Application of the PAMONO-Sensor for Quantification of Microvesicles and Determination of Nano-Particle Size Distribution
Abstract
:1. Introduction
2. Materials and Methods
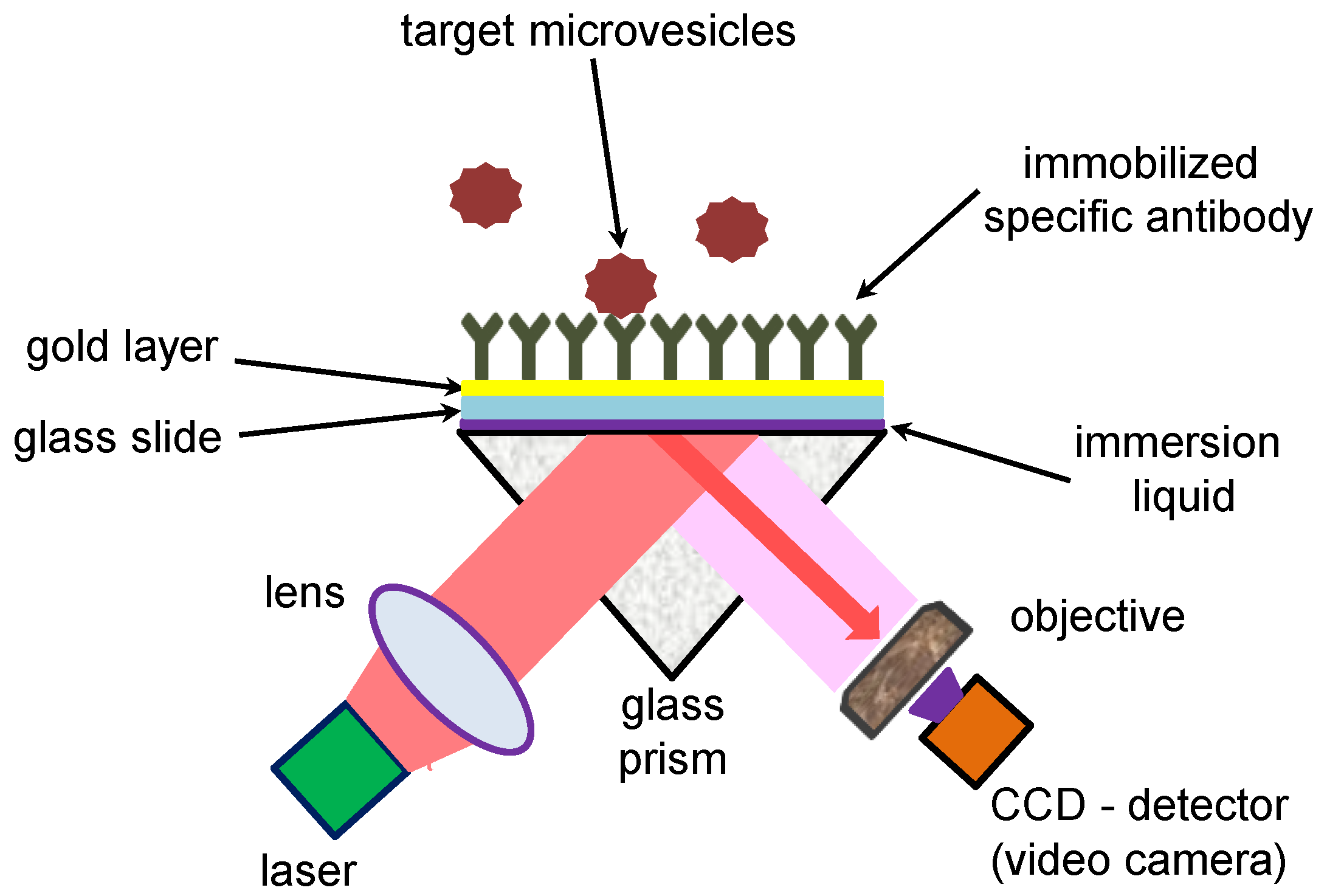
2.1. Surface Plasmon Resonance (SPR) Experimental Setup
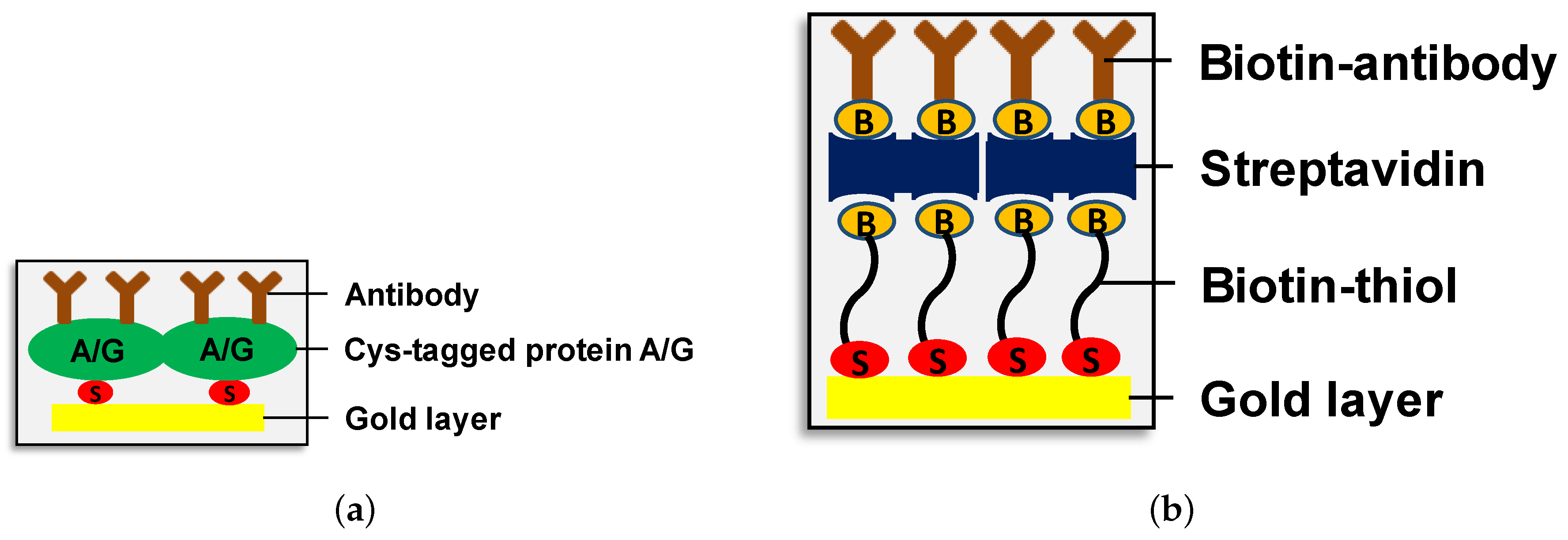
2.2. Chemical and Biological Materials: Preparation of Human Immunodeficiency Virus (HIV) Virus-Like Particles (VLPs) and Microvesicles (MVs) from SH SY5Y Neuroblastoma Cells
2.3. PAMONO Measurements of MVs Derived from SH-SY5Y Cells and Human Immunodeficiency Virus (HIV)-VLPs Produced by HEK293T Cells
2.4. PAMONO Measurements of Particle Size Distribution in the Mixtures of Particles
2.5. The Demonstration of Suitability of Convolutional Neural Networks for Real-Time Estimation of Nano-Particle Size Distributions
3. Results and Discussion
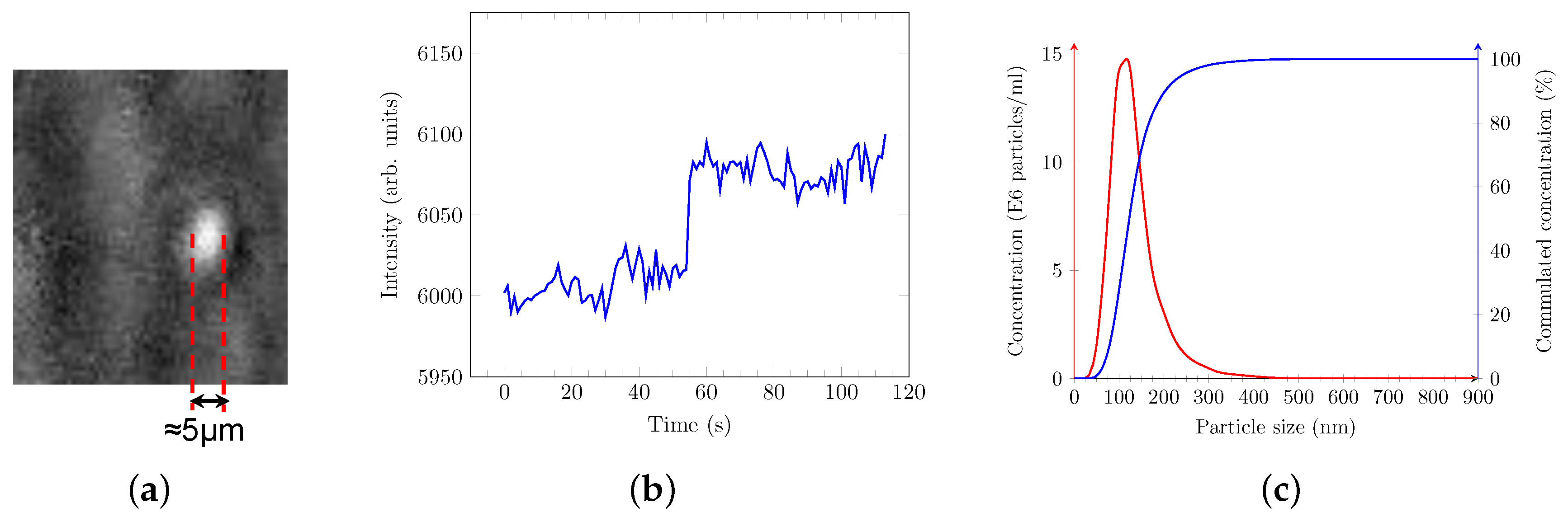
3.1. PAMONO-Sensor Enables Detection of Microvesicles (MVs) Derived from SH SY5Y Cells
3.2. PAMONO-Sensor Measurements Permit to Compare Relative Microvesicle Concentrations in Samples
3.3. Estimation the Specificity of Vesicle Detection by the PAMONO-Sensor
