Microbial Fuels Cell-Based Biosensor for Toxicity Detection: A Review
Abstract
1. Introduction
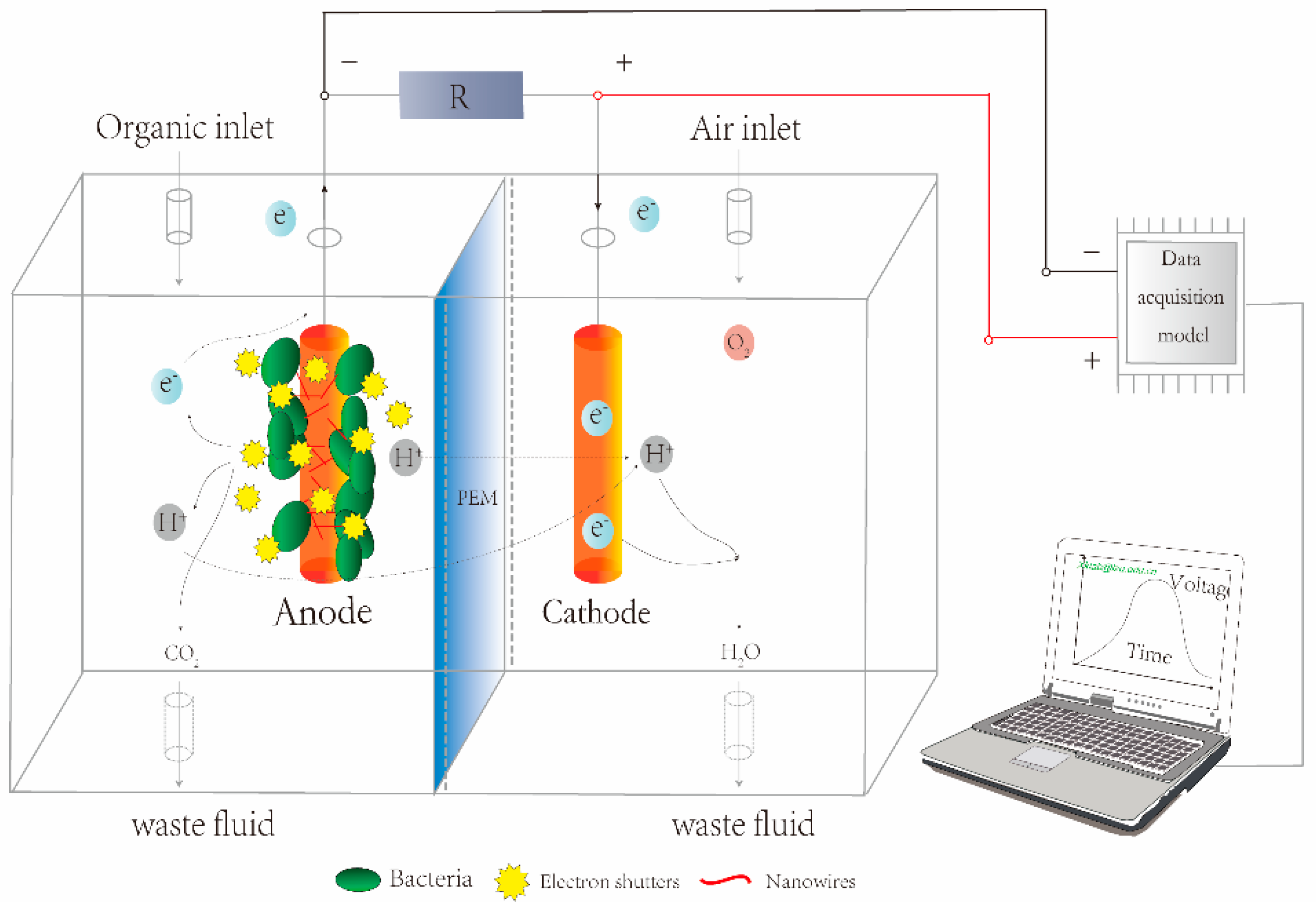
2. The Mechanisms Governing MFCs Used as Biosensors
3. Analytical Applications of Microbial Fuel Cell-Based Biosensors
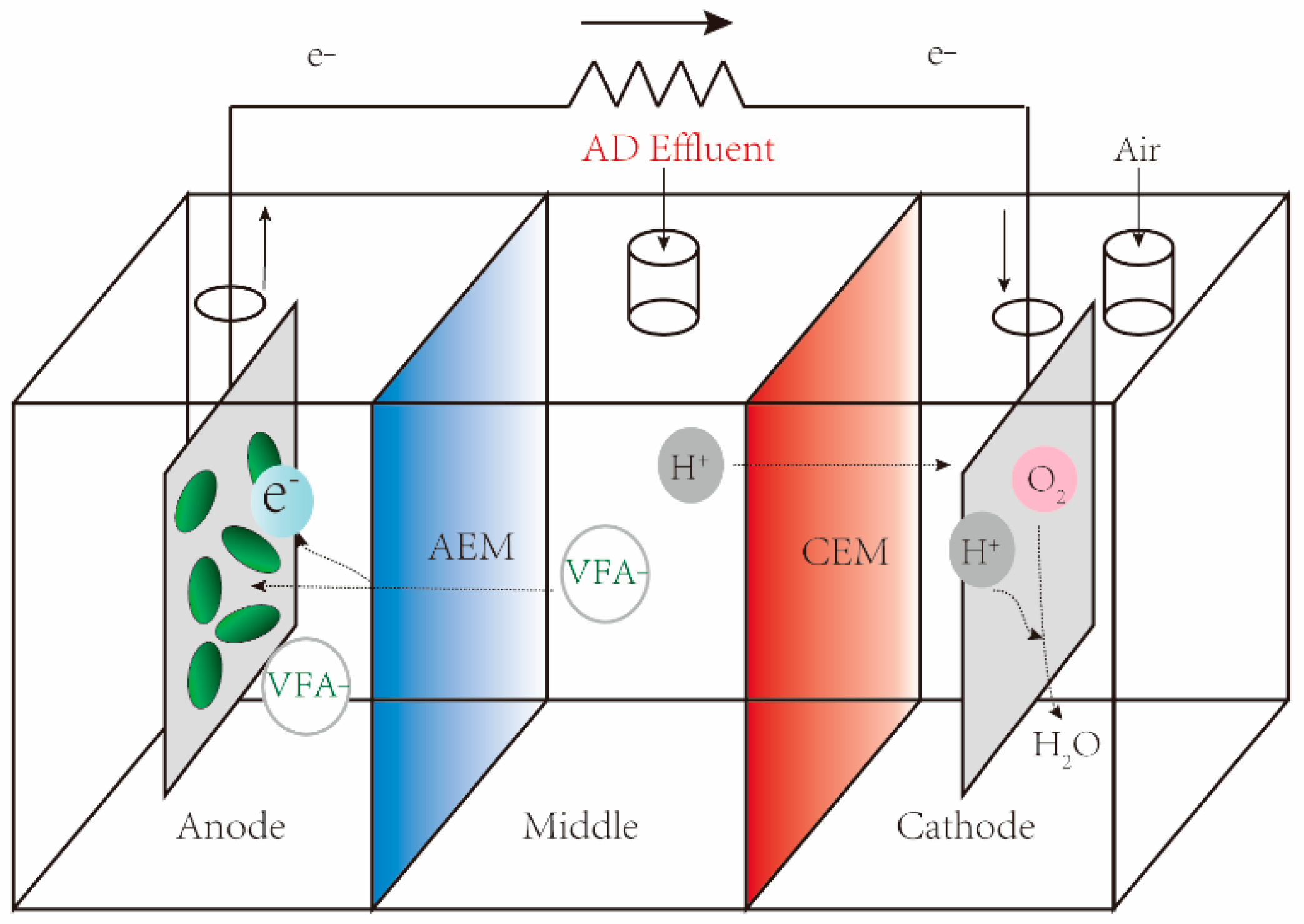
3.1. MFC as VFA Biosensor
3.2. MFC as BOD Biosensors
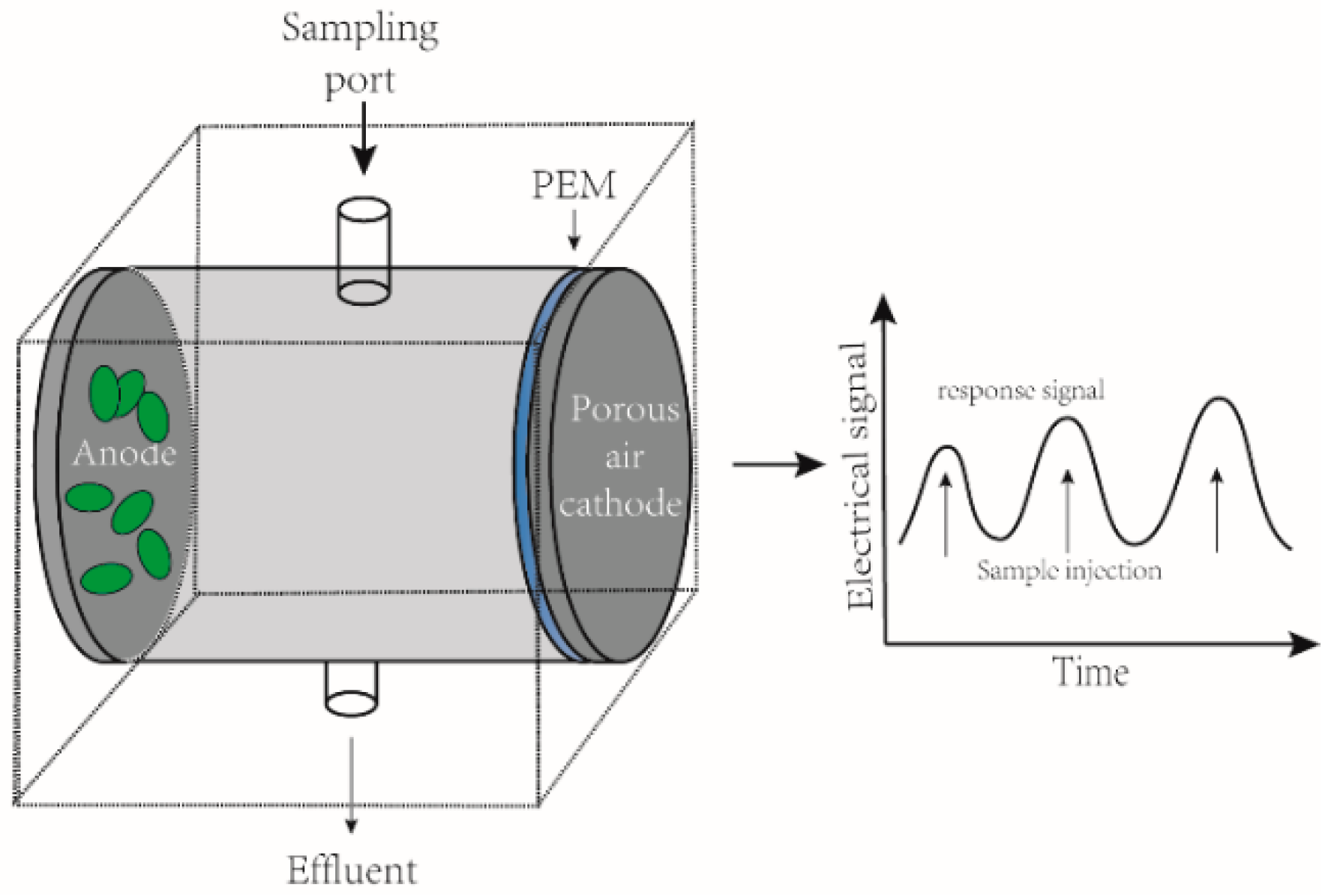
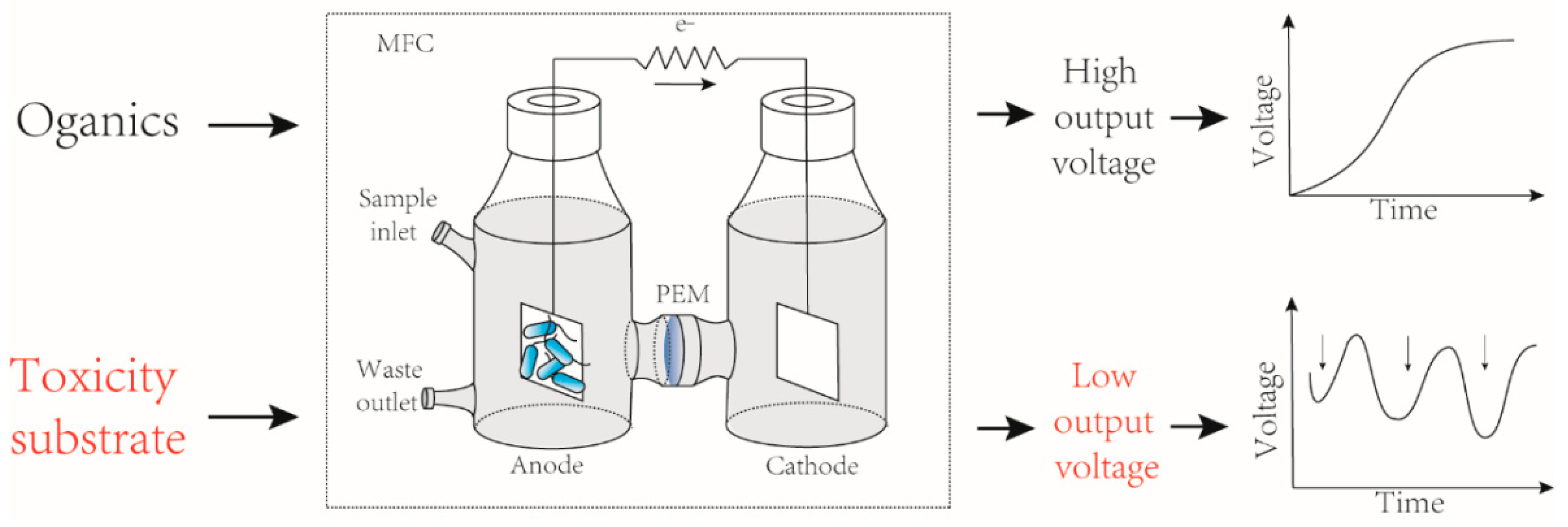
3.3. MFC as Toxicants Biosensors
3.3.1. MFCs as Heavy Metal Biosensors
3.3.2. MFCs as Organic Toxin Biosensors
3.4. Comparison of Different Biosensors
4. The Performance of MFC-Based Biosensors
4.1. Factors That Influence MFC-Based Biosensors
4.2. Performance Improvement of MFC-Based Biosensors
4.3. Modification of the MFC-Based Biosensors Model
5. Challenges and Future Prospects
6. Conclusions
Acknowledgments
Author contribution
Conflicts of Interest
References
- Awasthi, A.K.; Zeng, X.; Li, J. Environmental pollution of electronic waste recycling in India: A critical review. Environ. Pollut. 2016, 211, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Li, J. Environmental effects of heavy metals derived from the e-waste recycling activities in China: A systematic review. Waste Manag. 2014, 34, 2587–2594. [Google Scholar] [CrossRef] [PubMed]
- Fujimori, T.; Takigami, H. Pollution distribution of heavy metals in surface soil at an informal electronic-waste recycling site. Environ. Geochem. Health 2014, 36, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Baldé, C. The Global e-Waste Monitor 2014: Quantities, Flows and Resources; United Nations University: Tokyo, Japan, 2015. [Google Scholar]
- Riget, F.; Vorkamp, K.; Bossi, R.; Sonne, C.; Letcher, R.J.; Dietz, R. Twenty years of monitoring of persistent organic pollutants in Greenland biota. A review. Environ. Pollut. 2016, 217, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Abrevaya, X.C.; Sacco, N.J.; Bonetto, M.C.; Hilding-Ohlsson, A.; Cortón, E. Analytical applications of microbial fuel cells. Part II: Toxicity, microbial activity and quantification, single analyte detection and other uses. Biosens. Bioelectron. 2015, 63, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Su, L.; Jia, W.; Hou, C.; Lei, Y. Microbial biosensors: A review. Biosens. Bioelectron. 2011, 26, 1788–1799. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, B.D.; Chaubey, A. Biosensors for clinical diagnostics industry. Sens. Actuators B Chem. 2003, 91, 117–127. [Google Scholar] [CrossRef]
- Pasternak, G.; Greenman, J.; Ieropoulos, I. Self-powered, autonomous biological oxygen demand biosensor for online water quality monitoring. Sens. Actuators B Chem. 2017, 244, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Velasquez-Orta, S.B.; Werner, D.; Varia, J.C.; Mgana, S. Microbial fuel cells for inexpensive continuous in-situ monitoring of groundwater quality. Water Res. 2017, 117, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Jouanneau, S.; Recoules, L.; Durand, M.J.; Boukabache, A.; Picot, V.; Primault, Y.; Lakel, A.; Sengelin, M.; Barillon, B.; Thouand, G. Methods for assessing biochemical oxygen demand (BOD): A review. Water Res. 2014, 49, 62–82. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Call, D.; Cheng, S.; Hamelers, H.V.; Sleutels, T.H.; Jeremiasse, A.W.; Rozendal, R.A. Microbial electrolysis cells for high yield hydrogen gas production from organic matter. Environ. Sci. Technol. 2008, 42, 8630–8640. [Google Scholar] [CrossRef] [PubMed]
