Textile Concentric Ring Electrodes for ECG Recording Based on Screen-Printing Technology
Abstract
1. Introduction
2. Materials and Methods
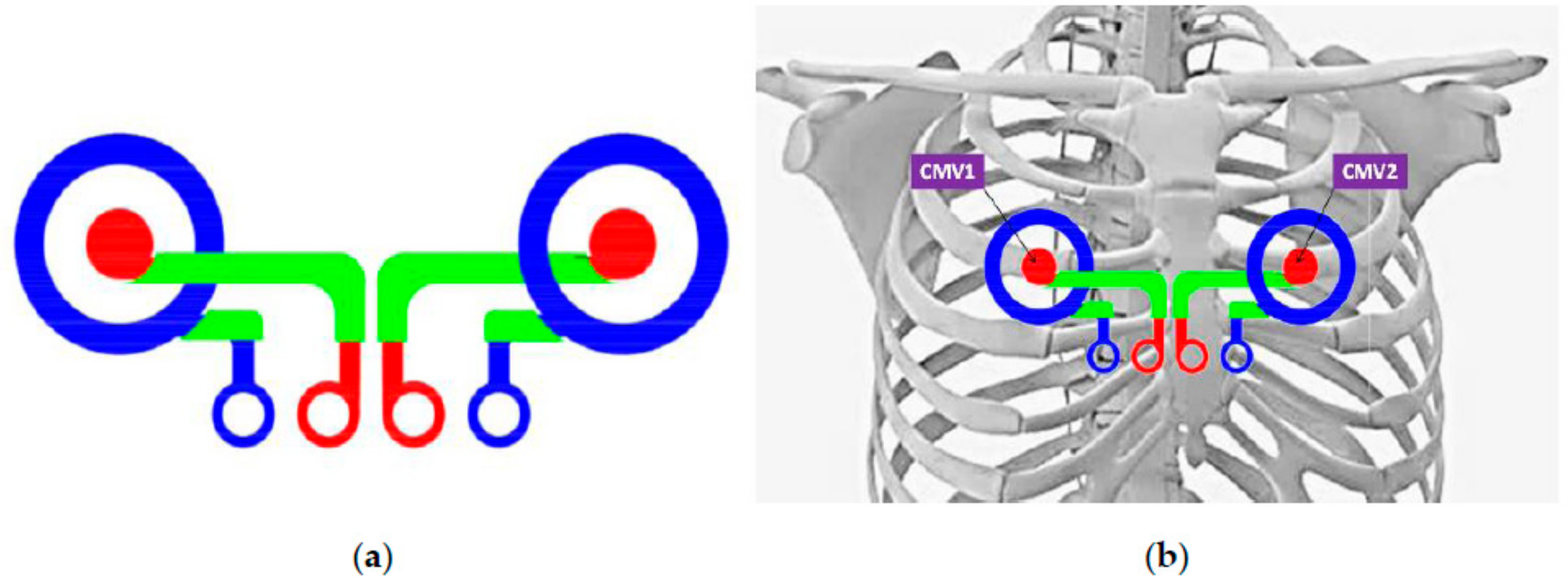
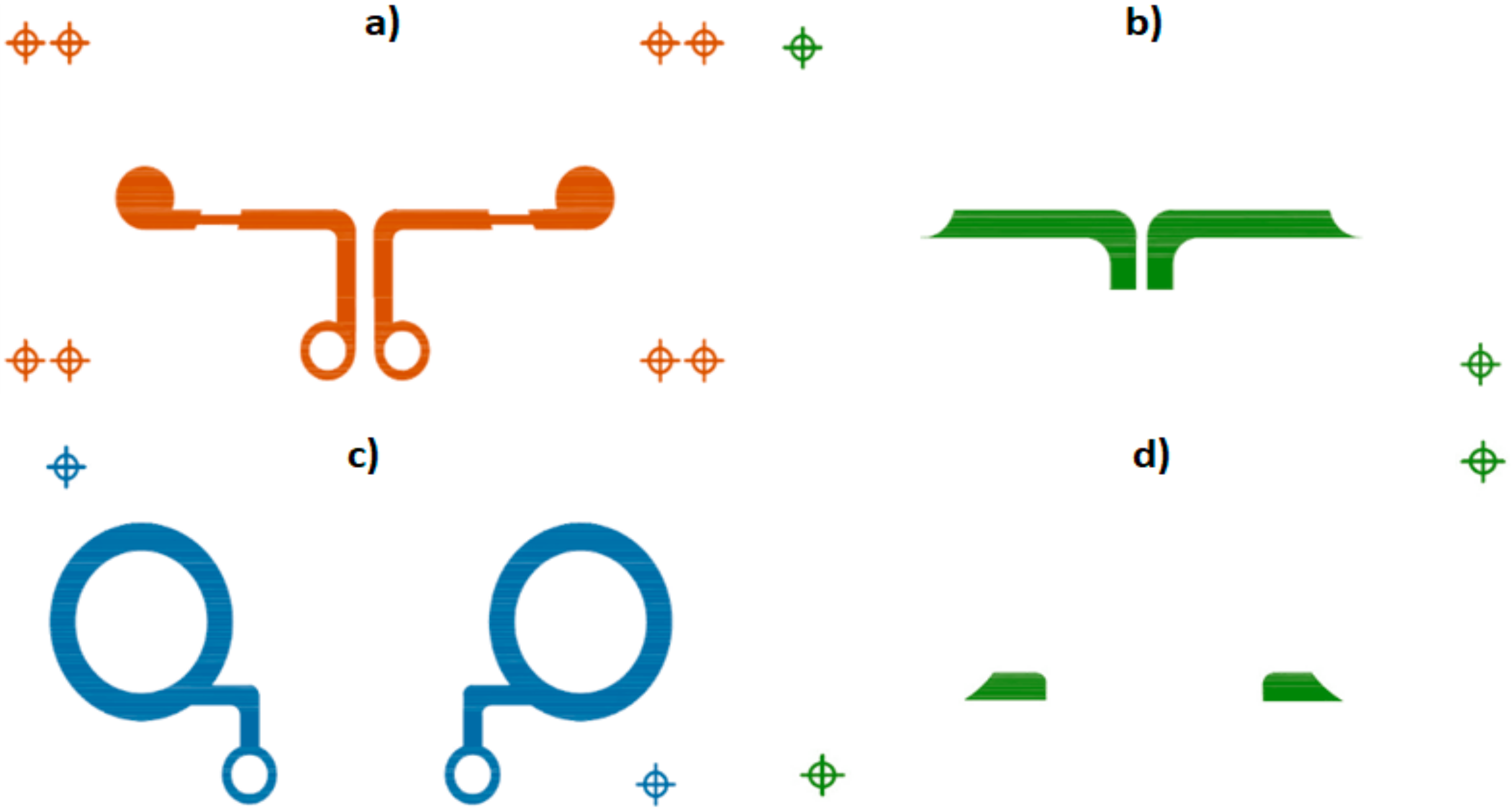
2.1. Textile Concentric Ring Electrodes (CRE): Design and Development
2.2. Physical and Electrical Electrode Characterization
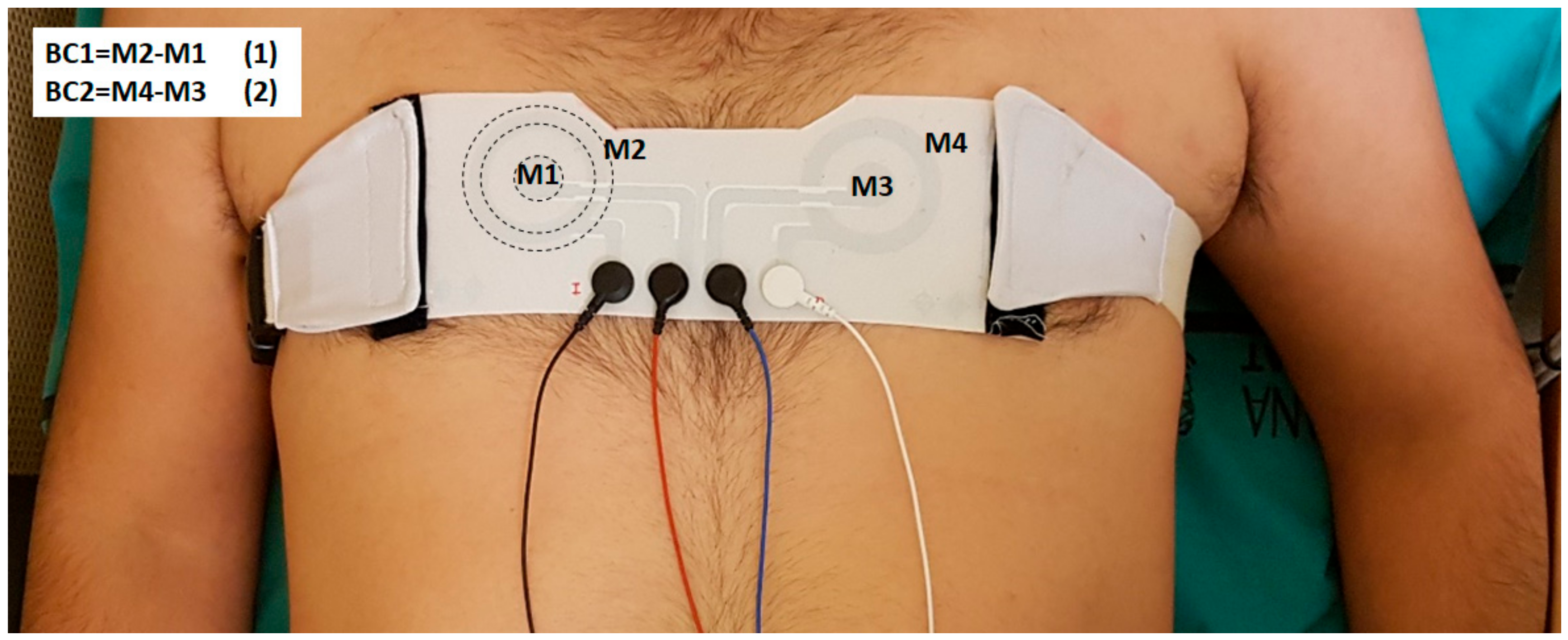
2.3. Recording Protocol
2.4. ECG Analysis
3. Results
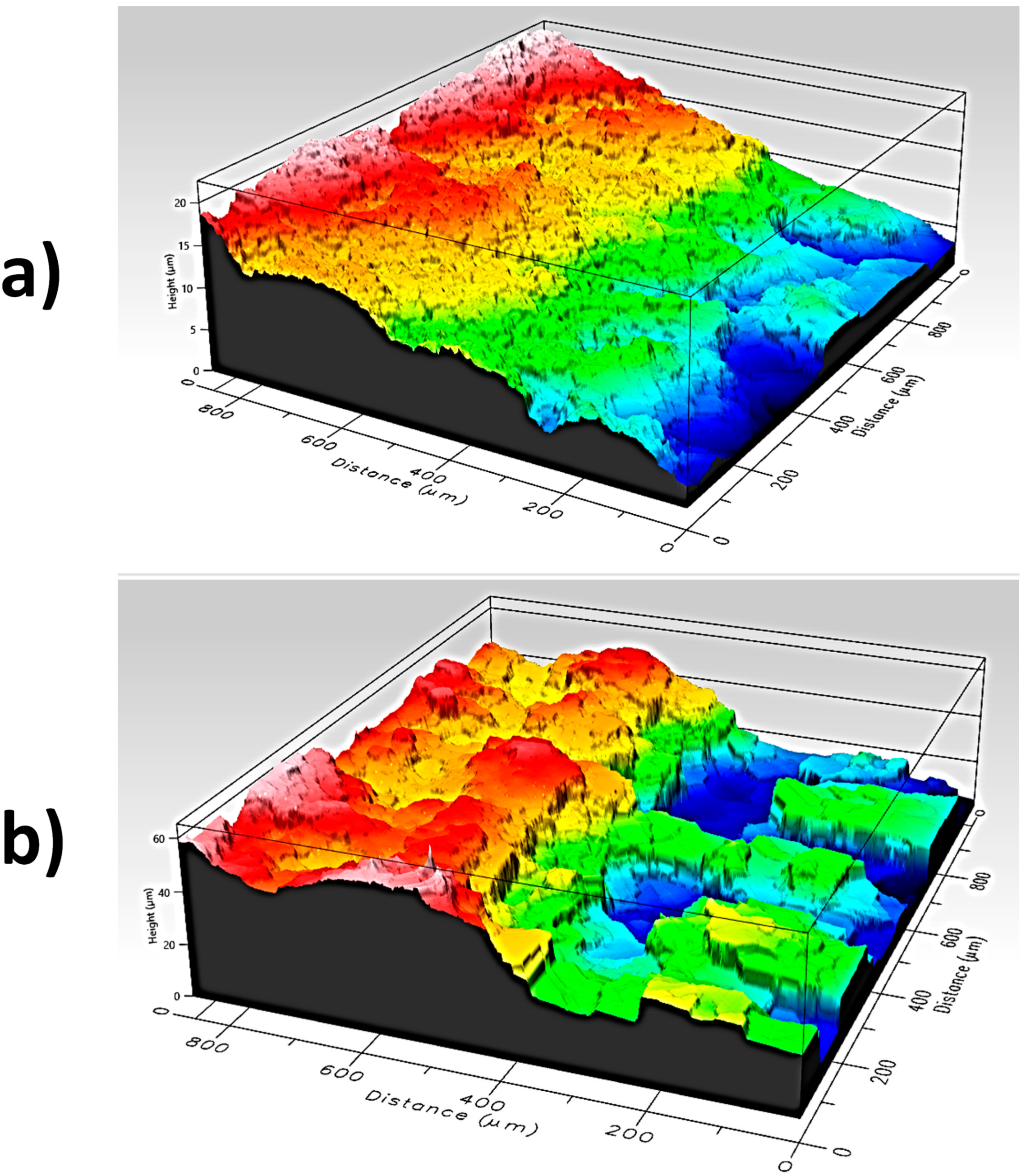
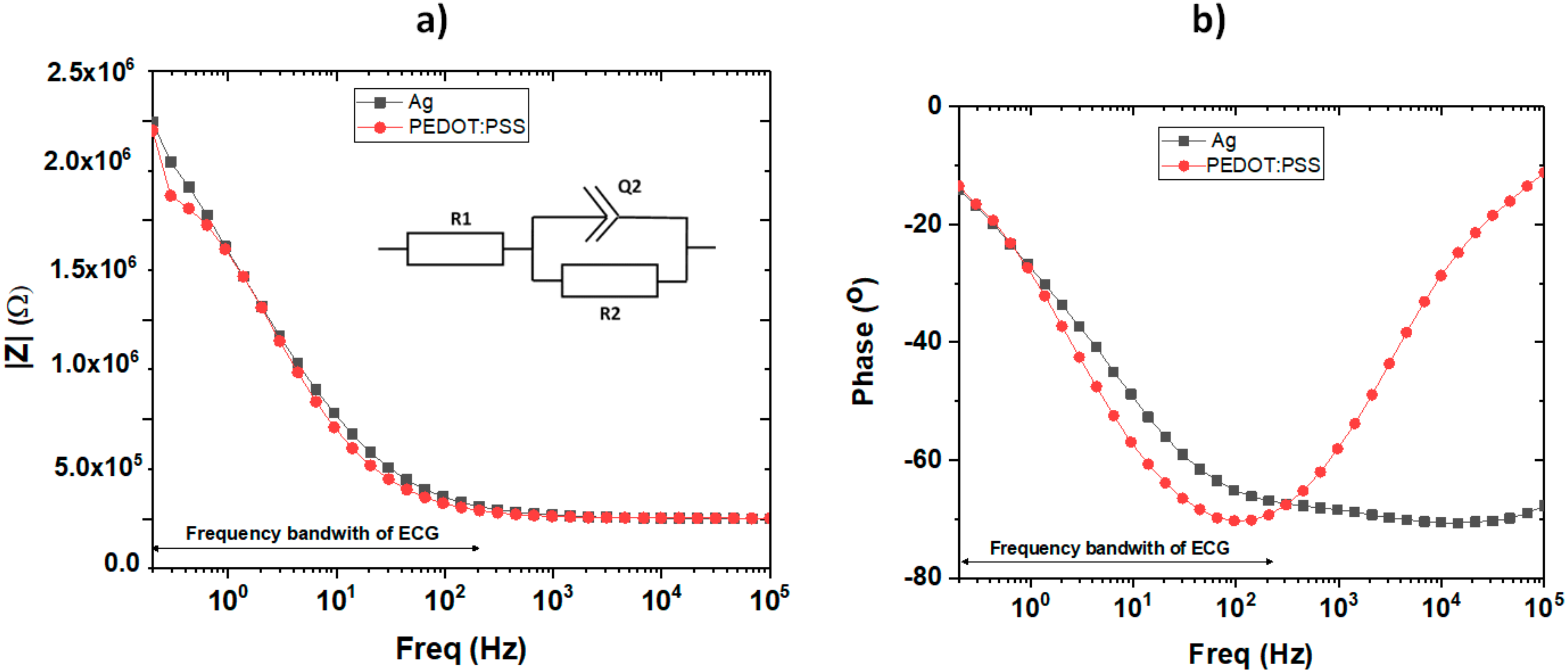
3.1. CREs Physical and Electrical Characteristics
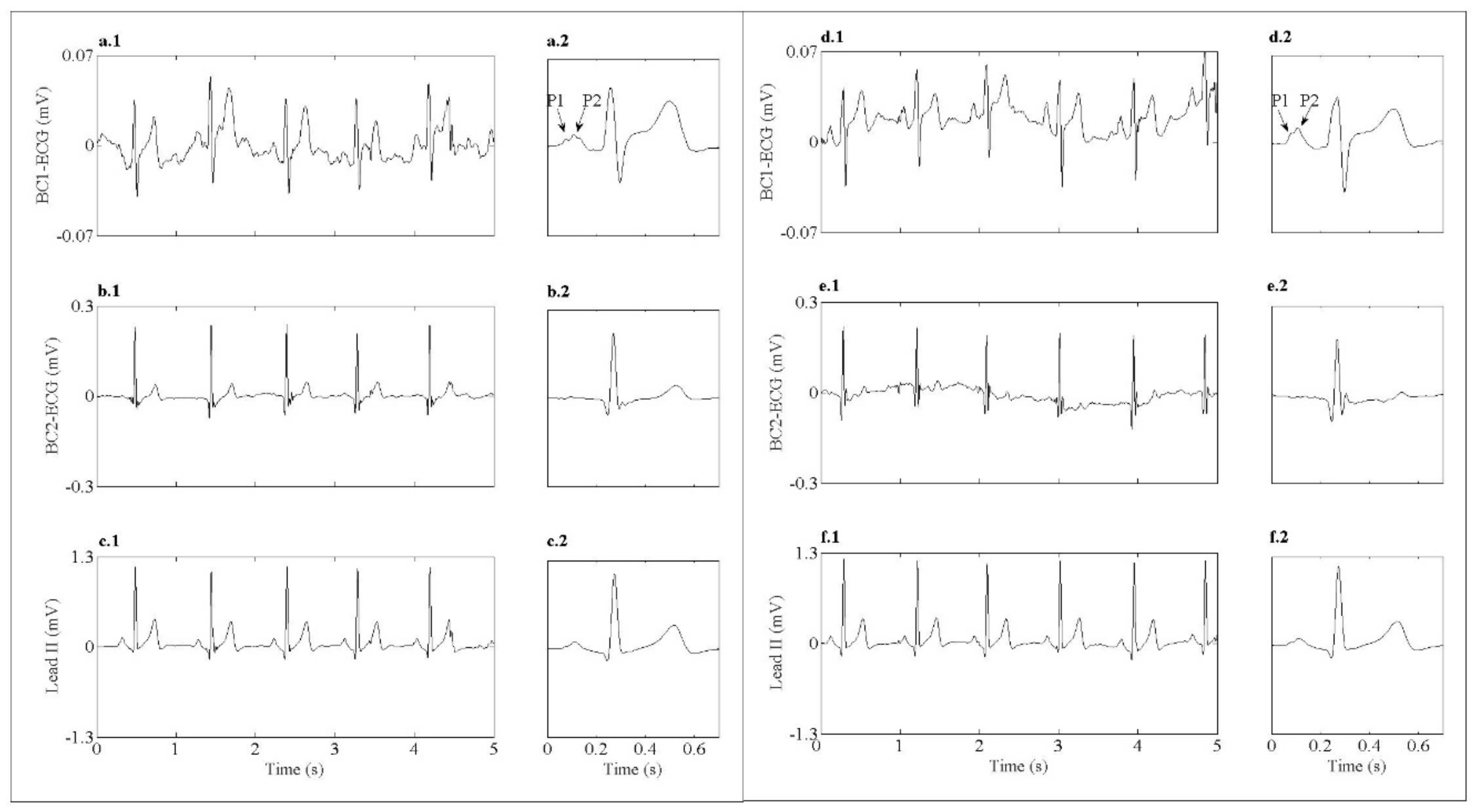
3.2. ECG Analysis
4. Discussion
5. Conclusions
- The use of silver on textiles presents better characteristics than PEDOT:PSS. Even so, techniques that improve the transfer of PEDOT:PSS to textiles could be used.
- Both textile silver and PEDOT:PSS CREs implemented on textile substrates are able to detect surface electrocardiographic activity in standard precordial recording positions, similar to V1 and V2 (CMV1 and CMV2).
- The amplitudes the BC-ECG signals detected with the textile CREs developed are of hundreds of microvolts, which is slightly lower than those of conventional bipolar ECG signals in precordial positions.
- BC-ECGs recorded with silver and PEDOT:PSS textile CREs presented similar signal to noise ratios with these values being similar to those of BC-ECGs from CREs implemented on plastic substrates published in the literature.
- Regarding the saturation and alterations of the BC-ECGs associated with movement of the subject, textile silver CRE showed a more stable response (fewer saturations and alterations) than PEDOT:PSS.
