Electrochemical Biosensor for Nitrite Based on Polyacrylic-Graphene Composite Film with Covalently Immobilized Hemoglobin
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instrumentation
2.3. Synthesis of Graphene Nanosheets
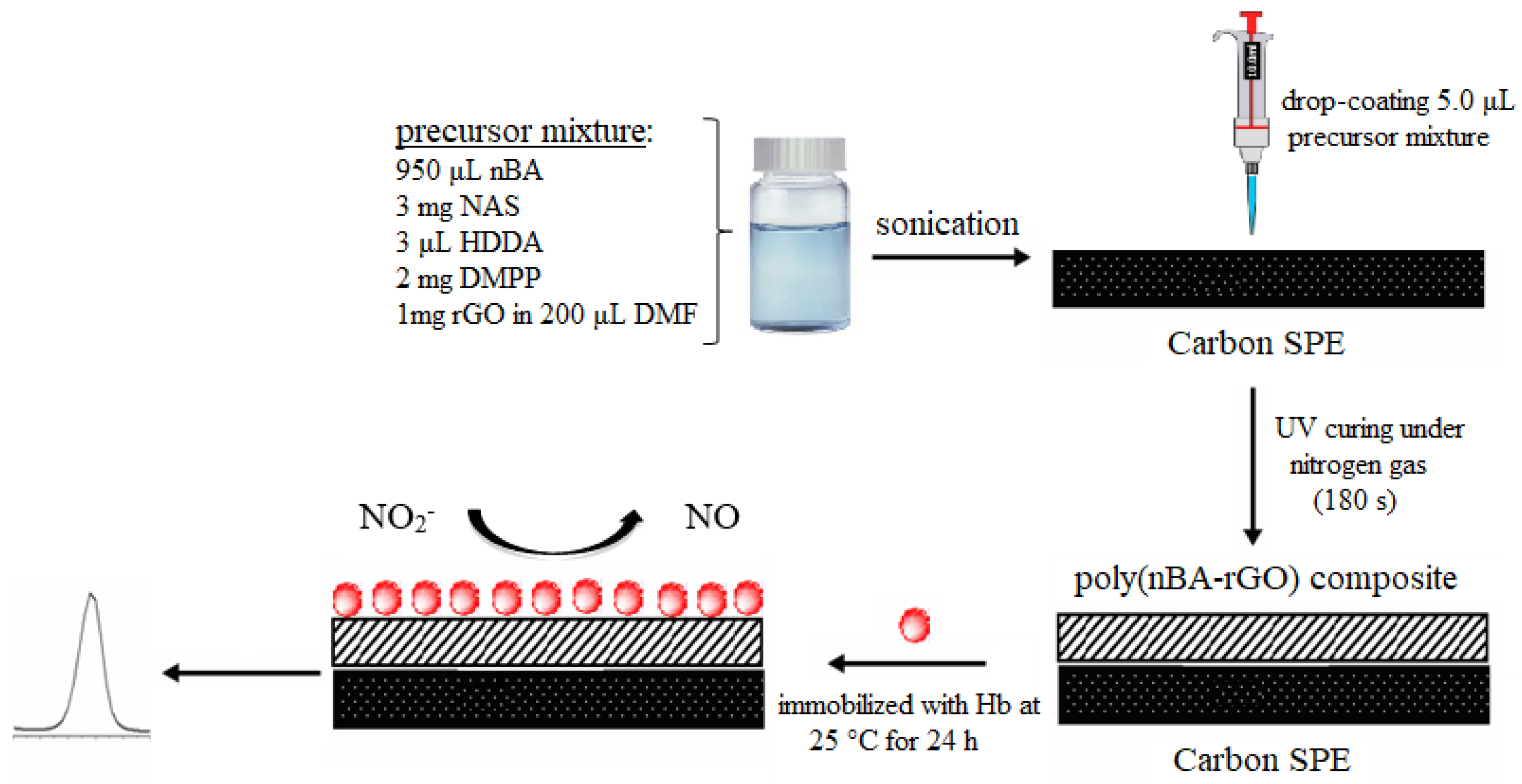
2.4. Fabrication and Characterization of Electrochemical Nitrite Biosensor
2.5. Optimizing the Electrochemical Nitrite Biosensor Based on Poly(nBA)-rGO Supporting Matrix
2.6. Validation and Recovery Studies
3. Results and Discussion
3.1. Physical and Chemical Characterizations of the Synthesized Graphene
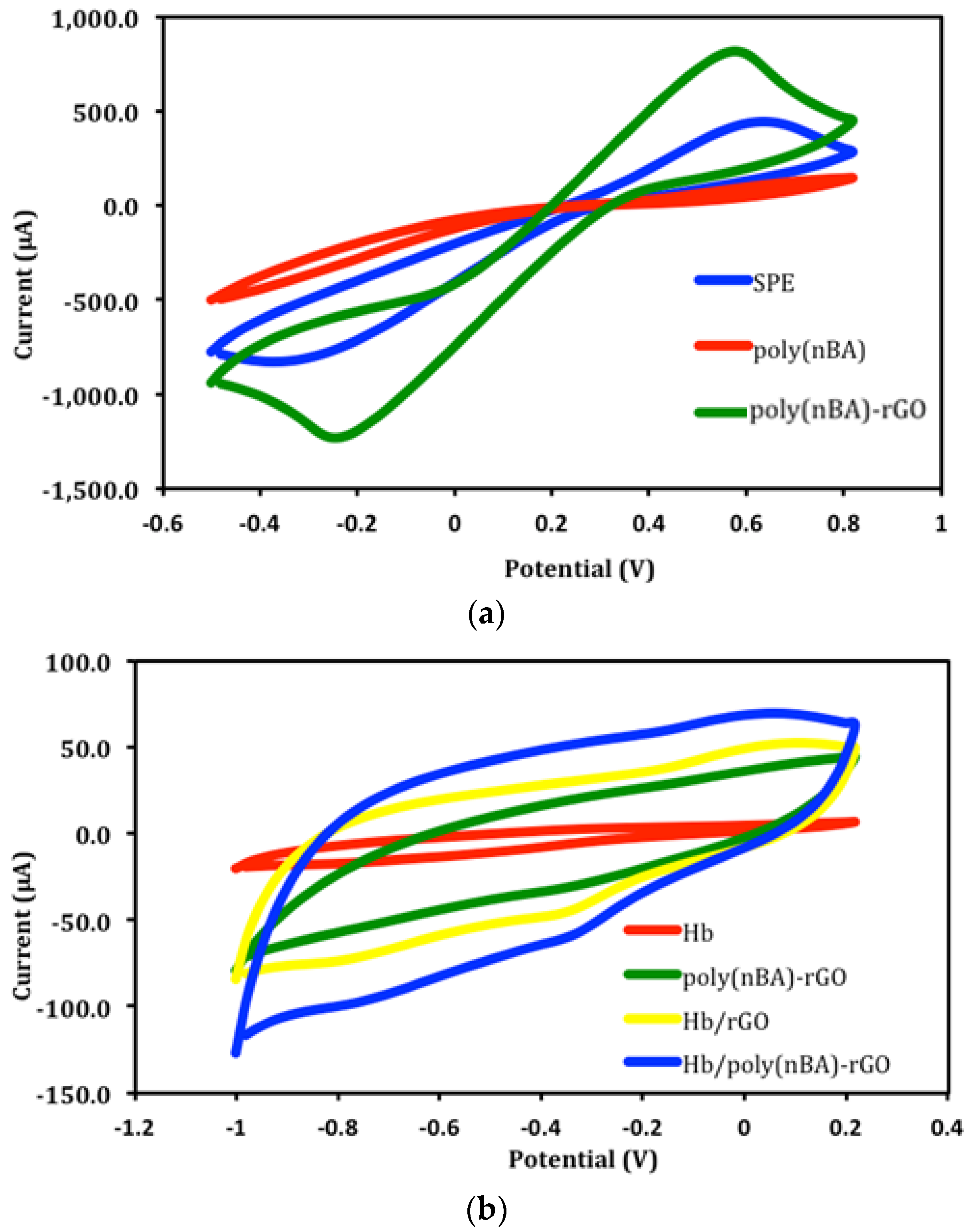
3.2. Electrochemical Characteristics of Polyacrylic-Graphene Composite Film Coated Electrodes
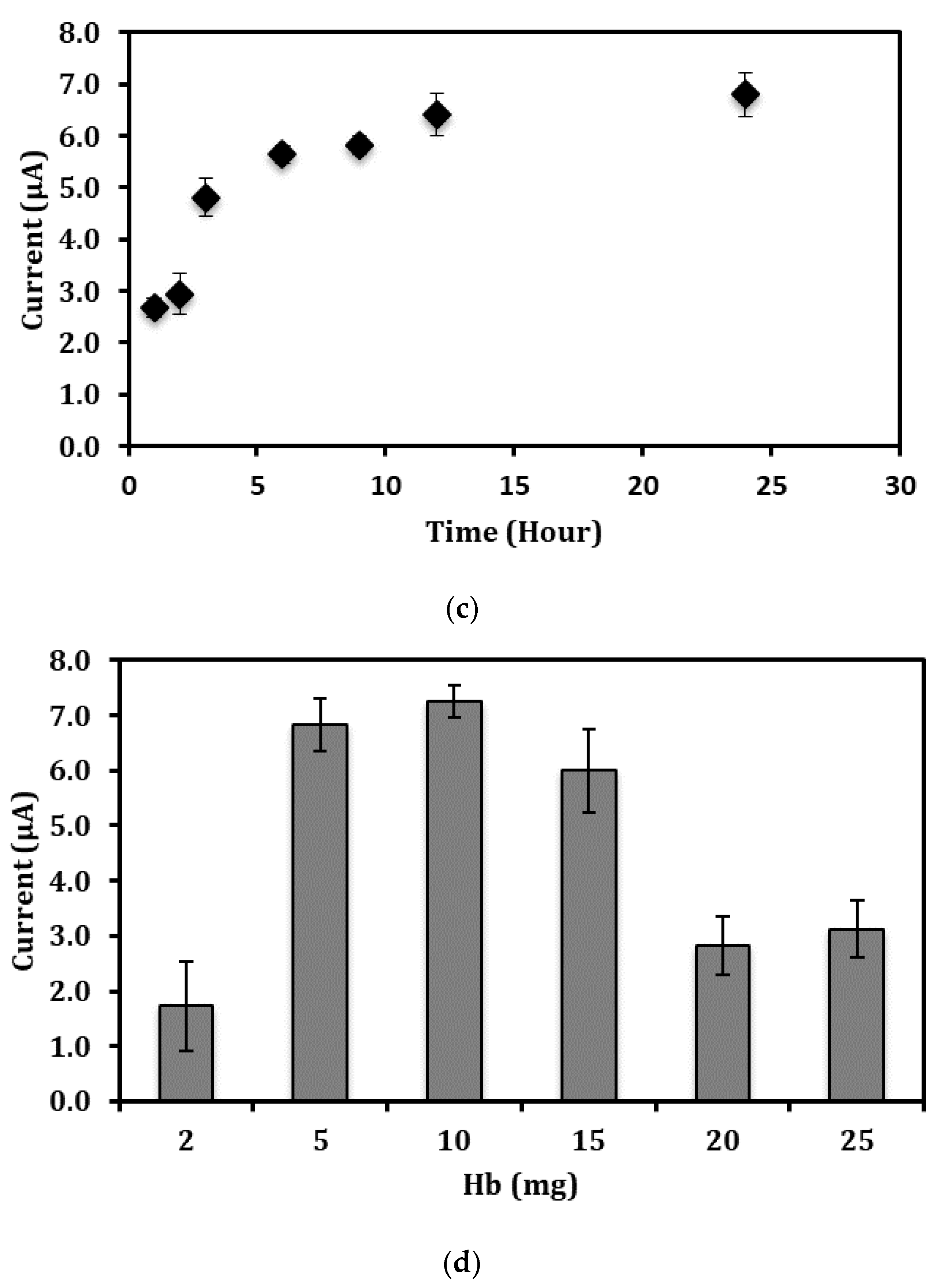
3.3. Effects of rGO, NAS, and Hb Loadings on the Nitrite Biosensor Response
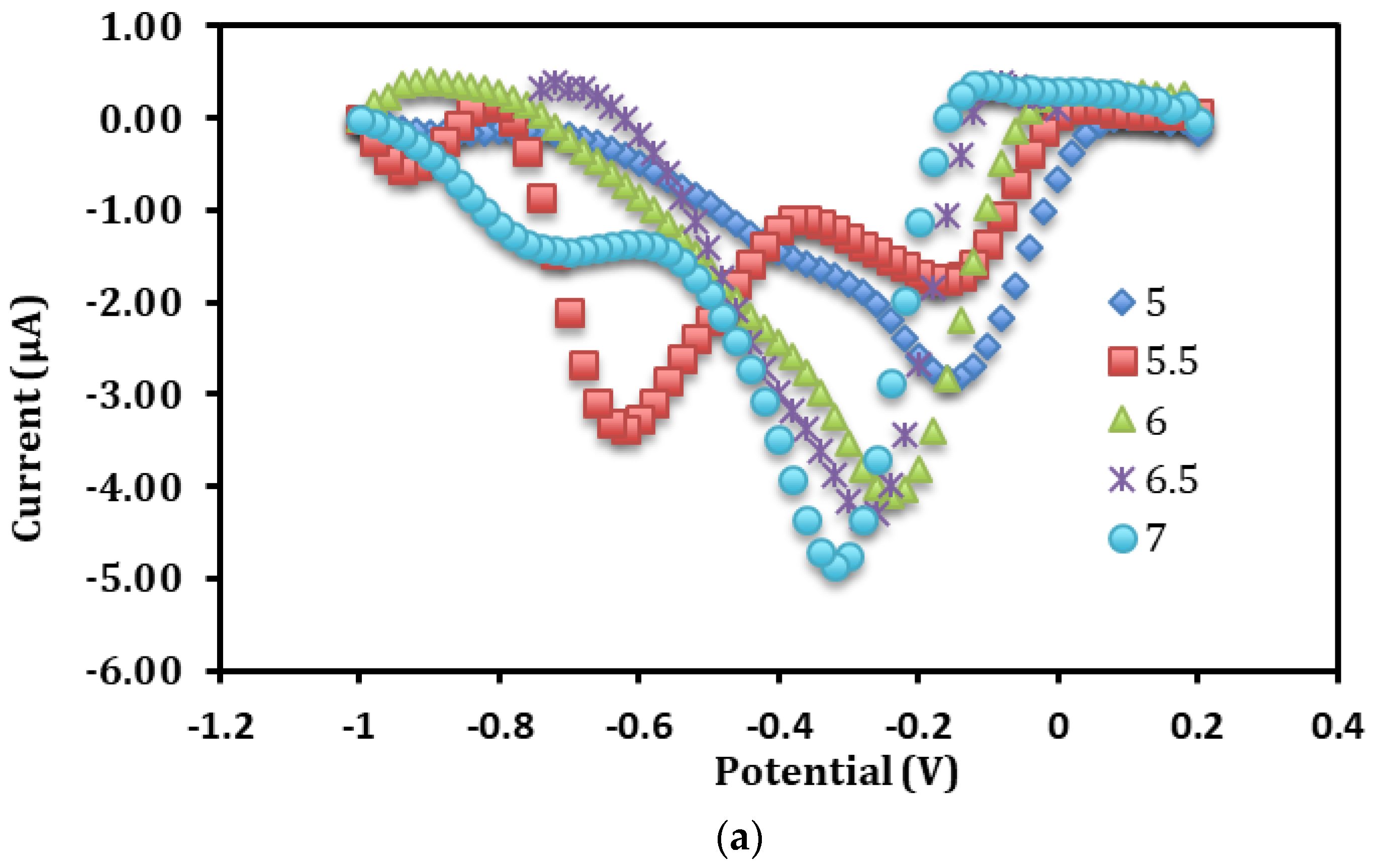
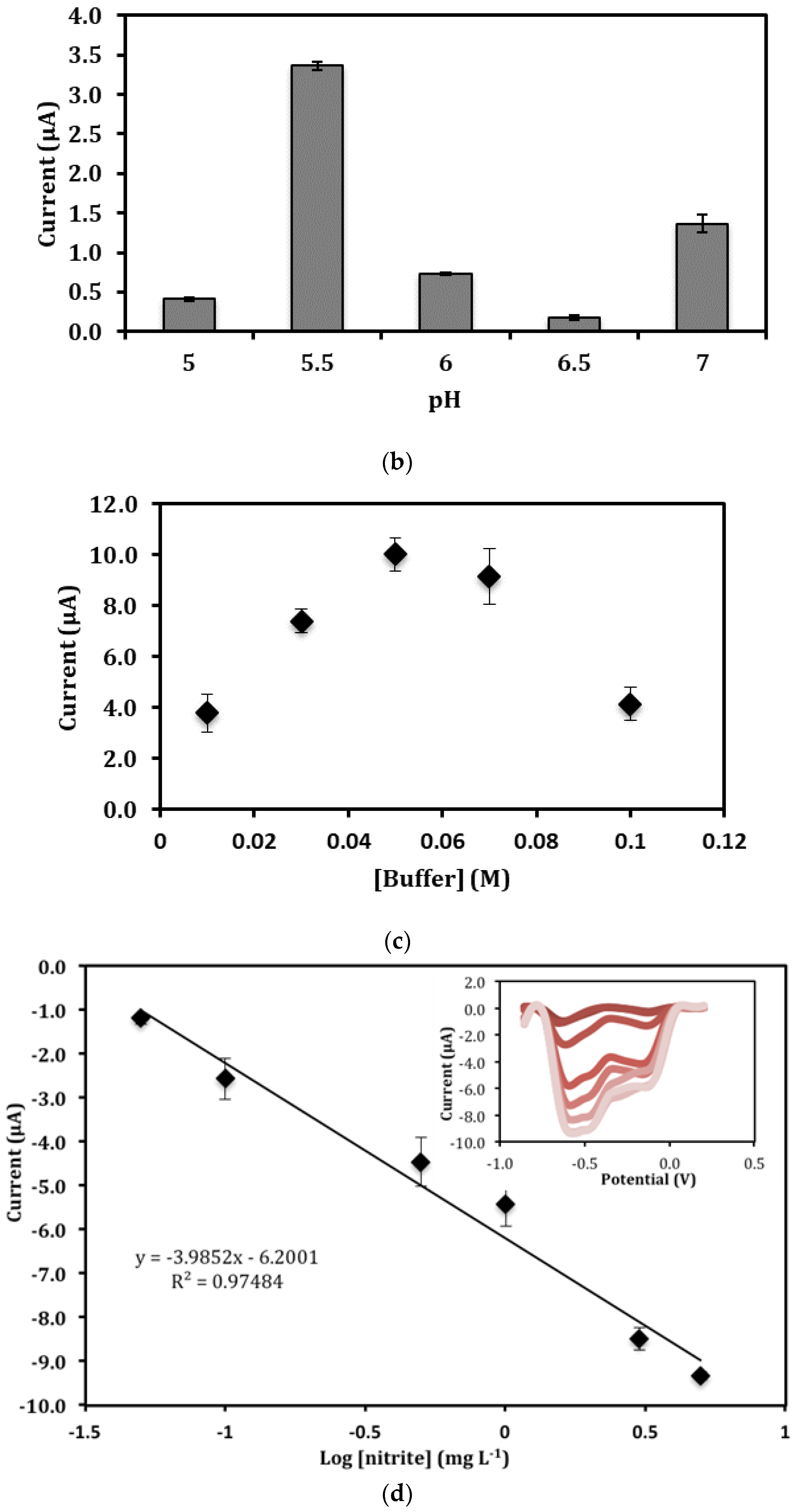
3.4. Influence of pH and Buffer Concentration on the Direct Electrochemistry of Immobilized Hb
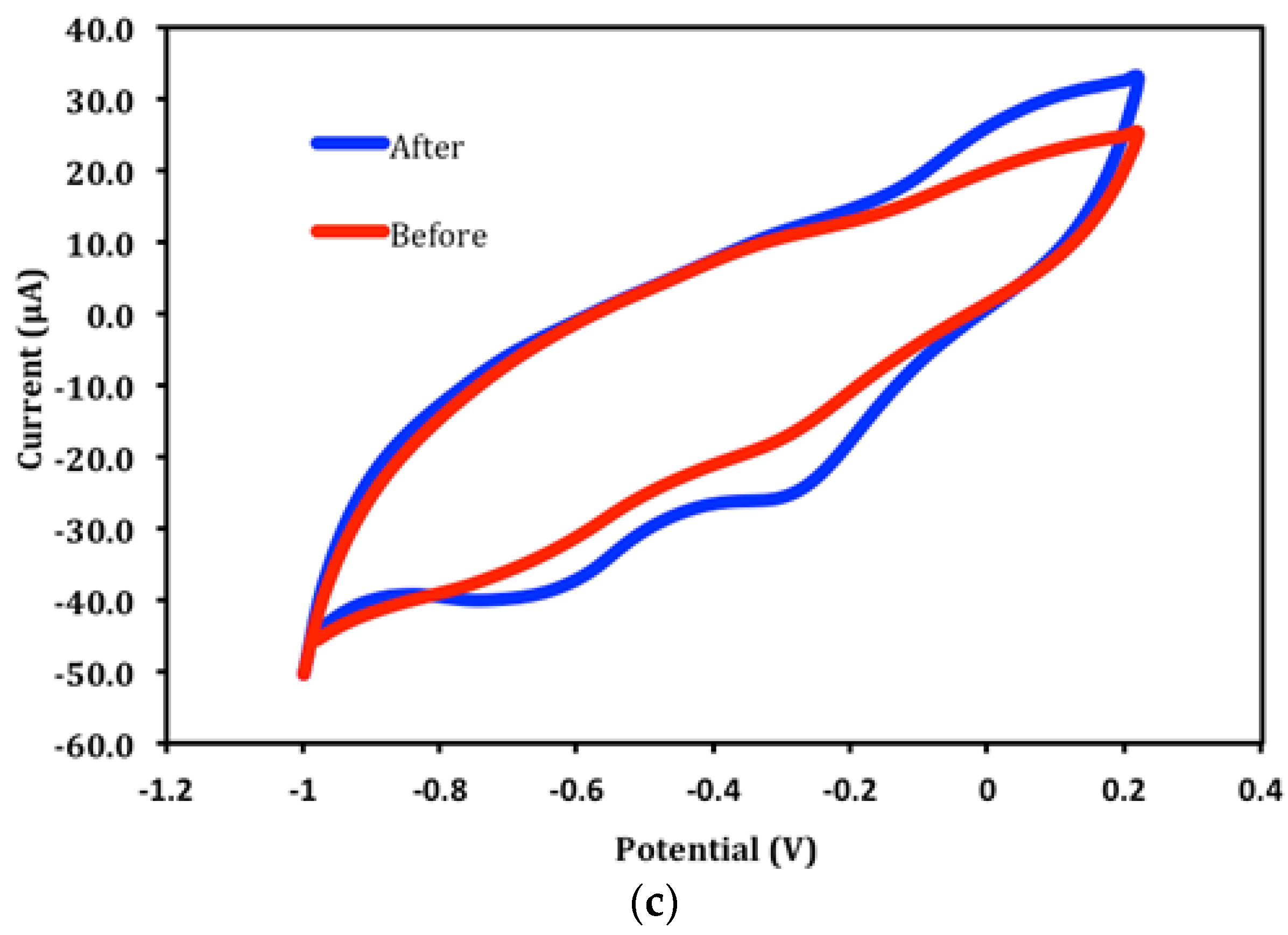
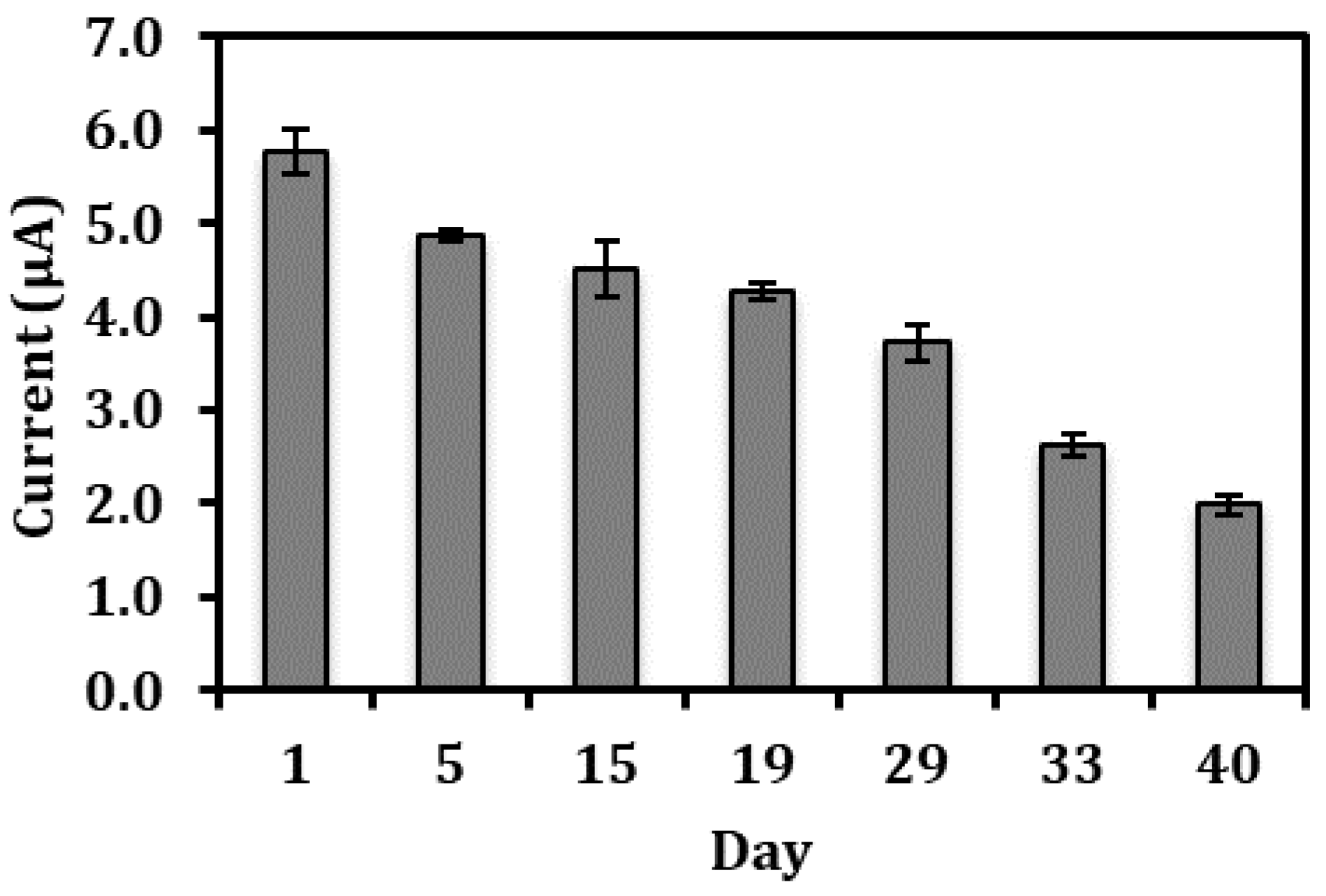
3.5. Long Term Stability of the Biosensor Response and Interference Studies
3.6. Validation of the Electrochemical Biosensor for the Detection of Nitrite in Food Samples
3.7. A Comparison of Analytical Performance of Nitrite Biosensor with Several Reported Electrochemical Nitrite Biosensors
