Fabrication of a Food Nano-Platform Sensor for Determination of Vanillin in Food Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of CPE/NiO-SWCNTs/BPrPF6
2.2. Preparation of Real Sample
3. Results
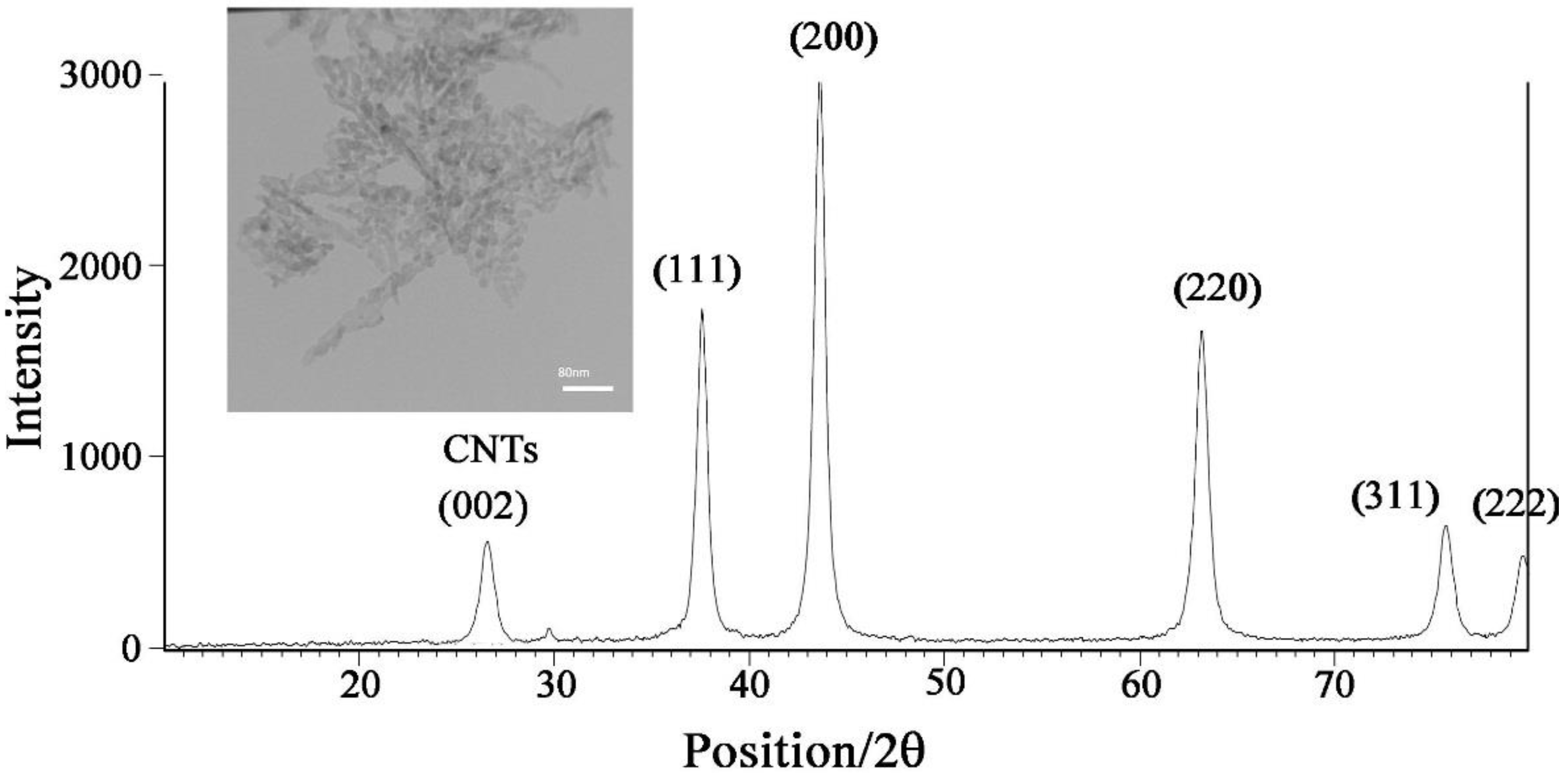
3.1. NiO-SWCNTs Morphological and Structure Investigation
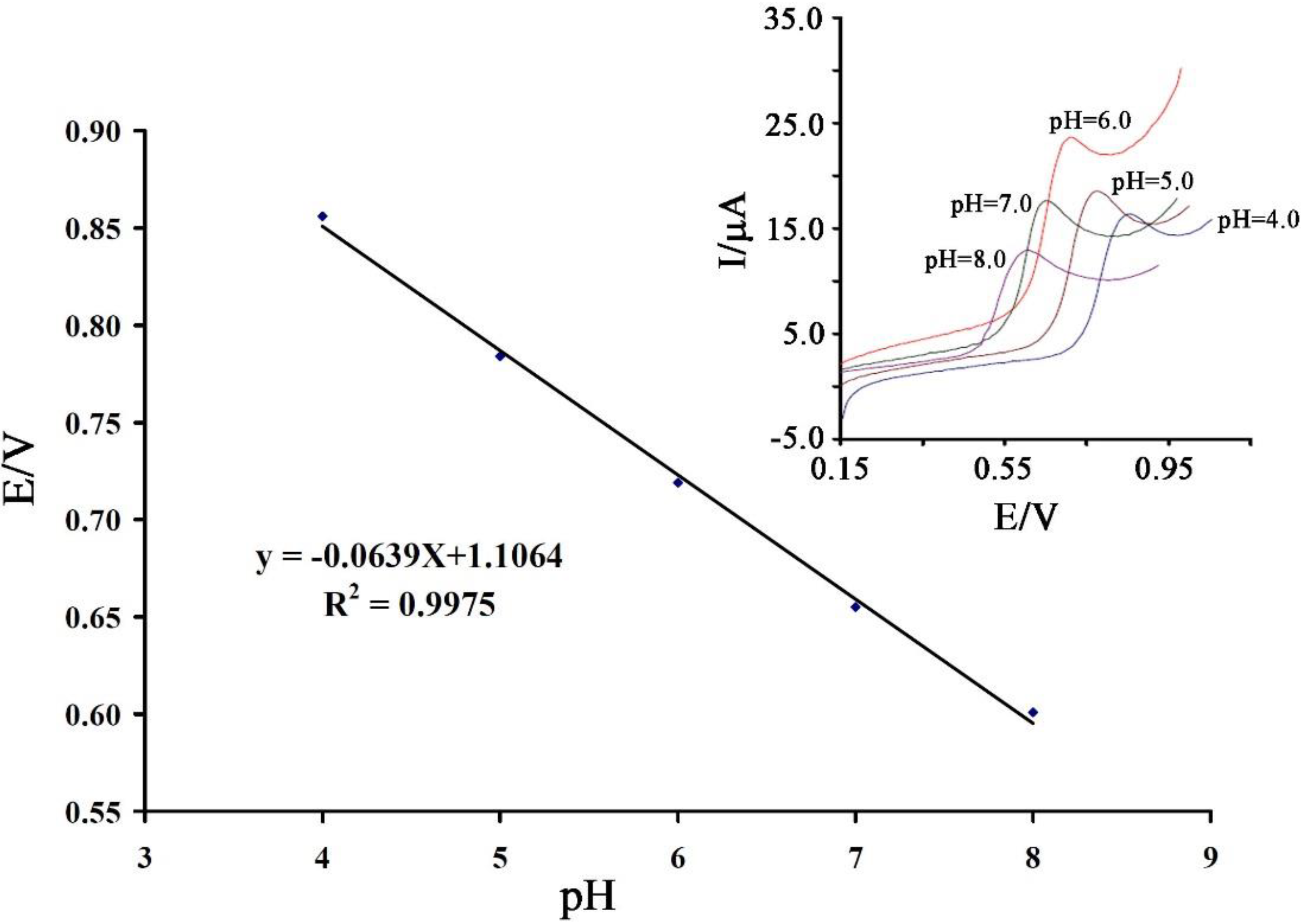
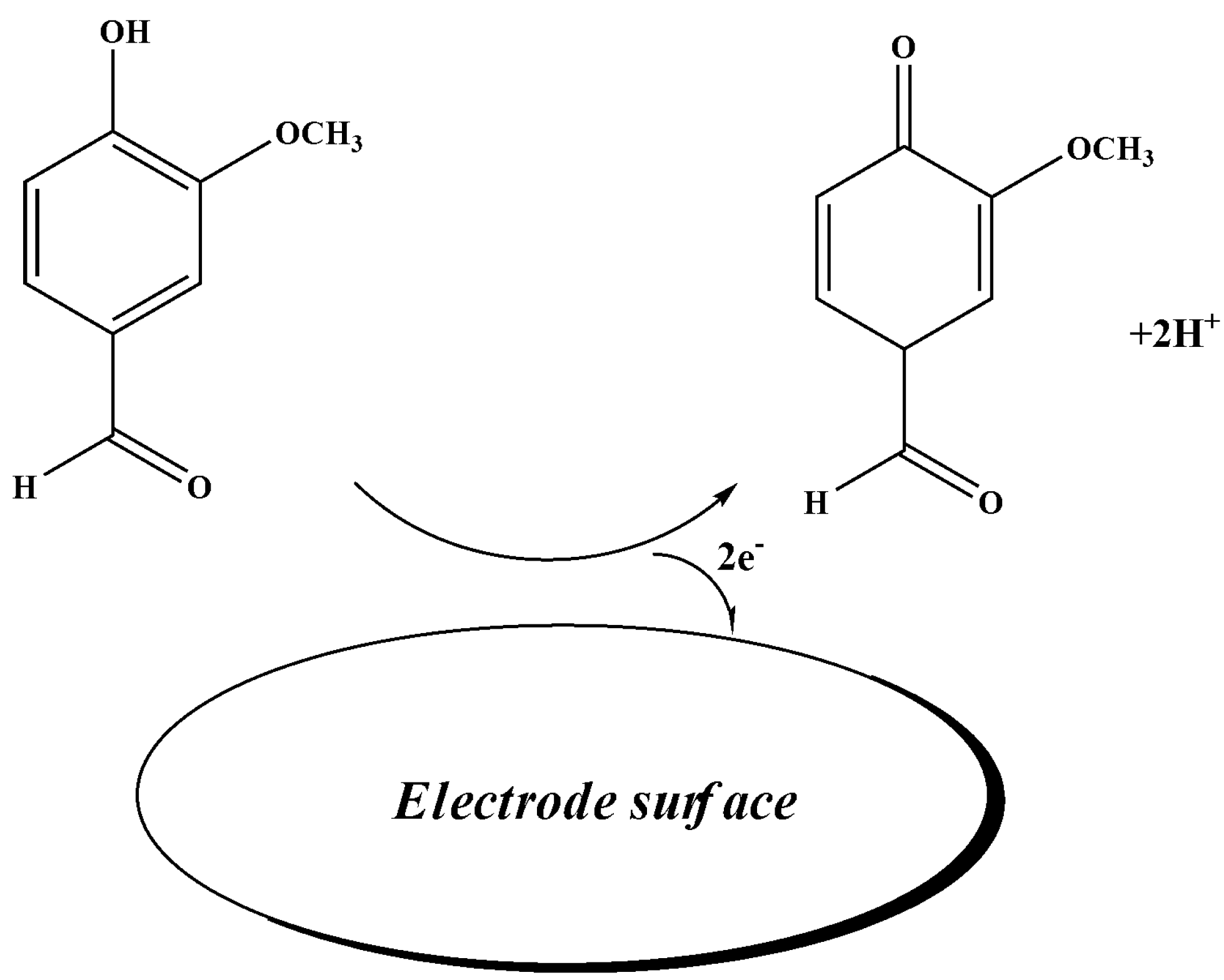
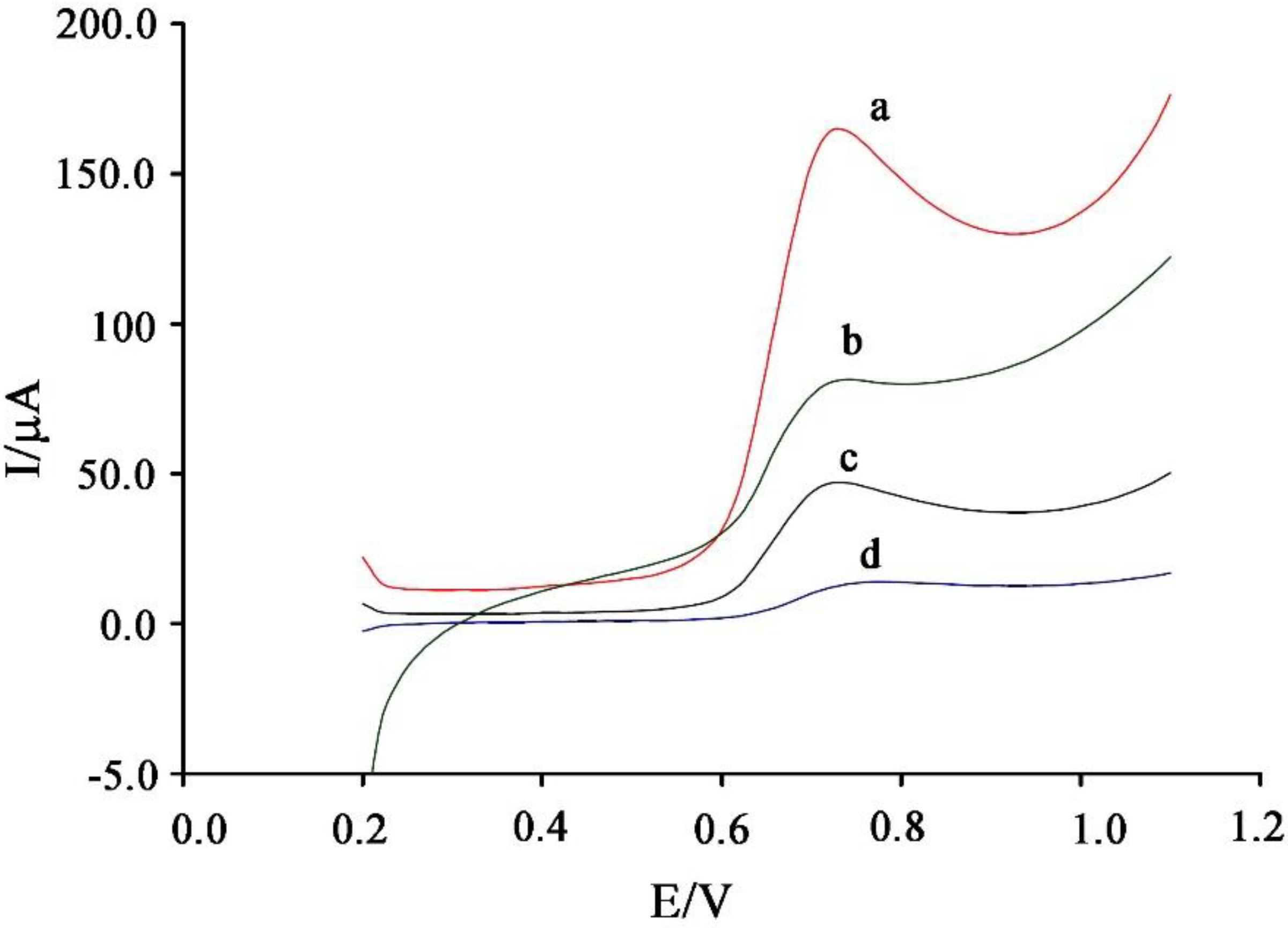
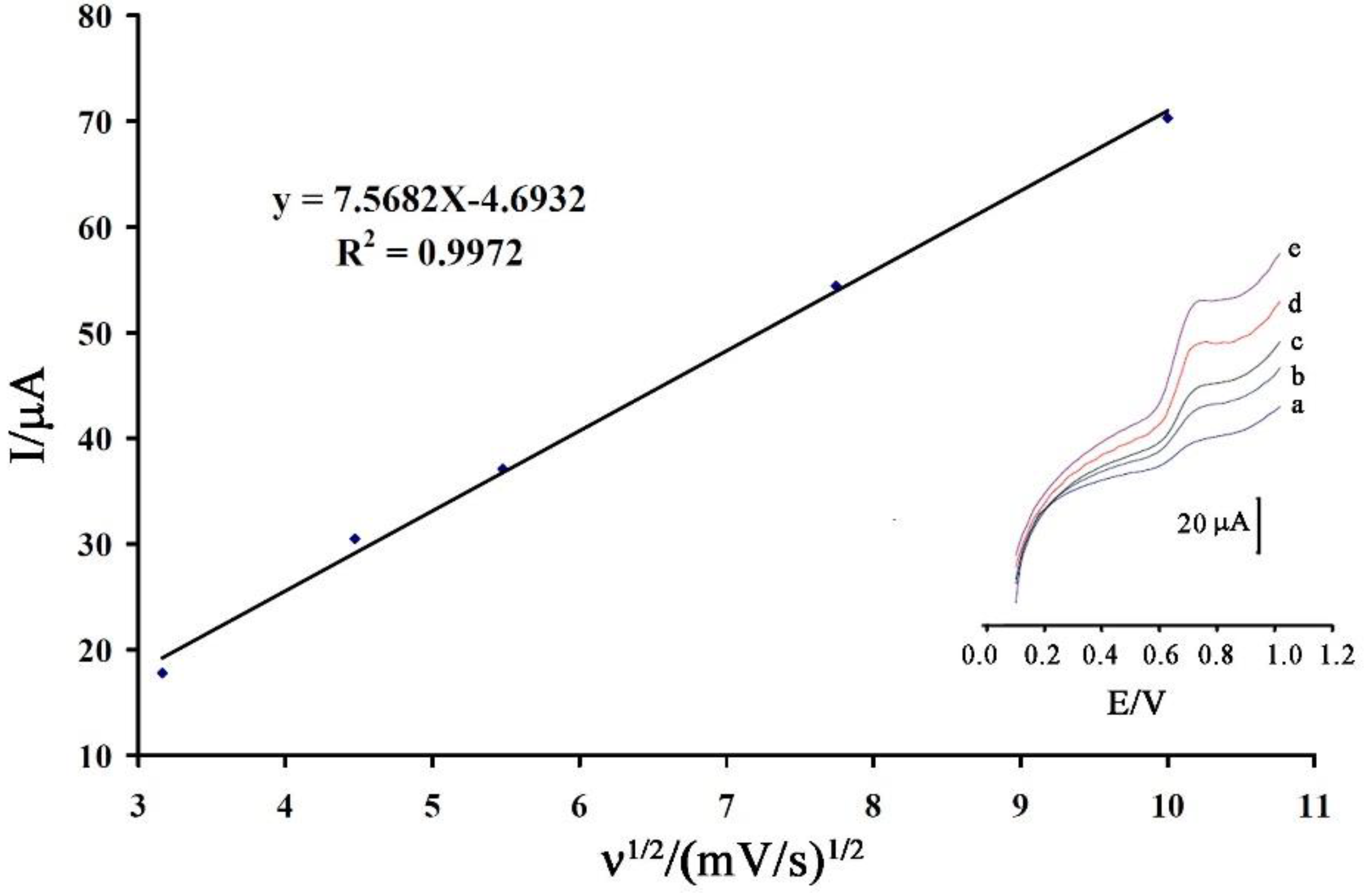
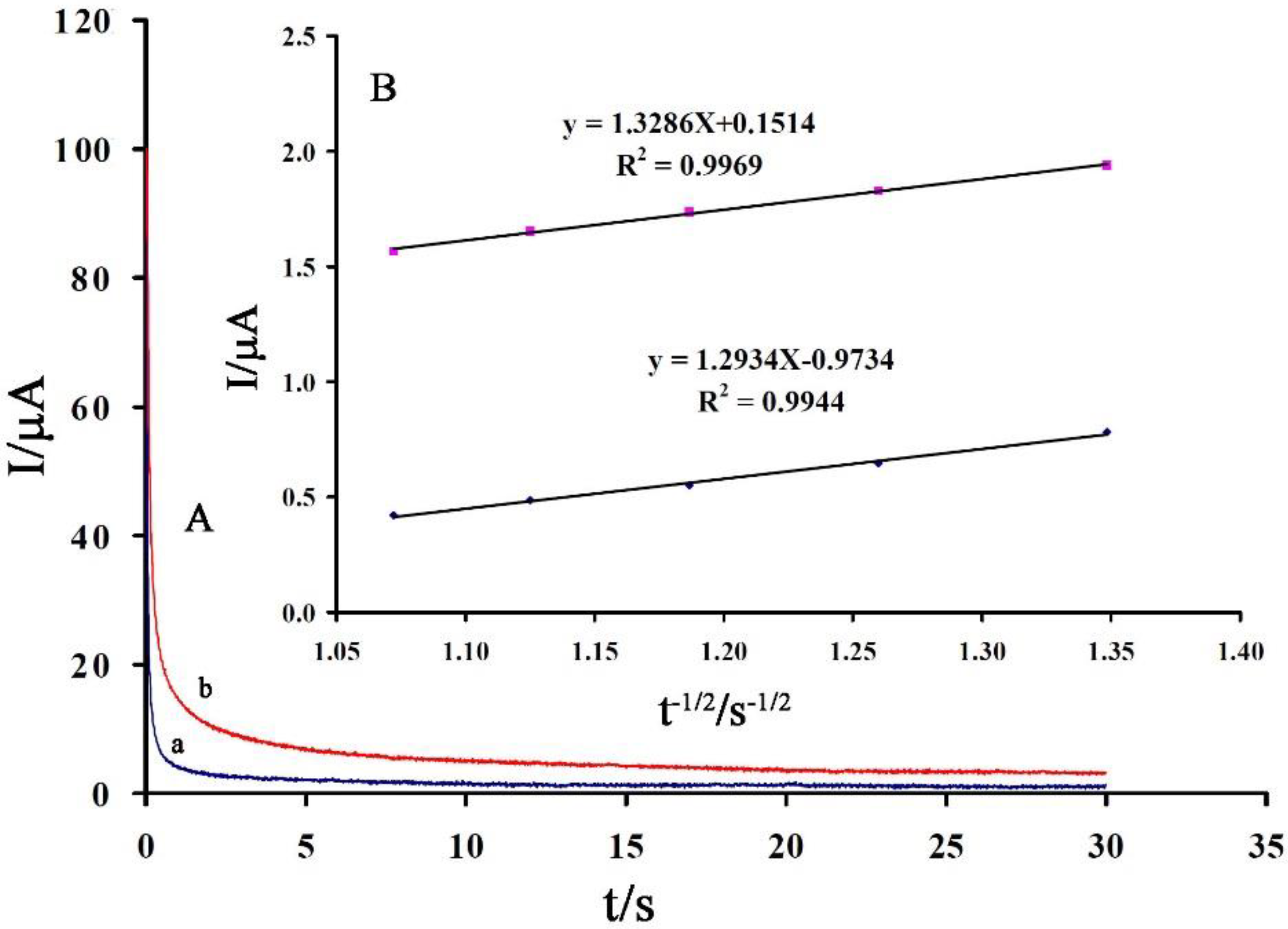
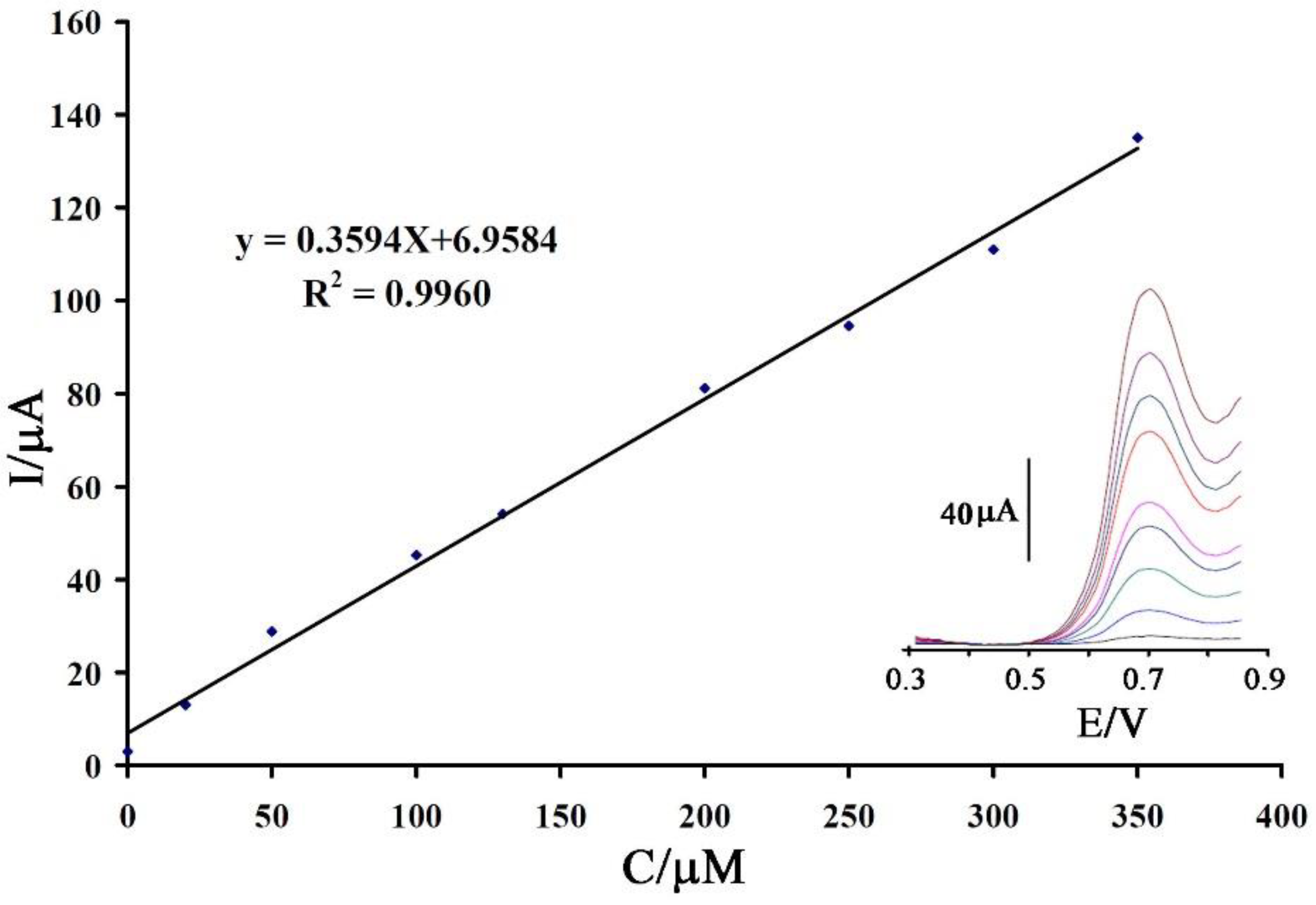
3.2. Electrochemical Behavior of Vanillin at the Surface of the Proposed Sensor
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cifuentes, A. Food analysis and Foodomics. J. Chromatogr. A 2009, 1216, 7109. [Google Scholar] [CrossRef] [PubMed]
- Worm, M.; Ehlers, I.; Sterry, W.; Zuberbier, T. Clinical relevance of food additives in adult patients with atopic dermatitis. Clin. Exp. Allergy 2000, 30, 404–417. [Google Scholar] [CrossRef]
- Charissou, A.; Ait-Ameur, L.; Birlouez-Aragon, I. Evaluation of a gas chromatography/mass spectrometry method for the quantification of carboxymethyllysine in food samples. J. Chromatogr. A 2007, 1140, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Kenney, B.F. Determination of organic acids in food samples by capillary electrophoresis. J. Chromatogr. A 1991, 546, 423–430. [Google Scholar] [CrossRef]
- Wen, X.; Yang, Q.; Yan, Z.; Deng, Q. Determination of cadmium and copper in water and food samples by dispersive liquid–liquid microextraction combined with UV–vis spectrophotometry. Microchem. J. 2011, 97, 249–254. [Google Scholar] [CrossRef]
- Yang, D.; Ying, Y. Applications of Raman Spectroscopy in Agricultural Products and Food Analysis: A Review. Appl. Spectrosc. Rev. 2011, 46, 539–560. [Google Scholar] [CrossRef]
- Shabir, G.A. Validation of high-performance liquid chromatography methods for pharmaceutical analysis: Understanding the differences and similarities between validation requirements of the US Food and Drug Administration, the US Pharmacopeia and the International Conference on Harmonization. J. Chromatogr. A 2003, 987, 57–66. [Google Scholar] [PubMed]
