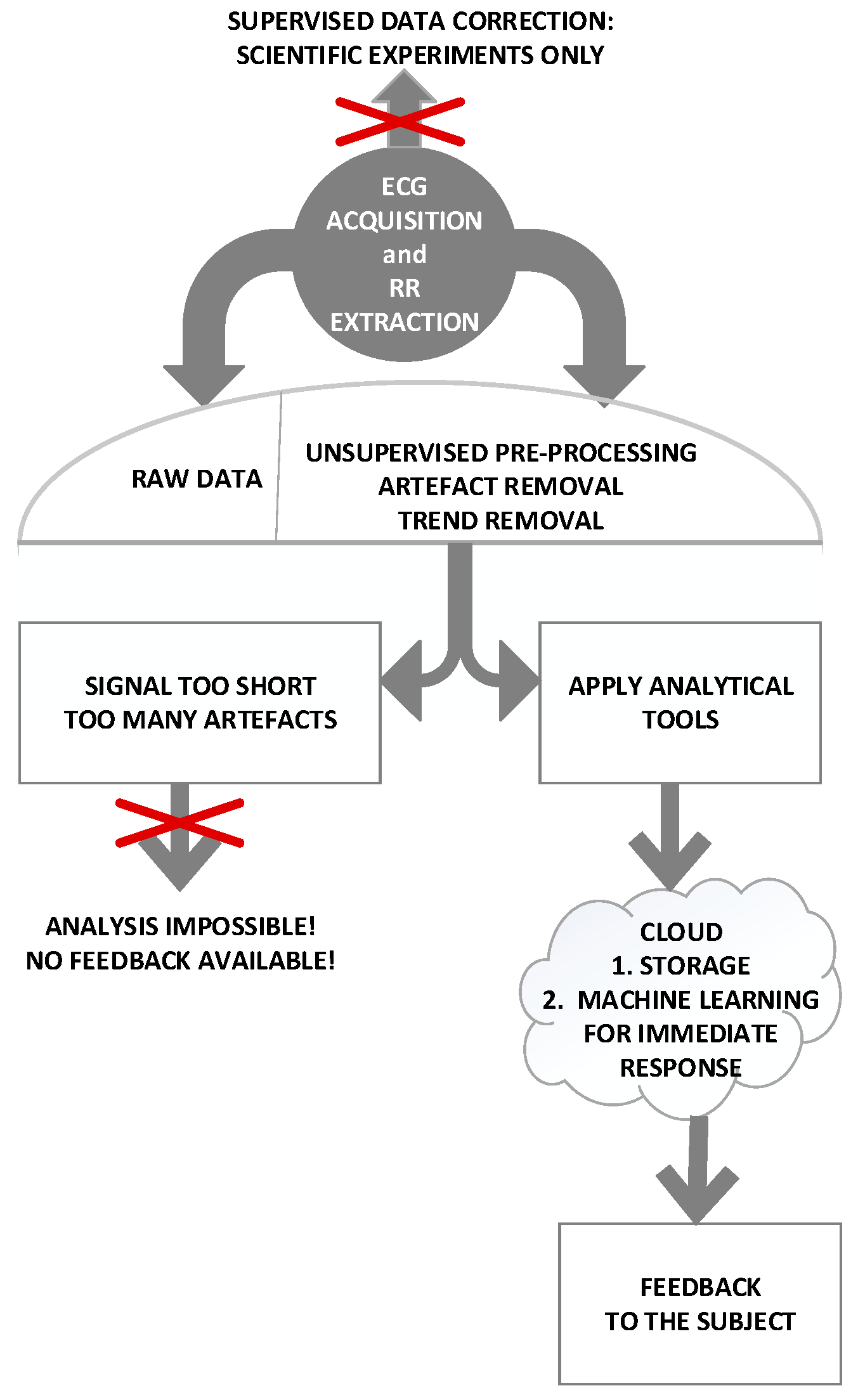
2.1. Data—Quality of Information
2.2. People—Minimal Effort Principle
2.3. Things—Devices and Bandwidth
2.3.1. Deployment of a TV White Space (TVWS) Network
2.3.2. Hardware and Android application
2.4. Process—Experimental Setting
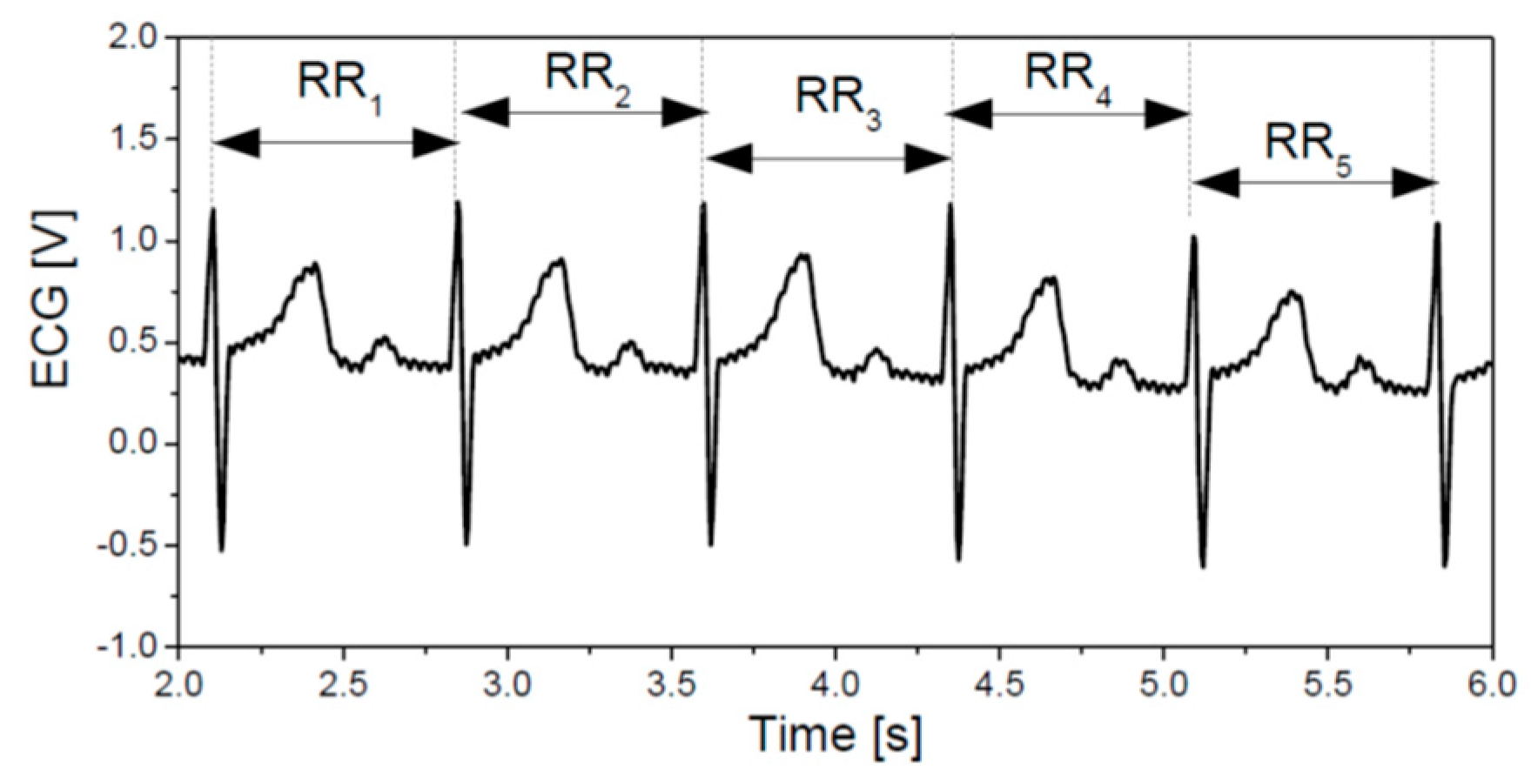
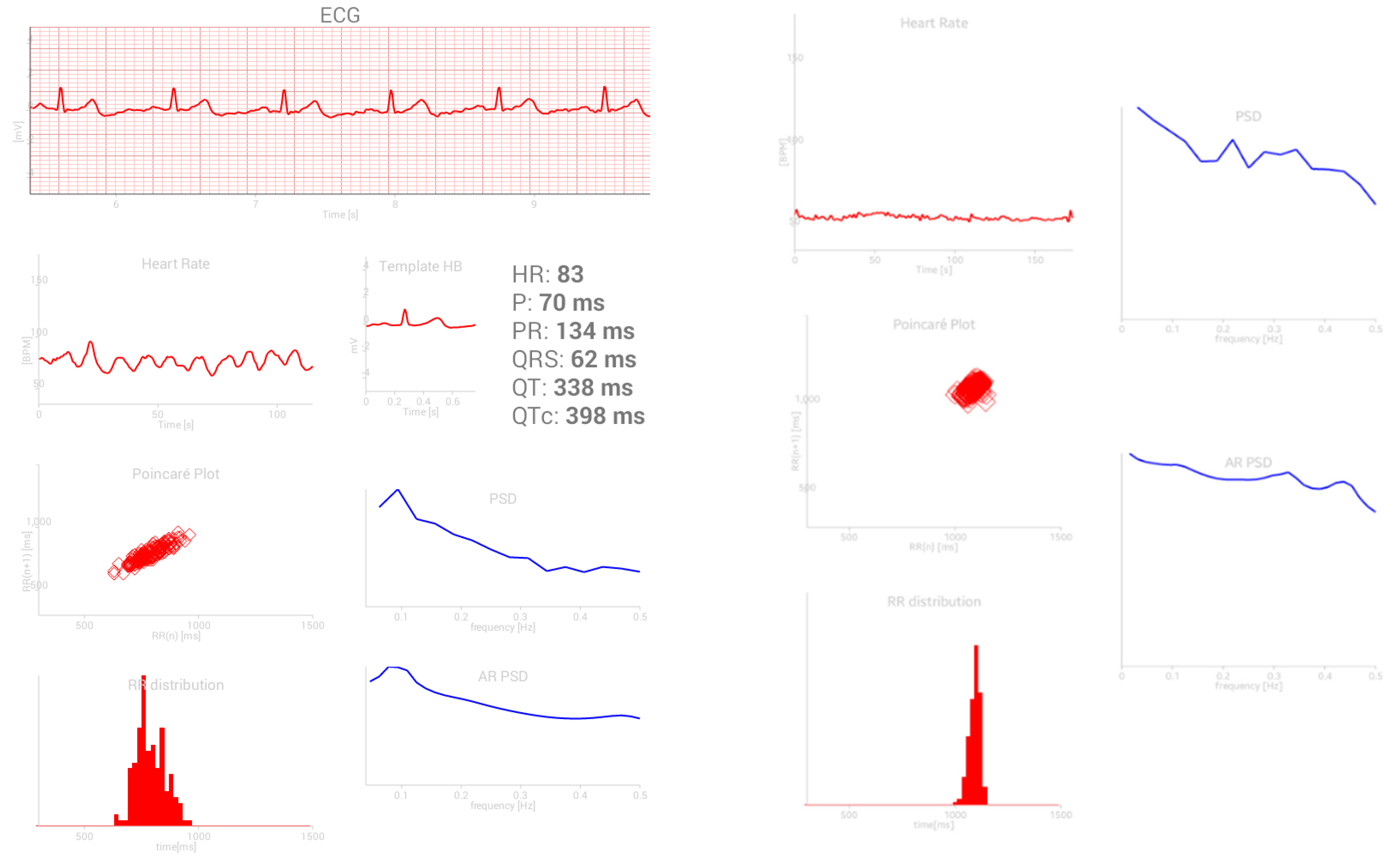
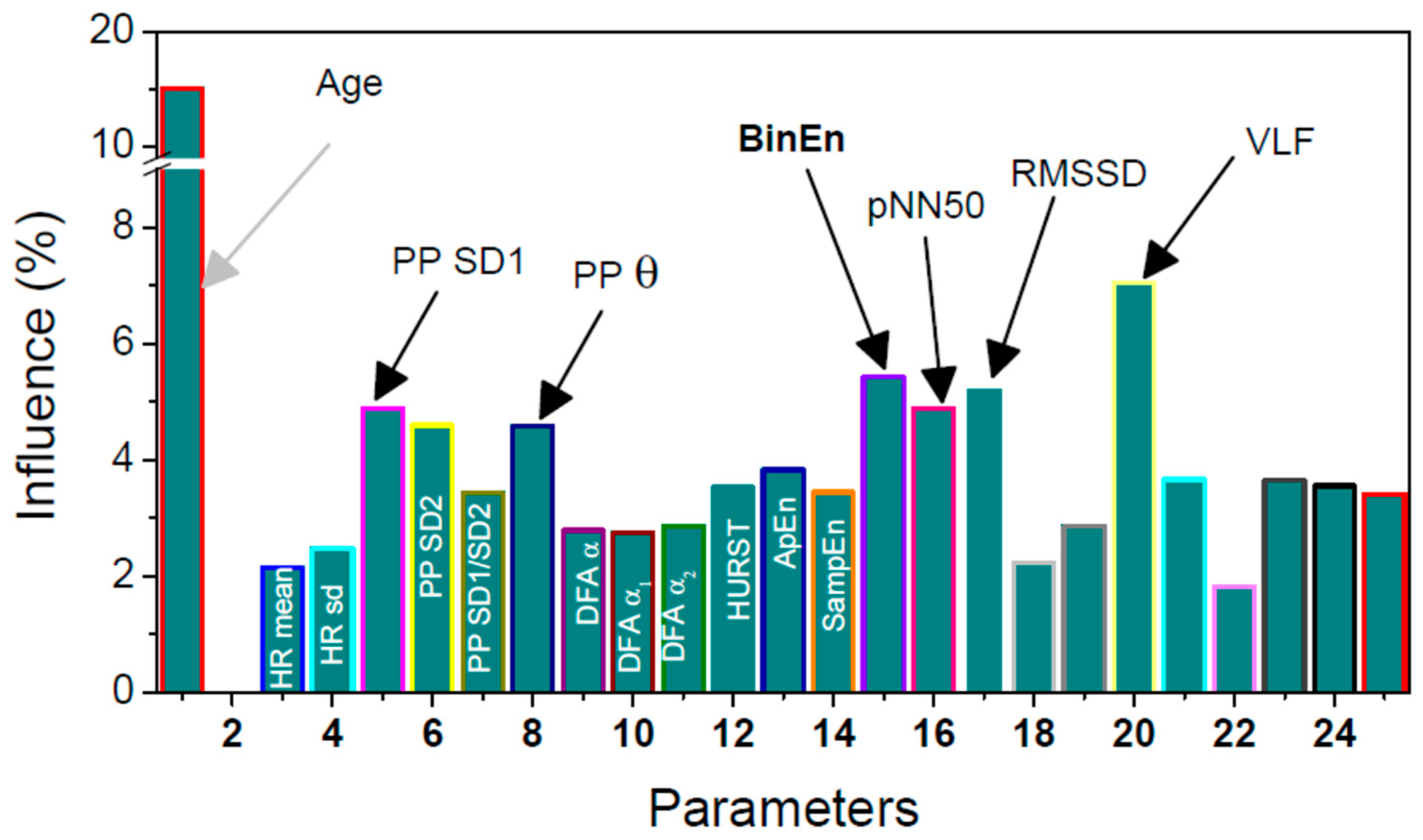
2.4.1. Signals and Features
- (1)
- Age
- (2)
- Heart rate mean and standard deviation;
- (3)
- Poincaré plots (PPlot or PP) of RR intervals parameters [31]:
- standard deviation SD1 across the identity line of PPlot shows short-term variability,
