Novel Competitive Fluorescence Sensing Platform for L-carnitine Based on Cationic Pillar[5]Arene Modified Gold Nanoparticles
Abstract
1. Introduction
2. Experiment Section
2.1. Reagents and Apparatus
2.2. Apparatus and Instruments
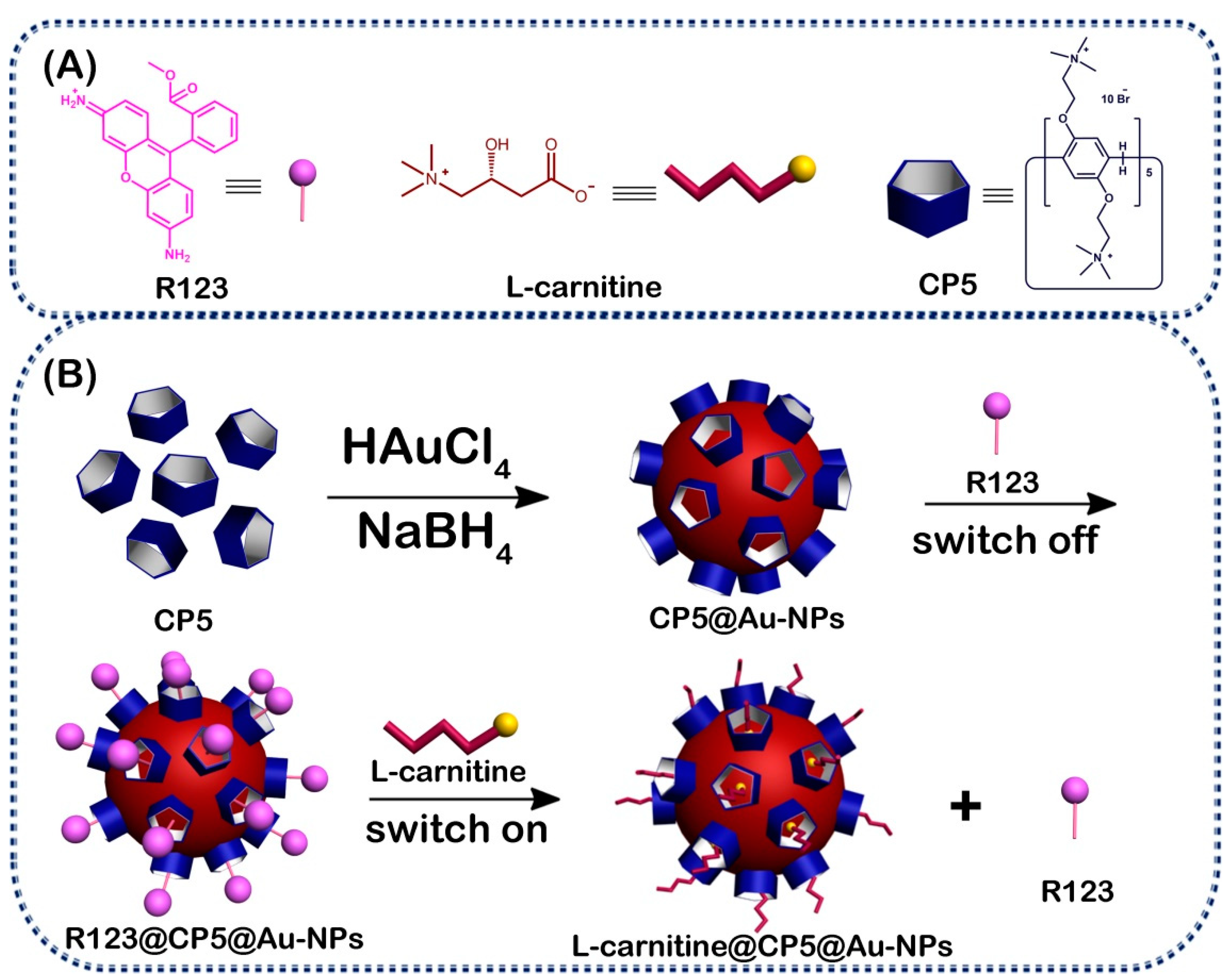
2.3. Synthesis of the CP5@Au-NPs
2.4. Experiments for Titration L-carnitine
3. Results and Discussion
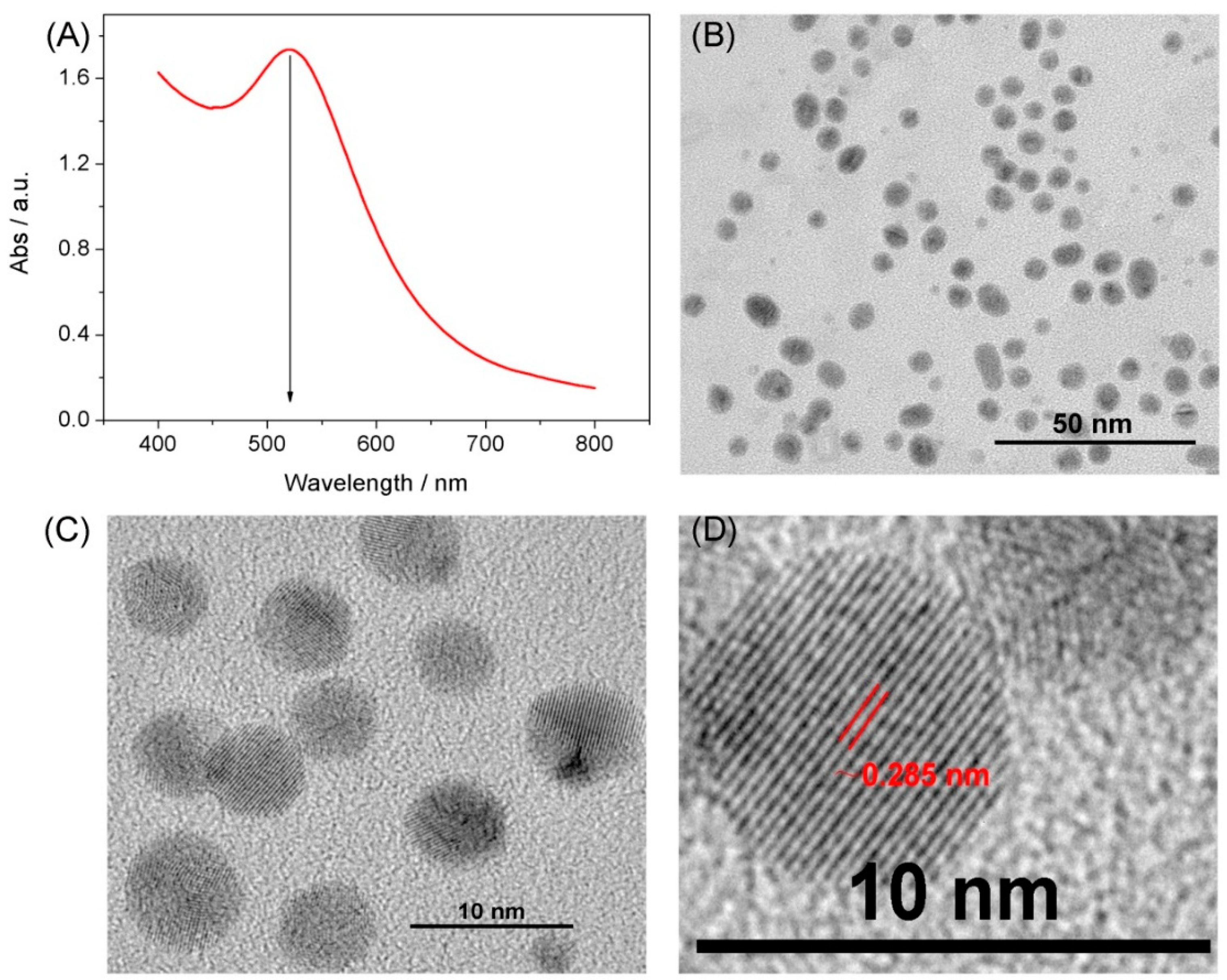
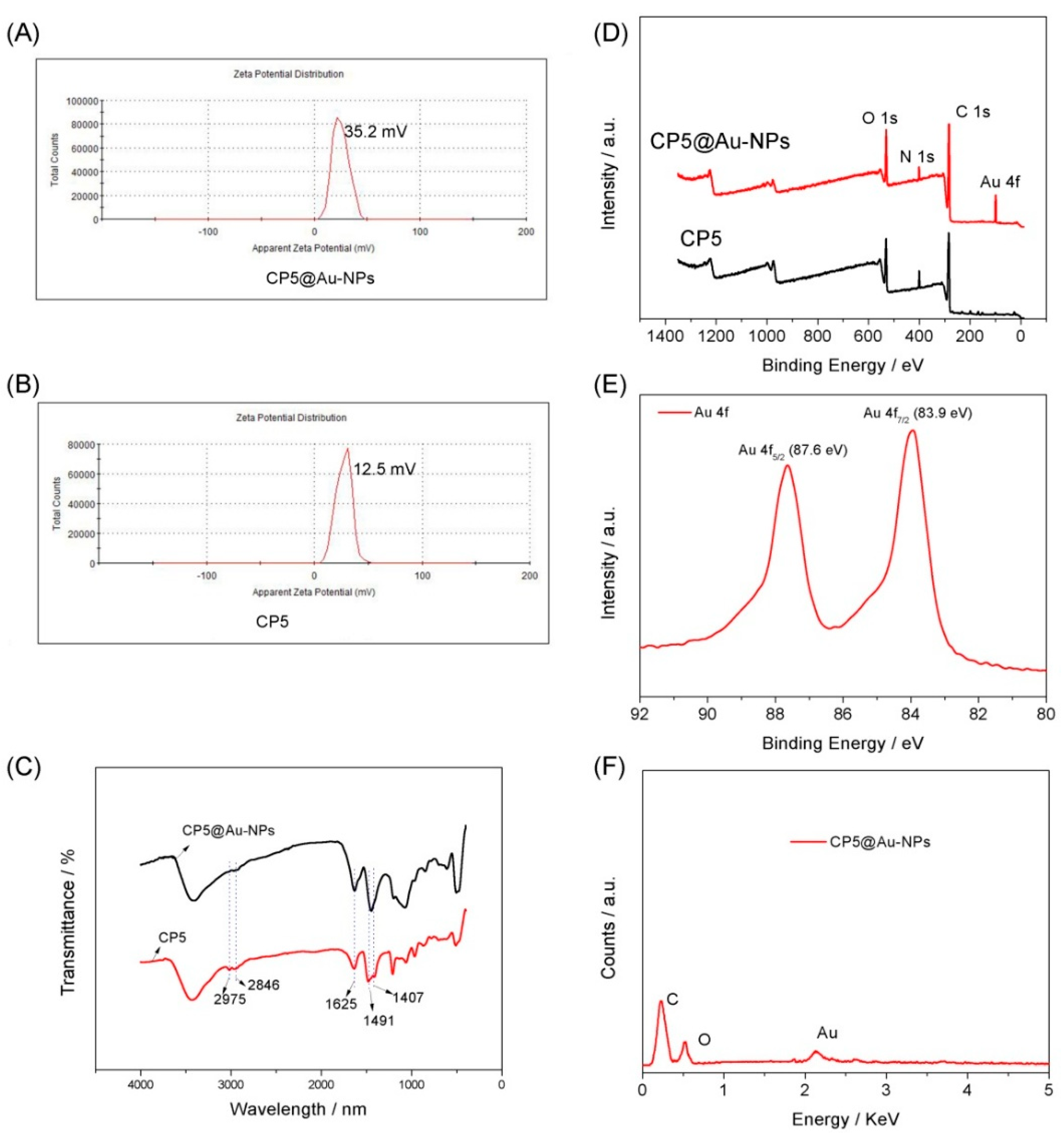
3.1. Characterization of the CP5@Au-NPs
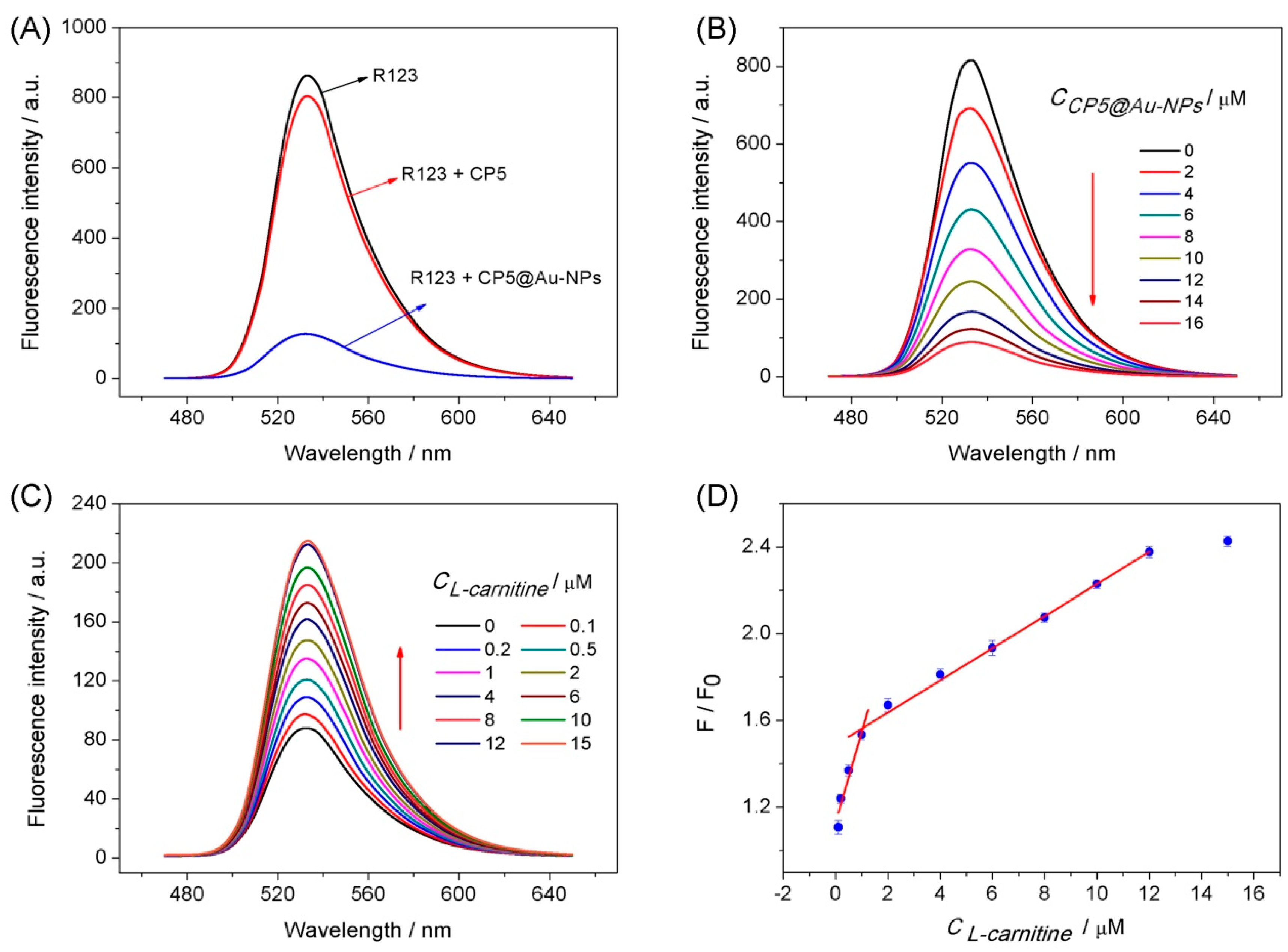
3.2. Fluorescence Spectra Analysis
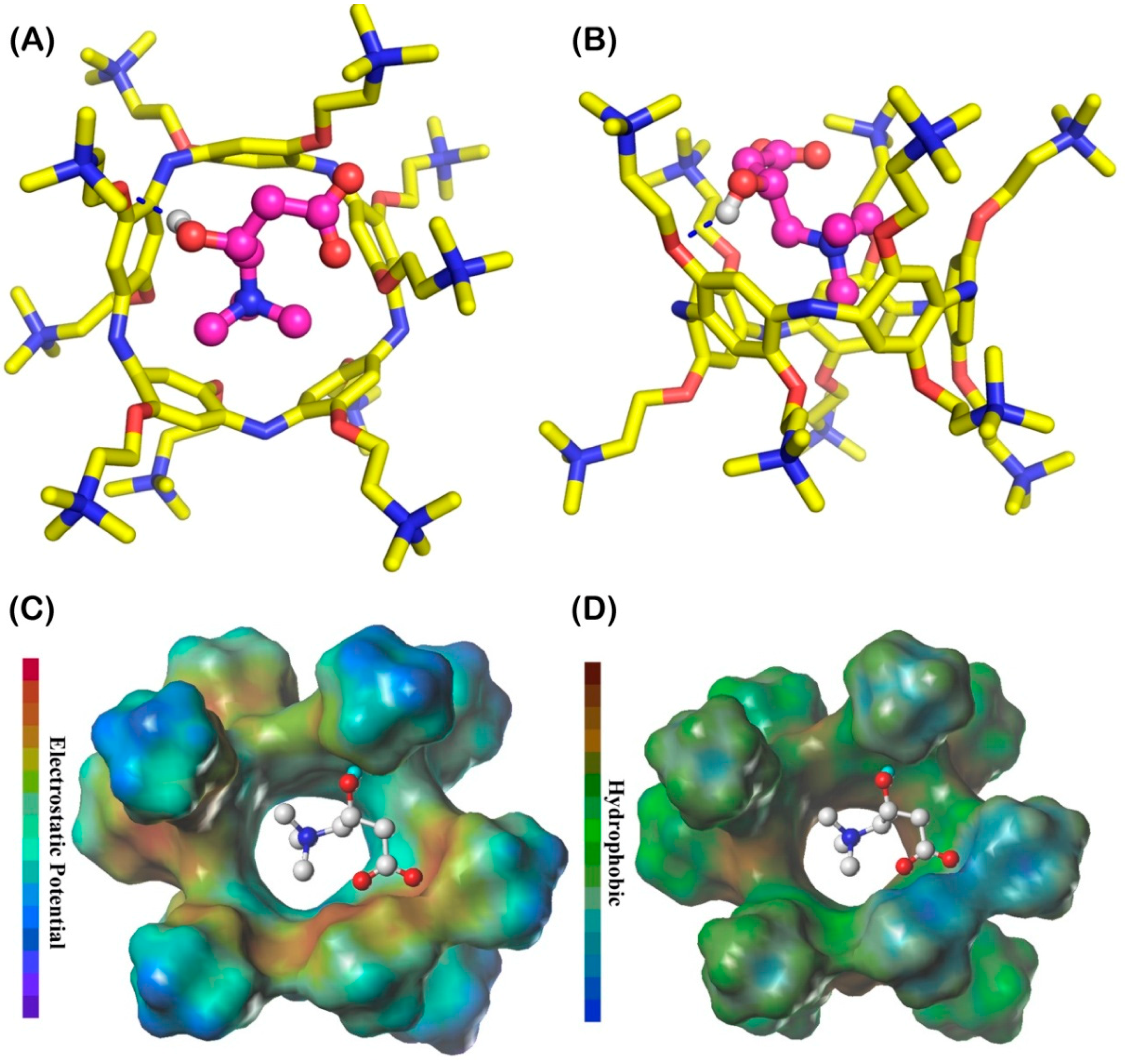
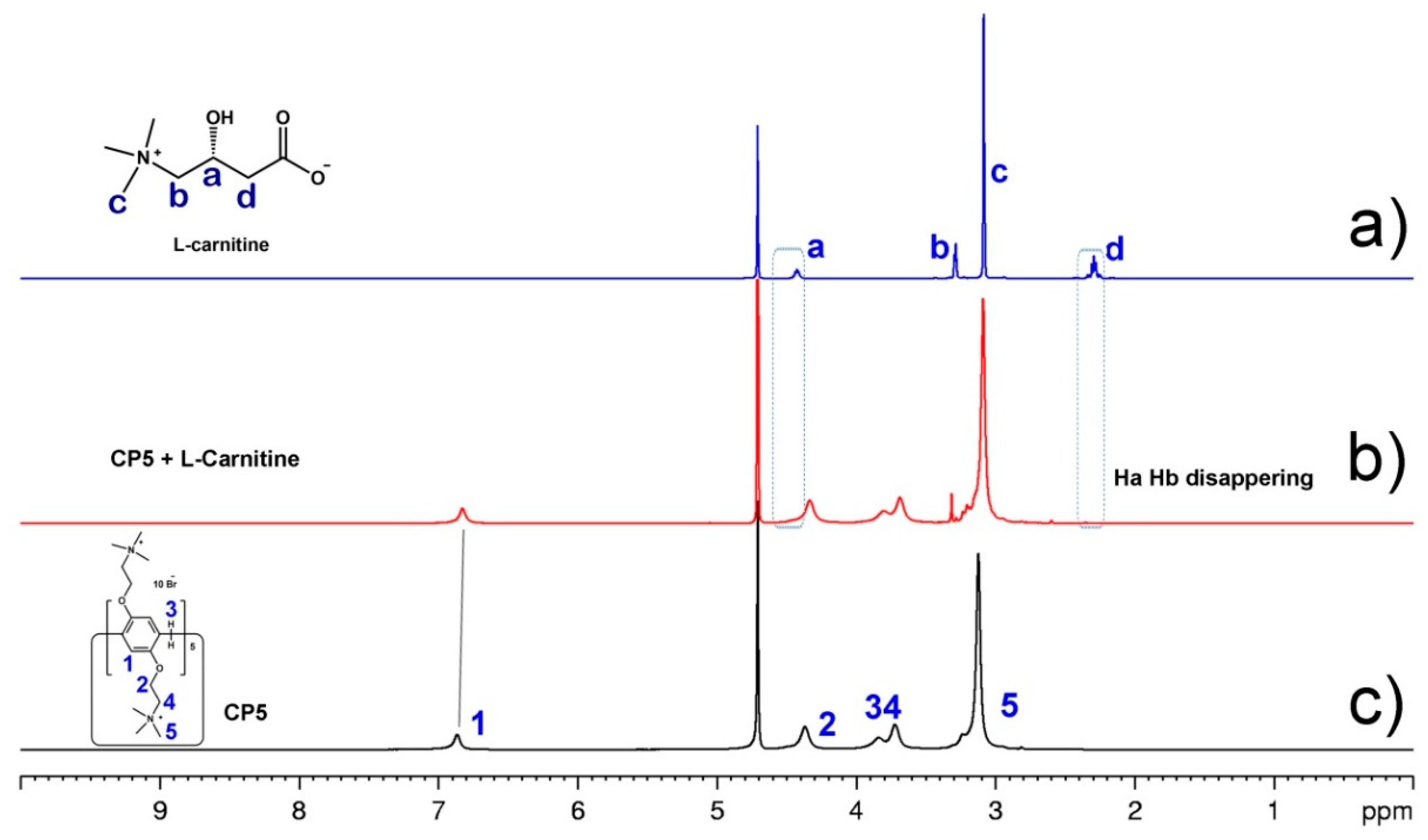
3.3. The Analysis of Host–Guest Recognition
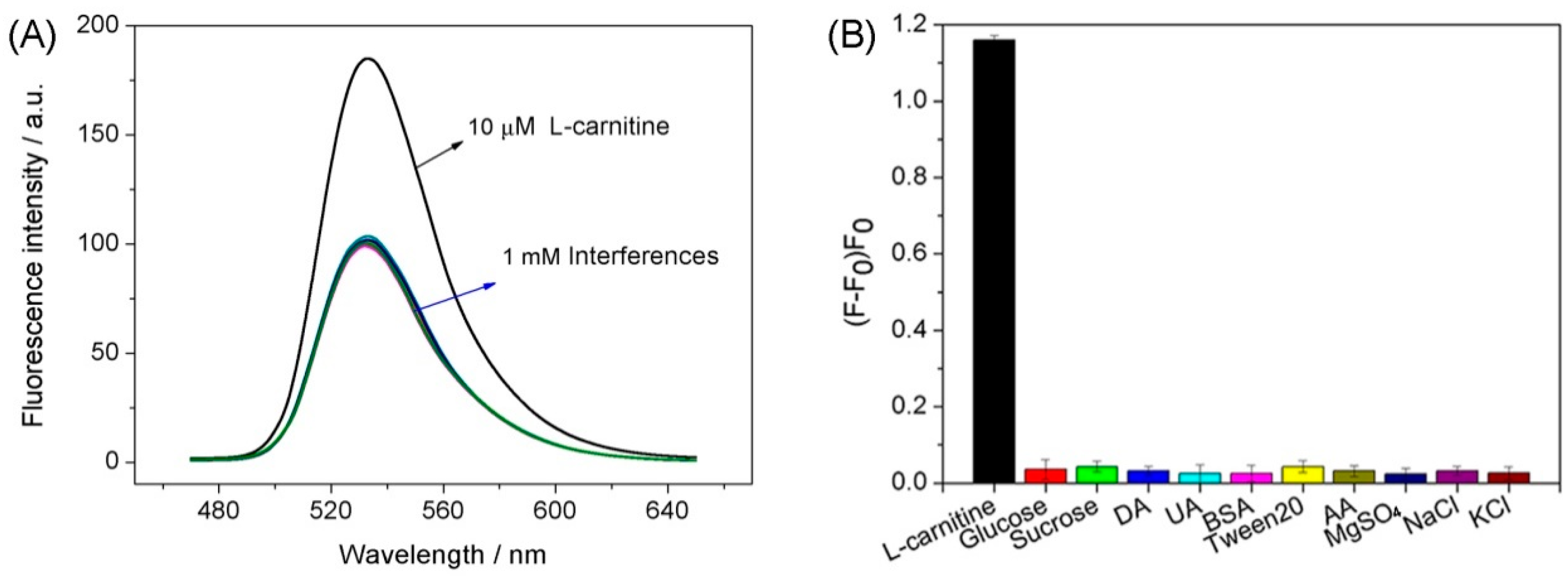
3.4. Selectivity and Practical Samples Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Siwy, Z.; Trofin, L.; Kohli, P.; Baker, L.A. Protein biosensors based on biofunctionalized conical gold nanotubes. J. Am. Chem. Soc. 2005, 127, 5000–5001. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Puebla, R.A.; Liz-Marzan, L.M. Nachweis kleiner anorganischer Moleküle durch oberflächenverstärkte Raman-Streuung. Angew. Chem. 2012, 124, 11376–11385. [Google Scholar] [CrossRef]
- Chen, M.S.; Goodman, D.W. Catalytically active gold on ordered Titania supports. Chem. Soc. Rev. 2008, 37, 1860–1870. [Google Scholar] [CrossRef] [PubMed]
- Hassenkam, T.; Moth-Poulsen, K.; Stuhr-Hansen, N.; Nørgaard, K.; Kabir, M.S.; Bjørnholm, T. Self-assembly and conductive properties of molecularly linked gold nanowires. Nano Lett. 2004, 4, 19–22. [Google Scholar] [CrossRef]
- Zheng, Y.B.; Kiraly, B.; Cheunkar, S.; Huang, T.J.; Weiss, P.S. Incident-angle-modulated molecular plasmonic switches: A case of weak exciton-plasmon coupling. Nano Lett. 2011, 11, 2061–2065. [Google Scholar] [CrossRef] [PubMed]
- Ofir, Y.; Samanta, B.; Rotello, V.M. Polymer and biopolymer mediated self-assembly of gold nanoparticles. Chem. Soc. Rev. 2008, 37, 1814–1825. [Google Scholar] [CrossRef] [PubMed]
