Fully Inkjet-Printed CuO Sensor on Flexible Polymer Substrate for Alcohol Vapours and Humidity Sensing at Room Temperature
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis and Characterization of CuO Nanoparticles
2.2. Ink Formulation and Characterization
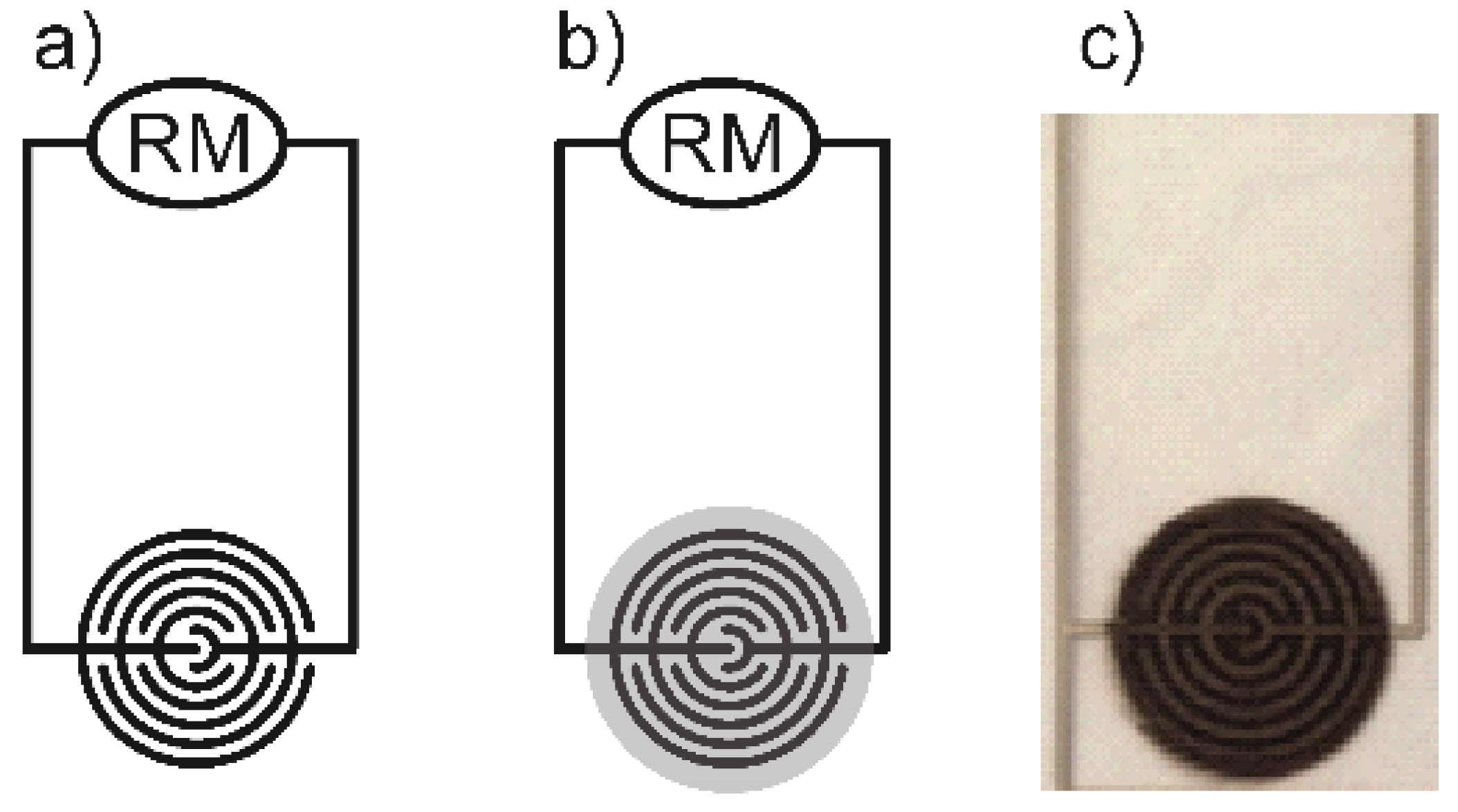
2.3. Printing of Thin Films, Patterns and Sensor Specimens
2.4. Analysis of Printed Patterns
2.5. Electrical and Sensing Properties
3. Results and Discussions
3.1. Results and Discussion of Material and Process Development and Device Fabrication Process
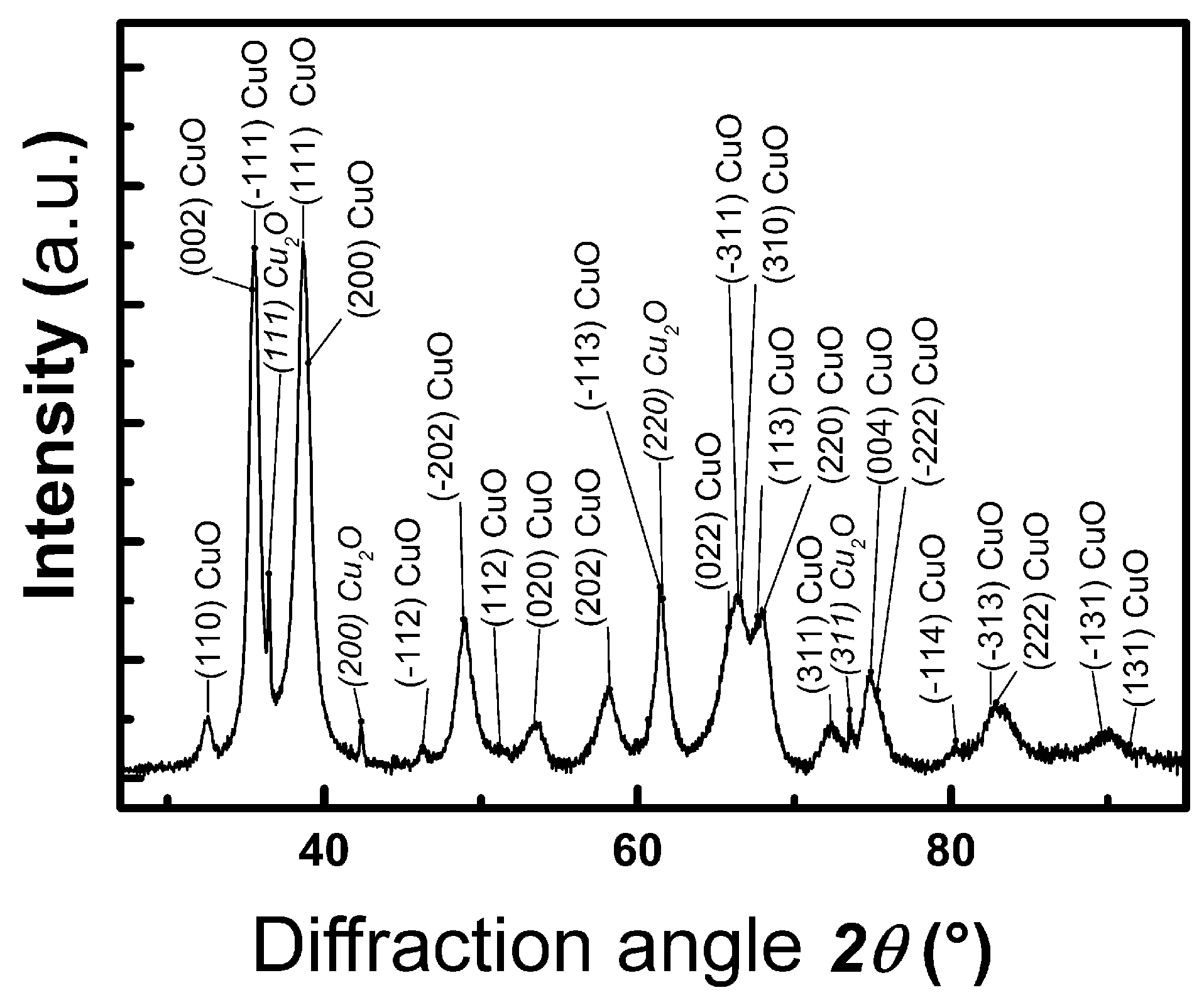
3.1.1. Characterization of CuO Particles
3.1.2. Ink Formulation and Characterization
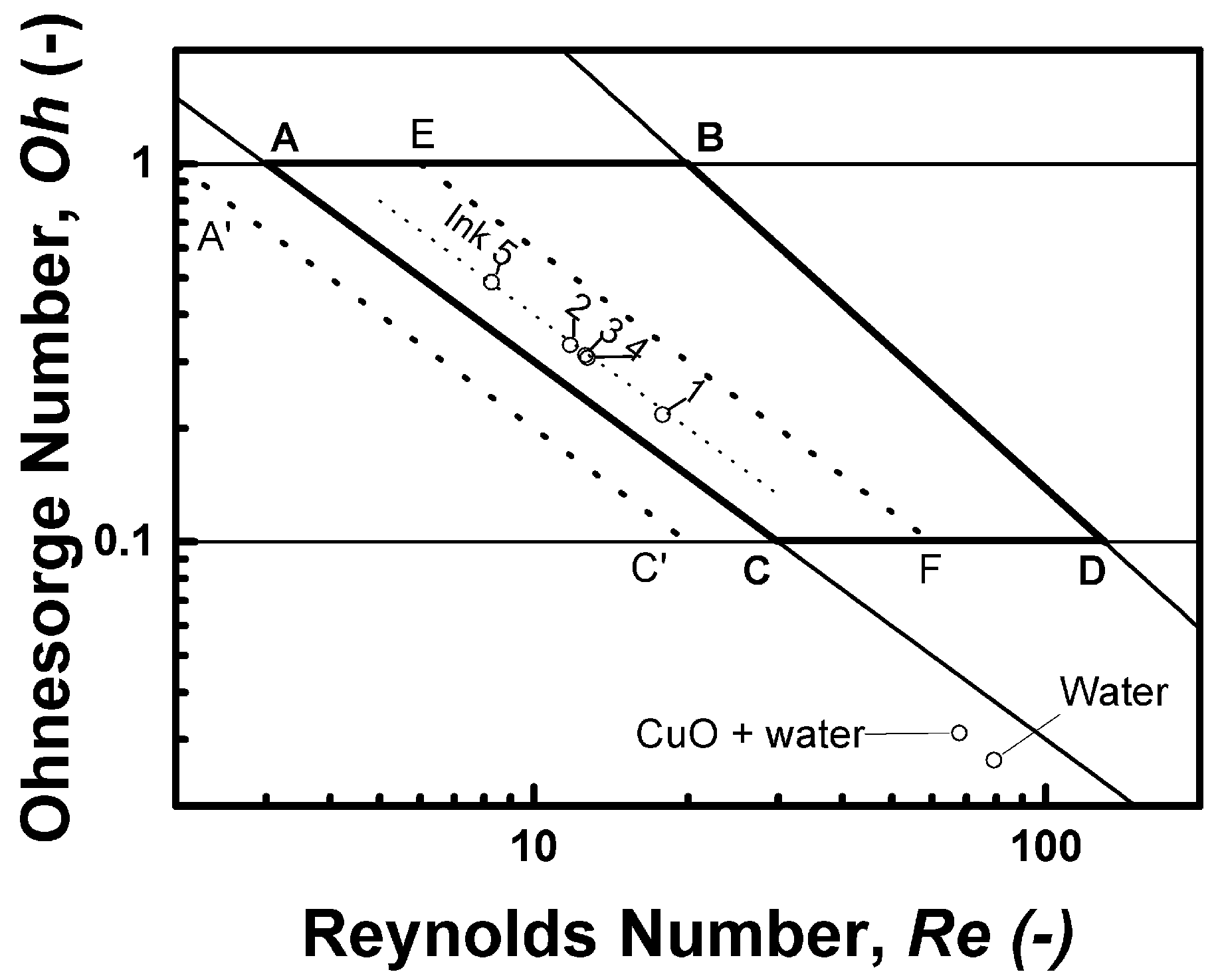
3.1.3. Refinement of the Ink and Inkjet Process Development by Dimensionless Criteria Analysis
3.1.4. Printing Process with a Dimatix DMP 2800 Series Device
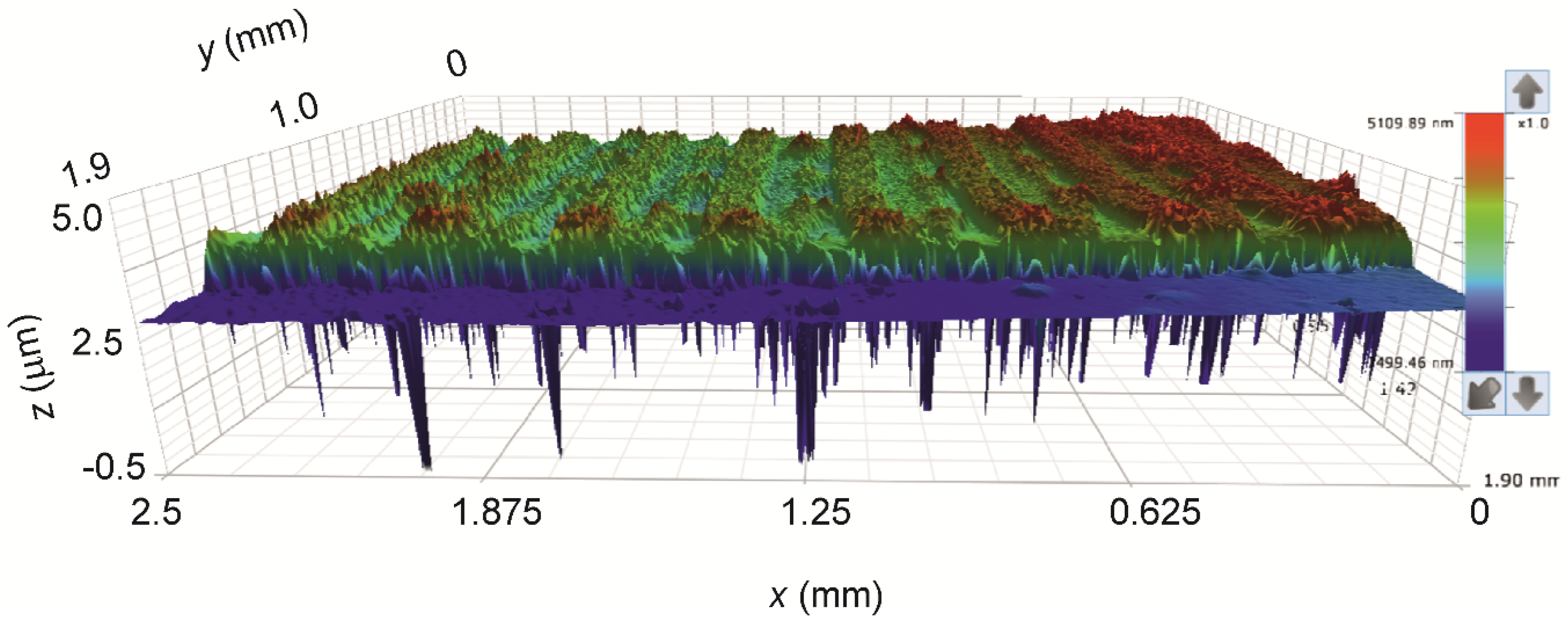
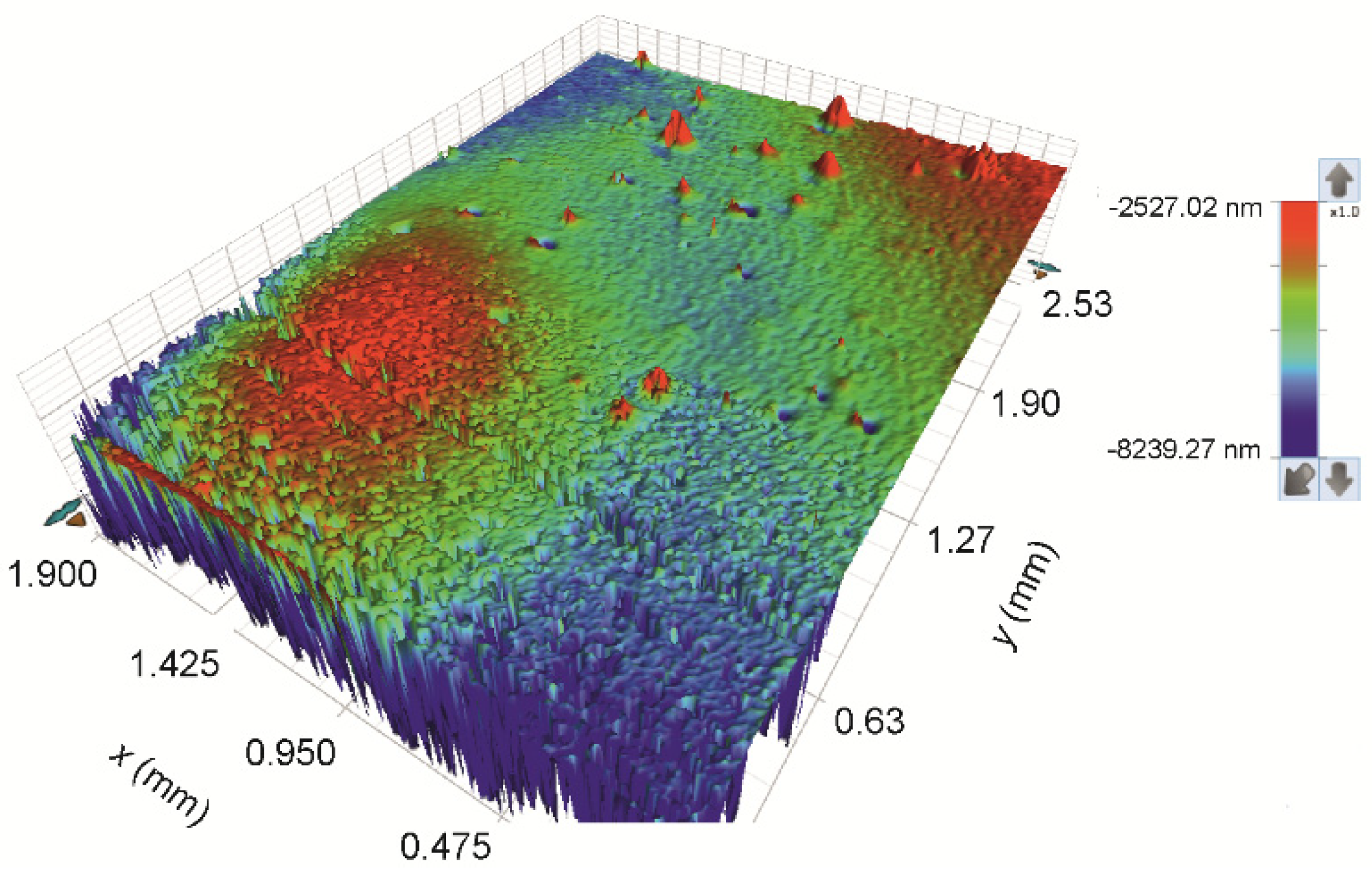
3.1.5. Analysis of the Printed Patterns
3.2. Results and Discussion of the Electrical Characterization and Sensing Properties
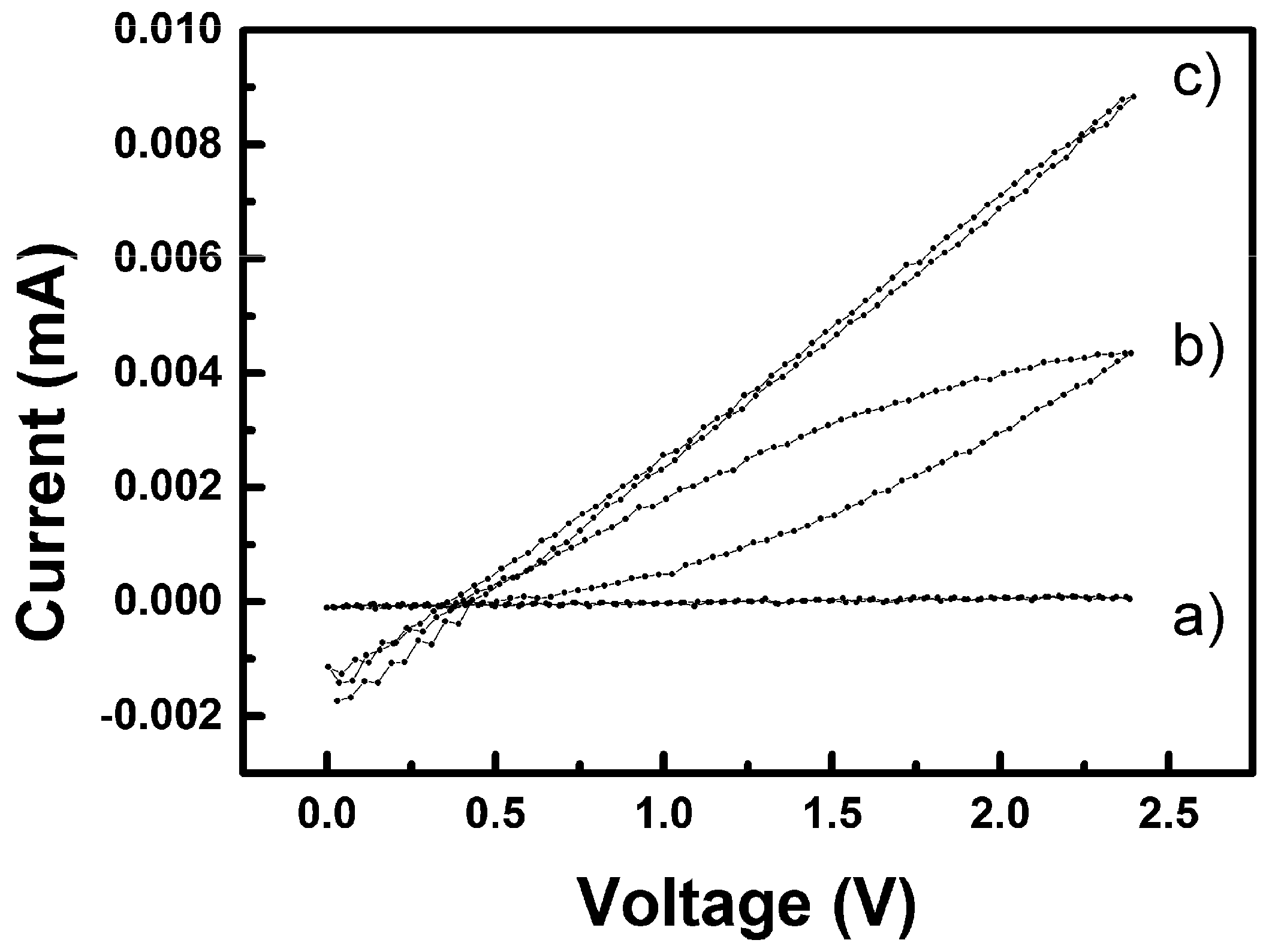
3.2.1. Preliminary Considerations and Experimental Verification of Printed Material Layers, Electrodes and Wirings
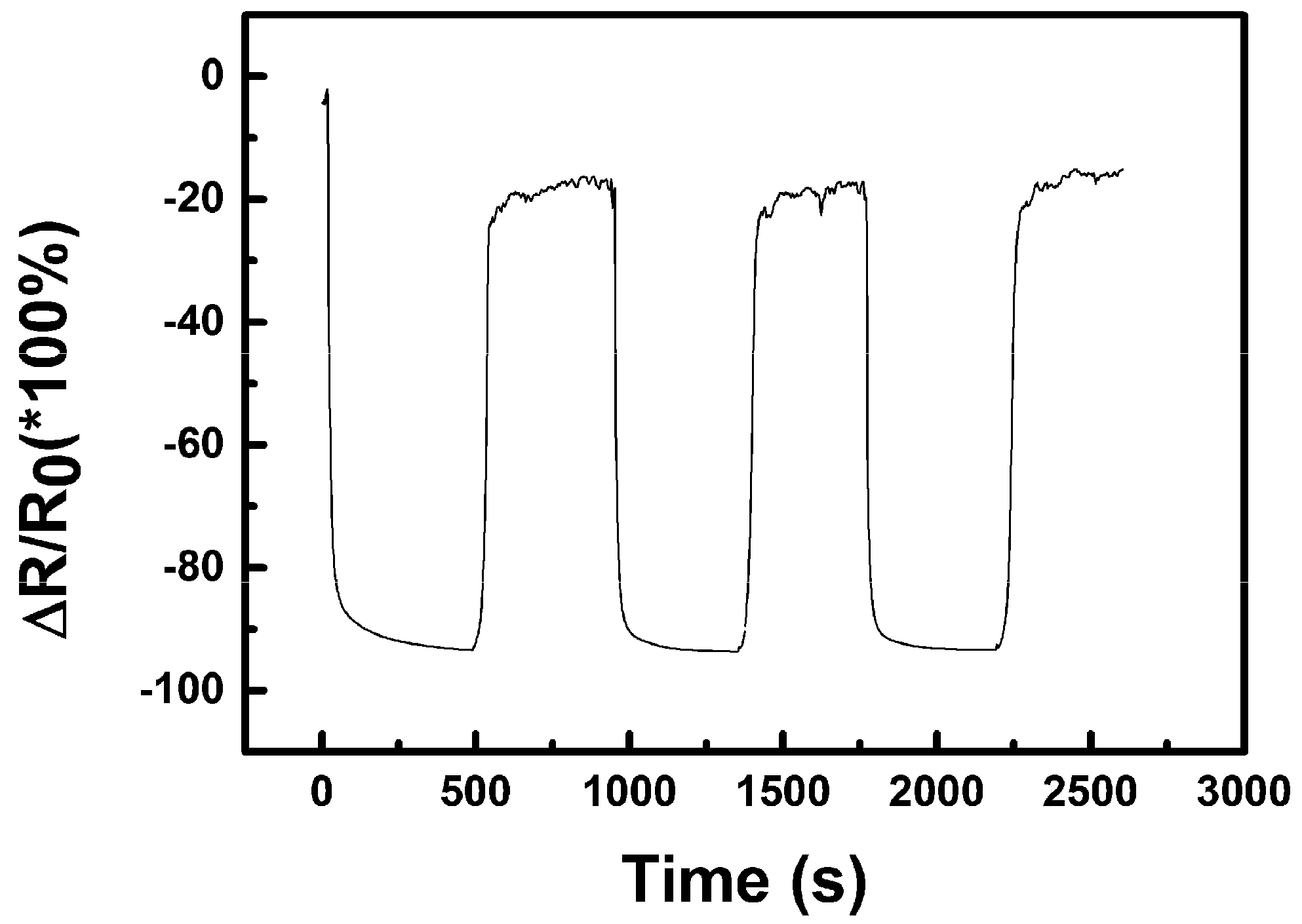
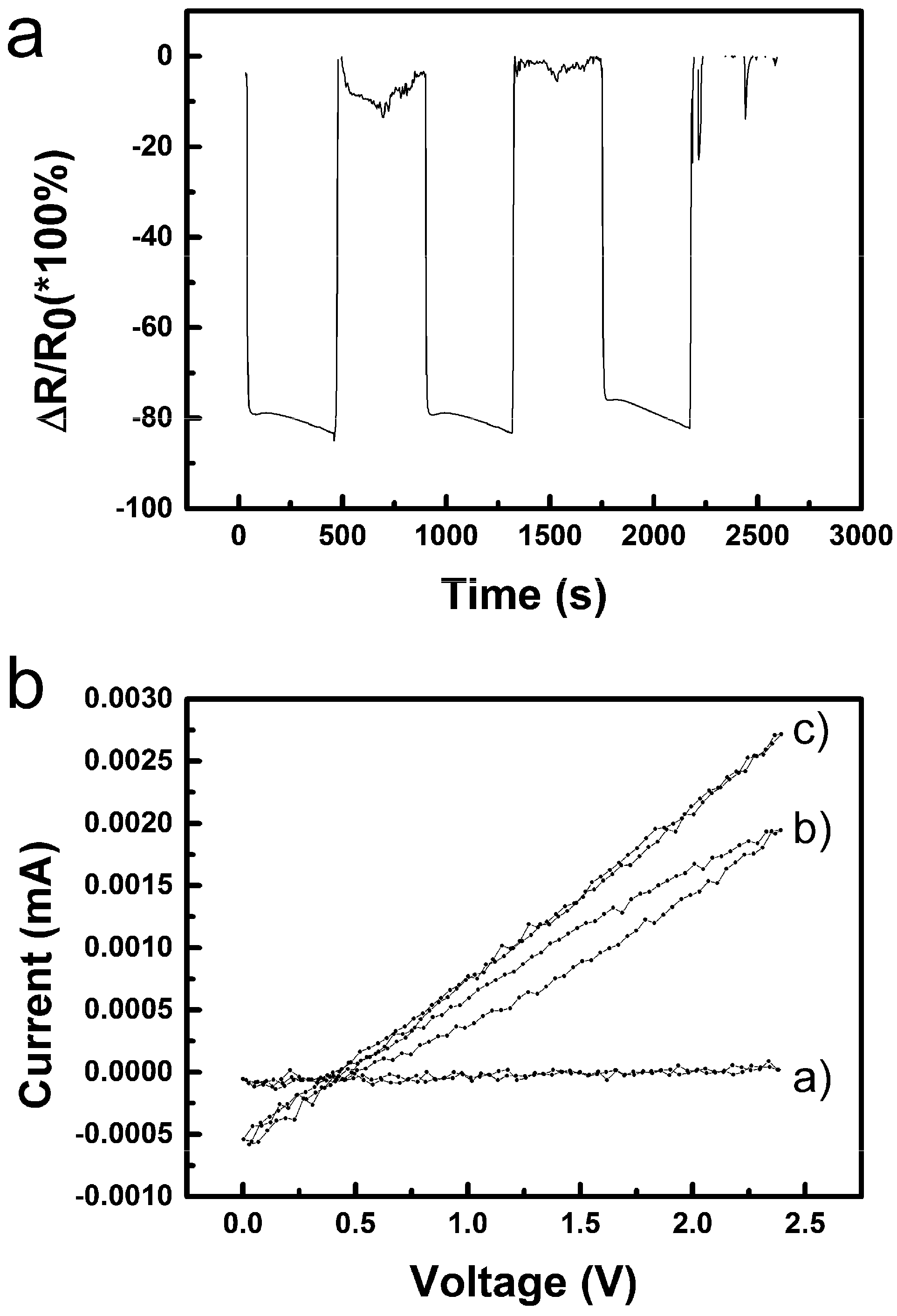
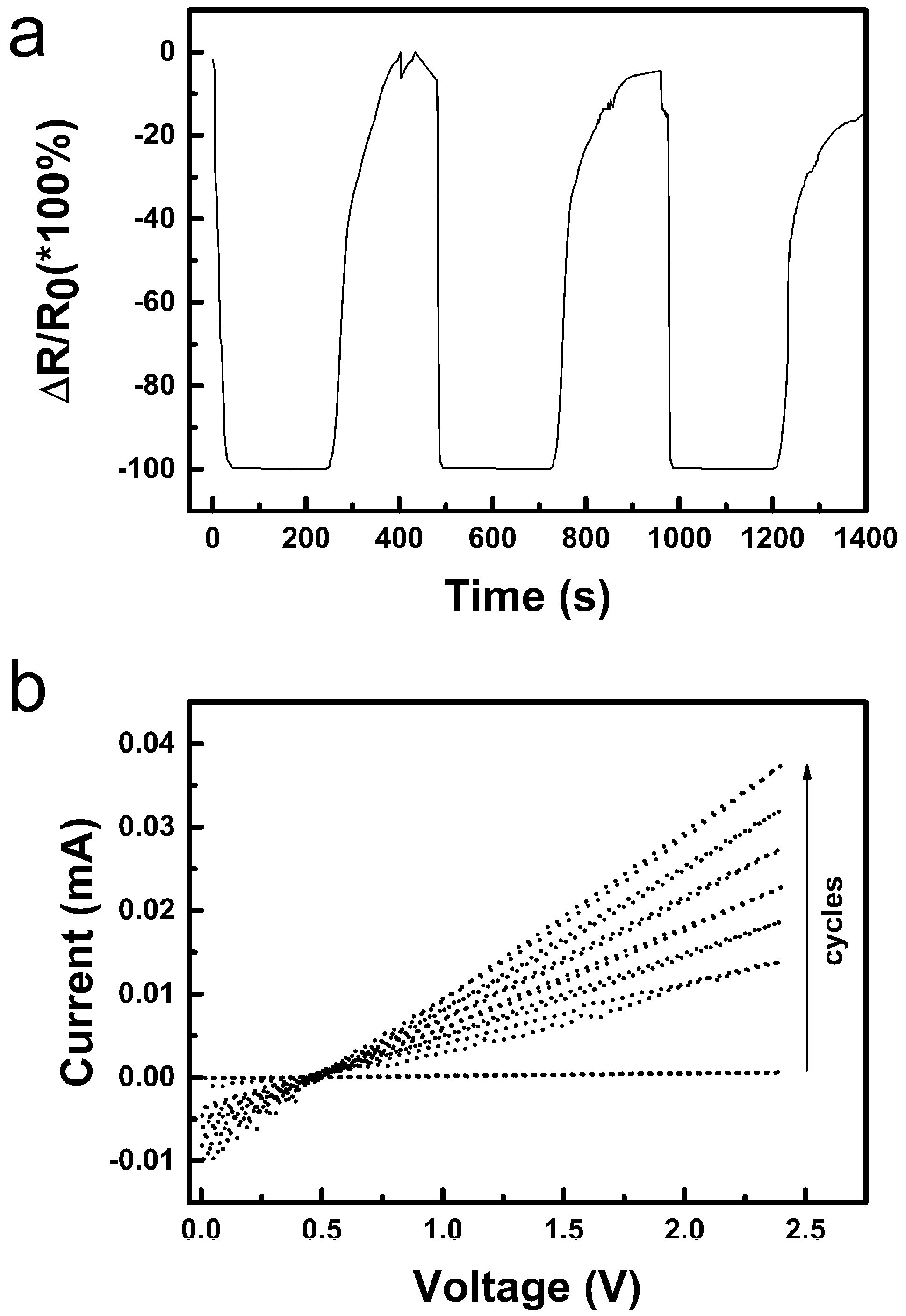
3.2.2. Humidity Sensing and Why Its Mechanism Differs from the Common p-Type one for CuO