- Bijad, M.; Karimi-Maleh, H.; Farsi, M.; Shahidi, S.A. An electrochemical-amplified-platform based on the nanostructure voltammetric sensor for the determination of carmoisine in the presence of tartrazine in dried fruit and soft drink samples. J. Food Meas. Charact. 2018, 12, 634–640. [Google Scholar] [CrossRef]
- Sheikhshoaie, M.; Karimi-Maleh, H.; Sheikhshoaie, I.; Ranjbar, M. Voltammetric amplified sensor employing RuO2 nano-road and room temperature ionic liquid for amaranth analysis in food samples. J. Mol. Liq. 2017, 229, 489–494. [Google Scholar] [CrossRef]
- Baghizadeh, A.; Karimi-Maleh, H.; Khoshnama, Z.; Hassankhani, A.; Abbasghorbani, M. A voltammetric sensor for simultaneous determination of vitamin C and vitamin B6 in food samples using ZrO2 nanoparticle/ionic liquids carbon paste electrode. Food Anal. Methods 2015, 8, 549–557. [Google Scholar] [CrossRef]
- Khaleghi, F.; Arab, Z.; Gupta, V.K.; Ganjali, M.R.; Norouzi, P.; Atar, N.; Yola, M.L. Fabrication of novel electrochemical sensor for determination of vitamin C in the presence of vitamin B9 in food and pharmaceutical samples. J. Mol. Liq. 2016, 221, 666–672. [Google Scholar] [CrossRef]
- Khaleghi, F.; Elyasi Irai, A.; Sadeghi, R.; Gupta, V.K.; Wen, Y. A fast strategy for determination of vitamin B9 in food and pharmaceutical samples using an ionic liquid-modified nanostructure voltammetric sensor. Sensors 2016, 16, 747. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Tahernejad-Javazmi, F.; Atar, N.; Yola, M.L.; Gupta, V.K.; Ensafi, A.A. A novel DNA biosensor based on a pencil graphite electrode modified with polypyrrole/functionalized multiwalled carbon nanotubes for determination of 6-mercaptopurine anticancer drug. Ind. Eng. Chem. Res. 2015, 54, 3634–3639. