Detection of Chloroalkanes by Surface-Enhanced Raman Spectroscopy in Microfluidic Chips
Abstract
1. Introduction
2. Materials and Methods
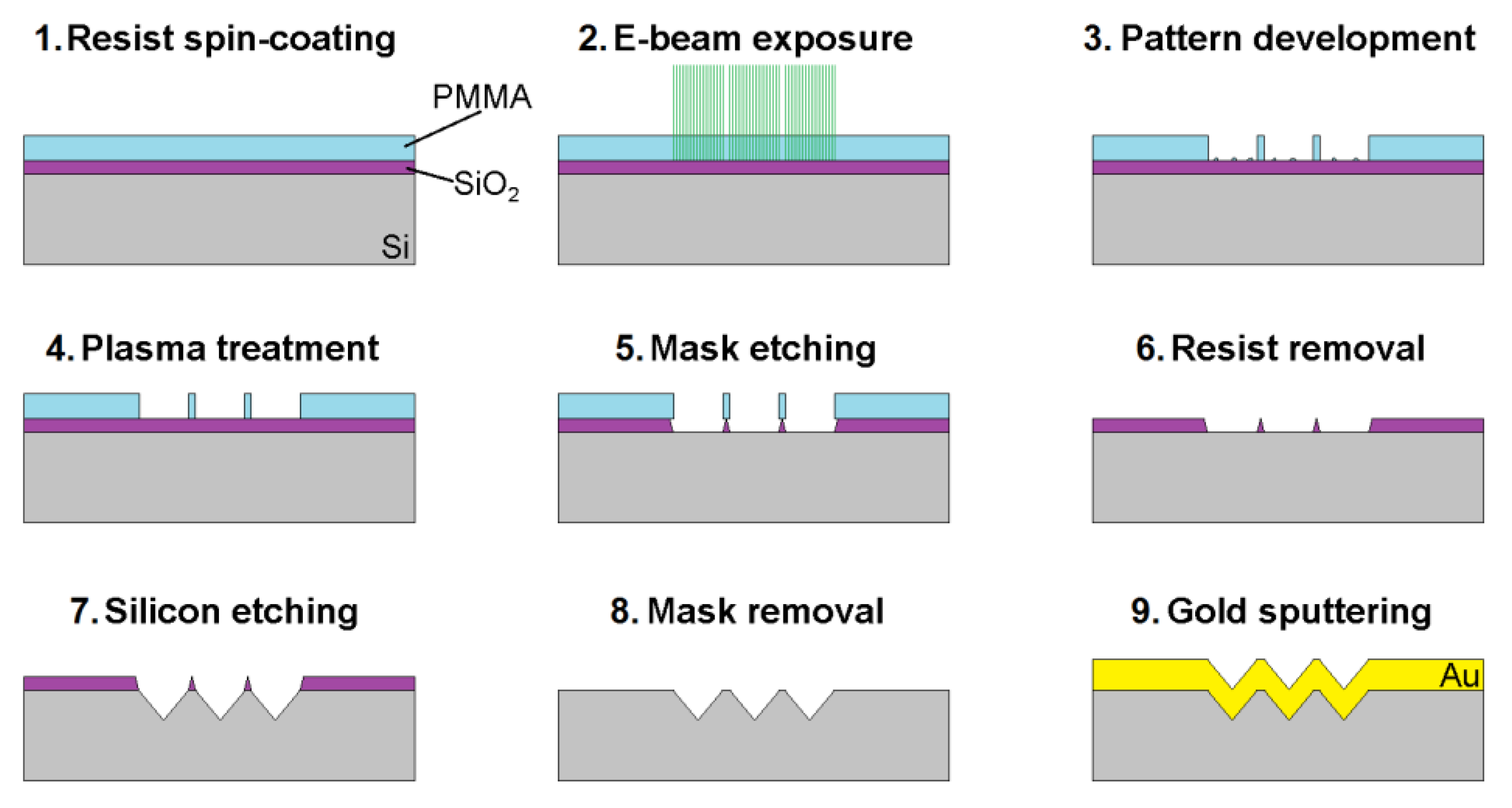
2.1. Fabrication of the SERS Substrate
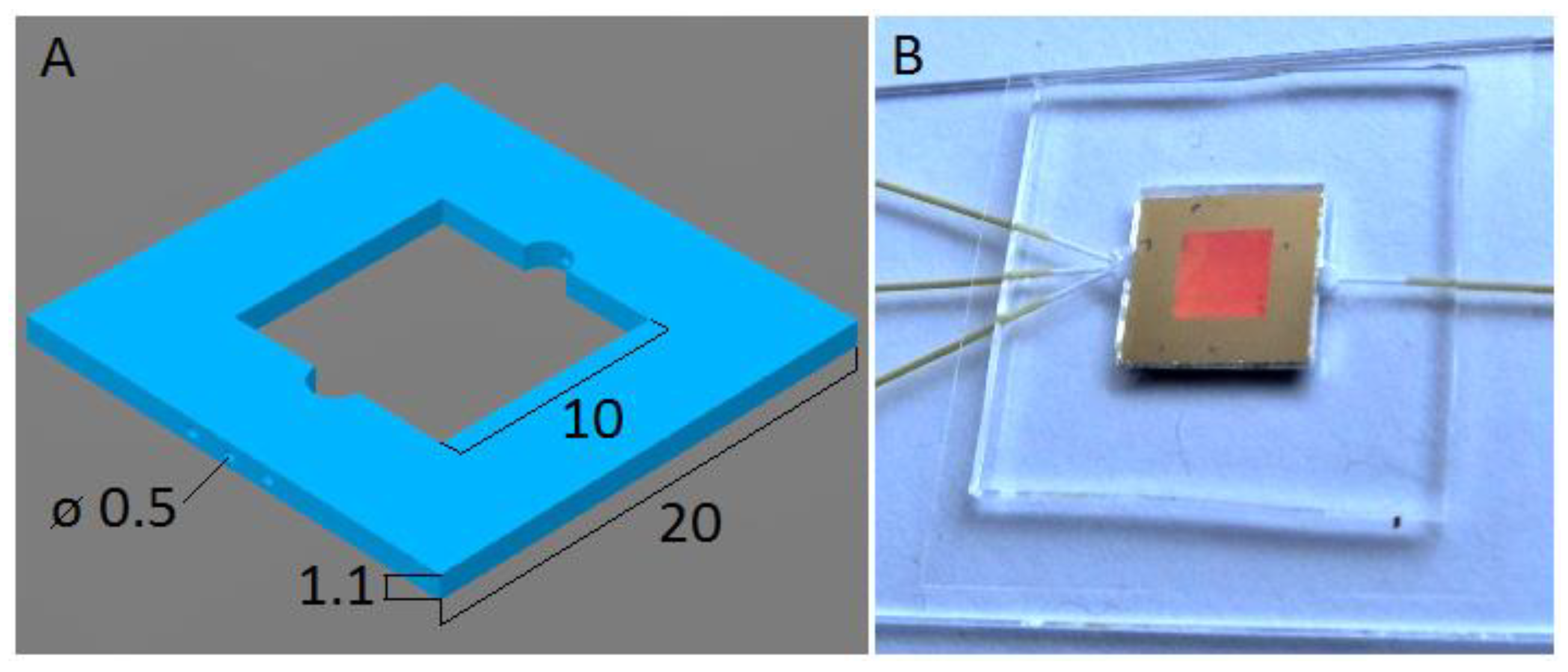
2.2. Fabrication of the Microfluidic Chip
2.3. Insertion of the Sers Substrate into the Microfluidic Chip and Completion of the Fabrication
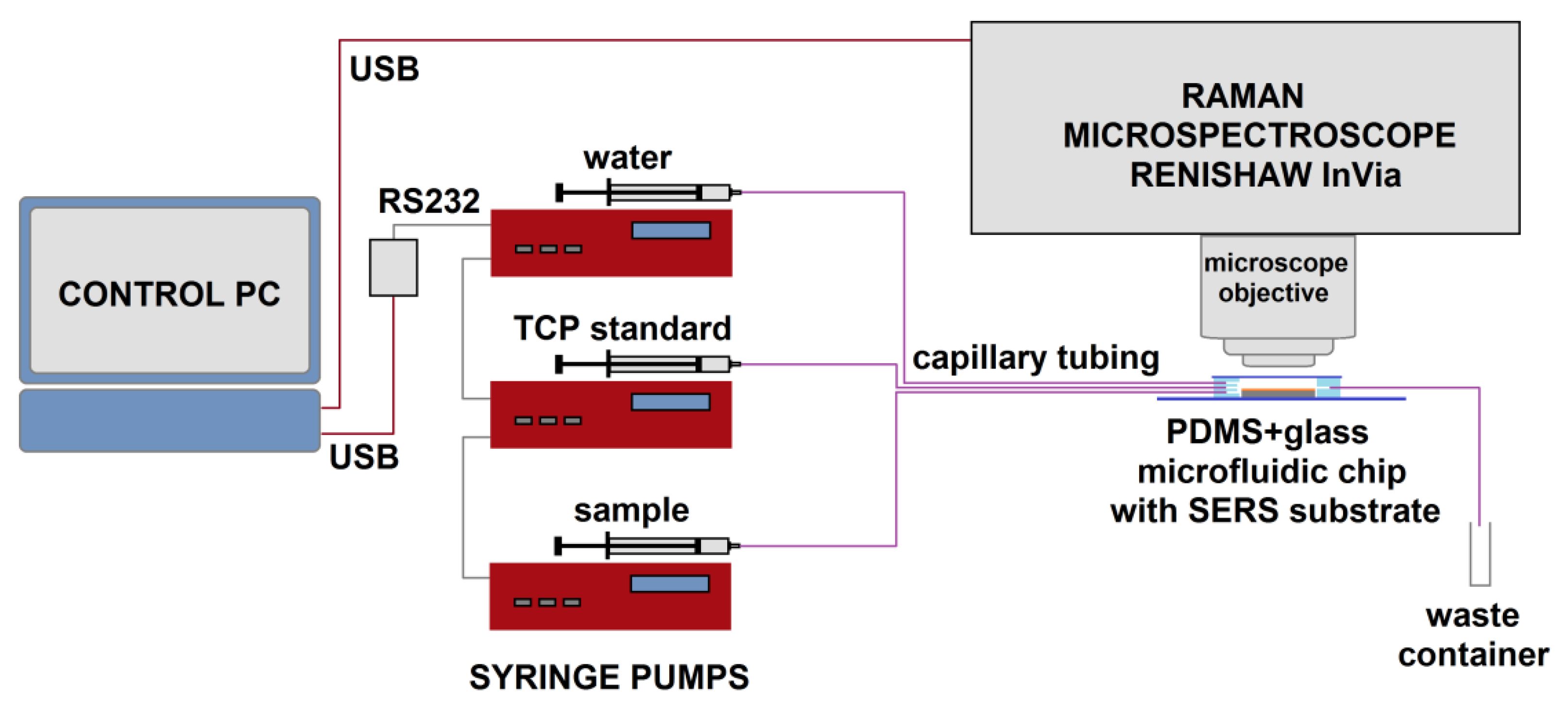
2.4. The Experimental Setup
2.5. Experimental Protocol
2.6. Spectra and Data Processing
2.7. Gas Chromatography-Mass Spectroscopy
3. Results
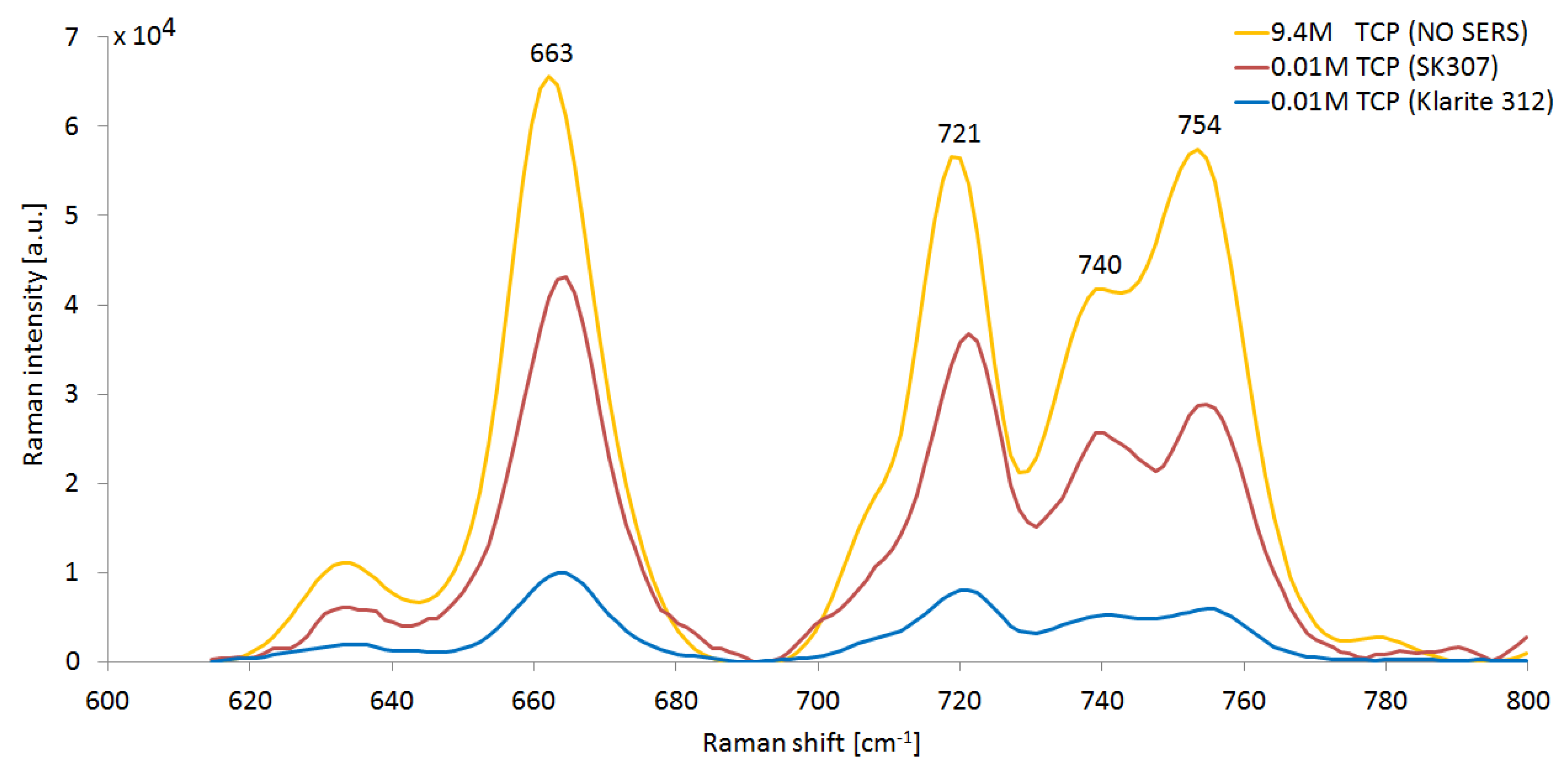
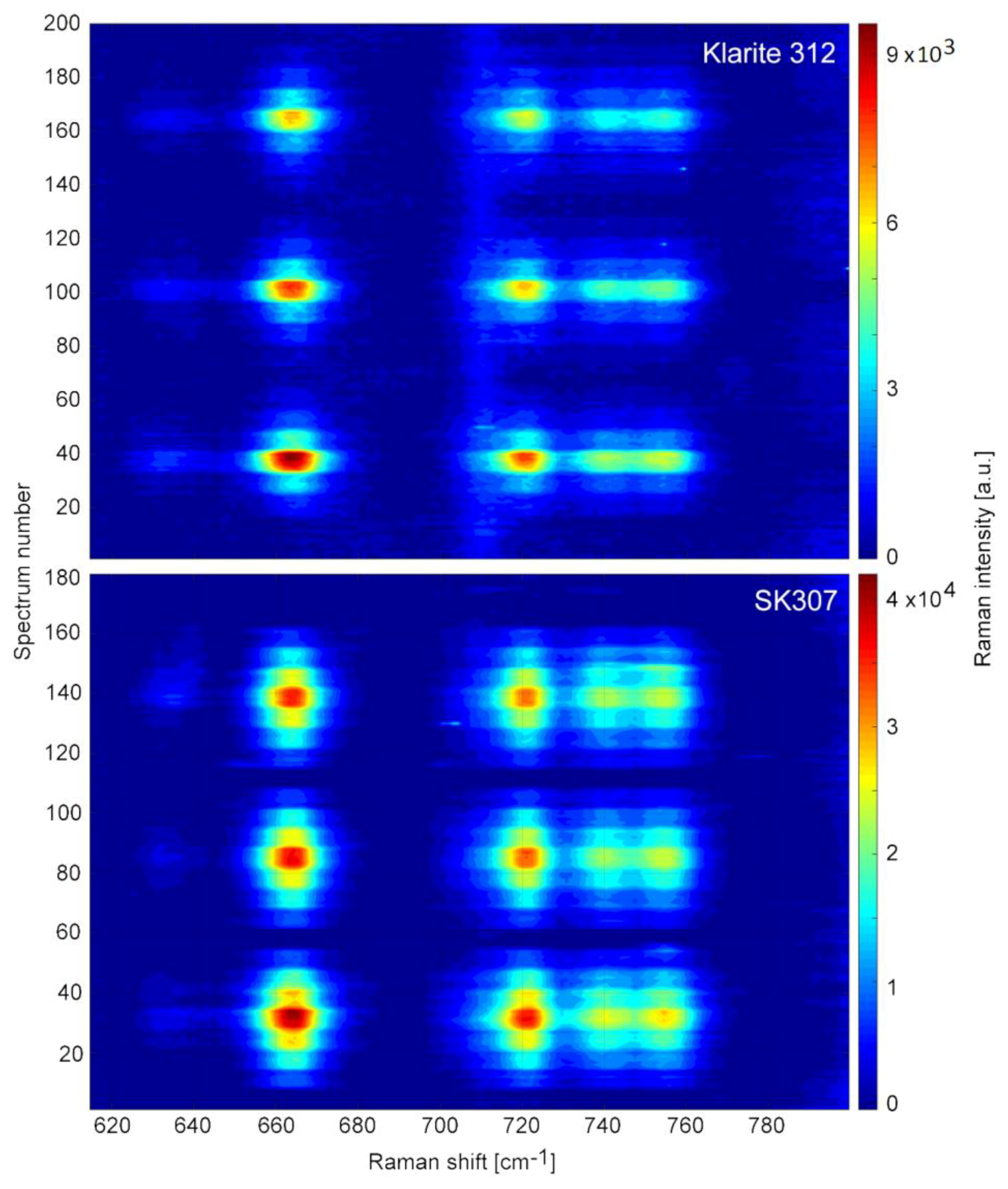
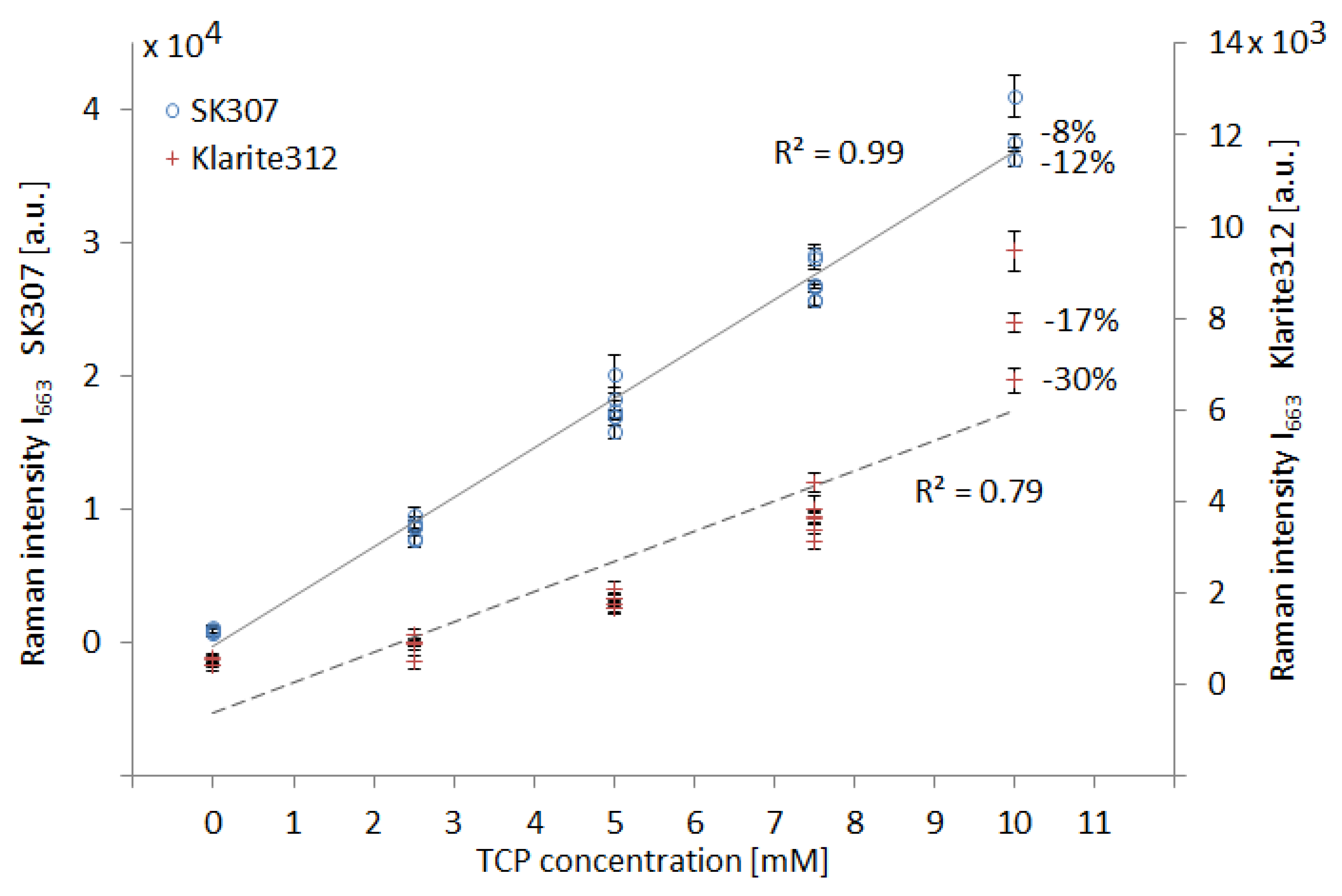
3.1. SERS Spectra of Tcp-Comparison of Sers Substrates SK307 with Klarite 312
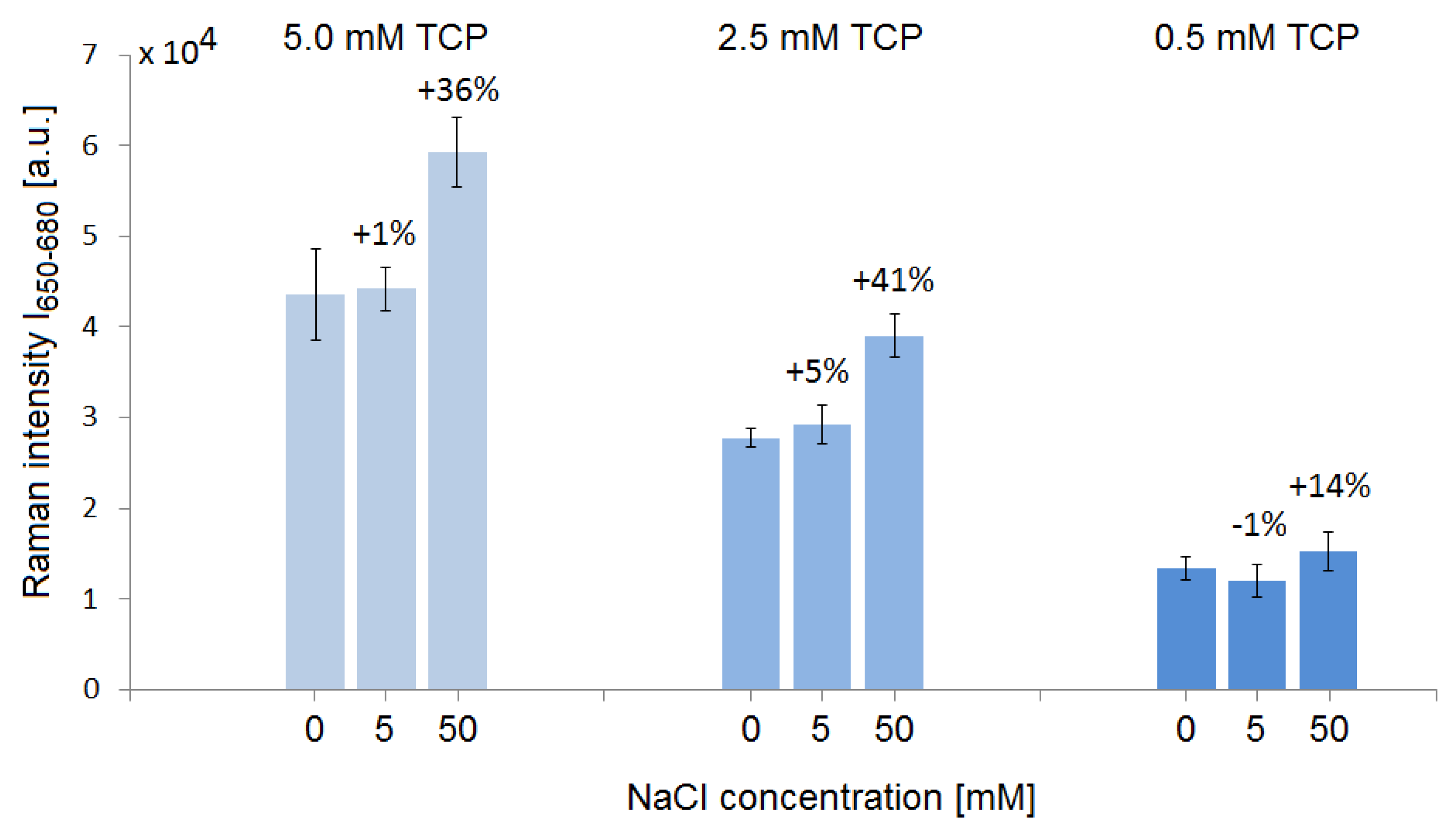
3.2. Influence of Dissolved Salt on the TCP SERS Signal from SK307
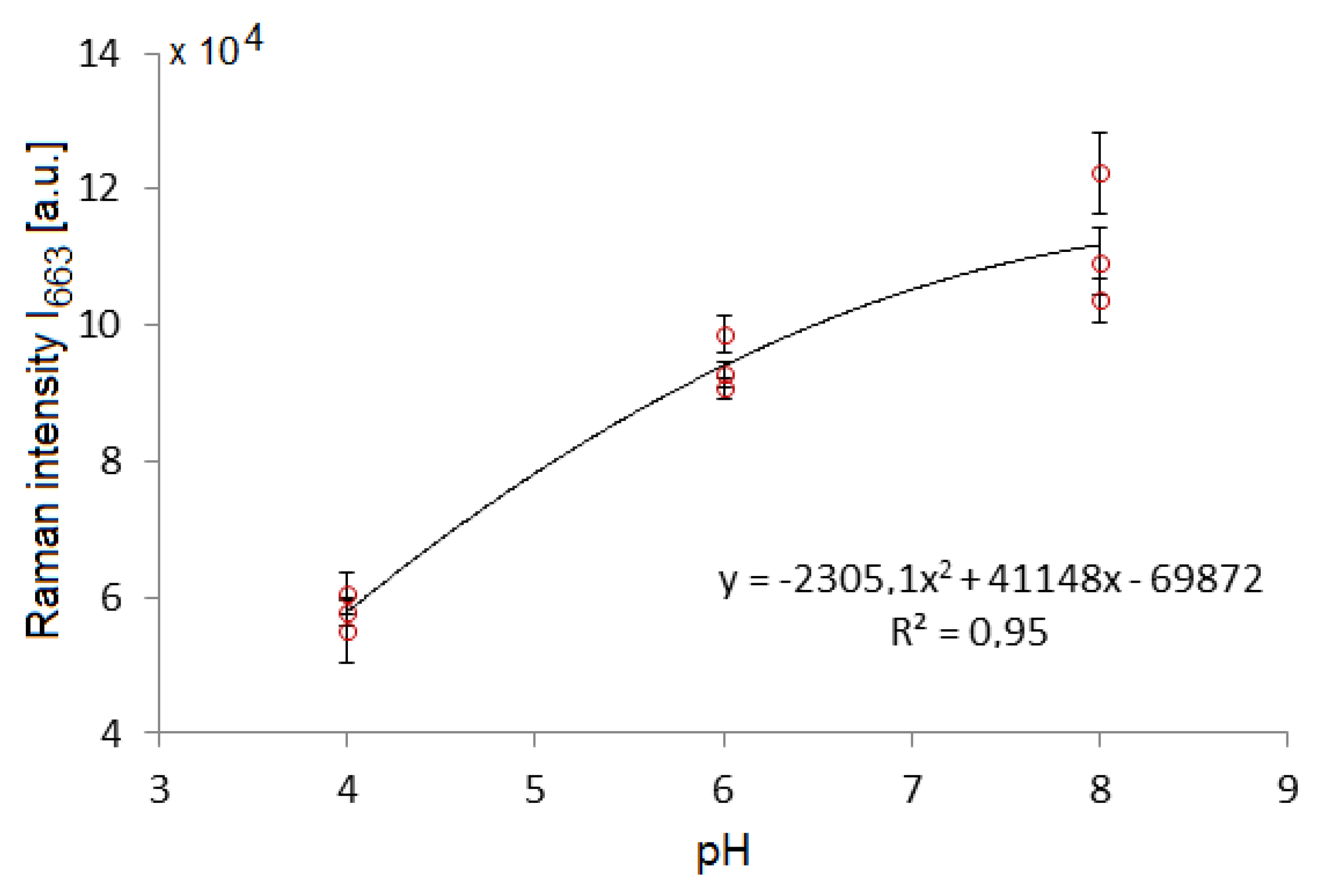
3.3. Influence of pH on the TCP SERS Signal from SK307
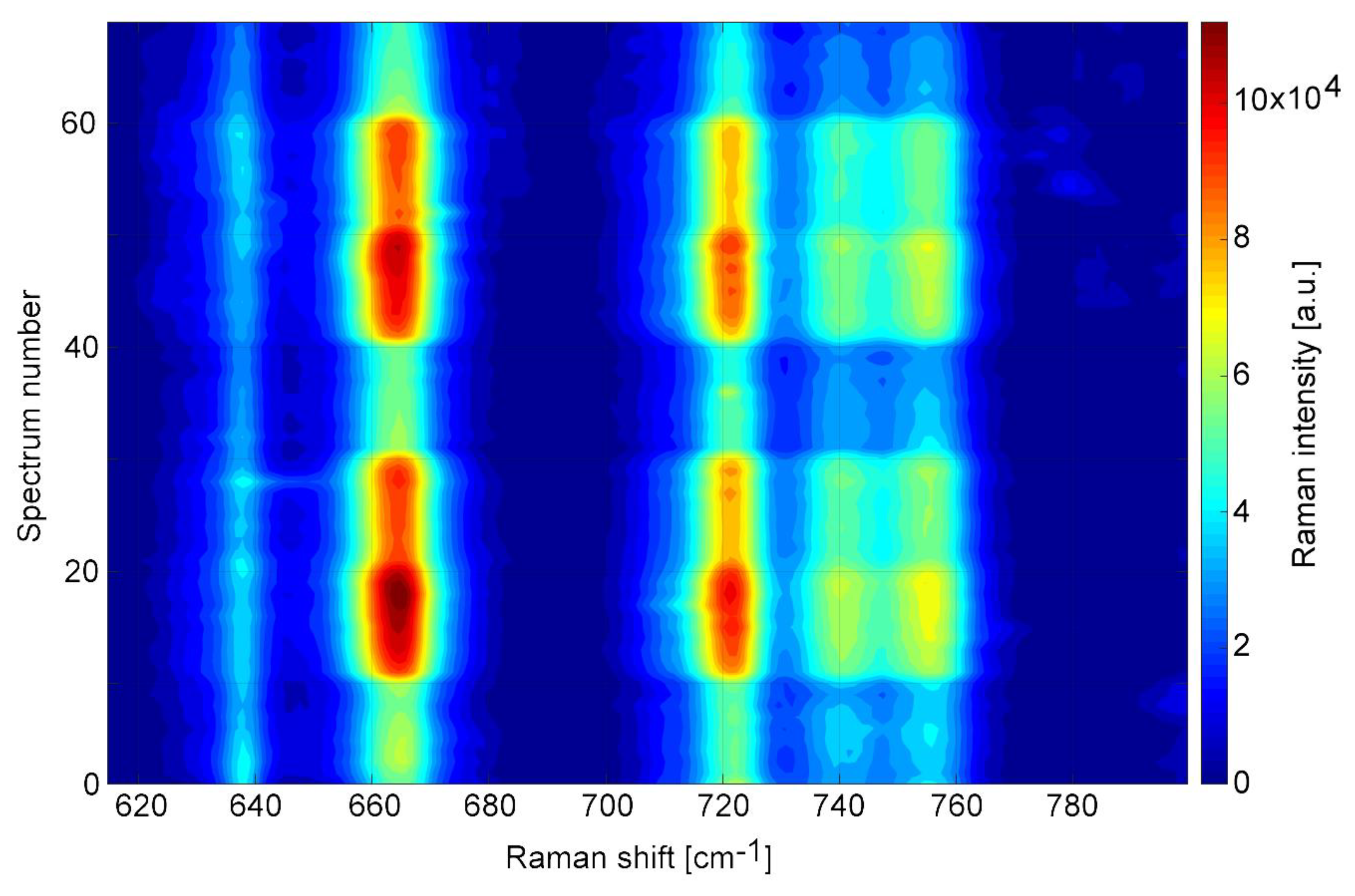
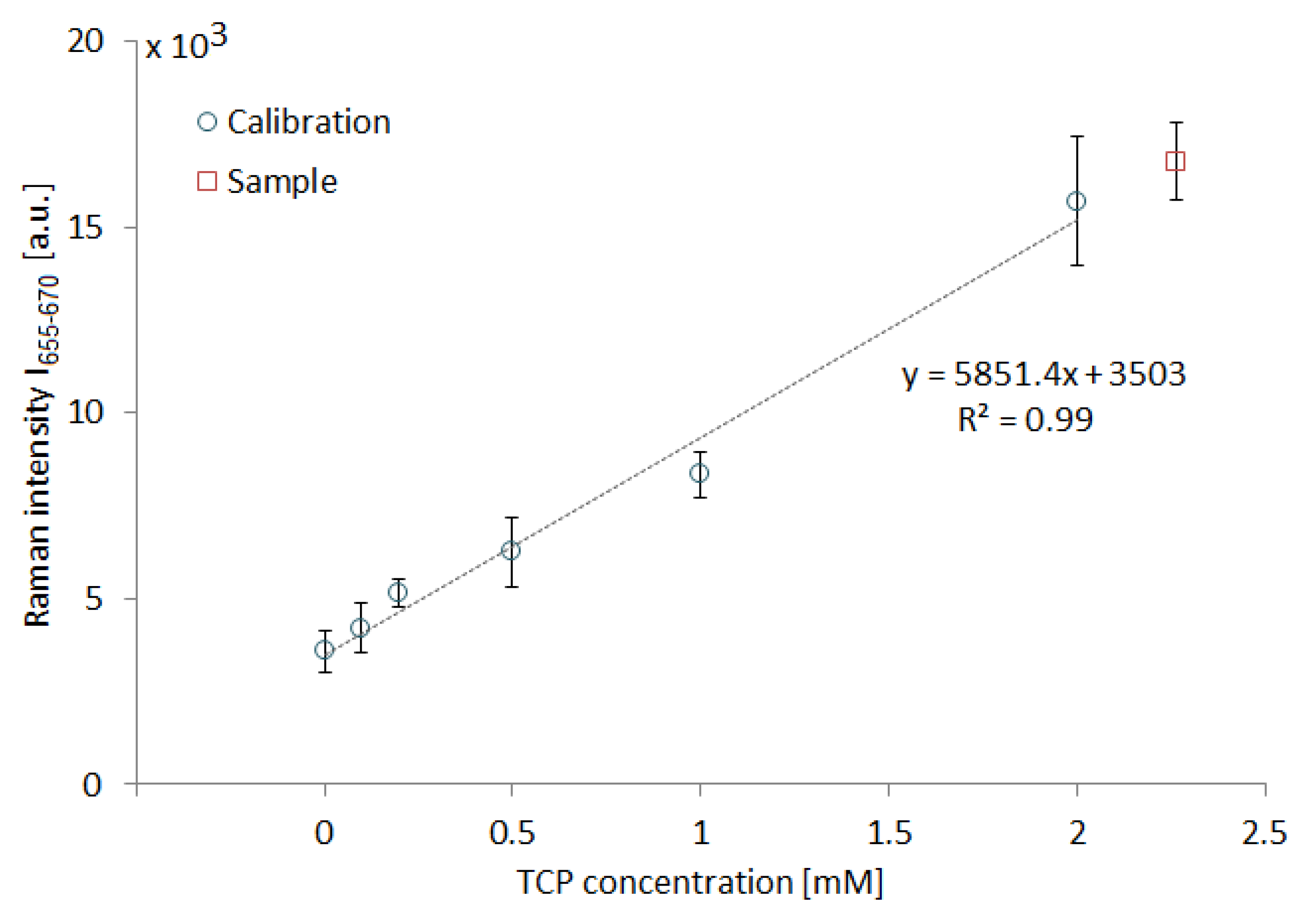
3.4. Measurement of Submillimolar Concentrations of TCP and a Simulated Sampling
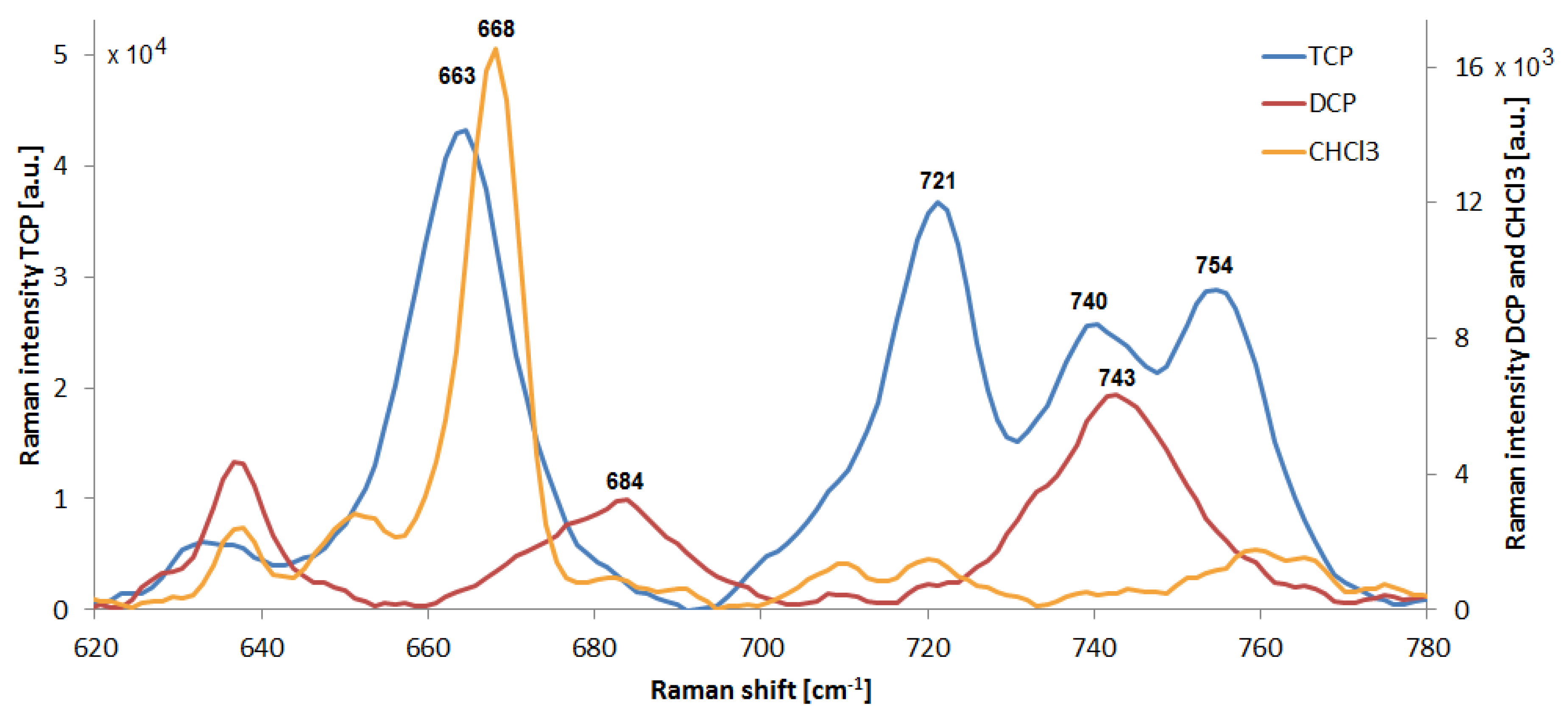
3.5. Other Analytes
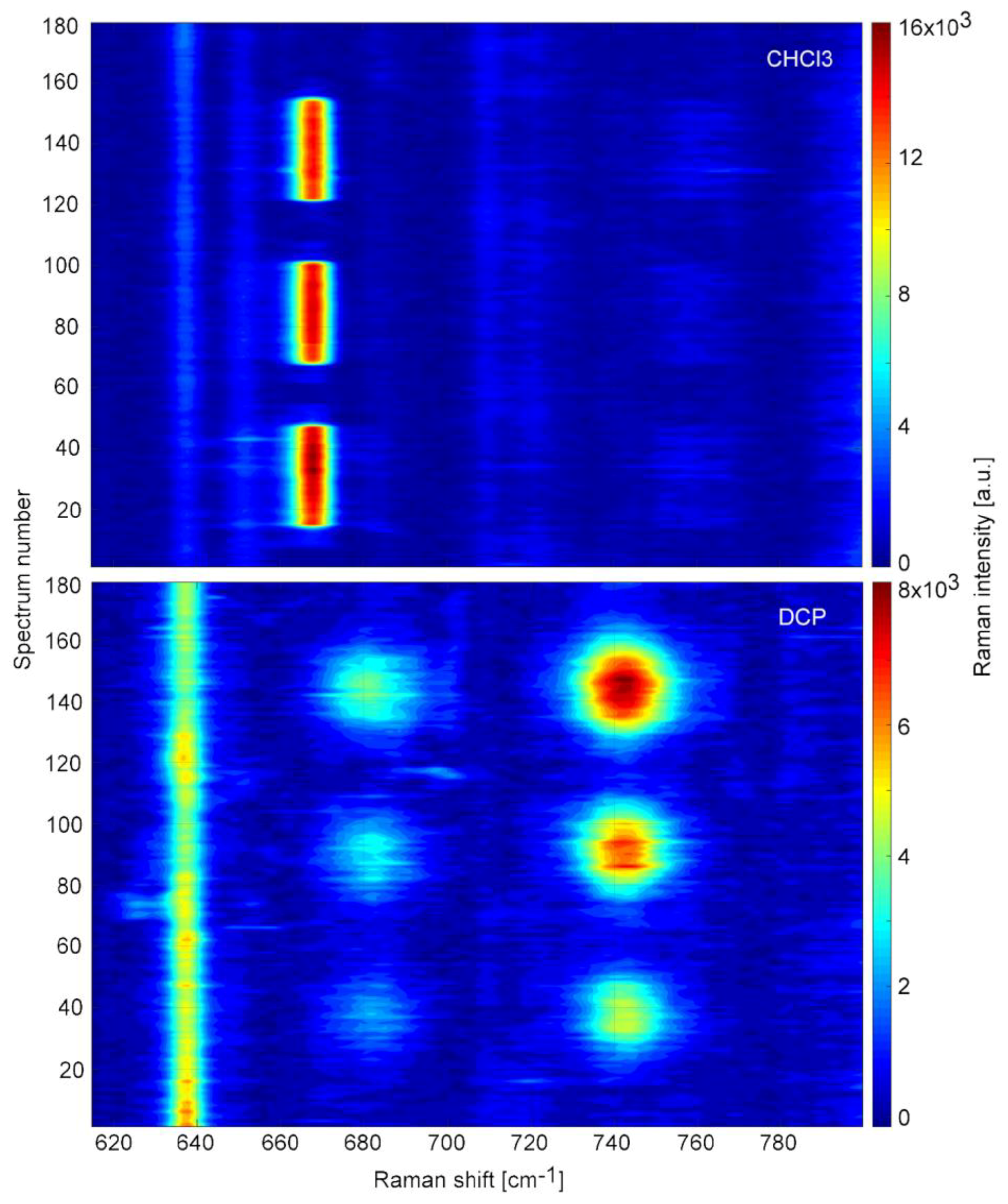
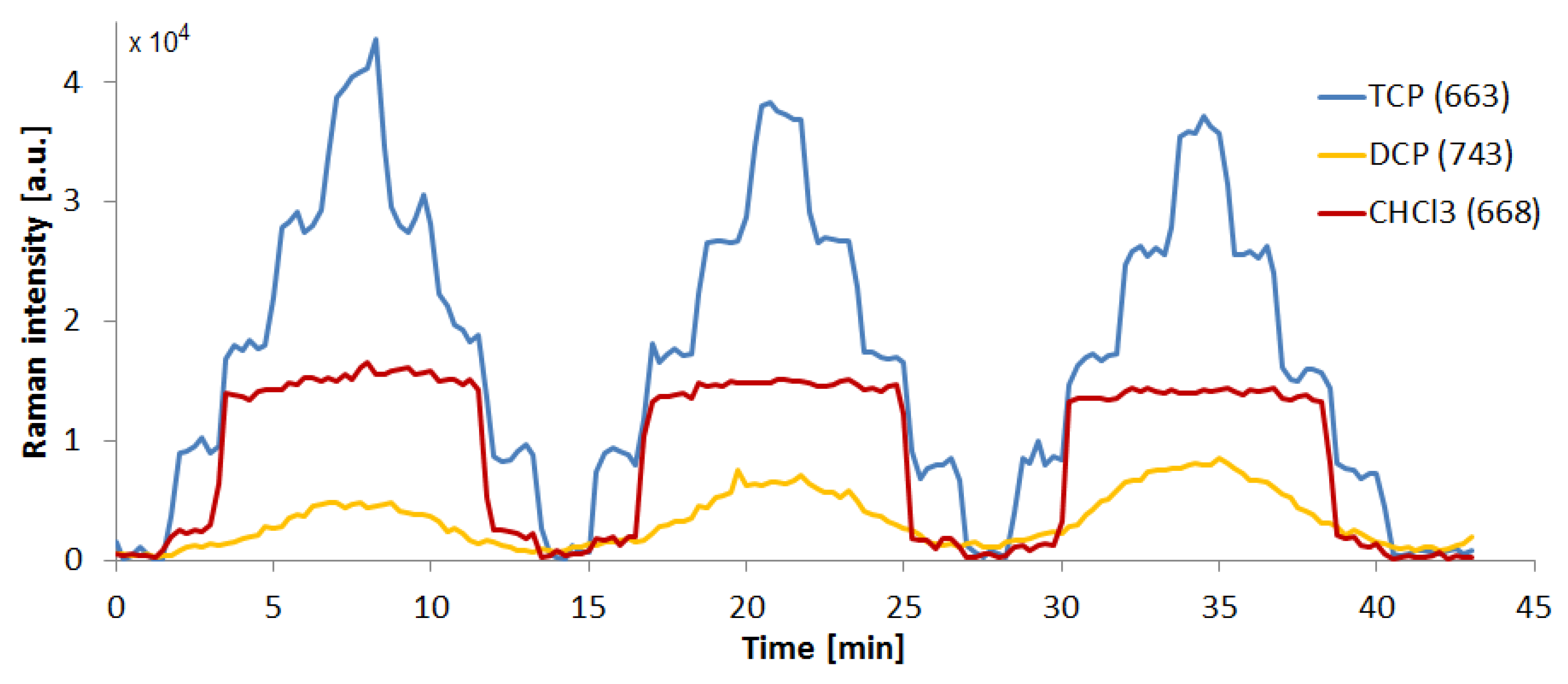
3.6. Retention of TCP in the Microfluidic System
4. Discussion
4.1. Using the Microfluidic SERS Detector with SK307 for Environmental Monitoring
4.2. Gradual Loss of the SERS Signal over Time
Author Contributions
Funding
Conflicts of Interest
References