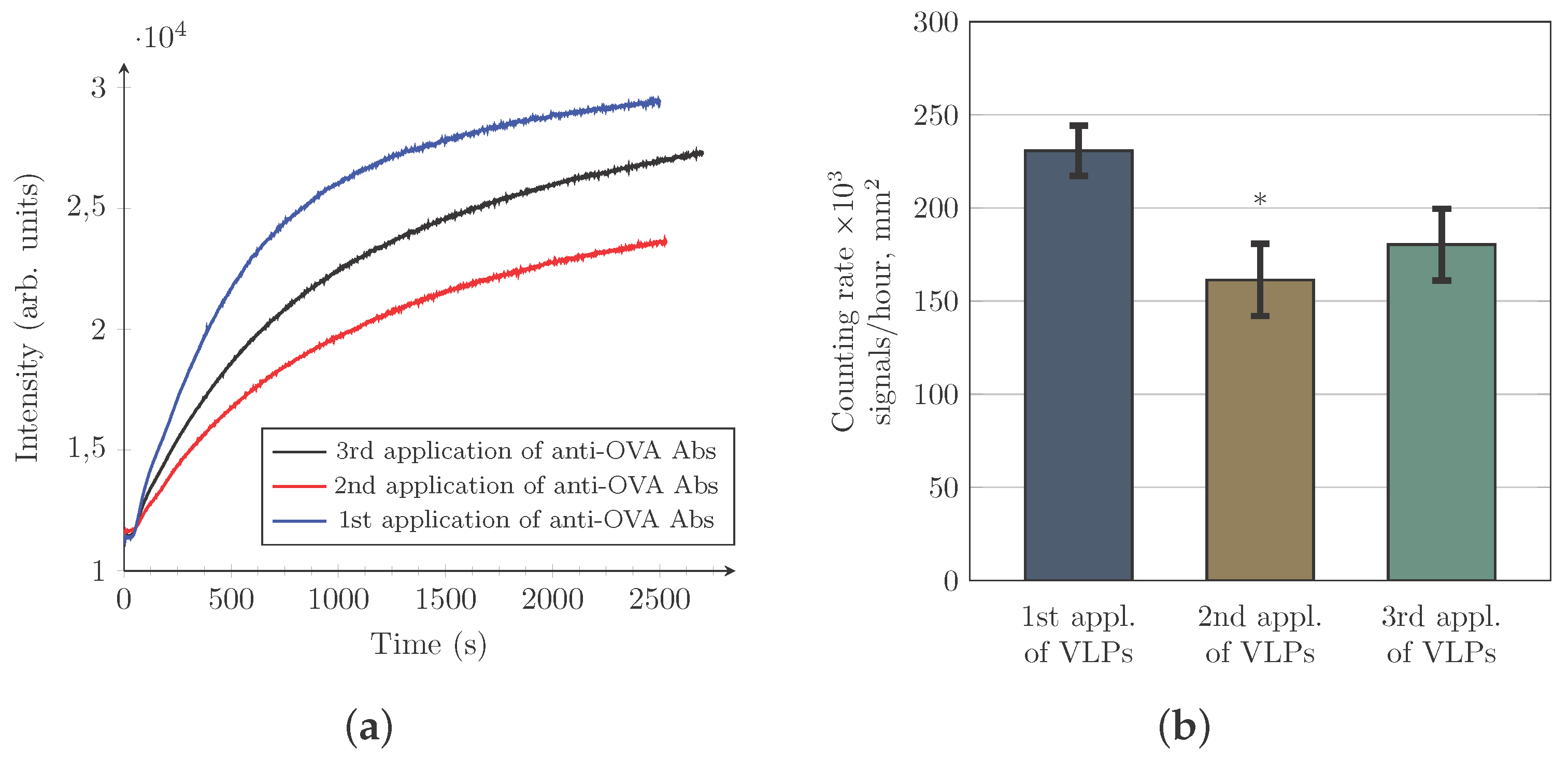
3.4. Evaluation of the Changes in the Counting Rate of HIV-VLPs after Repeated Use of the Same Gold Sensor
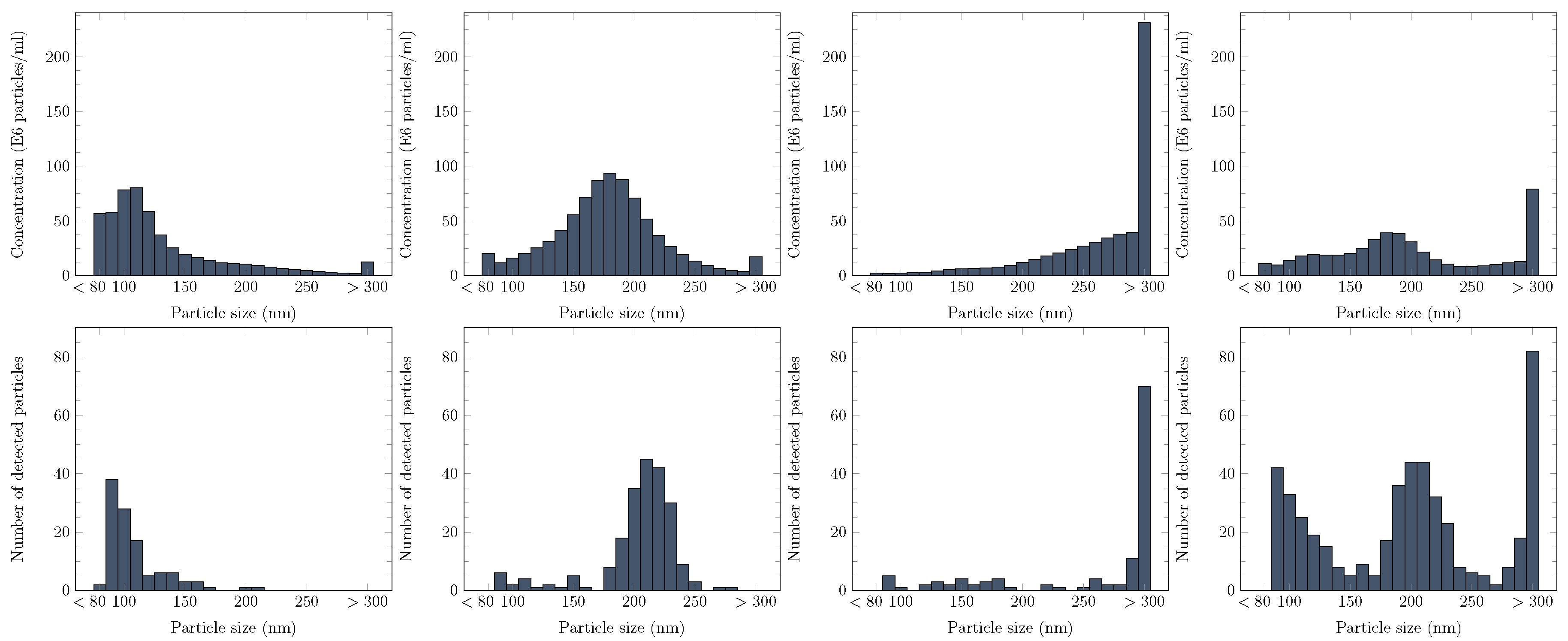
3.5. Monitoring of Particle Size Distribution Using the PAMONO-Sensor
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CNN | convolutional neural networks |
| ELISA | enzyme-linked immunosorbent assay |
| EV | extracellular vesicle |
| FBS | fetal bovine serum |
| FACS | fluorescence-activated cell sorting |
| HIV | human immunodeficiency virus |
| MV | microvesicle |
| NTA | nano-particle tracking analysis |
| OVA | ovalbumin |
| PAMONO | plasmon-assisted microscopy of nano-objects |
| PBS | phosphate-buffered saline |
| SPR | surface plasmon resonance |
| STR | short tandem repeats |
| VLP | virus-like particle |
References
- Zybin, A.; Kuritsyn, Y.A.; Gurevich, E.L.; Temchura, V.V.; Überla, K.; Niemax, K. Real-time Detection of Single Immobilized Nanoparticles by Surface Plasmon Resonance Imaging. Plasmonics 2010, 5, 31–35. [Google Scholar] [CrossRef]
