Disposable Potentiometric Sensory System for Skin Antioxidant Activity Evaluation
Abstract
1. Introduction
2. Materials and Methods
2.1. Instruments
2.2. Chemicals and Reagents
2.3. Materials
2.4. Investigated Persons
2.5. Methods and Calculations
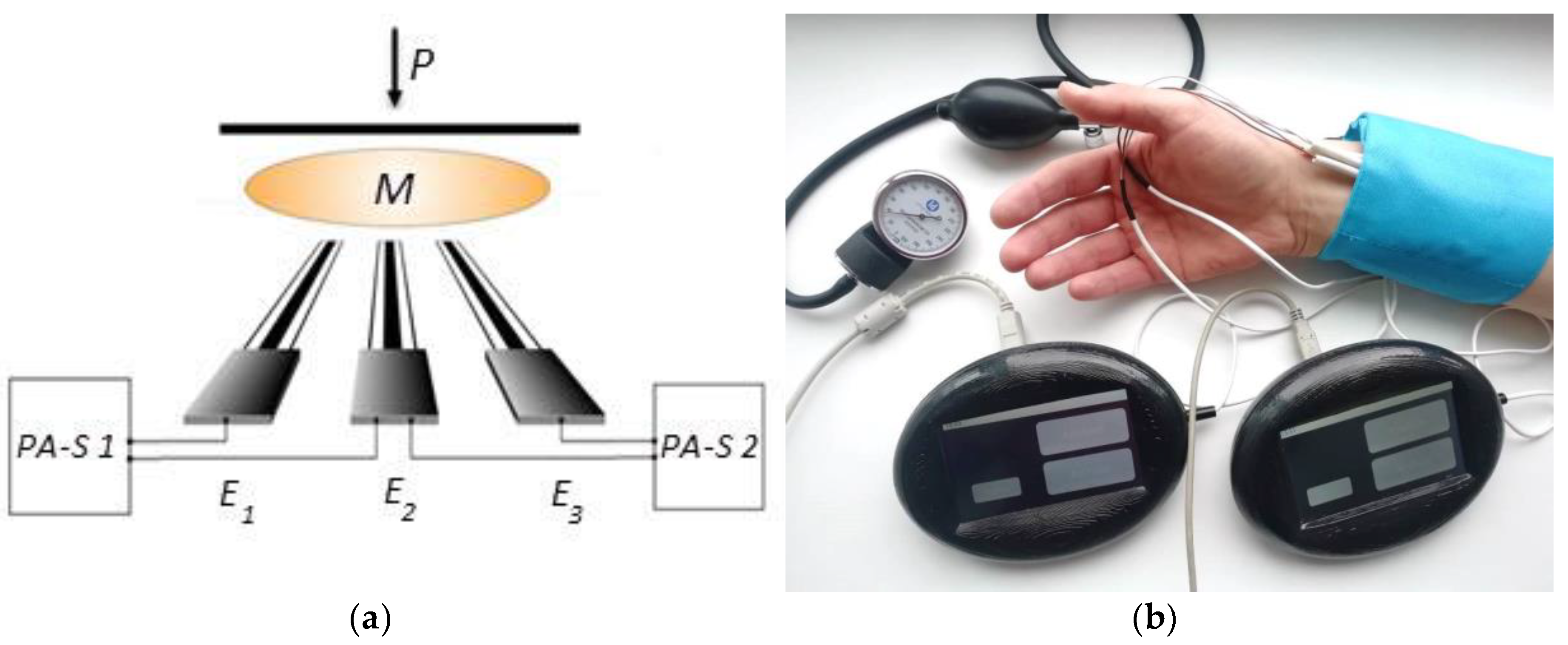
3. Sensory System Design
3.1. Fabrication of the Sensory System
3.2. Gold Nanoparticles Suspension Synthesis
3.3. Modified Carbon Screen-Printed Electrode
3.4. Modified Silver Screen-Printed Electrode
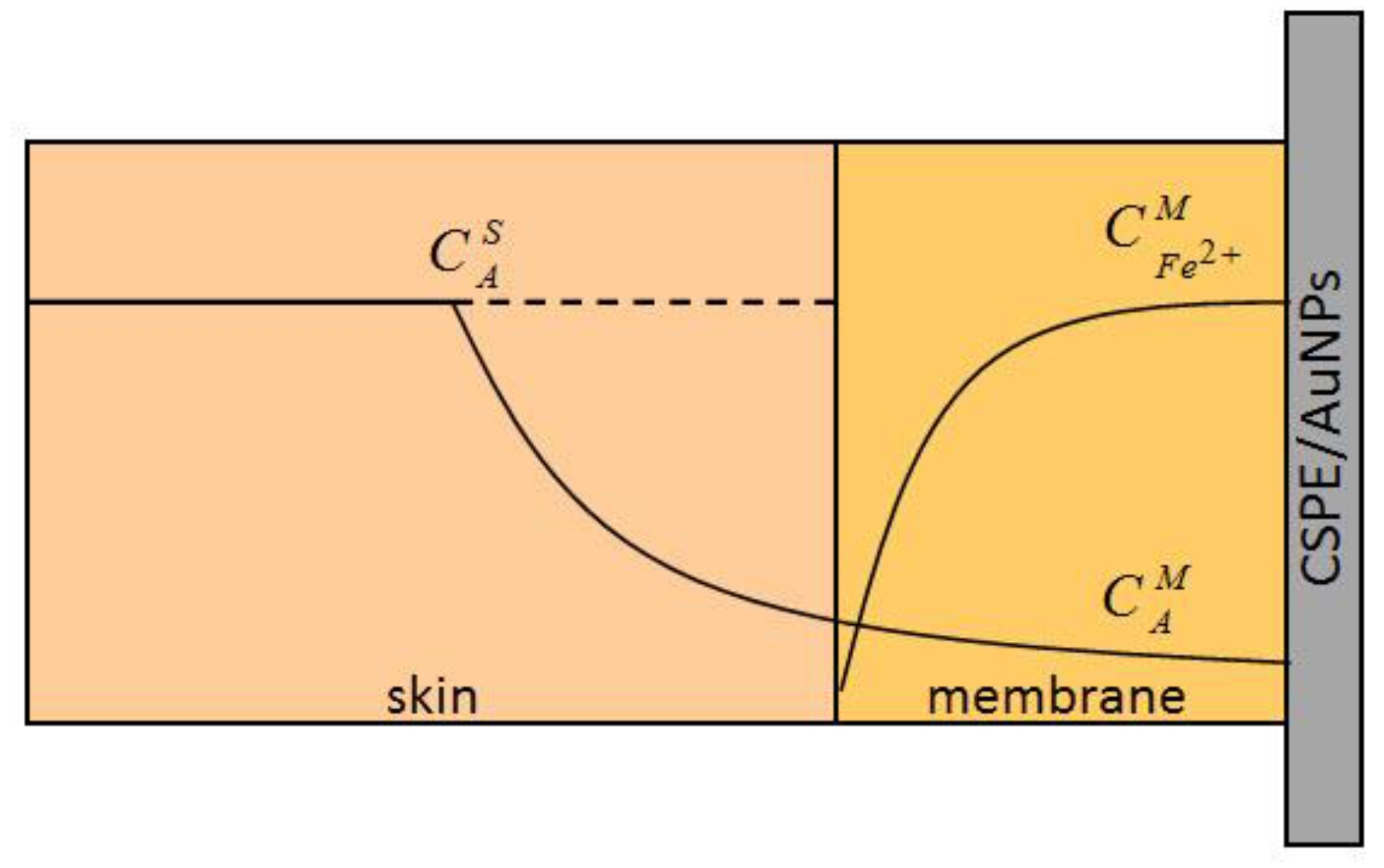
3.5. Membrane
3.6. Scheme and Measurement Technic
4. Results and Discussion
4.1. Study of the Kinetics of Stabilization and Reproducibility of the Potentials of the Electrodes Values
4.2. Investigation of Sensory System in Model Conditions
4.3. Sensory System in AOA of Skin Evaluation
4.3.1. Measurement of Skin AOA and L-Ascorbic Acid Recovery
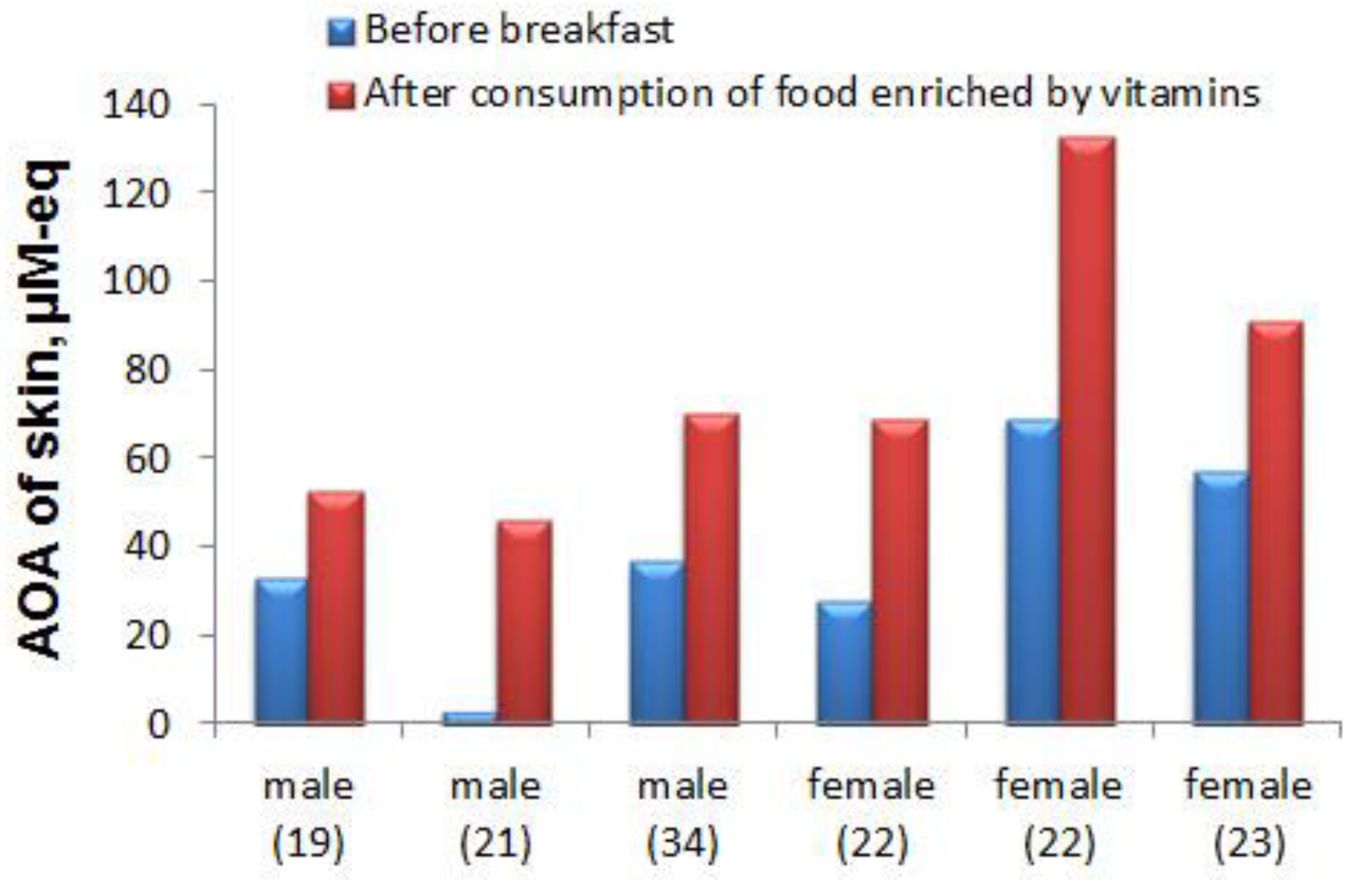
4.3.2. Practical Application of the Developed Sensory System
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kohen, R.; Gati, I. Skin low molecular weight antioxidants and their role in aging and in oxidative stress. Toxicology 2000, 148, 149–157. [Google Scholar] [CrossRef]
- Madison, K.C. Barrier function of the skin: “La raison d’etre" of the epidermis”. J. Invest. Dermatol. 2003, 121, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Ageing Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Liebel, F.; Kaur, S.; Ruvolo, E.; Kollias, N.; Southall, M.D. Irradiation of skin with visible light induces reactive oxygen species and matrix-degrading enzymes. J. Invest. Dermatol. 2012, 132, 1901–1907. