Assessment of Performance of the Industrial Process of Bulk Vacuum Packaging of Raw Meat with Nondestructive Optical Oxygen Sensing Systems
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Containers with Sensors and Meat
2.2. Storage and Measurement of O2 Levels
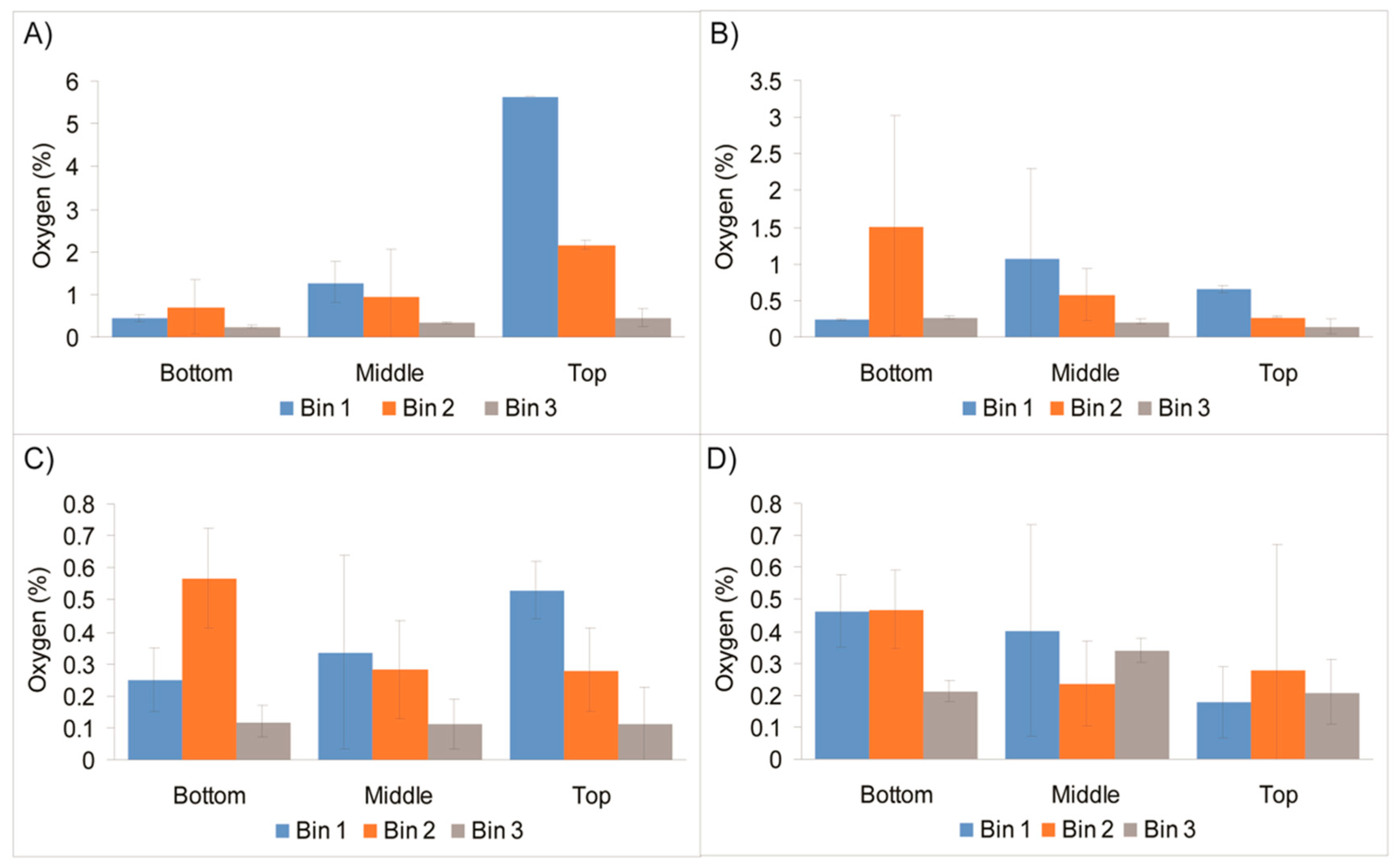
3. Results and Discussion
Design of the Sensor Trial with Bulk Vacuum-Packaged Meat
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kerry, J.P.; Butler, P. Smart Packaging Technologies for Fast Moving Consumer Goods; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Robertson, G.L. Food Packaging: Principles and Practice, 3rd ed.; Taylor & Francis: New York, NY, USA, 2012. [Google Scholar]
- Banerjee, S.; Kelly, C.; Kerry, J.P.; Papkovsky, D.B. High throughput non-destructive assessment of quality and safety of packaged food products using phosphorescent oxygen sensors. Trends Food Sci. Technol. 2016, 50, 85–102. [Google Scholar] [CrossRef]
- Smiddy, M.; Fitzgerald, M.; Kerry, J.P.; Papkovsky, D.B.; O’Sullivan, C.K.; Guilbault, G.G. Use of oxygen sensors to non-destructively measure the oxygen content in modified atmosphere and vacuum packed beef: Impact of oxygen content on lipid oxidation. Meat Sci. 2002, 61, 285–290. [Google Scholar] [CrossRef]
- Smiddy, M.; Papkovsky, D.; Kerry, J. Evaluation of oxygen content in commercial modified atmosphere packs (MAP) of processed cooked meats. Food Res. Int. 2002, 35, 571–575. [Google Scholar] [CrossRef]
- Papkovsky, D.B.; Dmitriev, R.I. Biological detection by optical oxygen sensing. Chem. Soc. Rev. 2013, 42, 8700–8732. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-D.; Wolfbeis, O.S. Optical methods for sensing and imaging oxygen: Materials, spectroscopies and applications. Chem. Soc. Rev. 2014, 43, 3666–3761. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.A.; Toncelli, C.; Kerry, J.P.; Papkovsky, D.B. Phosphorescent O2 sensors based on polyolefin fabric materials. J. Mater. Chem. C 2014, 2, 2169–2174. [Google Scholar] [CrossRef]
