Potential Applications and Limitations of Electronic Nose Devices for Plant Disease Diagnosis
Abstract
1. Introduction
2. Electronic Nose Plant Diagnostic Applications
3. Gas Sampling Techniques
4. Training, Data Elaboration, and Decision-Making
5. Effects of Disease Progression, Severity, and Incidence
6. Signal Drift Effects and Sample Humidity Control
7. Conclusions
Conflicts of Interest
References
- Martinelli, F.; Scalenghe, R.; Davino, S.; Panno, S.; Scuderi, G.; Ruisi, P.; Villa, P.; Stroppiana, D.; Boschetti, M.; Goulart, L.R.; et al. Advanced methods of plant disease detection. A review. Agron. Sustain. Dev. 2015, 35, 1–25. [Google Scholar] [CrossRef]
- Sankaran, S.; Mishra, A.; Ehsani, R.; Davis, C. A review of advanced techniques for detecting plant diseases. Comput. Electron. Agric. 2010, 72, 1–13. [Google Scholar] [CrossRef]
- Ray, M.; Ray, A.; Dash, S.; Mishra, A.; Achary, K.G.; Nayak, S.; Singh, S. Fungal disease detection in plants: Traditional assays, novel diagnostic techniques and biosensors. Biosens. Bioelectron. 2017, 87, 708–723. [Google Scholar] [CrossRef] [PubMed]
- Markom, M.A.; Shakaff, A.Y.M.; Adom, A.H.; Ahmad, M.N.; Hidayat, W.; Abdullah, A.H.; Fikri, N.A. Intelligent electronic nose system for basal stem rot disease detection. Comput. Electron. Agric. 2009, 66, 140–146. [Google Scholar] [CrossRef]
- Rizzolo, A.; Bianchi, G.; Lucido, P.; Cangelosi, B.; Pozzi, L.; Villa, G.; Clematis, F.; Pasini, C.; Curir, P. Electronic nose for the early detection of red palm weevil (Rhynchophorus ferrugineous Olivier) infestation in palms: Preliminary results. Acta Hortic. 2015, 1099, 347–355. [Google Scholar] [CrossRef]
- EPPO Website. Available online: http://archives.eppo.int/EPPOStandards/diagnostics.htm (accessed on 26 September 2017).
- Dudareva, N.; Negre, F.; Nagegowda, D.A.; Orlova, I. Plant volatiles: Recent advances and future perspectives. Crit. Rev. Plant Sci. 2006, 25, 417–440. [Google Scholar] [CrossRef]
- Spinelli, F.; Noferini, M.; Costa, G. Near Infrared spectroscopy (NIRS): Perspective of fire blight detection in asymptomatic plant material. Acta Hortic. 2006, 704, 87–91. [Google Scholar] [CrossRef]