- standard deviation SD2 along the identity line of PPlot shows long-term variability,
- ratio SD1/SD2;
- copula parameter θ shows the level of interconnection of adjacent RR samples in PPlot plane [32];
- (4)
- Detrended fluctuation analysis (DFA) is a method for determining the statistical self-affinity of a signal, with overall self-affinity α and its lower and upper segments α1 and α2 [33];
- (5)
- Hurst exponent is similar to DFA but requires stationary data. It is used to explore the long-term memory of the time series [34];
- (6)
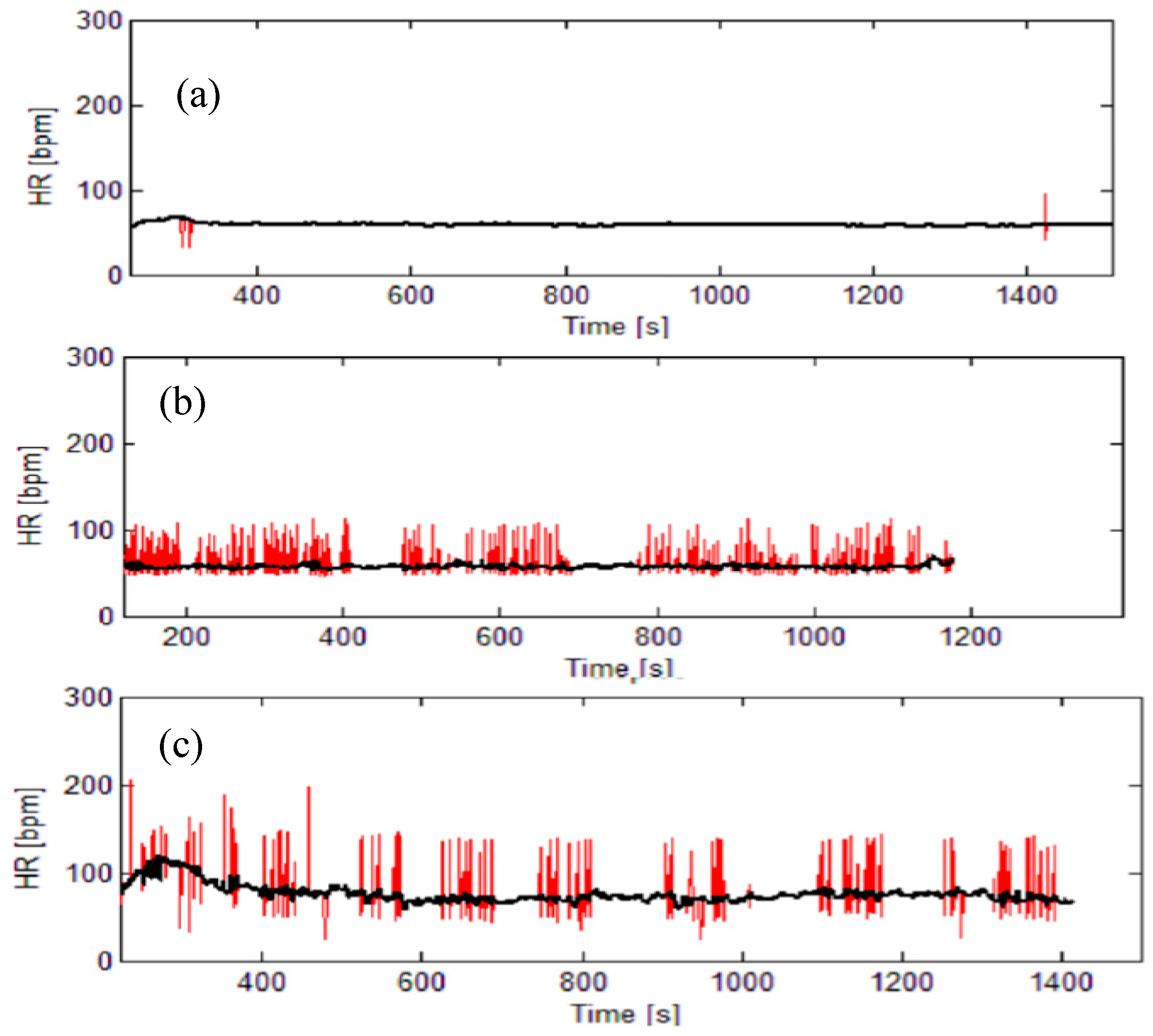
- pNN50 is a percentage of adjacent pairs of NN intervals that differ more than 50ms. It is a tricky parameter as it must be performed on NN intervals (emphasized by its name: pNN50). NN intervals are RR intervals without artifacts and pathologies. Therefore, the signal must be pre-processed prior to applying this analysis;
- (7)
- RMSSD is a root mean square of the adjacent NN intervals;
- (8)
- ApEn, SampEn, and BinEn: Approximate entropy, ApEn, [35] is one of the most quoted methods for estimating the self-regularity of the observed process. Its improved modification is Sample entropy, SampEn, [36]. Some evaluation of their respective thresholds is specified in [37]. Both methods require artifact-free stationary data. Binarized entropy, BinEn, is more robust. It is derived for crowd sensing [17].
- (9)
- Frequency parameters include the ratio of powers in low frequency and high frequency bands. The frequency bands range from 0.15 to 0.4 Hz–high (HF), from 0.04 to 0.15 Hz–low (LF), and from 0.0033 to 0.04 Hz–very low (VLF) [38].
2.4.2. The Central Application
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Crowdsensing: State of the Art and Privacy Aspects. Available online: https://resources.infosecinstitute.com/ crowdsensing-state-art-privacy-aspects/ (accessed on 28 November 2018).