- Toncelli, C.; Arzhakova, O.V.; Dolgova, A.; Volynskii, A.L.; Bakeev, N.F.; Kerry, J.P.; Papkovsky, D.B. Oxygen-sensitive phosphorescent nanomaterials produced from high-density polyethylene films by local solvent-crazing. Anal. Chem. 2014, 86, 1917–1923. [Google Scholar] [CrossRef] [PubMed]
- Papkovsky, D.B.; Smiddy, M.A.; Papkovskaia, N.Y.; Kerry, J.P. Nondestructive measurement of oxygen in modified atmosphere packaged hams using a phase-fluorimetric sensor system. J. Food Sci. 2002, 67, 3164–3169. [Google Scholar] [CrossRef]
- O’Mahony, F.C.; O’Riordan, T.C.; Papkovskaia, N.; Kerry, J.P.; Papkovsky, D.B. Non-destructive assessment of oxygen levels in industrial modified atmosphere packaged cheddar cheese. Food Control 2006, 17, 286–292. [Google Scholar] [CrossRef]
- Hempel, A.; Sullivan, M.; Papkovsky, D.; Kerry, J. Assessment and use of optical oxygen sensors as tools to assist in optimal product component selection for the development of packs of ready-to-eat mixed salads and for the non-destructive monitoring of in-pack oxygen levels using chilled storage. Foods 2013, 2, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Hempel, A.W.; O’Sullivan, M.G.; Papkovsky, D.B.; Kerry, J.P. Use of smart packaging technologies for monitoring and extending the shelf-life quality of modified atmosphere packaged (MAP) bread: Application of intelligent oxygen sensors and active ethanol emitters. Eur. Food Res. Technol. 2013, 237, 117–124. [Google Scholar] [CrossRef]
- O’Mahony, F.C.; O’Riordan, T.C.; Papkovskaia, N.; Ogurtsov, V.I.; Kerry, J.P.; Papkovsky, D.B. Assessment of oxygen levels in convenience-style muscle-based sous vide products through optical means and impact on shelf-life stability. Packag. Technol. Sci. 2004, 17, 225–234. [Google Scholar] [CrossRef]