- Wachsmann-Hogiu, S.; Weeks, T.; Huser, T. Chemical analysis in vivo and in vitro by Raman spectroscopy - from single cells to humans. Curr. Opin. Biotechnol. 2009, 20, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Paudel, A.; Raijada, D.; Rantanen, J. Raman spectroscopy in pharmaceutical product design. Adv. Drug Deliv. Rev. 2015, 89, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Mallick, B.; Lakshmanna, A.; Radhalakshmi, V.; Umapathy, S. Design and development of stimulated Raman spectroscopy apparatus using a femtosecond laser system. Curr. Sci. 2008, 95, 1551–1559. [Google Scholar]
- Smekal, A. Zur quantentheorie der dispersion. Naturwissenschaftliche 1923, 11, 873–875. [Google Scholar] [CrossRef]
- Raman, C.V.; Krishnan, K.S. A new type of secondary radiation. Nature 1928, 121, 501–502. [Google Scholar] [CrossRef]
- Jeanmaire, D.L.; Duyne, R.P.V. Surface Raman spectroelectrochemistry: Part I. heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. Interfaces Electrochem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Creighton, J.A.; Blatchford, C.G.; Albrecht, M.G. Plasma resonance enhancement of Raman scattering by pyridine adsorbed on silver or gold sol particles of size comparable to the excitation wavelength. J. Chem. Soc. Faraday Trans. 1979, 75, 790–798. [Google Scholar] [CrossRef]
- Kneipp, K.; Kneipp, H.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Ultrasensitive chemical analysis by Raman spectroscopy. Chem. Rev. 1999, 99, 2957–2976. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Frontiera, R.R.; Henry, A.I.; Ringe, E.; Duyne, R.P.V. SERS: Materials, applications, and the future. Mater. Today 2012, 15, 16–25. [Google Scholar] [CrossRef]
- Le Ru, E.C.; Blackie, E.; Meyer, M.; Etchegoin, P.G. Surface enhanced Raman scattering enhancement factors: A comprehensive study. J. Phys. Chem. C 2007, 111, 13794–13803. [Google Scholar] [CrossRef]