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kim-Shapiro, D.B.; Gladwin, M.T.; Patel, R.P.; Hogg, N. The reaction between nitrite and hemoglobin: The role of nitrite in hemoglobin-mediated hypoxic vasodilation. J. Inorg. Biochem. 2005, 99, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Yue, R.; Lu, Q.; Zhou, Y. A novel nitrite biosensor based on single-layer graphene nanoplatelet-protein composite film. Biosens. Bioelectron. 2011, 26, 4436–4441. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Dai, Z. Amperometric biosensor for hydrogen peroxide and nitrite based on hemoglobin immobilized on one-dimensional gold nanoparticle. Sens. Actuators B Chem. 2009, 140, 222–226. [Google Scholar] [CrossRef]
- Mani, V.; Periasamy, A.P.; Chen, S.M. Highly selective amperometric nitrite sensor based on chemically reduced graphene oxide modified electrode. Electrochem. Commun. 2012, 17, 75–78. [Google Scholar] [CrossRef]
- Nur Syarmim, M.N.; Tan, L.L.; Lee, Y.H.; Chong, K.F.; Saiful Nizam, T. Acrylic microspheres-based optosensor for visual detection of nitrite. Food Chem. 2016, 207, 132–138. [Google Scholar] [CrossRef]
- Chen, X.; Wang, F.; Chen, Z. An electropolymerized Nile Blue sensing film-based nitrite sensor and application in food analysis. Anal. Chim. Acta 2008, 623, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, S. An amperometric nitrite biosensor based on the bioelectrocatalysis of hemoglobin incorporated in sol-gel film. Am. J. Biomed. Sci. 2009, 1, 171–177. [Google Scholar] [CrossRef][Green Version]
- Liu, P.; Zhang, X.H.; Feng, L.J.; Xiong, H.Y.; Wang, S.F. Direct electrochemistry of hemoglobin on graphene nanosheet-based modified electrode and its electrocatalysis to nitrite. Am. J. Biomed. Sci. 2011, 3, 69–76. [Google Scholar] [CrossRef]