3.2.3. Sensing of Alcohol Vapours and Re-Discussion of the Sensing Mechanism
4. Conclusions
5. Patent
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Used Dimensionless Criteria
References
- Singh, M.; Haverinen, H.M.; Dhagat, P.; Jabbour, G.E. Inkjet Printing-Process and Its Applications. Adv. Mater. 2010, 22, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Magdassi, S. The Chemistry of Inkjet Inks; World Scientific Publishing: Singapore; Hackensack, NJ, USA; London, UK, 2010; p. 345. [Google Scholar]
- Alamán, J.; Alicante, R.; Peña, J.; Sánchez-Somolinos, C. Inkjet Printing of Functional Materials for Optical and Photonic Applications. Materials 2016, 9, 910. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Hui, C.; Liu, H.; Du, G.; He, X.; Xi, Y. Synthesis of CuO nanostructures and their application for nonenzymatic glucose sensing. Sens. Actuators B Chem. 2010, 144, 220–225. [Google Scholar] [CrossRef]
- Calvert, P. Inkjet printing for materials and devices. Chem. Mater. 2001, 13, 3299–3305. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, K.; Xu, D.; Yang, G.; Huang, H.; Nie, F.; Liu, C.; Yang, S. CuO nanostructures: Synthesis, characterization, growth mechanisms, fundamental properties, and applications. Prog. Mater. Sci. 2014, 60, 208–337. [Google Scholar] [CrossRef]
