A Portable Quantum Cascade Laser Spectrometer for Atmospheric Measurements of Carbon Monoxide
Abstract
1. Introduction
2. Materials and Methods
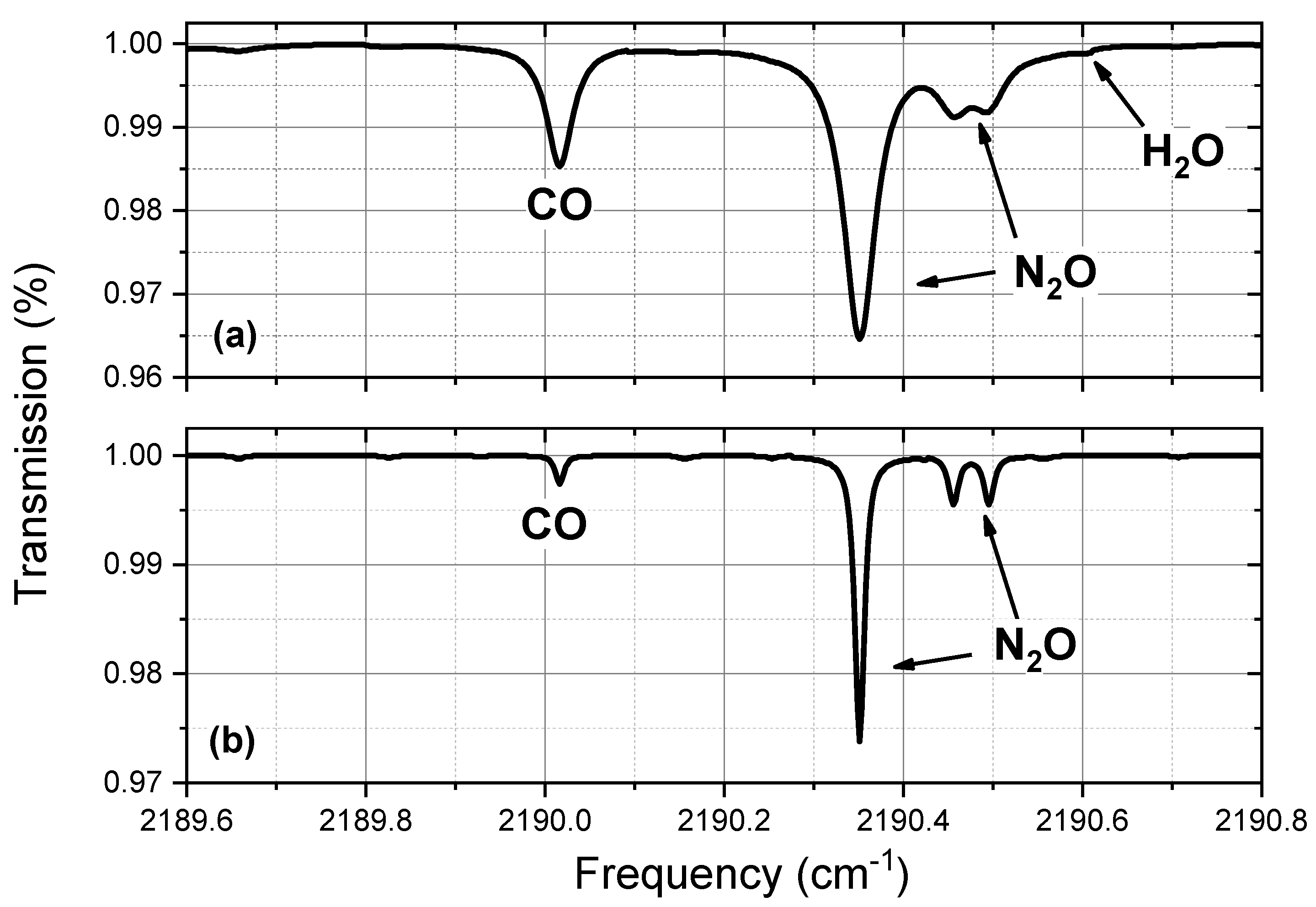
2.1. Selection of the Technique and of the CO Absorption Line
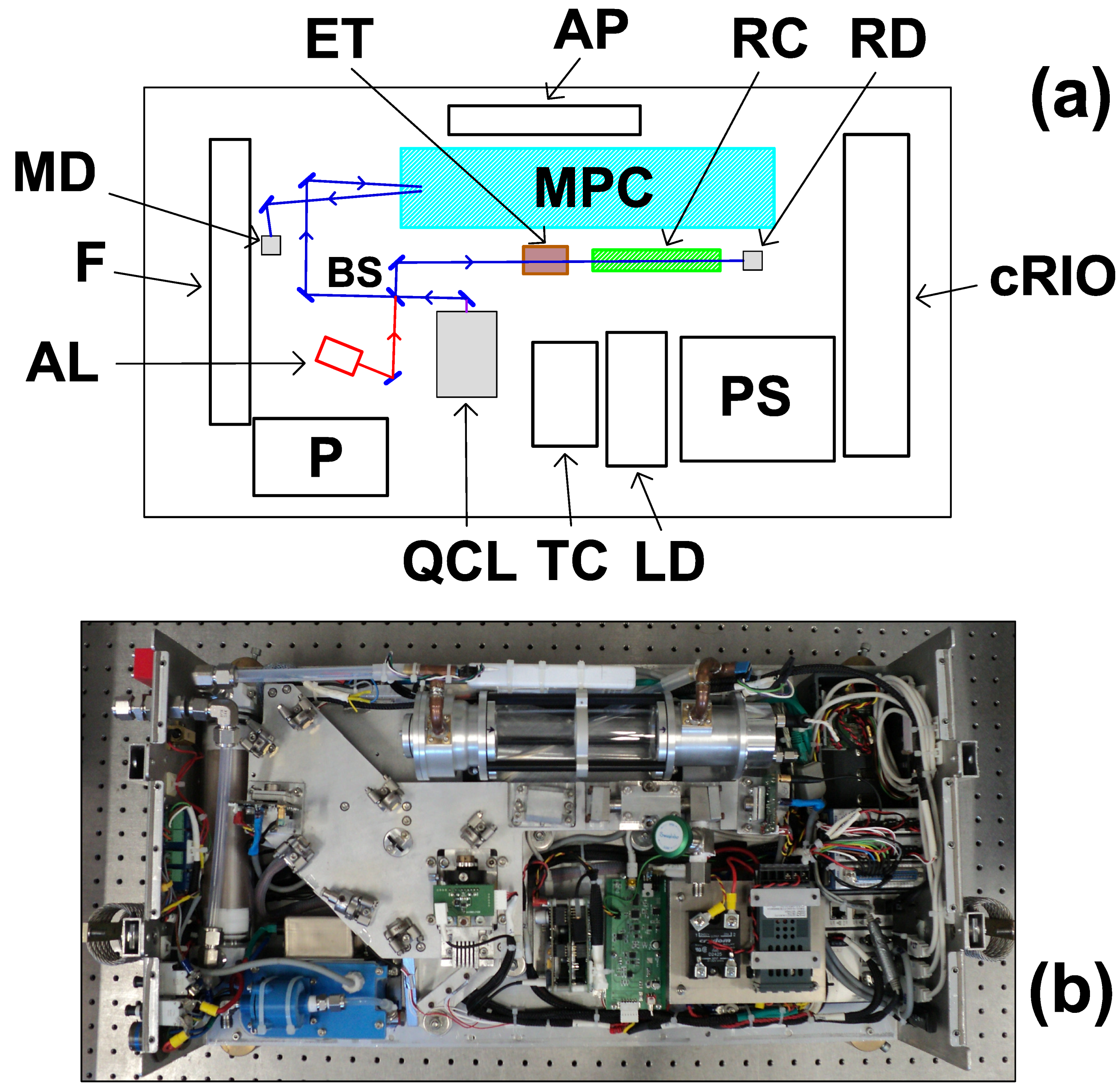
2.2. Opto-Mechanics
2.3. Electronics
2.4. Acquisition and Data Processing
3. Results
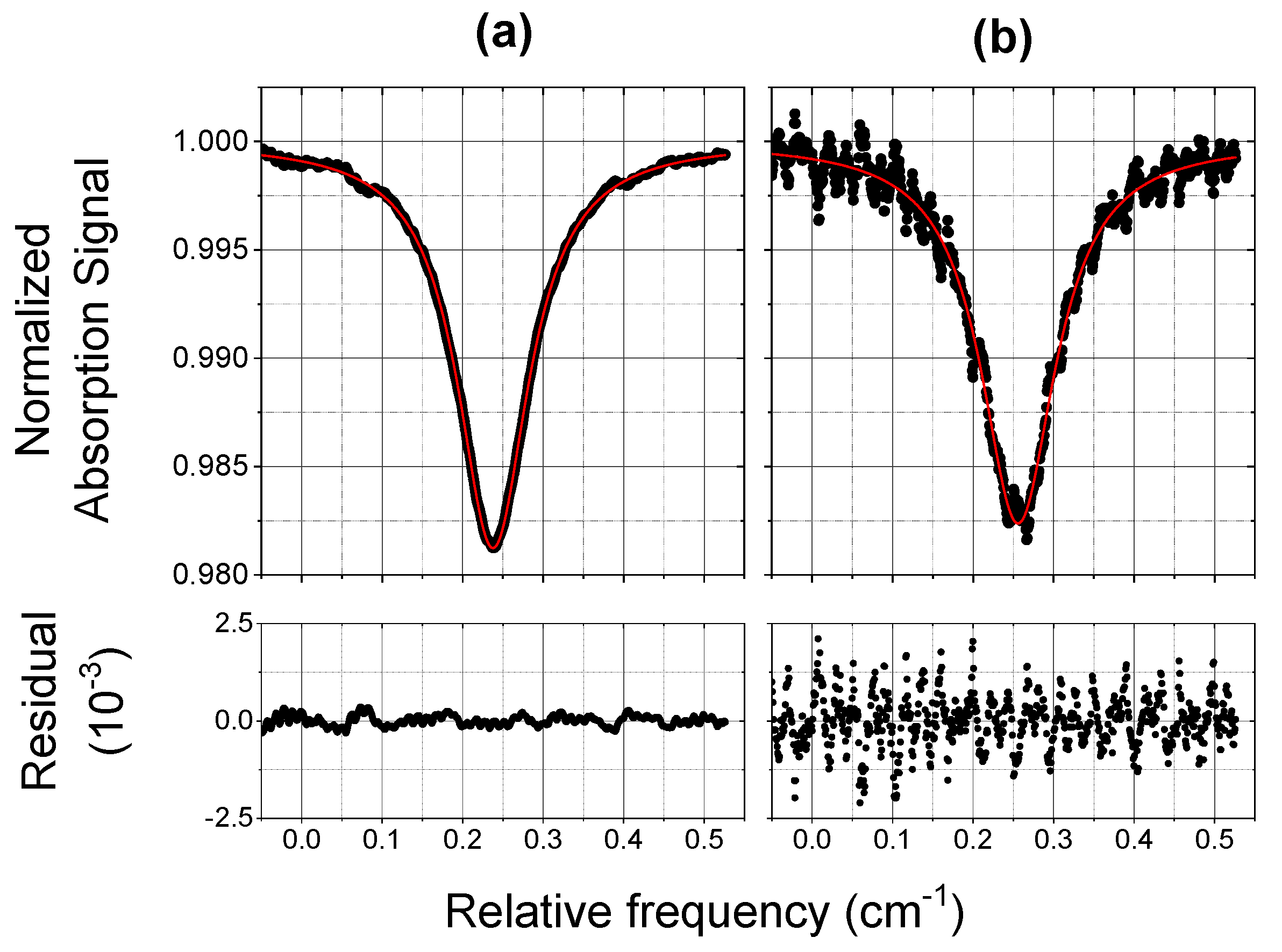
