Nanostructured Tip-Shaped Biosensors: Application of Six Sigma Approach for Enhanced Manufacturing
Abstract
:1. Introduction
2. Nanostructured Tip Bio-Sensors
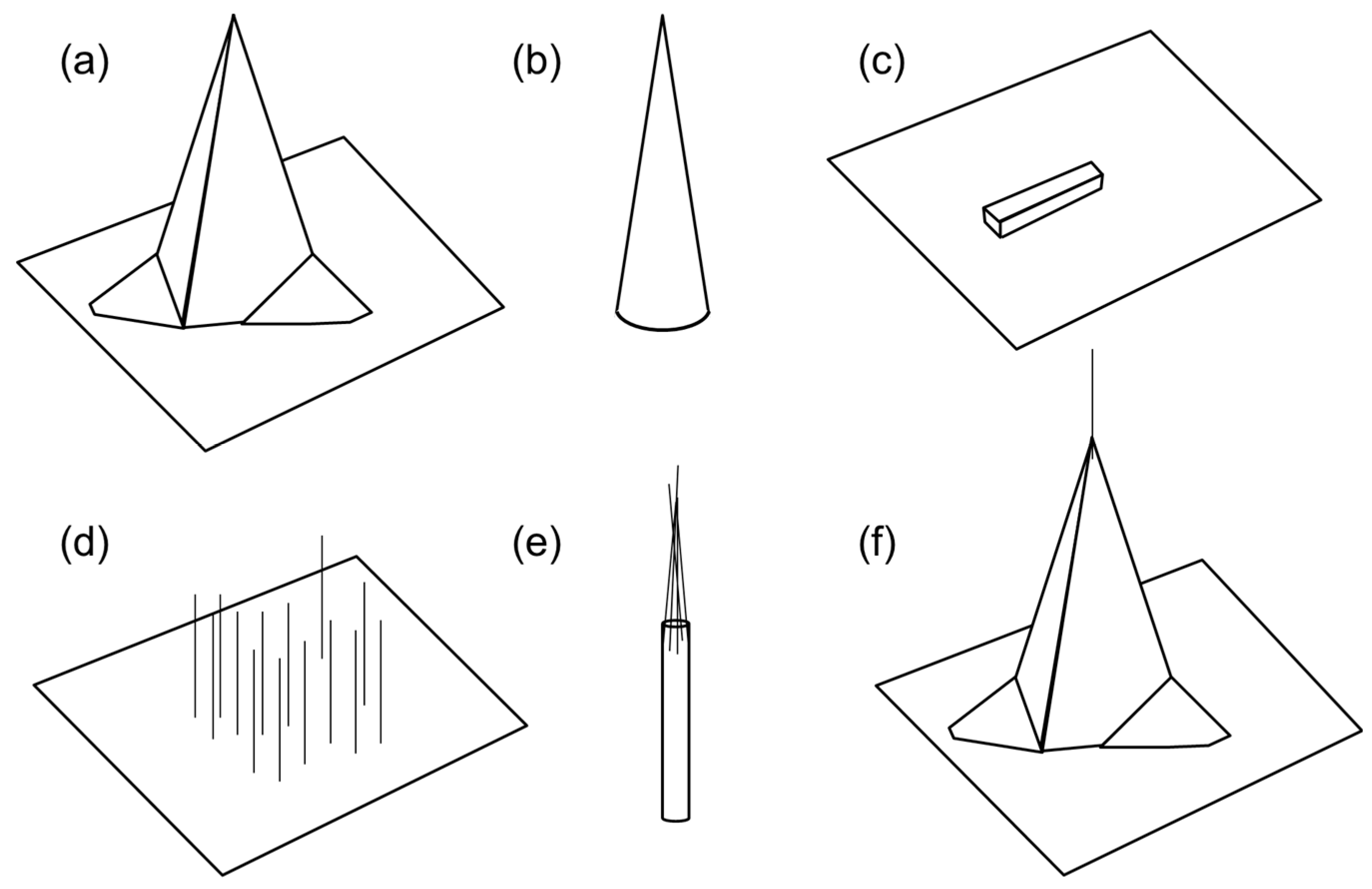
2.1. Fabrication and Nanotip Shapes
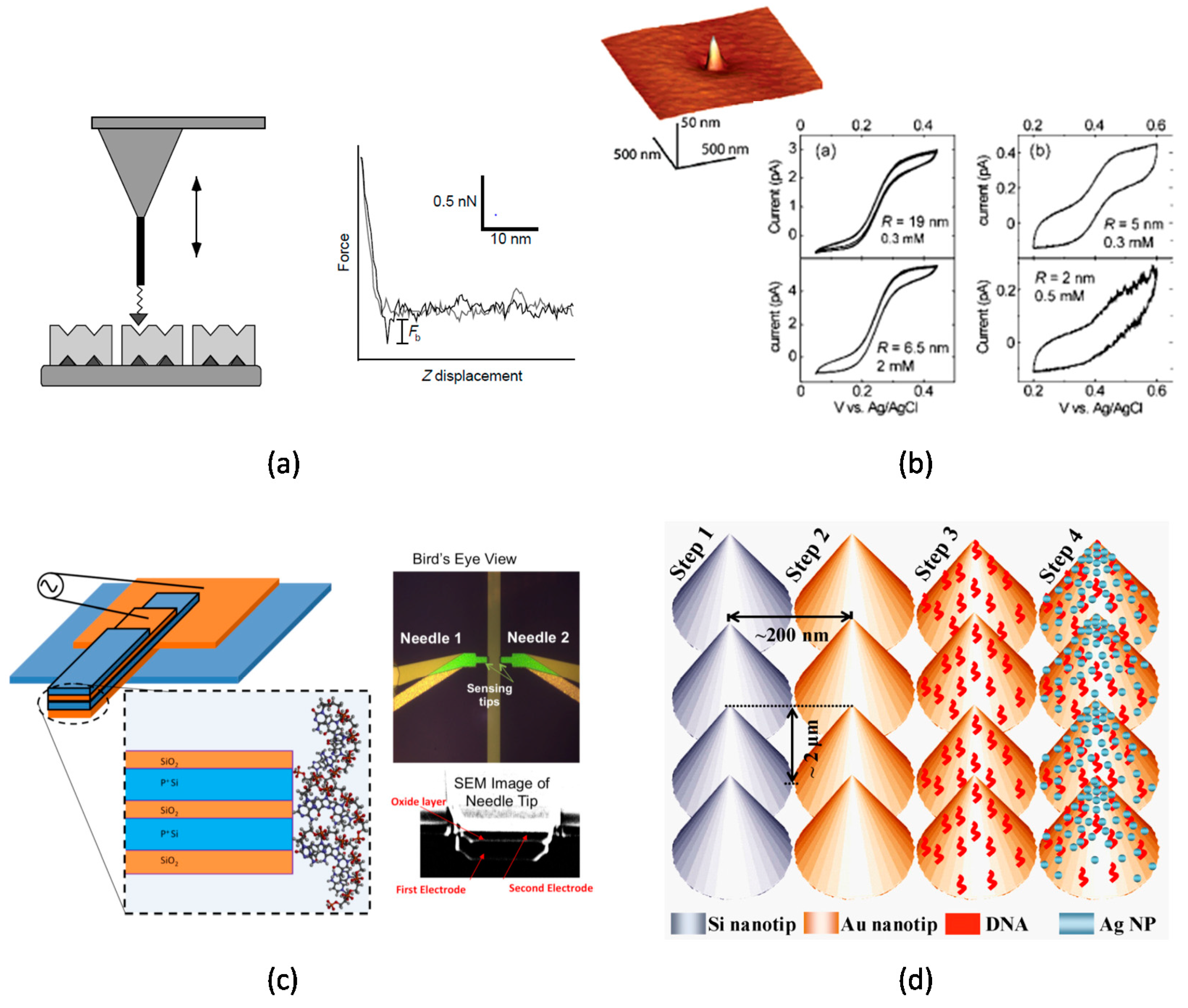