- Gao, H.; Jia, H.; Bierer, B.; Woellenstein, J.; Lu, Y.; Palzer, S. Scalable gas sensors fabrication to integrate metal oxide nanoparticles with well-defined shape and size. Sens. Actuators B Chem. 2017, 249, 639–646. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Song, L.; Chen, X.; Liu, W.; Sun, J.; She, X.; Zhong, Z.; Su, F. Flower-like ZnO grown on urchin-like CuO microspheres for catalytic synthesis of dimethyldichlorosilane. RSC Adv. 2013, 3, 9794–9802. [Google Scholar] [CrossRef]
- Sun, Z.P.; Liu, L.; Zhang, L.; Jia, D.Z. Rapid synthesis of ZnO nano-rods by one-step, room-temperature, solid-state reaction and their gas-sensing properties. Nanotechnology 2006, 17, 2266–2270. [Google Scholar] [CrossRef]
- Wan, Q.; Li, Q.H.; Chen, Y.J.; Wang, T.H.; He, X.L.; Li, J.P.; Lin, C.L. Fabrication and ethanol sensing characteristics of ZnO nanowire gas sensors. Appl. Phys. Lett. 2004, 84, 3654–3656. [Google Scholar] [CrossRef]
- Comini, E.; Faglia, G.; Sberveglieri, G.; Pan, Z.W.; Wang, Z.L. Stable and highly sensitive gas sensors based on semiconducting oxide nanobelts. Appl. Phys. Lett. 2002, 81, 1869–1871. [Google Scholar] [CrossRef]
- Chen, L.; Lee, C.; Cheng, W. MEMS-based humidity sensor with integrated temperature compensation mechanism. Sens. Actuators A Phys. 2008, 147, 522–528. [Google Scholar] [CrossRef]
- Yeo, T.L.; Sun, T.; Grattan, K.T.V. Fibre-optic sensor technologies for humidity and moisture measurement. Sens. Actuators A Phys. 2008, 144, 280–295. [Google Scholar] [CrossRef]
- Su, P.; Wang, C. Flexible humidity sensor based on TiO2 nanoparticles-polypyrrole-poly [3-(methacrylamino)propyl] trimethyl ammonium chloride composite materials. Sens. Actuators B Chem. 2008, 129, 538–543. [Google Scholar] [CrossRef]
