Wearable Sensors for Remote Health Monitoring
Abstract
:1. Introduction
2. Wearable Health Monitoring Systems
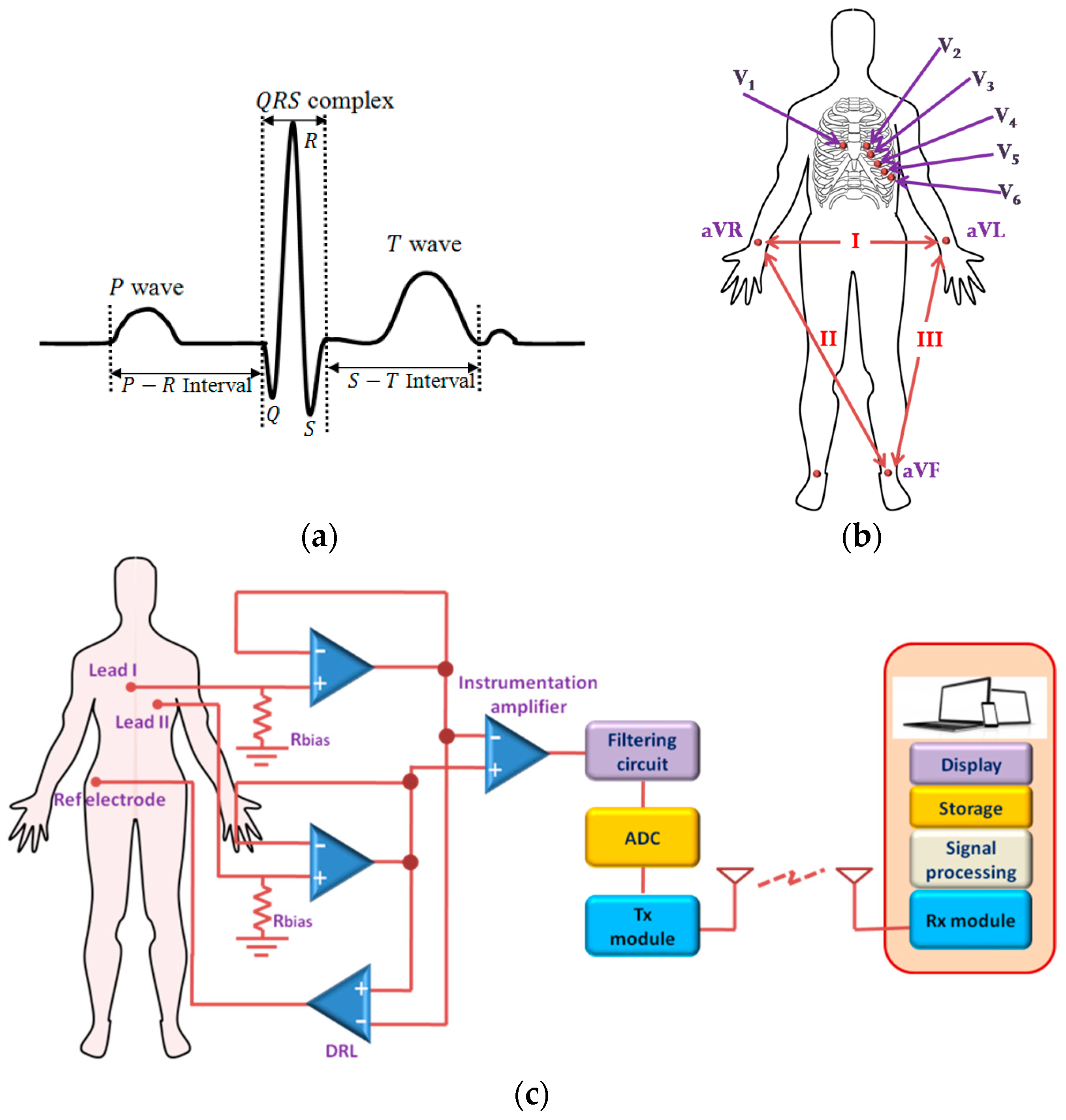
2.1. Cardiovascular Monitoring System
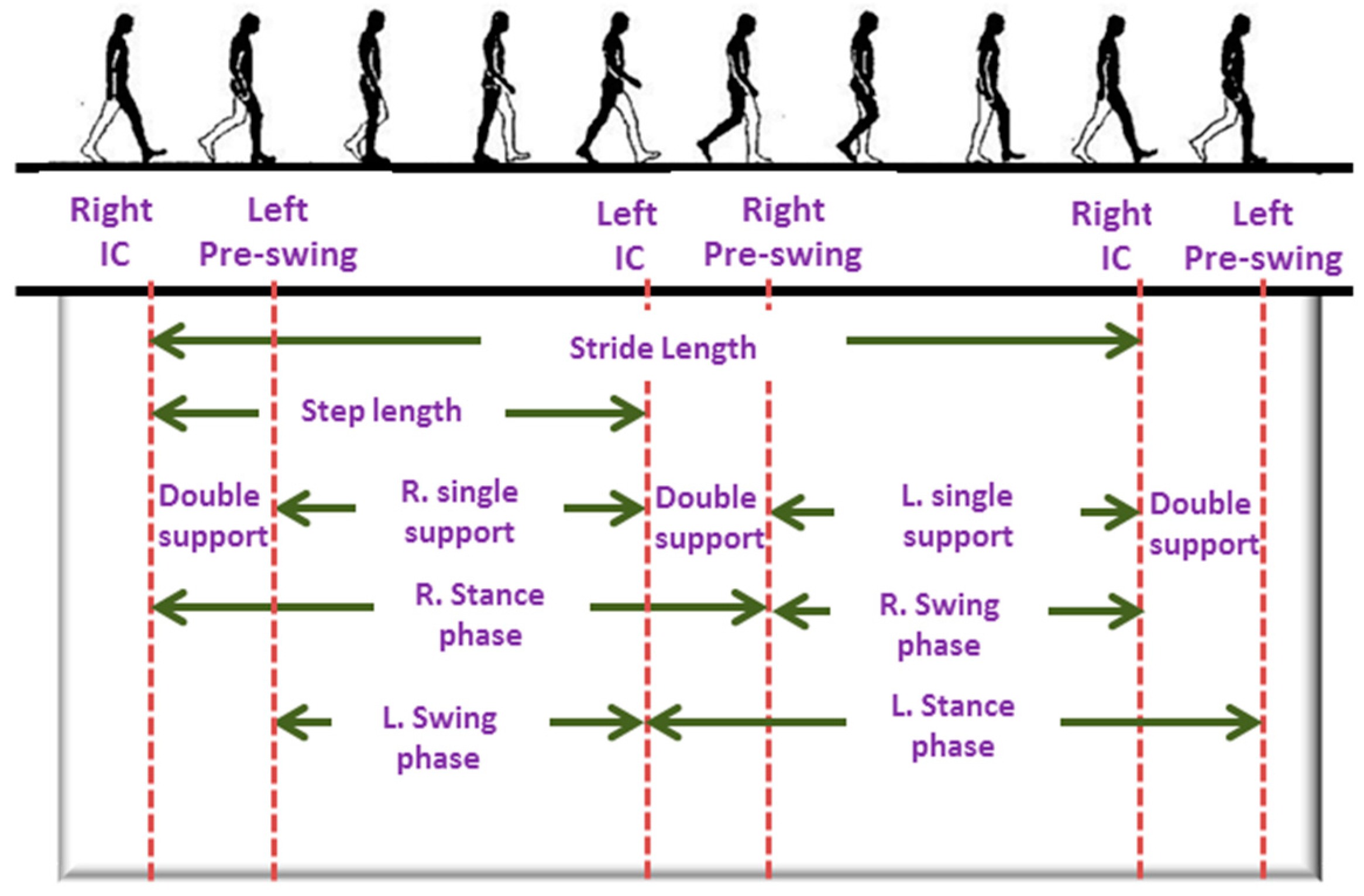
2.2. Activity Monitoring System
2.3. Body Temperature Monitoring System
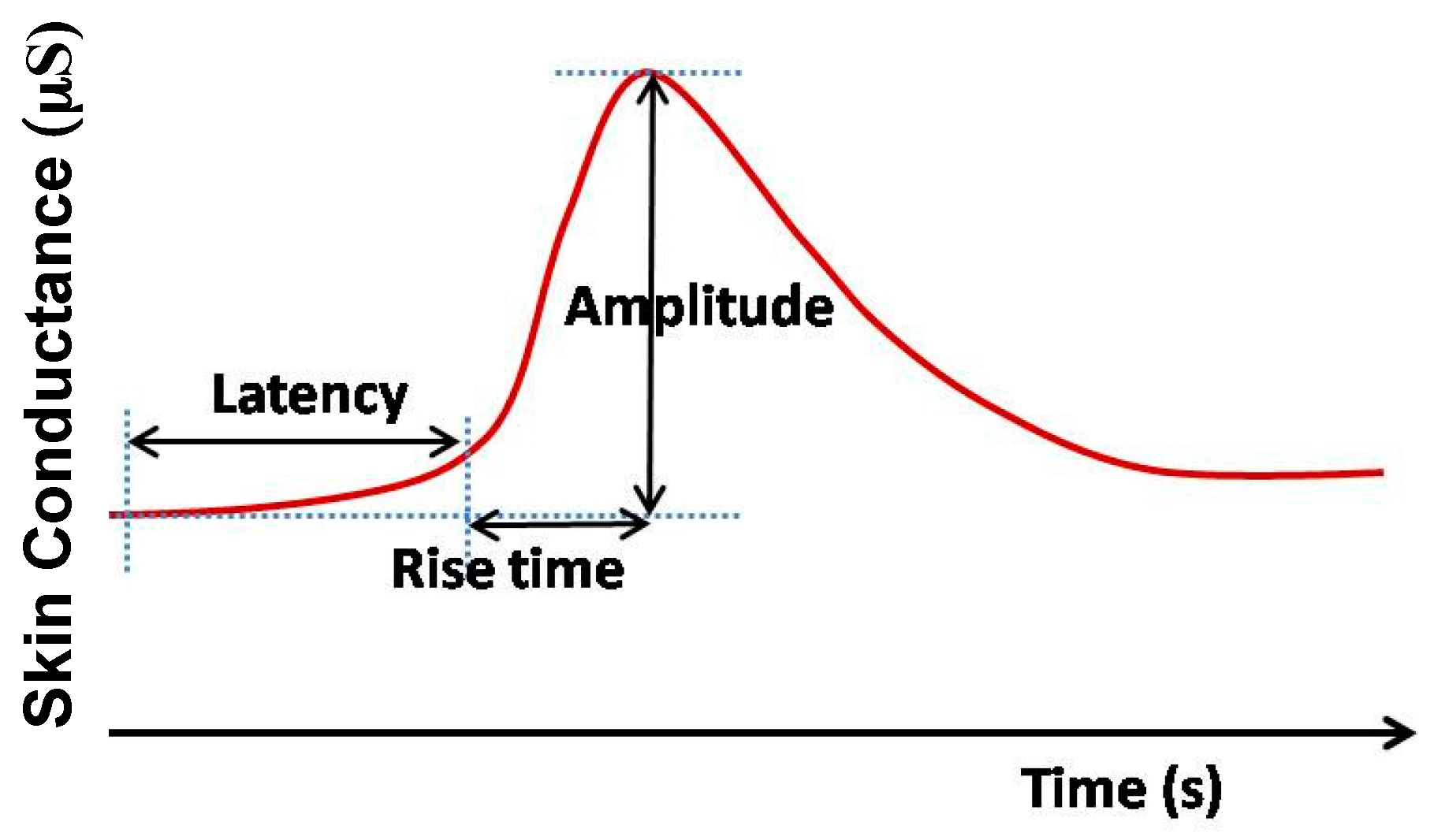
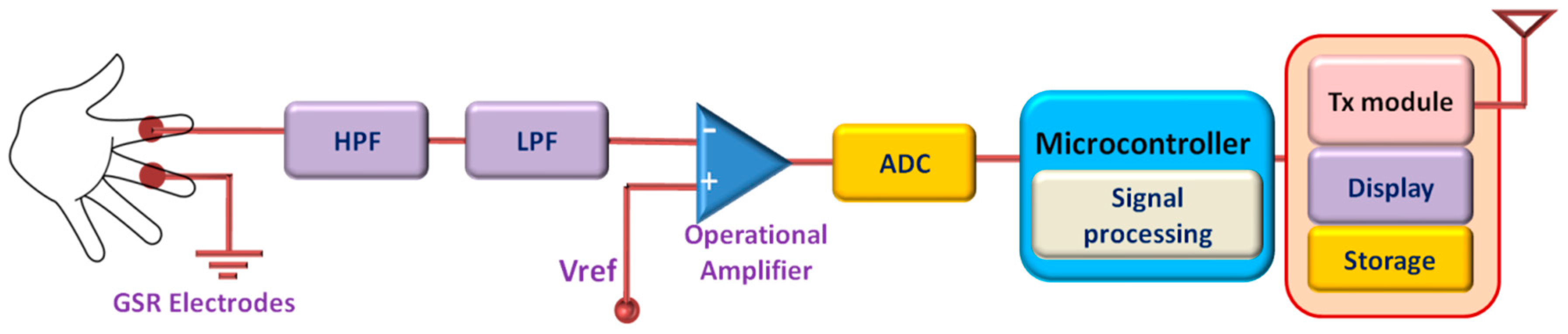