2.2. Nanotip-Based Biosensors and Potential Challenges
2.3. Six Sigma Approach and Its Application to Nanomanufacturing
3. Materials and Methods
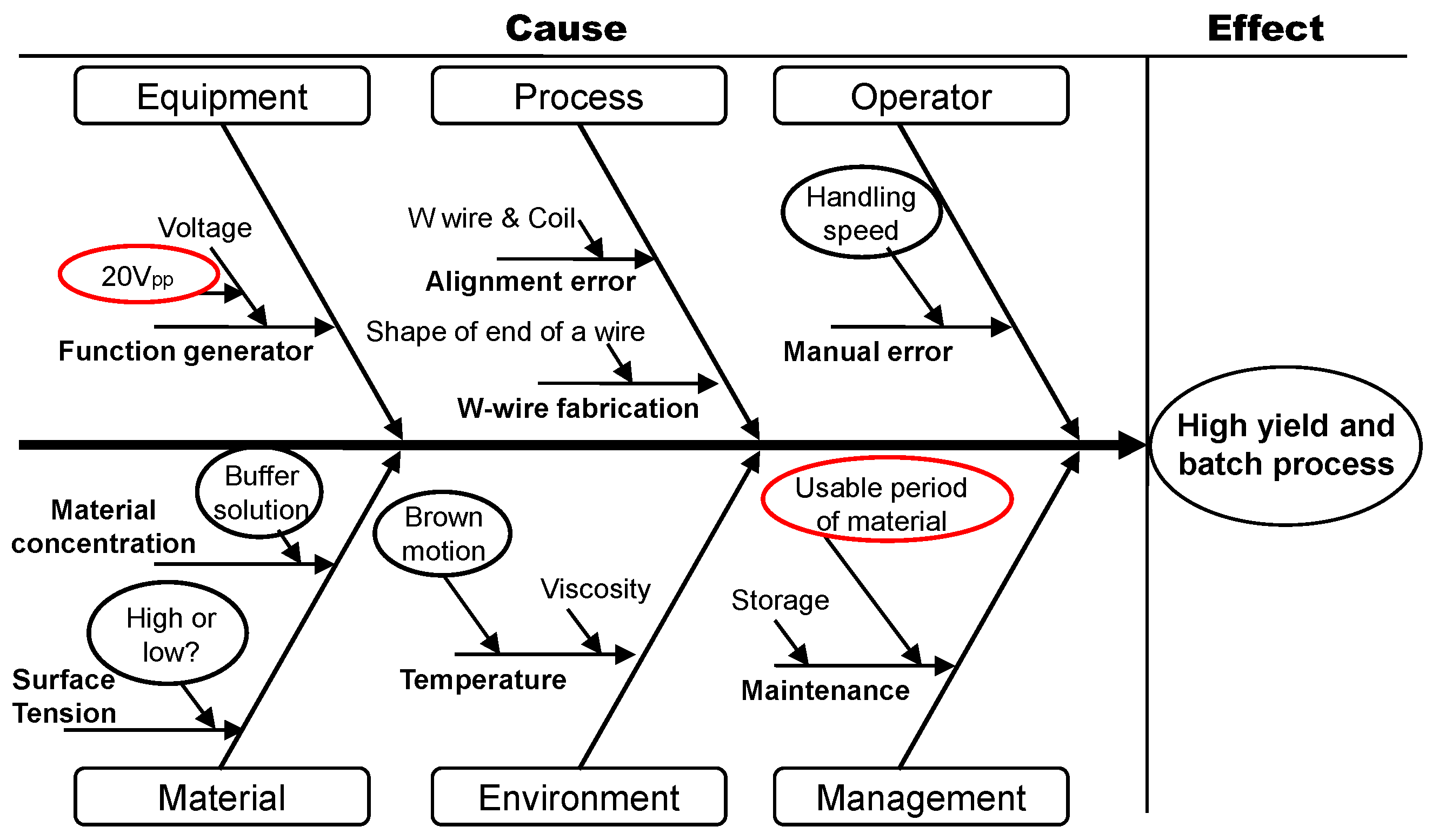
3.1. Application of the Six Sigma Approach to Nanotip Fabrication
- Define phase: The goal is to improve the yield of a dendritic nanotip.