- Regardless the use of silver or PEDOT:PSS, BC-ECG signals captured with the developed textile CREs have a better spatial resolution than that of conventional recordings (lead II). Specifically, BC-ECG signals have an improved capability of recording atrial activity on the surface, with P1 and P2 waves being associated with the activity of the left and right atria identified in most BC-ECG signals.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Trung, T.Q.; Lee, N.E. Flexible and stretchable physical sensor integrated platforms for wearable human-activity monitoring and personal healthcare. Adv. Mater. 2016, 28, 4338–4372. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Breijo, E.; Prats-Boluda, G.; Lidon-Roger, J.V.; Ye-Lin, Y.; Garcia-Casado, J. A comparative analysis of printing techniques by using an active concentric ring electrode for bioelectrical recording. Microelectron. Int. 2015, 32, 103–107. [Google Scholar] [CrossRef]
- Linti, C.; Horter, H.; Österreicher, P.; Planck, H. Sensory baby vest for the monitoring of infants. In Proceedings of the International Workshop on Wearable Implantable Body Sensor Networks (BSN 2006), Cambridge, MA, USA, 3–5 April 2006; pp. 135–137. [Google Scholar] [CrossRef]
- Wei, Y.; Torah, R.; Li, Y.; Tudor, J. Dispenser printed capacitive proximity sensor on fabric for applications in the creative industries. Sens. Actuators A Phys. 2016, 247, 239–246. [Google Scholar] [CrossRef]
- Takamatsu, S.; Lonjaret, T.; Ismailova, E.; Masuda, A.; Itoh, T.; Malliaras, G.G. Wearable keyboard using conducting polymer electrodes on textiles. Adv. Mater. 2016, 28, 4485–4488. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Kim, J.H.; Kwon, H.J.; Kwon, Y.H. A touchpad for force and location sensing. ETRI J. 2010, 32, 722–728. [Google Scholar] [CrossRef]
- Catrysse, M.; Puers, R.; Hertleer, C.; Van Langenhove, L.; Van Egmond, H.; Matthys, D. Fabric sensors for the measurement of physiological parameters. In Proceedings of the 12th International Conference on Transducers Solid-State Sensors, Actuators and Microsystems, Boston, MA, USA, 8ߝ12 June 2003; pp. 1758–1761. [Google Scholar] [CrossRef]
