Nanobiosensing with Arrays and Ensembles of Nanoelectrodes
Abstract
:1. Introduction
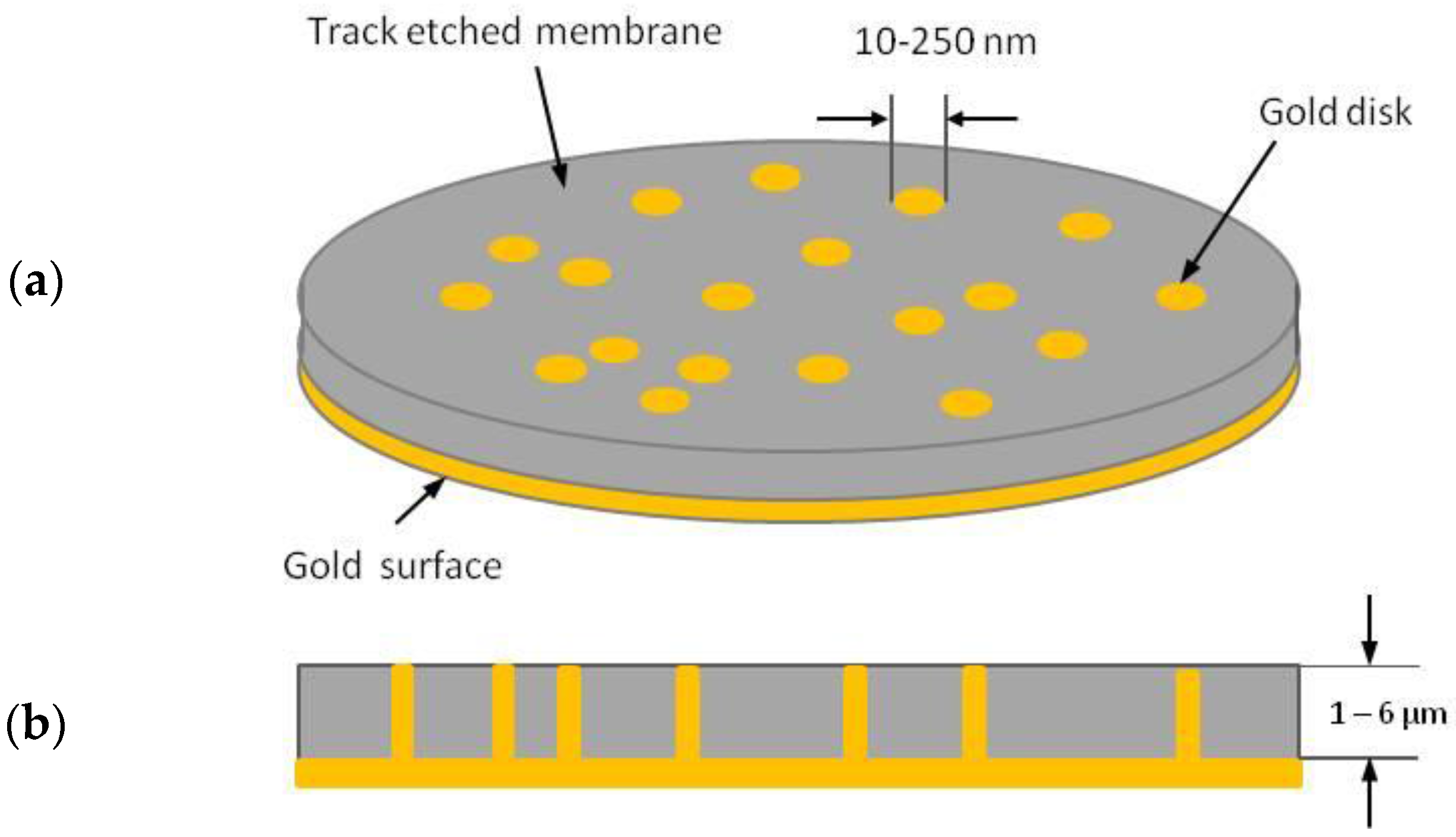
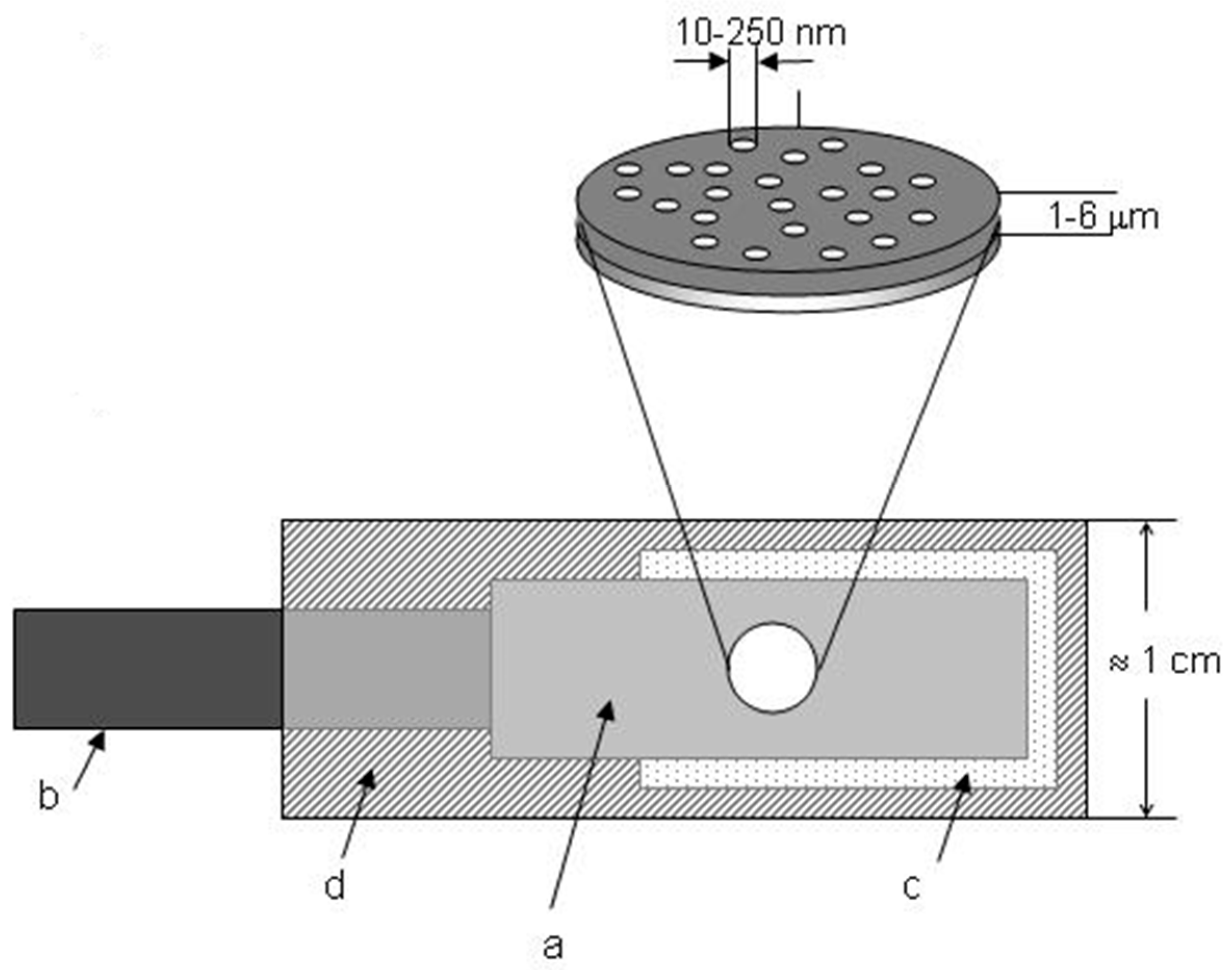
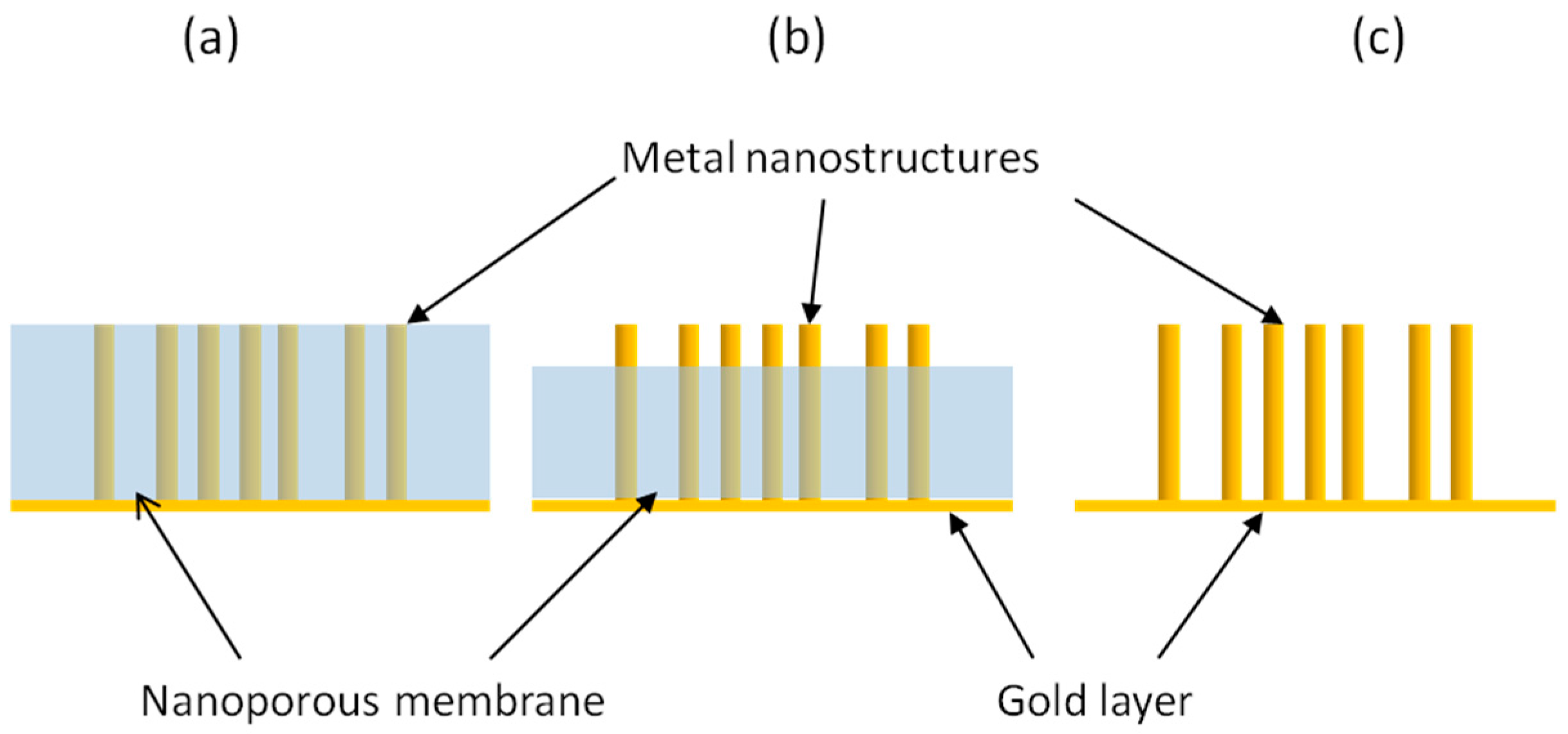
2. Template Ensemble of Nanoelectrodes
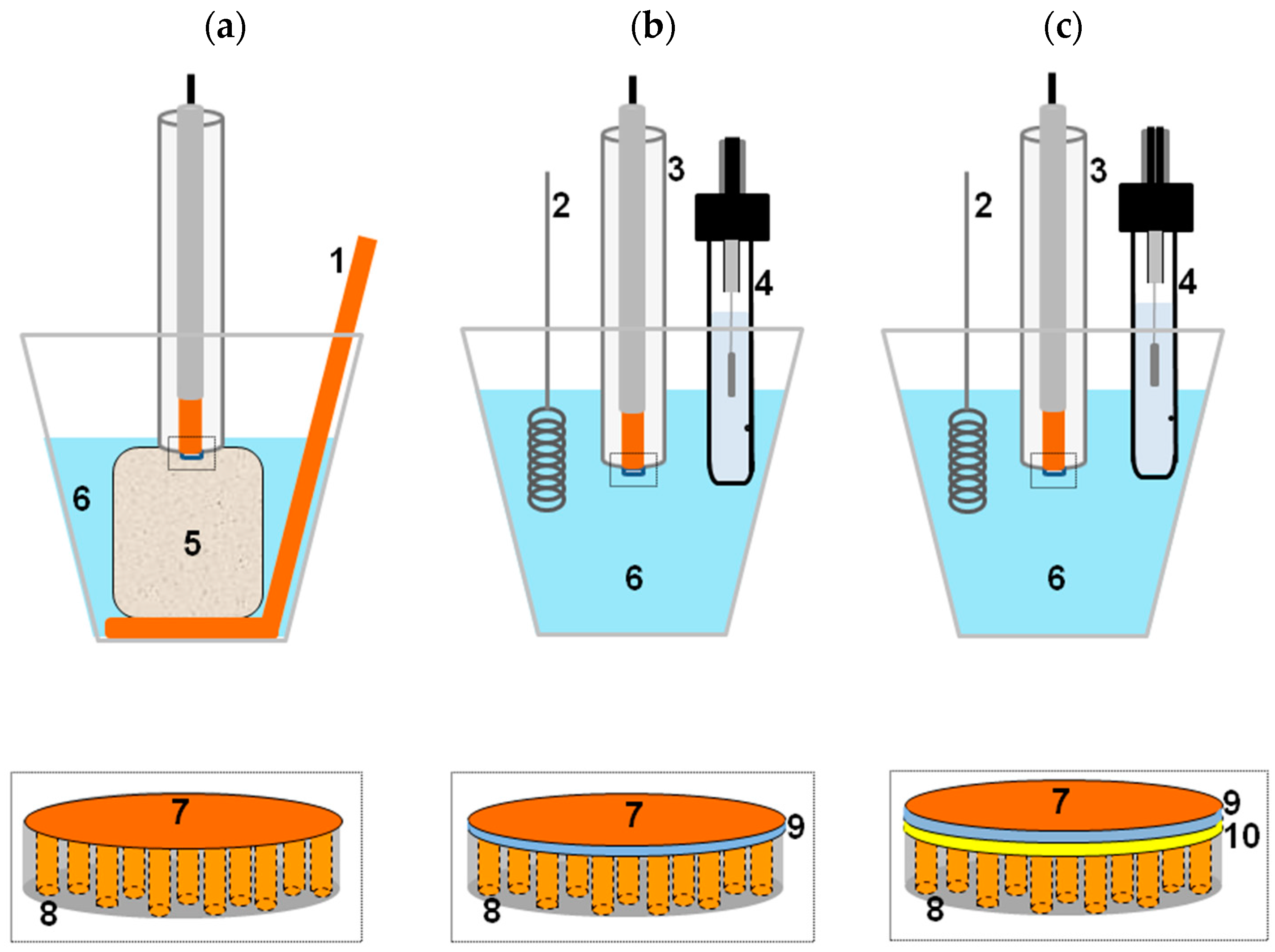
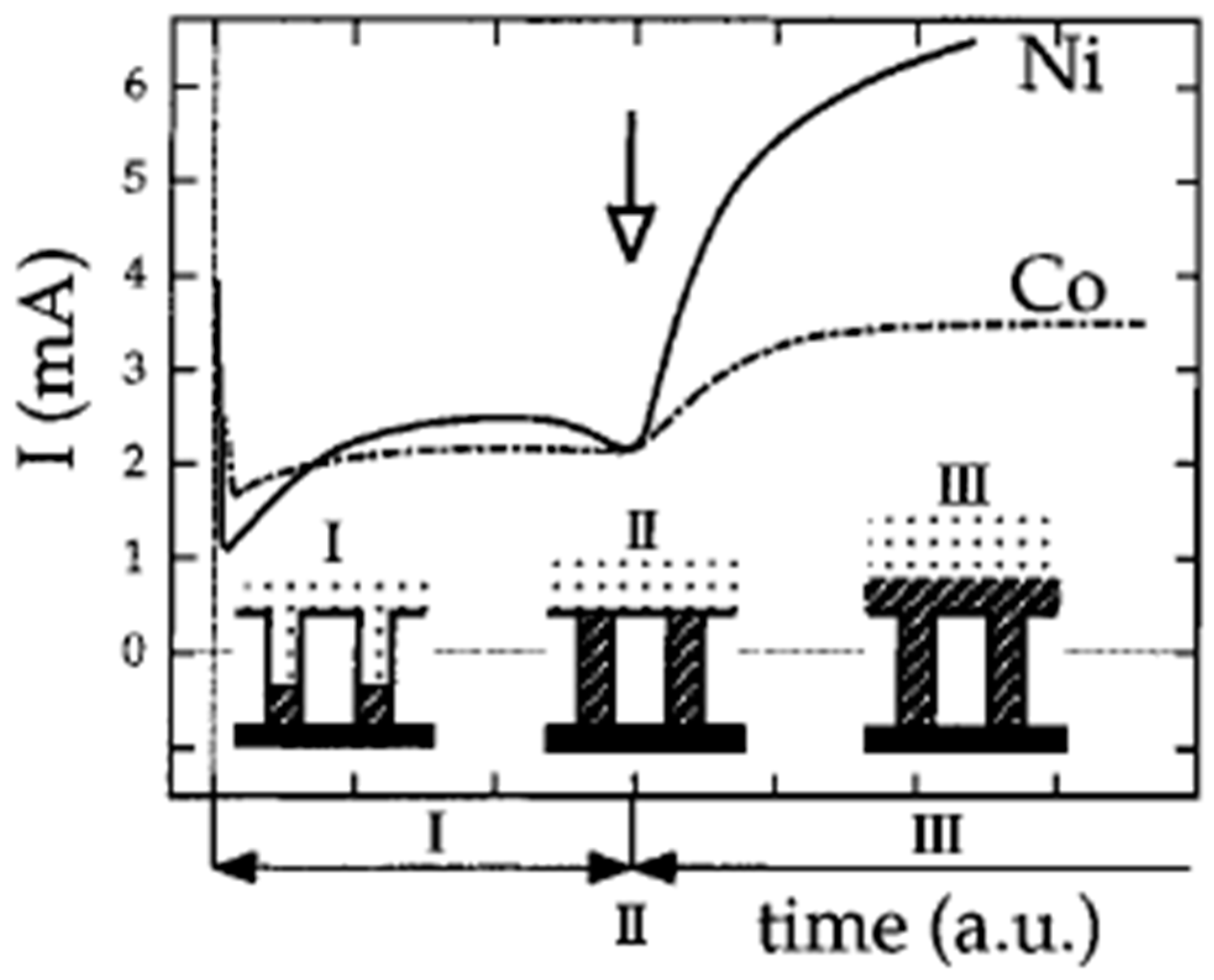
2.1. Template Electrochemical Deposition of Metals
2.2. Template Electroless Deposition
2.3. Combined Electroless-Electrochemical Deposition
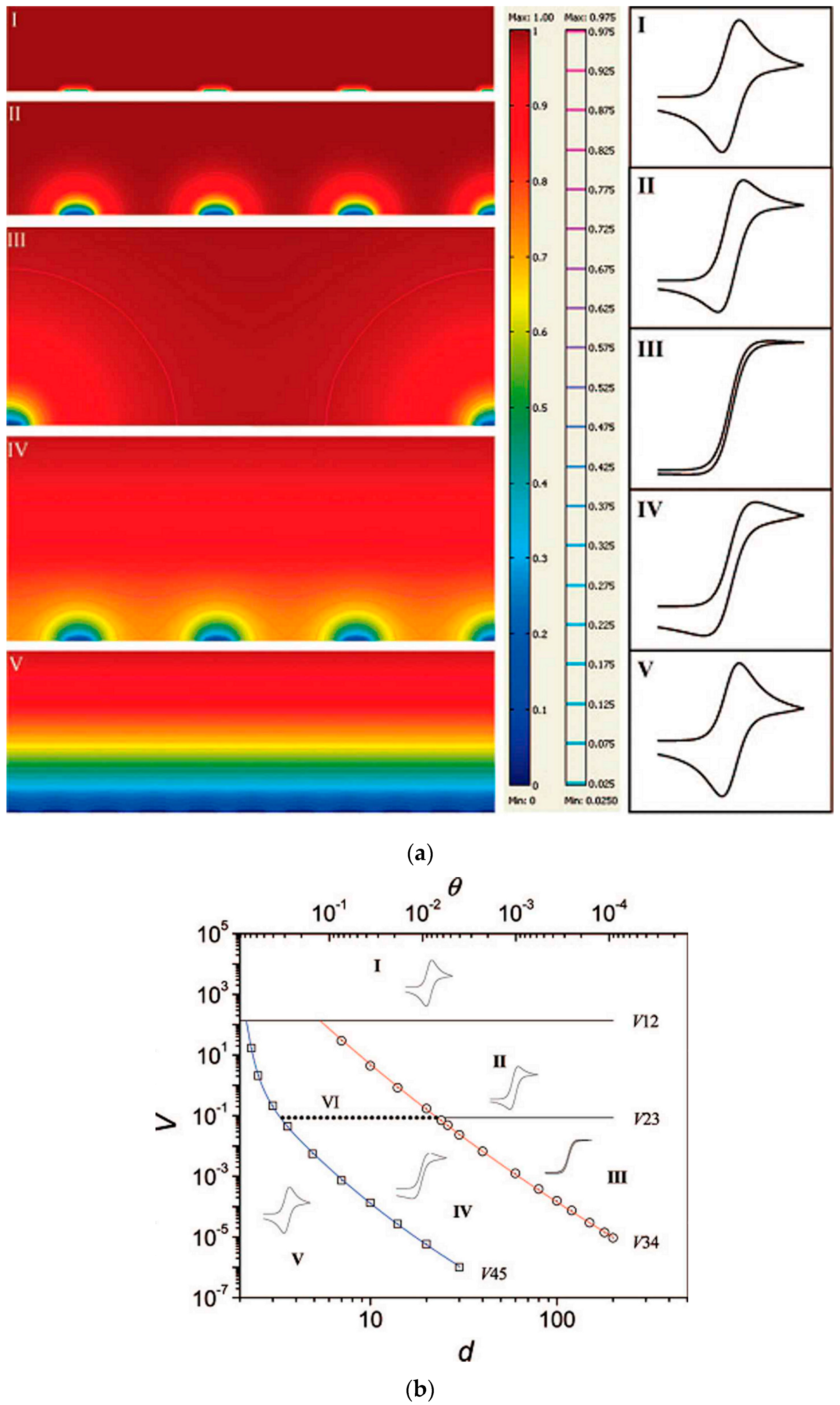
3. Diffusion at Arrays or Ensembles of Nanoelectrodes
4. Voltammetry with NEEs and NEAs
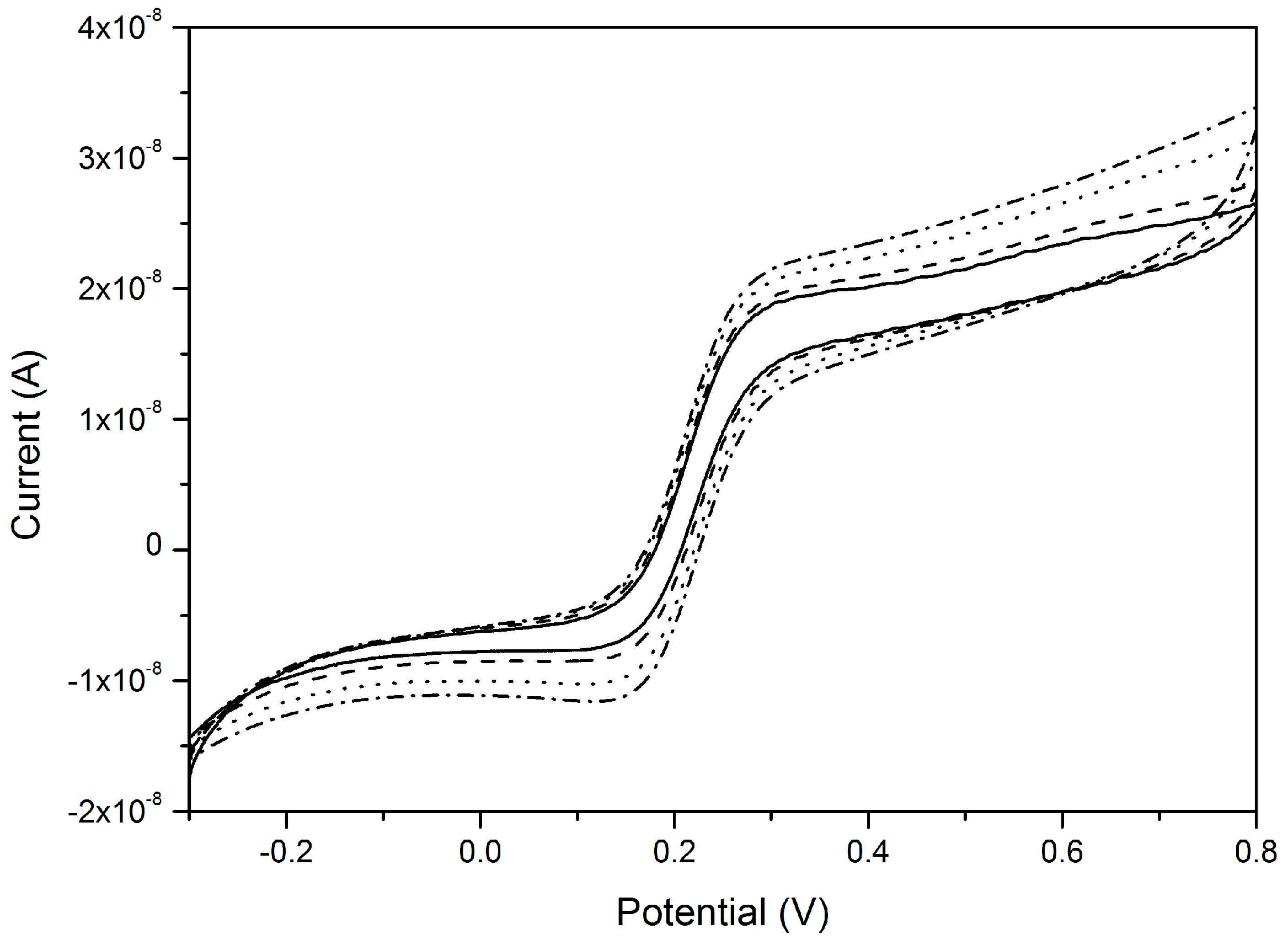
4.1. Voltammetry with NEEs
4.2. Ordered Arrays of Nanoelectrodes Fabricated by Nanolithography
4.3. Nanoelectrode Arrays of Boron-Doped Diamond
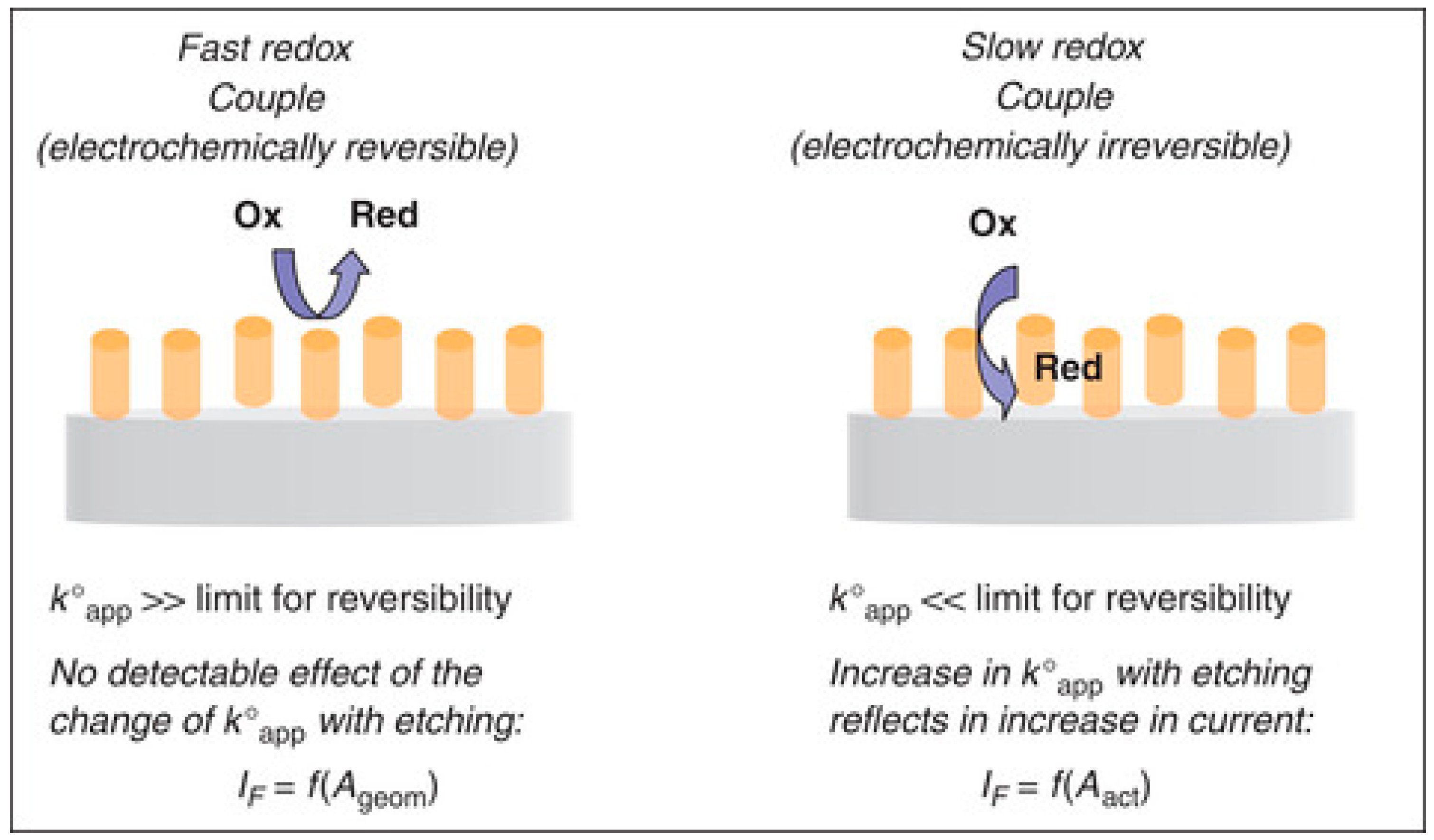
4.4. Electron Transfer Kinetics
5. From 2D- to 3D-NEAs
6. Bio-Analytical Applications of NEEs/NEAs: First Studies
7. Most Recent Advances in Biosensing with NEEs/NEAs
8. Conclusions and Prospects
Author Contributions
Conflicts of Interest
References
- Martin, C.R.; Mitchell, D.T. Template-synthesed nanomaterials in electrochemistry. In Electroanalytical Chemistry; Bard, A.J., Rubinstein, I., Eds.; Marcel Dekker: New York, NY, USA, 1999; Volume 21, pp. 1–74. [Google Scholar]
- Ugo, P.; Moretto, L.M.; Vezzà, F. Ionomer-coated electrodes and nanoelectrode ensembles as electrochemical environmental sensors: Recent advances and prospects. Chem. Phys. Chem. 2002, 3, 917–925. [Google Scholar] [CrossRef]
- Arrigan, D.W.M. Nanoelectrodes, nanoelectrode arrays and their application. Analyst 2004, 129, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Ongaro, M.; Ugo, P. Bioelectroanalysis with nanoelectrode ensembles and arrays. Anal. Bioanal. Chem. 2013, 405, 3715–3721. [Google Scholar] [CrossRef] [PubMed]
- De Leo, M.; Kuhn, A.; Ugo, P. 3D-ensembles of gold nanowires: Preparation, characterization and electroanalytical peculiarities. Electroanalysis 2007, 19, 227–236. [Google Scholar] [CrossRef]
- Heim, M.; Reculusa, S.; Ravaine, S.; Kuhn, A. Engineering of complex macroporous materials through controlled electrodeposition in colloidal superstructures. Adv. Funct. Mater. 2012, 22, 538–545. [Google Scholar] [CrossRef]
- Godino, N.; Borrise, X.; Munoz, F.X.; del Campo, F.J.; Compton, R.G. Mass transport to nanoelectrode arrays and limitations of the diffusion domain approach: Theory and experiment. J. Phys. Chem. C 2009, 113, 11119–11125. [Google Scholar] [CrossRef]
- Silvestrini, M.; Schiavuta, P.; Scopece, P.; Pecchielan, G.; Moretto, L.M.; Ugo, P. Modification of nanoelectrode ensembles by thiols and disulfides to prevent non specific adsorption of proteins. Electrochim. Acta 2011, 56, 7718–7724. [Google Scholar] [CrossRef]
- Pozzi Mucelli, S.; Zamuner, M.; Tormen, M.; Stanta, G.; Ugo, P. Nanoelectrode ensembles as recognition platform for electrochemical immunosensors. Biosens. Bioelectron. 2008, 23, 1900–1903. [Google Scholar] [CrossRef] [PubMed]
- Fayazfar, H.; Afshar, A.; Dolati, A. Electrodeposition of various Au nanostructures on aligned carbon nanotubes as highly sensitive nanoelectrode ensembles. J. Mater. Eng. Perform. 2015, 24, 2005–2015. [Google Scholar] [CrossRef]
- Mohamed, N.M.; Irshad, M.I.; Abdullah, M.Z.; Saheed, M.S.M. Novel growth of carbon nanotubes on nickel nanowires. Diamond Relat. Mater. 2016, 65, 59–64. [Google Scholar] [CrossRef]
- Malhotra, R.; Patel, V.; Vaqué, J.P.; Gutkind, J.S.; Rusling, J.F. Ultrasensitive electrochemical immunosensor for oral cancer biomarker IL-6 using carbon nanotube forest electrodes and multilabel amplification. Anal. Chem. 2010, 82, 3118–3123. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, R.; Papadimitrakopoulos, F.; Rusling, J.F. Sequential layer analysis of protein immunosensors based on single wall carbon nanotube forests. Langmuir 2010, 26, 15050–15056. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, A.; Shahrokhian, S.; Iraji zad, A.; Mohajerzadeh, S.; Vosoughi, M.; Darbari, S.; Koohsorkhi, J.; Mehran, M. Fabrication of sensitive glutamate biosensor based on vertically aligned CNT nanoelectrode array and investigating the effect of CNTs density on the electrode performance. Anal. Chem. 2012, 84, 5932–5938. [Google Scholar] [CrossRef] [PubMed]