- Spinelli, F.; Costa, G.; Rondelli, E.; Vanneste, J.L.; Rodriguez Estrada, M.T.; Busi, S.; Savioli, S.; Cristescu, S. Volatile compounds produced by Erwinia amylovora and their potential exploitation for bacterial identification. Acta Hortic. 2011, 896, 77–84. [Google Scholar] [CrossRef]
- Blasioli, S.; Biondi, E.; Braschi, I.; Mazzucchi, U.; Bazzi, C.; Gessa, C.E. Electronic nose as an innovative tool for the diagnosis of grapevine crown gall. Anal. Chim. Acta 2010, 672, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.D.; Lester, D.G.; Oberle, C.S. Development of conductive polymer analysis for the rapid detection and identification of phytopathogenic microbes. Phytopathology 2004, 94, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Baietto, M.; Wilson, A.D.; Bassi, D.; Ferrini, F. Evaluation of three electronic noses for detecting incipient wood decay. Sensors 2010, 10, 1062–1092. [Google Scholar] [CrossRef] [PubMed]
- Baietto, M.; Pozzi, L.; Wilson, A.D.; Bassi, D. Evaluation of a portable MOS electronic nose to detect root rots in shade tree species. Comput. Electron. Agric. 2013, 96, 117–125. [Google Scholar] [CrossRef]
- Vikram, A.; Prithiviraj, B.; Hamzehzarghani, H.; Kushalappa, A.C. Volatile metabolite profiling to discriminate diseases of McIntosh apple inoculated with fungal pathogens. J. Sci. Food Agric. 2004, 84, 1333–1340. [Google Scholar] [CrossRef]
- Prithiviraj, B.; Vikram, A.; Kushalappa, A.C.; Yaylayan, V. Volatile metabolite profiling for the discrimination of onion bulbs infected by Erwinia carotovora ssp. carotovora, Fusarium oxysporum and Botrytis allii. Eur. J. Plant Pathol. 2004, 110, 371–377. [Google Scholar] [CrossRef]
- De Lacy Costello, B.P.J.; Evans, P.; Ewen, R.J.; Gunson, H.E.; Jones, P.R.H.; Ratcliffe, N.M.; Spencer-Phillips, P.T.N. Gas chromatography-mass spectrometry analyses of volatile organic compounds from potato tubers inoculated with Phytophthora infestans or Fusarium coeruleum. Plant Pathol. 2001, 50, 489–496. [Google Scholar] [CrossRef]
- Kushalappa, A.C.; Lui, L.H.; Chen, C.R.; Lee, B. Volatile fingerprinting (SPME-GC FID) to detect and discriminate diseases of potato tubers. Plant Dis. 2002, 86, 131–137. [Google Scholar] [CrossRef]