2.4. Galvanic Skin Response (GSR) Monitoring System
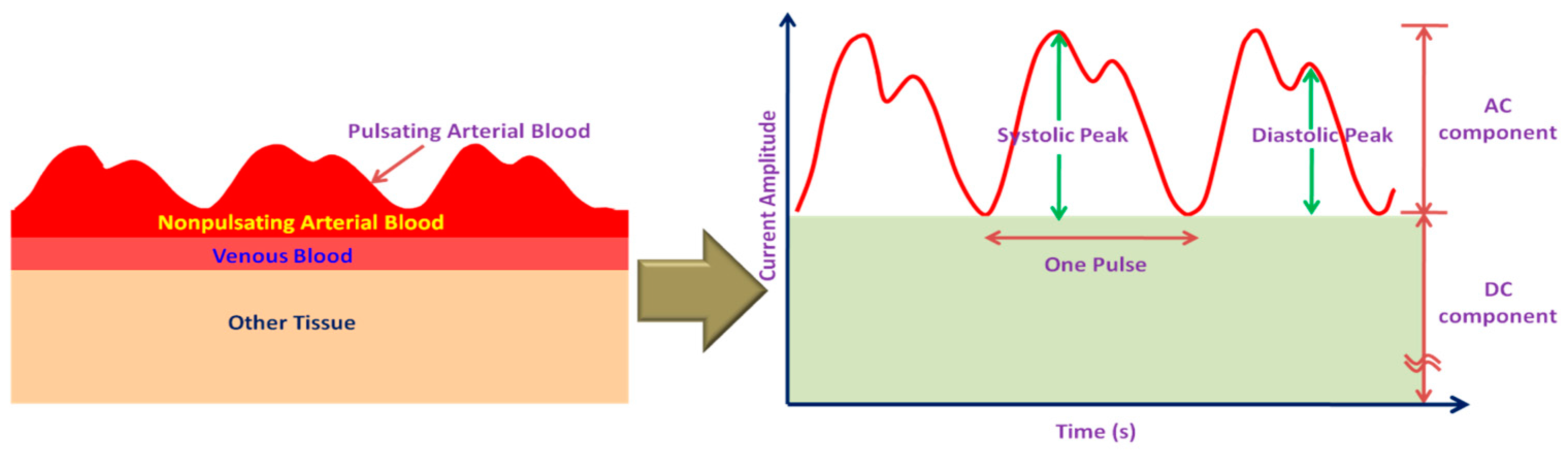
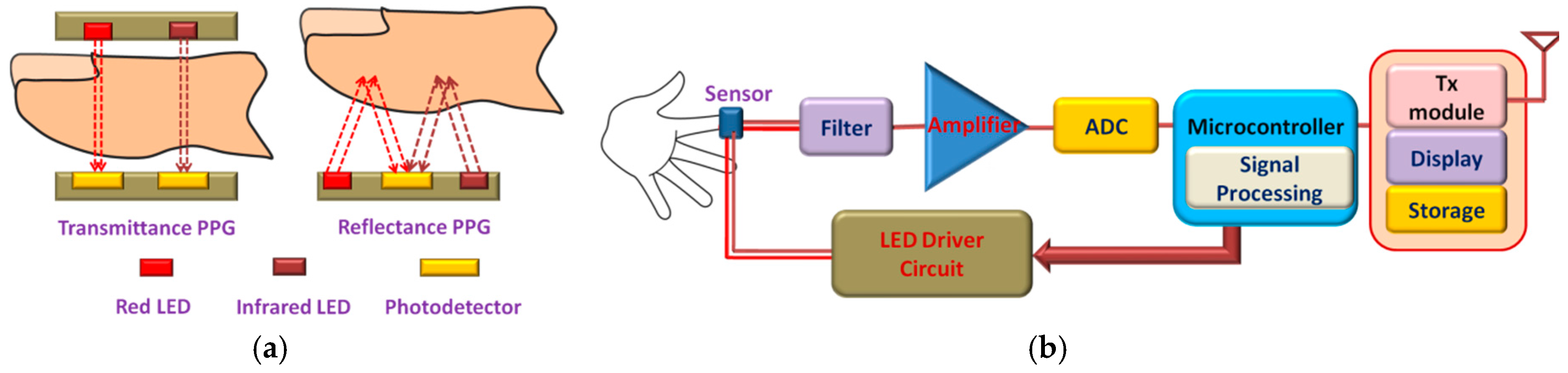
2.5. Blood Oxygen Saturation (SpO2) Monitoring Systems
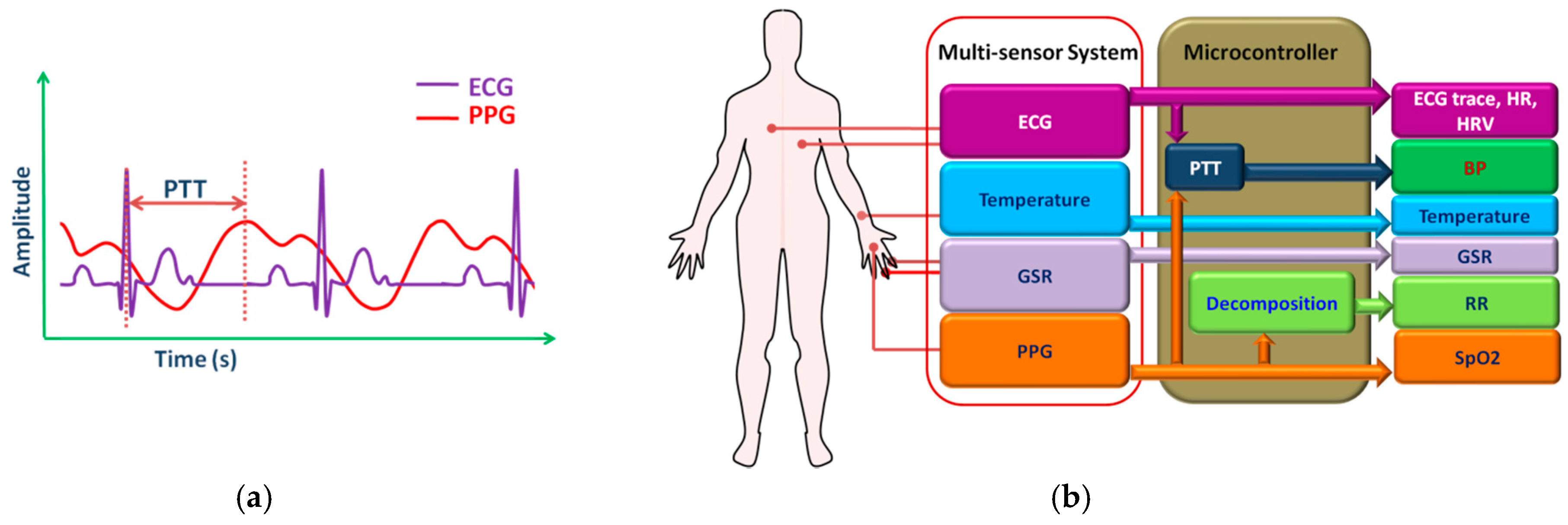
2.6. Multi-Sensor Monitoring System
3. Textile-Based Wearable Sensors
3.1. Textile Electrodes
3.2. Textile Based Temperature Sensors
3.3.Textile Sensors for Activity Measurement
4. Communication Technologies for Wearable Systems
5. Conclusions and Research Challenges
Acknowledgments
Conflicts of Interest
References
- Centers for Disease Control and Prevention. The State of Aging and Health in America 2013; Centers for Disease Control and Prevention, US Department of Health and Human Services: Atlanta, GA, USA, 2013.