- O’Callaghan, K.; Papkovsky, D.; Kerry, J. An assessment of the influence of the industry distribution chain on the oxygen levels in commercial modified atmosphere packaged cheddar cheese using non-destructive oxygen sensor technology. Sensors 2016, 16, 916. [Google Scholar] [CrossRef] [PubMed]
- Morsy, M.K.; Khalaf, H.H.; Sharoba, A.M.; El-Tanahi, H.H. Applicability of biosensor and oxygen sensor for monitoring spoilage and bacterial contaminants of packed minced beef and poultry. In Proceedings of the 2nd International Conference on Biotechnology Applications in Agriculture (ICBAA), Hurghada, Egypt, 8–12 April 2014. [Google Scholar]
- Fitzgerald, M.; Papkovsky, D.; Smiddy, M.; Kerry, J.; O’Sullivan, C.; Buckley, D.; Guilbault, G. Nondestructive monitoring of oxygen profiles in packaged foods using phase-fluorimetric oxygen sensor. J. Food Sci. 2001, 66, 105–110. [Google Scholar] [CrossRef]

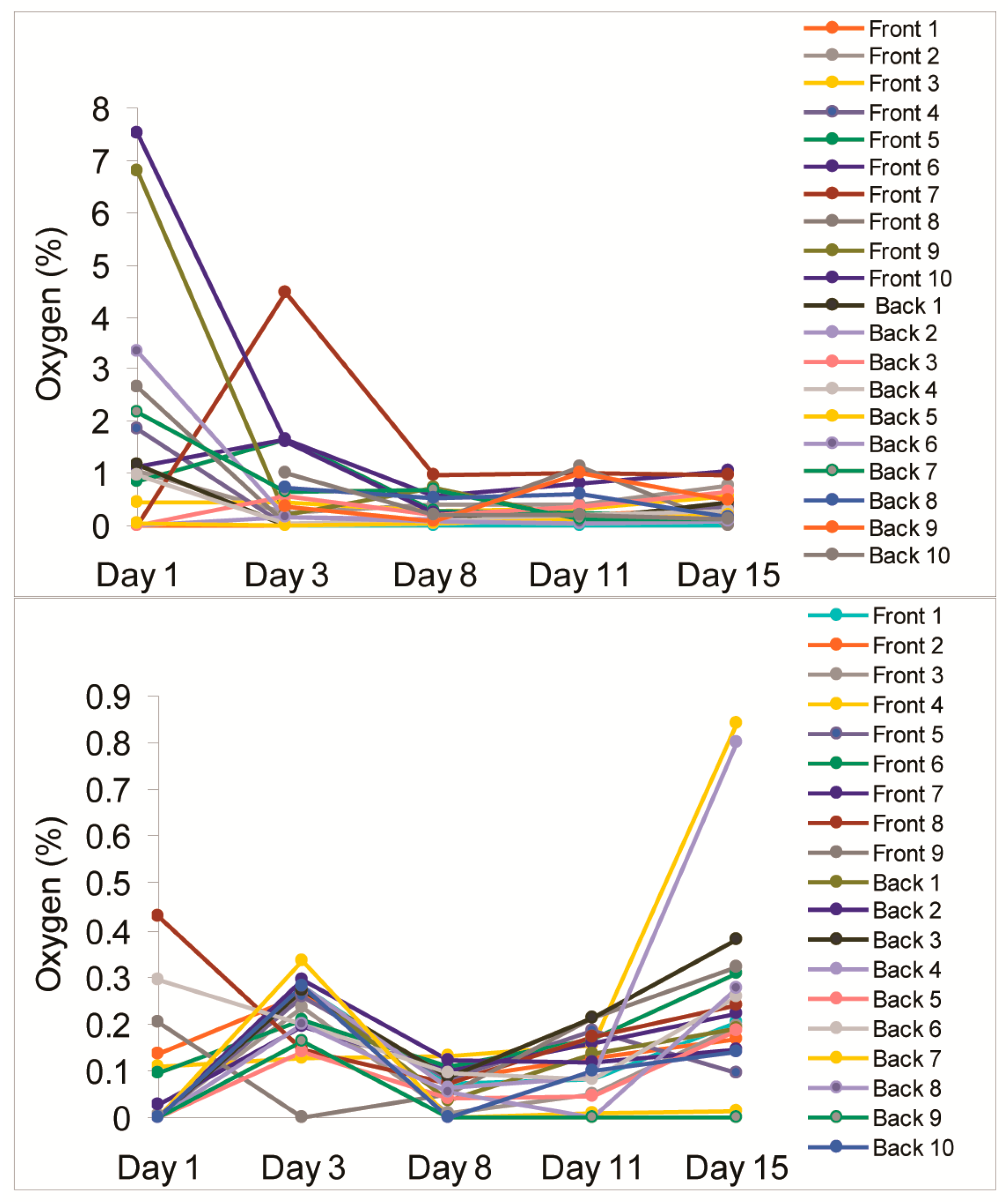
| Location | Day 1 | Day 3 | Day 8 | Day 11 | Day 15 |
|---|---|---|---|---|---|
| Front 1 | 0.00 | 0.00 | 0.11 | 0.22 | 0.32 |
| Front 2 | 1.05 | 0.25 | 0.42 | 0.42 | 0.75 |
| Front 3 | 0.45 | 0.46 | 0.29 | 0.33 | 0.57 |
| Front 4 | 1.85 | 0.01 | 0.13 | 0.14 | 0.36 |
| Front 5 | 0.83 | 1.63 | 0.27 | 0.23 | 0.16 |
| Front 6 | 1.13 | 1.64 | 0.56 | 0.81 | 1.06 |
| Front 7 | 0.00 | 4.47 | 0.97 | 1.02 | 0.98 |
| Front 8 | 2.64 | 0.03 | 0.08 | 1.13 | 0.00 |
| Front 9 | 6.80 | 0.21 | 0.72 | 0.12 | 0.08 |
| Front 10 | 7.53 | 1.63 | 0.25 | 0.15 | 0.23 |
| Back 1 | 1.17 | 0.00 | 0.11 | 0.14 | 0.43 |
| Back 2 | 0.00 | 0.17 | 0.13 | 0.07 | 0.10 |
| Back 3 | 0.00 | 0.56 | 0.19 | 0.34 | 0.63 |
| Back 4 | 0.98 | 0.05 | 0.08 | 0.21 | 0.29 |