- Biondi, E.; Blasioli, S.; Galeone, A.; Spinelli, F.; Cellini, A.; Lucchese, C.; Braschi, I. Detection of potato brown rot and ring rot by electronic nose: From laboratory to real scale. Talanta 2014, 129, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Blasioli, S.; Biondi, E.; Samudrala, D.; Spinelli, F.; Cellini, A.; Bertaccini, A.; Cristescu, S.M.; Braschi, I. Identification of volatile markers in potato brown rot and ring rot by combined GC-MS and PTR-MS techniques: Study on in vitro and in vivo samples. J. Agric. Food Chem. 2014, 62, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Cellini, A.; Biondi, E.; Blasioli, S.; Rocchi, L.; Farneti, B.; Braschi, I.; Savioli, S.; Rodriguez-Estrada, M.T.; Biasioli, F.; Spinelli, F. Early detection of bacterial diseases in apple plants by analysis of volatile organic compounds profiles and use of electronic nose. Ann. Appl. Biol. 2016, 168, 409–420. [Google Scholar] [CrossRef]
- Prior, P.; Ailloud, F.; Dalsing, B.L.; Remenant, B.; Sanchez, B.; Allen, C. Genomic and proteomic evidence supporting the division of the plant pathogen Ralstonia solanacearum into three species. BMC Genom. 2016, 17, 90. [Google Scholar] [CrossRef] [PubMed]
- Cellini, A.; Biondi, E.; Buriani, G.; Farneti, B.; Rodriguez-Estrada, M.T.; Braschi, I.; Savioli, S.; Blasioli, S.; Rocchi, L.; Biasioli, F.; et al. Characterization of volatile organic compounds emitted by kiwifruit plants infected with Pseudomonas syringae pv. actinidiae and their effects on host defences. Trees 2016, 30, 795–806. [Google Scholar] [CrossRef]
- Pardo, M.; Sberveglieri, G. Electronic olfactory systems based on metal oxide semiconductor sensor arrays. MRS Bull. 2004, 29, 703–708. [Google Scholar] [CrossRef]
- Baietto, M.; Wilson, A.D. Electronic-nose applications for fruit identification, ripeness and quality grading. Sensors 2015, 15, 899–931. [Google Scholar] [CrossRef] [PubMed]
- Loutfi, A.; Coradeschi, S.; Mani, G.K.; Shankar, P.; Rayappan, J.B.B. Electronic noses for food quality: A review. J. Food Eng. 2015, 144, 103–111. [Google Scholar] [CrossRef]
- Peris, M.; Escuder-Gilabert, L. A 21st century technique for food control: Electronic noses. Anal. Chim. Acta 2009, 638, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.D.; Baietto, M. Applications and advances in electronic-nose technologies. Sensors 2009, 9, 5099–5148. [Google Scholar] [CrossRef] [PubMed]
- Biondi, E.; Blasioli, S.; Fantini, M.; Braschi, I.; Bertaccini, A. Grapevine crown gall detection by electronic nose. In Proceedings of the 16th International Symposium on Olfaction and Electronic Noses, Dijon, France, 28 June–1 July 2015. [Google Scholar]