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Liu, W.; Li, L.; Pan, X.; Crawford, M.; Sore, G.; Seite, S. Pollution and skin: From epidemiological and mechanistic studies to clinical implications. J. Dermatol. Sci. 2014, 76, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Brand, R.M.; Wipf, P.; Durham, A.; Epperly, M.W.; Greenberger, J.S.; Falo, L.D., Jr. Targeting mitochondrial oxidative stress to mitigate UV-induced skin damage. Front. Pharmacol. 2018, 9, 920. [Google Scholar] [CrossRef] [PubMed]
- Portugal, M.; Barak, V.; Ginsburg, I.; Kohen, R. Interplay among oxidants, antioxidants, and cytokines in skin disorders: Present status and future considerations. Biomed. Pharmacother. 2007, 61, 412–422. [Google Scholar] [CrossRef] [PubMed]
- Farage, M.A.; Miller, K.W.; Elsner, P.; Maibach, H.I. Intrinsic and extrinsic factors in skin ageing: A review. Int. J. Cosmet. Sci. 2008, 30, 87–95. [Google Scholar] [CrossRef]
- Poljšak, B.; Dahmane, R.G.; Godić, A. Intrinsic skin aging: The role of oxidative stress. Acta Derm. Alp. Pannonica Adriat. 2012, 21, 33–36. [Google Scholar]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef]
- Masaki, H. Role of antioxidants in the skin: Anti-aging effects. J. Dermatol. Sci. 2010, 58, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Witt, E.; Han, D.; Epstein, W.; Packer, L. Enzymic and non-enzymic antioxidants in epidermis and dermis of human skin. J. Invest. Dermatol. 1994, 102, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Thiele, J.J.; Ekanayake-Mudiyanselage, S. Vitamin E in human skin: Organ-specific physiology and considerations for its use in dermatology. Mol. Aspects Med. 2007, 28, 646–667. [Google Scholar] [CrossRef] [PubMed]
- Starr, J.M.; Starr, R.J. Skin aging and oxidative stress. In Aging: Oxidative Stress and Dietary Antioxidants; Preedy, V.R., Ed.; Academic Press: Oxford, UK; Waltham, MA, USA, 2014; Chapter 2; pp. 15–22. [Google Scholar]
- Addor, F.A.S. Antioxidants in dermatology. An. Bras. Dermatol. 2017, 92, 356–362. [Google Scholar] [CrossRef] [PubMed]