- Gurevich, E.L.; Temchura, V.V.; Überla, K.; Zybin, A. Analytical features of particle counting sensor based on plasmon assisted microscopy of nano objects. Sens. Actuators B Chem. 2011, 160, 1210–1215. [Google Scholar] [CrossRef]
- Nguyen, H.H.; Park, J.; Kang, S.; Kim, M. Surface Plasmon Resonance: A Versatile Technique for Biosensor Applications. Sensors 2015, 15, 10481–10510. [Google Scholar] [CrossRef] [PubMed]
- Abdulhalim, I.; Zourob, M.; Lakhtakia, A. Surface Plasmon Resonance for Biosensing: A Mini-Review. Electromagnetics 2008, 28, 214–242. [Google Scholar] [CrossRef]
- Silvestry, D.; Sonato, A.; Ruffato, G.; Meneghello, A.; Antognoli, A.; Cretaio, E.; Dettin, M.; Zamuner, A.; Casarin, E.; Zacco, G.; et al. A peptide nucleic acid label-free biosensor for Mycobacterium tuberculosis DNA detection via azimuthally controlled grating-coupled SPR. Anal. Methods 2015, 7, 4173–4180. [Google Scholar] [CrossRef]
- Sonato, A.; Agostini, M.; Ruffato, G.; Gazzola, E.; Liuni, D.; Greco, G.; Travagliati, M.; Cecchini, M.; Romanato, F. A surface acoustic wave (SAW)-enhanced grating-coupling phase-interrogation surface plasmon resonance (SPR) microfluidic biosensor. Lab