- Eifler, J.; Martinelli, E.; Santonico, M.; Capuano, R.; Schild, D.; Di Natale, C. Differential detection of potentially hazardous Fusarium species in wheat grains by an electronic nose. PLoS ONE 2011, 6, e21026. [Google Scholar] [CrossRef] [PubMed]
- Henderson, W.G.; Khalilian, A.; Han, Y.J.; Greene, J.K.; Degenhardt, D.C. Detecting stink bugs/damage in cotton utilizing a portable electronic nose. Comput. Electron. Agric. 2010, 70, 157–162. [Google Scholar] [CrossRef]
- Laothawornkitkul, J.; Moore, J.P.; Taylor, J.E.; Possell, M.; Gibson, T.M.; Hewitt, C.N.; Paul, N.D. Discrimination of plant volatile signatures by an electronic nose: A potential technology for plant pest and disease monitoring. Environ. Sci. Technol. 2008, 42, 8433–8439. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Krewer, G.W.; Ji, P.; Scherm, H.; Kays, S.J. Gas sensor array for blueberry fruit disease detection and classification. Postharvest Biol. Technol. 2010, 55, 144–149. [Google Scholar] [CrossRef]
- Pallottino, F.; Costa, C.; Antonucci, F.; Strano, M.C.; Calandra, M.; Solaini, S.; Menesatti, P. Electronic nose application for determination of Penicillium digitatum in Valencia oranges. J. Sci. Food Agric. 2012, 92, 2008–2012. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Zhang, W.; Zhu, N.; Mao, S.; Tu, K. Early detection and classification of pathogenic fungal disease in post-harvest strawberry fruit by electronic nose and gas chromatography–mass spectrometry. Food Res. Int. 2014, 62, 162–168. [Google Scholar] [CrossRef]
- Prak Chang, K.P.; Zakaria, A.; Abdul Nasir, A.S.; Yusuf, N.; Thriumani, R.; Shakaff, A.Y.M.; Adorn, A.H. Analysis and feasibility study of plant disease using e-nose. In Proceedings of the IEEE International Conference on Control System, Computing and Engineering, Penang, Malaysia, 28–30 November 2014. [Google Scholar]
- Spinelli, F.; Noferini, M.; Vanneste, J.L.; Costa, G. Potential of the electronic-nose for the diagnosis of bacterial and fungal diseases in fruit trees. Bull. OEPP/EPPO Bull. 2010, 40, 59–67. [Google Scholar] [CrossRef]