- Measure phase: The production yield of a nanotip is 20%. The shape of a nanotip is not uniform.
- Analyze phase: The electric field for tip fabrication can be low, and the medium for carbon nanotubes can be degraded, which can lower the production yield of a nanotip.
- Improve phase: A higher electric field is applied, and the medium for carbon nanotube suspension is refreshed weekly.
- Control phase: When the factors of an electric field and carbon nanotube medium are controlled, the yield is improved from 20% to 80%. The uniform shape is improved.
3.2. Materials
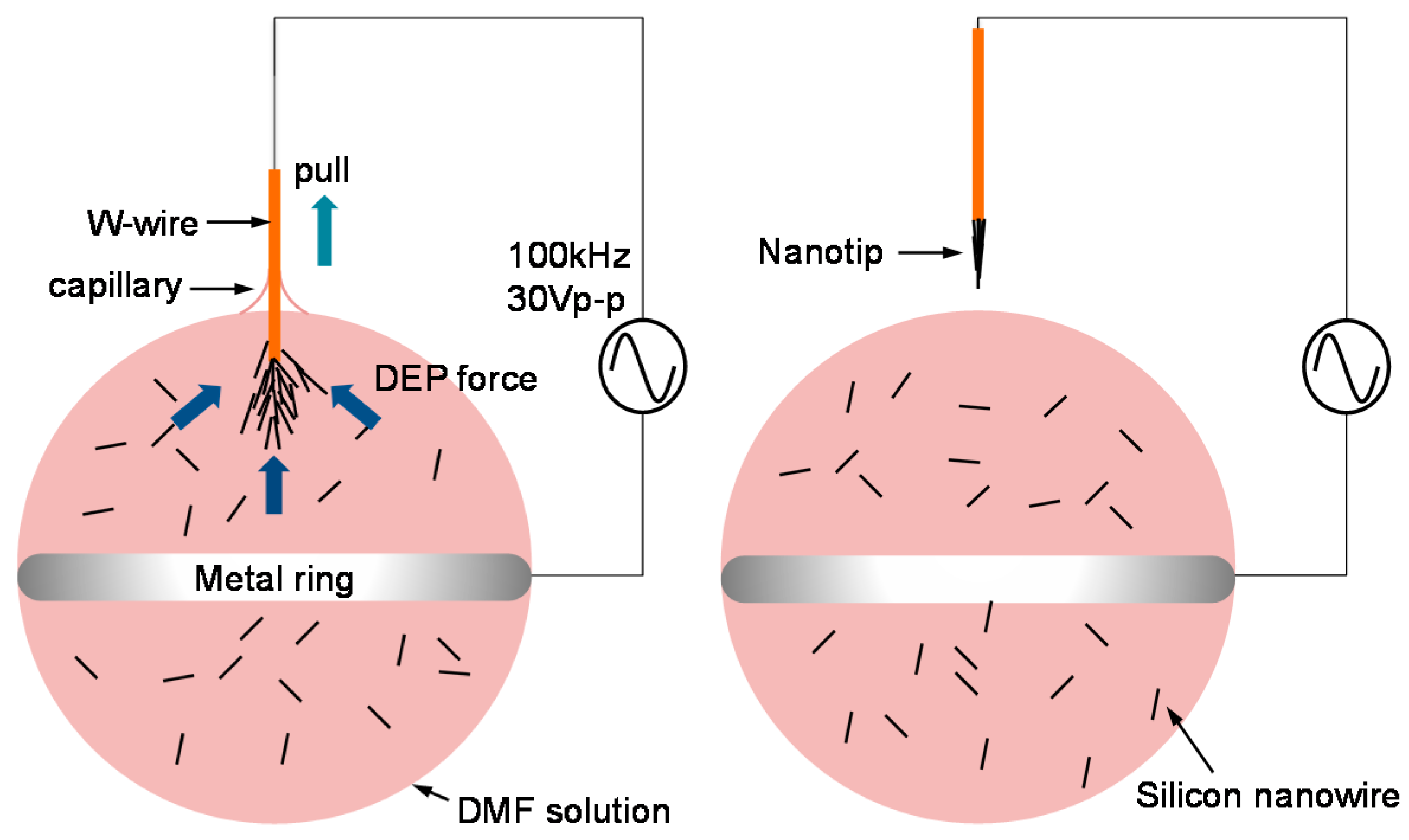
3.3. Nanotip Fabrication
3.4. Nanotip Funtionalization
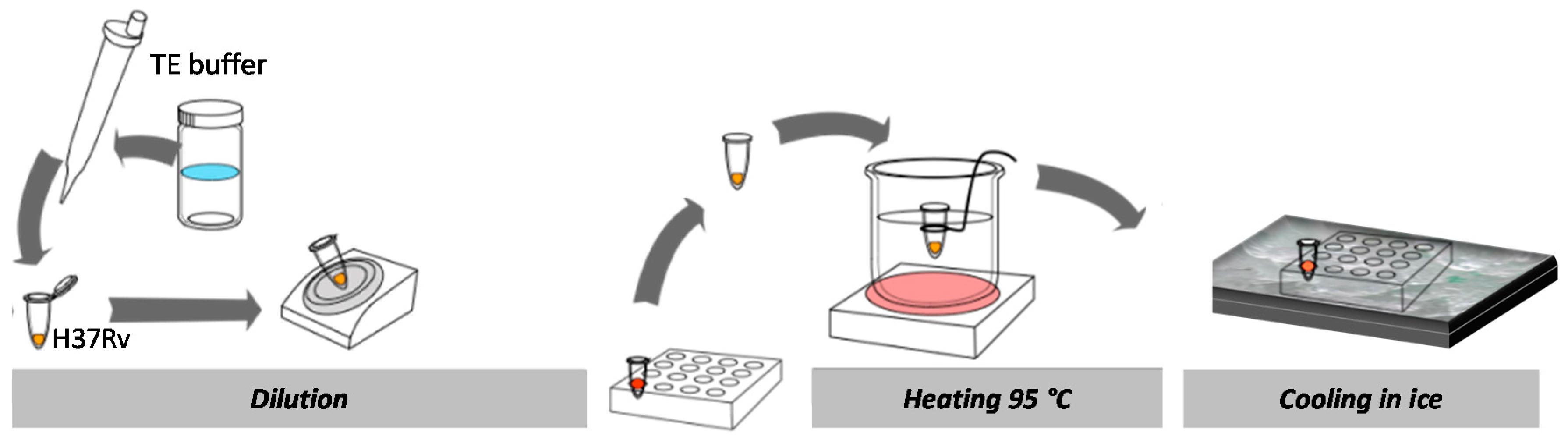
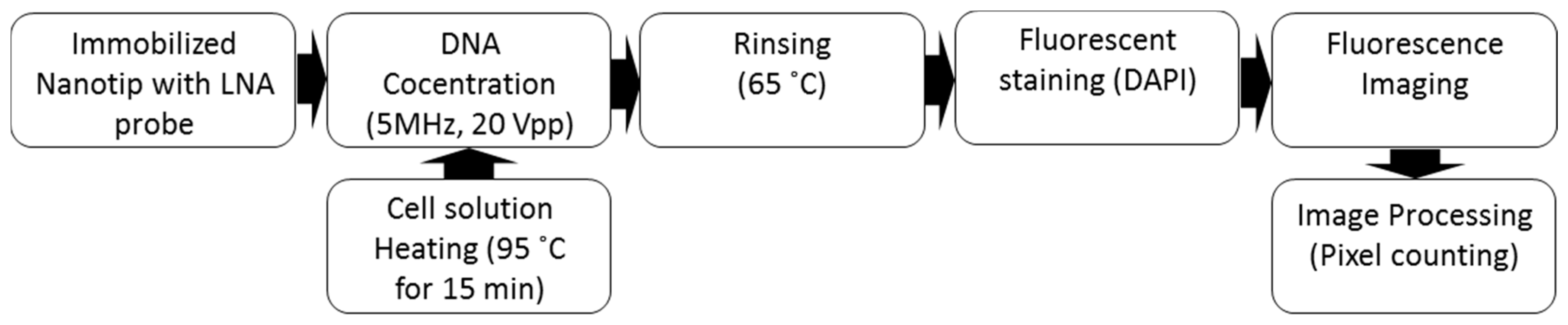
3.5. Sequence Specific DNA Detection
4. Results
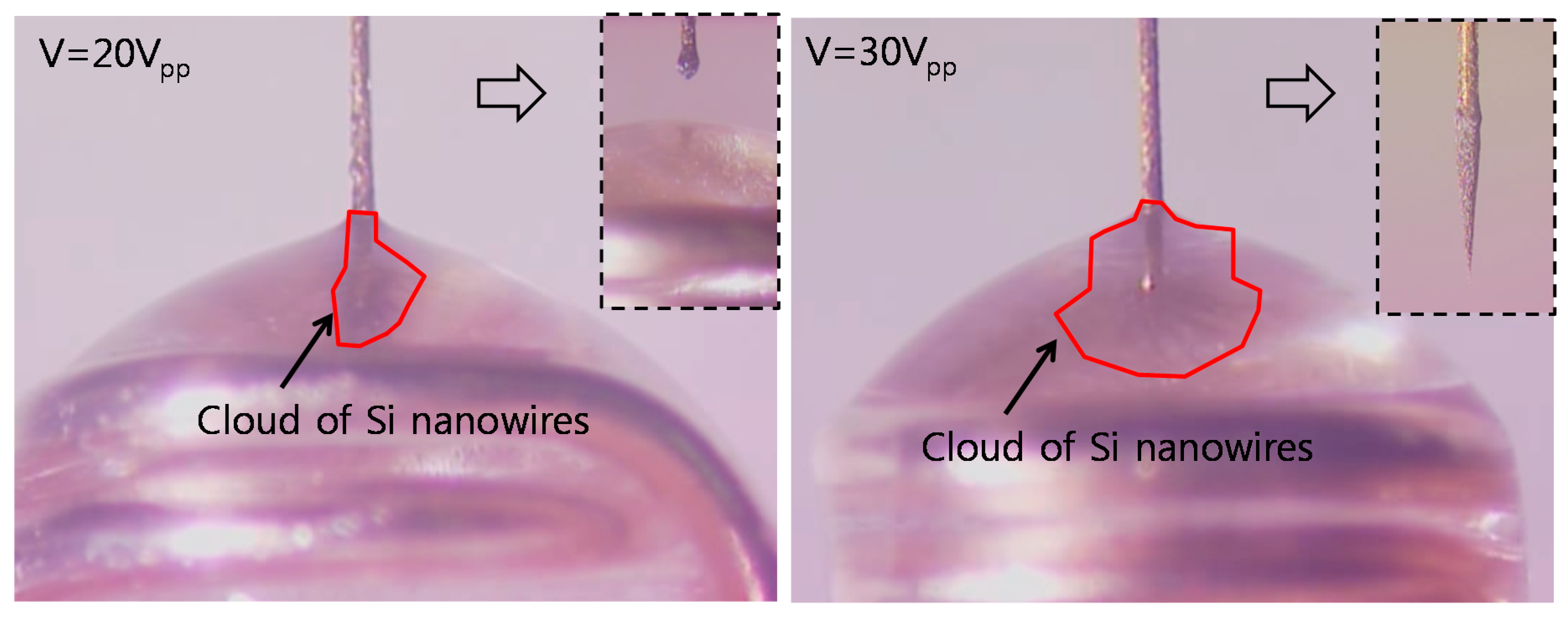
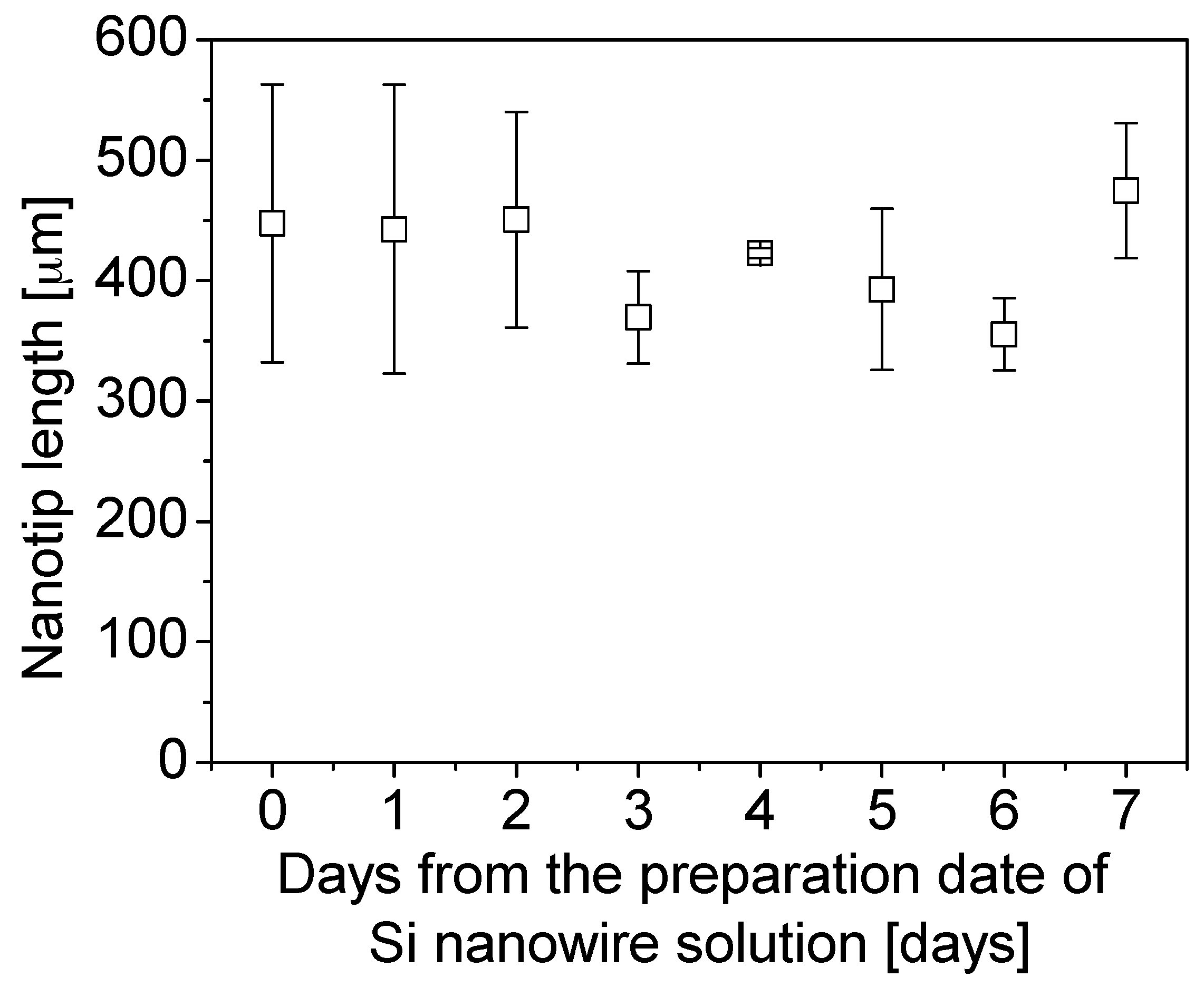
4.1. Dendritic Nanotip Fabrication with Six Sigma Approach
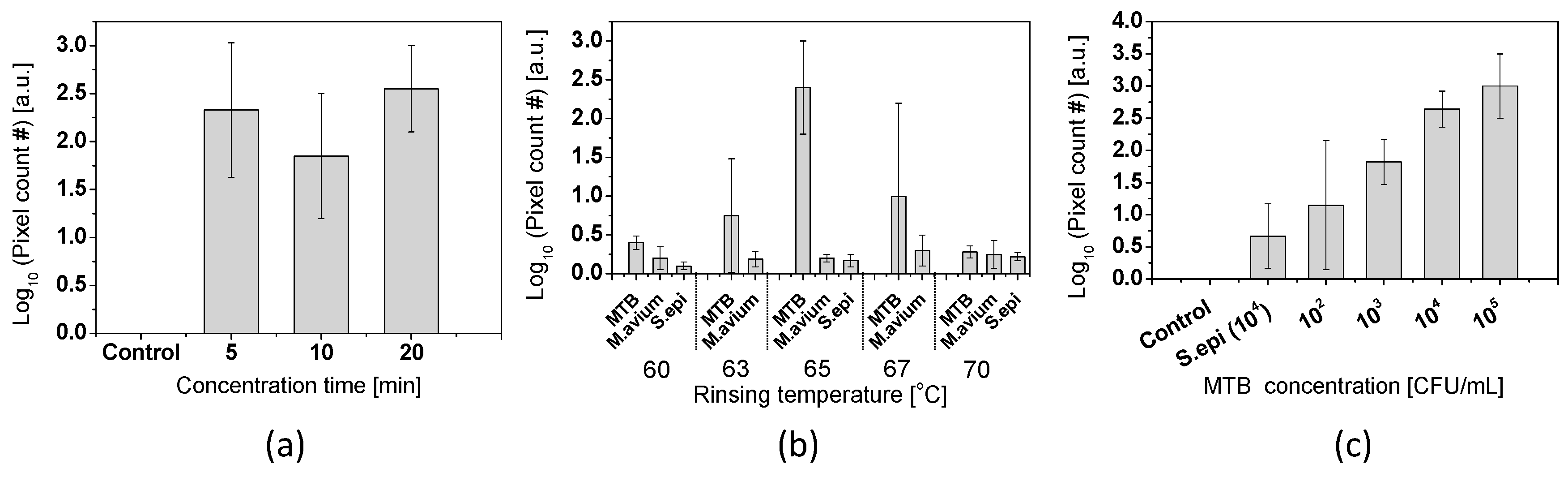
4.2. Sequence Specific DNA Ddetection
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ramanavicius, A.; Ramanaviciene, A.; Malinauskas, A. Electrochemical sensors based on conducting polymer-polypyrrole. Electrochim. Acta 2006, 51, 6025–6037. [Google Scholar] [CrossRef]
- Ferrier, D.C.; Shaver, M.P.; Hands, P.J.W. Micro- and nano-structure based oligonucleotide sensors. Biosens. Bioelectron. 2015, 68, 798–810. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Xu, H.X. Hot spots in different metal nanostructures for plasmon-enhanced raman spectroscopy. Nanoscale 2013, 5, 10794–10805. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.I.; Li, B.R.; Chen, Y.T. Silicon nanowire field-effect transistor-based biosensors for biomedical diagnosis and cellular recording investigation. Nano Today 2011, 6, 131–154. [Google Scholar] [CrossRef]
- Walcarius, A.; Minteer, S.D.; Wang, J.; Lin, Y.H.; Merkoci, A. Nanomaterials for bio-functionalized electrodes: Recent trends. J. Mater. Chem. B 2013, 1, 4878–4908. [Google Scholar] [CrossRef]