- Nevin, K.P.; Kim, B.C.; Glaven, R.H.; Johnson, J.P.; Woodard, T.L.; Methe, B.A.; Didonato, R.J.; Covalla, S.F.; Franks, A.E.; Liu, A.; et al. Anode biofilm transcriptomics reveals outer surface components essential for high density current production in Geobacter sulfurreducens fuel cells. PLoS ONE 2009, 4, e5628. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K.; Qian, X.; Morgado, L.; Kim, B.C.; Mester, T.; Izallalen, M.; Salgueiro, C.A.; Lovley, D.R. Purification and characterization of OmcZ, an outer-surface, octaheme c-type cytochrome essential for optimal current production by Geobacter sulfurreducens. Appl. Environ. Microbiol. 2010, 76, 3999–4007. [Google Scholar] [CrossRef] [PubMed]
- Malvankar, N.S.; Lovley, D.R. Microbial nanowires for bioenergy applications. Curr. Opin. Biotechnol. 2014, 27, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Reguera, G.; McCarthy, K.D.; Mehta, T.; Nicoll, J.S. Extracellular electron transfer via microbial nanowires. Nature 2005, 435, 1098–1101. [Google Scholar] [CrossRef] [PubMed]
- Kotloski, N.J.; Gralnick, J.A. Flavin electron shuttles dominate extracellular electron transfer by Shewanella oneidensis. mBio 2013, 4, e00553-12. [Google Scholar] [CrossRef] [PubMed]
- Yong, Y.C.; Cai, Z.; Yu, Y.Y.; Chen, P.; Jiang, R.; Cao, B.; Sun, J.Z.; Wang, J.Y.; Song, H. Increase of riboflavin biosynthesis underlies enhancement of extracellular electron transfer of Shewanella in alkaline microbial fuel cells. Bioresour. Technol. 2013, 130, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Rabaey, K.; Rodriguez, J.; Blackall, L.L.; Keller, J.; Gross, P.; Batstone, D.; Verstraete, W.; Nealson, K.H. Microbial ecology meets electrochemistry: Electricity-driven and driving communities. ISME J. 2007, 1, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Zeikus, J. Utilization of electrically reduced neutral Red byActinobacillus succinogenes: Physiological function of neutral Red in membrane-driven fumarate reduction and energy conservation. J. Bacteriol. 1999, 181, 2403–2410. [Google Scholar] [PubMed]
- Park, D.H.; Zeikus, J.G. Electricity generation in microbial fuel cells using neutral red as an electronophore. Appl. Environ. Microbiol. 2000, 66, 1292–1297. [Google Scholar] [CrossRef] [PubMed]
- Chouler, J.; Di Lorenzo, M. Water quality monitoring in developing countries; can microbial fuel cells be the answer? Biosensors 2015, 5, 450–470. [Google Scholar] [CrossRef] [PubMed]
- Abrevaya, X.C.; Sacco, N.J.; Bonetto, M.C.; Hilding-Ohlsson, A.; Cortón, E. Analytical applications of microbial fuel cells. Part I: Biochemical oxygen demand. Biosens. Bioelectron. 2015, 63, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, M.; Thomson, A.R.; Schneider, K.; Cameron, P.J.; Ieropoulos, I. A small-scale air-cathode microbial fuel cell for on-line monitoring of water quality. Biosens. Bioelectron. 2014, 62, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Sik Hyun, M.; Gadd, G.M.; Joo Kim, H. A novel biomonitoring system using microbial fuel cells. J. Environ. Monit. 2007, 9, 1323–1328. [Google Scholar] [CrossRef] [PubMed]
- Logan, B.E.; Hamelers, B.; Rozendal, R.; Schröder, U.; Keller, J.; Freguia, S.; Aelterman, P.; Verstraete, W.; Rabaey, K. Microbial fuel cells: Methodology and technology. Environ. Sci. Technol. 2006, 40, 5181–5192. [Google Scholar] [CrossRef] [PubMed]
- Stein, N.E.; Hamelers, H.M.V.; van Straten, G.; Keesman, K.J. On-line detection of toxic components using a microbial fuel cell-based biosensor. J. Process Control 2012, 22, 1755–1761. [Google Scholar] [CrossRef]