- Al-Jamal, M.S.; Griffith, J.L.; Lim, H.W. Photoprotection in ethnic skin. Dermatol. Sin. 2014, 32, 217–224. [Google Scholar] [CrossRef]
- Lephart, E.D. Skin aging and oxidative stress: Equol’s anti-aging effects via biochemical and molecular mechanisms. Ageing Res. Rev. 2016, 31, 36–54. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef]
- Afaq, F.; Katiyar, S.K. Polyphenols: Skin photoprotection and inhibition of photocarcinogenesis. Mini Rev. Med. Chem. 2011, 11, 1200–1215. [Google Scholar]
- Bragazzi, N.L.; Sellami, M.; Salem, I.; Conic, R.; Kimak, M.; Pigatto, P.D.M.; Damiani, G. Fasting and its impact on skin anatomy, physiology, and physiopathology: A comprehensive review of the literature. Nutrients 2019, 11, 249. [Google Scholar] [CrossRef]
- Kohen, R. Skin antioxidants: Their role in aging and in oxidative stress—New approaches for their evaluation. Biomed. Pharmacother. 1999, 53, 181–192. [Google Scholar] [CrossRef]
- Darvin, M.E.; Meinke, M.C.; Sterry, W.; Lademan, J. Optical methods for noninvasive determination of carotenoids in human and animal skin. J. Biomed. Opt. 2013, 18, 061230. [Google Scholar] [CrossRef] [PubMed]
- Markina, M.; Stozhko, N.; Krylova, V.; Vidrevich, M.; Brainina, K. Nanoparticle-based paper sensor for thiols evaluation in human skin. Talanta 2017, 165, 563–569. [Google Scholar] [CrossRef] [PubMed]
- De Jongh, C.M.; Verberk, M.M.; Spiekstra, S.W.; Gibbs, S.; Kezic, S. Cytokines at different stratum corneum levels in normal and sodium lauryl sulphate-irritated skin. Skin Res. Technol. 2007, 13, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Portugal-Cohen, M.; Oron, M.; Ma’or, Z.; Boaz, M.; Shtendik, L.; Biro, L.; Cernes, R.; Barnea, Z.; Kazir, Z.; Kohen, R. Noninvasive skin measurements to monitor chronic renal failure pathogenesis. Biomed. Pharmacother. 