- Xu, H.; Aizpurua, J.; Kall, M.; Apell, P. Electromagnetic contributions to single-molecule sensitivity in surface-enhanced Raman scattering. Phys. Rev. E 2000, 62, 4318–4324. [Google Scholar] [CrossRef]
- Yu, X.; Cai, H.; Zhang, W.; Li, X.; Pan, N.; Luo, Y.; Wang, X.; Hou, J.G. Tuning chemical enhancement of SERS by controlling the chemical reduction of graphene oxide nanosheets. ACS Nano 2011, 5, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.J.; Lin, M.; Hu, Z.; Li, H. Evaporation-controlled chemical enhancement of SERS using a soft polymer substrate. Chem. Commun. (Camb.) 2009, 41, 6246–6248. [Google Scholar] [CrossRef] [PubMed]
- Hakonen, A.; Svedendahl, M.; Ogier, R.; Yang, Z.J.; Lodewijks, K.; Verre, R.; Shegai, T.; Andersson, P.O.; Kall, M. Dimer-on-mirror SERS substrates with attogram sensitivity fabricated by colloidal lithography. Nanoscale 2015, 7, 9405–9410. [Google Scholar] [CrossRef] [PubMed]
- Maier, S.A. Plasmonic field enhancement and SERS in the effective mode volume picture. Opt. Express 2006, 14, 1957–1964. [Google Scholar] [CrossRef] [PubMed]
- De Luca, A.C.; Reader-Harris, P.; Mazilu, M.; Mariggio, S.; Corda, D.; Di Falco, A. Reproducible surface-enhanced Raman quantification of biomarkers in multicomponent mixtures. ACS Nano 2014, 8, 2575–2583. [Google Scholar] [CrossRef] [PubMed]
- King, M.D.; Khadka, S.; Craig, G.A.; Mason, M.D. Effect of local heating on the SERS efficiency of optically trapped prismatic nanoparticles. J. Phys. Chem. C 2008, 112, 11751–11757. [Google Scholar] [CrossRef]
- Hakonen, A.; Wang, F.C.; Andersson, P.O.; Wingfors, H.; Rindzevicius, T.; Schmidt, M.S.; Soma, V.R.; Xu, S.; Li, Y.Q.; Boisen, A.; et al. Hand-held femtogram detection of hazardous picric acid with hydrophobic Ag nanopillar SERS substrates and mechanism of elasto-capillarity. ACS Sens. 2017, 2, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Hakonen, A.; Wu, K.; Stenbek Schmidt, M.; Andersson, P.O.; Boisen, A.; Rindzevicius, T. Detecting forensic substances using commercially available SERS substrates and handheld Raman spectrometers. Talanta 2018, 189, 649–652. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Endo, T.; Imai, H.; Kido, M.; Jeong, H.; Ohno, Y. Effectiveness of surface enhanced Raman spectroscopy of tear fluid with soft substrate for point-of-care therapeutic drug monitoring. SPIE BiOS 2016, 97150E. [Google Scholar] [CrossRef]
- Wen, N.; Zhao, Z.; Fan, B.; Chen, D.; Men, D.; Wang, J.; Chen, J. Development of droplet microfluidics enabling high-throughput single-cell analysis. Molecules 2016, 21, 881. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; He, L. Surface-enhanced Raman spectroscopy for the chemical analysis of food. Compr. Rev. Food Sci. Food Saf. 2014, 13, 317–328. [Google Scholar] [CrossRef]
- Zhou, Q.; Meng, G.; Liu, J.; Huang, Z.; Han, F.; Zhu, C.; Kim, D.J.; Kim, T.; Wu, N. A hierarchical nanostructure-based surface-enhanced Raman scattering sensor for preconcentration and detection of antibiotic pollutants. Adv. Mat. Technol. 2017, 2, 1700028. [Google Scholar] [CrossRef]
- White, I.M.; Yazdi, S.H.; Yu, W.W. Optofluidic SERS: Synergizing photonics and microfluidics for chemical and biological analysis. Microfluid. Nanofluid. 2012, 13, 205–216. [Google Scholar] [CrossRef]
- Yazdi, S.H.; White, I.M. Multiplexed detection of aquaculture fungicides using a pump-free optofluidic SERS microsystem. Analyst 2013, 138, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jing, N.; Chou, I.H.; Cote, G.L.; Kameoka, J. An optofluidic device for surface enhanced Raman spectroscopy. Lab Chip 2007, 7, 630–632. [Google Scholar] [CrossRef] [PubMed]