- Catrysse, M.; Puers, R.; Hertleer, C.; Van Langenhove, L.; Van Egmond, H.; Matthys, D. Towards the integration of textile sensors in a wireless monitoring suit. Sens. Actuators A Phys. 2004, 114, 302–311. [Google Scholar] [CrossRef]
- Chi, Y.M.; Deiss, S.R.; Cauwenberghs, G. Non-contact low power EEG/ECG electrode for high density wearable biopotential sensor networks. In Proceedings of the 6th International Workshop on Wearable Implantable Body Sensor Networks (BSN 2009), Berkeley, CA, USA, 3–5 June 2009. [Google Scholar] [CrossRef]
- Al-huda Hamdan, N.; Heller, F.; Wacharamanotham, C.; Thar, J.; Borchers, J. Grabrics: A foldable two-dimensional textile input controller. CHI Ext. Abstr. Hum. Factors Comput. Syst. 2016. [CrossRef]
- Baig, M.M.; Gholamhosseini, H.; Connolly, M.J. A comprehensive survey of wearable and wireless ECG monitoring systems for older adults. Med. Biol. Eng. Comput. 2013, 51, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.L.; Ding, X.R.; Poon, C.C.Y.; Lo, B.P.L.; Zhang, H.; Zhou, X.L.; Yang, G.Z.; Zhao, N.; Zhang, Y.T. Unobtrusive sensing and wearable devices for health informatics. IEEE Trans. Biomed. Eng. 2014, 61, 1538–1554. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yu, X.B. Capacitive biopotential measurement for electrophysiological signal acquisition: a review. IEEE Sens. J. 2016, 16, 2832–2853. [Google Scholar] [CrossRef]
- Lin, C.T.; Liao, L.D.; Liu, Y.H.; Wang, I.J.; Lin, B.S.; Chang, J.Y. Novel dry polymer foam electrodes for long-term EEG measurement. IEEE Trans. Biomed. Eng. 2011, 58, 1200–1207. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, K.P.; Ruff, R.; Poppendieck, W. Long-term characterization of electrode materials for surface electrodes in biopotential recording. Annu. Int. Conf. IEEE Eng. Med. Biol.-Proc. 2006. [Google Scholar] [CrossRef]