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Karimi-Maleh, H.; Mallakpour, S. A new strategy for the selective determination of glutathione in the presence of nicotinamide adenine dinucleotide (NADH) using a novel modified carbon nanotube paste electrode. Colloids Surf. B Biointerfaces 2013, 104, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Moazampour, M.; Ensafi, A.A.; Mallakpour, S.; Hatami, M. An electrochemical nanocomposite modified carbon paste electrode as a sensor for simultaneous determination of hydrazine and phenol in water and wastewater samples. Environ. Sci. Pollut. Res. Int. 2014, 21, 5879–5888. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.K.; Atar, N.; Yola, M.L.; Üstündağ, Z.; Uzun, L. A novel magnetic Fe@Au core-shell nanoparticles anchored graphene oxide recyclable nanocatalyst for the reduction of nitrophenol compounds. Water Res. 2014, 48, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Sanghavi, B.J.; Mobin, S.M.; Mathur, P.; Lahiri, G.K.; Srivastava, A.K. Biomimetic sensor for certain catecholamines employing copper (II) complex and silver nanoparticle modified glassy carbon paste electrode. Biosens. Bioelectron. 2013, 39, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Yola, M.L.; Atar, N.; Eren, T.; Karimi-Maleh, H.; Wang, S. Sensitive and selective determination of aqueous triclosan based on gold nanoparticles on polyoxometalate/reduced graphene oxide nanohybrid. RSC Adv. 2015, 5, 65953–65962. [Google Scholar] [CrossRef]
- Sanghavi, B.J.; Sitaula, S.; Griep, M.H.; Karna, S.P.; Ali, M.F.; Swami, N.S. Real-time electrochemical monitoring of adenosine triphosphate in the picomolar to micromolar range using graphene-modified electrodes. Anal. Chem. 2013, 85, 8158–8165. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Gao, Y.; Zhan, D.; Liu, H.; Zhao, Q.; Kou, Y.; Shao, Y.; Li, M.; Zhuang, Q.; Zhu, Z. Selective detection of dopamine in the presence of ascorbic acid and uric acid by a carbon nanotubes-ionic liquid gel modified electrode. Talanta 2005, 66, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Beitollahi, H.; Raoof, J.B.; Karimi-Maleh, H.; Hosseinzadeh, R. Electrochemical behavior of isoproterenol in the presence of uric acid and folic acid at a carbon paste electrode modified with 2,7-bis(ferrocenyl ethyl)fluoren-9-one and carbon nanotubes. J. Solid State Electrochem. 2012, 16, 1701–1707. [Google Scholar] [CrossRef]