- Periyakaruppan, A.; Gandhiraman, R.P.; Meyyappan, M.; Koehne, J.E. Label-free detection of cardiac troponin I using carbon nanofiber based nanoelectrode arrays. Anal. Chem. 2013, 85, 3858–3863. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Rivera, M.; Zaibaq, N.G.; Wilson, L.J. Toward carbon nanotube-based imaging agents for the clinic. Biomaterials 2016, 101, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.I.; Jamshaid, U.; Jamshaid, T.; Zafar, N.; Fessi, H.; Elaissari, A. Carbon nanotubes from synthesis to in vivo biomedical applications. Int. J. Pharm. 2016, 501, 278–299. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Miao, J.; Yang, Z.; Xiao, F.X.; Yang, H.B.; Liu, B.; Yang, Y. Carbon nanotube catalysts: Recent advances in synthesis, characterization and applications. Chem. Soc. Rev. 2015, 44, 3295–3346. [Google Scholar] [CrossRef] [PubMed]
- Tîlmaciu, C.M.; Morris, M.C. Carbon nanotube biosensors. Front Chem. 2015, 3, 59–79. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.P.; Martin, C.R. Fabrication and evaluation of nanoelectrode ensembles. Anal. Chem. 1995, 67, 1920–1928. [Google Scholar] [CrossRef]
- Possin, G.E. A method for forming very small diameter wires. Rev. Sci. Instrum. 1970, 41, 772–774. [Google Scholar] [CrossRef]
- Williams, W.D.; Giordano, N. Fabrication of 80 A° metal wires. Rev. Sci. Instrum. 1984, 55, 410–412. [Google Scholar] [CrossRef]
- Routkevitch, D.; Bigioni, T.; Moskovits, M.; Xu, J.M. Electrochemical fabrication of CdS nanowire arrays in porous anodic aluminum oxide template. J. Phys. Chem. C 1996, 100, 14037–14047. [Google Scholar] [CrossRef]
- Schoenberger, C.; van der Zande, B.M.I.; Fokkink, L.G.J.; Henny, M.; Schmid, C.; Kruger, M.; Bachtold, A.; Huber, R.; Birk, H.; Staufer, U. Template synthesis of nanowires in porous polycarbonate membranes: Electrochemistry and morphology. J. Phys. Chem. B 1997, 101, 5497–5505. [Google Scholar] [CrossRef]
- Penner, R.M.; Martin, C.R. Preparation and electrochemical characterization of ultramicroelectrode ensembles. Anal. Chem. 1987, 59, 2625–2630. [Google Scholar] [CrossRef]
- De Leo, M.; Pereira, F.C.; Moretto, L.M.; Scopece, P.; Polizzi, S.; Ugo, P. Towards a better understanding of gold electroless deposition in track-etched templates. Chem. Mater. 2007, 19, 5955–5964. [Google Scholar] [CrossRef]
- Gilliam, R.J.; Thorpe, S.J.; Kirk, D.J.W. A nucleation and growth study of gold nanowires and nanotubes in polymeric membranes. Appl. Electrochem. 2006, 37, 233–239. [Google Scholar] [CrossRef]
- Konishi, Y.; Motoyama, M.; Matsushima, H.; Fukunaka, Y.; Ishii, R.; Ito, Y. Electrodeposition of Cu nanowire arrays with a template. J. Electroanal. Chem. 2003, 559, 149–153. [Google Scholar] [CrossRef]
- Stortini, A.M.; Moretto, L.M.; Mardegan, A.; Ongaro, M.; Ugo, P. Arrays of copper nanowire electrodes: Preparation, characterization and application as nitrate sensor. Sens. Actuators B Chem. 2015, 207, 186–192. [Google Scholar] [CrossRef]
- Gambirasi, A.; Cattarin, S.; Musiani, M.; Vázquez-Gómez, L.; Verlato, E. Direct electrodeposition of metal nanowires on electrode surface. Electrochim. Acta 2011, 56, 8582–8588. [Google Scholar] [CrossRef]
- Motoyama, M.; Fukunaka, Y.; Sakka, T.; Ogata, Y.H.; Kikuchi, S. Electrochemical processing of Cu and Ni nanowire arrays. J. Electroanal. Chem. 2005, 584, 84–91. [Google Scholar] [CrossRef]
- Piraux, L.; Duboix, S.; Champagne, S. Template synthesis of nanoscale materials using the membrane porosity. Nucl. Instrum. Methods Phys. Res. Sect. B 1997, 131, 357–363. [Google Scholar] [CrossRef]
- Chiriac, H.; Moga, A.E.; Urse, M.; Ovari, T.A. Preparation and magnetic properties of electrodeposited magnetic nanowires. Sens. Actuators A Phys. 2003, 106, 348–351. [Google Scholar] [CrossRef]
- Pirota, K.R.; Navas, D.; Hernandez-Vélez, M.; Nielsch, K.; Vasquez, M. Novel magnetic materials prepared by electrodeposition techniques: Arrays of nanowires and multi-layered microwires. J. Alloy Compd. 2004, 369, 18–26. [Google Scholar] [CrossRef]
- Platt, M.; Dryfeand, R.A.W.; Robaerts, E.P.L. Structural and electrochemical characterisation of Pt and Pd nanoparticles electrodeposited at the liquid/liquid interface. Electrochim. Acta 2004, 49, 3937–3945. [Google Scholar] [CrossRef]
- Prieto, A.L.; Sander, M.S.; Gonzalez, M.S.M.; Gronsky, R.; Sands, T.; Stacy, A.M. Electrodeposition of ordered Bi2Te3 nanowire arrays. J. Am. Chem. Soc. 2001, 123, 7160–7161. [Google Scholar] [CrossRef] [PubMed]