- Yang, W.; Bai, Y.; Li, Y.; Sun, C. Amperometric nitrite sensor based on hemoglobin/colloidal gold nanoparticles immobilized on a glassy carbon electrode by a titania sol-gel film. Anal. Bioanal. Chem. 2005, 382, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Gu, H.Y.; Yu, A.M.; Chen, H.Y. Direct electron transfer and characterization of hemoglobin immobilized on a Au colloid–cysteamine-modified gold electrode. J. Electroanal. Chem. 2001, 516, 119–126. [Google Scholar] [CrossRef]
- Huang, H.; Hu, N.; Zeng, Y.; Zhou, G. Electrochemistry and electrocatalysis with heme proteins in chitosan biopolymer films. Anal. Biochem. 2002, 308, 141–151. [Google Scholar] [CrossRef]
- Wang, Q.; Lu, Q.; Yang, B. Direct electrochemistry and electrocatalysis of hemoglobin immobilized on carbon paste electrode by silica sol-gel film. Biosens. Bioelectron. 2004, 19, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.X.; Lu, T.H.; Xia, Y.Y. Direct electrochemistry and bioelectrocatalysis of hemoglobin immobilized on carbon black. Bioelectrochemistry 2007, 71, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Zore, O.V.; Lenehan, P.J.; Kumar, C.V.; Kasi, R.M. Efficient biocatalysis in organic media with hemoglobin and poly(acrylic acid) nanogels. Langmuir 2014, 30, 5176–5184. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, A.; Zore, O.V.; Thilakarathne, V.K.; Briand, V.A.; Lenehan, P.J.; Lei, Y.; Kasi, R.M.; Kumar, C.V. “Stable-on-the-table” biosensors: Hemoglobin-poly (acrylic acid) nanogel bioelectrodes with high thermal stability and enhanced electroactivity. Sensors 2015, 15, 23868–23885. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.H.; Wan, Y.Q.; Zou, G.L. Direct electrochemistry and electrochemical catalysis of immobilized hemoglobin in an ethanol–water mixture. Anal. Bioanal. Chem. 2006, 385, 1470–1476. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Wu, H.; Chen, S.; Lu, L.; Shen, H.; Jia, N. Direct electrochemistry and electrocatalysis of hemoglobin immobilized in poly(ethylene glycol) grafted multi-walled carbon nanotubes. Electrochim. Acta 2009, 54, 7078–7084. [Google Scholar] [CrossRef]
- Wen, W.; Chen, W.; Ren, Q.Q.; Hu, X.Y.; Xiong, H.Y.; Zhang, X.H.; Zhao, Y.D. A highly sensitive nitric oxide biosensor based on hemoglobin–chitosan/graphene–hexadecyltrimethylammonium bromide nanomatrix. Sens. Actuators B Chem. 2012, 166–167, 444–450. [Google Scholar] [CrossRef]
- Jiang, J.J.; Fan, W.J.; Du, X.Z. Nitrite electrochemical biosensing based on coupled graphene and gold nanoparticles. Biosens. Bioelectron. 2014, 51, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Bi, C.Y. A novel nitrite biosensor based on direct electron transfer of hemoglobin immobilized on a graphene oxide/Au nanoparticles/multiwalled carbon nanotubes nanocomposite film. RSC Adv. 2014, 4, 31573–31580. [Google Scholar] [CrossRef]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Kovtyukhova, N.I.; Ollivier, P.J.; Martin, B.R.; Mallouk, T.E.; Chizhik, S.A.; Buzaneva, E.V.; Gorchinskiy, A.D. Layer-By-Layer Assembly of Ultrathin Composite Films from Micron-Sized Graphite Oxide Sheets and Polycations. Chem. Mater. 1999, 11, 771–778. [Google Scholar] [CrossRef]