Chip 2016, 16, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.P.; Shan, X.N.; Patel, U.; Huang, X.P.; Lu, J.; Li, J.H.; Tao, N.J. Label-free imaging, detection, and mass measurement of single viruses by surface plasmon resonance. Proc. Natl. Acad. Sci. USA 2010, 107, 16028–16032. [Google Scholar] [CrossRef] [PubMed]
- Halpern, A.R.; Wood, J.B.; Wang, Y.; Corn, R.M. Single-Nanoparticle Near-Infrared Surface Plasmon Resonance Microscopy for Real-Time Measurements of DNA Hybridization Adsorption. ACS Nano 2014, 8, 1022–1030. [Google Scholar] [CrossRef] [PubMed]
- Shpacovitch, V.; Temchura, V.; Matrosovich, M.; Hamacher, J.; Skolnik, J.; Libuschewski, P.; Siedhoff, D.; Weichert, F.; Marvedel, P.; Müller, H.; et al. Application of surface plasmon resonance imaging technique for the detection of single spherical biological submicrometer particles. Anal. Biochem. 2015, 486, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Im, H.; Shao, H.L.; Park, Y.I.; Peterson, V.M.; Castro, C.M.; Weissleder, R.; Lee, H. Label-free detection and molecular profiling of exosomes with a nano-plasmonic sensor. Nat. Biotechnol. 