- Bograchev, D.A.; Volgin, V.M.; Davydov, A.D. Simulation of inhomogeneous pores filling in template electrodeposition of ordered metal nanowire arrays. Electrochim. Acta 2013, 112, 279–286. [Google Scholar] [CrossRef]
- Bograchev, D.A.; Volgin, V.M.; Davydov, A.D. Modeling of metal electrodeposition in the pores of anodic aluminum oxide. Russ. J. Electrochem. 2015, 51, 799–806. [Google Scholar] [CrossRef]
- Bograchev, D.A.; Volgin, V.M.; Davydov, A.D. Simple model of mass transfer in template synthesis of metal ordered nanowire arrays. Electrochim. Acta 2013, 96, 1–7. [Google Scholar] [CrossRef]
- Volgin, V.M.; Davydov, A.D. Mass-transfer problems in the electrochemical systems. Russ. J. Electrochem. 2012, 48, 565–569. [Google Scholar] [CrossRef]
- Yu, C.; Xie, Y.; Xu, T.; Chen, Y.; Li, X.; Li, W.; Liu, B.; Zhang, X. Growth kinetics of nickel crystals in nanopores. Crystal Growth Des. 2009, 9, 3840–3843. [Google Scholar] [CrossRef]
- Philippe, L.; Kacem, N.; Michler, J. Electrochemical deposition of metals inside high aspect ratio nanoelectrode array: Analytical current expression and multidimensional kinetic model for cobalt nanostructure synthesis. J. Phys. Chem. C 2007, 111, 5229–5235. [Google Scholar] [CrossRef]
- Paunovic, M.; Schlesinger, M. Modern Electroplating; Wiley: New York, NY, USA, 2000. [Google Scholar]
- Pereira, F.C.; Moretto, L.M.; De Leo, M.; Boldrin Zanoni, M.V.; Ugo, P. Gold nanoelectrode ensembles for direct trace electroanalysis of iodide. Anal. Chim. Acta 2006, 575, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Jirage, K.B.; Hulteen, J.C.; Martin, C.R. Nanotubule-based molecular-filtration membranes. Science 1997, 278, 655–658. [Google Scholar] [CrossRef]
- Hulteen, J.C.; Jirage, K.B.; Martin, C.R. Introducing chemical transport selectivity into gold nanotubule membranes. J. Am. Chem. Soc. 1998, 120, 6603–6604. [Google Scholar] [CrossRef]
- Jirage, K.B.; Hulteen, J.C.; Martin, C.R. Effects of thiol chemisorptions on the transport properties of gold nanotubule membranes. Anal. Chem. 1999, 71, 4913–4918. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Martin, C.R. Highly-sensitive methods for electroanalytical chemistry based on nanotubule membranes. Anal. Chem. 1999, 71, 3665–3672. [Google Scholar] [CrossRef] [PubMed]
- Bercu, B.; Enculescu, I.; Spohr, R. Copper tubes prepared by electroless deposition in ion track templates. Nucl. Instrum. Methods Phys. Res. Sect. B 2004, 225, 497–502. [Google Scholar] [CrossRef]
- Dryfe, R.A.W.; Simm, A.O.; Kralj, B. Electroless deposition of palladium at bare and templated liquid/liquid interfaces. J. Am. Chem. Soc. 2003, 125, 13014–13015. [Google Scholar] [CrossRef] [PubMed]
- Tai, Y.L.; Teng, H. Template synthesis and electrochemical characterization of nickel based tubule electrode arrays. Chem. Mater. 2004, 16, 338–342. [Google Scholar] [CrossRef]
- Ugo, P.; Moretto, L.M. Template deposition of metals. In Handbook of Electrochemistry; Zoski, C.G., Ed.; Elsevier: Amsterdam, The Netherlands, 2007; pp. 678–709. [Google Scholar]
- Moretto, L.M.; Pepe, N.; Ugo, P. Voltammetry of redox analytes at trace concentrations with nanoelectrodes ensembles. Talanta 2004, 62, 1055–1060. [Google Scholar] [CrossRef] [PubMed]
- Habtamu, H.B.; Ugo, P. Miniaturized enzymatic biosensor via biofunctionalization of the insulator of nanoelectrode ensembles. Electroanalysis 2015, 27, 2187–2193. [Google Scholar] [CrossRef]
- Ongaro, M.; Gambirasi, A.; Favaro, M.; Ugo, P. Electrochemical synthesis and characterization of hierarchically branched ZnO nanostructures on ensembles of gold nanowires. Electrochim. Acta 2012, 78, 539–546. [Google Scholar] [CrossRef]
- Ongaro, M.; Gambirasi, A.; Ugo, P. Closed bipolar electrochemistry for the low-potential asymmetrical functionalization of micro- and nanowires. ChemElectroChem 2016, 3, 450–456. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L. Electrochemical Methods; VCH: Weinheim, Germany, 2000. [Google Scholar]
- Dickinson, E.J.F.; Compton, R.G. Diffuse double layer at nanoelectrodes. Phys. Chem. Lett. C 2009, 113, 17585–17589. [Google Scholar] [CrossRef]
- Chen, S.; Liu, Y. Electrochemistry at nanometer-sized electrodes. Phys. Chem. Chem. Phys. 2014, 16, 635–652. [Google Scholar] [CrossRef] [PubMed]