- Spinelli, F.; Cellini, A.; Vanneste, J.L.; Rodriguez-Estrada, M.T.; Costa, G.; Savioli, S.; Harren, F.J.M.; Cristescu, S.M. Emission of volatile compounds by Erwinia amylovora biological activity in vitro and possible exploitation for bacterial identification. Trees 2012, 26, 141–152. [Google Scholar] [CrossRef]
- Stinson, J.A.; Persaud, K.C.; Bryning, G. Generic system for the detection of statutory potato pathogens. Sens. Actuators B Chem. 2006, 116, 100–106. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Cheng, S. Predicting Attacked Time of Tomato Seedling by E-nose based on Kernel Principal Component Analysis. In Proceedings of the ASABE Annual International Meeting, Orlando, FL, USA, 17–20 July 2016. [Google Scholar]
- Xu, S.; Zhou, Z.; Lu, H.; Luo, X.; Lan, Y.; Zhang, Y.; Li, Y. Estimation of the age and amount of brown rice plant hoppers based on bionic electronic nose use. Sensors 2014, 14, 18114–18130. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, J. Detection of age and insect damage incurred by wheat, with an electronic nose. J. Stored Prod. Res. 2007, 43, 489–495. [Google Scholar] [CrossRef]
- Zhou, B.; Wang, J. Use of electronic nose technology for identifying rice infestation by Nilaparvata lugens. Sens. Actuators B Chem. 2011, 160, 15–21. [Google Scholar] [CrossRef]
- Ghaffari, R.; Laothawornkitkul, J.; Zhang, F.; Iliescu, D.; Hines, E.; Leeson, M.; Napier, R.; More, J.P.; Tayloer, J.E.; Possell, M.; et al. Plant pest and disease diagnosis: Electronic nose and support vector machine approach. J. Plant Dis. Protect. 2012, 119, 200–207. [Google Scholar] [CrossRef]
- Dettmer, K.; Engewald, W. Adsorbent materials commonly used in air analysis for adsorptive enrichment and thermal desorption of volatile organic compounds. Anal. Bioanal. Chem. 2002, 373, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Gallego, E.; Roca, F.J.; Perales, J.F.; Guardino, X. Comparative study of the adsorption performance of a multi-sorbent bed (Carbotrap, Carbopack X, Carboxen 569) and a Tenax TA adsorbent tube for the analysis of volatile organic compounds (VOCs). Talanta 2010, 81, 916–924. [Google Scholar] [CrossRef] [PubMed]
- Harper, M. Sorbent trapping of volatile organic compounds from air. J. Chromatogr. A 2000, 885, 129–151. [Google Scholar] [CrossRef]
- Li, G.; Fu, J.; Zhang, J.; Zheng, J. Progress in bionic information processing techniques for an electronic nose based on olfactory models. Chin. Sci. Bull. 2009, 54, 521–534. [Google Scholar] [CrossRef]