- Parisi, J.; Su, L.; Lei, Y. In situ synthesis of silver nanoparticle decorated vertical nanowalls in a microfluidic device for ultrasensitive in-channel SERS sensing. Lab Chip 2013, 13, 1501–1508. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Ye, J. Reproducibility in surface-enhanced Raman spectroscopy. J. Shanghai Jiaotong Univ. (Sci.) 2014, 19, 681–690. [Google Scholar] [CrossRef]
- Reyer, A.; Prinz, A.; Giancristofaro, S.; Schneider, J.; Bertoldo Menezes, D.; Zickler, G.; Bourret, G.R.; Musso, M.E. Investigation of mass-produced substrates for reproducible surface-enhanced Raman scattering measurements over large areas. ACS Appl. Mat. Interfaces 2017, 9, 25445–25454. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, P.; Kurumbang, N.P.; Bendl, J.; Brezovsky, J.; Prokop, Z.; Damborsky, J. Maximizing the efficiency of multienzyme process by stoichiometry optimization. ChemBioChem. 2014, 15, 1891–1895. [Google Scholar] [CrossRef] [PubMed]
- Kurumbang, N.P.; Dvorak, P.; Bendl, J.; Brezovsky, J.; Prokop, Z.; Damborsky, J. Computer-assisted engineering of the synthetic pathway for biodegradation of a toxic persistent pollutant. ACS Synth. Biol. 2014, 3, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, P.; Bidmanova, S.; Damborsky, J.; Prokop, Z. Immobilized synthetic pathway for biodegradation of toxic recalcitrant pollutant 1,2,3-trichloropropane. Environ. Sci. Technol. 2014, 48, 6859–6866. [Google Scholar] [CrossRef] [PubMed]
- Perney, N.M.B.; Baumberg, J.J.; Zoorob, M.E.; Charlton, M.D.B.; Mahnkopf, S.; Netti, C.M. Tuning localized plasmons in nanostructured substrates for surface-enhanced Raman scattering. Opt. Express 2006, 14, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Shiroma, L.S.; Piazzetta, M.H.O.; Duarte-Junior, G.F.; Coltro, W.K.T.; Carrilho, E.; Gobbi, A.L.; Lima, R.S. Self-regenerating and hybrid irreversible/reversible PDMS microfluidic devices. Sci. Rep. 2016, 6, 26032. [Google Scholar] [CrossRef] [PubMed]