- Hamelers, H.V.M.; ter Heijne, A.; Stein, N.; Rozendal, R.A.; Buisman, C.J.N. Butler–Volmer–Monod model for describing bio-anode polarization curves. Bioresour. Technol. 2011, 102, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Bai, L.; Zhai, J.; Wang, Y.; Dong, S. Toxicity detection in water containing heavy metal ions with a self-powered microbial fuel cell-based biosensor. Talanta 2017, 168, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Jiang, Y.; Zhou, Y.; Shen, K.; Zhong, W. Using electrical signals of microbial fuel cells to detect copper stress on soil microorganisms. Eur. J. Soil Sci. 2015, 66, 369–377. [Google Scholar] [CrossRef]
- Zeng, L.; Li, X.; Shi, Y.; Qi, Y.; Huang, D.; Tadé, M.; Wang, S.; Liu, S. FePO 4 based single chamber air-cathode microbial fuel cell for online monitoring levofloxacin. Biosens. Bioelectron. 2017, 91, 367–373. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.H.; Cheng, C.Y.; Liu, M.H.; Chen, T.Y.; Hsieh, M.C.; Chung, Y.C. Utility of Ochrobactrum anthropi YC152 in a Microbial Fuel Cell as an Early Warning Device for Hexavalent Chromium Determination. Sensors 2016, 16, 1272. [Google Scholar] [CrossRef] [PubMed]
- Davila, D.; Esquivel, J.; Sabate, N.; Mas, J. Silicon-based microfabricated microbial fuel cell toxicity sensor. Biosens. Bioelectron. 2011, 26, 2426–2430. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, N.; Zhou, Q. Concentration responses of toxicity sensor with Shewanella oneidensis MR-1 growing in bioelectrochemical systems. Biosens. Bioelectron. 2013, 43, 264–267. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Niu, Y.; Zhao, S.; Khan, A.; Ling, Z.; Chen, Y.; Liu, P.; Li, X. A novel biosensor for p-nitrophenol based on an aerobic anode microbial fuel cell. Biosens. Bioelectron. 2016, 85, 860–868. [Google Scholar] [CrossRef] [PubMed]
- Chae, K.J.; Choi, M.J.; Lee, J.W.; Kim, K.Y.; Kim, I.S. Effect of different substrates on the performance, bacterial diversity, and bacterial viability in microbial fuel cells. Bioresour. Technol. 2009, 100, 3518–3525. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Chen, W.; Mulchandani, A. Microbial biosensors. Anal. Chim. Acta 2006, 568, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhou, M.; Liu, M.; Yang, W.; Gu, T. Microbial fuel cells for biosensor applications. Biotechnol. Lett. 2015, 37, 2357–2364. [Google Scholar] [CrossRef] [PubMed]
- Falk, H.M.; Reichling, P.; Andersen, C.; Benz, R. Online monitoring of concentration and dynamics of volatile fatty acids in anaerobic digestion processes with mid-infrared spectroscopy. Bioprocess Biosyst. Eng. 2015, 38, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Raposo, F.; Borja, R.; Cacho, J.; Mumme, J.; Orupõld, K.; Esteves, S.; Noguerol-Arias, J.; Picard, S.; Nielfa, A.; Scherer, P. First international comparative study of volatile fatty acids in aqueous samples by chromatographic techniques: Evaluating sources of error. TrAC Trends Anal. Chem. 2013, 51, 127–143. [Google Scholar] [CrossRef]
- Siedlecka, E.; Kumirska, J.; Ossowski, T.; Glamowski, P.; Gołębiowski, M.; Gajdus, J.; Kaczyński, Z.; Stepnowski, P. Determination of volatile fatty acids in environmental aqueous samples. Pol. J. Environ. Stud. 2008, 17, 351–356. [Google Scholar]
- Kaur, A.; Kim, J.R.; Michie, I.; Dinsdale, R.M.; Guwy, A.J.; Premier, G.C. Microbial fuel cell type biosensor for specific volatile fatty acids using acclimated bacterial communities. Biosens. Bioelectron. 2013, 47, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Angelidaki, I.; Zhang, Y. Microbial electrochemical monitoring of volatile fatty acids during anaerobic digestion. Environ. Sci. Technol. 2016, 50, 4422–4429. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Li, X.; Zhao, N.; Angelidaki, I.; Zhang, Y. Bio-electrolytic sensor for rapid monitoring of volatile fatty acids in anaerobic digestion process. Water Res. 2017, 111, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.J.; Lefebvre, O.; Tan, Z.; Ng, H.Y. Microbial fuel-cell-based toxicity sensor for fast monitoring of acidic toxicity. Water Sci. Technol. 2012, 65, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Schievano, A.; Colombo, A.; Cossettini, A.; Goglio, A.; D’Ardes, V.; Trasatti, S.; Cristiani, P. Single-chamber microbial fuel cells as on-line shock-sensors for volatile fatty acids in anaerobic digesters. Waste Manag. 2017. [Google Scholar] [CrossRef] [PubMed]
- Penn, M.R.; Pauer, J.J.; Mihelcic, J.R. Biochemical oxygen demand. Environ. Ecol. Chem. 2009, 2, 278–297. [Google Scholar]
- Butkus, M.A.; Manous, J.D. Biochemical oxygen demand. In Water Encyclopedia; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Karube, I.; Matsunaga, T.; Mitsuda, S.; Suzuki, S. Microbial electrode BOD sensors. Biotechnol. Bioeng. 1977, 19, 1535–1547. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.H.; Chang, I.S.; Cheol Gil, G.; Park, H.S.; Kim, H.J. Novel BOD (biological oxygen demand) sensor using mediator-less microbial fuel cell. Biotechnol. Lett. 2003, 25, 541–545. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.H.; Jang, J.K.; Pham, T.H.; Moon, H.; Chang, I.S.; Kim, B.H. A microbial fuel cell with improved cathode reaction as a low biochemical oxygen demand sensor. Biotechnol. Lett. 2003, 25, 1357–1361. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Chang, I.S.; Kang, K.H.; Jang, J.K.; Kim, B.H. Improving the dynamic response of a mediator-less microbial fuel cell as a biochemical oxygen demand (BOD) sensor. Biotechnol. Lett. 2004, 26, 1717–1721. [Google Scholar] [CrossRef] [PubMed]
- Chang, I.S.; Jang, J.K.; Gil, G.C.; Kim, M.; Kim, H.J.; Cho, B.W.; Kim, B.H. Continuous determination of biochemical oxygen demand using microbial fuel cell type biosensor. Biosens. Bioelectron. 2004, 19, 607–613. [Google Scholar] [CrossRef]
- Moon, H.; Chang, I.S.; Jang, J.K.; Kim, K.S.; Lee, J.; Lovitt, R.W.; Kim, B.H. On-line monitoring of low biochemical oxygen demand through continuous operation of a mediator-less microbial fuel cell. J. Microbiol. Biotechnol. 2005, 15, 192–196. [Google Scholar]
- Kumlanghan, A.; Liu, J.; Thavarungkul, P.; Kanatharana, P.; Mattiasson, B. Microbial fuel cell-based biosensor for fast analysis of biodegradable organic matter. Biosens. Bioelectron. 2007, 22, 2939–2944. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, M.; Curtis, T.P.; Head, I.M.; Scott, K. A single-chamber microbial fuel cell as a biosensor for wastewaters. Water Res. 2009, 43, 3145–3154. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, L.; Min, B.; Martins, G.; Brito, A.G.; Kroff, P.; Parpot, P.; Angelidaki, I.; Nogueira, R. In situ microbial fuel cell-based biosensor for organic carbon. Bioelectrochemistry 2011, 81, 99–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Angelidaki, I. Submersible microbial fuel cell sensor for monitoring microbial activity and BOD in groundwater: Focusing on impact of anodic biofilm on sensor applicability. Biotechnol. Bioeng. 2011, 108, 2339–2347. [Google Scholar] [CrossRef] [PubMed]