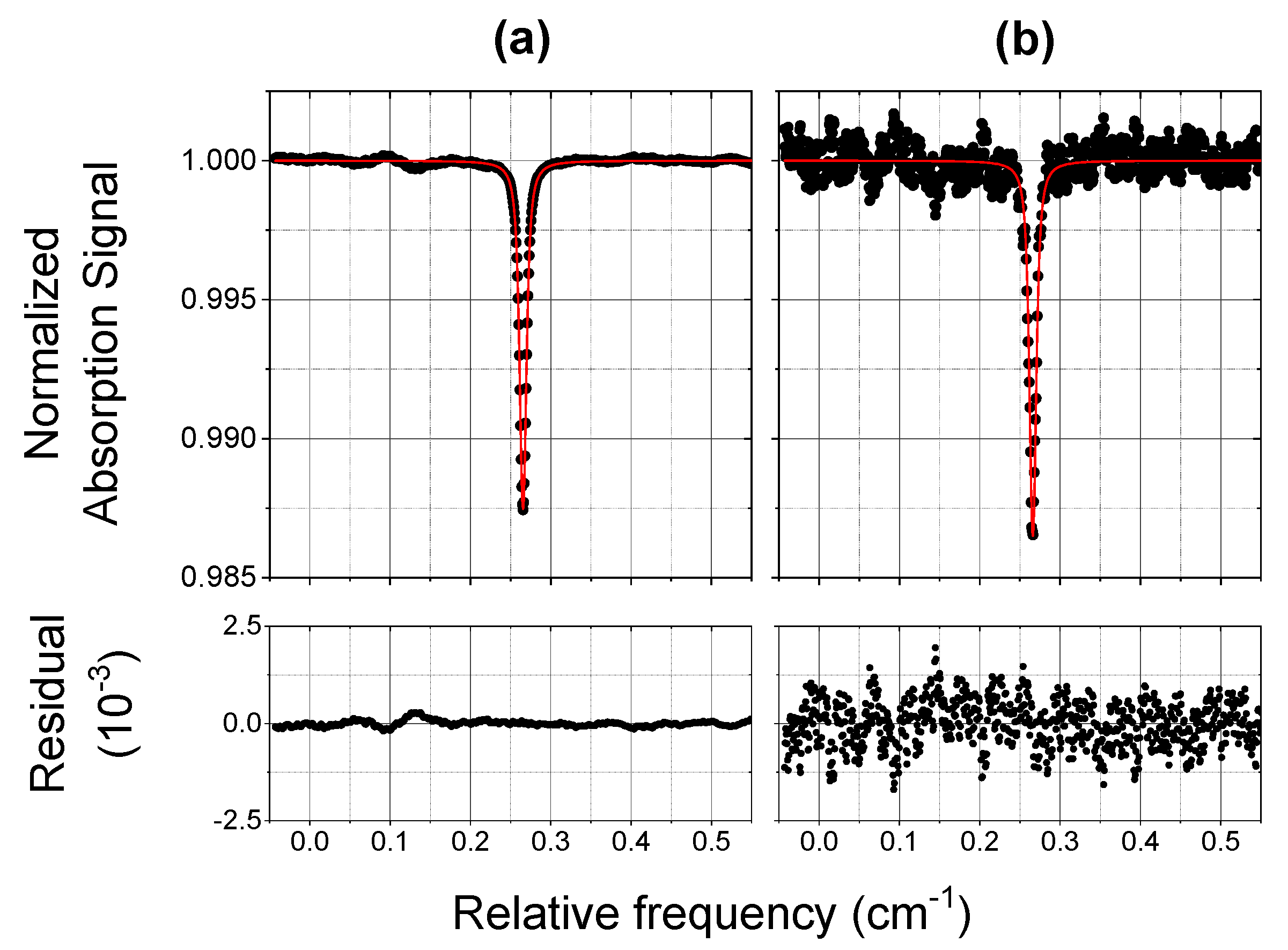
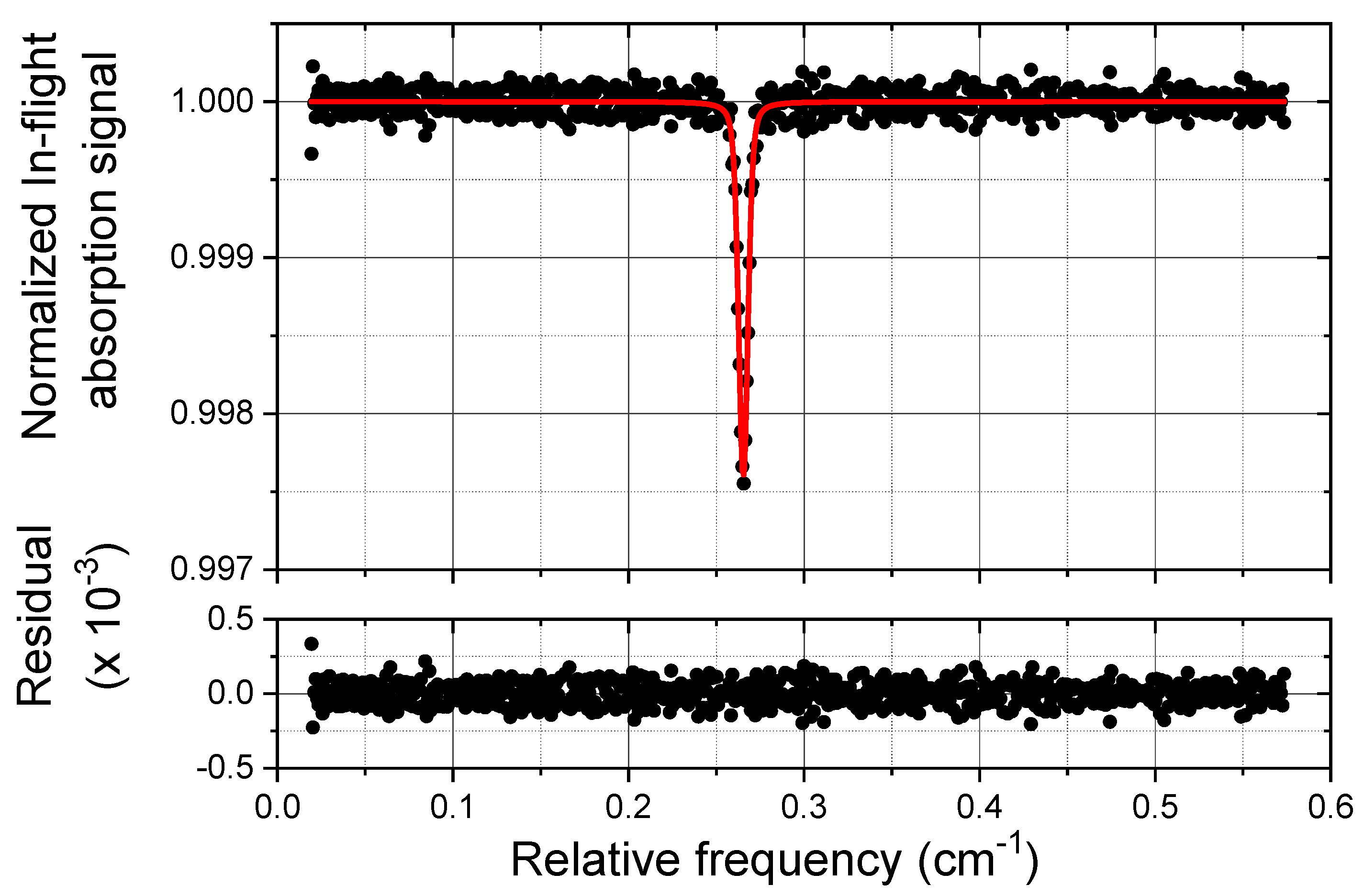
3.1. Laboratory Tests and Performances
3.2. In-Field Performances
4. Discussion
- (1)
- The selection of a calibration-free technique, as dTDLS, and the choice of optical and electronics components which work properly also without pressurization systems, allowed the realization of a portable device, entirely contained in one box only, that can be easily mounted on different platforms. The low weight, small size and low power consumption, with the only need of an access to sample the external air, make this instrument particularly suitable for installation on aircraft or balloons as piggyback in different missions.
- (2)
- The robustness of COLD2 is guaranteed by a mechanical structure designed to damp vibrations and to decouple the movements of the box from the optical breadboard. Its robustness was demonstrated by its deployment onboard the M55 stratospheric aircraft, where it operated, without need of realignment and without any kind of damages, during 8 tropical flights in harsh environment, with pressure down to 50 mbar and external temperature variable between C or higher (when the sensor was forced to stay for long periods inside the aircraft on the apron before take-off) and C (in the stratosphere).