- McIlvaine, T.C. A buffer solution for colorimetric comparison. J. Biol. Chem. 1921, 49, 183–186. [Google Scholar]
- Brandt, N.N.; Brovko, O.O.; Chikishev, A.Y.; Paraschuk, O.D. Optimization of the rolling-circle filter for Raman background subtraction. Appl. Spectrosc. 2006, 60, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.T.; Haes, A.J. Impacts of pH and intermolecular interactions on surface-enhanced Raman scattering chemical enhancements. J. Phys. Chem. C 2018, 122, 14846–14856. [Google Scholar] [CrossRef]
- Bi, L.; Wang, Y.; Yang, Y.; Li, Y.; Mo, S.; Zheng, Q.; Chen, L. Highly sensitive and reproducible SERS sensor for biological pH detection based on a uniform gold nanorod array platform. ACS Appl. Mater. Interf. 2018, 10, 15381–15387. [Google Scholar] [CrossRef] [PubMed]
- Hardy, M.; Doherty, M.D.; Krstev, I.; Maier, K.; Moller, T.; Muller, G.; Dawson, P. Detection of low-concentration contaminants in solution by exploiting chemical derivatization in surface-enhanced Raman spectroscopy. Anal. Chem. 2014, 86, 9006–9012. [Google Scholar] [CrossRef] [PubMed]
- Thorbjornsrud, J.; Ellestad, O.; Klaboe, P.; Torgrimsen, T. Substituted propanes: VI. the vibrational spectra and conformations of 1,3-dichloro-, 1,3-bromochloro-, 1,3-dibromo- and 1,3-diiodo-propane. J. Mol. Struct. 1973, 15, 61–76. [Google Scholar] [CrossRef]