- Henstridge, M.C.; Compton, R.G. Mass transport to micro- and nanoelectrodes and their arrays: A review. Chem. Rec. 2011, 12, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Beriet, C.; Ferrigno, R.; Girault, H.H. Cyclic voltammetry at a regular microdisc electrode array. J. Electroanal. Chem. 2001, 502, 138–145. [Google Scholar] [CrossRef]
- Hulteen, J.C.; Menon, V.P.; Martin, C.R. Template preparation of nanoelectrode ensembles achieving the ‘pure-radial’ electrochemical response limiting case. J. Chem. Soc. Faraday Trans. 1996, 92, 4029–4032. [Google Scholar] [CrossRef]
- Cheng, J.F.; Whitley, L.D.; Martin, C.R. Ultramicroelectrode ensembles. Comparison of experimental and theoretical responses and evaluation of electroanalytical detection limits. Anal. Chem. 1989, 61, 762–766. [Google Scholar] [CrossRef]
- Guo, J.; Lindner, E. Cyclic voltammograms at coplanar and shallow recessed microdisk electrode arrays: Guidelines for design and experiment. Anal. Chem. 2009, 81, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Davies, T.J.; Compton, R.G. The cyclic and linear sweep voltammetry of regular and random arrays of microdisc electrodes: Theory. J. Electroanal. Chem. 2005, 585, 63–82. [Google Scholar] [CrossRef]
- Huang, X.J.; O’Mahony, A.M.; Compton, R.G. Microelectrode arrays for electrochemistry: Approaches to fabrication. Small 2009, 7, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Amatore, C.; Oleinik, A.I.; Svir, I. Numerical simulation of diffusion processes at recessed disk microelectrode arrays using the quasi-conformal mapping approach. Anal. Chem. 2009, 81, 4397–4405. [Google Scholar] [CrossRef] [PubMed]
- Moretto, L.M.; Tormen, M.; De Leo, M.; Carpentiero, A.; Ugo, P. Polycarbonate-based ordered arrays of electrochemical nanoelectrodes obtained by e-beam lithography. Nanotechnology 2011, 22, 185305–185311. [Google Scholar] [CrossRef] [PubMed]
- Sliusarenko, O.; Oleinick, A.; Svir, I.; Amatore, C. Development and validation of an analytical model for predicting chronoamperometric responses of random arrays of micro- and nanodisk electrodes. ChemElectroChem 2015, 2, 1279–1291. [Google Scholar] [CrossRef]
- Ugo, P.; Moretto, L.M.; De Leo, M.; Doherty, A.P.; Vallese, C.; Pentlavalli, S. Diffusion regimes at nanoelectrode ensembles in different ionic liquids. Electrochim. Acta 2010, 55, 2865–2872. [Google Scholar] [CrossRef]
- Ugo, P.; Moretto, L.M.; Vezzà, F. Ionomer-coated electrodes and nanoelectrode ensembles as electrochemical environmental sensors: Recent advances and prospects. In Sensors Update; Baltes, H., Fedder, G.K., Korvink, J.G., Eds.; Wiley–VCH: Weinheim, Germany, 2003; Volume 12, pp. 121–140. [Google Scholar]
- Ugo, P.; Moretto, L.M.; Bellomi, S.; Menon, V.P.; Martin, C.R. Ion exchange voltammetry at polymer film coated nanoelectrode ensembles. Anal. Chem. 1996, 68, 4160–4165. [Google Scholar] [CrossRef] [PubMed]
- Brunetti, B.; Ugo, P.; Moretto, L.M.; Martin, C.R. Electrochemistry of phenothiazine and methylviologen biosensor electrontransfer mediators at nanoelectrode ensembles. J. Electroanal. Chem. 2000, 491, 166–174. [Google Scholar] [CrossRef]
- Errachid, A.; Mills, C.A.; Pla-Roca, M.; Lopez, M.J.; Villanueva, G.; Bausells, J.; Crespo, E.; Teixidor, F.; Samitier, J. Focused ion beam production of nanoelectrode arrays. Mater. Sci. Eng. C 2008, 28, 777–780. [Google Scholar] [CrossRef]
- Lanyon, Y.H.; De Marzi, G.; Watson, Y.E.; Quinn, A.J.; Gleeson, J.P.; Redmond, G.; Arrigan, D.W.M. Fabrication of nanopore array electrodes by focused ion beam milling. Anal. Chem. 2007, 79, 3048–3055. [Google Scholar] [CrossRef] [PubMed]
- Sandison, M.E.; Cooper, J.M. Nanofabrication of electrode arrays by electron-beam and nanoimprint lithographies. Lab Chip 2006, 6, 1020–1025. [Google Scholar] [CrossRef] [PubMed]
- Losilia, N.S.; Martinez, J.; Garcia, R. Large area nanoscale patterning of silicon surfaces by parallel local oxidation. Nanotechnology 2009, 20, 475304–475308. [Google Scholar] [CrossRef] [PubMed]
- Losilia, N.S.; Oxtoby, N.S.; Martinez, J.; Garcia, F.; Garcia, R.; Mas-Torrent, M.; Vecciana, J.; Rovia, C. Sub-50 nm positioning of organic compounds onto silicon oxide patterns fabricated by local oxidation nanolithography. Nanotechnology 2008, 19, 455308–455313. [Google Scholar] [CrossRef] [PubMed]
- Albonetti, C.; Martinez, J.; Losilia, N.S.; Greco, P.; Cavallini, M.; Borgatti, F.; Montecchi, M.; Pasquali, L.; Garcia, R.; Biscarini, F. Parallel-local anodic oxidation of silicon surfaces by soft stamps. Nanotechnology 2008, 19, 435303–435311. [Google Scholar] [CrossRef] [PubMed]