- Pylatiuk, C.; Müller-Riederer, M.; Kargov, A.; Schulz, S.; Schill, O.; Reischl, M.; Bretthauer, G. Comparison of surface EMG monitoring electrodes for long term use in rehabilitation device control. In Proceedings of the 2009 IEEE 11th International Conference on Rehabilitation Robotics, Kyoto, Japan, 23–26 June 2009; pp. 300–304. [Google Scholar]
- Jung, H.; Moon, J.; Baek, D.; Lee, J.; Choi, Y.; Hong, J. CNT/PDMS composite flexible dry electrodes for long-term ECG monitoring. IEEE Trans. Biomed. Eng. 2012, 59, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Otley, M.T.; Li, M.; Zhang, X.; Sinha, S.K.; Treich, G.M.; Sotzing, G.A. PEDOT:PSS “wires” printed on textile for wearable electronics. ACS Appl. Mater. Interfaces. 2016, 8, 26998–27005. [Google Scholar] [CrossRef] [PubMed]
- Papaiordanidou, M.; Takamatsu, S.; Rezaei-Mazinani, S.; Lonjaret, T.; Martin, A.; Ismailova, E. Cutaneous recording and stimulation of muscles using organic electronic textiles. Adv. Healthc. Mater. 2016, 5, 2001–2006. [Google Scholar] [CrossRef] [PubMed]
- Rivnay, J.; Leleux, P.; Ferro, M.; Sessolo, M.; Williamson, A.; Koutsouras, D.A.; Khodagholy, D.; Ramuz, M.; Strakosas, X.; Owens, R.M.; Benar, C.; Badier, J.-M.; Bernard, C.; Malliaras, G.G. High-performance transistors for bioelectronics through tuning of channel thickness. Sci. Adv. 2015, 1, e1400251. [Google Scholar] [CrossRef] [PubMed]
- Takamatsu, S.; Lonjaret, T.; Crisp, D.; Badier, J.-M.; Malliaras, G.G.; Ismailova, E. Direct patterning of organic conductors on knitted textiles for long-term electrocardiography. Sci. Rep. 2015, 5, 15003. [Google Scholar] [CrossRef] [PubMed]
- Pandian, P.S.; Mohanavelu, K.; Safeer, K.P.; Kotresh, T.M.; Shakunthala, D.T.; Gopal, P.; Padaki, V.C. Smart vest: wearable multi-parameter remote physiological monitoring system. Med. Eng. Phys. 2008, 30, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Bradshaw, L.A.; Richards, W.O.; Wikswo, J.P. Volume conductor effects on the spatial resolution of magnetic fields and electric potentials from gastrointestinal electrical activity. Med. Biol. Eng. Comput. 