- Vijayan, A.; Fuke, M.; Hawaldar, R.; Kulkarni, M.; Amalnerkar, D.; Aiyer, R.C. Optical fibre based humidity sensor using Co-polyaniline clad. Sens. Actuators B Chem. 2008, 129, 106–112. [Google Scholar] [CrossRef]
- Hsueh, H.T.; Hsueh, T.J.; Chang, S.J.; Hung, F.Y.; Tsai, T.Y.; Weng, W.Y.; Hsu, C.L.; Dai, B.T. CuO nanowire-based humidity sensors prepared on glass substrate. Sens. Actuators B Chem. 2011, 156, 906–911. [Google Scholar] [CrossRef]
- Xia, S.; Zhu, H.; Cai, H.; Zhang, J.; Yu, J.; Tang, Z. Hydrothermally synthesized CuO based volatile organic compound gas sensor. RSC Adv. 2014, 4, 57975–57982. [Google Scholar] [CrossRef]
- Danesh, E.; Molina-Lopez, F.; Camara, M.; Bontempi, A.; Quintero, A.V.; Teyssieux, D.; Thiery, L.; Briand, D.; de Rooij, N.F.; Persaud, K.C. Development of a New Generation of Ammonia Sensors on Printed Polymeric Hotplates. Anal. Chem. 2014, 86, 8951–8958. [Google Scholar] [CrossRef] [PubMed]
- Rieu, M.; Camara, M.; Tournier, G.; Viricelle, J.; Pijolat, C.; de Rooij, N.F.; Briand, D. Fully inkjet printed SnO2 gas sensor on plastic substrate. Sens. Actuators B Chem. 2016, 236, 1091–1097. [Google Scholar] [CrossRef]
- Zhang, D.; Jiang, C.; Liu, J.; Cao, Y. Carbon monoxide gas sensing at room temperature using copper oxide-decorated graphene hybrid nanocomposite prepared by layer-by-layer self-assembly. Sens. Actuators B Chem. 2017, 247, 875–882. [Google Scholar] [CrossRef]
