The Effect of UVB Irradiation and Oxidative Stress on the Skin Barrier—A New Method to Evaluate Sun Protection Factor Based on Electrical Impedance Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Skin Membranes
2.3. Narrowband UVB Irradiation
2.4. Electrical Impedance Spectroscopy (EIS) Measurements of Skin Membranes
2.5. Experimental Design
- Exposure to UVB irradiation (without additional oxidative stress from H2O2 and NaN3)
- Exposure to UVB irradiation with presence of 10 mM NaN3 in the donor and receptor solution
- Exposure to UVB irradiation with presence of 1 mM H2O2 in the donor and receptor solution
- Exposure to UVB irradiation with presence of 10 mM NaN3 and varying concentrations of H2O2 (i.e., 0.5, 1.0, 5.0, 50, 980 mM H2O2) in the donor and receptor solution
- Exposure to UVB irradiation with presence of topically applied sunscreen formulation with SPF varying between 0, 10, 20 30, 50, 70.
2.6. Histology and Microscopy
2.7. Statistical Analysis
3. Results
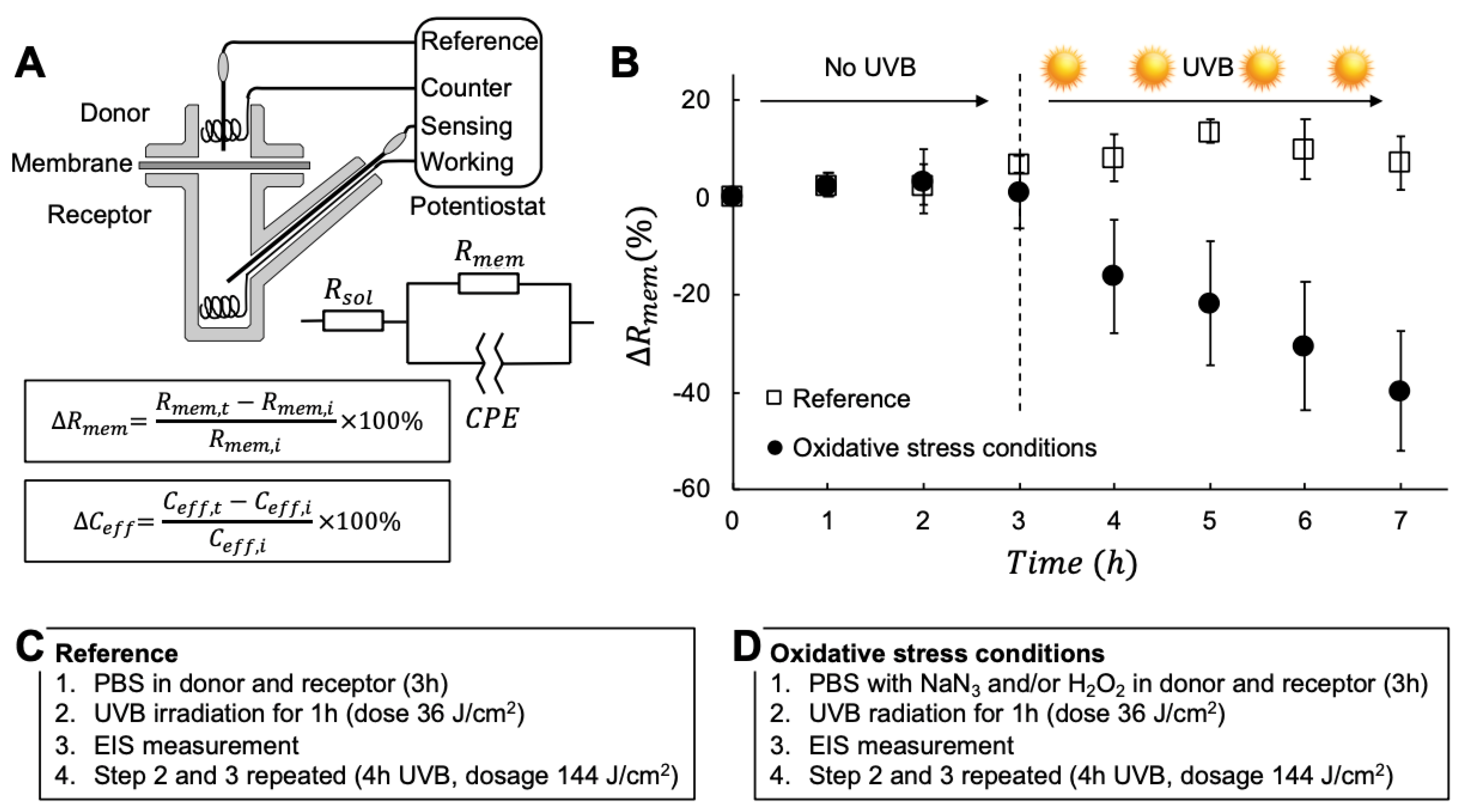
3.1. A New Protocol for Investigating UVB and Oxidative Stress with Electrical Impedance Spectroscopy
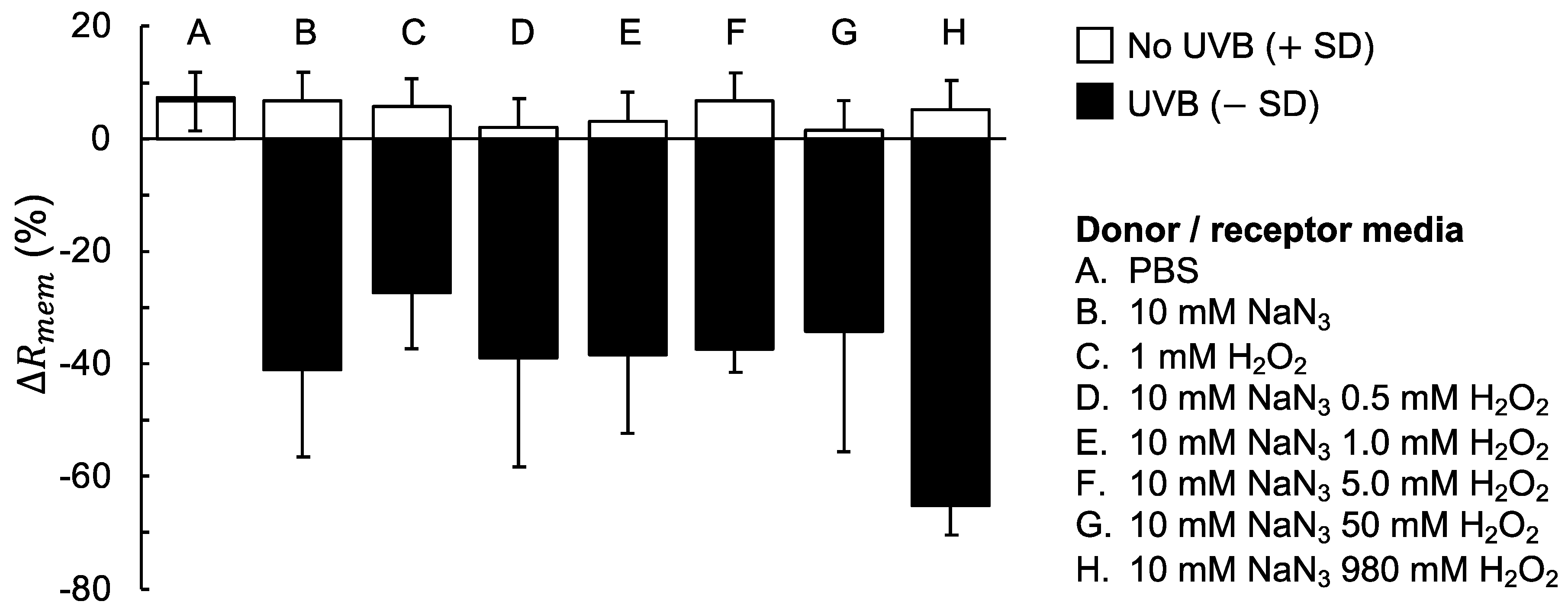
3.2. The Combined Effect of UVB Irradiation And Oxidative Stress on the Skin Barrier Electrical Properties
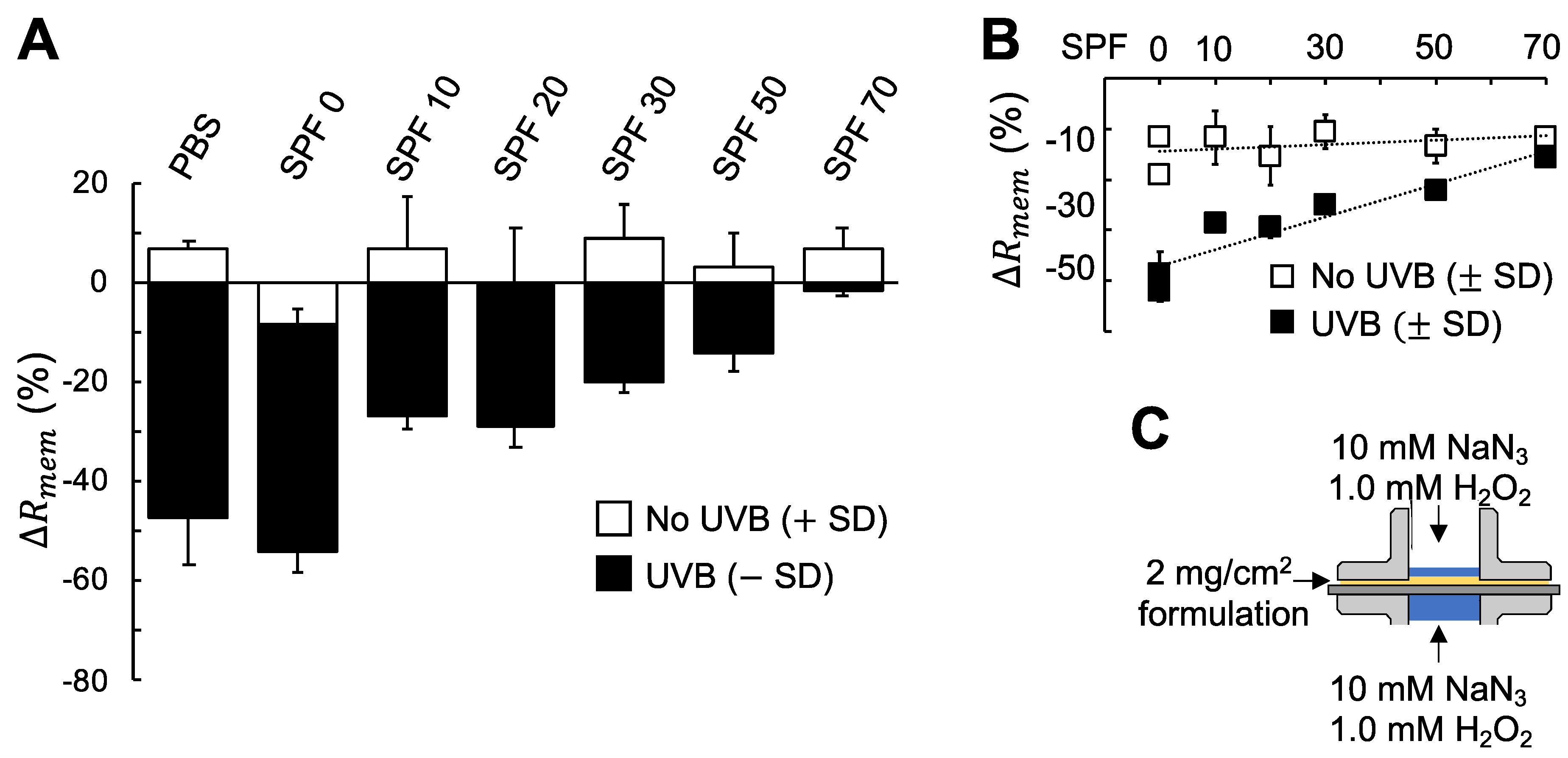
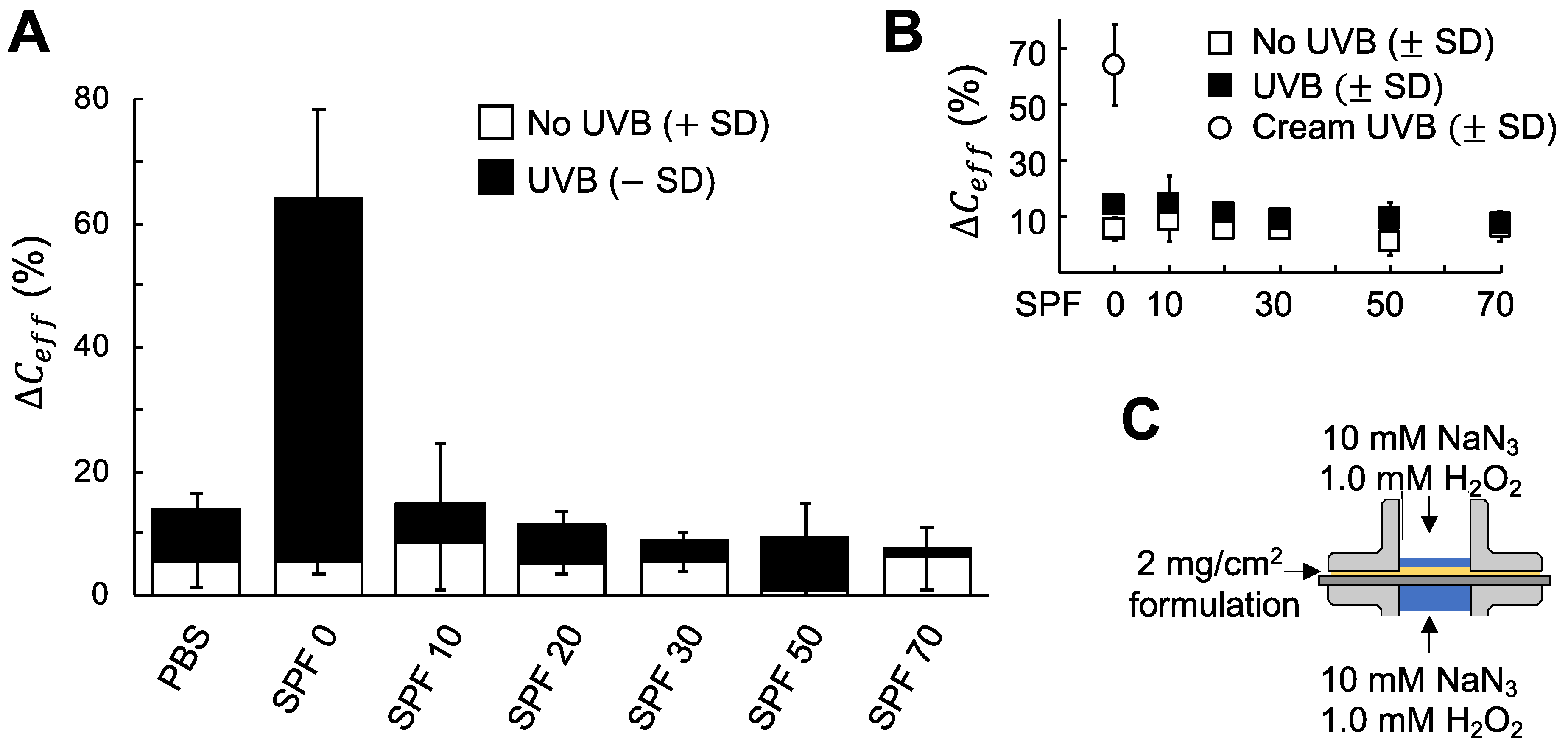
3.3. A New Method to Evaluate Sun Protection Factor (SPF) based on Electrical Impedance Spectroscopy (EIS)