- (3)
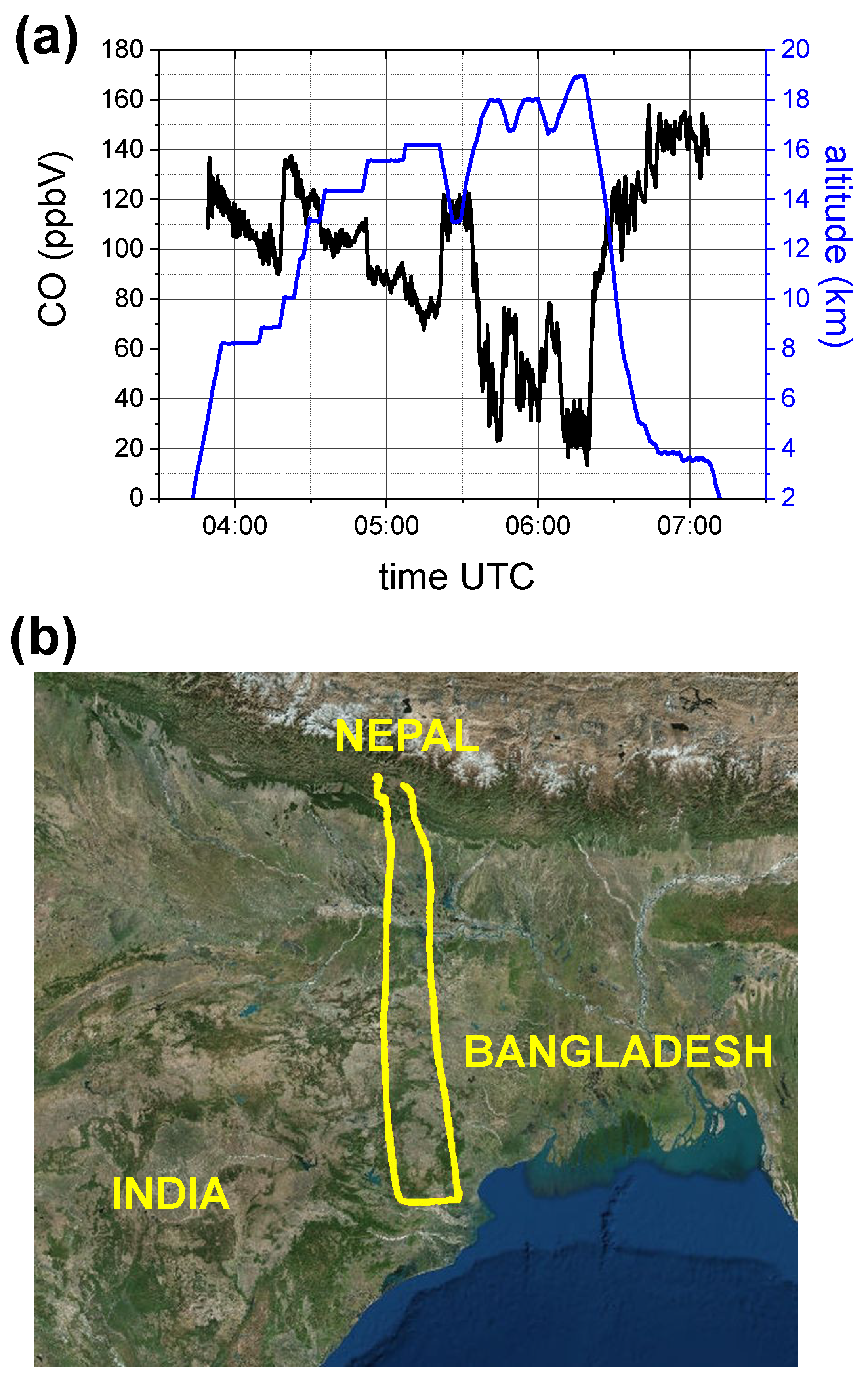
- For all the period of the tropical campaign in Nepal, the sensor worked completely unattended without failure and without need of external maintenance, as calibration procedures. Although the sensor experienced high humidity during each aircraft descent, the signal-to-noise ratio did not suffer significant degradation and a procedure of mirror cleaning was not necessary.
- (4)
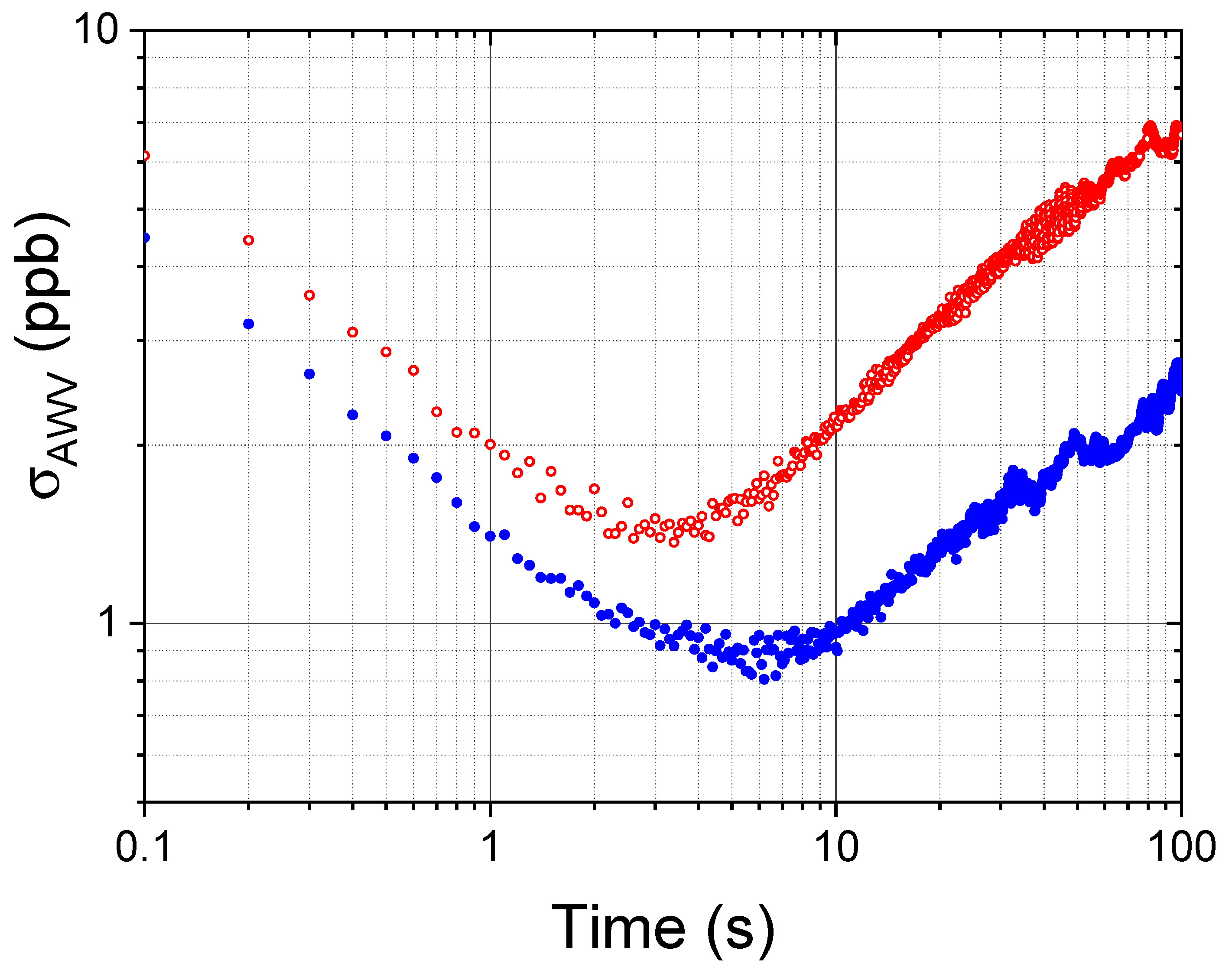
- The COLD2 acquisition time for each 1000 points spectrum is 1 ms. The device time resolution depends on the number of averages and can be easily set to fulfill the requirements of different applications. An Allan-Werle analysis demonstrated that an integration time of 1 s allows a sensitivity of 2 ppbV at atmospheric pressure and that this sensitivity increases at lower pressure. COLD2 is able to work at 10 Hz with a sensitivity between 5 and 8 ppbV and can reach a sensitivity lower than 1 ppb for integration time of 6 s. For ballonborne operation, when the time resolution can be smaller with respect to an aircraft, COLD2 can reach sub-ppbV performance.