- Klajn, R.; Stoddart, J.F.; Grzybowski, B.A. Nanoparticles functionalised with reversible molecular and supramolecular switches. Chem. Soc. Rev. 2010, 39, 2203–2237. [Google Scholar] [CrossRef] [PubMed]
- Dalgarno, S.J.; Thallapally, P.K.; Barbour, L.J.; Atwood, J.L. Engineering void space in organic van der Waals crystals: Calixarenes lead the way. Chem. Soc. Rev. 2007, 36, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.-Y.; Rambo, B.M.; Karnas, E.; Lynch, V.M.; Sessler, J.L. A ‘Texas-sized’ molecular box that forms an anion-induced supramolecular necklace. Nat. Chem. 2010, 2, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Zhao, Y. Biomedical applications of supramolecular systems based on host–guest interactions. Chem. Rev. 2015, 115, 7794–7839. [Google Scholar] [CrossRef] [PubMed]
- Shurpik, D.N.; Yakimova, L.S.; Gorbachuk, V.V.; Sevastyanov, D.A.; Padnya, P.L.; Bazanova, O.B.; Rizvanov, I.K.; Stoikov, I.I. Hybrid multicyclophanes based on thiacalix[4]arene and pillar[5]arene: Synthesis and influence on the formation of polyaniline. Org. Chem. Front. 2018, 5, 2780–2786. [Google Scholar] [CrossRef]
- Crini, G. Review: A history of cyclodextrins. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef] [PubMed]
- Cragg, P.J.; Sharma, K. Pillar[5]arenes: Fascinating cyclophanes with a bright future. Chem. Soc. Rev. 2012, 41, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Ogoshi, T.; Kanai, S.; Fujinami, S.; Yamagishi, T.; Nakamoto, Y. para-Bridged symmetrical pillar[5]arenes: Their Lewis acid catalyzed synthesis and host–guest property. J. Am. Chem. Soc. 2008, 130, 5022–5023. [Google Scholar] [CrossRef] [PubMed]
- Yakimova, L.S.; Shurpik, D.N.; Guralnik, E.G.; Evtugyn, V.G.; Osin, Y.N.; Stoikov, I.I. Fluorescein-loaded solid lipid nanoparticles based on monoamine pillar[5]arene: Synthesis and interaction with DNA. ChemNanoMat 2018, 4, 919–923. [Google Scholar] [CrossRef]