- Stankovich, S.; Piner, R.D.; Chen, X.Q.; Wu, N.Q.; Nguyen, S.T.; Ruoff, R.S. Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly(sodium 4-styrenesulfonate). J. Mater. Chem. 2006, 16, 155–158. [Google Scholar] [CrossRef]
- Raja Zaidatul, A.R.J.; Lee, Y.H.; Tan, L.L.; Chong, K.F. A Biosensor for Genetic Modified Soybean DNA Determination via Adsorption of Anthraquinone-2-sulphonic Acid in Reduced Graphene Oxide. Electroanalysis 2018, 30, 250–258. [Google Scholar] [CrossRef]
- Ulianas, A.; Lee, Y.H.; Musa, A.; Lau, H.Y.; Zamri, I.; Tan, L.L. A regenerable screen-printed DNA biosensor based on acrylic microsphere–gold nanoparticle composite for genetically modified soybean determination. Sens. Actuators B Chem. 2014, 190, 694–701. [Google Scholar] [CrossRef]
- Ulianas, A.; Lee, Y.H.; Musa, A. A biosensor for urea from succinimide-modified acrylic microspheres based on reflectance transduction. Sensors 2011, 11, 8323–8338. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Alva, S.; Musa, A. Ammonium ion sensor based on photocured and self-plasticising acrylic films for the analysis of sewage. Sens. Actuators B Chem. 2004, 98, 160–165. [Google Scholar] [CrossRef]
- Lee, Y.H.; Hall, E. Methacrylic–acrylic polymers in ion-selective membranes: Achieving the right polymer recipe. Anal. Chim. Acta 2000, 403, 77–89. [Google Scholar] [CrossRef]
- Sharina, A.H.; Lee, Y.H.; Musa, A. Effects of Gold Nanoparticles on the Response of Phenol Biosensor Containing Photocurable Membrane with Tyrosinase. Sensors 2008, 8, 6408–6416. [Google Scholar] [CrossRef] [PubMed]
- Ulianas, A.; Lee, Y.H.; Sharina, A.H.; Tan, L.L. An electrochemical DNA microbiosensor based on succinimide-modified acrylic microspheres. Sensors 2012, 12, 5445–5460. [Google Scholar] [CrossRef] [PubMed]
