Preparation of Quasi-Three-Dimensional Porous Ag and Ag-NiO Nanofibrous Mats for SERS Application
Abstract
1. Introduction
2. Experimental Section
2.1. Reagents and Chemicals
2.2. Instruments and Apparatus
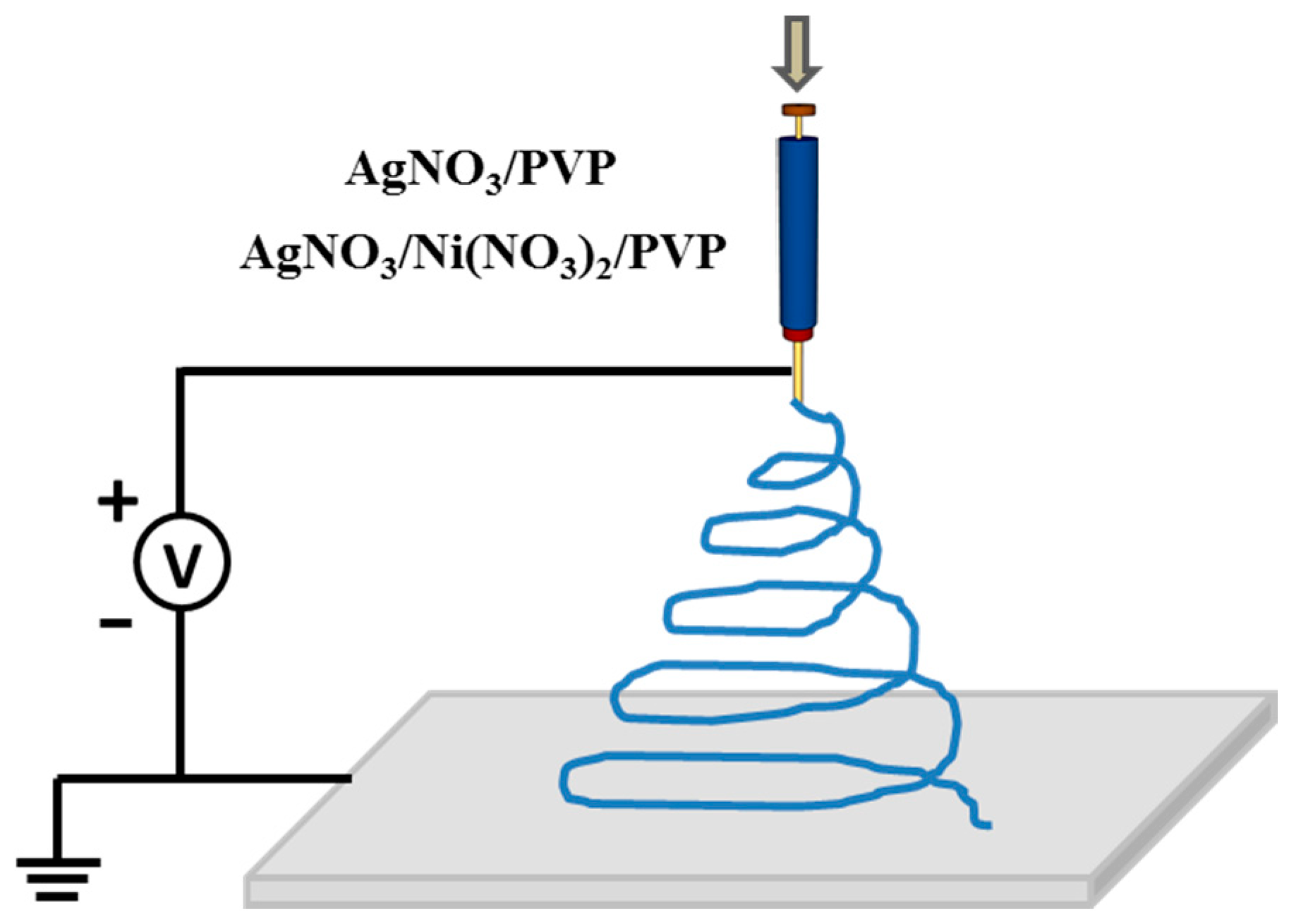
2.3. Preparation of Quasi-3D Porous Ag and Ag-NiO Nanofibers Mats
2.4. Sample Preparation and Measurement Procedure
3. Results and Discussions
3.1. Nanofber Characterization
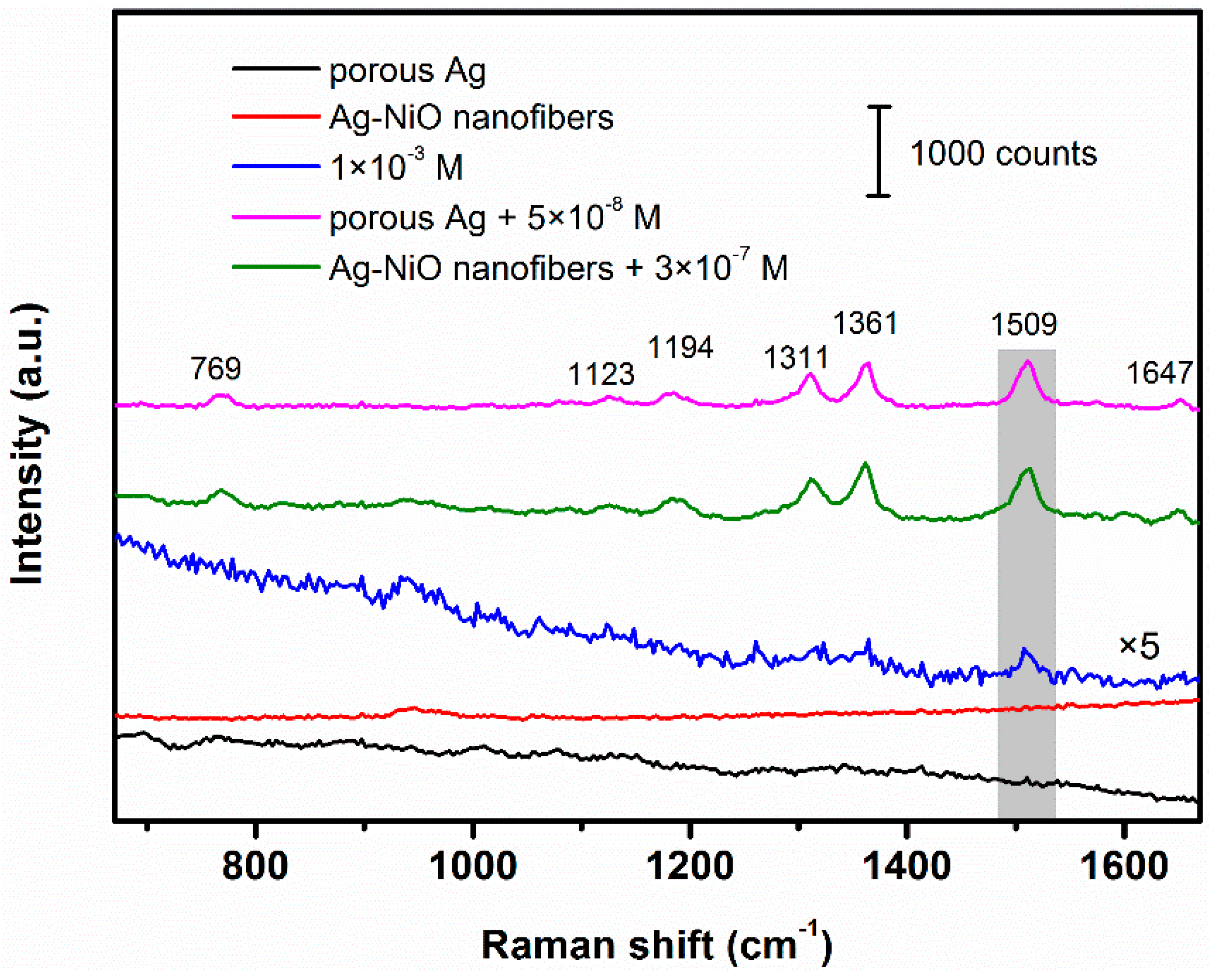
3.2. SERS Performance of Porous Ag and Ag-NiO Nanofibers
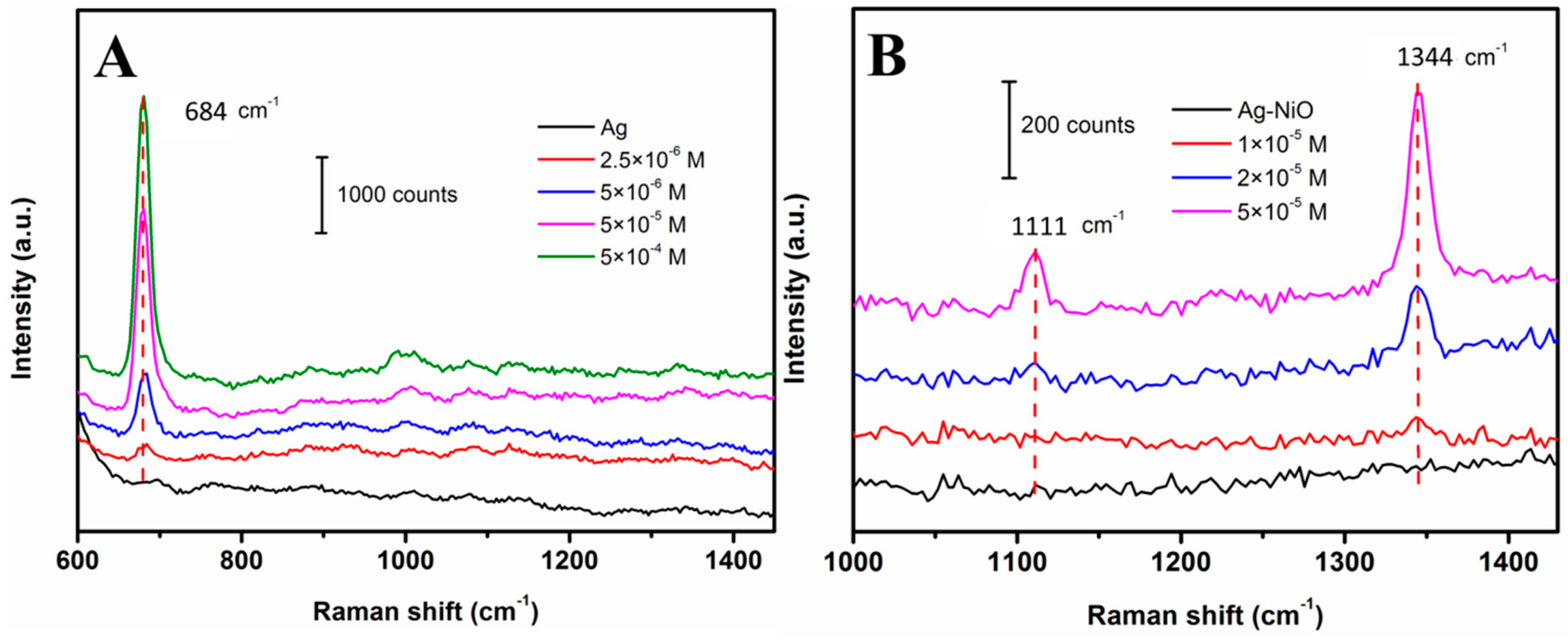
3.3. SERS Detection for Melamine and Methyl Parathion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, W.; Fang, Z.; Zhu, X. Near-Field Raman Spectroscopy with Aperture Tips. Chem. Rev. 2016, 117, 5095–5109. [Google Scholar] [CrossRef] [PubMed]
- Zrimsek, A.B.; Chiang, N.; Mattei, M.; Zaleski, S.; McAnally, M.O.; Chapman, C.T.; Henry, A.I.; Schatz, G.C.; Van Duyne, R.P. Single-Molecule Chemistry with Surface- and Tip-Enhanced Raman Spectroscopy. Chem. Rev. 2017, 117, 7583–7613. [Google Scholar] [CrossRef] [PubMed]
- Madzharova, F.; Heiner, Z.; Kneipp, J. Surface Enhanced Hyper-Raman Scattering of the Amino Acids Tryptophan, Histidine, Phenylalanine, and Tyrosine. J. Phys. Chem. C 2017, 121, 1235–1242. [Google Scholar] [CrossRef]
- Fazio, B.; D’Andrea, C.; Foti, A.; Messina, E.; Irrera, A.; Donato, M.G.; Villari, V.; Micali, N.; Marago, O.M.; Gucciardi, P.G. SERS detection of Biomolecules at Physiological pH via aggregation of Gold Nanorods mediated by Optical Forces and Plasmonic Heating. Sci. Rep. 2016, 6, 26952. [Google Scholar] [CrossRef] [PubMed]
- Fleischmann, M.; Hendra, P.J.; Mcquillan, A.J. Raman spectra of pyridine adsorbed at a silver electrode. Chem. Phys. Lett. 1974, 26, 163–166. [Google Scholar] [CrossRef]