- Yakimova, L.S.; Shurpik, D.N.; Stoikov, I.I. Amide-functionalized pillar[5]arenes as a novel class of macrocyclic receptors for the sensing of H2PO4− anion. Chem. Commun. 2016, 52, 12462–12465. [Google Scholar] [CrossRef] [PubMed]
- Yakimova, L.S.; Shurpik, D.N.; Gilmanova, L.H.; Makhmutova, A.R.; Rakhimbekova, A.; Stoikov, I.I. Highly selective binding of methyl orange dye by cationic water-soluble pillar[5]arenes. Org. Biomol. Chem. 2016, 14, 4233–4238. [Google Scholar] [CrossRef] [PubMed]
- Nazarova, A.A.; Yakimova, L.S.; Klochkov, V.V.; Stoikov, I.I. Monoaminophosphorylated pillar[5]arenes as hosts for alkaneamines. New J. Chem. 2017, 41, 1820–1826. [Google Scholar] [CrossRef]
- Zhou, X.; Yang, L.; Tan, X.P.; Zhao, G.F.; Xie, X.G.; Du, G.B. A robust electrochemical immunosensor based on hydroxyl pillar[5]arene@ AuNPs@g-C3N4 hybrid nanomaterial for ultrasensitive detection of prostate specific antigen. Biosens. Bioelectron. 2018, 112, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Ogoshi, T.; Yamagishi, T.A.; Nakamoto, Y. Pillar-shaped macrocyclic hosts pillar[n]arenes: New key players for supramolecular chemistry. Chem. Rev. 2016, 116, 7937–8002. [Google Scholar] [CrossRef] [PubMed]
- Strutt, N.L.; Zhang, H.C.; Schneebeli, S.T.; Stoddart, J.F. Functionalizing pillar[n]arenes. Acc. Chem. Res. 2014, 47, 2631–2642. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Yang, Y.; Chi, X.; Zhang, Z.; Huang, F. Pillararenes, A new class of macrocycles for supramolecular chemistry. Acc. Chem. Res. 2012, 45, 1294–1308. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Pei, Y.X.; Pei, Z.C. Recent advances in pillar[n]arenes: synthesis and applications based on host-guest interactions. Chem. Commun. 2016, 52, 9316–9326. [Google Scholar] [CrossRef] [PubMed]