- Domke, K.F.; Zhang, D.; Pettinger, B. Tip-enhanced raman spectra of picomole quantities of DNA nucleobases at Au(111). J. Am. Chem. Soc. 2007, 129, 6708–6709. [Google Scholar] [CrossRef] [PubMed]
- Dimaki, M.; Vazquez, P.; Olsen, M.H.; Sasso, L.; Rodriguez-Trujillo, R.; Vedarethinam, I.; Svendsen, W.E. Fabrication and characterization of 3D micro- and nanoelectrodes for neuron recordings. Sensors 2010, 10, 10339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, R.; Ramakrishna, S.N.; Naik, V.V.; Chu, Z.; Drew, M.E.; Spencer, N.D.; Yamakoshi, Y. Versatile method for afm-tip functionalization with biomolecules: Fishing a ligand by means of an in situ click reaction. Nanoscale 2015, 7, 6599–6606. [Google Scholar] [CrossRef] [PubMed]
- Borzenets, I.V.; Yoon, I.; Prior, M.M.; Donald, B.R.; Mooney, R.D.; Finkelstein, G. Ultra-sharp metal and nanotube-based probes for applications in scanning microscopy and neural recording. J. Appl. Phys. 2012, 111, 074703. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Lian, X. Rapid fabrication of high-aspect-ratio platinum microprobes by electrochemical discharge etching. Materials 2016, 9, 233. [Google Scholar] [CrossRef]
- Tang, J.; Gao, B.; Geng, H.Z.; Velev, O.D.; Qin, L.C.; Zhou, O. Assembly of id nanostructures into sub-micrometer diameter fibrils with controlled and variable length by dielectrophoresis. Adv. Mater. 2003, 15, 1352–1355. [Google Scholar] [CrossRef]
- Xia, Y.; Rogers, J.A.; Paul, K.E.; Whitesides, G.M. Unconventional methods for fabricating and patterning nanostructures. Chem. Rev. 1999, 99, 1823–1848. [Google Scholar] [CrossRef] [PubMed]
- Jeon, H.-J.; Kim, K.H.; Baek, Y.-K.; Kim, D.W.; Jung, H.-T. New top-down approach for fabricating high-aspect-ratio complex nanostructures with 10 nm scale features. Nano Lett. 2010, 10, 3604–3610. [Google Scholar] [CrossRef] [PubMed]
- Lo, H.C.; Hsiung, H.I.; Chattopadhyay, S.; Han, H.C.; Chen, C.F.; Leu, J.P.; Chen, K.H.; Chen, L.C. Label free sub-picomole level DNA detection with ag nanoparticle decorated au nanotip arrays as surface enhanced raman spectroscopy platform. Biosens. Bioelectron. 2011, 26, 2413–2418. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.-H.; Lee, G.-J.; Kim, W.-S.; Lim, H.-E.; Mativenga, M.; Park, K.-C.; Park, H.-K. All-carbon electrode consisting of carbon nanotubes on graphite foil for flexible electrochemical applications. Materials 2014, 7, 1975. [Google Scholar] [CrossRef]
