Membrane-Based Microwave-Mediated Electrochemical Immunoassay for the In Vitro, Highly Sensitive Detection of Osteoporosis-Related Biomarkers
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Materials
2.2. Preparation of Membrane Disks
2.3. Custom-Made Well Titer
2.4. Custom-Made Gold Electrode
2.5. In Vitro Measurement
3. Results
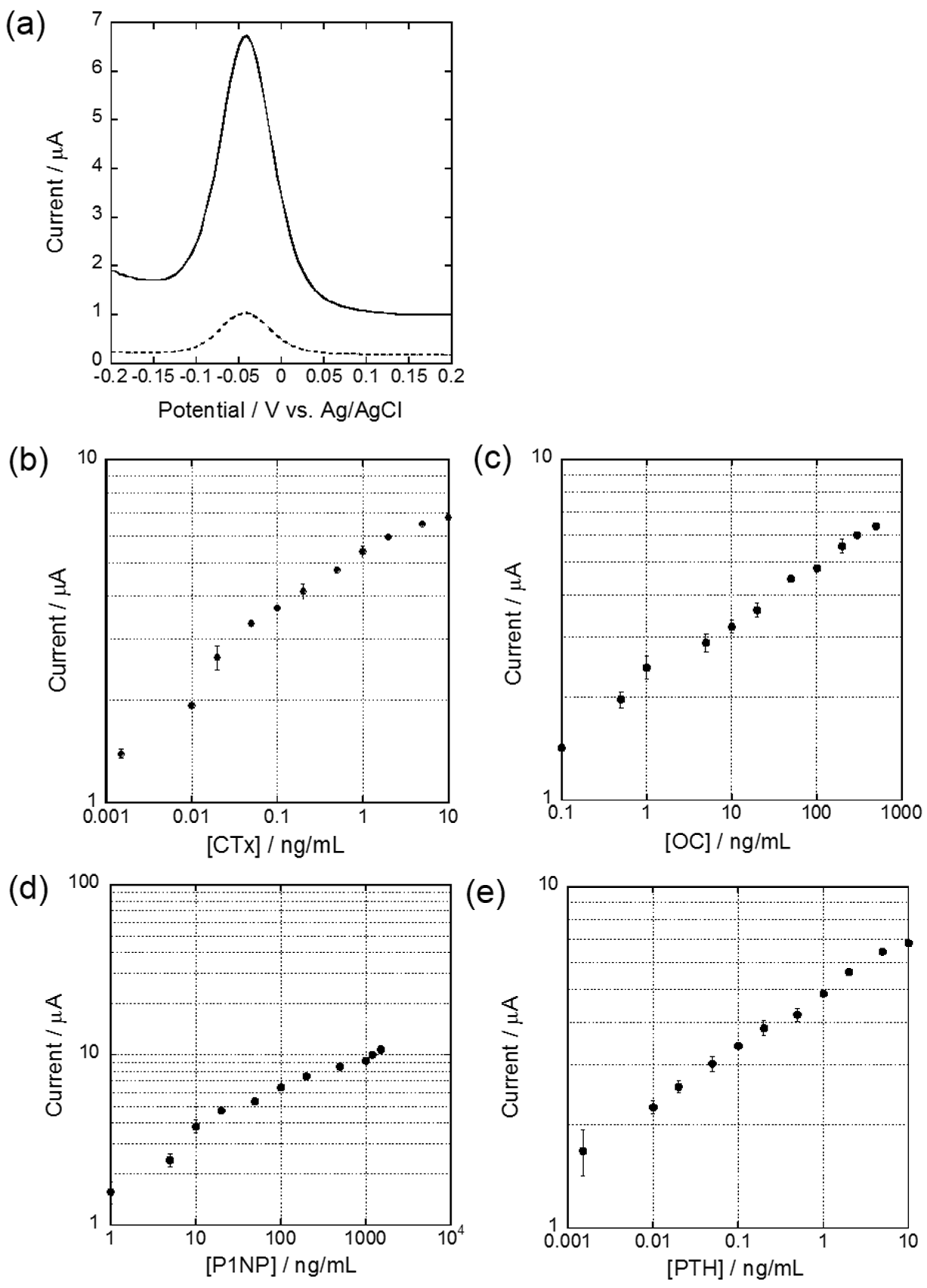
3.1. DPV Measurement
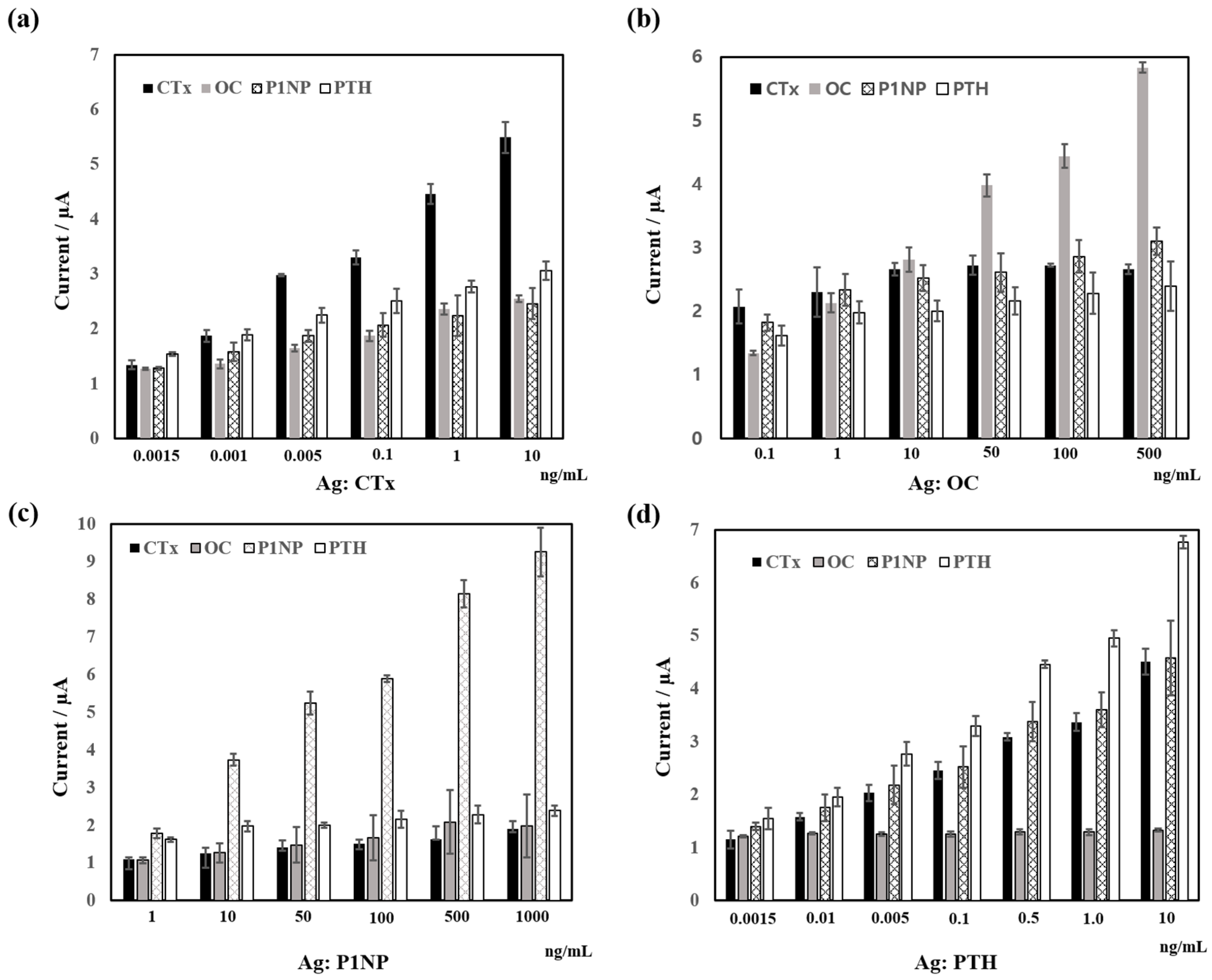
3.2. Cross Reactivity
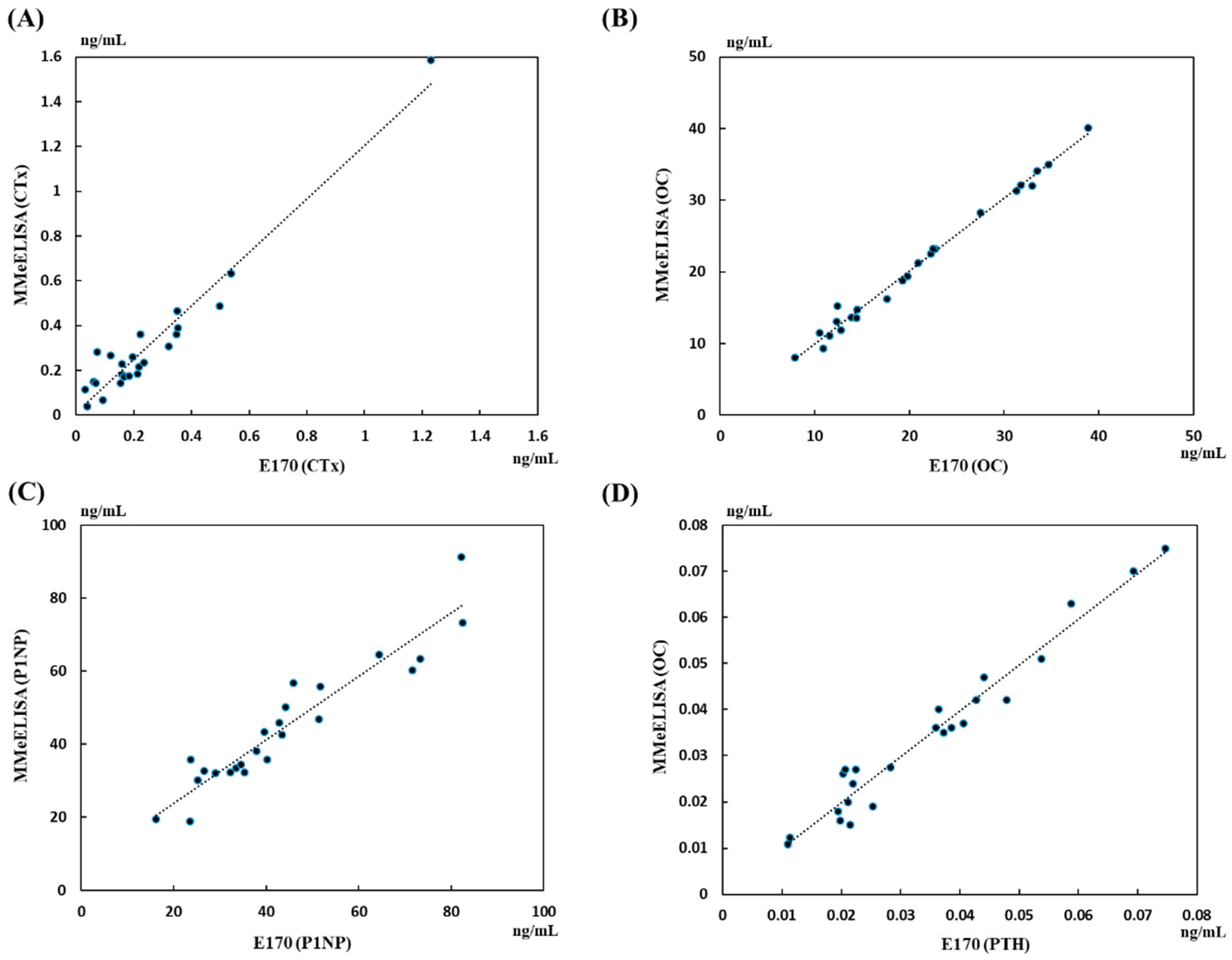
3.3. Analysis of Biomedical Markers in Human Serum
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aloia, J.F.; Feuerman, M.; Yeh, J.K. Reference range for serum parathyroid hormone. Endocr. Pract. 2006, 12, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Ramos, R.M.L.; Armann, J.A.; Galeano, N.A.; Hernandez, A.M.; Gomez, J.M.G.; Molinero, J.G. Dual energy X-ray absorptiometry: Fundamentals, methodology, and clinical applications. Radiologia 2012, 54, 410–423. [Google Scholar]
- Barco, C.M.R.; Arjia, S.M.; Perez, M.R. Biochemical markers in osteoporosis: Usefulness in clinical practice. Reumatol. Clin. 2012, 8, 149–152. [Google Scholar]
- Biver, E.; Chopin, F.; Coiffier, G.; Brentano, T.F.; Bouvard, B.; Garnero, P.; Cortet, B. Bone turnover markers for osteoporotic status assessment? A systematic review of their diagnosis value at baseline in osteoporosis. Jt. Bone Spine 2012, 79, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Chopin, F.; Biver, E.; Funck-Brentano, T.; Bouvard, B.; Coiffier, G.; Garnero, P.; Thomas, T. Prognostic