2011, 65, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Portugal-Cohen, M.; Kohen, R. Non-invasive evaluation of skin cytokines secretion: An innovative complementary method for monitoring skin disorders. Methods 2013, 61, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Kohen, R.; Fanberstein, D.; Tirosh, O. Non-invasive device and method for quantitative determination of oxidants and/or antioxidants in the skin. U.S. Patent No. 6,108,570, 10 October 1995. [Google Scholar]
- Brainina, K.Z.; Galperin, L.G.; Gerasimova, E.L.; Khodos, M.Y. Noninvasive potentiometric method of determination of skin oxidant/antioxidant activity. IEEE Sens. J. 2012, 12, 527–532. [Google Scholar] [CrossRef]
- Brainina, K.Z.; Varzakova, D.P.; Kazakov, Y.E.; Vidrevich, M.B. Noninvasive electrochemical antioxidant activity estimation: saliva analysis. Biointerface Res. Appl. Chem. 2018, 8, 3381–3387. [Google Scholar]
- Brainina, K.Z.; Markina, M.G.; Stozhko, N.Y. Optimized potentiometric assay for non-invasive investigation of skin antioxidant activity. Electroanalysis 2018, 30, 2405–2412. [Google Scholar] [CrossRef]
- Fitzpatrick, T.B. Soleil et peau. J. Méd. Esthét. 1975, 2, 33–34. (In French) [Google Scholar]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. The formation of colloidal gold. J. Phys. Chem. 1953, 57, 670–673. [Google Scholar] [CrossRef]
| Electrode | τ, s | E, mV | R, mV |
|---|---|---|---|
| CSPE | 360 ± 55 | 5 ± 3 | 7 |
| CSPE/AuNPs | 168 ± 72 | 4 ± 2 | 5 |
| AgSPE/mod | 70 ± 43 | 1 ± 1 | 1 |
| Introduced, μM-eq | Found, μM-eq | RSD, % | Recovery, % |
|---|---|---|---|
| 20.0 | 31.0 ± 16.8 | 17 | 155 ± 84 |
| 30.0 | 34.6 ± 3.0 | 8 | 115 ± 10 |
| 40.0 | 40.8 ± 2.1 | 5 | 102 ± 5 |
| 100.0 | 104.2 ± 2.4 | 2 | 104 ± 2 |
| 900.0 | 929.5 ± 22.1 | 2 | 103 ± 2 |