2001, 39, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Besio, W.; Aakula, R.; Dai, W. Comparison of bipolar vs. tripolar concentric ring electrode Laplacian estimates. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2004, 3, 2255–2258. [Google Scholar] [CrossRef]
- Makeyev, O.; Ding, Q.; Besio, W.G. Improving the accuracy of Laplacian estimation with novel multipolar concentric ring electrodes. Measurement 2016, 80, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, B. An online transformation of EEG scalp potentials into orthogonal source derivations. Electroencephalogr. Clin. Neurophysiol. 1975, 39, 526–530. [Google Scholar] [CrossRef]
- Wu, D.; Tsai, H.C.; He, B. On the estimation of the Laplacian electrocardiogram during ventricular activation. Ann. Biomed. Eng. 1999, 27, 731–745. [Google Scholar] [CrossRef] [PubMed]
- Tandonnet, C.; Burle, B.; Hasbroucq, T.; Vidal, F. Spatial enhancement of EEG traces by surface Laplacian estimation: comparison between local and global methods. Clin. Neurophysiol. 2005, 116, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.C.; Tarjan, P.P. An ultra high common mode rejection ratio (CMRR) AC instrumentation amplifier for Laplacian electrocardiographic measurement. Biomed. Instrum. Technol. 1999, 33, 76–83. [Google Scholar] [PubMed]
- Besio, W.; Chen, T. Tripolar Laplacian electrocardiogram and moment of activation isochronal mapping. Physiol. Measurement 2007, 28, 515–529. [Google Scholar] [CrossRef] [PubMed]
- Prats-Boluda, G.; Ye-Lin, Y.; Garcia-Breijo, E.; Ibañez, J.; Garcia-Casado, J. Active flexible concentric ring electrode for non-invasive surface bioelectrical recordings. Meas. Sci. Technol. 2012, 23, 125703. [Google Scholar] [CrossRef]
- Prats-Boluda, G.; Ye-Lin, Y.; Bueno Barrachina, J.M.; Senent, E.; Rodriguez de Sanabria, R.; Garcia-Casado, J. Development of a portable wireless system for bipolar concentric ECG recording. Meas. Sci. Technol. 2015, 26, 75102. [Google Scholar] [CrossRef]
