Organophosphorous Pesticide Detection in Olive Oil by Using a Miniaturized, Easy-to-Use, and Cost-Effective Biosensor Combined with QuEChERS for Sample Clean-Up
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Apparatus
2.2. Preparation of SPE
2.3. Preparation of BChE Biosensor
2.4. Paraoxon Determination Using Biosensor
2.5. Paraoxon Measurement in Olive Oil Extract Using HPLC/MS
3. Results and Discussion
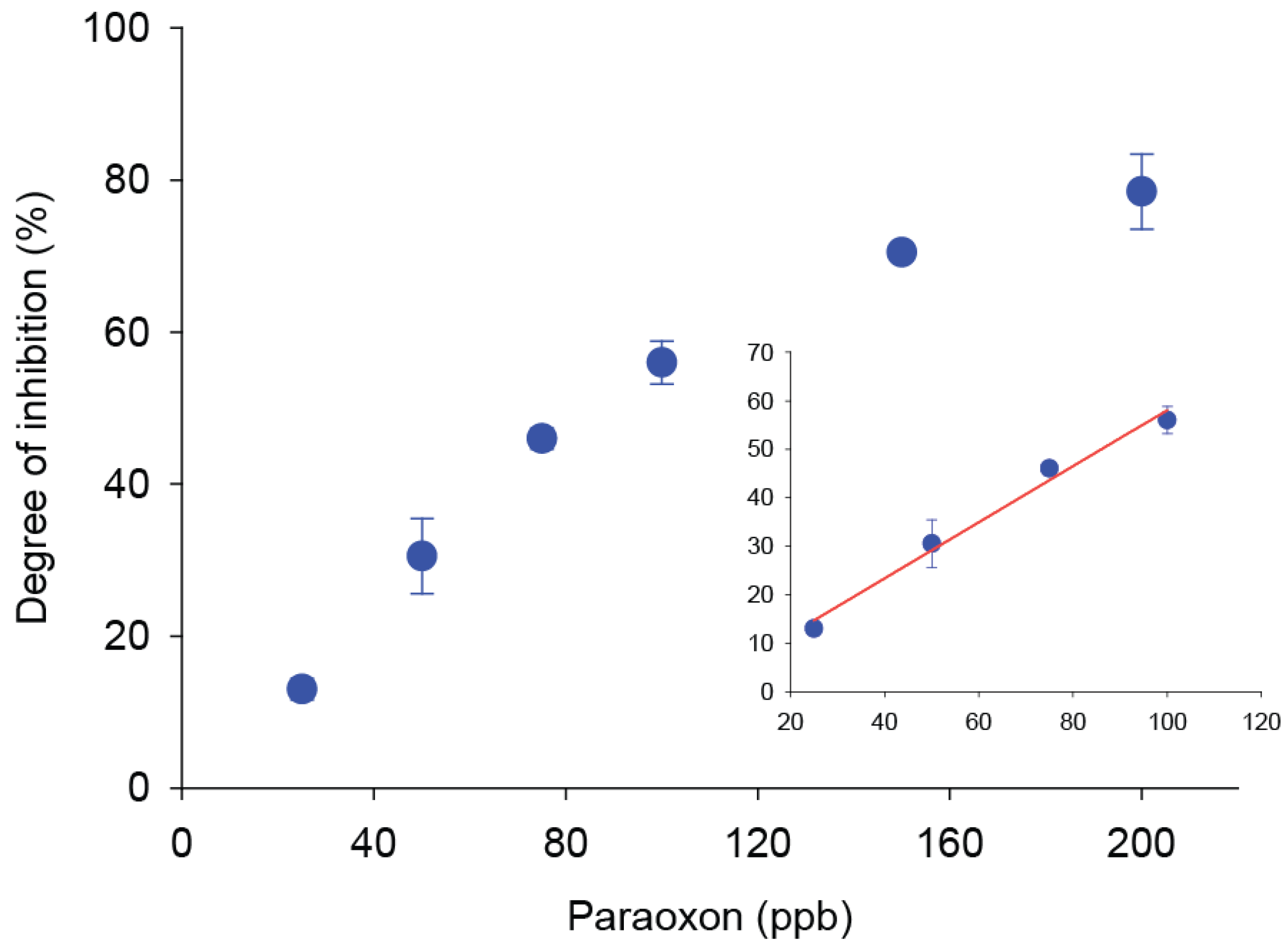
3.1. Optimization of Paraoxon Analysis in Standard Solutions
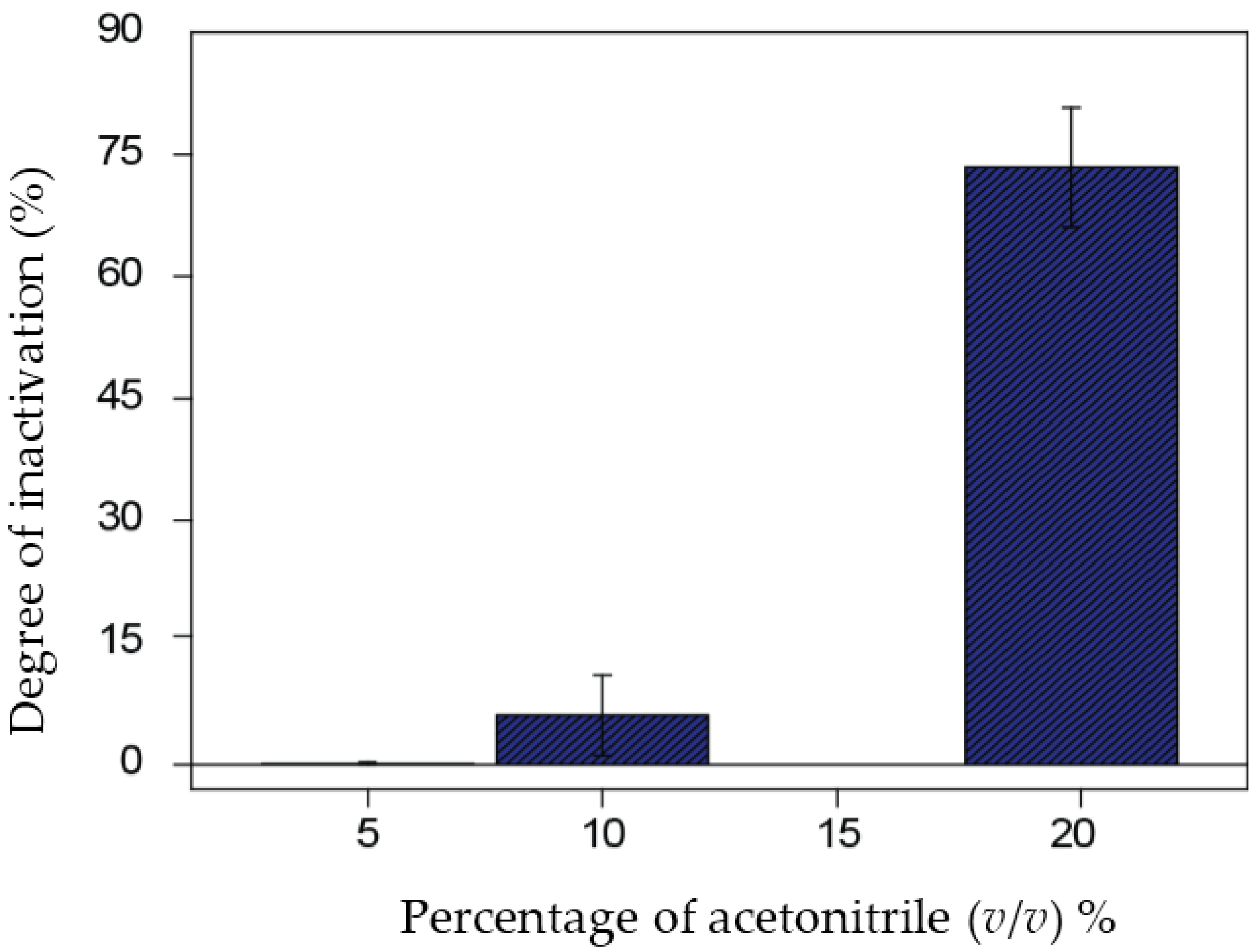