- Wei, P.; Yan, X.; Huang, F.H. Supramolecular polymers constructed by orthogonal self-assembly based on host-guest and metal-ligand interactions. Chem. Soc. Rev. 2015, 44, 815–832. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Yang, Y.-W.; Zhang, S.X.-A. Progress on the synthesis and host-guest chemistry of pillararenes. Chem. J. Chin. Univ. 2012, 33, 1–13. [Google Scholar] [CrossRef]
- Wang, K.; Tan, L.-L.; Chen, D.-X.; Song, N.; Xi, G.; Zhang, S.X.-A.; Li, C.; Yang, Y.-W. One-pot synthesis of pillar[n]arenes catalyzed by a minimum amount of TfOH and a solution-phase mechanistic study. Org. Biomol. Chem. 2012, 10, 9405–9409. [Google Scholar] [CrossRef] [PubMed]
- Montes-Garcia, V.; Perez-Juste, J.; Pastoriza-Santos, I.; Liz-Marzan, L.M. Metal nanoparticles and supramolecular macrocycles: A tale of synergy. Chem. Eur. J. 2014, 20, 10874–10883. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xue, M.; Chi, X.D.; Ma, Y.J.; He, J.M.; Abliz, Z.; Huang, F.H. A new water-soluble pillar[5]arene: Synthesis and application in the preparation of gold nanoparticles. Chem. Commun. 2012, 48, 6505–6507. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.H.; Song, X.N.; Bai, Y.B.; Yang, W.S.; Peng, X.G. Size control of gold nanocrystals in citrate reduction: the third role of citrate. J. Am. Chem. Soc. 2007, 129, 13939–13948. [Google Scholar] [CrossRef] [PubMed]
- Rowe, M.P.; Plass, K.E.; Kim, K.; Kurdak, Ç.; Zellers, E.T.; Matzger, A.J. Single-phase synthesis of functionalized gold nanoparticles. Chem. Mater. 2004, 16, 3513–3517. [Google Scholar] [CrossRef]
- Zheng, Y.B.; Payton, J.L.; Song, T.-B.; Pathem, B.K.; Zhao, Y.; Ma, H.; Yang, Y.; Jensen, L.; Jen, A.K.-Y.; Weiss, P.S. Surface-enhanced Raman spectroscopy to probe photoreaction pathways and kinetics of isolated reactants on surfaces: Flat versus curved substrates. Nano Lett. 2012, 12, 5362–5368. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, D.X.; Sun, Y.L.; Zheng, Y.B.; Tan, L.L.; Weiss, P.S.; Yang, Y.W. Viologen-mediated assembly of and sensing with arboxylatopillar[5]arene-modified gold nanoparticles. J. Am. Chem. Soc. 2013, 135, 1570–1576. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xue, M.; Zhang, Z.B.; Zhang, M.M.; Wang, Y.; Huang, F.H. Gold nanoparticles stabilized by an amphiphilic pillar[5]arene: Preparation, self-assembly into composite microtubes in water and application in green catalysis. Chem. Sci. 2013, 4, 3667–3672. [Google Scholar] [CrossRef]
- Montes-Garcia, V.; Fernandez-Lopez, C.; Gomez, B.; Perez-Juste, I.; Garcia-Rio, L.; Liz-Marzan, L.M.; Perez-Juste, J.; Pastoriza-Santos, I. Pillar[5]arene-mediated synthesis of gold nanoparticles: Size control and sensing capabilities. Chem. Eur. J. 2014, 20, 8404–8409. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.F.; Ran, X.; Zhou, X.; Tan, X.P.; Lei, H.; Xie, X.G.; Yang, L.; Du, G.B. Green synthesis of hydroxylatopillar[5]arene-modified gold nanoparticles and their self-assembly, sensing, and catalysis application. ACS Sustain. Chem. Eng. 2018, 6, 3938–3947. [Google Scholar] [CrossRef]
- Seline, K.G.; Johein, H. The determination of l-carnitine in several food samples. Food Chem. 2007, 105, 793–804. [Google Scholar] [CrossRef]
- Cao, Q.R.; Ren, S.; Park, M.J.; Choi, Y.J.; Lee, B.J. Determination of highly soluble l-carnitine in biological samples by reverse phase high performance liquid chromatography with fluorescent derivatization. Arch. Pharm. Res. 2007, 30, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Wang, X.; Wang, N.N.; Zhu, Y. A rapid ion chromatography column-switching method for online sample pretreatment and determination of l-carnitine, choline and mineral ions in milk and powdered infant formula. RSC Adv. 2017, 7, 5920–5927. [Google Scholar] [CrossRef]
- Kiessig, S.; Vogt, C. Separation of carnitine and acylcarnitines by capillary electrophoresis. J. Chromatogr. A 1997, 781, 475–479. [Google Scholar] [CrossRef]