- (5)
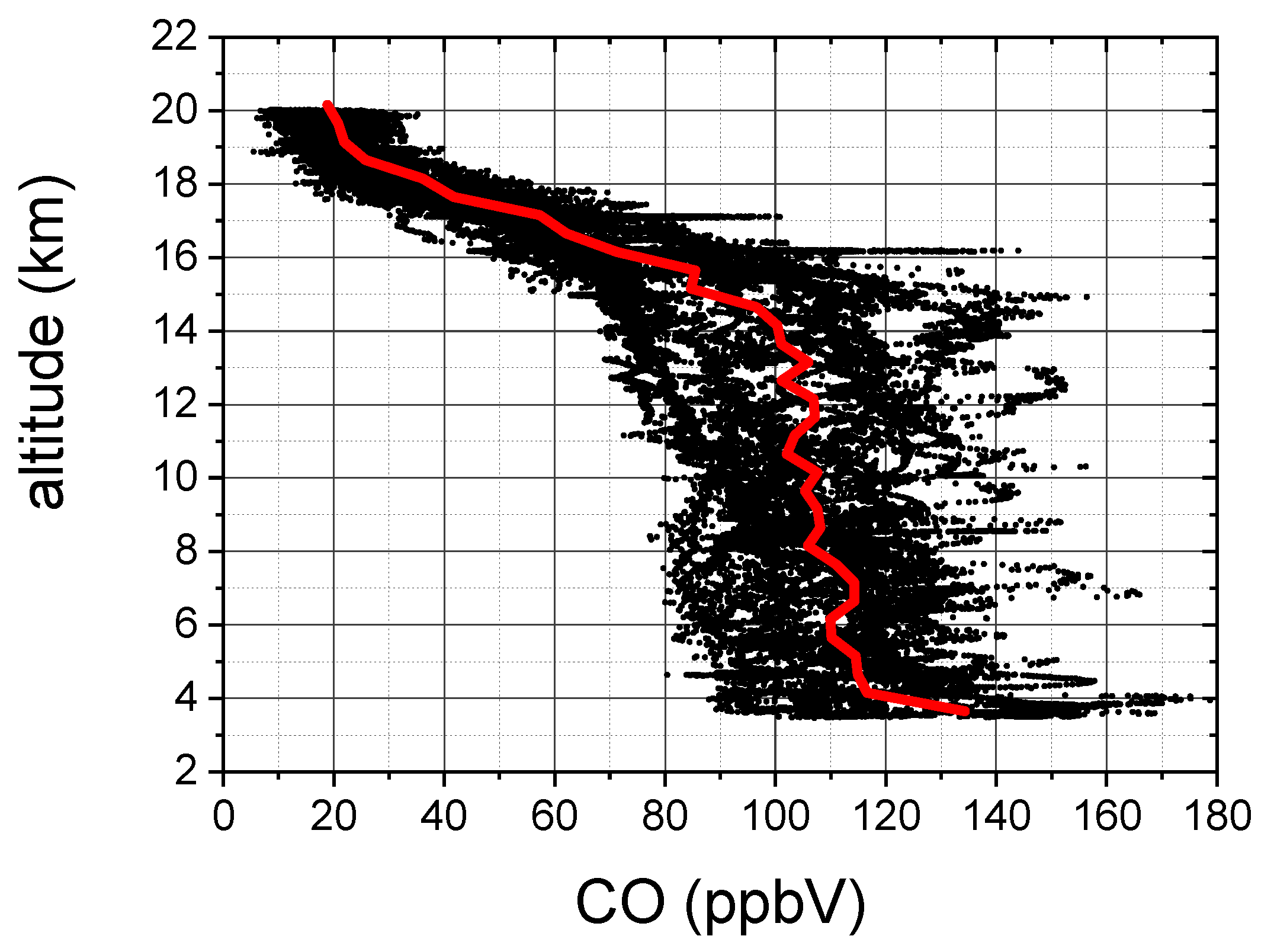
- The dTDLS technique in conjunction with a 16 bit acquisition system guarantees enough vertical resolution to register and analyze the CO absorption spectrum in a large range of pressure, temperature and concentration values. The COLD2 large dynamic range is demonstrated by the measurement of the CO vertical profiles during the campaign on board the M55 stratospheric aircraft (as shown in Figure 12) .