interest of bone turnover markers in the management of postmenopausal osteoporosis. Jt. Bone Spine 2012, 79, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Piatek, S.; Adolf, D.; Wex, T.; Halangk, W.; Klose, S.; Westphal, S.; Amthauer, H.; Winckler, S. Multiparameter analysis of serum levels of c-telopeptide crosslaps, bone-specific alkaline phosphatase, cathepsin k, osteoprotegerin and receptor activator of nuclear factor κb ligand in the diagnosis of osteoporosis. Maturitas 2013, 74, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.C.; Garnero, P.; Bilezikian, J.P.; Greenspan, S.L.; Ensrud, K.E.; Rosen, C.J.; Palermo, L.; Black, D.M. Short-term changes in bone turnover markers and bone mineral density response to parathyroid hormone in postmenopausal women with osteoporosis. J. Clin. Endocrinol. Metab. 2006, 91, 1370–1375. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Kumar, D.; Lal, A.K. Serum osteocalcin as a diagnostic biomarker for primary osteoporosis in women. J. Clin. Diagn. Res. 2015, 9, RC04–RC07. [Google Scholar] [CrossRef] [PubMed]
- Watts, N.B. Clinical utility of biochemical markers of bone remodeling. Clin. Chem. 1999, 45, 1359–1368. [Google Scholar] [PubMed]
- Kalaiselvi, V.S.; Prabhu, K.; Mani Ramesh, V.V. The association of serum osteocalcin with the bone mineral density in post menopausal women. J. Clin. Diagn. Res. 2013, 7, 814–816. [Google Scholar]
- Stokes, F.J.; Ivanov, P.; Bailey, L.M.; Fraser, W.D. The effects of sampling procedures and storage conditions on short-term stability of blood-based biochemical markers of bone metabolism. Clin. Chem. 2010, 57, 138–140. [Google Scholar] [CrossRef] [PubMed]