- Zamuner, M.; Pozzi Mucelli, S.; Tormen, M.; Stanta, G.; Ugo, P. Electrochemical nanobiosensors and protein detection. Eur. J. Nanomed. 2008, 1, 33–36. [Google Scholar] [CrossRef]
- Yang, N.; Foord, J.S.; Jiang, X. Diamond electrochemistry at the nanoscale: A review. Carbon 2016, 99, 90–110. [Google Scholar] [CrossRef]
- Xiao, L.; Streeter, I.; Wildgoose, G.G.; Compton, R.G. Fabricating random arrays of boron doped diamond nano-disc electrodes: Towards achieving maximum faradaic current with minimum capacitive charging. Sens. Actuators B Chem. 2008, 33, 118–127. [Google Scholar] [CrossRef]
- Hees, J.; Hoffmann, R.; Kriele, A.; Smirnov, W.; Obloh, H.; Glorer, K.; Raynor, B.; Driad, R.; Yang, N.; Williams, O.A.; et al. Nanocrystalline diamond nanoelectrode arrays and ensembles. ACS Nano 2011, 5, 3339–3346. [Google Scholar] [CrossRef] [PubMed]
- Virgilio, F.; Prasciolu, M.; Ugo, P.; Tormen, M. Development of electrochemical biosensors by e-beam lithography for medical diagnostics. Microelectron. Eng. 2013, 111, 320–324. [Google Scholar] [CrossRef]
- Liu, Y.; Holzinger, A.; Knittel, P.; Poltorak, L.; Gamero-Quijano, A.; Rickard, W.D.A.; Walcarius, A.; Herzog, G.; Kranz, C.; Arrigan, D.W.M. Visualization of diffusion within nanoarrays. Anal. Chem. 2016, 88, 6689–6695. [Google Scholar] [CrossRef] [PubMed]
- Dincer, C.; Laubender, E.; Hees, J.; Nebel, C.E.; Urban, G.; Heinze, J. SECM detection of single boron doped diamond nanodes and nanoelectrode arrays using phase-operated shear force technique. Electrochem. Commun. 2012, 24, 123–127. [Google Scholar] [CrossRef]
- Hees, J.; Hoffmann, R.; Yang, N.; Nebel, C.E. Diamond nanoelectrode arrays for the detection of surface sensitive adsorption. Chem. Eur. J. 2013, 19, 11287–11292. [Google Scholar] [CrossRef] [PubMed]
- Sentic, M.; Virgilio, F.; Zanut, A.; Manojlovic, D.; Arbault, S.; Tormen, M.; Sojic, N.; Ugo, P. Microscopic imaging and tuning of electrogenerated chemiluminescence with boron-doped diamond nanoelectrode arrays. Anal. Bioanal. Chem. 2016, 408, 7085–7094. [Google Scholar] [CrossRef] [PubMed]
- Amatore, C.; Saveant, J.M.; Tessier, D. Charge transfer at partially blocked surfaces. A model for the case of microscopic active and inactive sites. J. Electroanal. Chem. Interfacial Electrochem. 1983, 147, 39–51. [Google Scholar] [CrossRef]
- Greef, R.; Pea, R.; Peter, L.M.; Pletcher, D.; Robinson, J. Instrumental Methods in Electrochemistry; Ellis Horwood Ltd.: Chester, UK, 1985. [Google Scholar]
- Nicholson, R.S. Theory and application of cyclic voltammetry for measurement of electrode reaction kinetics. Anal. Chem. 1965, 37, 1351–1355. [Google Scholar] [CrossRef]
- Yu, S.; Li, N.; Wharton, J.; Martin, C.R. Nano wheat fields prepared by plasma-etching gold nanowire-containing membranes. Nano Lett. 2003, 3, 815–818. [Google Scholar] [CrossRef]
- Gasparac, R.; Taft, B.J.; Lapierre-Devlin, M.A.; Lazareck, A.D.; Xu, J.M.; Kelley, S.O. Ultrasensitive electrocatalytic DNA detection at two- and three-dimensional nanoelectrodes. J. Am. Chem. Soc. 2004, 126, 12270–12271. [Google Scholar] [CrossRef] [PubMed]
- Zoski, C.G.; Yang, N. Addressable nanoelectrode membrane arrays: Fabrication and steady-state behavior. Anal. Chem. 2007, 79, 1474–1484. [Google Scholar] [CrossRef] [PubMed]
- Ugo, P.; Moretto, L.M.; Scrosati, B. Electrodes|nanoelectrodes. In Encyclopedia of Electrochemical Power Sources; Garche, J., Dyer, C., Moseley, P., Ogumi, Z., Rand, D., Scrosati, B., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; Volume 2, pp. 92–102. [Google Scholar]
- Krishnamoorthy, K.; Zoski, C.G. Fabrication of 3D gold nanoelectrode ensembles by chemical etching. Anal. Chem. 2005, 77, 5068–5071. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Yan, P.; Sun, K.; Kirk, D.W. Tailor-made gold brush nanoelectrode ensembles modified with L-cysteine for the detection of daunorubicine. Electrochim. Acta 2008, 53, 8144–8148. [Google Scholar] [CrossRef]
- Cao, L.; Yan, P.; Sun, K.; Kirk, D.W. Gold 3D brush nanoelectrode ensembles with enlarged active area for the direct voltammetry of daunorubicin. Electroanalysis 2009, 21, 1183–1188. [Google Scholar] [CrossRef]