- Xu, J.; Yu, K.; Wu, J.; Shang, D.; Li, L.; Xu, Y.; Zhu, Z. Synthesis, field emission and humidity sensing characteristics of honeycomb-like CuO. J. Phys. D Appl. Phys. 2009, 42, 075417. [Google Scholar] [CrossRef]
- Tanvir, N.B.; Yurchenko, O.; Laubender, E.; Urban, G. Investigation of low temperature effects on work function based CO2 gas sensing of nanoparticulate CuO films. Sens. Actuators B Chem. 2017, 247, 968–974. [Google Scholar] [CrossRef]
- Hong, J.; Li, J.; Ni, Y. Urchin-like CuO microspheres: Synthesis, characterization, and properties. J. Alloys Compd. 2009, 481, 610–615. [Google Scholar] [CrossRef]
- Mirzaei, A.; Neri, G. Microwave-assisted synthesis of metal oxide nanostructures for gas sensing application: A review. Sens. Actuators B Chem. 2016, 237, 749–775. [Google Scholar] [CrossRef]
- Lupan, O.; Postica, V.; Ababii, N.; Hoppe, M.; Cretu, V.; Tiginyanu, I.; Sontea, V.; Pauporte, T.; Viana, B.; Adelung, R. Influence of CuO nanostructures morphology on hydrogen gas sensing performances. Microelectron. Eng. 2016, 164, 63–70. [Google Scholar] [CrossRef]
- Cretu, V.; Postica, V.; Mishra, A.K.; Hoppe, M.; Tiginyanu, I.; Mishra, Y.K.; Chow, L.; de Leeuw, N.H.; Adelung, R.; Lupan, O. Synthesis, characterization and DFT studies of zinc-doped copper oxide nanocrystals for gas sensing applications. J. Mater. Chem. A 2016, 4, 6527–6539. [Google Scholar] [CrossRef]