- Ghane Motlagh, B.; Choueib, M.; Hajhosseini Mesgar, A.; Hasanuzzaman, M.; Sawan, M. Direct growth of carbon nanotubes on new high-density 3D pyramid-shaped microelectrode arrays for brain-machine interfaces. Micromachines 2016, 7, 163. [Google Scholar] [CrossRef]
- Yoon, Y.; Lee, G.; Yoo, K.; Lee, J.-B. Fabrication of a microneedle/CNT hierarchical micro/nano surface electrochemical sensor and its in-vitro glucose sensing characterization. Sensors 2013, 13, 16672. [Google Scholar] [CrossRef] [PubMed]
- An, T.; Choi, W.; Lee, E.; Kim, I.-T.; Moon, W.; Lim, G. Fabrication of functional micro- and nanoneedle electrodes using a carbon nanotube template and electrodeposition. Nanoscale Res. Lett. 2011, 6, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Hafner, J.H.; Cheung, C.-L.; Oosterkamp, T.H.; Lieber, C.M. High-yield assembly of individual single-walled carbon nanotube tips for scanning probe microscopies. J. Phys. Chem. B 2001, 105, 743–746. [Google Scholar] [CrossRef]
- Hafner, J.H.; Cheung, C.L.; Woolley, A.T.; Lieber, C.M. Structural and functional imaging with carbon nanotube afm probes. Prog. Biophys. Mol. Biol. 2001, 77, 73–110. [Google Scholar] [CrossRef]
- Cheung, C.L.; Hafner, J.H.; Lieber, C.M. Carbon nanotube atomic force microscopy tips: Direct growth by chemical vapor deposition and application to high-resolution imaging. Proc. Natl. Acad. Sci. USA 2000, 97, 3809–3813. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Florin, E.L.; Rief, M.; Lehmann, H.; Ludwig, M.; Dornmair, C.; Moy, V.T.; Gaub, H.E. Sensing specific molecular-interactions with the atomic-force microscope. Biosens. Bioelectron. 1995, 10, 895–901. [Google Scholar] [CrossRef]