- Ayyaru, S.; Dharmalingam, S. Enhanced response of microbial fuel cell using sulfonated poly ether ether ketone membrane as a biochemical oxygen demand sensor. Anal. Chim. Acta 2014, 818, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Chang, I.S.; Moon, H.; Jang, J.K.; Kim, B.H. Improvement of a microbial fuel cell performance as a BOD sensor using respiratory inhibitors. Biosens. Bioelectron. 2005, 20, 1856–1859. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.; Luong, T.T.T.; Tran, P.H.N.; Bui, H.T.V.; Nguyen, H.Q.; Dinh, H.T.; Kim, B.H.; Pham, H.T. A lithotrophic microbial fuel cell operated with pseudomonads-dominated iron-oxidizing bacteria enriched at the anode. Microbial Biotechnol. 2015, 8, 579–589. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.H.; Luong, T.T.; Nguyen, T.T.; Nguyen, H.Q.; Duong, H.V.; Kim, B.H.; Pham, H.T. Possibility of using a lithotrophic iron-oxidizing microbial fuel cell as a biosensor for detecting iron and manganese in water samples. Environ. Sci. Process. Impacts 2015, 17, 1806. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Lei, Y.; Li, B. A batch-mode cube microbial fuel cell based “shock” biosensor for wastewater quality monitoring. Biosens. Bioelectron. 2014, 62, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Li, X.; Jiang, X.; Xie, B.; Liu, H.; Liang, D.; Zhu, Y. Assessing the solitary and joint biotoxicities of heavy metals and acephate using microbial fuel cell. In Proceedings of the 2015 International Conference on Advances in Energy, Environment and Chemical Engineering, Changsha, China, 26–27 September 2015. [Google Scholar]
- Lee, J.S.; Kim, D.S.; Jeon, H.J.; Park, B.S. Microbial fuel cell as a biosensor to monitor various toxic metal substances in water. In Proceedings of the 9th International Conference on Sensing Technology, Auckland, New Zealand, 8–10 December 2015; pp. 416–419. [Google Scholar]
- Li, T.; Wang, X.; Zhou, L.; An, J.; Li, J.; Li, N.; Sun, H.; Zhou, Q. Bioelectrochemical sensor using living biofilm to in situ evaluate flocculant toxicity. ACS Sens. 2016, 1, 1374–1379. [Google Scholar] [CrossRef]
- Becaria, A.; Campbell, A.; Bondy, S. Aluminum as a toxicant. Toxicol. Ind. Health 2002, 18, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Laureanti, J.A.; Jones, A.K. Photosynthetic Microbial Fuel Cells. In Biophotoelectrochemistry: From Bioelectrochemistry to Biophotovoltaics; Springer International Publishing: Switzerland, 2016; pp. 159–175. [Google Scholar]
- Strik, D.P.; Terlouw, H.; Hamelers, H.V.; Buisman, C.J. Renewable sustainable biocatalyzed electricity production in a photosynthetic algal microbial fuel cell (PAMFC). Appl. Microbiol. Biotechnol. 2008, 81, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Labro, J.; Craig, T.; Wood, S.A.; Packer, M.A. Demonstration of the use of a photosynthetic microbial fuel cell as an environmental biosensor. Int. J. Nanotechnol. 2017, 14, 213–225. [Google Scholar] [CrossRef]
- Shen, Y.; Wang, M.; Chang, I.S.; Ng, H.Y. Effect of shear rate on the response of microbial fuel cell toxicity sensor to Cu(II). Bioresour. Technol. 2013, 136, 707–710. [Google Scholar] [CrossRef] [PubMed]
- Tao, X.; Gao, Y.-M.; Zheng, Q.; Wang, X.-H.; Yuan, L.; Nan, L.; Rui, L. A Double-microbial Fuel Cell Heavy Metals Toxicity Sensor. DEStech Trans. Environ. Energy Earth Sci. 2017. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, H.S.; Hyun, M.S.; Chang, I.S.; Kim, M.; Kim, B.H. A mediator-less microbial fuel cell using a metal reducing bacterium, Shewanella putrefaciens. Enzym. Microb. Technol. 2002, 30, 145–152. [Google Scholar] [CrossRef]
- Ringeisen, B.R.; Henderson, E.; Wu, P.K.; Pietron, J.; Ray, R.; Little, B.; Biffinger, J.C.; Jones-Meehan, J.M. High power density from a miniature microbial fuel cell using Shewanella oneidensis DSP10. Environ. Sci. Technol. 2006, 40, 2629–2634. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, W.; Kimura, N.; Kamagata, Y. A novel p-nitrophenol degradation gene cluster from a gram-positive bacterium, Rhodococcus opacus SAO101. J. Bacteriol. 2004, 186, 4894–4902. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Yang, W.; Wei, X.; Fraiwan, A.; Choi, S. A microsized microbial fuel cell based biosensor for fast and sensitive detection of toxic substances in water. In Proceedings of the 2015 28th IEEE International Conference on Micro Electro Mechanical Systems (MEMS), Estoril, Portugal, 18–22 January 2015; IEEE: Piscataway, NJ, USA, 2015; pp. 573–576. [Google Scholar]