- Moretto, L.M.; Panero, S.; Scrosati, B.; Ugo, P. Templated ensembles of nanoelectrodes. In Handbook of Electrochemical Nanotechnology; Lin, Y., Nalwa, H.S., Eds.; American Scientific: Stevenson Ranch, CA, USA, 2009; Volume 1, pp. 87–105. [Google Scholar]
- Sliusarenko, O.; Oleinick, A.; Svir, I.; Amatore, C. Validating a central approximation in theories of regular electrode electrochemical arrays of various common geometries. Electroanalysis 2015, 27, 980–991. [Google Scholar] [CrossRef]
- Ugo, P.; Moretto, L.M.; Ongaro, M.; Scrosati, B. Electrodes|Nanoelectrodes; Elsevier: Waltham, MA, USA, 2015. [Google Scholar]
- Ugo, P.; Moretto, L.M.; Silvestrini, M.; Pereira, F.C. Nanoelectrode ensembles for the direct voltammetric determination of trace iodide in water. Int. J. Environ. Anal. Chem. 2010, 90, 747–759. [Google Scholar] [CrossRef]
- Ugo, P.; Pepe, N.; Moretto, L.M.; Battagliarin, M. Direct voltammetry of cytochrome c at trace concentrations with nanoelectrode ensembles. J. Electroanal. Chem. 2003, 560, 51–58. [Google Scholar] [CrossRef]
- Mardegan, A.; Scopece, P.; Lamberti, F.; Meneghetti, M.; Moretto, L.M.; Ugo, P. Electroanalysis of trace inorganic arsenic with gold nanoelectrode ensembles. Electroanalysis 2012, 24, 798–806. [Google Scholar] [CrossRef]
- Mardegan, A.; Borgo, S.D.; Scopece, P.; Moretto, L.M.; Hočevar, S.B.; Ugo, P. Bismuth modified gold nanoelectrode ensemble for stripping voltammetric determination of lead. Electrochem. Commun. 2012, 24, 28–31. [Google Scholar] [CrossRef]
- Viswanathan, S.; Rani, C.; Delerue-Matos, C. Ultrasensitive detection of ovarian cancer marker using immunoliposomes and gold nanoelectrodes. Anal. Chim. Acta 2012, 726, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Silvestrini, M.; Fruk, L.; Ugo, P. Functionalized ensembles of nanoelectrodes as affinity biosensors for DNA hybridization detection. Biosens. Bioelectron. 2013, 40, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Fruk, L.; Müller, J.; Weber, G.; Narvaez, A.; Dominguez, E.; Niemeyer, C.M. DNA-directed immobilization of horseradish peroxidase–DNA conjugates on microelectrode arrays: Towards electrochemical screening of enzyme libraries. Chem. Eur. J. 2007, 13, 5223–5231. [Google Scholar] [CrossRef] [PubMed]
- Bottari, F.; Oliveri, P.; Ugo, P. Electrochemical immunosensor based on ensemble of nanoelectrodes for immunoglobulin IgY detection: Application to identify hen’s egg yolk in tempera paintings. Biosens. Bioelectron. 2014, 52, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Mollamahale, Y.B.; Ghorbani, M.; Ghalkhani, M.; Vossoughi, M.; Dolati, A. Highly sensitive 3D gold nanotube ensembles: Application to electrochemical determination of metronidazole. Electrochim. Acta 2013, 106, 288–292. [Google Scholar] [CrossRef]
- Mardegan, A.; Scopece, P.; Ugo, P.; Moretto, L.M. Ensembles of gold nanowires for the anodic stripping voltammetric determination of inorganic arsenic. J. Nanosci. Nanotechnol. 2014, 15, 1–6. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, Y.; Chenb, J.; Zeng, Y. Amperometric biosensor based on 3D ordered freestanding porous Pt nanowire array electrode. Nanoscale 2012, 4, 6025–6031. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, Q.; Zhao, W.; Chu, W.; Zheng, J. Array of recessed gold nanoelectrodes formed with polymethylmethacrylate for individual detection of ascorbic acid, dopamine and uric acid. Electrochim. Acta 2016, 212, 25–31. [Google Scholar] [CrossRef]
- Dincer, C.; Ktaich, R.; Laubender, E.; Hees, J.J.; Kieninger, J.; Nebel, C.E.; Heinze, J.; Urban, G.A. Nanocrystalline boron-doped diamond nanoelectrode arrays for ultrasensitive dopamine detection. Electrochim. Acta 2015, 185, 101–106. [Google Scholar] [CrossRef]
- De Oliveira, R.A.G.; Zanoni, M.V.B. Highly ordered TiO2 nanotubes for electrochemical sensing of hair dye basic brown 17. Electroanalysis 2013, 25, 2507–2514. [Google Scholar] [CrossRef]
- Lee, J.; Hong, M.H.; Han, S.; Na, J.; Kim, I.; Kwon, Y.J.; Lim, Y.b.; Choi, H.J. Sensitive and selective detection of HIV-1 RRE RNA using vertical silicon nanowire electrode array. Nanoscale Res. Lett. 2016, 11, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.M.; Kim, Y.H.; Cho, Y. A nanowire-based label-free immunosensor: Direct incorporation of a PSA antibody in electropolymerized polypyrrole. Biosens. Bioelectron. 2014, 57, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Cao, L.; Li, Q.; Ma, K.; Yanb, P.; Kirk, D.W. Fabrication and modeling of an ultrasensitive label free impedimetric immunosensor for Aflatoxin B1 based on poly(ophenylenediamine) modified gold 3D nano electrode ensembles. RSC Adv. 2015, 5, 55209–55217. [Google Scholar] [CrossRef]