- (6)
- The COLD2 sensitivity is not significantly reduced by in-flight operation and it was demonstrated to be around 2 ppbV @ 1 s of integration time.
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CO | Carbon Monoxide |
| NO | Nitrous Oxide |
| QCL | Quantum Cascade Laser |
| CRDS | Cavity Ring Down Spectroscopy |
| CEAS | Cavity-Enhanced Absorption Spectroscopy |
| ICOS | Integrated Cavity Output Spectroscopy |
| WMS | Wavelength Modulation Spectroscopy |
| dTDLS | Direct Tunable Diode Laser Spectroscopy |
| FWHM | Full Width at Half Maximum |
| FSR | Free Spectral Range |
| FPGA | Field Programmable Gate Array |
| PZT | piezoelectric transducer |
References
- Hegglin, M.I.; Brunner, D.; Wernli, H.; Schwierz, C.; Martius, O.; Hoor, P.; Fischer, H.; Parchatka, U.; Spelten, N.; Schiller, C.; et al. Tracing troposphere-to-stratosphere transport above a mid-latitude deep convective system. Atmos. Chem. Phys. 2004, 4, 741–756. [Google Scholar] [CrossRef]
- Pickering, K.E.; Scala, J.R.; Thompson, A.M.; Tao, W.K.; Simpson, J.; Dickerson, R.R. Free tropospheric ozone production following entrainment of urban plumes into deep convection. J. Geophys. Res. 1992, 97, 17985–18000. [Google Scholar] [CrossRef]
- Crutzen, P.J.; Andreae, M.O. Biomass burning in the tropics: Impact on atmospheric chemistry and biogeochemical cycles. Science 1990, 250, 1669–1678. [Google Scholar] [CrossRef] [PubMed]
- Crutzen, P.J.; Fishman, J. Average concentrations of OH in the troposphere, and the budgets of CH4, CO, H2 and CH3CCl3. Geophys. Res. Lett. 1977, 4, 321–324. [Google Scholar] [CrossRef]
- Logan, J.A.; Prather, M.J.; Wofsy, S.C.; McElroy, M.B. Tropospheric chemistry: A global perspective. J. Geophys. Res. 1981, 86, 7210–7254. [Google Scholar] [CrossRef]
- Fishman, J.; Crutzen, P.J. The origin of ozone in the troposphere. Nature 1978, 274, 855–858. [Google Scholar] [CrossRef]
- Zahn, A.; Brenninkmeijer, C.A.M.; Asman, W.A.H.; Crutzen, P.J.; Heinrich, G.; Fischer, H.; Cuijpers, J.W.M.; van Velthoven, P.F.J. Budgets of O3 and CO in the upper troposphere: CARIBIC passenger aircraft results 1997–2001. J. Geophys. Res. 2002, 107, 6.1–6.19. [Google Scholar] [CrossRef]
- Podolske, J.; Leowenstein, M.; Blackburn, T. High Altitude Aircraft Measurements of Carbon Monoxide Using Tunable Diode Lasers. In Monitoring of Gaseous Pollutants by Tunable Diode Lasers; Grisar, R., Preier, H., Schmidtke, G., Restelli, G., Eds.; Air Pollution Research Reports; Springer: Dordrecht, The Netherlands, 1987; p. 80. [Google Scholar]
- Herman, R.L.; Webster, C.R.; May, R.D.; Scott, D.C.; Hu, H.; Moyer, E.J.; Wennberg, P.O.; Hanisco, T.F.; Lanzendorf, E.J.; Salawitch, R.J.; et al. Measurements of CO in the upper troposphere and lower stratosphere. Chemosp. Glob. Chang. Sci. 1999, 1, 173–183. [Google Scholar] [CrossRef]
- Nedelec, P.; Cammas, J.-P.; Thouret, V.; Athier, G.; Cousin, J.-M.; Legrand, C.; Abonnel, C.; Lecoeur, F.; Cayez, G.; Mariz, C. An improved infrared carbon monoxide analyser for routine measurements aboard commercial Airbus aircraft: Technical validation and first scientific results of the MOZAIC III programme. Atmos. Chem. Phys. 2003, 3, 1551–1564. [Google Scholar] [CrossRef]
- Lopez, J.P.; Luo, M.; Christensen, L.E.; Loewenstein, M.; Jost, H.-J.; Webster, C.R.; Osterman, G. TES carbon monoxide validation during two AVE campaigns using the Argus and ALIAS instruments on NASA’s WB-57F. J. Geophys. Res. 2008, 113, D16S47. [Google Scholar] [CrossRef]
- Santoni, G.W.; Daube, B.C.; Kort, E.A.; Jiménez, R.; Park, S.; Pittman, J.V.; Gottlieb, E.; Xiang, B.; Zahniser, M.S.; Nelson, D.D.; et al. Evaluation of the airborne quantum cascade laser spectrometer (QCLS) measurements of the carbon and greenhouse gas suite—CO2, CH4, N2O, and CO—During the CalNex and HIPPO campaigns. Atmos. Meas. Tech. 2014, 7, 1509–1526. [Google Scholar] [CrossRef]
- Tadic, I.; Parchatka, U.; Königstedt, R.; Fischer, H. In-flight stability of quantum cascade laser-based infrared absorption spectroscopy measurements of atmospheric carbon monoxide. Appl. Phys. B 2017, 123, 146. [Google Scholar] [CrossRef]