- Jeanmaire, D.L.; Duyne, R.P.V. Surface raman spectroelectrochemistry: Part I. Heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. Interfacial Electrochem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Yang, L.; Liu, H.; Wang, J.; Zhou, F.; Tian, Z.; Liu, J. Metastable state nanoparticle-enhanced Raman spectroscopy for highly sensitive detection. Chem. Commun. 2011, 47, 3583–3585. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Wang, F.; Lan, T.; Zhang, Y.; Shao, Z. Convenient fabrication of carboxymethyl cellulose electrospun nanofibers functionalized with silver nanoparticles. Cellulose 2016, 23, 1899–1909. [Google Scholar] [CrossRef]
- Aoki, P.H.B.; Furini, L.N.; Alessio, P.; Aliaga, A.E.; Constantino, C.J.L. Surface-enhanced Raman scattering (SERS) applied to cancer diagnosis and detection of pesticides, explosives, and drugs. Rev. Anal. Chem. 2013, 32, 55–76. [Google Scholar] [CrossRef]
- Cañamares, M.V.; Feis, A. Surface-enhanced Raman spectra of the neonicotinoid pesticide thiacloprid. J. Raman Spectrosc. 2013, 44, 1126–1135. [Google Scholar] [CrossRef]
- Craig, A.P.; Franca, A.S.; Irudayaraj, J. Surface-Enhanced Raman Spectroscopy Applied to Food Safety. Annu. Rev. Food Sci. Technol. 2013, 4, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Grasseschi, D.; Parussulo, A.L.A.; Zamarion, V.M.; Guimarães, R.R.; Araki, K.; Toma, H.E. How relevant can the SERS effect in isolated nanoparticles be? RSC Adv. 2013, 3, 24465–24472. [Google Scholar] [CrossRef]