- Fouladgar, M.; Karimi-Maleh, H. Ionic liquid/multiwall carbon nanotubes paste electrode for square wave voltammetric determination of methyldopa. Ionics 2013, 19, 1163–1170. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Karimi Maleh, H. A multiwall carbon nanotubes paste electrode as a sensor and ferrocenemonocarboxylic acid as a mediator for electrocatalytic determination of isoproterenol. Int. J. Electrochem. Sci. 2010, 5, 1484–1495. [Google Scholar]
- Sun, W.; Yang, M.; Jiao, K. Electrocatalytic oxidation of dopamine at an ionic liquid modified carbon paste electrode and its analytical application. Anal. Bioanal. Chem. 2007, 389, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Safavi, A.; Maleki, N.; Moradlou, O.; Tajabadi, F. Simultaneous determination of dopamine, ascorbic acid, and uric acid using carbon ionic liquid electrode. Anal. Biochem. 2006, 359, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Kuskur, C.M.; Swamy, B.E.K.; Jayadevappa, H. Electrochemical behaviour of norepinephrine in the presence of paracetamol and folic acid at poly (Congo red) modified carbon paste electrode. Anal. Bioanal. Electrochem. 2018, 10, 658–674. [Google Scholar]
- Malhotra, S.; Tang, Y.; Varshney, P.K. Non-enzymatic glucose sensor based on electrodeposition of platinum particles on polyaniline modified Pt electrode. Anal. Bioanal. Electrochem. 2018, 10, 699–715. [Google Scholar]
- Khalilzadeh, M.A.; Karimi-Maleh, H.; Amiri, A.; Gholami, F. Determination of captopril in patient human urine using ferrocenemonocarboxylic acid modified carbon nanotubes paste electrode. Chin. Chem. Lett. 2010, 21, 1467–1470. [Google Scholar] [CrossRef]
- Elyasi, M.; Khalilzadeh, M.A.; Karimi-Maleh, H. High sensitive voltammetric sensor based on Pt/CNTs nanocomposite modified ionic liquid carbon paste electrode for determination of Sudan I in food samples. Food Chem. 2013, 141, 4311–4317. [Google Scholar] [CrossRef] [PubMed]