- Jiang, Y.; Liang, P.; Liu, P.; Wang, D.; Miao, B.; Huang, X. A novel microbial fuel cell sensor with biocathode sensing element. Biosens. Bioelectron. 2017, 94, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Chen, A. Functionalization of carbon buckypaper for the sensitive determination of hydrogen peroxide in human urine. Biosens. Bioelectron. 2012, 35, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Schreiter, P.P.Y.; Gillor, O.; Post, A.; Belkin, S.; Schmid, R.D.; Bachmann, T.T. Monitoring of phosphorus bioavailability in water by an immobilized luminescent cyanobacterial reporter strain. Biosens. Bioelectron. 2001, 16, 811–818. [Google Scholar] [CrossRef]
- Casavant, N.C.; Thompson, D.; Beattie, G.A.; Phillips, G.J.; Halverson, L.J. Use of a site-specific recombination-based biosensor for detecting bioavailable toluene and related compounds on roots. Environ. Microbiol. 2003, 5, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Wei, X.; Fraiwan, A.; Coogan, C.G.; Lee, H.; Choi, S. Fast and sensitive water quality assessment: A μL-scale microbial fuel cell-based biosensor integrated with an air-bubble trap and electrochemical sensing functionality. Sens. Actuators B Chem. 2016, 226, 191–195. [Google Scholar] [CrossRef]
- Xu, X.; Ying, Y. Microbial biosensors for environmental monitoring and food analysis. Food Rev. Int. 2011, 27, 300–329. [Google Scholar] [CrossRef]
- Liu, Y.; Ding, M.; Ling, W.; Yang, Y.; Zhou, X.; Li, B.-Z.; Chen, T.; Nie, Y.; Wang, M.; Zeng, B.; et al. A three-species microbial consortium for power generation. Energy Environ. Sci. 2017, 10, 1600–1609. [Google Scholar] [CrossRef]
- Choudhury, P.; Prasad Uday, U.S.; Bandyopadhyay, T.K.; Ray, R.N.; Bhunia, B. Performance improvement of microbial fuel cell (MFC) using suitable electrode and Bioengineered organisms: A review. Bioengineered 2017, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kong, X.; Zhou, X.; Tian, Y.; Wu, X.; Zhang, J.; Zuo, W. Niobium doped lanthanum calcium ferrite perovskite as a novel electrode material for symmetrical solid oxide fuel cells. J. Power Sources 2016, 326, 35–42. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, X.; Li, S.; Lv, X.; Cheng, Y.; Wang, X. Microbial biofuel cell operating effectively through carbon nanotube blended with gold–titania nanocomposites modified electrode. Electrochim. Acta 2013, 109, 328–332. [Google Scholar] [CrossRef]
- Liang, P.; Wang, H.; Xia, X.; Huang, X.; Mo, Y.; Cao, X.; Fan, M. Carbon nanotube powders as electrode modifier to enhance the activity of anodic biofilm in microbial fuel cells. Biosens. Bioelectron. 2011, 26, 3000–3004. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.C.; Cheng, C.Y.; Liu, M.H.; Chung, Y.C. Effects of operating parameters on measurements of biochemical oxygen demand using a mediatorless microbial fuel cell biosensor. Sensors 2015, 16, 35. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Yang, G.; Wang, J.; Ngo, H.H.; Guo, W.; Zhang, H.; Zhang, X. Performance of a microbial fuel cell-based biosensor for online monitoring in an integrated system combining microbial fuel cell and upflow anaerobic sludge bed reactor. Bioresour. Technol. 2016, 218, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Stein, N.E.; Hamelers, H.V.M.; Buisman, C.N.J. Influence of membrane type, current and potential on the response to chemical toxicants of a microbial fuel cell based biosensor. Sens. Actuators B Chem. 2012, 163, 1–7. [Google Scholar] [CrossRef]