- Vasikaran, S.; Eastell, R.; Bruyère, O.; Foldes, A.J.; Garnero, P.; Griesmacher, A.; McClung, M.; Morris, H.A.; Silverman, S.; Trenti, T.; et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: A need for international reference standards. Osteoporous. Int. 2011, 22, 391–420. [Google Scholar] [CrossRef] [PubMed]
- Warsinke, A.; Benkert, A.; Scheller, F.W. Electrochemical immunoassays. Fresenius J. Anal. Chem. 2000, 366, 622–634. [Google Scholar] [CrossRef] [PubMed]
- Qiana, Y.; Fana, T.; Wang, P.; Zhang, X.; Luo, J.; Zhou, F.; Yao, Y.; Liao, X.; Li, Y.; Gao, F. A novel label-free homogeneous electrochemical immunosensorbased on proximity hybridization-triggered isothermal exponentialamplification induced g-quadruplex formation. Sens. Actuators B Chem. 2017, 248, 187–194. [Google Scholar] [CrossRef]
- Gao, F.; Zhou, F.; Chen, S.; Yao, Y.; Wu, J.; Yin, D.; Geng, D.; Wang, P. Proximity hybridization triggered rolling-circle amplification for sensitive electrochemical homogeneous immunoassay. Analysts 2017, 142, 4308–4316. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Yao, Y.; Luo, J.; Zhang, X.; Zhang, Y.; Yin, D.; Gao, F.; Wang, P. Proximity hybridization-regulated catalytic DNA hairpin assembly for electrochemical immunoassay based on in situ DNA templatesynthesized pd nanoparticles. Anal. Chim. Acta 2017, 969, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Khashayar, P.; Amoabediny, G.; Hosseini, M.; Verplancke, R.; Razi, F.; Vanfleteren, J.; Larijani, B. An electrochemical biosensor based on aunp-modified gold electrodes for selective determination of serum levels of osteocalcin. IEEE Sens. J. 2017, 17, 3367–3374. [Google Scholar] [CrossRef]