- Global Age Watch Index 2015. Available online: http://www.helpage.org/global-agewatch/ (accessed on 20 June 2016).
- World Health Organization. Family Planning/Contraception. 2015. Available online: http://www.who.int/mediacentre/factsheets/fs351/en/ (accessed on 20 June 2016).
- World Health Organization. Are You Ready? What You Need to Know about Ageing. World Health Day. 2012. Available online: http://www.who.int/world-health-day/2012/toolkit/background/en/ (accessed on 20 June 2016).
- U.S. Health Care Costs Rise Faster Than Inflation. Available online: http://www.forbes.com/sites/mikepatton/2015/06/29/u-s-health-care-costs-rise-faster-than-inflation/#1384765c6ad2 (accessed on 20 June 2016).
- Deen, M.J. Information and communications technologies for elderly ubiquitous healthcare in a smart home. Pers. Ubiquitous Comput. 2015, 19, 573–599. [Google Scholar] [CrossRef]
- Agoulmine, N.; Deen, M.; Lee, J.-S.; Meyyappan, M. U-Health Smart Home. IEEE Nanotechnol. Mag. 2011, 5, 6–11. [Google Scholar] [CrossRef]
- Wang, H.; Choi, H.-S.; Agoulmine, N.; Deen, M.J.; Hong, J.W.-K. Information-based sensor tasking wireless body area networks in U-health systems. In Proceedings of the 2010 International Conference on Network and Service Management, Niagara Falls, ON, Canada, 25–29 October 2010; pp. 517–522.
- Pantelopoulos, A.; Bourbakis, N. A Survey on Wearable Sensor-Based Systems for Health Monitoring and Prognosis. IEEE Trans. Syst. Man Cybern. C 2010, 40, 1–12. [Google Scholar] [CrossRef]
- Nemati, E.; Deen, M.; Mondal, T. A wireless wearable ECG sensor for long-term applications. IEEE Commun. Mag. 2012, 50, 36–43. [Google Scholar] [CrossRef]
- Hong, Y.; Kim, I.; Ahn, S.; Kim, H. Mobile health monitoring system based on activity recognition using accelerometer. Simul. Model. Pract. Theory 2010, 18, 446–455. [Google Scholar] [CrossRef]
- Ullah, S.; Higgins, H.; Braem, B.; Latre, B.; Blondia, C.; Moerman, I.; Saleem, S.; Rahman, Z.; Kwak, K. A Comprehensive Survey of Wireless Body Area Networks. J. Med. Syst. 2012, 36, 1065–1094. [Google Scholar] [CrossRef] [PubMed]
- Al Ameen, M.; Liu, J.; Kwak, K. Security and Privacy Issues in Wireless Sensor Networks for Healthcare Applications. J. Med. Syst. 2012, 36, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Castillejo, P.; Martinez, J.; Rodriguez-Molina, J.; Cuerva, A. Integration of wearable devices in a wireless sensor network for an E-health application. IEEE Wirel. Commun. 2013, 20, 38–49. [Google Scholar] [CrossRef]
- Dementyev, A.; Hodges, S.; Taylor, S.; Smith, J. Power consumption analysis of Bluetooth Low Energy, ZigBee and ANT sensor nodes in a cyclic sleep scenario. In Proceedings of the 2013 IEEE International Wireless Symposium (IWS), Beijing, China, 14–18 April 2013; pp. 1–4.
- Suzuki, T.; Tanaka, H.; Minami, S.; Yamada, H.; Miyata, T. Wearable wireless vital monitoring technology for smart health care. In Proceedings of the 2013 7th International Symposium on Medical Information and Communication Technology (ISMICT), Tokyo, Japan, 6–8 March 2013; pp. 1–4.
- Malhi, K.; Mukhopadhyay, S.; Schnepper, J.; Haefke, M.; Ewald, H. A Zigbee-Based Wearable Physiological Parameters Monitoring System. IEEE Sens. J. 2012, 12, 423–430. [Google Scholar] [CrossRef]
- Valchinov, E.; Antoniou, A.; Rotas, K.; Pallikarakis, N. Wearable ECG System for Health and Sports Monitoring. In Proceedings of the 4th International Conference on Wireless Mobile Communication and Healthcare—“Transforming Healthcare through Innovations in Mobile and Wireless Technologies”, Athens, Greece, 3–5 November 2014; pp. 63–66.
- Mehmood, N.Q.; Culmone, R. An ANT Protocol Based Health Care System. In Proceedings of the 2015 IEEE 29th International Conference on Advanced Information Networking and Applications Workshops, Guwangiu, Korea, 24–27 March 2015; pp. 193–198.
- Coskun, V.; Ozdenizci, B.; Ok, K. A Survey on Near Field Communication (NFC) Technology. Wirel. Pers. Commun. 2013, 71, 2259–2294. [Google Scholar] [CrossRef]
- Pang, Z.; Zheng, L.; Tian, J.; Kao-Walter, S.; Dubrova, E.; Chen, Q. Design of a terminal solution for integration of in-home health care devices and services towards the Internet-of-Things. Enterp. Inf. Syst. 2013, 9, 86–116. [Google Scholar] [CrossRef]
- Corchado, J.; Bajo, J.; Abraham, A. GerAmi: Improving Healthcare Delivery in Geriatric Residences. IEEE Intell. Syst. 2008, 23, 19–25. [Google Scholar] [CrossRef]
- Stav, E.; Walderhaug, S.; Mikalsen, M.; Hanke, S.; Benc, I. Development and evaluation of SOA-based AAL services in real-life environments: A case study and lessons learned. Int. J. Med. Inform. 2013, 82, e269–e293. [Google Scholar] [CrossRef] [PubMed]
- Vaishnav, S.; Stevenson, R.; Marchant, B.; Lagi, K.; Ranjadayalan, K.; Timmis, A.D. Relation between heart rate variability early after acute myocardial infarction and long-term mortality. Am. J. Cardiol. 1994, 73, 653–657. [Google Scholar] [CrossRef]
- Bigger, J.T.; Fleiss, J.L.; Kleiger, R.; Miller, J.P.; Rolnitzky, L.M. The relationships among ventricular arrhythmias, left ventricular dysfunction, and mortality in the 2 years after myocardial infarction. Circulation 1984, 69, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Kleiger, R.E.; Miller, J.; Bigger, J.; Moss, A.J. Decreased heart rate variability and its association with increased mortality after acute myocardial infarction. Am. J. Cardiol. 1987, 59, 256–262. [Google Scholar] [CrossRef]
- Hadjem, M.; Salem, O.; Nait-Abdesselam, F. An ECG monitoring system for prediction of cardiac anomalies using WBAN. In Proceedings of the 2014 IEEE 16th International Conference on e-Health Networking, Applications and Services (Healthcom), Natal, Brazil, 15–18 October 2014.
- Andreoni, G.; Perego, P.; Standoli, C. Wearable monitoring of elderly in an ecologic setting: The SMARTA project. Available online: https://sciforum.net/conference/ecsa-2/paper/3192/download/pdf (accessed on 5 January 2017).