- Viciani, S.; D’Amato, F.; Mazzinghi, P.; Castagnoli, F.; Toci, G.; Werle, P. A cryogenically operated laser diode spectrometer for airborne measurement of stratospheric trace gases. Appl. Phys. B 2008, 90, 581–592. [Google Scholar] [CrossRef]
- Zellweger, C.; Steinbacher, M.; Buchmann, B. Evaluation of New Laser Spectrometer Techniques for in-Situ Carbon Monoxide Measurements. Atmos. Meas. Tech. 2012, 5, 2555–2567. [Google Scholar] [CrossRef]
- Nwaboh, J.A.; Qu, Z.; Werhahn, O.; Ebert, V. Interband cascade laser-based optical transfer standard for atmospheric carbon monoxide measurements. Appl. Opt. 2017, 56, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Gordon, I.E.; Rothman, L.S.; Hill, C.; Kochanov, R.V.; Tan, Y.; Bernath, P.F.; Birk, M.; Boudon, V.; Campargue, A.; Chance, K.V.; et al. The HITRAN 2016 Molecular Spectroscopic Database. J. Quant. Spectrosc. Radiat. Transf. 2017, 203, 3–69. [Google Scholar] [CrossRef]
- Li, G.; Gordon, I.E.; Rothman, L.S.; Tan, Y.; Hu, S.-M.; Kassi, S.; Campargue, A.; Medvedev, E.S. Rovibrational Line Lists for Nine Isotopologues of the CO Molecule in the X1Σ+ Ground Electronic State. Astrophys. J. Suppl. Ser. 2015, 216, 1–18. [Google Scholar] [CrossRef]
- Bartalini, S.; Borri, S.; Galli, I.; Giusfredi, G.; Mazzotti, D.; Edamura, T.; Akikusa, N.; Yamanishi, M.; De Natale, P. Measuring frequency noise and intrinsic linewidth of a room-temperature DFB quantum cascade laser. Opt. Expr. 2011, 19, 17996–18003. [Google Scholar] [CrossRef] [PubMed]
- Radio Technical Commission for Aeronautics. RTCA/DO-160D; Radio Technical Commission for Aeronautics: Washington, DC, USA, 1997; Section 7 and 8. [Google Scholar]
- Puerta, J.; Martin, P. Three and four generalized Lorentzian approximations for the Voigt line shape. Appl. Opt. 1981, 20, 3923–3928. [Google Scholar] [CrossRef] [PubMed]
- Martin, P.; Puerta, J. Generalized Lorentzian approximations for the Voigt line shape. Appl. Opt. 1981, 20, 259–263. [Google Scholar] [CrossRef] [PubMed]
- McManus, J.B.; Kebabian, P.L.; Zahniser, M.S. Astigmatic mirror multipass absorption cells for long-path-length spectroscopy. Appl. Opt. 1995, 34, 3336–3348. [Google Scholar] [CrossRef] [PubMed]
- Werle, P.; Mücke, R.; Slemr, F. The Limits of Signal Averaging in Atmospheric Trace-Gas Monitoring by Tunable Diode-Laser Absorption Spectroscopy (TDLAS). Appl. Phys. B 1993, 57, 131–139. [Google Scholar] [CrossRef]
- Park, M.; Randel, W.J.; Emmons, L.K.; Livesey, N.J. Transport pathways of carbon monoxide in the Asian summer monsoon diagnosed from Model of Ozone and Related Tracers (MOZART). J. Geophys. Res. 2009, 114, 1–11. [Google Scholar] [CrossRef]
- Barret, B.; Sauvage, B.; Bennouna, Y.; Le Flochmoen, E. Upper-tropospheric CO and O3 budget during the Asian summer monsoon. Atmos. Chem. Phys. 2016, 16, 9129–9147. [Google Scholar] [CrossRef]
- Waters, J.W. The Earth Observing System Microwave Limb Sounder (EOS MLS) on the Aura satellite. IEEE Trans. Geosci. Remote Sens. 2006, 44, 1075–1092. [Google Scholar] [CrossRef]
Sample Availability: Experimental data used in this paper are saved in the STRATOCLIM Project Wiki, available to Project partners only, according to the STRATOCLIM Data Protocol. They can be obtained upon request from the National Institute of Optics (CNR-INO). |




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Viciani, S.; Montori, A.; Chiarugi, A.; D’Amato, F. A Portable Quantum Cascade Laser Spectrometer for Atmospheric Measurements of Carbon Monoxide. Sensors 2018, 18, 2380. https://doi.org/10.3390/s18072380
Viciani S, Montori A, Chiarugi A, D’Amato F. A Portable Quantum Cascade Laser Spectrometer for Atmospheric Measurements of Carbon Monoxide. Sensors. 2018; 18(7):2380. https://doi.org/10.3390/s18072380
Chicago/Turabian StyleViciani, Silvia, Alessio Montori, Antonio Chiarugi, and Francesco D’Amato. 2018. "A Portable Quantum Cascade Laser Spectrometer for Atmospheric Measurements of Carbon Monoxide" Sensors 18, no. 7: 2380. https://doi.org/10.3390/s18072380
APA StyleViciani, S., Montori, A., Chiarugi, A., & D’Amato, F. (2018). A Portable Quantum Cascade Laser Spectrometer for Atmospheric Measurements of Carbon Monoxide. Sensors, 18(7), 2380. https://doi.org/10.3390/s18072380