- Bijad, M.; Karimi-Maleh, H.; Khalilzadeh, M.A. Application of ZnO/CNTs nanocomposite ionic liquid paste electrode as a sensitive voltammetric sensor for determination of ascorbic acid in food samples. Food Anal. Methods 2013, 6, 1639–1647. [Google Scholar] [CrossRef]
- Najafi, M.; Khalilzadeh, M.A.; Karimi-Maleh, H. A new strategy for determination of bisphenol A in the presence of Sudan I using a ZnO/CNTs/ionic liquid paste electrode in food samples. Food Chem. 2014, 158, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Sheikholeslami, M. Solidification of NEPCM under the effect of magnetic field in a porous thermal energy storage enclosure using CuO nanoparticles. J. Mol. Liq. 2018, 263, 303–315. [Google Scholar] [CrossRef]
- Sheikholeslami, M. Numerical modeling of nano enhanced PCM solidification in an enclosure with metallic fin. J. Mol. Liq. 2018, 259, 424–438. [Google Scholar] [CrossRef]
- .Sheikholeslami, M. Numerical investigation for CuO-H2O nanofluid flow in a porous channel with magnetic field using mesoscopic method. J. Mol. Liq. 2018, 249, 739–746. [Google Scholar] [CrossRef]
- Arabali, V.; Ebrahimi, M.; Abbasghorbani, M.; Gupta, V.K.; Farsi, M.; Ganjali, M.; Karimi, F. Electrochemical determination of vitamin C in the presence of NADH using a CdO nanoparticle/ionic liquid modified carbon paste electrode as a sensor. J. Mol. Liq. 2016, 213, 312–316. [Google Scholar] [CrossRef]
- Goyal, R.N.; Gupta, V.K.; Chatterjee, S. Simultaneous determination of adenosine and inosine using single-wall carbon nanotubes modified pyrolytic graphite electrode. Talanta 2008, 76, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Tahernejad-Javazmi, F.; Ensafi, A.A.; Moradi, R.; Mallakpour, S.; Beitollahi, H. A high sensitive biosensor based on fept/cnts nanocomposite/N-(4-hydroxyphenyl)-3,5-dinitrobenzamide modified carbon paste electrode for simultaneous determination of glutathione and piroxicam. Biosens. Bioelectron. 