- Kim, H.-U.; Kim, H.Y.; Kulkarni, A.; Ahn, C.; Jin, Y.; Kim, Y.; Lee, K.-N.; Lee, M.-H.; Kim, T. A sensitive electrochemical sensor for in vitro detection of parathyroid hormone based on a mos2-graphene composite. Sci. Rep. 2016, 6, 34587. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-E.; Cortés-Salazar, F.; Lesch, A.; Qiao, L.; Bondarenko, A.; Girault, H.H. Multiple scanning electrochemical microscopy mapping of tyrosinase in micro-contact printed fruit samples on polyvinylidene fluoride membrane. Electrochim. Acta 2015, 179, 57–64. [Google Scholar] [CrossRef]
- Holstein, C.A.; Chevalier, A.; Bennett, S.; Anderson, C.E.; Keniston, K.; Olsen, C.; Li, B.; Bales, B.; Moore, D.R.; Fu, E.; et al. Immobilizing affinity proteins to nitrocellulose: A toolbox for paper-based assay developers. Anal. Bioanal. Chem. 2015, 408, 1335–1346. [Google Scholar] [CrossRef] [PubMed]
- Diala, I.; Sato, S.; Usui, M.; Nakashima, K.; Nishihara, T.; Takenaka, S. Development of a membrane-based microwave-mediated electrochemical elisa method for tnf-alpha detection in patients with periodontitis. Anal. Sci. 2013, 29, 927–930. [Google Scholar] [CrossRef] [PubMed]
- Frew, J.E.; Foulds, N.C.; Wilshere, J.M.; Forrow, N.J.; Green, M.J. Measurement of alkaline phosphatase activity by electrochemical detection of phosphate esters: Application to amperometric enzyme immunoassay. J. Electr. Chem. Interfacial Electrochem. 1989, 266, 309–316. [Google Scholar] [CrossRef]