- Stein, N.E.; Hamelers, H.V.; Buisman, C.N. Stabilizing the baseline current of a microbial fuel cell-based biosensor through overpotential control under non-toxic conditions. Bioelectrochemistry 2010, 78, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Fraiwan, A.; Sundermier, S.; Han, D.; Steckl, A.; Hassett, D.; Choi, S. Enhanced Performance of Micro-Electro-Mechanical-Systems (MEMS) Microbial Fuel Cells Using Electrospun Microfibrous Anode and Optimizing Operation. Fuel Cells 2013, 13, 336–341. [Google Scholar] [CrossRef]
- Bergel, A.; Féron, D.; Mollica, A. Catalysis of oxygen reduction in PEM fuel cell by seawater biofilm. Electrochem. Commun. 2005, 7, 900–904. [Google Scholar] [CrossRef]
- Si, R.-W.; Zhai, D.-D.; Liao, Z.-H.; Gao, L.; Yong, Y.-C. A whole-cell electrochemical biosensing system based on bacterial inward electron flow for fumarate quantification. Biosens. Bioelectron. 2015, 68, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liang, P.; Zhang, C.; Bian, Y.; Yang, X.; Huang, X.; Girguis, P.R. Enhancing the response of microbial fuel cell based toxicity sensors to Cu(II) with the applying of flow-through electrodes and controlled anode potentials. Bioresour. Technol. 2015, 190, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Kaur, A.; Ibrahim, S.; Pickett, C.J.; Michie, I.S.; Dinsdale, R.M.; Guwy, A.J.; Premier, G.C. Anode modification to improve the performance of a microbial fuel cell volatile fatty acid biosensor. Sens. Actuators B Chem. 2014, 201, 266–273. [Google Scholar] [CrossRef]
- Stein, N.E.; Keesman, K.J.; Hamelers, H.V.M.; van Straten, G. Kinetic models for detection of toxicity in a microbial fuel cell based biosensor. Biosens. Bioelectron. 2011, 26, 3115–3120. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Kayode, O.; Harper, W.F., Jr. Using microbial fuel cell output metrics and nonlinear modeling techniques for smart biosensing. Sci. Total Environ. 2013, 449, 223–228. [Google Scholar] [CrossRef] [PubMed]

| Source Inoculum | MFC Configuration | Electrode Material | Detection Range (BOD, mg L−1) | Saturation Signal | Response Time (min) | Reference |
|---|---|---|---|---|---|---|
| Clostridium butyricum | Double chamber | Anode: Pt; cathode: Carbon | 10–300 | 0.120 mA | 70 | [49] |
| MFC effluent | Double chamber | Graphite felt | 2.58–206.4 | 1.1 mA a | 30–600 | [50] |
| River sediment | Double chamber | Graphite felt | 5 | ND | 180 | [51] |
| MFC effluent | Double-chamber | ND | 50–100 | 1.85 mA a | 36 | [52] |
| Activated sludge | Double chamber | Graphite felt | 23–200 | 6 mA a | 60 | [53] |
| River sediments | Double chamber | Graphite felt | 2–10 | 6 mA | 60 | [54] |
| Activated sludge | Single chamber | Graphite roll | Glucose: 1000–25,000 b | 1.6 mv a | 60 | [55] |
| Primary wastewater | Single chamber | Carbon cloth | COD: 50–1000 b | 0.4 mA | 40 | [56] |
| Domestic wastewater | Double chamber | Carbon paper | 17–183 | 222 mA | 30 | [57] |
| Underground water | Single chamber | Carbon paper | 10–250 | 233 mA | <40.2 | [58] |
| Activated sludge | Double chamber | Carbon cloth | 50–650 | 0.6 mA a | 80 | [59] |
| Neat human urine | Single chamber | Carbon fibre | Urine: 67–813 b | 297 mV | 69–960 | [9] |
| Heavy Metals | Source Inoculum | MFC Configuration | Electrode Material | Voltage or Current | Inhibition Ratio | Detection Range (mg L−1) | Reference |
|---|---|---|---|---|---|---|---|