2014, 60, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Biparva, P.; Hatami, M. A novel modified carbon paste electrode based on nio/cnts nanocomposite and (9,10-dihydro-9,10-ethanoanthracene-11,12-dicarboximido)-4-ethylbenzene-1,2-diol as a mediator for simultaneous determination of cysteamine, nicotinamide adenine dinucleotide and folic acid. Biosens. Bioelectron. 2013, 48, 270–275. [Google Scholar] [PubMed]
- Karimi-Maleh, H.; Shojaei, A.F.; Tabatabaeian, K.; Karimi, F.; Shakeri, S.; Moradi, R. Simultaneous determination of 6-mercaptopruine, 6-thioguanine and dasatinib as three important anticancer drugs using nanostructure voltammetric sensor employing Pt/MWCNTs and 1-butyl-3-methylimidazolium hexafluoro phosphate. Biosens. Bioelectron. 2016, 86, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Sanati, A.L.; Karimi-Maleh, H.; Abbasghorbani, M. Synthesis of NiO nanoparticle and application of its in the preparation of electrochemical sensor for voltammetric determination of Nalbuphine. J. Appl. Chem. 2015, 9, 35–40. [Google Scholar]
- Sanati, A.L.; Faridbod, F.; Ganjali, M.R. Synergic effect of graphene quantum dots and room temperature ionic liquid for the fabrication of highly sensitive voltammetric sensor for levodopa determination in the presence of serotonin. J. Mol. Liq. 2017, 241, 316–320. [Google Scholar] [CrossRef]
- Sanati, A.L.; Faridbod, F. Electrochemical Determination of Methyldopa by Graphene Quantum Dot/1-butyl-3-methylimidazolium hexafluoro phosphate Nanocomposite Electrode. Int. J. Electrochem. Sci. 2017, 12, 7997–8005. [Google Scholar] [CrossRef]
- Ashjari, M.; Karimi-Maleh, H.; Ahmadpour, F.; Shabani-Nooshabadi, M.; Sadrnia, A.; Khalilzadeh, M.A. Voltammetric analysis of mycophenolate mofetil in pharmaceutical samples via electrochemical nanostructure based sensor modified with ionic liquid and MgO/SWCNTs. J. Taiwan Inst. Chem. Eng. 2017, 80, 989–996. [Google Scholar] [CrossRef]
- Alavi-Tabari, S.A.R.; Khalilzadeh, M.A.; Karimi-Maleh, H.; Zareyee, D. An amplified platform nanostructure sensor for the analysis of epirubicin in the presence of topotecan as two important chemotherapy drugs for breast cancer therapy. New J. Chem. 2018, 42, 3828–3832. [Google Scholar] [CrossRef]