3.2. Inactivation Studies in the Presence of Acetonitrile
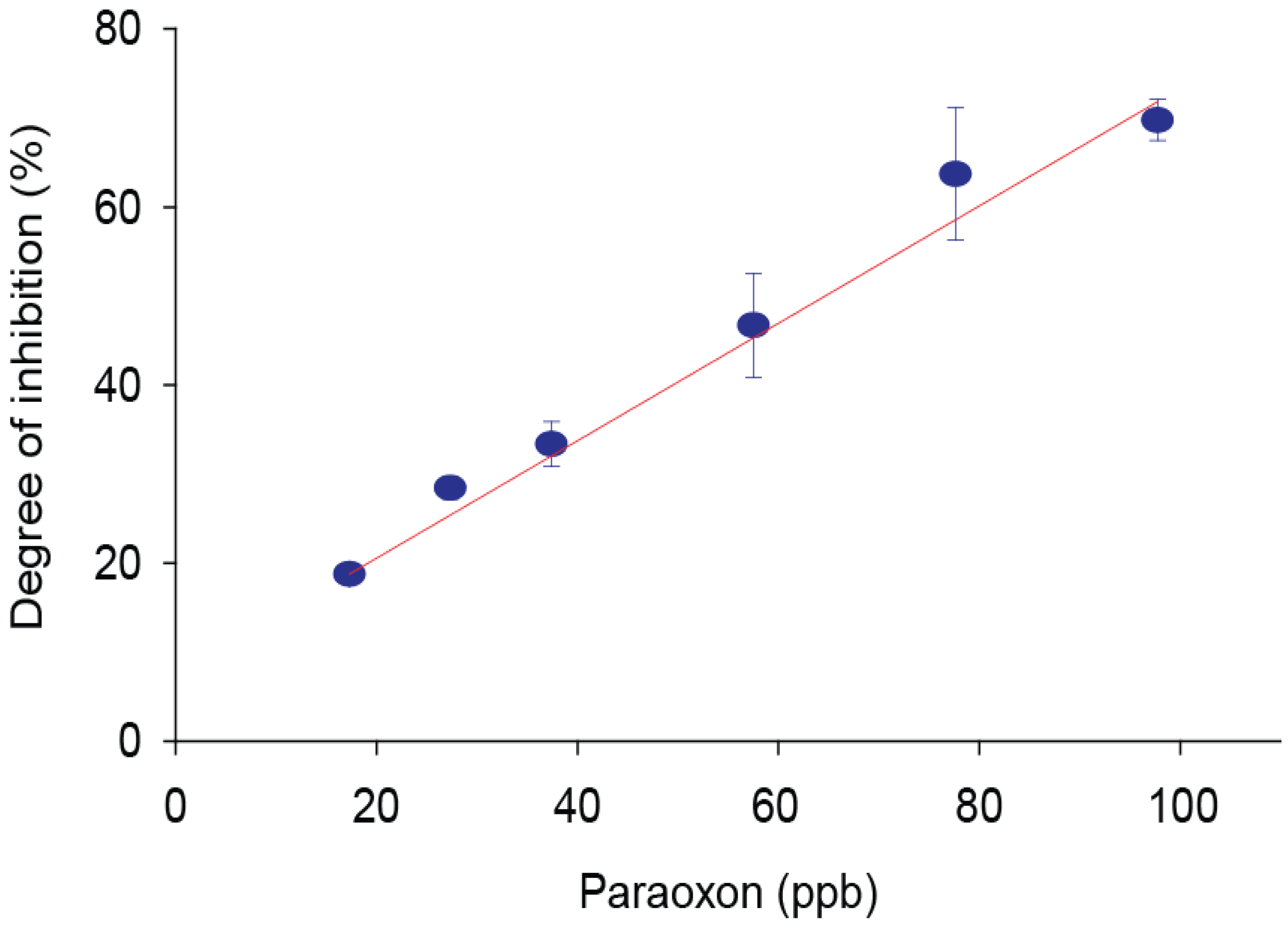
3.3. Inhibitory Effect of Paraoxon Extracted from Olive Oil Samples
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Agriculture and Rural Development. Olive Oil. Available online: http://ec.europa.eu/agriculture/olive-oil/index_en.htm (accessed on 12 December 2016).
- EC Regulation No 1183/2008. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:32008R1183 (accessed on 12 December 2016).
- Codex Alimentarius Commission 2105. Available online: http://www.fao.org/fao-who-codexalimentarius/standards/pestres/commodities-detail/en/?c_id=212 (accessed on 12 December 2016).
- EC Regulation No 396/2005. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2005:070:0001:0016:en:PDF (accessed on 12 December 2016).
- Commission Implementing Regulation (EU). No 400/2014 of 22 April 2014. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32014R0400&from=EN (accessed on 12 December 2016).
- EU Regulation No 2013/39/EC. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:32013L0039 (accessed on 12 December 2016).