- Kong, Y.; Yang, G.F.; Chen, S.M.; Hou, Z.W.; Du, X.M.; Li, H.; Kong, L.H. Rapid and sensitive determination of l-carnitine and acetyl-L-carnitine in liquid milk samples with capillary zone electrophoresis using indirect UV detection. Food Anal. Methods 2018, 11, 170–177. [Google Scholar] [CrossRef]
- Vais, R.D.; Yadegari, H.; Sattarahmady, N.; Helia, H. An anodized nanostructure of Ni/Cu alloy synthesized in ethaline for electrocatalytic oxidation and amperometric determination of l-carnitine. J. Electroanal. Chem. 2018, 815, 134–142. [Google Scholar] [CrossRef]
- Li, H.B.; Zhang, Y.; Wang, X.Q. l-Carnitine capped quantum dots as luminescent probes for cadmium ions. Sens. Actuators B 2007, 127, 593–597. [Google Scholar] [CrossRef]
- Wang, M.H.; Du, J.A.; Mani, V.; Wu, Y.C.; Lin, Y.J.; Chia, Y.M.; Huang, S.T. A rapid fluorescence detecting platform: Applicable to sense carnitine and chloramphenicol in food samples. RSC Adv. 2014, 4, 64112–64118. [Google Scholar] [CrossRef]
- Mao, X.W.; Tian, D.M.; Li, H.B. p-Sulfonated calix[6]arene modified graphene as a ‘turn on’ fluorescent probe for l-carnitine in living cells. Chem. Commun. 2012, 48, 4851–4853. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.F.; Lu, H.Z. Ratiometric fluorescence and mesoporous structure dual signal amplification for sensitive and selective detection of TNT based on MIP@QD fluorescence sensors. Chem. Commun. 2015, 51, 3200–3203. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.; Naugolny, A.; Feldman, M.; Herzog, I.M.; Fridman, M.; Cohen, Y. Cationic Pillararenes potently inhibit biofilm formation without affecting bacterial growth and viability. J. Am. Chem. Soc. 2016, 138, 754–757. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.J.; Ji, X.F.; Xiang, F.; Chi, X.D.; Han, C.Y.; He, J.M.; Abliz, Z.; Chen, W.X.; Huang, F.H. A cationic water-soluble pillar[5]arene: Synthesis and host–guest complexation with sodium 1-octanesulfonate. Chem. Commun. 2011, 47, 12340–12342. [Google Scholar] [CrossRef] [PubMed]