| Hg, Pb | Activated sludge | Double chamber | Carbon felt | 0.026–0.040 mA | -- | 1–10 | [25] |
| Fe, Mn | Iron-oxidizing bacterial consortia | Double chamber | Graphite rod | 0.4–0.6 mA 0.1–0.3 mA | -- | Fe: 168–1120 Mn: 5.5–165 | [62] |
| KAl(SO4)2·12H2O | MFC effluent | Double chamber | Glassy carbon | 6–6.75 A m2−1 a | -- | 50–500 | [66] |
| Cu | Soil | Double chamber | Carbon felt | 52–354 mV | -- | 50–400 | [30] |
| Cr, Fe | Fresh wastewater | Single chamber | Carbon felt | 53–125 mV | -- | Cr: 1–8 | [63] |
| 118–121 mV | Fe: 1–48 | ||||||
| Cr | Ochrobactrum anthropi YC152 | Double chamber | Plain porous carbon paper | 81–258 mV a | -- | 0.0125–5 | [32] |
| Cu | Domestic wastewater | Single chamber | Carbon felt | -- | 30–85% | 5–7 | [71] |
| Cu, Ni, Cd | Activated sludge | Single chamber | Carbon cloth | -- | Cu: 7.5–22.5% | Cu: 1–10 | [64] |
| Cd: 10–60% | Cd: 0.1–1.0 | ||||||
| Ni: 3–10% a | Ni: 0.1–1.0 | ||||||
| Cu, Zn | Paulschulzia pseudovolvox; Cyanobactera CAWBG64 | Double chamber | Carbon cloth | -- | Cu: 0–115% | Cu: 0.063–0.189 | [70] |
| Zn: 0–100% b | Zn: 0.065–0.195 | ||||||
| Cu,Hg Zn, Cd Pb, Cr | Anaerobic sludge | Double chamber | Graphite felts | -- | Cu: 7.9–18.48% | Cu: 1–4 | [29] |
| Hg: 13.99% | Other metals: 0–2 | ||||||
| Zn: 8,81% | |||||||
| Cd: 9.29% | |||||||
| Pb: 5.59% | |||||||
| Cr: 1.95% | |||||||
| Cu, Zn Cr, Cd | Anaerobic sludge | Double chamber | Carbon felt | -- | Cu: 1.02–9.31% | Cu: 1–25 | [72] |
| Zn: 0.70–4.16% | Zn: 15–80 | ||||||
| No data for Cr and Cd | Cr: 0.3–1Cd: 0.4–10 |
| Organic Substrate | Source Inoculum | MFC Configuration | Electrode Material | Voltage or Current | Inhibition Ratio | Detection Range (mg L−1) | Reference |
|---|---|---|---|---|---|---|---|
| Diazinon | Activated sludge | Double chamber | Carbon felt | -- | 55–61% | 1–10 | [25] |
| Polychlorinated biphenyls | Activated sludge | Double chamber | Carbon felt | -- | 29–38% | 1–5 | [25] |
| Acephate | Activated sludge | Single chamber | Carbon cloth | -- | 8.54–13.34% | 1–7 | [64] |
| Glyphosate | Cyanobacteria CAWBG64 Paulschulzia pseudovolvox | Double chamber | Carbon cloth | 0–125% | -- | 0.169–0.507 | [70] |
| Formaldehyde | Geobacter sulfurreducens | Double chamber | Ti/Ni/Au layer | 0–200 mV | -- | 100 | [33] |
| Formaldehyde | Shewanella oneidensis MR-1 | Single chamber | Graphite rod | 0–200 mV | -- | 100–1000 | [34] |
| p-Nitrophenol | Pseudomonas monteilii LZU-3 | Double chamber | Carbon felt | 115–150 mV | -- | 50–200 | [35] |
| Formaldehyde | Wild-type Shewanella oneidensis | Single chamber | Carbon cloth | 0.014–0.023 mA | -- | 10–100 | [76] |
| Levofloxacin | No Data | Single chamber | Carbon felt | 0.41–0.2 mA | -- | 0.0001–1 | [31] |
| Formaldehyde | MFC effluent | Double chamber | Graphite felt | 0.22–0.5 mA | -- | 5–100 | [77] |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, T.; Han, H.; Liu, P.; Xiong, J.; Tian, F.; Li, X. Microbial Fuels Cell-Based Biosensor for Toxicity Detection: A Review. Sensors 2017, 17, 2230. https://doi.org/10.3390/s17102230
Zhou T, Han H, Liu P, Xiong J, Tian F, Li X. Microbial Fuels Cell-Based Biosensor for Toxicity Detection: A Review. Sensors. 2017; 17(10):2230. https://doi.org/10.3390/s17102230
Chicago/Turabian StyleZhou, Tuoyu, Huawen Han, Pu Liu, Jian Xiong, Fake Tian, and Xiangkai Li. 2017. "Microbial Fuels Cell-Based Biosensor for Toxicity Detection: A Review" Sensors 17, no. 10: 2230. https://doi.org/10.3390/s17102230
APA StyleZhou, T., Han, H., Liu, P., Xiong, J., Tian, F., & Li, X. (2017). Microbial Fuels Cell-Based Biosensor for Toxicity Detection: A Review. Sensors, 17(10), 2230. https://doi.org/10.3390/s17102230