2014, 32, U490–U495. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Wang, K.; Cui, J.; Liu, H.; Bu, X.L.; Ma, H.L.; Wang, W.Z.; Gong, H.; Lausted, C.; Hood, L.; et al. Label-Free Quantitative Detection of Tumor-Derived Exosomes through Surface Plasmon Resonance Imaging. Anal. Chem. 2014, 86, 8857–8864. [Google Scholar] [CrossRef] [PubMed]
- Yanez-Mo, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borras, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 2015, 4. [Google Scholar] [CrossRef] [PubMed]
- Gyorgy, B.; Szabo, T.G.; Pasztoi, M.; Pal, Z.; Misjak, P.; Aradi, B.; Laszlo, V.; Pallinger, E.; Pap, E.; Kittel, A.; et al. Membrane vesicles, current state-of-the-art: emerging role of extracellular vesicles. Cell Mol. Life Sci. 2011, 68, 2667–2688. [Google Scholar] [CrossRef] [PubMed]
- Erdbrugger, U.; Lannigan, J. Analytical challenges of extracellular vesicle detection: A comparison of different techniques. Cytometry A 2016, 89, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Kretschmann, E. Determination of Optical Constants of Metals by Excitation of Surface Plasmons. Z. Phys. 1971, 241, 313–324. [Google Scholar] [CrossRef]