- Pavlova, M.; Klvana, M.; Prokop, Z.; Chaloupkova, R.; Banas, P.; Otyepka, M.; Wade, R.C.; Tsuda, M.; Nagata, Y.; Damborsky, J. Redesigning dehalogenase access tunnels as a strategy for degrading an anthropogenic substrate. Nat. Chem. Biol. 2009, 5, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, P.; Chrast, L.; Nikel, P.I.; Fedr, R.; Soucek, K.; Sedlackova, M.; Chaloupkova, R.; de Lorenzo, V.; Prokop, Z.; Damborsky, J. Exacerbation of substrate toxicity by IPTG in Escherichia coli BL21(de3) carrying a synthetic metabolic pathway. Microb. Cell. Factories 2015, 14, 201. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, P.; Nikel, P.I.; Damborsky, J.; de Lorenzo, V. Bioremediation 3.0: Engineering pollutant-removing bacteria in the times of systemic biology. Biotechnol. Adv. 2017, 35, 845–866. [Google Scholar] [CrossRef]
- Huang, W.; Jing, Q.; Du, Y.; Zhang, B.; Meng, X.; Sun, M.; Schanze, K.S.; Gao, H.; Xu, P. An in situ SERS study of substrate-dependent surface plasmon induced aromatic nitration. J. Mater. Chem. C 2015, 3, 5285–5291. [Google Scholar] [CrossRef]
- Ankudze, B.; Pakkanen, T.T. Gold nanoparticle decorated Au-Ag alloy tubes: A bifunctional substrate for label-free and in situ surface-enhanced Raman scattering based reaction monitoring. Appl. Surf. Sci. 2018, 453, 341–349. [Google Scholar] [CrossRef]
- Oo, S.Z.; Charlton, M.D.B.; Eustace, D.; Chen, R.Y.; Pearce, S.J.; Pollard, M.E. Optimization of SERS enhancement from nanostructured metallic substrate based on arrays of inverted rectangular pyramids and investigation of effect of lattice non-symmetry. Proc. SPIE 2012, 8234, 8234–8237. [Google Scholar] [CrossRef]
- Fukami, K.; Chourou, M.L.; Miyagawa, R.; Munoz Noval, A.; Sakka, T.; Manso-Silvan, M.; Martin-Palma, R.J.; Ogata, Y.H. Gold nanostructures for surface-enhanced Raman spectroscopy, prepared by electrodeposition in porous silicon. Materials 2011, 4, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Bandarenka, H.V.; Girel, K.V.; Zavatski, S.A.; Panarin, A.; Terekhov, S.N. Progress in the development of SERS-active substrates based on metal-coated porous silicon. Materials 2018, 11, 852. [Google Scholar] [CrossRef] [PubMed]