3.4. UVB Irradiation in the Presence of Oxidative Stress Conditions leads to Substantial Damage of the Skin Membrane
4. Discussion
4.1. The Skin Membrane Electrical Resistance Is not Influenced by UVB Irradiation
4.2. The Combined Effect of UVB Irradiation and Oxidative Stress Results in a Significant Decrease of the Skin Membrane Electrical Resistance
4.3. Comprehensive Evaluation of the Protecting Capacity of Sunscreen Formulations against the Combined Assault Of UVB Irradiation And Oxidative Stress
5. Conclusions
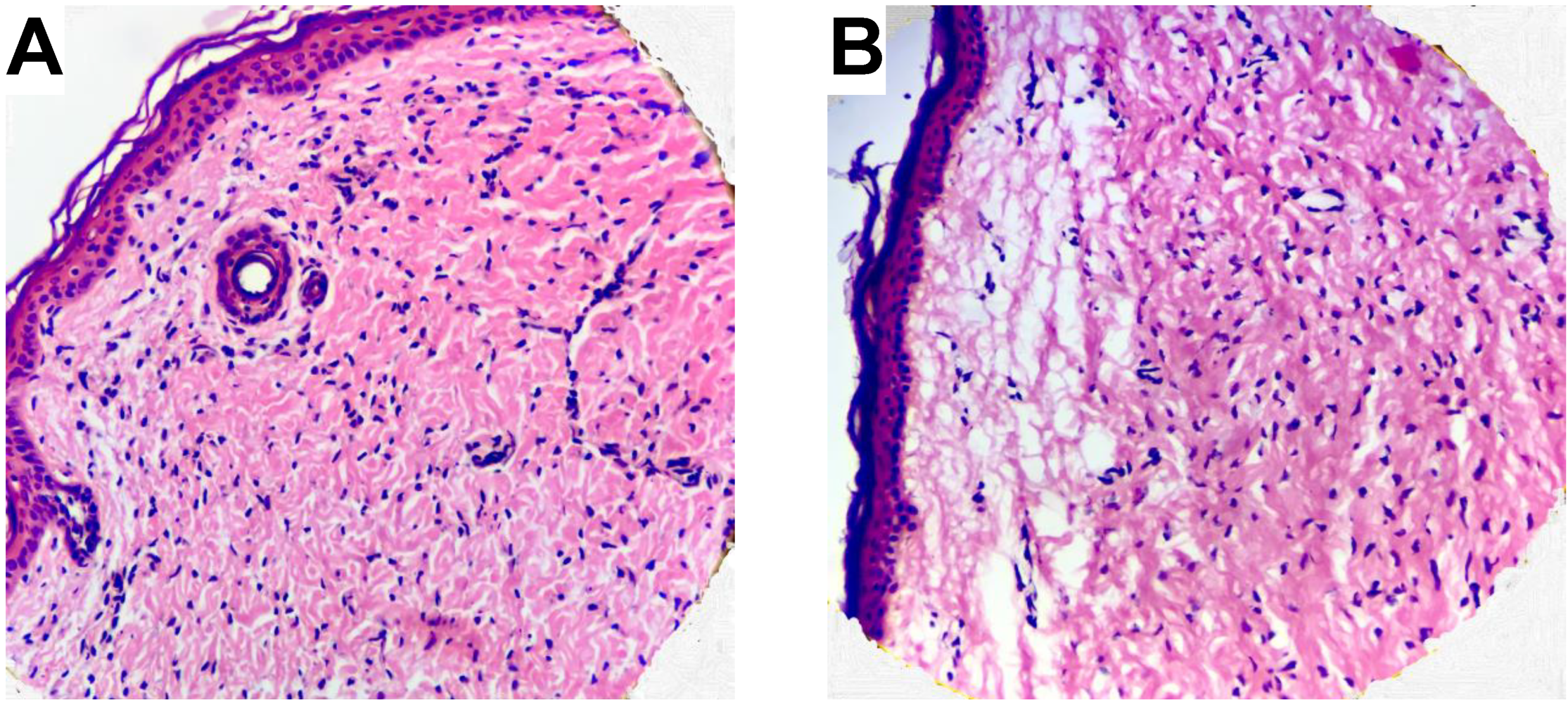
- If no UVB irradiation is applied to the skin membrane, and are not significantly affected by exposure to oxidative stress from 10 mM NaN3 and H2O2 in concentrations ranging between 0.5 mM and 980 mM (data without UVB irradiation in Figure 1, Figure 2, Figure 3 and Figure 4). This conclusion is supported by the relatively intact skin integrity observed by microscopy imaging after exposure to oxidative stress conditions (Figure 5A).