- Tseng, K.C.; Lin, B.-S.; Liao, L.-D.; Wang, Y.-T.; Wang, Y.-L. Development of a Wearable Mobile Electrocardiogram Monitoring System by Using Novel Dry Foam Electrodes. IEEE Syst. J. 2014, 8, 900–906. [Google Scholar] [CrossRef]
- Lee, J.; Heo, J.; Lee, W.; Lim, Y.; Kim, Y.; Park, K. Flexible Capacitive Electrodes for Minimizing Motion Artifacts in Ambulatory Electrocardiograms. Sensors 2014, 14, 14732–14743. [Google Scholar] [CrossRef] [PubMed]
- Komensky, T.; Jurcisin, M.; Ruman, K.; Kovac, O.; Laqua, D.; Husar, P. Ultra-wearable capacitive coupled and common electrode-free ECG monitoring system. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 1594–1597.
- Park, J.-H.; Jang, D.-G.; Park, J.; Youm, S.-K. Wearable Sensing of In-Ear Pressure for Heart Rate Monitoring with a Piezoelectric Sensor. Sensors 2015, 15, 23402–23417. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Li, C.; Wang, Z.; Mi, W.; Li, Y.; Ren, T.-L. A Pressure sensing system for heart rate monitoring with polymer-based pressure sensors and an anti-interference post processing circuit. Sensors 2015, 15, 3224–3235. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.; Cho, Y.-H. A Skin-attachable Flexible Piezoelectric Pulse Wave Energy Harvester. J. Phys. Conf. Ser. 2014, 557, 012026. [Google Scholar] [CrossRef]
- Tajitsu, Y. Piezoelectret sensor made from an electro-spun fluoropolymer and its use in a wristband for detecting heart-beat signals. IEEE Trans. Dielect. Electr. Insul. 2015, 22, 1355–1359. [Google Scholar] [CrossRef]
- Izumi, S.; Yamashita, K.; Nakano, M.; Kawaguchi, H.; Kimura, H.; Marumoto, K.; Fuchikami, T.; Fujimori, Y.; Nakajima, H.; Shiga, T.; et al. A Wearable Healthcare System With a 13.7 µA Noise Tolerant ECG Processor. IEEE Trans. Biomed. Circuits Syst. 2015, 9, 733–742. [Google Scholar] [CrossRef] [PubMed]
- He, D.D.; Sodini, C.G. A 58 nW ECG ASIC With Motion-Tolerant Heartbeat Timing Extraction for Wearable Cardiovascular Monitoring. IEEE Trans. Biomed. Circuits Syst. 2015, 9, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Helleputte, N.V.; Kim, S.; Kim, H.; Kim, J.P.; Hoof, C.V.; Yazicioglu, R.F. A 160 μA biopotential acquisition ASIC with fully integrated IA and motion-artifact suppression. In Proceedings of the 2012 IEEE International Solid-State Circuits Conference, San Francisco, CA, USA, 19–23 February 2012; pp. 552–561.
- Mulroy, S.; Gronley, J.; Weiss, W.; Newsam, C.; Perry, J. Use of cluster analysis for gait pattern classification of patients in the early and late recovery phases following stroke. Gait Posture 2003, 18, 114–125. [Google Scholar] [CrossRef]
- Snijders, A.H.; Warrenburg, B.P.V.D.; Giladi, N.; Bloem, B.R. Neurological gait disorders in elderly people: Clinical approach and classification. Lancet Neurol. 2007, 6, 63–74. [Google Scholar] [CrossRef]
- Coutinho, E.S.F.; Bloch, K.V.; Coeli, C.M. One-year mortality among elderly people after hospitalization due to fall-related fractures: Comparison with a control group of matched elderly. Cadernos de Saúde Pública 2012, 28, 801–805. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Dai, W.; Eggert, J.; Giger, J.; Keller, J.; Rantz, M.; He, Z. A real-time system for in-home activity monitoring of elders. In Proceedings of the 2009 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Minneapolis, MN, USA, 3–6 September 2009; pp. 6115–6118.
- Ni, B.; Wang, G.; Moulin, P. RGBD-HuDaAct: A Color-Depth Video Database for Human Daily Activity Recognition. In Consumer Depth Cameras for Computer Vision; Springer: London, UK, 2013; pp. 193–208. [Google Scholar]
- Derawi, M.; Bours, P. Gait and activity recognition using commercial phones. Comput. Secur. 2013, 39, 137–144. [Google Scholar] [CrossRef]
- De, D.; Bharti, P.; Das, S.K.; Chellappan, S. Multimodal Wearable Sensing for Fine-Grained Activity Recognition in Healthcare. IEEE Internet Comput. 2015, 19, 26–35. [Google Scholar] [CrossRef]
- Bertolotti, G.M.; Cristiani, A.M.; Colagiorgio, P.; Romano, F.; Bassani, E.; Caramia, N.; Ramat, S. A Wearable and Modular Inertial Unit for Measuring Limb Movements and Balance Control Abilities. IEEE Sens. J. 2016, 16, 790–797. [Google Scholar] [CrossRef]
- Panahandeh, G.; Mohammadiha, N.; Leijon, A.; Handel, P. Continuous Hidden Markov Model for Pedestrian Activity Classification and Gait Analysis. IEEE Trans. Instrum. Meas. 2013, 62, 1073–1083. [Google Scholar] [CrossRef]
- Bejarano, N.C.; Ambrosini, E.; Pedrocchi, A.; Ferrigno, G.; Monticone, M.; Ferrante, S. A Novel Adaptive, Real-Time Algorithm to Detect Gait Events from Wearable Sensors. IEEE Trans. Neural Syst. Rehabil. Eng. 2015, 23, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Ngo, T.T.; Makihara, Y.; Nagahara, H.; Mukaigawa, Y.; Yagi, Y. Similar gait action recognition using an inertial sensor. Pattern Recognit. 2015, 48, 1289–1301. [Google Scholar] [CrossRef]
- Alshurafa, N.; Xu, W.; Liu, J.J.; Huang, M.-C.; Mortazavi, B.; Roberts, C.K.; Sarrafzadeh, M. Designing a Robust Activity Recognition Framework for Health and Exergaming Using Wearable Sensors. IEEE J. Biomed. Health Inform. 2014, 18, 1636–1646. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, H.; Amini, N.; Saeedi, R.; Sarrafzadeh, M. Power-Aware Computing in Wearable Sensor Networks: An Optimal Feature Selection. IEEE Trans. Mob. Comput. 2015, 14, 800–812. [Google Scholar] [CrossRef]
- Chen, B.; Zheng, E.; Wang, Q.; Wang, L. A new strategy for parameter optimization to improve phase-dependent locomotion mode recognition. Neurocomputing 2015, 149, 585–593. [Google Scholar] [CrossRef]
- Cristiani, A.M.; Bertolotti, G.M.; Marenzi, E.; Ramat, S. An Instrumented Insole for Long Term Monitoring Movement, Comfort, and Ergonomics. IEEE Sens. J. 2014, 14, 1564–1572. [Google Scholar] [CrossRef]
- Tang, W.; Sazonov, E.S. Highly Accurate Recognition of Human Postures and Activities through Classification with Rejection. IEEE J. Biomed. Health Inform. 2014, 18, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Friedman, N.; Rowe, J.B.; Reinkensmeyer, D.J.; Bachman, M. The Manumeter: A Wearable Device for Monitoring Daily Use of the Wrist and Fingers. IEEE J. Biomed. Health Inform. 2014, 18, 1804–1812. [Google Scholar] [CrossRef] [PubMed]
- El-Gohary, M.; Mcnames, J. Human Joint Angle Estimation with Inertial Sensors and Validation with a Robot Arm. IEEE Trans. Biomed. Eng. 2015, 62, 1759–1767. [Google Scholar] [CrossRef] [PubMed]
- Wan, E.; Merwe, R.V.D. The unscented Kalman filter for nonlinear estimation. In Proceedings of the IEEE 2000 Adaptive Systems for Signal Processing, Communications, and Control Symposium (Cat. No. 00EX373), Lake Louise, AB, Canada, 1–4 October 2000; pp. 153–158.