| Respondent No | AOA of Skin, μM-eq | RSD, % | Introduced L-ascorbic Acid, μM-eq | Total AOA, μM-eq | Recovery, % |
|---|---|---|---|---|---|
| 1 | 26.0 ± 5.2 | 20 | 50.0 | 81.2 ± 8.8 | 110 ± 7 |
| 2 | 43.5 ± 6.5 | 15 | 50.0 | 96.0 ± 12.2 | 105 ± 11 |
| 3 | 64.8 ± 7.8 | 12 | 50.0 | 115.9 ± 9.2 | 102 ± 3 |
| 4 | 95.4 ± 9.2 | 9 | 50.0 | 145.3 ± 8.0 | 100 ± 3 |
| Respondent | AOA, μM-eq | ΔAOA, μM-eq | |||
|---|---|---|---|---|---|
| No | Sex | Age, years | Before Breakfast | After 100 ± 10 min After Breakfast | |
| 1 | m a l e | 19 | 8 | 77 | +69 |
| 2 | 18 | 10 | 25 | +15 | |
| 3 | 21 | 12 | 40 | +28 | |
| 4 | 19 | 73 | 77 | +4 | |
| 5 | 18 | 91 | 95 | +4 | |
| 6 | 18 | 106 | 73 | –33 | |
| 7 | f e m a l e | 18 | 12 | 69 | +57 |
| 8 | 18 | 22 | 50 | +28 | |
| 9 | 22 | 28 | 43 | +15 | |
| 10 | 18 | 57 | 40 | –17 | |
| 11 | 22 | 69 | 60 | –9 | |
| 12 | 23 | 91 | 61 | –30 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brainina, K.; Tarasov, A.; Khamzina, E.; Kazakov, Y.; Stozhko, N. Disposable Potentiometric Sensory System for Skin Antioxidant Activity Evaluation. Sensors 2019, 19, 2586. https://doi.org/10.3390/s19112586
Brainina K, Tarasov A, Khamzina E, Kazakov Y, Stozhko N. Disposable Potentiometric Sensory System for Skin Antioxidant Activity Evaluation. Sensors. 2019; 19(11):2586. https://doi.org/10.3390/s19112586
Chicago/Turabian StyleBrainina, Khiena, Aleksey Tarasov, Ekaterina Khamzina, Yan Kazakov, and Natalia Stozhko. 2019. "Disposable Potentiometric Sensory System for Skin Antioxidant Activity Evaluation" Sensors 19, no. 11: 2586. https://doi.org/10.3390/s19112586
APA StyleBrainina, K., Tarasov, A., Khamzina, E., Kazakov, Y., & Stozhko, N. (2019). Disposable Potentiometric Sensory System for Skin Antioxidant Activity Evaluation. Sensors, 19(11), 2586. https://doi.org/10.3390/s19112586