- Michelsen, J.; Wallaschofski, H.; Friedrich, N.; Spielhagen, C.; Rettig, R.; Ittermann, T.; Nauck, M.; Hannemann, A. Reference intervals for serum concentrations of three bone turnover markers for men and women. Bone 2013, 57, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Hannemann, A.; Friedrich, N.; Spielhagen, C.; Rettig, R.; Ittermann, T.; Nauck, M.; Wallaschofski, H. Reference intervals for serum osteocalcin concentrations in adult men and women from the study of health in pomerania. BMC Endocr. Disord. 2013, 13, 11. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Lein, M. Bone turnover markers in serum and urine as diagnostic, prognostic and monitoring biomarkers of bone metastasis. Biochim. Biophys. Acta 2014, 1846, 425–438. [Google Scholar] [CrossRef] [PubMed]
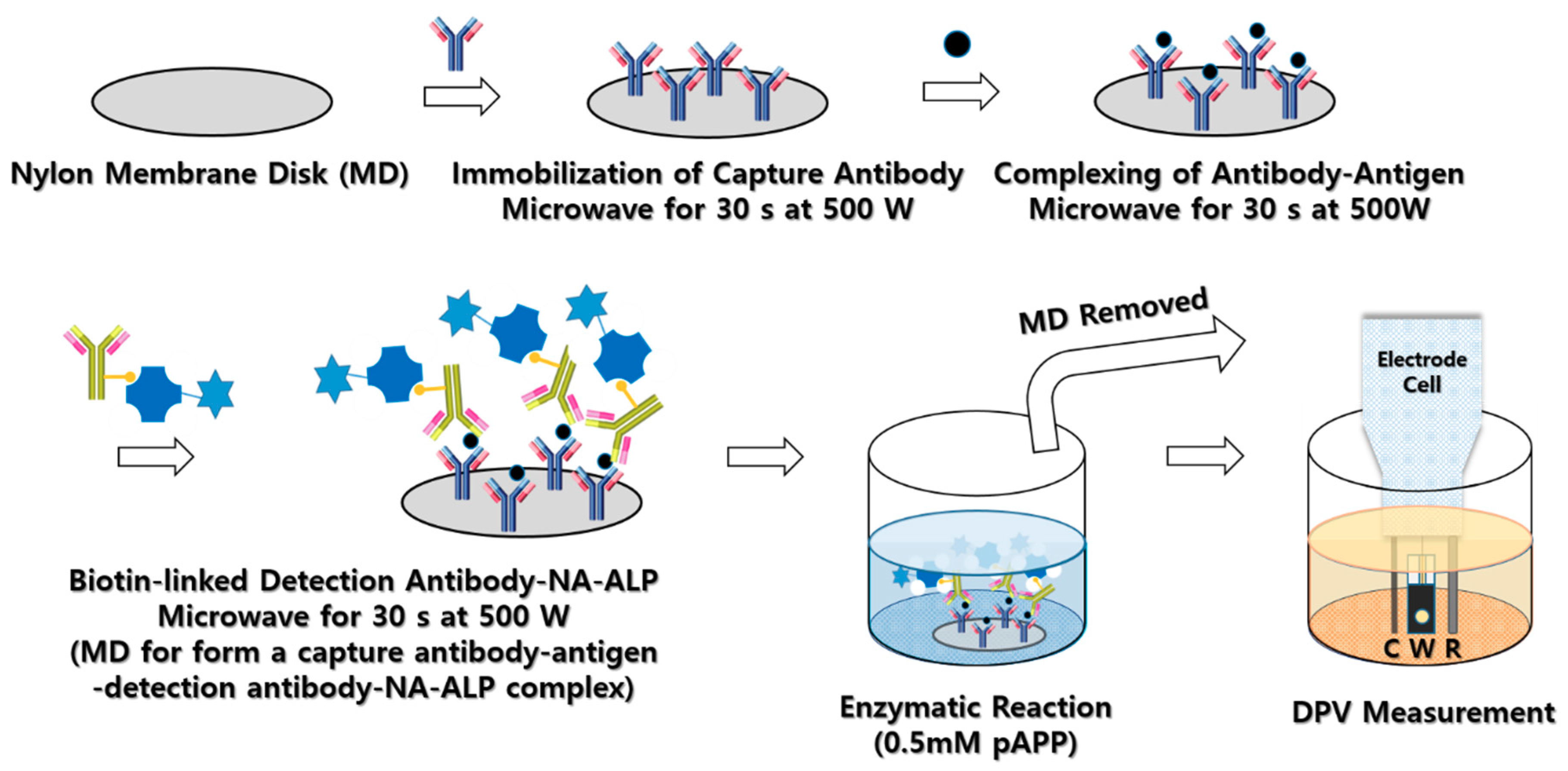
| Capture Antibodies | Antigen | Detection Antibodies | |
|---|---|---|---|
| CTx | Monoclonal antibody to cross-linked CTx | BSA conjugated CTx | Biotin-linked polyclonal antibody to CTx |
| OC | OC monoclonal antibody | Human OC full length protein | OC polyclonal antibody, biotin conjugated |
| P1NP | Monoclonal antibody to P1NP | OVA conjugated P1NP | Biotin-linked polyclonal antibody to P1NP |
| PTH | Mouse monoclonal PTH antibody | Recombinant human PTH protein | Anti-PTH antibody (Biotin) |
| Intra-Assay CV (%) | Inter-Assay CV (%) | LLOD | HLOQ | |
|---|---|---|---|---|
| CTx | 1.25–5.69 | 1.87–7.85 | 1 pg/mL | 10 ng/mL |