- Le Ru, E.C.; Meyer, M.; Etchegoin, P.G. Proof of single-molecule sensitivity in surface enhanced Raman scattering (SERS) by means of a two-analyte technique. J. Phys. Chem. B 2006, 110, 1944–1948. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Andrade, G.F.; Brolo, A.G. A review on the fabrication of substrates for surface enhanced Raman spectroscopy and their applications in analytical chemistry. Anal. Chim. Acta 2011, 693, 7–25. [Google Scholar] [CrossRef] [PubMed]
- Banholzer, M.J.; Millstone, J.E.; Qin, L.; Mirkin, C.A. Rationally designed nanostructures for surface-enhanced Raman spectroscopy. Chem. Soc. Rev. 2008, 37, 885–897. [Google Scholar] [CrossRef] [PubMed]
- Schwartzberg, A.M.; Grant, C.D.; Wolcott, A.; Talley, C.E.; Huser, T.R.; Bogomolni, R.; Zhang, J.Z. Unique Gold Nanoparticle Aggregates as a Highly Active Surface-Enhanced Raman Scattering Substrate. J. Phys. Chem. B 2004, 108, 19191–19197. [Google Scholar] [CrossRef]
- Blakey, I.; Merican, Z.; Thurecht, K.J. A method for controlling the aggregation of gold nanoparticles: Tuning of optical and spectroscopic properties. Langmuir ACS J. Surf. Colloids 2013, 29, 8266–8274. [Google Scholar] [CrossRef] [PubMed]
- Khoury, C.G.; Vo-Dinh, T. Gold nanostars for surface-enhanced Raman scattering: Synthesis, characterization and optimization. J. Phys. Chem. C 2008, 112, 18849–18859. [Google Scholar] [CrossRef]
- Zhang, X.; Zhao, J.; Whitney, A.V.; Elam, J.W.; Duyne, R.P.V. Ultrastable Substrates for Surface-Enhanced Raman Spectroscopy: Al2O3 Overlayers Fabricated by Atomic Layer Deposition Yield Improved Anthrax Biomarker Detection. J. Am. Chem. Soc. 2006, 128, 10304–10309. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Stagon, S.; Huang, H.; Chen, J.; Lei, Y. Functionalized aligned silver nanorod arrays for glucose sensing through surface enhanced Raman scattering. RSC Adv. 2014, 4, 23382–23388. [Google Scholar] [CrossRef]