- Anastassiades, M.; Lehotay, S.J.; Štajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [PubMed]
- Arduini, F.; Forchielli, M.; Amine, A.; Neagu, D.; Cacciotti, I.; Nanni, F.; Moscone, D.; Palleschi, G. Screen-printed biosensor modified with carbon black nanoparticles for the determination of paraoxon based on the inhibition of butyrylcholinesterase. Microchim. Acta 2015, 182, 643–651. [Google Scholar] [CrossRef]
- Arduini, F.; Neagu, D.; Scognamiglio, V.; Patarino, S.; Moscone, D.; Palleschi, G. Automatable flow system for paraoxon detection with an embedded screen-printed electrode tailored with butyrylcholinesterase and prussian blue nanoparticles. Chemosensors 2015, 3, 129–145. [Google Scholar] [CrossRef]
- Arduini, F.; Amine, A.; Majorani, C.; Di Giorgio, F.; De Felicis, D.; Cataldo, F.; Moscone, D.; Palleschi, G. High performance electrochemical sensor based on modified screen-printed electrodes with cost-effective dispersion of nanostructured carbon black. Electrochem. Commun. 2010, 12, 346–350. [Google Scholar] [CrossRef]
- Arduini, F.; Di Nardo, F.; Amine, A.; Micheli, L.; Palleschi, G.; Moscone, D. Carbon black-modified screen-printed electrodes as electroanalytical tools. Electroanalysis 2012, 24, 743–751. [Google Scholar] [CrossRef]
- Wong, C.H.A.; Ambrosi, A.; Pumera, M. Thermally reduced graphenes exhibiting a close relationship to amorphous carbon. Nanoscale 2012, 4, 4972–4977. [Google Scholar] [CrossRef] [PubMed]
- Lo, T.W.; Aldous, L.; Compton, R.G. The use of nano-carbon as an alternative to multi-walled carbon nanotubes in modified electrodes for adsorptive stripping voltammetry. Sens. Actuators B Chem. 2012, 162, 361–368. [Google Scholar] [CrossRef]
- Suprun, E.V.; Arduini, F.; Moscone, D.; Palleschi, G.; Shumyantseva, V.V.; Archakov, A.I. Direct electrochemistry of heme proteins on electrodes modified with Didodecyldimethyl Ammonium Bromide and carbon black. Electroanalysis 2012, 24, 1923–1931. [Google Scholar] [CrossRef]
- Talarico, D.; Arduini, F.; Constantino, A.; Del Carlo, M.; Compagnone, D.; Moscone, D.; Palleschi, G. Carbon black as successful screen-printed electrode modifier for phenolic compound detection. Electrochem. Commun. 2015, 60, 78–82. [Google Scholar] [CrossRef]
- Vicentini, F.C.; Raymundo-Pereira, P.A.; Janegitz, B.C.; Machado, S.A.; Fatibello-Filho, O. Nanostructured carbon black for simultaneous sensing in biological fluids. Sens. Actuators B Chem. 2016, 227, 610–618. [Google Scholar] [CrossRef]
- Oujji, N.B.; Bakas, I.; Istamboulié, G.; Ait-Ichou, I.; Ait-Addi, E.; Rouillon, R.; Noguer, T. Sol-gel immobilization of acetylcholinesterase for the determination of organophosphate pesticides in olive oil with biosensors. Food Control 2013, 30, 657–661. [Google Scholar] [CrossRef]
- Oujji, N.B.; Bakas, I.; Istamboulié, G.; Ait-Ichou, I.; Ait-Addi, E.; Rouillon, R.; Noguer, T. A Simple Colorimetric Enzymatic-Assay, based on immobilization of acetylcholinesterase by adsorption, for sensitive detection of organophosphorus insecticides in olive oil. Food Control 2014, 46, 75–80. [Google Scholar] [CrossRef]
- Arduini, F.; Palleschi, G. Disposable electrochemical biosensor based on cholinesterase inhibition with improved shelf-life and working stability for nerve agent detection. In Portable Chemical Sensors; Springer: Berlin, Germany, 2012; pp. 261–278. [Google Scholar]
- Amine, A.; Arduini, F.; Moscone, D.; Palleschi, G. Recent advances in biosensors based on enzyme inhibition. Biosens. Bioelectron. 2016, 76, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Lin, Y. Biosensor based on self-assembling acetylcholinesterase on carbon nanotubes for flow injection/amperometric detection of organophosphate pesticides and nerve agents. Anal. Chem. 2006, 78, 835–843. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Bai, Y.; Han, G.; Li, M. Porous-reduced graphene oxide for fabricating an amperometric acetylcholinesterase biosensor. Sens. Actuators B Chem. 2013, 185, 706–712. [Google Scholar] [CrossRef]