- Viswanathan, S.; Rani, C.; Ribeiro, S.; Matos, C.D. Molecular imprinted nanoelectrodes for ultra sensitive detection of ovarian cancer marker. Biosens. Bioelectron. 2012, 33, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Silvestrini, M.; Fruk, L.; Moretto, L.M.; Ugo, P. Detection of DNA hybridization by methylene blue electrochemistry at activated nanoelectrode ensembles. J. Nanosci. Nanotechnol. 2015, 15, 3437–3442. [Google Scholar] [CrossRef] [PubMed]
- Habtamu, H.B.; Sentic, M.; Silvestrini, M.; Leo, L.D.; Not, T.; Arbault, S.; Manojlovic, D.; Sojic, N.; Ugo, P. A sensitive electrochemiluminescence immunosensor for celiac disease diagnosis based on nanoelectrode ensembles. Anal. Chem. 2015, 87, 12080–12087. [Google Scholar] [CrossRef] [PubMed]
- Sultana, R.; Reza, N.; Kay, N.J.; Schmueser, I.; Walton, A.J.; Terry, J.G.; Mount, A.R.; Freeman, N.J. Practical implications of using nanoelectrodes for bioanalytical measurements. Electrochim. Acta 2014, 126, 98–103. [Google Scholar] [CrossRef]
- Yu, J.; Zhang, Y.; Liu, S. Enzymatic reactivity of glucose oxidase confined in nanochannels. Biosens. Bioelectron. 2014, 55, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Luo, P.; Xin, C.; Cao, X.; Zhang, Y.; Liu, S. Quantitative evaluation of biological reaction kinetics in confined nanospaces. Anal. Chem. 2014, 86, 8129–8135. [Google Scholar] [CrossRef] [PubMed]
- Selvam, A.P.; Prasad, S.; Barrett, T.W.; Kazmierczak, S.C. Electrical nanowell diagnostics sensors for rapid and ultrasensitive detection of prostate-specific antigen. Nanomedicine (Lond.) 2015, 10, 2527–2536. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Li, H.; Cui, D.; Zhang, Y.; Liu, S. Enhanced enzymatic reactivity for electrochemically driven drug metabolism by confining cytochrome P450 enzyme in TiO2 nanotube arrays. Anal. Chem. 2014, 86, 8003–8009. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Tian, Y.W.; Shao, W.; Yuan, C.G.; Wang, K.; Xia, X.H. Solution pH regulating mass transport in highly ordered nanopore array electrode. Electrochem. Commun. 2014, 42, 1–5. [Google Scholar] [CrossRef]
- Dickey, M.D.; Lipomi, D.J.; Bracher, P.J.; Whitesides, G.M. Electrically addressable parallel nanowires with 30 nm spacing from micromolding and nanoskiving. Nano Lett. 2008, 8, 4568–4573. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, P.U.; Chen, H.; Siddiqui, S.; Weinrich, J.A.P.; Jejelowo, A.; Li, J.; Meyyappan, M. Wafer-scale fabrication of patterned carbon nanofiber nanoelectrode arrays: A route for development of multiplexed, ultrasensitive disposable biosensors. Biosens. Bioelectron. 2009, 24, 2818–2824. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Keilbach, A.; Codinachs, L.M.; Nielsch, K.; Abstreiter, G.; Morral, A.F.I.; Bein, T. Multiple nanowire species synthesized on a single chip by selectively addressable horizontal nanochannels. Nano Lett. 2010, 10, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Dawson, K.; Wahl, A.; Barry, S.; Barrett, C.; Sassiat, N.; Quinn, A.J.; O’Riordan, A. Fully integrated on-chip nano-electrochemical devices for electroanalytical applications. Electrochim. Acta 2014, 115, 239–246. [Google Scholar] [CrossRef]




| Nanosensor Type | Nanosense Platform | Target | Notes | Ref. |
|---|---|---|---|---|
| NEEs | Ensembles of copper nanowire electrodes | NO3− | Sensor durability and reproducibility are achieved by using a thin Nafion interlayer | [29] |
| NEEs | 3D-gold nanotubes | Metronidazole | DL 0.1 nM | [110] |
| NEEs | 3D-ensembles of gold nanowires electrodes | Inorganic Arsenic | Anodic stripping voltammetric determination of As(III), DL 0.08 µg·L−1, linear range up to 20 µg·L−1 | [111] |
| NEAs | 3D- ordered freestanding porous platinum (Pt) nanowire array electrode | Glucose, H2O2 | Effect of granular and rougher porous nanowire surface on the bioactivity of glucose oxidase is examined | [112] |
| NEAs | Recessed NEAs with polymethylmethacrylate coated gold planar electrodes | Dopamine, Ascorbic acid, Uric acid | Sensing platform for detection of components in a mixture of analytes | [113] |