- Maslik, J.; Kuritka, I.; Urbanek, P.; Krcmar, P.; Suly, P.; Masar, M.; Machovsky, M. Water-Based Indium Tin Oxide Nanoparticle Ink for Printed Toluene Vapours Sensor Operating at Room Temperature. Sensors 2018, 18, 3246. [Google Scholar] [CrossRef]
- McKinley, G.H.; Renardy, M. Wolfgang von Ohnesorge. Phys. Fluids 2011, 23, 127101. [Google Scholar] [CrossRef]
- Derby, B. Inkjet Printing of Functional and Structural Materials: Fluid Property Requirements, Feature Stability, and Resolution. Annu. Rev. Mater. Res. 2010, 40, 395–414. [Google Scholar] [CrossRef]
- Derby, B. Additive Manufacture of Ceramics Components by Inkjet Printing. Engineering 2015, 1, 113–123. [Google Scholar] [CrossRef]
- Kim, H.; Jin, C.; Park, S.; Kim, S.; Lee, C. H2S gas sensing properties of bare and Pd-functionalized CuO nanorods. Sens. Actuators B Chem. 2012, 161, 594–599. [Google Scholar] [CrossRef]
- Hansen, B.J.; Kouklin, N.; Lu, G.; Lin, I.; Chen, J.; Zhang, X. Transport, Analyte Detection, and Opto-Electronic Response of p-Type CuO Nanowires. J. Phys. Chem. C 2010, 114, 2440–2447. [Google Scholar] [CrossRef]
- Wang, C.; Fu, X.Q.; Xue, X.Y.; Wang, Y.G.; Wang, T.H. Surface accumulation conduction controlled sensing characteristic of p-type CuO nanorods induced by oxygen adsorption. Nanotechnology 2007, 18, 145506. [Google Scholar] [CrossRef]