- Kueng, A.; Kranz, C.; Lugstein, A.; Bertagnolli, E.; Mizaikoff, B. Afm-tip-integrated amperometric microbiosensors: High-resolution imaging of membrane transport. Angew. Chem. Int. Ed. 2005, 44, 3419–3422. [Google Scholar] [CrossRef] [PubMed]
- Yum, K.; Wang, N.; Yu, M.F. Nanoneedle: A multifunctional tool for biological studies in living cells. Nanoscale 2010, 2, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Krapf, D.; Wu, M.Y.; Smeets, R.M.M.; Zandbergen, H.W.; Dekker, C.; Lemay, S.G. Fabrication and characterization of nanopore-based electrodes with radii down to 2 nm. Nano Lett. 2006, 6, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Esfandyarpour, R.; Javanmard, M.; Koochak, Z.; Esfandyarpour, H.; Harris, J.S.; Davis, R.W. Label-free electronic probing of nucleic acids and proteins at the nanoscale using the nanoneedle biosensor. Biomicrofluidics 2013, 7, 044114. [Google Scholar] [CrossRef] [PubMed]
- Esfandyarpour, R.; Yang, L.; Koochak, Z.; Harris, J.S.; Davis, R.W. Nanoelectronic three-dimensional (3D) nanotip sensing array for real-time, sensitive, label-free sequence specific detection of nucleic acids. Biomed. Microdevices 2016, 18, 7. [Google Scholar] [CrossRef] [PubMed]
- Giannetti, A.; Barucci, A.; Cosi, F.; Pelli, S.; Tombelli, S.; Trono, C.; Baldini, F. Optical fiber nanotips coated with molecular beacons for DNA detection. Sensors 2015, 15, 9666–9680. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Gan, Z.H.; Zhuang, Q.K. Electrochemical sensors based on carbon nanotubes. Electroanalysis 2002, 14, 1609–1613. [Google Scholar] [CrossRef]
- Campbell, J.K.; Sun, L.; Crooks, R.M. Electrochemistry using single carbon nanotubes. J. Am. Chem. Soc. 1999, 121, 3779–3780. [Google Scholar] [CrossRef]
- Wong, S.S.; Joselevich, E.; Woolley, A.T.; Cheung, C.L.; Lieber, C.M. Covalently functionalized nanotubes as nanometre-sized probes in chemistry and biology. Nature 1998, 394, 52–55. [Google Scholar] [PubMed]
- Boo, H.; Jeong, R.A.; Park, S.; Kim, K.S.; An, K.H.; Lee, Y.H.; Han, J.H.; Kim, H.C.; Chung, T.D. Electrochemical nanoneedle biosensor based on multiwall carbon nanotube. Anal. Chem. 2006, 78, 617–620. [Google Scholar] [CrossRef] [PubMed]