- Abu Hatab, N.A.; Oran, J.M.; Sepaniak, M.J. Surface-enhanced Raman spectroscopy substrates created via electron beam lithography and nanotransfer printing. ACS Nano 2008, 2, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Mcfarland, A.D.; Young, M.A.; Dieringer, J.A.; Duyne, R.P.V. Wavelength-scanned surface-enhanced Raman excitation spectroscopy. J. Phys. Chem. B 2005, 109, 11279–11285. [Google Scholar] [CrossRef] [PubMed]
- Brolo, A.G.; Arctander, E.; Gordon, R.; Leathem, B.; Kavanagh, K.L. Nanohole-enhanced Raman scattering. Nano Lett. 2004, 4, 2015–2018. [Google Scholar] [CrossRef]
- Ding, Y.; Wang, Y.; Zhang, L.; Zhang, H.; Li, C.M.; Lei, Y. Preparation of TiO2–Pt Hybrid nanofibers and their application for sensitive hydrazine detection. Nanoscale 2011, 3, 1149–1157. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.L.; Lv, K.P.; Cong, H.P.; Yu, S.H. Controlled Assemblies of Gold Nanorods in PVA Nanofiber Matrix as Flexible Free-Standing SERS Substrates by Electrospinning. Small 2012, 8, 648–653. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yan, Z.; Jia, L.; Song, P.; Mei, L.; Bai, L.; Liu, Y. Gold nanoparticle decorated electrospun nanofibers: A 3D reproducible and sensitive SERS substrate. Appl. Surf. Sci. 2017, 403, 29–34. [Google Scholar] [CrossRef]
- Li, D.; Xia, Y. Electrospinning of nanofibers: Reinventing the wheel? Adv. Mater. 2004, 16, 1151–1170. [Google Scholar] [CrossRef]
- Bhardwaj, N.; Kundu, S.C. Electrospinning: A fascinating fiber fabrication technique. Biotechnol. Adv. 2010, 28, 325–347. [Google Scholar] [CrossRef] [PubMed]
- Roque-Ruiz, J.H.; Martínez-Máynez, H.; Zalapa-Garibay, M.A.; Arizmendi-Moraquecho, A.; Farias, R.; Reyes-López, S.Y. Surface enhanced Raman spectroscopy in nanofibers mats of SiO2-TiO2-Ag. Results Phys. 2017, 7, 2520–2527. [Google Scholar] [CrossRef]