- Mullen, K.; Carron, K. Adsorption of chlorinated ethylenes at 1-octadecanethiol-modified silver surfaces. Anal. Chem. 1994, 66, 478–483. [Google Scholar] [CrossRef]
- Storey, J.M.E.; Shelton, R.D.; Barber, T.E.; Wachter, E.A. Electrochemical SERS detection of chlorinated hydrocarbons in aqueous solutions. Appl. Spectrosc. 1994, 48, 1265–1271. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pilát, Z.; Kizovský, M.; Ježek, J.; Krátký, S.; Sobota, J.; Šiler, M.; Samek, O.; Buryška, T.; Vaňáček, P.; Damborský, J.; et al. Detection of Chloroalkanes by Surface-Enhanced Raman Spectroscopy in Microfluidic Chips. Sensors 2018, 18, 3212. https://doi.org/10.3390/s18103212
Pilát Z, Kizovský M, Ježek J, Krátký S, Sobota J, Šiler M, Samek O, Buryška T, Vaňáček P, Damborský J, et al. Detection of Chloroalkanes by Surface-Enhanced Raman Spectroscopy in Microfluidic Chips. Sensors. 2018; 18(10):3212. https://doi.org/10.3390/s18103212
Chicago/Turabian StylePilát, Zdeněk, Martin Kizovský, Jan Ježek, Stanislav Krátký, Jaroslav Sobota, Martin Šiler, Ota Samek, Tomáš Buryška, Pavel Vaňáček, Jiří Damborský, and et al. 2018. "Detection of Chloroalkanes by Surface-Enhanced Raman Spectroscopy in Microfluidic Chips" Sensors 18, no. 10: 3212. https://doi.org/10.3390/s18103212
APA StylePilát, Z., Kizovský, M., Ježek, J., Krátký, S., Sobota, J., Šiler, M., Samek, O., Buryška, T., Vaňáček, P., Damborský, J., Prokop, Z., & Zemánek, P. (2018). Detection of Chloroalkanes by Surface-Enhanced Raman Spectroscopy in Microfluidic Chips. Sensors, 18(10), 3212. https://doi.org/10.3390/s18103212