- Alavi-Tabari, S.A.R.; Khalilzadeh, M.A.; Karimi-Maleh, H. Simultaneous determination of doxorubicin and dasatinib as two breast anticancer drugs uses an amplified sensor with ionic liquid and ZnO nanoparticle. J. Electroanal. Chem. 2018, 811, 84–88. [Google Scholar] [CrossRef]
- Van Assendelft, H. Adverse drug reactions checklist. Br. Med. J. 1987, 294, 576–577. [Google Scholar] [CrossRef]
- Cheraghi, S.; Taher, M.A.; Karimi-Maleh, H. Highly sensitive square wave voltammetric sensor employing CdO/SWCNTs and room temperature ionic liquid for analysis of vanillin and folic acid in food samples. J. Food Compos. Anal. 2017, 62, 254–259. [Google Scholar] [CrossRef]
- Khalilzadeh, M.A.; Arab, Z. High sensitive nanostructure square wave voltammetric sensor for determination of vanillin in food samples. Curr. Anal. Chem. 2017, 13, 81–86. [Google Scholar] [CrossRef]
- Jiang, L.; Ding, Y.; Jiang, F.; Li, L.; Mo, F. Electrodeposited nitrogen-doped graphene/carbon nanotubes nanocomposite as enhancer for simultaneous and sensitive voltammetric determination of caffeine and vanillin. Anal. Chim. Acta 2014, 833, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Yardım, Y.; Gülcan, M.; Şentürk, Z. Determination of vanillin in commercial food product by adsorptive stripping voltammetry using a boron-doped diamond electrode. Food Chem. 2013, 141, 1821–1827. [Google Scholar] [CrossRef] [PubMed]
- Deng, P.; Xu, Z.; Zeng, R.; Ding, C. Electrochemical behavior and voltammetric determination of vanillin based on an acetylene black paste electrode modified with grapheme-polyvinylpyrrolidone composite film. Food Chem. 2015, 180, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Sanati, A.; Karimi-Maleh, H.; Badiei, A.; Biparva, P.; Ensafi, A.A. A voltammetric sensor based on NiO/CNTs ionic liquid carbon paste electrode for determination of morphine in the presence of diclofenac. Mater. Sci. Eng. C 2014, 35, 379–385. [Google Scholar] [CrossRef] [PubMed]