- Chaix, C.; Pacard, E.; Elaı̈ssari, A.; Hilaire, J.-F.; Pichot, C. Surface functionalization of oil-in-water nanoemulsion with a reactive copolymer: Colloidal characterization and peptide immobilization. Colloids Surf. B Biointerfaces 2003, 29, 39–52. [Google Scholar] [CrossRef]
- Chen, J.; Chiu, S. A poly(N-isopropylacrylamide-co-N-acryloxysuccinimide-co-2-hydroxyethyl methacrylate) composite hydrogel membrane for urease immobilization to enhance urea hydrolysis rate by temperature swing. Enzyme Microb. Technol. 2000, 26, 359–367. [Google Scholar] [CrossRef]
- D’Agosto, F.; Charreyre, M.T.; Pichot, C. Side-Product of N-Acryloyloxysuccinimide Synthesis or Useful New Bifunctional Monomer? Macromol. Biosci. 2001, 1, 322–328. [Google Scholar] [CrossRef]
- Yew, P.L.; Lee, Y.H. A Potentiometric Formaldehyde Biosensor Based on Immobilization of Alcohol Oxidase on Acryloxysuccinimide-modified Acrylic Microspheres. Sensors 2010, 10, 9963–9981. [Google Scholar] [CrossRef]
- Liu, X.Q.; Guan, Y.P.; Liu, H.Z.; Ma, Z.Y.; Yang, Y.; Wu, X.B. Preparation and characterization of magnetic polymer nanospheres with high protein binding capacity. J. Magn. Magn. Mater. 2005, 293, 111–118. [Google Scholar] [CrossRef]
- Ngounou, B.; Neugebauer, S.; Frodl, A.; Reiter, S.; Schuhmann, W. Combinatorial synthesis of a library of acrylic acid-based polymers and their evaluation as immobilisation matrix for amperometric biosensors. Electrochim. Acta 2004, 49, 3855–3863. [Google Scholar] [CrossRef]