- Noureen, J.; Asif, M. Crowdsensing: Socio-Technical Challenges and Opportunities. IJACSA 2017, 8, 363–369. [Google Scholar] [CrossRef]
- Ganti, R.K.; Ye, F.; Lei, H. Mobile crowdsensing: Current state and future challenges. IEEE Commun. Mag. 2011, 49, 32–39. [Google Scholar] [CrossRef]
- Chessa, S.; Corradi, A.; Foschini, L.; Girolami, M.E. Mobile Crowdsensing through Social and Ad Hoc Networking. IEEE Commun. Mag. 2016, 54, 108–114. [Google Scholar] [CrossRef]
- Talasila, M.; Curtmola, R.; Borcea, C. Ilr: Improving location reliability in mobile crowd sensing. IJBDCN 2013, 9, 65–85. [Google Scholar] [CrossRef]
- Jaimes, L.G.; Steele, R. Incentivization for Health Crowdsensing. In Proceedings of the IEEE 15th Intl Conf on Dependable, Autonomic and Secure Computing, 15th Intl Conf on Pervasive Intelligence and Computing, 3rd Intl Conf on Big Data Intelligence and Computing and Cyber Science and Technology Congress, Orlando, FL, USA, 6–10 November 2017. [Google Scholar]
- Pryss, R.; Reichert, M.; Langguth, B.; Schlee, W. Mobile Crowd Sensing Services for Tinnitus Assessment, Therapy, and Research. In Proceedings of the IEEE International Conference on Mobile Services, New York, NY, USA, 27 June–2 July 2015. [Google Scholar]
- Pryss, R.; Reichert, M.; Herrmann, J.; Langguth, B.; Schlee, W. Mobile Crowd Sensing in Clinical and Psychological Trials—A Case Study. In Proceedings of the IEEE 28th International Symposium on Computer-Based Medical Systems, Sao Carlos, Brazil, 22–25 June 2015. [Google Scholar]
- Mehdi, M.; MÃhlmeier, G.; Agrawal, K.; Pryss, R.; Reichert, M.; Hauck, F.J. Referenceable mobile crowdsensing architecture: A healthcare use case. Procedia Comput. Sci. 2018, 134, 445–451. [Google Scholar] [CrossRef]
- Mahdi, M.; Ali, M.; Excell, P.S.; Picking, R. A review on Internet of Things (IoT), Internet of Everything (IoE) and Internet of Nano Things (IoNT). Futur. Internet 2018, 10, 1–28. [Google Scholar]
- Vanderbroek, S.V. Three Pillars Enabling IoE. In Proceedings of the IEEE International Solid-State Circuits Conference (ISSCC), San Francisco, CA, USA, 31 January–4 February 2016. [Google Scholar]
- Wang, X.; Liu, Z.; Tian, X.; Gan, X.; Guan, Y.; Wang, X. Incentivizing Crowdsensing With Location-Privacy Preserving. IEEE Trans. Wirel. Commun. 2017, 16, 6940–6952. [Google Scholar] [CrossRef]
- Jin, H.; Su, L.; Chen, D.; Nahrstedt, K.; Xu, J. Quality of Information Aware Incentive Mechanisms for Mobile Crowd Sensing Systems. In Proceedings of the 16th ACM International Symposium on Mobile Ad Hoc Networking and Computing, Hangzhou, China, 22–25 June 2015. [Google Scholar]
- Jin, H.; Su, L.; Xiao, H.; Nahrstedt, K. Incentive Mechanism for Privacy-Aware Data Aggregation in Mobile Crowd Sensing Systems. IEEE/ACM Trans. Netw 2019, 26, 2019–2032. [Google Scholar] [CrossRef]
- Gandini, S.; Palli, D.; Spadola, G.; Bendinelli, B.; Cocorocchio, E.; Stanganelli, I.; Miligi, L.; Masala, G.; Caini, S. Anti-hypertensive drugs and skin cancer risk: A review of the literature and meta-analysis. Crit. Rev. Oncol. Hematol. 2018, 122, 1–9. [Google Scholar] [CrossRef]
- Skoric, T.; Mohamoud, O.; Milovanovic, B.; Japundzic-Zigon, N.; Bajic, D. Binarized cross-approximate entropy in crowdsensing environment. Comput. Biol. Med. 2017, 80, 137–147. [Google Scholar] [CrossRef]
- Mozetić, M.; Škorić, T.; Antelj, J.; Otašević, K.; Milovanović, B.; Bajić, D. Artifacts in dependency structures of Portapres® signals. IJEEC 2018, 2, 57–65. [Google Scholar]
- Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 1, 1043–1065.