- Zybin, A.; Shpacovitch, V.; Skolnik, J.; Hergenröder, R. Optimal conditions for SPR-imaging of nano-objects. Sens. Actuators B Chem. 2017, 239, 338–342. [Google Scholar] [CrossRef]
- Sidorenko, I.; Nizamov, S.; Hergenröder, R.; Zybin, A.; Kuzmichev, A.; Kiwull, B.; Niessner, R.; Mirsky, V.M. Computer assisted detection and quantification of single adsorbing nanoparticles by differential surface plasmon microscopy. Microchim. Acta 2016, 183, 101–109. [Google Scholar] [CrossRef]
- Siedhoff, D.; Libuschewski, P.; Weichert, F.; Zybin, A.; Marwedel, P.; Müller, H. Modellierung und Optimierung eines Biosensors zur Detektion viralen Strukturen. In Bildverarbeitung für die Medizin. Lecture Notes in Informatics; Springer: Berlin, Germany, 2014; pp. 108–113. (In Germany) [Google Scholar]
- Libuschewski, P.; Siedhoff, D.; Timm, C.; Gelenberg, A.; Weichert, F. Fuzzy-enhanced real-time capable detection of biological viruses using a portable biosensor. In Proceedings of the International Joint Conference on Biomedical Engineering Systems and Technologies (Biosignals), Barcelona, Spain, 11–14 February 2013.