- The combined assault from UVB irradiation and oxidative stress conditions results in a significant decrease of (Figure 2). This conclusion is supported by the severe tissue damage observed by microscopy imaging after exposure to UVB irradiation in the presence of oxidative stress conditions (Figure 5B).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kupper, T.S.; Fuhlbrigge, R.C. Immune surveillance in the skin: Mechanisms and clinical consequences. Nat. Rev. Immunol. 2004, 4, 211. [Google Scholar] [CrossRef]
- Madison, K.C. Barrier Function of the Skin: “La Raison d’Être” of the Epidermis. J. Investig. Dermatol. 2003, 121, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Bickers, D.R.; Athar, M. Oxidative Stress in the Pathogenesis of Skin Disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef] [PubMed]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans—Solar and Ultraviolet Radiation. Available online: https://monographs.iarc.fr/wp-content/uploads/2018/06/mono55.pdf (accessed on 2 March 2019).
- Bulat, V.; Situm, M.; Dediol, I.; Ljubicić, I.; Bradić, L. The mechanisms of action of phototherapy in the treatment of the most common dermatoses. Coll. Antropol. 2011, 35, 147–151. [Google Scholar]
- Benedix, F.; Berneburg, M.; Röcken, M. Phototherapy with Narrowband vs Broadband UVB. Acta Derm. Venereol. 2005, 85, 98–108. [Google Scholar] [CrossRef]
- Briganti, S.; Picardo, M. Antioxidant activity, lipid peroxidation and skin diseases. What’s new. J. Eur. Acad. Dermatol. Venereol. 2003, 17, 663–669. [Google Scholar] [CrossRef]
- Biniek, K.; Levi, K.; Dauskardt, R.H. Solar UV radiation reduces the barrier function of human skin. Proc. Acad. Sci. 2012, 109, 17111–17116. [Google Scholar] [CrossRef] [PubMed]
- Wagener, F.A.D.T.G.; Carels, C.E.; Lundvig, D.M.S. Targeting the Redox Balance in Inflammatory Skin Conditions. Int. J. Mol. Sci. 2013, 14, 9126–9167. [Google Scholar] [CrossRef]
- Podda, M.; Grundmann-Kollmann, M. Low molecular weight antioxidants and their role in skin ageing. Clin. Exp. Dermatol. 2001, 26, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Glorieux, C.; Calderon, P.B. Catalase, a remarkable enzyme: Targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Boil. Chem. 2017, 398, 1095–1108. [Google Scholar] [CrossRef] [PubMed]
- Hellemans, L.; Corstjens, H.; Neven, A.; Declercq, L.; Maes, D. Antioxidant Enzyme Activity in Human Stratum Corneum Shows Seasonal Variation with an Age-Dependent Recovery. J. Investig. Dermatol. 2003, 120, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Nocchi, S.; Björklund, S.; Svensson, B.; Engblom, J.; Ruzgas, T. Electrochemical monitoring of native catalase activity in skin using skin covered oxygen electrode. Biosens. Bioelectron. 2017, 93, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Schallreuter, K.U.; Moore, J.; Wood, J.M.; Beazley, W.D.; Gaze, D.C.; Tobin, D.J.; Marshall, H.S.; Panske, A.; Panzig, E.; Hibberts, N.A. In Vivo and In Vitro Evidence for Hydrogen Peroxide (H2O2) Accumulation in the Epidermis of Patients with Vitiligo and its Successful Removal by a UVB-Activated Pseudocatalase. J. Investig. Dermatol. Symp. Proc. 1999, 4, 91–96. [Google Scholar] [CrossRef] [PubMed]
- ISO 24444: 2010 Cosmetics—Sun Protection Test Methods—In Vivo Determination of the Sun Protection Factor (SPF); ISO: Geneva, Switzerland, 2010.