- Jouzi, M.; Kerby, M.B.; Tripathi, A.; Xu, J. Nanoneedle method for high-sensitivity low-background monitoring of protein activity. Langmuir 2008, 24, 10786–10790. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Ibupoto, Z.H.; Abbasi, M.A.; Liu, X.J.; Nur, O.; Willander, M. Synthesis of three dimensional nickel cobalt oxide nanoneedles on nickel foam, their characterization and glucose sensing application. Sensors 2014, 14, 5415–5425. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Lu, F.; Tu, Y.; Ren, Z.F. Glucose biosensors based on carbon nanotube nanoelectrode ensembles. Nano Lett. 2004, 4, 191–195. [Google Scholar] [CrossRef]
- Vitol, E.A.; Orynbayeva, Z.; Friedman, G.; Gogotsi, Y. Nanoprobes for intracellular and single cell surface-enhanced raman spectroscopy (SERS). J. Raman Spectrosc. 2012, 43, 817–827. [Google Scholar] [CrossRef]
- Yeo, W.H.; Lee, H.B.; Kim, J.H.; Lee, K.H.; Chung, J.H. Nanotip analysis for dielectrophoretic concentration of nanosized viral particles. Nanotechnology 2013, 24, 185502. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.-H.; Kopacz, A.M.; Kim, J.-H.; Chen, X.; Wu, J.; Gao, D.; Lee, K.-H.; Liu, W.-K.; Chung, J.-H. Dielectrophoretic concentration of low-abundance nanoparticles using a nanostructured tip. Nanotechnology 2012, 23, 485707. [Google Scholar] [CrossRef] [PubMed]
- Kopacz, A.M.; Yeo, W.-H.; Chung, J.-H.; Liu, W.K. Nanoscale sensor analysis using the immersed molecular electrokinetic finite element method. Nanoscale 2012, 4, 5189–5194. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.H.; Chung, J.H.; Liu, Y.L.; Lee, K.H. Size-specific concentration of DNA to a nanostructured tip using dielectrophoresis and capillary action. J. Phys. Chem. B 2009, 113, 10849–10858. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.-H.; Chou, F.-L.; Fotouhi, G.; Oh, K.; Stevens, B.T.; Tseng, H.-Y.; Gao, D.; Shen, A.Q.; Chung, J.-H.; Lee, K.-H. Size-selective immunofluorescence of mycobacterium tuberculosis cells by capillary- and viscous forces. Lab Chip 2010, 10, 3178–3181. [Google Scholar] [CrossRef] [PubMed]