- Prats-Boluda, G.; Ye-Lin, Y.; Bueno-Barrachina, J.; Rodriguez de Sanabria, R.; Garcia-Casado, J. Towards the clinical use of concentric electrodes in ECG recordings: influence of ring dimensions and electrode position. Meas. Sci. Technol. 2016, 27, 25705. [Google Scholar] [CrossRef]
- Ye-Lin, Y.; Alberola-Rubio, J.; Prats-Boluda, G.; Perales, A.; Desantes, D.; Garcia-Casado, J. Feasibility and analysis of bipolar concentric recording of electrohysterogram with flexible active electrode. Ann. Biomed. Eng. 2014, 4, 968–976. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Parekh, U.; Pailla, T. Stretchable dry electrodes with concentric ring geometry for enhancing spatial resolution in electrophysiology. Adv. Healthc. Mater. 2017, 6, 1700552. [Google Scholar] [CrossRef] [PubMed]
- Junwei, M.; Han, Y.; Sunderam, S.; Besio, W.; Lei, D. Computation of surface Laplacian for tri-polar ring electrodes on high-density realistic geometry head model. In Proceedings of the 39th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Seogwipo, South Korea, 11–15 July 2017. [Google Scholar] [CrossRef]
- Besio, W.G.; Martínez-Juárez, I.E.; Makeyev, O.; Gaitanis, J.N.; Blum, A.S.; Fisher, R.S.; Medvedev, A.V. High-frequency oscillations recorded on the scalp of patients with epilepsy using tripolar concentric ring electrodes. IEEE J. Transl. Eng. Health Med. 2014, 2, 2000111. [Google Scholar] [CrossRef] [PubMed]
- Kaufer, M.; Rasquinha, L.; Tarjan, P. Optimization of multi-ring sensing electrode set. In Proceedings of the Annual Conference on Engineering in Medicine and Biology, Philadelphia, PA, USA, 1–4 November 1990; pp. 612–613. [Google Scholar]
- Besio, W.; Prasad, A. Analysis of skin-electrode impedance using concentric ring electrode. Proceedings 28th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA , 30 August 2006. [Google Scholar] [CrossRef]