| Back 5 | 0.06 | 0.02 | 0.04 | 0.14 | 0.19 |
| Back 6 | 3.35 | 0.18 | 0.07 | 0.04 | 0.07 |
| Back 7 | 2.17 | 0.65 | 0.70 | 0.10 | 0.12 |
| Back 8 | N/A | 0.72 | 0.53 | 0.59 | 0.17 |
| Back 9 | N/A | 0.35 | 0.07 | 0.99 | 0.49 |
| Back 10 | N/A | 1.02 | 0.21 | 0.20 | 0.11 |
| Mean, % O2 | 1.76 | 0.70 | 0.30 | 0.37 | 0.35 |
| SD, % O2 | 2.26 | 1.05 | 0.27 | 0.35 | 0.31 |
| Location | Day 1 | Day 3 | Day 8 | Day 11 | Day 15 |
|---|---|---|---|---|---|
| Front 1 | 0.33 | 0.50 | 0.00 | 0.47 | 0.36 |
| Front 2 | 0.00 | 0.41 | 0.11 | 0.76 | 0.79 |
| Front 3 | 0.45 | 0.46 | 0.11 | 0.15 | 0.00 |
| Front 4 | 0.00 | 0.36 | 0.14 | 0.28 | 0.00 |
| Front 5 | 0.00 | 0.27 | 0.08 | 0.31 | 0.00 |
| Front 6 | 0.23 | 0.31 | 0.59 | 0.00 | 0.23 |
| Front 7 | 0.51 | 0.36 | 0.13 | 0.11 | 0.34 |
| Front 8 | 3.52 | 0.42 | 0.08 | 0.82 | 0.32 |
| Front 9 | 0.63 | 0.41 | 0.17 | 0.30 | 0.69 |
| Front 10 | 2.61 | 0.00 | 0.17 | 0.00 | 0.66 |
| Back 1 | 1.24 | 0.38 | 0.18 | 0.39 | 0.43 |
| Back 2 | 2.23 | 7.19 | 3.36 | 1.65 | 0.50 |
| Back 3 | 0.00 | 0.17 | 0.00 | 0.00 | 0.74 |
| Back 4 | 0.66 | 0.49 | 0.00 | 0.55 | 0.43 |
| Back 5 | 3.32 | 0.80 | 0.26 | 0.50 | 0.21 |
| Back 6 | 0.73 | 1.71 | 0.08 | 0.19 | 0.43 |
| Back 7 | 2.19 | 0.30 | 0.28 | 0.32 | 0.25 |
| Back 8 | 0.00 | 0.28 | 0.14 | 0.19 | 0.00 |
| Back 9 | 3.17 | N/A | N/A | N/A | N/A |
| Back 10 | 3.07 | N/A | N/A | N/A | N/A |
| Mean, % O2 | 1.24 | 0.82 | 0.33 | 0.39 | 0.35 |
| SD, % O2 | 1.30 | 1.63 | 0.77 | 0.40 | 0.26 |
| Location | Day 1 | Day 3 | Day 8 | Day 11 | Day 15 |
|---|---|---|---|---|---|
| Front 1 | 0.00 | 0.28 | 0.07 | 0.08 | 0.20 |
| Front 2 | 0.14 | 0.26 | 0.08 | 0.13 | 0.17 |
| Front 3 | 0.01 | 0.24 | 0.01 | 0.05 | 0.20 |
| Front 4 | 0.11 | 0.13 | 0.13 | 0.16 | 0.84 |
| Front 5 | 0.00 | 0.26 | 0.08 | 0.19 | 0.10 |
| Front 6 | 0.10 | 0.21 | 0.11 | 0.17 | 0.31 |