- Ling, T.L.; Ahmad, M.; Heng, L.Y. An amperometric biosensor based on alanine dehydrogenase for the determination of low level of ammonium ion in water. J. Sens. 2011, 2011, 980709. [Google Scholar] [CrossRef]
- Turdean, G.L.; Szabo, G. Nitrite detection in meat products samples by square-wave voltammetry at a new single walled carbon naonotubes–myoglobin modified electrode. Food Chem. 2015, 179, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, G.; Nevin, O.; Ayca, O.; Filiz, S. Voltammetric determination of nitrite in meat products using polyvinylimidazole modified carbon paste electrode. Food Chem. 2014, 152, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Ensafi, A.A.; Maryam, A. Highly selective optical nitrite sensor for food analysis based on Lauth’s violet–triacetyl cellulose membrane film. Food Chem. 2012, 13, 1600–1606. [Google Scholar] [CrossRef] [PubMed]



| Interfering Ions | Biosensor Response (%) | ||
|---|---|---|---|
| 1:50 | 1:100 | 1:200 | |
| Na+ | 4.26 ± 0.07 | 8.78 ± 0.04 | 16.85 ± 0.25 |
| Ca2+ | 3.17± 0.46 | 4.39 ± 0.25 | 7.23 ± 0.10 |
| K+ | 4.81 ± 0.07 | 10.92 ± 0.51 | 16.74 ± 0.17 |
| Mg2+ | 3.17 ± 0.46 | 3.88 ± 0.30 | 4.20 ± 0.77 |
| NH4− | 1.31 ± 0.01 | 2.13 ± 0.26 | 3.17± 0.46 |
| NO3− | 1.68 ± 0.03 | 3.92 ± 0.32 | 4.68 ± 0.03 |
| Spiked Nitrite (mg L−1) | Nitrite Concentration Determined by Biosensor (mg L−1) | Recovery (%) | Nitrite Concentration Determined by Biosensor (mg L−1) | Recovery (%) |
|---|---|---|---|---|
| Processed EBN | Unprocessed EBN | |||
| 0.3 | 0.31 | 103 | 0.30 | 100 |
| 0.5 | 0.51 | 102 | 0.52 | 104 |
| 0.7 | 0.72 | 103 | 0.71 | 101 |
| 1.0 | 1.02 | 102 | 1.09 | 109 |
| 2.0 | 2.15 | 108 | 2.04 | 102 |