- Hsu, Y.-L.; Chung, P.-C.; Wang, W.-H.; Pai, M.-C.; Wang, C.-Y.; Lin, C.-W.; Wu, H.-L.; Wang, J.-S. Gait and Balance Analysis for Patients with Alzheimer’s Disease Using an Inertial-Sensor-Based Wearable Instrument. IEEE J. Biomed. Health Inform. 2014, 18, 1822–1830. [Google Scholar] [CrossRef] [PubMed]
- Pierleoni, P.; Belli, A.; Palma, L.; Pellegrini, M.; Pernini, L.; Valenti, S. A High Reliability Wearable Device for Elderly Fall Detection. IEEE Sens. J. 2015, 15, 4544–4553. [Google Scholar] [CrossRef]
- Lack, L.C.; Gradisar, M.; Someren, E.J.V.; Wright, H.R.; Lushington, K. The relationship between insomnia and body temperatures. Sleep Med. Rev. 2008, 12, 307–317. [Google Scholar] [CrossRef] [PubMed]
- Kräuchi, K.; Konieczka, K.; Roescheisen-Weich, C.; Gompper, B.; Hauenstein, D.; Schoetzau, A.; Fraenkl, S.; Flammer, J. Diurnal and menstrual cycles in body temperature are regulated differently: A 28-day ambulatory study in healthy women with thermal discomfort of cold extremities and controls. Chronobiol. Int. 2013, 31, 102–113. [Google Scholar] [PubMed]
- Coyne, M.D.; Kesick, C.M.; Doherty, T.J.; Kolka, M.A.; Stephenson, L.A. Circadian rhythm changes in core temperature over the menstrual cycle: Method for noninvasive monitoring. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, R1316–R1320. [Google Scholar] [PubMed]
- Reith, J.; Jorgensen, H.S.; Pedersen, P.M.; Nakamaya, H.; Jeppesen, L.L.; Olsen, T.S.; Raaschou, H.O. Body temperature in acute stroke: Relation to stroke severity, infarct size, mortality, and outcome. Lancet 1996, 347, 422–425. [Google Scholar] [CrossRef]
- Wright, K.P.; Hull, J.T.; Czeisler, C.A. Relationship between alertness, performance, and body temperature in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2002, 283, R1370–R1377. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, K.; Suzuki, M.; Mizuno, A.; Tominaga, M. Effects of Body Temperature on Neural Activity in the Hippocampus: Regulation of Resting Membrane Potentials by Transient Receptor Potential Vanilloid 4. J. Neurosci. 2007, 27, 1566–1575. [Google Scholar] [CrossRef] [PubMed]
- Buller, M.J.; Tharion, W.J.; Cheuvront, S.N.; Montain, S.J.; Kenefick, R.W.; Castellani, J.; Latzka, W.A.; Roberts, W.S.; Richter, M.; Jenkins, O.C.; et al. Estimation of human core temperature from sequential heart rate observations. Physiol. Meas. 2013, 34, 781–798. [Google Scholar] [CrossRef] [PubMed]
- Buller, M.J.; Tharion, W.J.; Hoyt, R.W.; Jenkins, O.C. Estimation of human internal temperature from wearable physiological sensors. In Proceedings of the 22nd Conference on Innovative Applications of Artificial Intelligence (IAAI), Atlanta, GA, USA, 11–15 July 2010; pp. 1763–1768.
- Oguz, P.; Ertas, G. Wireless dual channel human body temperature measurement device. In Proceedings of the 2013 International Conference on Electronics, Computer and Computation (ICECCO), Ankara, Turkey, 7–9 November 2013; pp. 52–55.
- Boano, C.A.; Lasagni, M.; Romer, K.; Lange, T. Accurate Temperature Measurements for Medical Research Using Body Sensor Networks. In Proceedings of the 2011 14th IEEE International Symposium on Object/Component/Service-Oriented Real-Time Distributed Computing Workshops, Newport Beach, CA, USA, 28–31 March 2011; pp. 189–198.
- Boano, C.A.; Lasagni, M.; Romer, K. Non-invasive measurement of core body temperature in Marathon runners. In Proceedings of the 2013 IEEE International Conference on Body Sensor Networks, Cambridge, MA, USA, 6–9 May 2013; pp. 1–6.
- Chen, W.; Dols, S.; Oetomo, S.B.; Feijs, L. Monitoring body temperature of newborn infants at neonatal intensive care units using wearable sensors. In Proceedings of the Fifth International Conference on Body Area Networks—BodyNets ′10, Corfu, Greece, 10–12 September 2010; pp. 188–194.
- Mansor, H.; Shukor, M.H.A.; Meskam, S.S.; Rusli, N.Q.A.M.; Zamery, N.S. Body temperature measurement for remote health monitoring system. In Proceedings of the 2013 IEEE International Conference on Smart Instrumentation, Measurement and Applications (ICSIMA), Kuala Lumpur, Malaysia, 25–27 November 2013; pp. 1–5.
- Rahman, M.A.; Barai, A.; Islam, M.A.; Hashem, M.A. Development of a device for remote monitoring of heart rate and body temperature. In Proceedings of the 2012 15th International Conference on Computer and Information Technology (ICCIT), Chittagong, Bangladesh, 22–24 December 2012; pp. 411–416.
- Miah, M.A.; Kabir, M.H.; Tanveer, M.S.R.; Akhand, M.A.H. Continuous heart rate and body temperature monitoring system using Arduino UNO and Android device. In Proceedings of the 2015 2nd International Conference on Electrical Information and Communication Technologies (EICT), Khulna, Bangladesh, 10–12 December 2015; pp. 183–188.
- Vaz, A.; Ubarretxena, A.; Zalbide, I.; Pardo, D.; Solar, H.; Garcia-Alonso, A.; Berenguer, R. Full Passive UHF Tag With a Temperature Sensor Suitable for Human Body Temperature Monitoring. IEEE Trans. Circuits Syst. II 2010, 57, 95–99. [Google Scholar] [CrossRef]
- Milici, S.; Amendola, S.; Bianco, A.; Marrocco, G. Epidermal RFID passive sensor for body temperature measurements. In Proceedings of the 2014 IEEE RFID Technology and Applications Conference (RFID-TA), Tampere, Finland, 8–9 September 2014; pp. 140–144.