- Wilson, A.D. Diverse applications of electronic-nose technologies in agriculture and forestry. Sensors 2013, 13, 2295–2348. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, N.; Khan, N.A.; Ferrante, A.; Trivellini, A.; Francini, A.; Khan, M.I.R. Ethylene role in plant growth, development and senescence: Interaction with other phytohormones. Front. Plant Sci. 2017, 8, 475. [Google Scholar] [CrossRef] [PubMed]
- Von Dahl, C.C.; Baldwin, I.T. Deciphering the role of ethylene in plant–herbivore interactions. J. Plant Growth Regul. 2007, 26, 201–209. [Google Scholar] [CrossRef]
- Niinemets, U.; Loreto, F.; Reichstein, M. Physiological and physicochemical controls on foliar volatile organic compound emissions. Trends Plant Sci. 2004, 9, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Tothill, I.E. Biosensors developments and potential applications in the agricultural diagnosis sector. Comput. Electron. Agric. 2001, 30, 205–218. [Google Scholar] [CrossRef]
- Williams, D.E. Semiconducting oxides as gas-sensitive resistors. Sens. Actuators B Chem. 1999, 57, 1–16. [Google Scholar] [CrossRef]
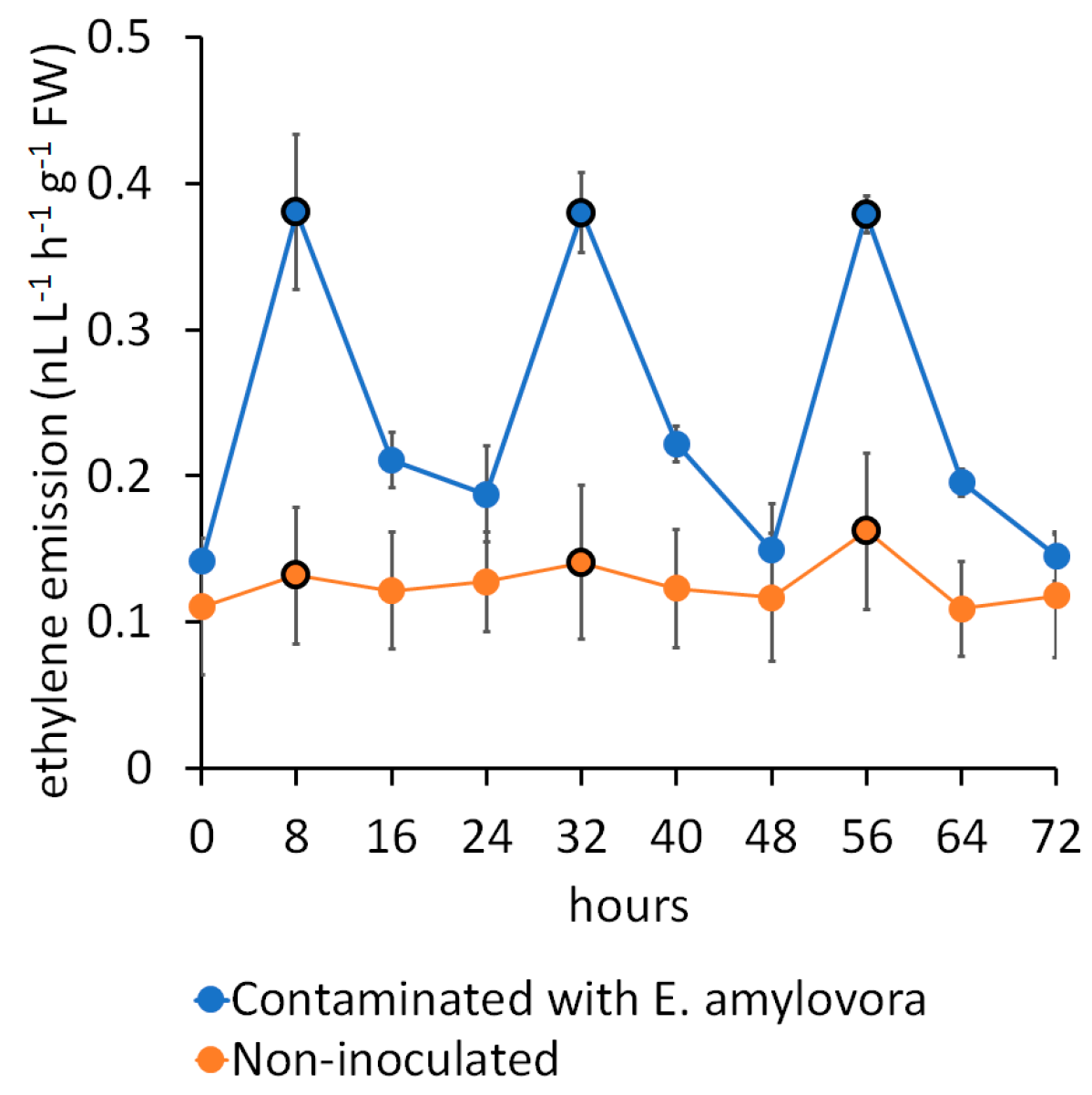
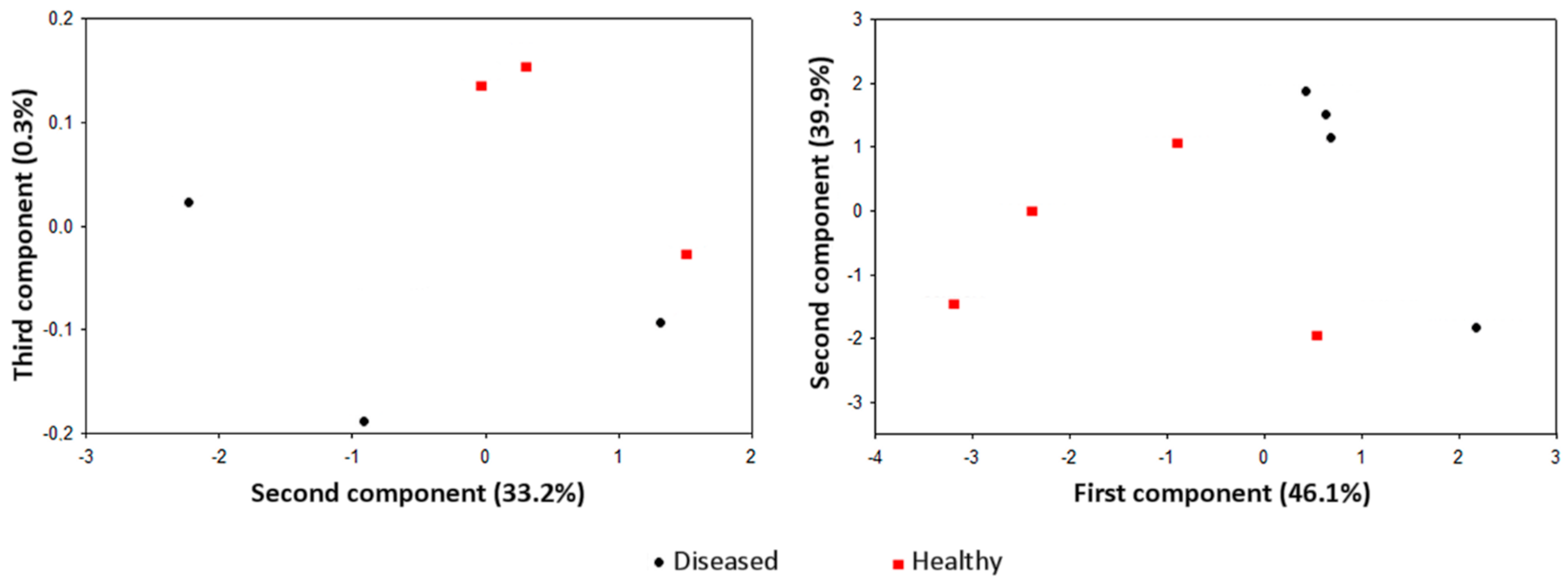
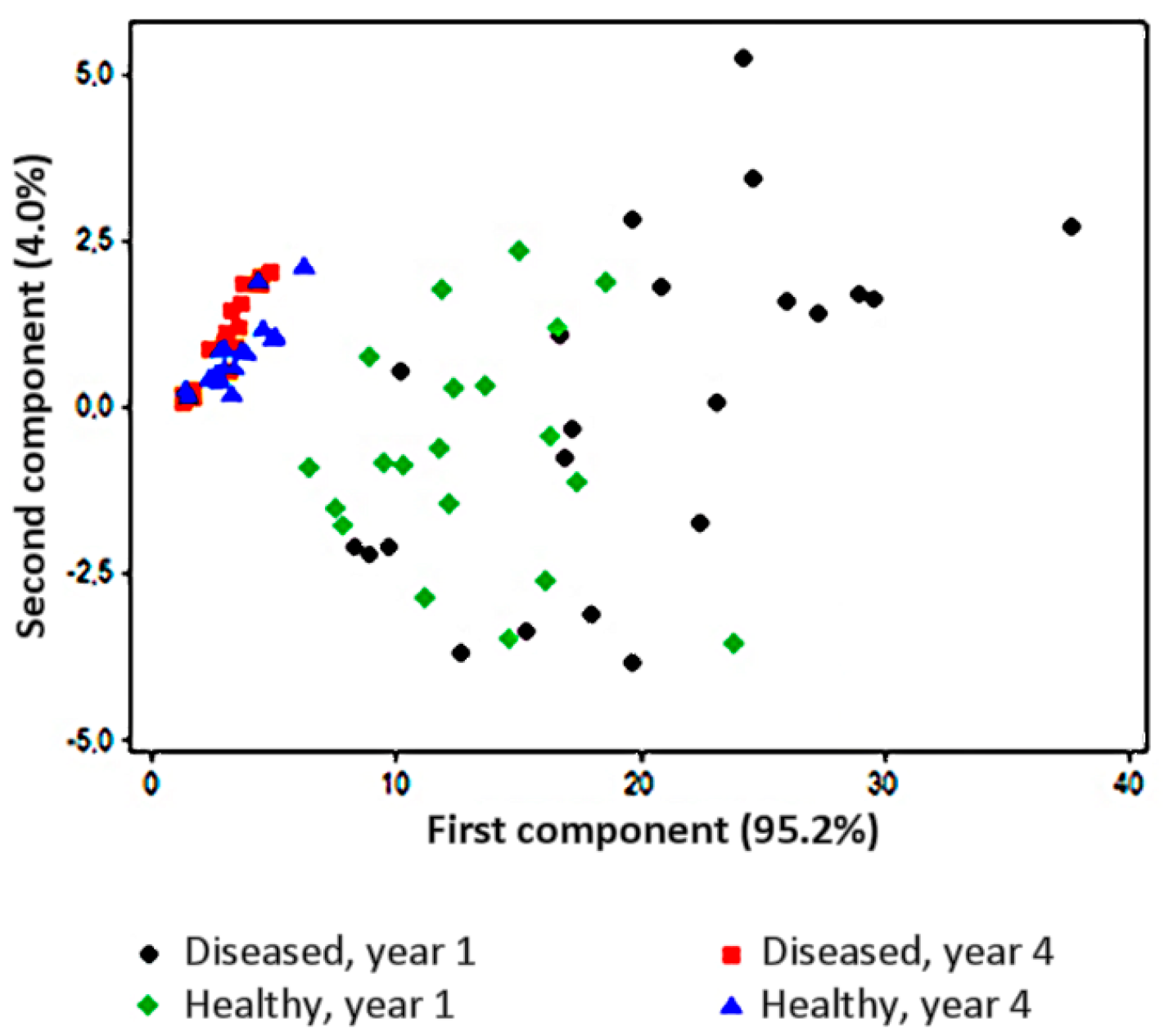
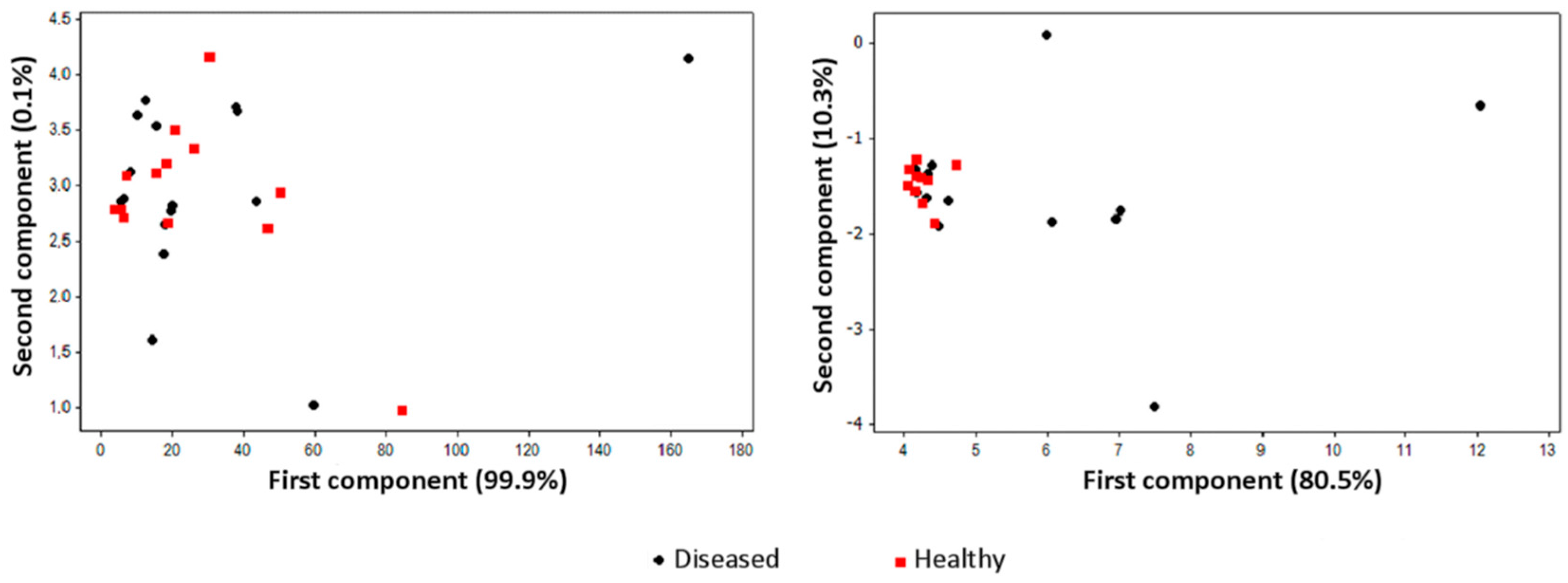

| Pathosystem and Plant Material | Analytical and Technical Solutions | E-Nose Model(s) |
|---|---|---|
| Multiple wood decay fungi on wood samples of several tree species [12] | Static headspace analysis Multiple-class recognition | AromaScan A32 SLibraNose 2.1 PEN3 |
| Multiple root decay fungi on root segments of several shade tree species [13] | Static headspace analysis Multiple-class recognition | PEN3 |
| Agrobacterium vitis on grapevine rootstock cuts [10] | Static headspace analysis | PEN3 |
| Ralstonia solanacearum and Clavibacter michiganensis ssp. sepedonicus on potato tubers [18] | Lab to real scale VOCs concentration on adsorbents Sample air desiccation with SiO2 | PEN3 |
| Agrobacterium vitis on grapevine rootstock cuts [28] | Static headspace analysis Evaluation of sensor drift | PEN3 |
| Erwinia amylovora and Pseudomonas syringae pv. syringae on in vitro and dormant apple plants [20] | Lab to real scale VOCs concentration on adsorbents Dilution effects Three-class recognition | EOS507C PEN3 |