- Gardt, O.; Grewe, B.; Tippler, B.G.; Überla, K.; Temchura, V.V. HIV-derived lentiviral particles promote T-cell independent activation and differentiation of naive cognate conventional B2-cells in vitro. Vaccine 2013, 31, 5088–5098. [Google Scholar] [CrossRef] [PubMed]
- Wagner, R.; Graf, M.; Bieler, K.; Wolf, H.; Grunwald, T.; Foley, P.; Überla, K. Rev-independent expression of synthetic gag-pol genes of human immunodeficiency virus type 1 and simian immunodeficiency virus: Implications for the safety of lentiviral vectors. Hum. Gene Ther. 2000, 11, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Temchura, V.V.; Tenbusch, M.; Nchinda, G.; Nabi, G.; Tippler, B.; Zelenyuk, M.; Wildner, O.; Überla, K.; Kuate, S. Enhancement of immunostimulatory properties of exosomal vaccines by incorporation of fusion-competent G protein of vesicular stomatitis virus. Vaccine 2008, 26, 3662–3672. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.X.; Li, Y.; Kang, C.Y. Budding and secretion of HIV Gag-Env virus-like particles from recombinant human adenovirus infected cells. Virus Res. 2003, 92, 75–82. [Google Scholar] [CrossRef]
- Grgacic, E.V.L.; Anderson, D.A. Virus-like particles: Passport to immune recognition. Methods 2006, 40, 60–65. [Google Scholar] [CrossRef] [PubMed]
- Schramm, A.; Schulte, J.H.; Klein-Hitpass, L.; Havers, W.; Sieverts, H.; Berwanger, B.; Christiansen, H.; Warnat, P.; Brors, B.; Eils, J.; et al. Prediction of clinical outcome and biological characterization of neuroblastoma by expression profiling. Oncogene 2005, 24, 7902–7912. [Google Scholar] [CrossRef] [PubMed]
- Hornik, K. Approximation Capabilities of Multilayer Feedforward Networks. Neural Netw. 1991, 4, 251–257. [Google Scholar] [CrossRef]
- Rosenblatt, F. The Perceptron: A Probabilistic Model for Information Storage and Organization in the Brain. Psychol. Rev. 1958, 65, 386–408. [Google Scholar] [CrossRef] [PubMed]
- Krizhevsky, A.; Sutskever, I.; Hinton, G.E. ImageNet Classification with Deep Convolutional Neural Networks. In Advances in Neural Information Processing Systems 25; Pereira, F., Burges, C.J.C., Bottou, L., Weinberger, K.Q., Eds.; Curran Associates, Inc.: Red Hook, NY, USA, 2012; pp. 1097–1105. [Google Scholar]
- Farabet, C.; Couprie, C.; Najman, L.; LeCun, Y. Learning Hierarchical Features for Scene Labeling. IEEE Trans. Pattern Anal. Mach. Intell. 2013, 35, 1915–1929. [Google Scholar] [CrossRef] [PubMed]