- O’Mahony, C.; Grygoryev, K.; Ciarlone, A.; Giannoni, G.; Kenthao, A.; Galvin, P. Design, fabrication and skin-electrode contact analysis of polymer microneedle-based ECG electrodes. J. Micromech. Microeng. 2016, 26, 1–11. [Google Scholar] [CrossRef]
- Carr, J.J.; Brown, J.M. Introduction to Biomedical Equipment Technology, 4th ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2001; ISBN 0-13-010492-2. [Google Scholar]
- Pan, J.; Tompkins, W.J. A real-time qrs detection algorithm. IEEE Trans. Biomed. Eng. 1985, 32, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, P.S.; Tompkins, W.J. Quantitative investigation of QRS detection rules using the MIT/BIH arrhythmia database. IEEE Trans. Biomed. Eng. 1986, 33, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Ye-Lin, Y.; Bueno-Barrachina, J.M.; Prats-boluda, G.; de Sanabria, R.R.; Garcia-Casado, J. Wireless sensor node for non-invasive high precision electrocardiographic signal acquisition based on a multi-ring electrode. Measurement 2017, 97, 195–202. [Google Scholar] [CrossRef]


| Parameter | Units (mm) |
|---|---|
| Inner disc diameter | 16 |
| Ring internal diameter | 36 |
| Ring external diameter | 50 |
| Distance (between the discs’ centers) | 120 |
| Property | Ag C2131014D3 | PEDOT:PSS C2100629D1 |
|---|---|---|
| Solids Content (%) | 57.00–59.75 | - |
| Vicosity (Pa.s) | 6.5–13.5 | 0.5–2.0 |
| Curing condition (°C) | 130°/3 min | 130°/15 min |
| Sheet resistivity (25 µm) | 100 mΩ/sq | 500–700 Ω/sq |
| Property | Ag C2131014D3 | PEDOT:PSS C2100629D1 |
|---|---|---|
| R1 (Ω) | 4.80 × 102 | 2.73 × 103 |
| R2 (Ω) | 1.81 × 106 | 1.85 × 106 |
| Q2 (F·s(α−1)) | 69.79 × 10−9 | 79.96 × 10−9 |
| α | 0.76 | 0.79 |
| Left Ag | PEDOT:PSS | Right Ag | PEDOT:PSS | |
|---|---|---|---|---|
| External ring impedance (kΩ) | 18.3 ± 20.5 | 27.3 ± 22.3 | 21.3 ± 22.3 | 32.0 ± 21.0 |
| Inner disc impedance (kΩ) | 25.0 ± 20.1 | 25.3 ± 24.0 | 24.0 ± 19.3 | 32.0 ± 20.1 |