- Li, D.; Hu, J.; Wu, R.; Lu, J.G. Conductometric chemical sensor based on individual CuO nanowires. Nanotechnology 2010, 21, 485502. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, P.; Annapoorni, S.; Trikha, S. Humidity-sensing properties of nanocrystalline haematite thin films prepared by sol-gel processing. Thin Solid Films 1999, 346, 266–268. [Google Scholar] [CrossRef]
- Pitois, A.; Pilenga, A.; Pfrang, A.; Tsotridis, G. Temperature-dependent CO desorption kinetics on supported gold nanoparticles: Relevance to clean hydrogen production and fuel cell systems. Int J Hydrogen. Energy 2011, 36, 4375–4385. [Google Scholar] [CrossRef]
- Chen, Z.; Lu, C. Humidity sensors: A review of materials and mechanisms. Sens. Lett. 2005, 3, 274–295. [Google Scholar] [CrossRef]




| Ink | Surfactant (wt%) | Dispersant (wt%) | CuO (wt%) | Viscosity (mPa∙s) | Surface Tension (mNm−1) | Density (g∙cm−3) |
|---|---|---|---|---|---|---|
| Ink 1 | 21.60 | 7.20 | 3.40 | (4.66 ± 0.04) | (22.13 ± 0.09) | (1.04 ± 0.04) |
| Ink 2 | 14.20 | 14.20 | 4.80 | (7.13 ± 0.02) | (21.98 ± 0.04) | (1.05 ± 0.06) |
| Ink 3 | 13.90 | 13.90 | 6.60 | (6.53 ± 0.01) | (21.73 ± 0.08) | (1.04 ± 0.03) |
| Ink 4 | 7.20 | 21.60 | 3.40 | (6.59 ± 0.02) | (21.42 ± 0.08) | (1.04 ± 0.07) |
| Ink 5 | 12.88 | 19.32 | 7.20 | (10.54 ± 0.01) | (21.52 ± 0.05) | (1.09 ± 0.04) |
| Water | - | - | - | (1.00 ± 0.02) | (72.22 ± 0.08) | (0.99 ± 0.01) |
| CuO + water | - | - | 3.00 | (1.18 ± 0.02) | (71.56 ± 0.01) | (1.00 ± 0.02) |
| Ink | Dimensionless Criterion | ||||
|---|---|---|---|---|---|
| Re | We | Oh | Z | Ca | |
| Ink 1 | 18.7 | 15.8 | 0.212 | 4.72 | 0.84 |
| Ink 2 | 12.4 | 16.1 | 0.324 | 3.09 | 1.30 |