- Liou, P.; Nayigiziki, F.X.; Kong, F.; Mustapha, A.; Lin, M. Cellulose nanofibers coated with silver nanoparticles as a SERS platform for detection of pesticides in apples. Carbohydr. Polym. 2017, 157, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Camposeo, A.; Spadaro, D.; Magri, D.; Moffa, M.; Gucciardi, P.G.; Persano, L.; Marago, O.M.; Pisignano, D. Surface-enhanced Raman spectroscopy in 3D electrospun nanofiber mats coated with gold nanorods. Anal. Bioanal. Chem. 2016, 408, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Tang, Y.; Vlahovic, B.; Yan, F. Electrospun Polymer Nanofibers Decorated with Noble Metal Nanoparticles for Chemical Sensing. Nanoscale Res. Lett. 2017, 12, 451. [Google Scholar] [CrossRef] [PubMed]
- Prikhozhdenko, E.; Lengert, E.; Parakhonskiy, B.; Gorin, D.A.; Sukhorukov, G.B.; Yashchenok, A. Biocompatible chitosan nanofibers functionalized with silver nanoparticles for SERS based detection. Acta Phys. Pol. A 2016, 129, 247–249. [Google Scholar] [CrossRef]
- He, D.; Hu, B.; Yao, Q.-F.; Wang, K.; Yu, S.-H. Large-scale synthesis of flexible free-standing SERS substrates with high sensitivity: Electrospun PVA nanofibers embedded with controlled alignment of silver nanoparticles. ACS Nano 2009, 3, 3993–4002. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wang, Y.; Su, L.; Zhang, H.; Lei, Y. Preparation and characterization of NiO–Ag nanofibers, NiO nanofibers, and porous Ag: Towards the development of a highly sensitive and selective non-enzymatic glucose sensor. J. Mater. Chem. 2010, 20, 9918–9926. [Google Scholar] [CrossRef]
- Wu, H.; Lin, D.; Zhang, R.; Pan, W. Facile synthesis and assembly of Ag/NiO nanofibers with high electrical conductivity. Chem. Mater. 2007, 19, 1895–1897. [Google Scholar] [CrossRef]