- Sim, S.Y.; Lee, W.K.; Baek, H.J.; Park, K.S. A nonintrusive temperature measuring system for estimating deep body temperature in bed. In Proceedings of the 2012 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, San Diego, CA, USA, 28 August–1 September 2012; pp. 3460–3463.
- Kimberger, O.; Thell, R.; Schuh, M.; Koch, J.; Sessler, D.I.; Kurz, A. Accuracy and precision of a novel non-invasive core thermometer. Br. J. Anaesth. 2009, 103, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, K.-I.; Zhu, X.; Chen, W.; Nemoto, T. Development of a new method for the noninvasive measurement of deep body temperature without a heater. Med. Eng. Phys. 2010, 32, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jänig, W. Integrative Action of the Autonomic Nervous System: Neurobiology of Homeostasis; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar]
- Critchley, H.D. Book Review: Electrodermal Responses: What Happens in the Brain? Neuroscientist 2002, 8, 132–142. [Google Scholar] [CrossRef]
- Bakker, J.; Pechenizkiy, M.; Sidorova, N. What's your current stress level? Detection of stress patterns from GSR sensor data. In 2011 IEEE 11th International Conference on Data Mining Workshops, Vancouver, BC, Canada, 11–14 December 2011; pp. 573–580.
- Bonato, P. Wearable sensors/systems and their impact on biomedical engineering. IEEE Eng. Med. Biol. Mag. 2003, 22, 18–20. [Google Scholar] [CrossRef] [PubMed]
- Sano, A.; Picard, R.W. Stress Recognition Using Wearable Sensors and Mobile Phones. In Proceedings of the 2013 Humaine Association Conference on Affective Computing and Intelligent Interaction, Geneva, Switzerland, 2–5 September 2013; pp. 671–676.
- Poh, M.-Z.; Swenson, N.C.; Picard, R.W. A Wearable Sensor for Unobtrusive, Long-Term Assessment of Electrodermal Activity. IEEE Trans. Biomed. Eng. 2010, 57, 1243–1252. [Google Scholar] [PubMed]
- Sugathan, A.; Roy, G.G.; Kirthyvijay, G.J.; Thomson, J. Application of arduino based platform for wearable health monitoring system. In Proceedings of the 2013 IEEE 1st International Conference on Condition Assessment Techniques in Electrical Systems (CATCON), Kolkata, India, 6–8 December 2013; pp. 1–5.
- Kim, J.; Kwon, S.; Seo, S.; Park, K. Highly wearable galvanic skin response sensor using flexible and conductive polymer foam. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 6631–6634.
- Garbarino, M.; Lai, M.; Tognetti, S.; Picard, R.; Bender, D. Empatica E3—A wearable wireless multi-sensor device for real-time computerized biofeedback and data acquisition. In Proceedings of the 4th International Conference on Wireless Mobile Communication and Healthcare—Transforming healthcare through innovations in mobile and wireless technologies, Athens, Greece, 3–5 November 2014; pp. 39–42.
- Guo, R.; Li, S.; He, L.; Gao, W.; Qi, H.; Owens, G. Pervasive and Unobtrusive Emotion Sensing for Human Mental Health. In Proceedings of the 7th International Conference on Pervasive Computing Technologies for Healthcare, Venice, Italy, 5–8 May 2013; pp. 436–439.
- Setz, C.; Arnrich, B.; Schumm, J.; Marca, R.L.; Troster, G.; Ehlert, U. Discriminating Stress From Cognitive Load Using a Wearable EDA Device. IEEE Trans. Inform. Technol. Biomed. 2010, 14, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Crifaci, G.; Billeci, L.; Tartarisco, G.; Balocchi, R.; Pioggia, G.; Brunori, E.; Maestro, S.; Morales, M.A. ECG and GSR measure and analysis using wearable systems: Application in anorexia nervosa adolescents. In Proceedings of the 2013 8th International Symposium on Image and Signal Processing and Analysis (ISPA), Trieste, Italy, 4–6 September 2013; pp. 499–504.
- Subramanya, K.; Bhat, V.V.; Kamath, S. A wearable device for monitoring galvanic skin response to accurately predict changes in blood pressure indexes and cardiovascular dynamics. In Proceedings of the 2013 Annual IEEE India Conference (INDICON), Mumbai, India, 13–15 December 2013; pp. 1–4.
- Yoon, Y.; Cho, J.H.; Yoon, G. Non-constrained Blood Pressure Monitoring Using ECG and PPG for Personal Healthcare. J. Med. Syst. 2008, 33, 261–266. [Google Scholar] [CrossRef]
- Blacher, J.; Staessen, J.A.; Girerd, X.; Gasowski, J.; Thijs, L.; Liu, L.; Wang, J.G.; Fagard, R.H.; Safar, M.E. Pulse Pressure Not Mean Pressure Determines Cardiovascular Risk in Older Hypertensive Patients. Arch. Intern. Med. 2000, 160, 1085–1089. [Google Scholar] [CrossRef] [PubMed]
- Baker, C. Method and System for Controlled Maintenance of Hypoxia for Therapeutic or Diagnostic Purposes. U.S. Patent No. US 11/241,062, 30 September 2005. [Google Scholar]
- O’driscoll, B.R.; Howard, L.S.; Davison, A.G. BTS guideline for emergency oxygen use in adult patients. Thorax 2008, 63, vi1–vi68. [Google Scholar] [CrossRef] [PubMed]
- Duun, S.B.; Haahr, R.G.; Birkelund, K.; Thomsen, E.V. A Ring-Shaped Photodiode Designed for Use in a Reflectance Pulse Oximetry Sensor in Wireless Health Monitoring Applications. IEEE Sens. J. 2010, 10, 261–268. [Google Scholar] [CrossRef]
- Chen, W.; Ayoola, I.; Oetomo, S.B.; Feijs, L. Non-invasive blood oxygen saturation monitoring for neonates using reflectance pulse oximeter. In Proceedings of the 2010 Design, Automation & Test in Europe Conference & Exhibition (DATE 2010), Dresden, Germany, 8–12 March 2010; pp. 1530–1535.
- Li, K.; Warren, S. A Wireless Reflectance Pulse Oximeter with Digital Baseline Control for Unfiltered Photoplethysmograms. IEEE Trans. Biomed. Circuits Syst. 2012, 6, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Tay, F.E.; Xu, L.; Yu, L.; Nyan, M.; Chong, F.; Yap, K.; Xu, B. A Long-term Wearable Vital Signs Monitoring System using BSN. In Proceedings of the 2008 11th EUROMICRO Conference on Digital System Design Architectures, Methods and Tools, Parma, Italy, 3–5 September 2008; pp. 825–830.