- Pelizzo, M.; Zattra, E.; Nicolosi, P.; Peserico, A.; Garoli, D.; Alaibac, M. In Vitro Evaluation of Sunscreens: An Update for the Clinicians. ISRN Dermatol. 2012, 2012, 1–4. [Google Scholar] [CrossRef]
- Tatullo, M.; Marrelli, M.; Amantea, M.; Paduano, F.; Santacroce, L.; Gentile, S.; Scacco, S. Bioimpedance Detection of Oral Lichen Planus Used as Preneoplastic Model. J. Cancer 2015, 6, 976–983. [Google Scholar] [CrossRef]
- Geladi, P.; Holmgren, U.; Ollmar, S.; Aberg, P.; Nicander, I.; Hansson, J. Skin cancer identification using multifrequency electrical impedance-a potential screening tool. IEEE Trans. Biomed. Eng. 2004, 51, 2097–2102. [Google Scholar]
- Björklund, S.; Nowacka, A.; Bouwstra, J.A.; Sparr, E.; Topgaard, D. Characterization of stratum corneum molecular dynamics by natural-abundance 13C solid-state NMR. PloS ONE 2013, 8, e61889. [Google Scholar] [CrossRef]
- Björklund, S.; Ruzgas, T.; Nowacka, A.; Dahi, I.; Topgaard, D.; Sparr, E.; Engblom, J. Skin Membrane Electrical Impedance Properties under the Influence of a Varying Water Gradient. Biophys. J. 2013, 104, 2639–2650. [Google Scholar] [CrossRef]
- Hirschorn, B.; Orazem, M.E.; Tribollet, B.; Vivier, V.; Frateur, I.; Musiani, M. Determination of effective capacitance and film thickness from constant-phase-element parameters. Electrochim. Acta 2010, 55, 6218–6227. [Google Scholar] [CrossRef]
- Björklund, S.; Pham, Q.D.; Jensen, L.B.; Knudsen, N.Ø.; Nielsen, L.D.; Ekelund, K.; Ruzgas, T.; Engblom, J.; Sparr, E. The effects of polar excipients transcutol and dexpanthenol on molecular mobility, permeability, and electrical impedance of the skin barrier. J. Colloid Interface Sci. 2016, 479, 207–220. [Google Scholar] [CrossRef]
- Orazem, M.E.; Pébère, N.; Tribollet, B. Enhanced Graphical Representation of Electrochemical Impedance Data. J. Electrochem. Soc. 2006, 153, B129–B136. [Google Scholar] [CrossRef]
- Potts, R. Routes of ionic permeability through mammalian skin. Solid State Ionics 1992, 53, 165–169. [Google Scholar] [CrossRef]
- Boddé, H.; Brink, I.V.D.; Koerten, H.; De Haan, F. Visualization of in vitro percutaneous penetration of mercuric chloride; transport through intercellular space versus cellular uptake through desmosomes. J. Control. Release 1991, 15, 227–236. [Google Scholar] [CrossRef]
- Brandner, J.; Zorn-Kruppa, M.; Yoshida, T.; Moll, I.; Beck, L.; De Benedetto, A. Epidermal tight junctions in health and disease. Tissue Barriers 2014, 3, e974451. [Google Scholar] [CrossRef]
- Jeon, S.-Y.; Lee, C.-Y.; Song, K.-H.; Kim, K.-H. Spectrophotometric Measurement of Minimal Erythema Dose Sites after Narrowband Ultraviolet B Phototesting: Clinical Implication of Spetrophotometric Values in Phototherapy. Ann. Dermatol. 2014, 26, 17–25. [Google Scholar] [CrossRef][Green Version]