| Left | Right | |||
|---|---|---|---|---|
| Ag | PEDOT:PSS | Ag | PEDOT:PSS | |
| Peak to peak amplitude (μV) | 330.2 ± 126.2 | 363.9 ± 148.0 | 124.8 ± 133.6 | 143.8 ± 128.3 |
| SNR (dB) | 22.4 ± 6.3 | 20.1 ± 6.8 | 20.8 ± 6.6 | 20.2 ± 3.9 |
| Left | Right | |||||||
|---|---|---|---|---|---|---|---|---|
| Altered (%) | Saturated (%) | Altered (%) | Saturated (%) | |||||
| Ag | PEDOT:PSS | Ag | PEDOT:PSS | Ag | PEDOT:PSS | Ag | PEDOT:PSS | |
| Head | 3.2 | 10.1 | 0.0 | 3.1 | 5.9 | 9.6 | 0.0 | 1.1 |
| Arm | 13.9 | 28.8 | 0.9 | 17.7 | 13.6 | 31.6 | 0.9 | 12.2 |
| Leg | 18.9 | 36.9 | 0.1 | 13.1 | 24.2 | 28.1 | 0.0 | 10.7 |
| Laughing | 18.1 | 41.4 | 2.1 | 26.9 | 22.9 | 24.9 | 4.2 | 7.1 |
| Breathing | 13.9 | 44.5 | 6.1 | 36.1 | 25.5 | 29.9 | 3.5 | 13.3 |
| μ ± σ | 13.6 ± 6.2 | 32.3 ± 13.8 | 1.8 ± 2.6 | 19.4 ± 12.7 | 18.4 ± 8.4 | 24.9 ± 8.9 | 1.7 ± 2.0 | 8.9 ± 5.0 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lidón-Roger, J.V.; Prats-Boluda, G.; Ye-Lin, Y.; Garcia-Casado, J.; Garcia-Breijo, E. Textile Concentric Ring Electrodes for ECG Recording Based on Screen-Printing Technology. Sensors 2018, 18, 300. https://doi.org/10.3390/s18010300
Lidón-Roger JV, Prats-Boluda G, Ye-Lin Y, Garcia-Casado J, Garcia-Breijo E. Textile Concentric Ring Electrodes for ECG Recording Based on Screen-Printing Technology. Sensors. 2018; 18(1):300. https://doi.org/10.3390/s18010300
Chicago/Turabian StyleLidón-Roger, José Vicente, Gema Prats-Boluda, Yiyao Ye-Lin, Javier Garcia-Casado, and Eduardo Garcia-Breijo. 2018. "Textile Concentric Ring Electrodes for ECG Recording Based on Screen-Printing Technology" Sensors 18, no. 1: 300. https://doi.org/10.3390/s18010300
APA StyleLidón-Roger, J. V., Prats-Boluda, G., Ye-Lin, Y., Garcia-Casado, J., & Garcia-Breijo, E. (2018). Textile Concentric Ring Electrodes for ECG Recording Based on Screen-Printing Technology. Sensors, 18(1), 300. https://doi.org/10.3390/s18010300