- Mullin, J.; Schatz, G.C. Combined linear response quantum mechanics and classical electrodynamics (QM/ED) method for the calculation of surface-enhanced Raman spectra. J. Phys. Chem. A 2012, 116, 1931–1938. [Google Scholar] [CrossRef] [PubMed]
- Shim, S.; Stuart, C.M.; Mathies, R.A. Resonance Raman cross-sections and vibronic analysis of rhodamine 6G from broadband stimulated Raman spectroscopy. ChemPhysChem 2008, 9, 697–699. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.; Schatz, G.C. Resonance Raman scattering of rhodamine 6G as calculated using time-dependent density functional theory. J. Phys. Chem. A 2006, 110, 5973–5977. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.-L.; Li, D.-W.; Qu, L.-L.; Fossey, J.S.; Long, Y.-T. Multiple depositions of Ag nanoparticles on chemically modified agarose films for surface-enhanced Raman spectroscopy. Nanoscale 2012, 4, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Wustholz, K.L.; Henry, A.-I.; McMahon, J.M.; Freeman, R.G.; Valley, N.; Piotti, M.E.; Natan, M.J.; Schatz, G.C.; Duyne, R.P.V. Structure-Activity Relationships in Gold Nanoparticle Dimers and Trimers for Surface-Enhanced Raman Spectroscopy. J. Am. Chem. Soc. 2010, 132, 10903–10910. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Guo, X.; Xu, L.; Ying, Y.; Wu, Y.; Wen, Y.; Yang, H. Template-free synthesis of SERS-active gold nanopopcorn for rapid detection of chlorpyrifos residues. Sens. Actuators B Chem. 2017, 241, 1008–1013. [Google Scholar] [CrossRef]
- Mircescu, N.E.; Oltean, M.; Chiş, V.; Leopold, N. FTIR, FT-Raman, SERS and DFT study on melamine. Vib. Spectrosc. 2012, 62, 165–171. [Google Scholar] [CrossRef]