- Wessel, N.; Malberg, H.; Bauernshmitt, R.; Kuths, J. Nonlinear methods of cardiovascular physics and their clinical applicability. IJBC 2007, 17, 3325–3371. [Google Scholar] [CrossRef]
- Tarvainen, M.P.; Ranta-aho, P.O.; Karjalainen, P.A. An advanced detrending approach with application to HRV analysis. IEEE Trans. Biomed. Eng. 2002, 42, 172–174. [Google Scholar] [CrossRef] [PubMed]
- Luo, T.; Das, S.K.; Tan, H.P.; Xia, L. Incentive Mechanism Design for Crowdsourcing: An All-Pay Auction Approach. ACM TIST 2016, 7, 35. [Google Scholar] [CrossRef]
- Zubairi, J.A.; Misbahuddin, S.; Tasadduq, I. Emergency Medical Data Transmission Systems and Techniques; IGI Global: Hershey, PA, USA, 2009; pp. 1–187. [Google Scholar]
- Manikandan, M.S.; Dandapat, S. Wavelet-based electrocardiogram signal compression methods and their performances: A prospective review. Biomed. Signal Process. Control 2014, 14, 73–107. [Google Scholar] [CrossRef]
- Adamo, A.; Grossi, G.; Lanzarotti, R.; Lin, J. ECG compression retaining the best natural basis k-coefficients via sparse decomposition. Biomed. Signal Process Control 2015, 15, 11–17. [Google Scholar] [CrossRef]
- Padhy, S.; Sharma, L.N.; Dandapat, S. Multilead ECG data compression using SVD in multiresolution domain. Biomed. Signal Process. Control 2016, 23, 10–18. [Google Scholar] [CrossRef]
- Němcová, A.; Smíšek, R.; Maršánová, L.; Smital, L.; Vítek, M. A Comparative Analysis of Methods for Evaluation of ECG Signal Quality after Compression. BioMed. Res. Int. 2018, 2018, 1868519. [Google Scholar] [CrossRef]
- Lin, Z.; Demir, A. Link adaptation on aggregated TVWS channels. In Proceedings of the IEEE Wireless Communications and Networking Conference (WCNC), Shanghai, China, 7–10 April 2013. [Google Scholar]
- Katzis, K.; Jones, R.W.; Despotou, G. Totally Connected Healthcare with TV White Spaces. Stud. Health Technol. Inform. 2017, 238, 68–71. [Google Scholar]
- Bochow, B.; Holland, O.; Katzis, K. Spectrum sensing infrastructure support for IEEE 1900.6b sensing-assisted spectrum databases. In Proceedings of the IEEE Conference on Standards for Communications and Networking (CSCN), Berlin, Germany, 31 October–2 November 2016. [Google Scholar]
- ECG for Everybody: Mobile Heart Care Platform. Available online: http://www.ecg4everybody.com/ (accessed on 28 November 2018).
- Brennan, M.; Palaniswami, M.; Kamen, P. Do Existing Measures of Poincaré Plot Geometry Reflect Nonlinear Features of Heart Rate Variability? IEEE Trans. Biomed. Eng. 2001, 48, 1342–1347. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, S.; Skoric, T.; Sarenac, O.; Milutinovic-Smiljanic, S.; Japundzic-Zigon, N.; Bajic, D. Copula as a dynamic measure of cardiovascular signal interactions. Biomed. Signal Process. Control 2018, 43, 250–264. [Google Scholar] [CrossRef]
- Peng, C.K.; Havlin, S.; Stanley, H.E.; Goldberger, A.L. Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series. Chaos Interdisciplin. J. Nonlinear Sci. 1995, 49, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Hurst, H.E.; Black, R.P.; Simaika, Y.M. Long-Term Storage: An Experimental Study; Constable: London, UK, 1965; pp. 1–145. [Google Scholar]