| Electrode | Mediator | pH | LDR (μM) | LOD (μM) | Ref. |
|---|---|---|---|---|---|
| carbon paste | CdO/SWCNTs and ionic liquid | 6.0 | 0.03–1200 | 0.009 | [47] |
| carbon paste | CuFe2O4 nanoparticles and ionic liquid | 7.0 | 0.1–700 | 0.07 | [48] |
| glassy carbon | AuPd nanoparticles–graphene | 0.1 M H2SO4 | 0.1–40 | 0.02 | [49] |
| boron-doped diamond | anodically pre-treated | 2.5 | 3.3–9.8 | 0.167 | [50] |
| acetylene black paste | graphene–polyvinylpyrrolidone | 0.1 M H3PO4 | 0.02–400 | 0.01 | [51] |
| carbon paste | NiO-SWCNTs and ionic liquid | 6.0 | 0.01–350 | 0.007 | This work |
| Sample | Added (μM) | Expected (μM) | Founded (μM) | Recovery % |
|---|---|---|---|---|
| Coffee milk | --- | --- | 4.12 ± 0.44 | --- |
| 10.00 | 14.12 | 14.43 ± 0.65 | 102.19 | |
| Chocolate | --- | --- | 1.95 ± 0.24 | --- |
| 10.00 | 11.95 | 11.75 ± 0.59 | 98.32 | |
| Biscuit | --- | --- | 4.56 ± 0.67 | --- |
| 10.00 | 14.56 | 14.98 ± 0.87 | 102.88 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gupta, V.K.; Karimi-Maleh, H.; Agarwal, S.; Karimi, F.; Bijad, M.; Farsi, M.; Shahidi, S.-A. Fabrication of a Food Nano-Platform Sensor for Determination of Vanillin in Food Samples. Sensors 2018, 18, 2817. https://doi.org/10.3390/s18092817
Gupta VK, Karimi-Maleh H, Agarwal S, Karimi F, Bijad M, Farsi M, Shahidi S-A. Fabrication of a Food Nano-Platform Sensor for Determination of Vanillin in Food Samples. Sensors. 2018; 18(9):2817. https://doi.org/10.3390/s18092817
Chicago/Turabian StyleGupta, Vinod Kumar, Hassan Karimi-Maleh, Shilpi Agarwal, Fatemeh Karimi, Majede Bijad, Mohammad Farsi, and Seyed-Ahmad Shahidi. 2018. "Fabrication of a Food Nano-Platform Sensor for Determination of Vanillin in Food Samples" Sensors 18, no. 9: 2817. https://doi.org/10.3390/s18092817
APA StyleGupta, V. K., Karimi-Maleh, H., Agarwal, S., Karimi, F., Bijad, M., Farsi, M., & Shahidi, S.-A. (2018). Fabrication of a Food Nano-Platform Sensor for Determination of Vanillin in Food Samples. Sensors, 18(9), 2817. https://doi.org/10.3390/s18092817