| Pseudomonas syringae pv. actinidiae on in vitro kiwifruit plants [22] | Static headspace analysis at high RH | EOS507C PEN3 |
| Fusarium spp. on wheat grain [29] | Static headspace analysis Air sample filtration on CaCO3 | Prototype |
| Anthonomus grandis grandis on cotton bolls [30] | VOCs concentration on adsorbents Exclusion of water-sensitive sensors Dilution effects | Cyranose 320 |
| Multiple pests on cucumber, pepper and tomato plants [31] | VOCs concentration on adsorbents | Bloodhound ST214 |
| Botrytis cinerea, Colletotrichum gloeosporioides and Alternaria sp. on blueberry fruit [32] | Static headspace analysis Four-class recognition | Cyranose 320 |
| Aspergillus, Fusarium and Penicillium spp. on oil palm [3] | Real scale | Cyranose 320 |
| Penicilium spp. on orange fruit [33] | Static headspace analysis Dilution effects | LibraNose 2.1 |
| Botrytis, Fusarium and Penicillium spp. on strawberry fruit [34] | Static headspace analysis External control of T and RH | PEN3 |
| Multiple bacterial pathogens [35] | Static headspace analysis | PEN3 |
| Rhynchophorus ferrugineous on ornamental palm [5] | 8-day time course | PEN3 |
| Erwinia amylovora on apple and pear plants; Botrytis cinerea and Sclerotinia sclerotiorum on kiwi fruit [8,36] | Static headspace analysis RH raised to 100% | EOS835 |
| Multiple bacterial pathogens [37] | Static headspace analysis | EOS507C |
| Ralstonia solanacearum and Clavibacter michiganensis ssp. sepedonicus on potato tubers [38] | VOCs concentration on adsorbents Three-class recognition | Prototype |
| Botrytis cinerea on tomato plants [39] | Static headspace analysis Severity effects | PEN2 |
| Multiple fungi and bacteria; Ceratocystis fagacearum on oak sapwood [11] | External control of RH | Aromascan A32S |
| Nilaparvata lugens [40] | Static headspace analysis | PEN2 |
| Rhyzopertha dominica on wheat grain [41] | Static headspace analysis Dilution and time effects | PEN2 |
| Nilaparvata lugens on rice plants [42] | Static headspace analysis Dilution effects | PEN2 |
| Unclassified spider mites on cucumber; powdery mildew on tomato [43] | Static headspace analysis External control of T and RH | Bloodhound ST214 |
| E-Nose Model | Characteristics | Producer |