| OC | 2.63–7.67 | 1.20–6.59 | 100 pg/mL | 500 ng/mL |
| P1NP | 0.51–7.01 | 0.29–4.94 | 1 ng/mL | 1500 ng/mL |
| PTH | 1.02–8.16 | 1.20–3.45 | 1 pg/mL | 10 ng/mL |
| Cross Reactivity Rate 1 (%) | |||||
|---|---|---|---|---|---|
| Target Antigen | CTx | OC | P1NP | PTH | |
| Pair Antibody | |||||
| CTx | 100 | 30.8 | 28.4 | 36.6 | |
| OC | 13.1 | 100 | 28.5 | 17.1 | |
| P1NP | 10.7 | 12.2 | 100 | 19.0 | |
| PTH | 64.4 | 2.3 | 60.9 | 100 | |
| No. | 1st | 2nd | 3rd | 4th | 5th | 6th | 7th | 8th |
|---|---|---|---|---|---|---|---|---|
| 1 | 2.0 | 2.1 | 2.1 | 2.6 | −0.3 | −0.6 | 1.1 | 0.9 |
| 2 | −2.8 | −2.2 | −1.4 | −0.7 | −1.9 | −2.2 | −0.7 | −1.2 |
| 3 | −0.9 | −0.3 | −0.6 | −1.2 | −1.2 | −1.6 | −0.3 | −1.2 |
| 4 | −1.3 | −1.2 | −0.7 | −0.7 | −0.6 | −1.5 | 0.1 | 0.1 |
| 5 | −1.9 | −2.6 | −3.0 | −2.5 | −1.4 | −1.6 | −0.3 | −0.8 |
| 6 | −0.6 | −0.4 | −−0.1 | −0.8 | −1.0 | −1.2 | 0.4 | 0.2 |
| 7 | −1.3 | −1.4 | −0.8 | 0.0 | −1.2 | −1.6 | 0.0 | −0.1 |
| 8 | −2.7 | −3.7 | −3.2 | −2.8 | −1.1 | −1.7 | −1.2 | −0.8 |
| 9 | −1.0 | −0.1 | −0.1 | −0.1 | −1.4 | −2.2 | −0.1 | −0.5 |
| 10 | −1.0 | −1.1 | −1.2 | −1.1 | −0.9 | −1.7 | −0.9 | −0.7 |
| 11 | −2.2 | −2.4 | −2.6 | −1.1 | −2.0 | −2.5 | −1.0 | −1.4 |
| 12 | −0.6 | 0.0 | −0.5 | 0.1 | −1.0 | −1.4 | 0.3 | 0.2 |
| 13 | −1.2 | −1.5 | −1.4 | −1.0 | −1.7 | −2.4 | −0.8 | −1.0 |
| 14 | −1.6 | −1.2 | −1.7 | −0.8 | −0.6 | −1.6 | 0.0 | 0.1 |
| 15 | −1.6 | 0.0 | 0.5 | −1.6 | −1.6 | −2.1 | −0.9 | −1.1 |
| 16 | −1.5 | −0.8 | −0.3 | −1.0 | −0.5 | −2.5 | −1.1 | −1.1 |