| Immobilization Matrix Materials | Linear Range (mM) | Limit of Detection (mM) | Long Term Stability (Day) | Reference |
|---|---|---|---|---|
| Poly(nBA)-rGO | 0.07–0.7 | 0.04 | 14 | This study |
| Graphene oxide/Au nanoparticles/multiwalled carbon nanotubes nanocomposite film | 5.0–1700 | 0.2 | 14 | [21] |
| Graphene-AuNP composites | 0.05–1000 | 0.01 | 30 | [19] |
| PNM modified glass carbon electrodes | 0.11–1.88 | 0.1 | 30 | [7] |
| Graphene nanosheet modified electrode by CS-DMF | 0.3–55 | 0.18 | - | [20] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raja Jamaluddin, R.Z.A.; Yook Heng, L.; Tan, L.L.; Chong, K.F. Electrochemical Biosensor for Nitrite Based on Polyacrylic-Graphene Composite Film with Covalently Immobilized Hemoglobin. Sensors 2018, 18, 1343. https://doi.org/10.3390/s18051343
Raja Jamaluddin RZA, Yook Heng L, Tan LL, Chong KF. Electrochemical Biosensor for Nitrite Based on Polyacrylic-Graphene Composite Film with Covalently Immobilized Hemoglobin. Sensors. 2018; 18(5):1343. https://doi.org/10.3390/s18051343
Chicago/Turabian StyleRaja Jamaluddin, Raja Zaidatul Akhmar, Lee Yook Heng, Ling Ling Tan, and Kwok Feng Chong. 2018. "Electrochemical Biosensor for Nitrite Based on Polyacrylic-Graphene Composite Film with Covalently Immobilized Hemoglobin" Sensors 18, no. 5: 1343. https://doi.org/10.3390/s18051343
APA StyleRaja Jamaluddin, R. Z. A., Yook Heng, L., Tan, L. L., & Chong, K. F. (2018). Electrochemical Biosensor for Nitrite Based on Polyacrylic-Graphene Composite Film with Covalently Immobilized Hemoglobin. Sensors, 18(5), 1343. https://doi.org/10.3390/s18051343