| NEAs | Nanocrystalline boron-doped diamond nanoelectrode arrays(BDD-NEAs) | Dopamine | Appropriate termination by choosing oxygen (O-) terminated BDD-NEAs, DL 100 nM | [114] |
| NEAs | Self-organized Ti/TiO2 nanotubular array | Hair dye basic brown 17 | DL 1.3 × 10−7 M | [115] |
| NEAs | Pt nanoband electrode | Detection of, ferrocene carboxylic acid, hydrogen peroxide and 4-aminophenol | Chronoamperometric and cyclic voltammetric detection | [122] |
| Nanosensor Type | Nanosense Platform | Target | Notes | Ref. |
|---|---|---|---|---|
| NEEs | Nonconductive PC component of the NEE is used for immobilizing glucose oxidase | Glucose | DL 36 µM | [54] |
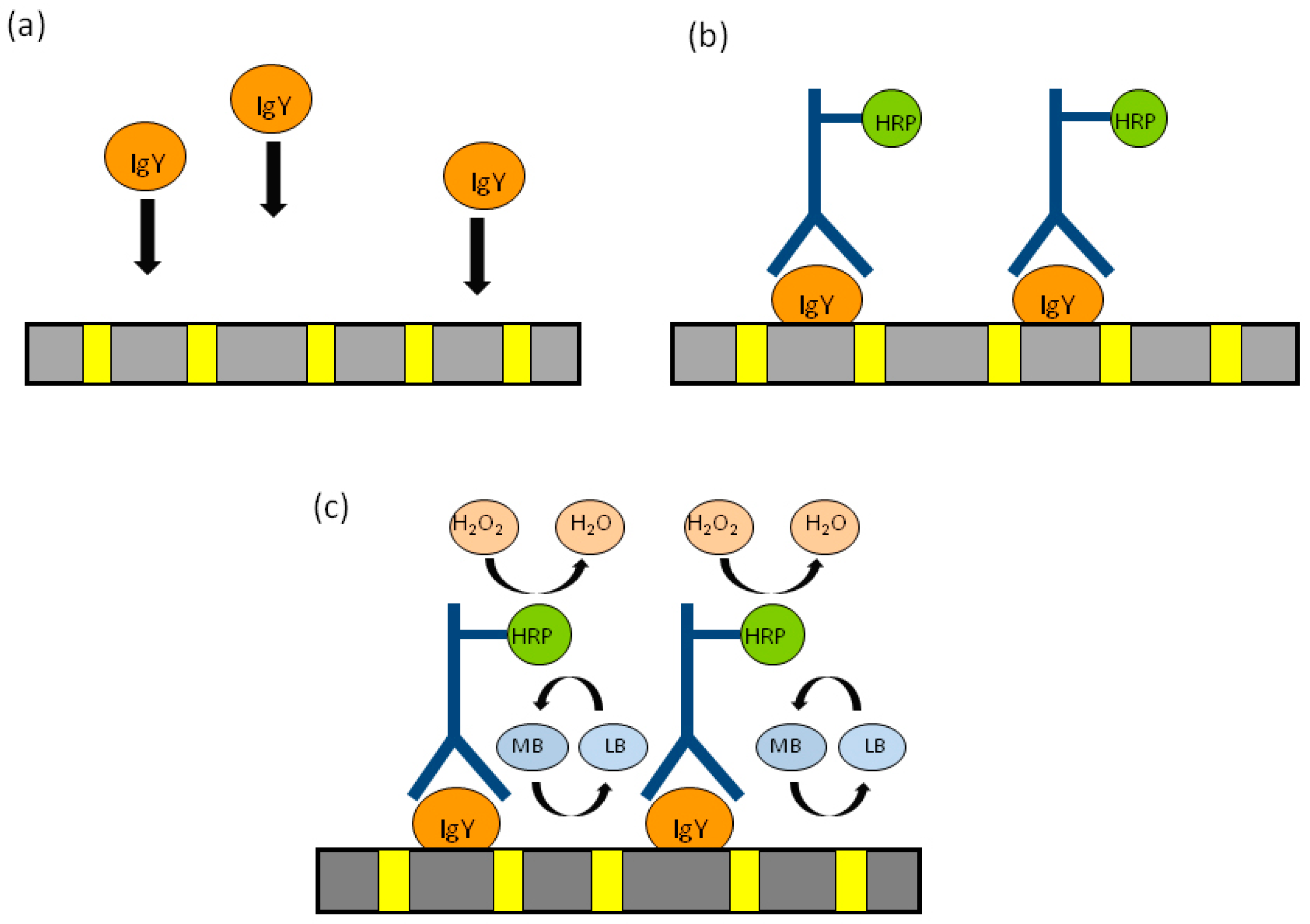
| NEEs | Capturing proteins by interaction with the PC membrane of the NEE | Immunoglobulin IgY | Application to identify hen’s egg yolk in tempera paintings | [109] |
| NEAs | Au-coated vertical silicon nanowire electrode array | HIV-1 Rev response element (RRE) RNA | Immobilized artificial peptides for the recognition of HIV-1 RRE. DL 1.513 fM | [116] |
| NEAs | 3D -gold nanowire array modified with electrodeposition of anti-PSA-doped Ppy polymers | Prostate-specific antigen | Linear response: 10 fg·mL−1 to 10 ng·mL−1, DL 0.3 fg·mL−1 | [117] |
| NEEs | 3D-gold nanoelectrode ensembles modified with poly- (o-phenylenediamine) | Aflatoxin B1 | Cyclic voltammetry and electrochemical impedance spectroscopy have been employed. DL 0.019 ng·mL−1 | [118] |
| NEEs | 3D-gold nanoelectrode ensembles (3D-NEEs) modified with molecular imprinted polyphenol | Epithelial ovarian cancer antigen-125 (CA 125) | DL 0.5 U·mL−1 | [119] |
| NEEs | Polymer surface of nanoelectrode ensembles bio-functionalized with DNA | DNA hybridization | Effect of the functionalization of the NEEs with the ss-DNA probe by measuring the changes in the methylene blue reduction signal is studied | [120] |
| EEs | Au nanodisk electrodes act as electrochemical transducers for initiating the ECL emission while PC is exploited for biorecogniton and to bind the luminescent label | Celiac disease diagnosis | Direct oxidation of tri-n-propylamine at NEEs generates ECL by a ruthenium label bound on antibody, DL 1.5 ng·mL−1. Application to human serum analysis | [121] |
| Nanosensor Type | Nanosense Platform | Target | Notes | Ref. |
|---|---|---|---|---|
| Nano channel | Inner walls of the PAA nanochannels are functionalized with GOx; a bottom Au layer acts as working electrode. | Enzymatic reactivity of glucose oxidase | Activity and stability of the glucose oxidase immobilized in the nanochannels is largely enhanced | [123] |
| Nano channel | Functionalization of PAA nanochannels with DNA | To monitor online immunological reactions and biosensing process in the nanochannels | Study to evaluate the speed of antibody and the immunological reaction progress in nanochannels | [124] |
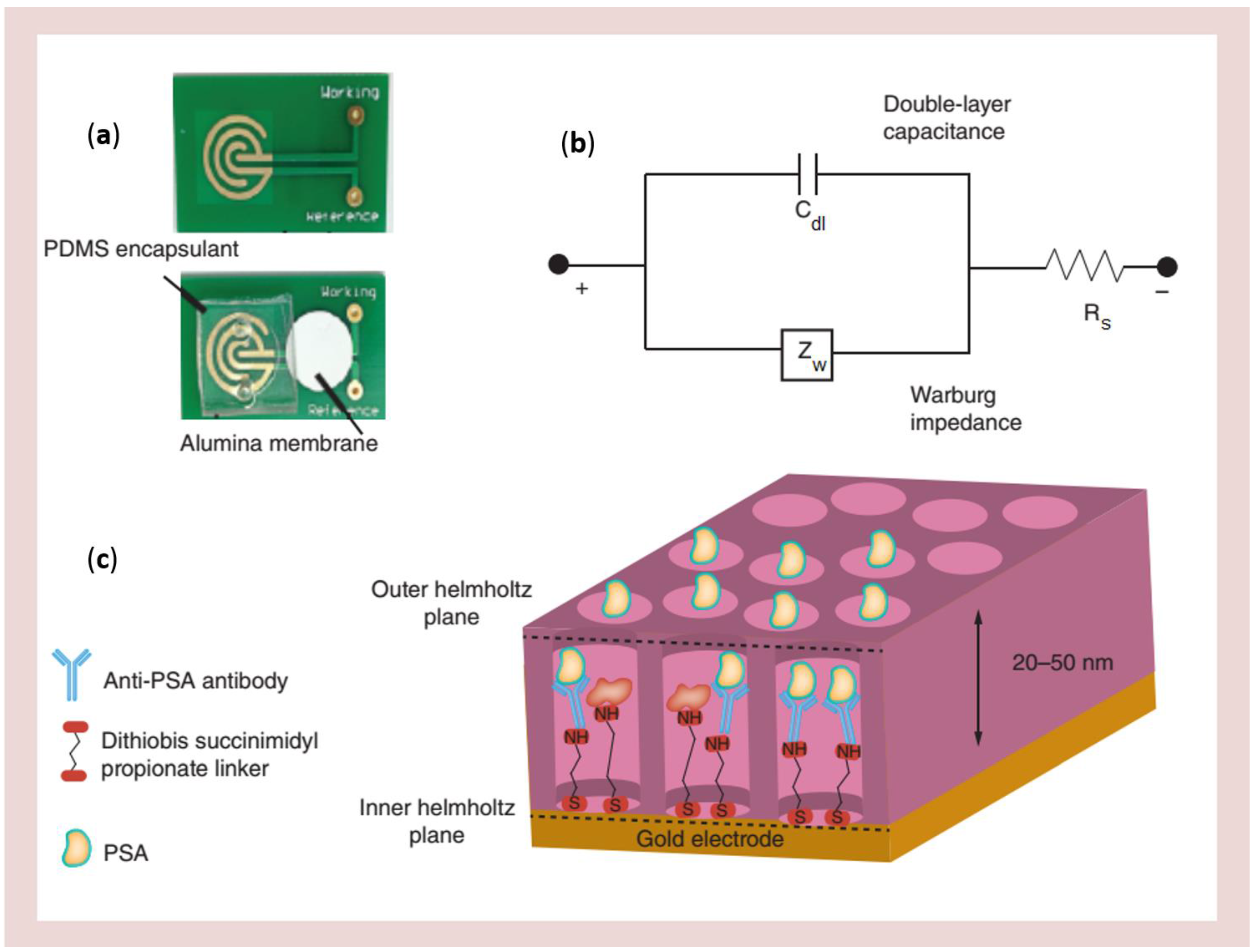
| Nanowell array | Integration of PAA membranes on printed circuit board platforms | Prostate-specific antigen (PSA) | PSA detection between 0.01 and 1000 ng·mL−1 | [125] |
| Nanotube array | Electrodepositing Au nanoparticles on the inner wall of TiO2 nanotube arrays | Cytochrome P450 2C9 enzyme as a model enzyme and tolbutamide as a model substrate | Excellent enzymatic activity, high affinity, and metabolic efficiency for tolbutamide | [126] |
| Nanopore array | Electrodeposition of gold nanowires in porous anodic alumina membranes by alternating current | Investigation of the electrochemistry property of nanopore array electrode | Voltammetric limiting current can be regulated by surface charge changes on the PAA walls by changing the solution pH | [127] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karimian, N.; Moretto, L.M.; Ugo, P. Nanobiosensing with Arrays and Ensembles of Nanoelectrodes. Sensors 2017, 17, 65. https://doi.org/10.3390/s17010065
Karimian N, Moretto LM, Ugo P. Nanobiosensing with Arrays and Ensembles of Nanoelectrodes. Sensors. 2017; 17(1):65. https://doi.org/10.3390/s17010065
Chicago/Turabian StyleKarimian, Najmeh, Ligia M. Moretto, and Paolo Ugo. 2017. "Nanobiosensing with Arrays and Ensembles of Nanoelectrodes" Sensors 17, no. 1: 65. https://doi.org/10.3390/s17010065
APA StyleKarimian, N., Moretto, L. M., & Ugo, P. (2017). Nanobiosensing with Arrays and Ensembles of Nanoelectrodes. Sensors, 17(1), 65. https://doi.org/10.3390/s17010065