| 17 | −1.4 | −2.9 | −2.8 | −1.7 | −0.5 | −1.7 | −0.2 | −0.3 |
| 18 | −0.8 | −0.7 | 0.5 | 0.7 | −1.9 | −2.7 | 0.4 | −0.8 |
| 19 | −1.6 | −1.7 | −1.6 | −1.5 | −1.3 | −1.7 | −1.1 | −0.9 |
| 20 | −2.8 | −2.2 | −1.7 | −2.1 | −1.5 | −2.5 | −0.9 | −1.2 |
| 21 | −3.2 | −2.7 | −3.0 | −2.7 | −1.7 | −1.9 | −1.3 | −1.4 |
| 22 | −2.3 | −2.7 | −2.9 | −1.3 | −2.1 | −2.4 | −1.4 | −1.3 |
| 23 | −2.3 | −3.0 | −3.0 | −2.7 | −1.3 | −2.1 | −1.3 | −1.1 |
| 24 | −1.3 | −2.4 | −2.0 | −2.2 | −1.9 | −2.5 | −0.6 | −0.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.Y.; Sato, S.; Takenaka, S.; Lee, M.-H. Membrane-Based Microwave-Mediated Electrochemical Immunoassay for the In Vitro, Highly Sensitive Detection of Osteoporosis-Related Biomarkers. Sensors 2018, 18, 2933. https://doi.org/10.3390/s18092933
Kim HY, Sato S, Takenaka S, Lee M-H. Membrane-Based Microwave-Mediated Electrochemical Immunoassay for the In Vitro, Highly Sensitive Detection of Osteoporosis-Related Biomarkers. Sensors. 2018; 18(9):2933. https://doi.org/10.3390/s18092933
Chicago/Turabian StyleKim, Hye Youn, Shinobu Sato, Shigeori Takenaka, and Min-Ho Lee. 2018. "Membrane-Based Microwave-Mediated Electrochemical Immunoassay for the In Vitro, Highly Sensitive Detection of Osteoporosis-Related Biomarkers" Sensors 18, no. 9: 2933. https://doi.org/10.3390/s18092933
APA StyleKim, H. Y., Sato, S., Takenaka, S., & Lee, M.-H. (2018). Membrane-Based Microwave-Mediated Electrochemical Immunoassay for the In Vitro, Highly Sensitive Detection of Osteoporosis-Related Biomarkers. Sensors, 18(9), 2933. https://doi.org/10.3390/s18092933