- Pande, S.; Ghosh, S.K.; Praharaj, S.; Panigrahi, S.; Basu, S.; Jana, S.; Pal, A.; Tsukuda, T.; Pal, T. Synthesis of normal and inverted gold-silver core-shell architectures in β-cyclodextrin and their applications in SERS. J. Phys. Chem. C 2007, 111, 10806–10813. [Google Scholar] [CrossRef]
- Kong, B.S.; Geng, J.; Jung, H.T. Layer-by-layer assembly of graphene and gold nanoparticles by vacuum filtration and spontaneous reduction of gold ions. Chem. Commun. 2009, 0, 2174–2176. [Google Scholar] [CrossRef] [PubMed]
- Shan, C.; Yang, H.; Han, D.; Zhang, Q.; Ivaska, A.; Niu, L. Graphene/AuNPs/chitosan nanocomposites film for glucose biosensing. Biosens. Bioelectron. 2010, 25, 1070–1074. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Huang, Y.C.; Zhu, H.; Zhu, Q.Q.; Xia, Y.S. Three-in-one: Sensing, self-assembly, and cascade catalysis of cyclodextrin modified gold nanoparticles. J. Am. Chem. Soc. 2016, 138, 16645–16654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Liu, Y.; Tong, L.; Xu, K.; Zhuo, L.; Tang, B. A novel assembly of Au NPs–β-CDs–FL for the fluorescent probing of cholesterol and its application in blood serum. Analyst 2008, 133, 1176–1181. [Google Scholar] [CrossRef] [PubMed]
- Mondal, A.; Jana, N.R. Fluorescent detection of cholesterol using β-cyclodextrin functionalized grapheme. Chem. Commun. 2012, 48, 7316–7318. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.M.; Huey, R.; Lindstrom, W.; Sanner, M.F.; Takada, Y.; Olson, A.J. AutoDock4 and AutoDockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- Ogoshi, T.; Takashima, S.; Yamagishi, T.A. Molecular recognition with microporous multi-layer films prepared by layer-by-layer assembly of pillar[5]arenes. J. Am. Chem. Soc. 2015, 137, 10962–10964. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Yang, L.; Li, Y.C.; Ran, X.; Ye, H.Z.; Zhao, G.F.; Zhang, Y.Q.; Li, F.; Li, C.P. A comparison study of macrocyclic hosts functionalized reduced graphene oxide for electrochemical recognition of tadalafil. Biosens. Bioelectron. 