- Lecun, Y.; Bottou, L.; Bengio, Y.; Haffner, P. Gradient-based learning applied to document recognition. Proc. IEEE 1998, 86, 2278–2324. [Google Scholar] [CrossRef]
- Rumelhart, D.E.; Hinton, G.E.; Williams, R.J. Learning representations by back-propagating errors. Nature 1986, 323, 533–536. [Google Scholar] [CrossRef]
- Lee, J.M.; Park, H.K.; Jung, Y.; Kim, J.K.; Jung, S.O.; Chung, B.H. Direct immobilization of protein G variants with various numbers of cysteine residues on a gold surface. Anal. Chem. 2007, 79, 2680–2687. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Erviti, L.; Seow, Y.; Schapira, A.H.; Gardiner, C.; Sargent, I.L.; Wood, M.J.A.; Cooper, J.M. Lysosomal dysfunction increases exosome-mediated alpha-synuclein release and transmission. Neurobiol. Dis. 2011, 42, 360–367. [Google Scholar] [CrossRef] [PubMed]
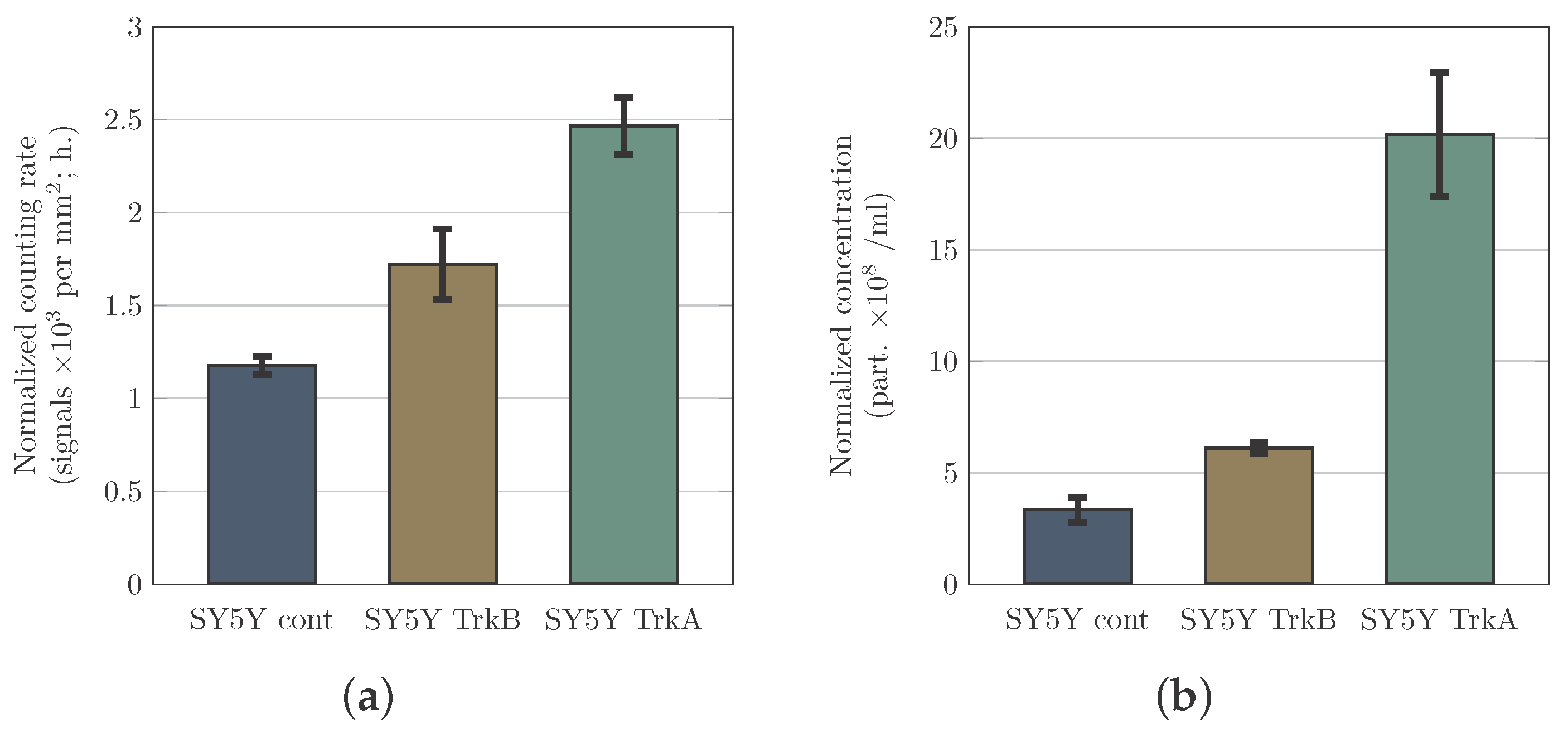
| Protein A/G | Biotin-Thiol | |
|---|---|---|
| Ratio: VLPs (virus-like particles) with OVA (ovalbumin) to VLPs without OVA (normalized counting rates were used) | 21 | |
| Specificity (%) | 88–94 | 95 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shpacovitch, V.; Sidorenko, I.; Lenssen, J.E.; Temchura, V.; Weichert, F.; Müller, H.; Überla, K.; Zybin, A.; Schramm, A.; Hergenröder, R. Application of the PAMONO-Sensor for Quantification of Microvesicles and Determination of Nano-Particle Size Distribution. Sensors 2017, 17, 244. https://doi.org/10.3390/s17020244
Shpacovitch V, Sidorenko I, Lenssen JE, Temchura V, Weichert F, Müller H, Überla K, Zybin A, Schramm A, Hergenröder R. Application of the PAMONO-Sensor for Quantification of Microvesicles and Determination of Nano-Particle Size Distribution. Sensors. 2017; 17(2):244. https://doi.org/10.3390/s17020244
Chicago/Turabian StyleShpacovitch, Victoria, Irina Sidorenko, Jan Eric Lenssen, Vladimir Temchura, Frank Weichert, Heinrich Müller, Klaus Überla, Alexander Zybin, Alexander Schramm, and Roland Hergenröder. 2017. "Application of the PAMONO-Sensor for Quantification of Microvesicles and Determination of Nano-Particle Size Distribution" Sensors 17, no. 2: 244. https://doi.org/10.3390/s17020244
APA StyleShpacovitch, V., Sidorenko, I., Lenssen, J. E., Temchura, V., Weichert, F., Müller, H., Überla, K., Zybin, A., Schramm, A., & Hergenröder, R. (2017). Application of the PAMONO-Sensor for Quantification of Microvesicles and Determination of Nano-Particle Size Distribution. Sensors, 17(2), 244. https://doi.org/10.3390/s17020244