- Wang, K.; Liu, Q.; Dai, L.; Yan, J.; Ju, C.; Qiu, B.; Wu, X. A highly sensitive and rapid organophosphate biosensor based on enhancement of CdS-decorated graphene nanocomposite. Anal. Chim. Acta 2011, 695, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Zamfir, L.G.; Rotariu, L.; Bala, C. A novel, sensitive, reusable andlow potential acetylcholinesterase biosensor for chlorpyrifos based on 1-butyl-3-methylimidazolium tetrafluoroborate/multiwalled carbon nanotubes gel. Biosens. Bioelectron. 2011, 26, 3692–3695. [Google Scholar] [CrossRef] [PubMed]
- Ovalle, M.; Stoytcheva, M.; Zlatev, R.; Valdez, B.; Velkova, Z. Electrochemical study on the type of immobilized acetylcholinesterase inhibition by sodium fluoride. Electrochim. Acta 2008, 53, 6344–6350. [Google Scholar] [CrossRef]
- Evtugyn, G.A.; Ivanov, A.N.; Gogol, E.V.; Marty, J.L.; Budnikov, H.C. Amperometric flow-through biosensor for the determination of cholinesterase inhibitors. Anal. Chim. Acta 1999, 385, 13–21. [Google Scholar] [CrossRef]
- Guilhermino, L.; Barros, P.; Silva, M.C.; Soares, A.M. Short Communication. Should the use of inhibition of cholinesterases as a specific biomarker for organophosphate and carbamate pesticides be questioned. Biomarkers 1998, 3, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Kukla, A.L.; Kanjuk, N.I.; Starodub, N.F.; Shirshov, Y.M. Multienzyme electrochemical sensor array for determination of heavy metal ions. Sens. Actuators B Chem. 1999, 57, 213–218. [Google Scholar] [CrossRef]
- Frasco, M.F.; Fournier, D.; Carvalho, F.; Guilhermino, L. Do metals inhibit acetylcholinesterase (AChE)? Implementation of assay conditions for the use of AChE activity as a biomarker of metal toxicity. Biomarkers 2005, 10, 360–375. [Google Scholar] [CrossRef] [PubMed]
- QuEChERS Informational Booklet. Available online: https://www.chromspec.com/pdf/e/uct19.pdf (accessed on 12 December 2016).
- Document No. SANCO/10684/2009, Method Validation and Quality Control Procedures for Pesticides Residues Analysis in Food and Feed. Available online: http://www.crl-pesticides.eu/library/docs/fv/SANCO12495-2011.pdf (accessed on 12 December 2016).

| Sample | Paraoxon Added (ppb) | Paraoxon Detected (ppb) | Recovery % Biosensor | Recovery % HPLC |
|---|---|---|---|---|
| extra virgin olive oil | 100 | 60 ± 2 | 60 ± 2 | 63 ± 2 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arduini, F.; Forchielli, M.; Scognamiglio, V.; Nikolaevna, K.A.; Moscone, D. Organophosphorous Pesticide Detection in Olive Oil by Using a Miniaturized, Easy-to-Use, and Cost-Effective Biosensor Combined with QuEChERS for Sample Clean-Up. Sensors 2017, 17, 34. https://doi.org/10.3390/s17010034
Arduini F, Forchielli M, Scognamiglio V, Nikolaevna KA, Moscone D. Organophosphorous Pesticide Detection in Olive Oil by Using a Miniaturized, Easy-to-Use, and Cost-Effective Biosensor Combined with QuEChERS for Sample Clean-Up. Sensors. 2017; 17(1):34. https://doi.org/10.3390/s17010034
Chicago/Turabian StyleArduini, Fabiana, Matteo Forchielli, Viviana Scognamiglio, Kozitsina Alisa Nikolaevna, and Danila Moscone. 2017. "Organophosphorous Pesticide Detection in Olive Oil by Using a Miniaturized, Easy-to-Use, and Cost-Effective Biosensor Combined with QuEChERS for Sample Clean-Up" Sensors 17, no. 1: 34. https://doi.org/10.3390/s17010034
APA StyleArduini, F., Forchielli, M., Scognamiglio, V., Nikolaevna, K. A., & Moscone, D. (2017). Organophosphorous Pesticide Detection in Olive Oil by Using a Miniaturized, Easy-to-Use, and Cost-Effective Biosensor Combined with QuEChERS for Sample Clean-Up. Sensors, 17(1), 34. https://doi.org/10.3390/s17010034