- Pincus, S.M. Approximate entropy (ApEn) as a complexity measure. Chaos 1995, 5, 110–117. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2008, 278, H2039–H2049. [Google Scholar] [CrossRef] [PubMed]
- Škorić, T.; Šarenac, O.; Japundžić-Žigon, N.; Milovanović, B.; Bajić, D. On consistency of cross-approximate entropy in cardiovascular and artificial environments. Complexity 2017, 2017, 1–15. [Google Scholar] [CrossRef]
- Berntson, G.G.; Bigger, J.T.; Eckberg, D.L.; Grossman, P.; Kaufmann, P.G.; Malik, M.; Nagaraja, H.N.; Porges, S.W.; Saul, J.P.; Stone, P.H.; et al. Heart rate variability: Origins, methods, and interpretive caveats. Pyschophysiology 1997, 43, 623–648. [Google Scholar] [CrossRef]
- Grobauer, B.; Walloschek, T.; Stocker, E. Understanding Cloud Computing Vulnerabilities. IEEE Secur. Priv. 2011, 9, 50–57. [Google Scholar] [CrossRef]
- William, Y.C.; Abu-Amara, H.; Feng Sanford, J. Transforming Enterprise Cloud Services; Springer: Dordrecht, The Netherlands, 2010; pp. 55–56. [Google Scholar]
- Oresko, J.J.; Jin, Z.; Cheng, J. A Wearable Smartphone-Based Platform for Real-Time Cardiovascular Disease Detection Via Electrocardiogram Processing. IEEE Trans. Inf. Technol. Biomed. 2010, 14, 734–740. [Google Scholar] [CrossRef]
- Weng, S.F.; Reps, J.; Kai, J.; Garibaldi, J.M.; Qureshi, N. Can machine-learning improve cardiovascular risk prediction using routine clinical data? PLoS ONE 2017. [Google Scholar] [CrossRef]
- Krittanawong, C.; Zhang, H.J.; Wang, Z.; Aydar, M.; Kitai, T. Artificial Intelligence in Precision Cardiovascular Medicine. J. Am. Coll. Cardiol. 2017, 69, 2658–2664. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.W.; Soto, J.T.; Glicksberg, B.S.; Shameer, K.; Miotto, R.; Ali, M.; Ashley, E.; Dudley, J.T. Artificial Intelligence in Cardiology. J. Am. Coll. Cardiol. 2018, 71, 2668–2678. [Google Scholar] [CrossRef] [PubMed]
- Haykin, S. Neural Networks, and Learning Machines, 3rd ed.; Prentice Hall Publishing: Englewood Cliffs, NJ, USA, 2008; pp. 1–936. [Google Scholar]
- Joo, S.; Choi, K.-J.; Huh, S.-J. Prediction of spontaneous ventricular tachyarrhythmia by an artificial neural network using parameters gleaned from short-term heart rate variability. Expert Syst. Appl. 2012, 39, 3862–3866. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Fawcett, T. An Introduction to ROC Analysis. Pattern Recognit. Lett. 2006, 27, 861–874. [Google Scholar] [CrossRef]
- Wu, F.-J.; Luo, T. WiFiScout: A Crowdsensing WiFi Advisory System with Gamification-Based Incentive. In Proceedings of the IEEE 11th International Conference on Mobile Ad Hoc and Sensor Systems, Philadelphia, PA, USA, 28–30 October 2014. [Google Scholar]


| No. of Subjects | Average Duration [s] | |
|---|---|---|
| Day 1: | 10 | 260 ± 52 |
| Day 10: | 3 | 120 ± 14 |
| Sensor Type | Sample Rate | Bandwidth Consumed |
|---|---|---|
| ECG | 125–500 sample/s. | 2 kbps to 8 kbps based on 2 bytes per sample |
| Blood Pressure | 1 sample/2 min | 16 bits/1 min |
| Pulse | 2 sample/s. | 32 bps |
| Respiration | 50 sample/s. | 800 bps |
| SpO2 | 2 samples/s. | 32 bps |
| CNTRL-R | CNTRL-C | HYP-R | HYP-C | CR vs CC | HR vs HC | CR vs HR | CC vs HC | ||
|---|---|---|---|---|---|---|---|---|---|