2017, 89, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xue, M.; Chen, J.; Zhang, M.; Huang, F. An amphiphilic pillar[5]arene: Synthesis, controllable self-assembly in water, and application in Calcein release and TNT adsorption. J. Am. Chem. Soc. 2012, 134, 15712–15715. [Google Scholar] [CrossRef] [PubMed]
| Sample | Added (µM) | Founded (µM) | RSD (%) | Recovery (%) |
|---|---|---|---|---|
| serum | 0 | 0.00 | - | - |
| 2 | 1.87 ± 0.04 | 2.1 | 93.5 | |
| 4 | 4.07 ± 0.17 | 4.1 | 101.7 | |
| 6 | 5.82 ± 0.22 | 3.8 | 97.0 | |
| milk | 0 | 0.00 | - | - |
| 2 | 1.91 ± 0.03 | 1.6 | 95.5 | |
| 4 | 3.97 ± 0.13 | 3.3 | 99.2 | |
| 6 | 6.02 ± 0.24 | 3.9 | 100.3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, X.; Yang, Y.; Luo, S.; Zhang, Z.; Zeng, W.; Zhang, T.; Su, F.; Zhou, L. Novel Competitive Fluorescence Sensing Platform for L-carnitine Based on Cationic Pillar[5]Arene Modified Gold Nanoparticles. Sensors 2018, 18, 3927. https://doi.org/10.3390/s18113927
Tan X, Yang Y, Luo S, Zhang Z, Zeng W, Zhang T, Su F, Zhou L. Novel Competitive Fluorescence Sensing Platform for L-carnitine Based on Cationic Pillar[5]Arene Modified Gold Nanoparticles. Sensors. 2018; 18(11):3927. https://doi.org/10.3390/s18113927
Chicago/Turabian StyleTan, Xiaoping, Yang Yang, Shasha Luo, Zhong Zhang, Wenjie Zeng, Tingying Zhang, Fawu Su, and Linzong Zhou. 2018. "Novel Competitive Fluorescence Sensing Platform for L-carnitine Based on Cationic Pillar[5]Arene Modified Gold Nanoparticles" Sensors 18, no. 11: 3927. https://doi.org/10.3390/s18113927
APA StyleTan, X., Yang, Y., Luo, S., Zhang, Z., Zeng, W., Zhang, T., Su, F., & Zhou, L. (2018). Novel Competitive Fluorescence Sensing Platform for L-carnitine Based on Cationic Pillar[5]Arene Modified Gold Nanoparticles. Sensors, 18(11), 3927. https://doi.org/10.3390/s18113927