|---|---|---|
| AromaScan A32S | organic matrix-coated polymer-type 32-sensor array | Osmetech Inc., Wobum, MA, USA |
| LibraNose 2.1 | quartz crystal microbalance 8-sensor array | Technobiochip, Pozzuoli, Italy |
| PEN2 | Array of 10 metal-oxide semiconductor sensors (obsolete) | Airsense Analytics, Schwering, Germany |
| PEN3 | Array of 10 metal-oxide semiconductor sensors, accumulation unit (EDU3) | Airsense Analytics, Schwering, Germany |
| EOS835 | Array of 6 metal-oxide semiconductor sensors (obsolete) | Sacmi Scrl, Imola, Italy |
| EOS507C | Array of 6 metal-oxide semiconductor sensors, RH compensation system | Sacmi Scrl, Imola, Italy |
| Cyranose 320 | Thin-film carbon-black polymer composite 32-sensor array | Smiths Detection Inc., Pasadena, CA, USA |
| Bloodhound ST214 | 14 organic polymer sensors (obsolete) | Scensive Technologies Ltd., Normanton, UK |
| VOCs-Sorbent Material | Ralstonia solanacearum | Clavibacter michiganensis ssp. sepedonicus | ||
|---|---|---|---|---|
| Symptomatic Tubers (%) | Discrimination Power * (%) | Symptomatic Tubers (%) | Discrimination Power * (%) | |
| Tenax-TA | 57 | 8 | 20 | 46 |
| Carbotrap | 67 | 23 | 0 | 25 |
| Tenax-GR | 57 | 0.5 | n.a. | 3 |
| Carboxen | 80 | 18 | n.a. | 46 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cellini, A.; Blasioli, S.; Biondi, E.; Bertaccini, A.; Braschi, I.; Spinelli, F. Potential Applications and Limitations of Electronic Nose Devices for Plant Disease Diagnosis. Sensors 2017, 17, 2596. https://doi.org/10.3390/s17112596
Cellini A, Blasioli S, Biondi E, Bertaccini A, Braschi I, Spinelli F. Potential Applications and Limitations of Electronic Nose Devices for Plant Disease Diagnosis. Sensors. 2017; 17(11):2596. https://doi.org/10.3390/s17112596
Chicago/Turabian StyleCellini, Antonio, Sonia Blasioli, Enrico Biondi, Assunta Bertaccini, Ilaria Braschi, and Francesco Spinelli. 2017. "Potential Applications and Limitations of Electronic Nose Devices for Plant Disease Diagnosis" Sensors 17, no. 11: 2596. https://doi.org/10.3390/s17112596
APA StyleCellini, A., Blasioli, S., Biondi, E., Bertaccini, A., Braschi, I., & Spinelli, F. (2017). Potential Applications and Limitations of Electronic Nose Devices for Plant Disease Diagnosis. Sensors, 17(11), 2596. https://doi.org/10.3390/s17112596