- Clayton, T.H.; Clark, S.M.; Turner, D.; Goulden, V. The treatment of severe atopic dermatitis in childhood with narrowband ultraviolet B phototherapy. Clin. Exp. Dermatol. Clin. Dermatol. 2007, 32, 28–33. [Google Scholar] [CrossRef]
- Jiang, S.J.; Chen, J.Y.; Lu, Z.F.; Yao, J.; Che, D.F.; Zhou, X.J. Biophysical and morphological changes in the stratum corneum lipids induced by UVB irradiation. J. Dermatol. Sci. 2006, 44, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Cezar, T.L.C.; Martinez, R.M.; Da Rocha, C.; Melo, C.P.B.; Vale, D.L.; Borghi, S.M.; Fattori, V.; Vignoli, J.A.; Camilios-Neto, D.; Baracat, M.M.; et al. Treatment with maresin 1, a docosahexaenoic acid-derived pro-resolution lipid, protects skin from inflammation and oxidative stress caused by UVB irradiation. Sci. Rep. 2019, 9, 3062. [Google Scholar] [CrossRef]
- Kalia, Y.N.; Guy, R.H. The Electrical Characteristics of Human Skin in Vivo. Pharm. Res. 1995, 12, 1605–1613. [Google Scholar] [CrossRef]
- Penven, K.; Leroy, D.; Verneuil, L.; Faguer, K.; Dompmartin, A. Evaluation of vaseline oil applied prior to UVB TL01 phototherapy in the treatment of psoriasis. Photodermatol. Photoimmunol. Photomed. 2005, 21, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Tatullo, M.; Simone, G.M.; Tarullo, F.; Irlandese, G.; De Vito, D.; Marrelli, M.; Santacroce, L.; Cocco, T.; Ballini, A.; Scacco, S. Antioxidant and Antitumor Activity of a Bioactive Polyphenolic Fraction Isolated from the Brewing Process. Sci. Rep. 2016, 6, 36042. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández, A.R.; Vallejo, B.; Ruzgas, T.; Björklund, S. The Effect of UVB Irradiation and Oxidative Stress on the Skin Barrier—A New Method to Evaluate Sun Protection Factor Based on Electrical Impedance Spectroscopy. Sensors 2019, 19, 2376. https://doi.org/10.3390/s19102376
Hernández AR, Vallejo B, Ruzgas T, Björklund S. The Effect of UVB Irradiation and Oxidative Stress on the Skin Barrier—A New Method to Evaluate Sun Protection Factor Based on Electrical Impedance Spectroscopy. Sensors. 2019; 19(10):2376. https://doi.org/10.3390/s19102376
Chicago/Turabian StyleHernández, Aura Rocio, Bibiana Vallejo, Tautgirdas Ruzgas, and Sebastian Björklund. 2019. "The Effect of UVB Irradiation and Oxidative Stress on the Skin Barrier—A New Method to Evaluate Sun Protection Factor Based on Electrical Impedance Spectroscopy" Sensors 19, no. 10: 2376. https://doi.org/10.3390/s19102376
APA StyleHernández, A. R., Vallejo, B., Ruzgas, T., & Björklund, S. (2019). The Effect of UVB Irradiation and Oxidative Stress on the Skin Barrier—A New Method to Evaluate Sun Protection Factor Based on Electrical Impedance Spectroscopy. Sensors, 19(10), 2376. https://doi.org/10.3390/s19102376