| Ink 3 | 13.4 | 16.1 | 0.300 | 3.34 | 1.20 |
| Ink 4 | 13.3 | 16.3 | 0.305 | 3.28 | 1.23 |
| Ink 5 | 8.7 | 17.0 | 0.475 | 2.11 | 1.96 |
| Water | 83.2 | 4.6 | 0.026 | 38.75 | 0.06 |
| CuO + water | 71.2 | 4.7 | 0.030 | 32.85 | 0.07 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krcmar, P.; Kuritka, I.; Maslik, J.; Urbanek, P.; Bazant, P.; Machovsky, M.; Suly, P.; Merka, P. Fully Inkjet-Printed CuO Sensor on Flexible Polymer Substrate for Alcohol Vapours and Humidity Sensing at Room Temperature. Sensors 2019, 19, 3068. https://doi.org/10.3390/s19143068
Krcmar P, Kuritka I, Maslik J, Urbanek P, Bazant P, Machovsky M, Suly P, Merka P. Fully Inkjet-Printed CuO Sensor on Flexible Polymer Substrate for Alcohol Vapours and Humidity Sensing at Room Temperature. Sensors. 2019; 19(14):3068. https://doi.org/10.3390/s19143068
Chicago/Turabian StyleKrcmar, Petr, Ivo Kuritka, Jan Maslik, Pavel Urbanek, Pavel Bazant, Michal Machovsky, Pavol Suly, and Petr Merka. 2019. "Fully Inkjet-Printed CuO Sensor on Flexible Polymer Substrate for Alcohol Vapours and Humidity Sensing at Room Temperature" Sensors 19, no. 14: 3068. https://doi.org/10.3390/s19143068
APA StyleKrcmar, P., Kuritka, I., Maslik, J., Urbanek, P., Bazant, P., Machovsky, M., Suly, P., & Merka, P. (2019). Fully Inkjet-Printed CuO Sensor on Flexible Polymer Substrate for Alcohol Vapours and Humidity Sensing at Room Temperature. Sensors, 19(14), 3068. https://doi.org/10.3390/s19143068