| HR mean | [bpm] | 72.04 ± 0.96 | 71.92 ± 0.97 | 75.25 ± 0.95 | 74.94 ± 0.94 | 0 | 0 | 1 | 0 |
| HR st. dev. | [bpm] | 6.06 ± 0.25 | 4.67 ± 0.17 | 5.77 ± 0.29 | 3.86 ± 0.13 | 1 | 1 | 1 | 1 |
| PP SD1 | [ms] | 43.87 ± 2.57 | 25.74 ± 1.38 | 36.72 ± 2.87 | 16.61 ± 0.86 | 1 | 1 | 1 | 1 |
| PP SD2 | [ms] | 87.75 ± 3.37 | 73.18 ± 2.93 | 67.88 ± 2.47 | 56.28 ± 1.77 | 1 | 1 | 1 | 1 |
| PP SD1/SD2 | 0.48 ± 0.02 | 0.35 ± 0.01 | 0.47 ± 0.02 | 0.29 ± 0.01 | 1 | 1 | 1 | 1 | |
| PP q | 7.97 ± 0.35 | 8.12 ± 0.36 | 10.35 ± 0.40 | 11.04 ± 0.39 | 0 | 0 | 1 | 1 | |
| DFA a | 0.85 ± 0.01 | 0.91 ± 0.01 | 0.90 ± 0.01 | 0.97 ± 0.01 | 1 | 1 | 1 | 1 | |
| DFA a1 | 0.80 ± 0.02 | 0.88 ± 0.02 | 0.90 ± 0.01 | 1.01 ± 0.01 | 1 | 1 | 1 | 1 | |
| DFA a2 | 0.81 ± 0.02 | 0.85 ± 0.02 | 0.84 ± 0.84 | 0.87 ± 0.02 | 0 | 0 | 0 | 0 | |
| HURST | 0.77 ± 0.01 | 0.81 ± 0.01 | 0.80 ± 0.01 | 0.84 ± 0.01 | 1 | 1 | 1 | 1 | |
| SampEn | 1.03 ± 0.03 | 1.15 ± 0.02 | 0.86 ± 0.02 | 0.99 ± 0.02 | 1 | 1 | 1 | 1 | |
| ApEn | 1.04 ± 0.02 | 1.13 ± 0.02 | 0.90 ± 0.02 | 1.02 ± 0.01 | 1 | 1 | 1 | 1 | |
| BinEn | 0.62 ± 0.01 | 0.62 ± 0.01 | 0.65 ± 0.00 | 0.65 ± 0.00 | 0 | 0 | 1 | 1 | |
| pNN50 | [%] | 18.01 ± 1.80 | 16.35 ± 1.66 | 8.38 ± 0.95 | 6.17 ± 0.75 | 0 | 1 | 1 | 1 |
| RMSSD | [ms] | 62.01 ± 3.63 | 36.38 ± 1.95 | 51.89 ± 4.05 | 23.48 ± 1.22 | 1 | 1 | 1 | 1 |
| ULF% | [%] | 98.28 ± 0.16 | 99.70 ± 0.02 | 94.00 ± 1.44 | 98.59 ± 0.39 | 1 | 1 | 1 | 1 |
| VLF% | [%] | 0.44 ± 0.04 | 0.10 ± 0.01 | 4.86 ± 1.40 | 1.32 ± 0.39 | 1 | 1 | 1 | 1 |
| LF% | [%] | 0.71 ± 0.07 | 0.14 ± 0.01 | 0.40 ± 0.05 | 0.06 ± 0.00 | 1 | 1 | 1 | 1 |
| HF% | [%] | 0.57 ± 0.08 | 0.07 ± 0.01 | 0.74 ± 0.13 | 0.03 ± 0.00 | 1 | 1 | 1 | 1 |
| LF/HF | 2.42 ± 0.22 | 2.98 ± 0.24 | 3.16 ± 0.27 | 3.94 ± 0.28 | 1 | 1 | 1 | 0 | |
| %LF/(LF+HF) | [%] | 60.98 ± 1.66 | 66.13 ± 1.55 | 60.09 ± 1.67 | 69.92 ± 1.10 | 1 | 1 | 1 | 0 |
| %HF/(LF+HF) | [%] | 39.02 ± 1.66 | 33.87 ± 1.55 | 39.91 ± 1.67 | 30.08 ± 1.10 | 1 | 1 | 1 | 0 |
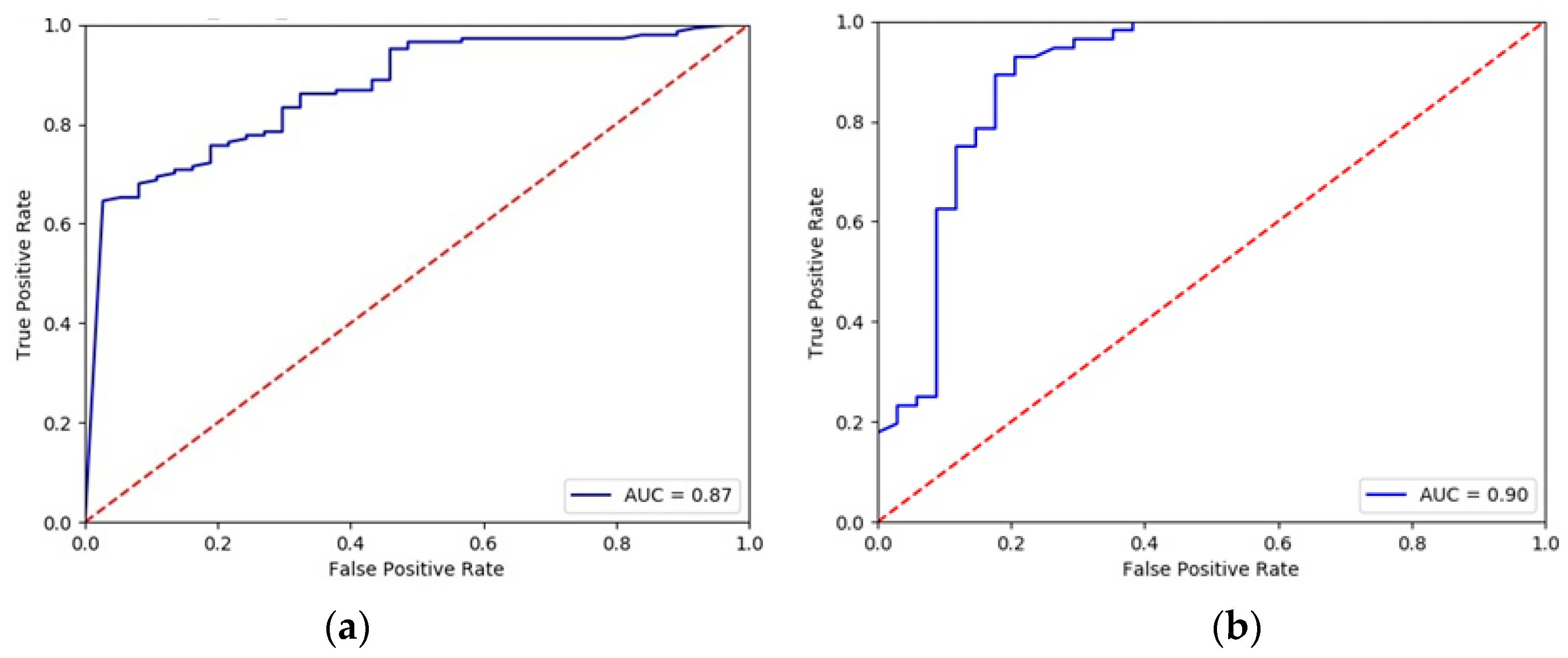
| ML Techniques | Accuracy | Sensitivity | Specificity | Positive Prediction | Negative Prediction |
|---|---|---|---|---|---|
| MLP | 85.6 | 87.3 | 82.9 | 88.9 | 80.0 |
| RF | 87.8 | 88.1 | 87.1 | 92.9 | 79.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jovanović, S.; Jovanović, M.; Škorić, T.; Jokić, S.; Milovanović, B.; Katzis, K.; Bajić, D. A Mobile Crowd Sensing Application for Hypertensive Patients. Sensors 2019, 19, 400. https://doi.org/10.3390/s19020400
Jovanović S, Jovanović M, Škorić T, Jokić S, Milovanović B, Katzis K, Bajić D. A Mobile Crowd Sensing Application for Hypertensive Patients. Sensors. 2019; 19(2):400. https://doi.org/10.3390/s19020400
Chicago/Turabian StyleJovanović, Slađana, Milan Jovanović, Tamara Škorić, Stevan Jokić, Branislav Milovanović, Konstantinos Katzis, and Dragana Bajić. 2019. "A Mobile Crowd Sensing Application for Hypertensive Patients" Sensors 19, no. 2: 400. https://doi.org/10.3390/s19020400
APA StyleJovanović, S., Jovanović, M., Škorić, T., Jokić, S., Milovanović, B., Katzis, K., & Bajić, D. (2019). A Mobile Crowd Sensing Application for Hypertensive Patients. Sensors, 19(2), 400. https://doi.org/10.3390/s19020400