- Li, Z.; Meng, G.; Huang, Q.; Hu, X.; He, X.; Tang, H.; Wang, Z.; Li, F. Ag Nanoparticle-Grafted PAN-Nanohump Array Films with 3D High-Density Hot Spots as Flexible and Reliable SERS Substrates. Small 2015, 11, 5452–5459. [Google Scholar] [CrossRef] [PubMed]
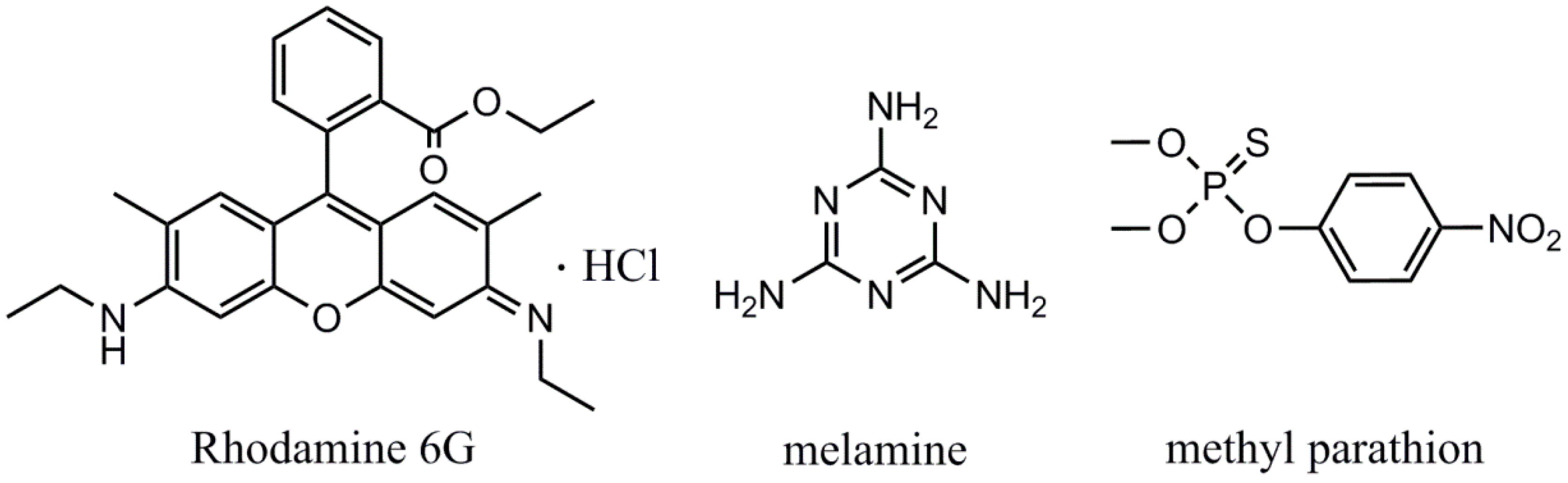



| Chemicals | Raman Shift (cm−1) | Assignment |
|---|---|---|
| Rodamine 6G | 769 | ip XRD and op C-H bend |
| 1123 | C-H str | |
| 1194 | ip XRD, C-H bend, N-H bend | |
| 1311 | ip XRB, N-H bend, CH2 wag | |
| 1361 | XRS, ip C-H bend, C-C str | |
| 1509 | XRS, C-N str, C-H bend, N-H bend, C-C str | |
| 1647 | XRS, ip C-H bend, C-C str | |
| melamine | 684 | Ring breathing |
| methyl parathion | 1111 | stretching vibration of C-N |
| 1344 | bending vibration of C-H |
| SERS Substrates | EF | ||||
|---|---|---|---|---|---|
| Porous Ag | 541 | 68 | 5 × 10−8 | 1 × 10−3 | 1.59 × 105 |
| Ag-NiO nanofibers | 589 | 3 × 10−7 | 2.89 × 104 |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.; Sun, X.; Hou, C.; Hou, J.; Lei, Y. Preparation of Quasi-Three-Dimensional Porous Ag and Ag-NiO Nanofibrous Mats for SERS Application. Sensors 2018, 18, 2862. https://doi.org/10.3390/s18092862
Wu H, Sun X, Hou C, Hou J, Lei Y. Preparation of Quasi-Three-Dimensional Porous Ag and Ag-NiO Nanofibrous Mats for SERS Application. Sensors. 2018; 18(9):2862. https://doi.org/10.3390/s18092862
Chicago/Turabian StyleWu, Huixiang, Xiangcheng Sun, Changjun Hou, Jingzhou Hou, and Yu Lei. 2018. "Preparation of Quasi-Three-Dimensional Porous Ag and Ag-NiO Nanofibrous Mats for SERS Application" Sensors 18, no. 9: 2862. https://doi.org/10.3390/s18092862
APA StyleWu, H., Sun, X., Hou, C., Hou, J., & Lei, Y. (2018). Preparation of Quasi-Three-Dimensional Porous Ag and Ag-NiO Nanofibrous Mats for SERS Application. Sensors, 18(9), 2862. https://doi.org/10.3390/s18092862