| Front 7 | 0.03 | 0.19 | 0.10 | 0.16 | 0.22 |
| Front 8 | 0.43 | 0.15 | 0.07 | 0.17 | 0.24 |
| Front 9 | 0.20 | 0.00 | 0.05 | 0.21 | 0.32 |
| Back 1 | 0.00 | 0.27 | 0.04 | 0.13 | 0.19 |
| Back 2 | 0.00 | 0.30 | 0.12 | 0.12 | 0.14 |
| Back 3 | 0.00 | 0.27 | 0.09 | 0.21 | 0.38 |
| Back 4 | 0.00 | 0.28 | 0.06 | 0.09 | 0.80 |
| Back 5 | 0.00 | 0.14 | 0.04 | 0.04 | 0.19 |
| Back 6 | 0.30 | 0.20 | 0.10 | 0.08 | 0.26 |
| Back 7 | 0.00 | 0.34 | 0.00 | 0.01 | 0.02 |
| Back 8 | 0.00 | 0.20 | 0.10 | 0.00 | 0.27 |
| Back 9 | 0.00 | 0.16 | 0.00 | 0.00 | 0.00 |
| Back 10 | 0.00 | 0.28 | 0.00 | 0.10 | 0.14 |
| Mean, % O2 | 0.07 | 0.22 | 0.07 | 0.11 | 0.26 |
| SD, % O2 | 0.12 | 0.08 | 0.04 | 0.07 | 0.22 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kelly, C.A.; Cruz-Romero, M.; Kerry, J.P.; Papkovsky, D.P. Assessment of Performance of the Industrial Process of Bulk Vacuum Packaging of Raw Meat with Nondestructive Optical Oxygen Sensing Systems. Sensors 2018, 18, 1395. https://doi.org/10.3390/s18051395
Kelly CA, Cruz-Romero M, Kerry JP, Papkovsky DP. Assessment of Performance of the Industrial Process of Bulk Vacuum Packaging of Raw Meat with Nondestructive Optical Oxygen Sensing Systems. Sensors. 2018; 18(5):1395. https://doi.org/10.3390/s18051395
Chicago/Turabian StyleKelly, Caroline A., Malco Cruz-Romero, Joseph P. Kerry, and Dmitri P. Papkovsky. 2018. "Assessment of Performance of the Industrial Process of Bulk Vacuum Packaging of Raw Meat with Nondestructive Optical Oxygen Sensing Systems" Sensors 18, no. 5: 1395. https://doi.org/10.3390/s18051395
APA StyleKelly, C. A., Cruz-Romero, M., Kerry, J. P., & Papkovsky, D. P. (2018). Assessment of Performance of the Industrial Process of Bulk Vacuum Packaging of Raw Meat with Nondestructive Optical Oxygen Sensing Systems. Sensors, 18(5), 1395. https://doi.org/10.3390/s18051395