- Kalyanasundaram, D.; Kim, J.-H.; Fotouhi, G.; Lee, H.-B.; Hiraiwa, M.; Oh, K.; Lee, K.-H.; Chung, J.-H. Nanotips for single-step preparation of DNA for qpcr analysis. Analyst 2013, 138, 3135–3138. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Hiraiwa, M.; Lee, H.-B.; Lee, K.-H.; Cangelosi, G.A.; Chung, J.-H. Electrolyte-free amperometric immunosensor using a dendritic nanotip. RSC Adv. 2013, 3, 4281–4287. [Google Scholar] [CrossRef] [PubMed]
- Shankar, R. Process Improvement Using Six Sigma a Dmaic Guide; ASQ Quality Press: Milwaukee, WI, USA, 2009. [Google Scholar]
- Genis, V.; Mauk, M.; Gogotsi, Y.; Sakalley, D.; Hagarman, J.; Burnside, H. Lean six sigma nanomanufacturing course for undergraduate engineering technology and engineering programs. J. Eng. Technol. 2010, 27, 18–29. [Google Scholar]
- Kim, J.-H.; Yeo, W.-H.; Shu, Z.; Soelberg, S.D.; Inoue, S.; Kalyanasundaram, D.; Ludwig, J.; Furlong, C.E.; Riley, J.J.; Weigel, K.M.; et al. Immunosensor towards low-cost, rapid diagnosis of tuberculosis. Lab Chip 2012, 12, 1437–1440. [Google Scholar] [CrossRef] [PubMed]
- Braasch, D.A.; Corey, D.R. Locked nucleic acid (LNA): Fine-tuning the recognition of DNA and RNA. Chem. Biol. 2001, 8, 1–7. [Google Scholar] [CrossRef]
- Vester, B.; Wengel, J. LNA (locked nucleic acid): High-affinity targeting of complementary RNA and DNA. Biochemistry 2004, 43, 13233–13241. [Google Scholar] [CrossRef] [PubMed]




© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahng, S.-J.; Kim, J.-H.; Chung, J.-H. Nanostructured Tip-Shaped Biosensors: Application of Six Sigma Approach for Enhanced Manufacturing. Sensors 2017, 17, 17. https://doi.org/10.3390/s17010017
Kahng S-J, Kim J-H, Chung J-H. Nanostructured Tip-Shaped Biosensors: Application of Six Sigma Approach for Enhanced Manufacturing. Sensors. 2017; 17(1):17. https://doi.org/10.3390/s17010017
Chicago/Turabian StyleKahng, Seong-Joong, Jong-Hoon Kim, and Jae-Hyun Chung. 2017. "Nanostructured Tip-Shaped Biosensors: Application of Six Sigma Approach for Enhanced Manufacturing" Sensors 17, no. 1: 17. https://doi.org/10.3390/s17010017