- Petersen, C.; Chen, T.; Ansermino, J.; Dumont, G. Design and Evaluation of a Low-Cost Smartphone Pulse Oximeter. Sensors 2013, 13, 16882–16893. [Google Scholar] [CrossRef] [PubMed]
- Sola, J.; Castoldi, S.; Chetelat, O.; Correvon, M.; Dasen, S.; Droz, S.; Jacob, N.; Kormann, R.; Neumann, V.; Perrenoud, A.; et al. SpO2 Sensor Embedded in a Finger Ring: Design and implementation. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 31 August–3 September 2006; pp. 4295–4298.
- Cai, Q.; Sun, J.; Xia, L.; Zhao, X. Implementation of a wireless pulse oximeter based on wrist band sensor. In Proceedings of the 2010 3rd International Conference on Biomedical Engineering and Informatics, Yantai, China, 16–18 October 2010; pp. 1897–1900.
- Huang, C.-Y.; Chan, M.-C.; Chen, C.-Y.; Lin, B.-S. Novel Wearable and Wireless Ring-Type Pulse Oximeter with Multi-Detectors. Sensors 2014, 14, 17586–17599. [Google Scholar] [CrossRef] [PubMed]
- Deen, M.J.; Basu, P.K. Silicon Photonics—Fundamentals and Devices; John Wiley and Sons: Chichester, UK, 2012. [Google Scholar]
- Palubiak, D.; El-Desouki, M.M.; Marinov, O.; Deen, M.J.; Fang, Q. High-speed, single-photon avalanche-photodiode imager for biomedical applications. IEEE Sens. J. 2011, 11, 2401–2412. [Google Scholar] [CrossRef]
- Tavakoli, M.; Turicchia, L.; Sarpeshkar, R. An Ultra-Low-Power Pulse Oximeter Implemented With an Energy-Efficient Transimpedance Amplifier. IEEE Trans. Biomed. Circuits Syst. 2010, 4, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Mendelson, Y.; Duckworth, R.J.; Comtois, G. A Wearable Reflectance Pulse Oximeter for Remote Physiological Monitoring. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 31 August–3 September 2006; pp. 912–915.
- Haahr, R.G.; Duun, S.B.; Toft, M.H.; Belhage, B.; Larsen, J.; Birkelund, K.; Thomsen, E.V. An Electronic Patch for Wearable Health Monitoring by Reflectance Pulse Oximetry. IEEE Trans. Biomed. Circuits Syst. 2012, 6, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Scully, C.G.; Lee, J.; Meyer, J.; Gorbach, A.M.; Granquist-Fraser, D.; Mendelson, Y.; Chon, K.H. Physiological Parameter Monitoring from Optical Recordings With a Mobile Phone. IEEE Trans. Biomed. Eng. 2012, 59, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Buxi, D.; Redouté, J.M.; Yuce, M.R. A survey on signals and systems in ambulatory blood pressure monitoring using pulse transit time. Physiol. Meas. 2015, 36, R1–R26. [Google Scholar] [CrossRef] [PubMed]
- Mccombie, D.B.; Reisner, A.T.; Asada, H.H. Adaptive blood pressure estimation from wearable PPG sensors using peripheral artery pulse wave velocity measurements and multi-channel blind identification of local arterial dynamics. In Proceedings of the 2006 International Conference of the IEEE Engineering in Medicine and Biology Society, New York, NY, USA, 31 August–3 September 2006; pp. 3521–3524.
- Puke, S.; Suzuki, T.; Nakayama, K.; Tanaka, H.; Minami, S. Blood pressure estimation from pulse wave velocity measured on the chest. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013; pp. 6107–6110.
- Zheng, Y.-L.; Yan, B.P.; Zhang, Y.-T.; Poon, C.C.Y. An Armband Wearable Device for Overnight and Cuff-Less Blood Pressure Measurement. IEEE Trans. Biomed. Eng. 2014, 61, 2179–2186. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Xu, W.; Guan, N.; Ji, D.; Wei, Y.; Yi, W. Noninvasive and Continuous Blood Pressure Monitoring Using Wearable Body Sensor Networks. IEEE Intell. Syst. 2015, 30, 38–48. [Google Scholar] [CrossRef]
- Allen, J. Photoplethysmography and its application in clinical physiological measurement. Physiol. Meas. 2007, 28, R1–R39. [Google Scholar] [CrossRef] [PubMed]
- Madhav, K.V.; Ram, M.R.; Krishna, E.H.; Komalla, N.R.; Reddy, K.A. Estimation of respiration rate from ECG, BP and PPG signals using empirical mode decomposition. In Proceedings of the 2011 IEEE International Instrumentation and Measurement Technology Conference, Hangzhou, China, 10–12 May 2011; pp. 1–4.
- Prathyusha, B.; Rao, T.S.; Asha, D. Extraction of Respiratory Rate from Ppg Signals Using Pca and Emd. Int. J. Res. Eng. Technol. 2012, 1, 164–184. [Google Scholar]
- Madhav, K.V.; Raghuram, M.; Krishna, E.H.; Komalla, N.R.; Reddy, K.A. Use of multi scale PCA for extraction of respiratory activity from photoplethysmographic signals. In Proceedings of the 2012 IEEE International Instrumentation and Measurement Technology Conference, Graz, Austria, 13–16 May 2012; pp. 1784–1787.
- Leonard, P.A.; Douglas, J.G.; Grubb, N.R.; Clifton, D.; Addison, P.S.; Watson, J.N. A Fully Automated Algorithm for the Determination of Respiratory Rate from the Photoplethysmogram. J. Clin. Monit. Comput. 2006, 20, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Clifton, D.; Douglas, J.G.; Addison, P.S.; Watson, J.N. Measurement of Respiratory Rate from the Photoplethysmogram in Chest Clinic Patients. J. Clin. Monit. Comput. 2006, 21, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Cherenack, K.; Pieterson, L.V. Smart textiles: Challenges and opportunities. J. Appl. Phys. 2012, 112, 091301. [Google Scholar] [CrossRef]
- Chan, M.; Estève, D.; Fourniols, J.-Y.; Escriba, C.; Campo, E. Smart wearable systems: Current status and future challenges. Artif. Intell. Med. 2012, 56, 137–156. [Google Scholar] [CrossRef] [PubMed]
- Sibinski, M.; Jakubowska, M.; Sloma, M. Flexible Temperature Sensors on Fibers. Sensors 2010, 10, 7934–7946. [Google Scholar] [CrossRef] [PubMed]
- Zeng, W.; Shu, L.; Li, Q.; Chen, S.; Wang, F.; Tao, X.-M. Fiber-based wearable electronics: A review of materials, fabrication, devices, applications. Adv. Mater. 2014, 26, 5310–5336. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Vosguerichian, M.; Bao, Z. A review of fabrication and applications of carbon nanotube film-based flexible electronics. Nanoscale 2013, 5, 1727–1752. [Google Scholar] [CrossRef] [PubMed]
- Rai, P.; Kumar, P.S.; Oh, S.; Kwon, H.; Mathur, G.N.; Varadan, V.K.; Agarwal, M.P. Smart healthcare textile sensor system for unhindered-pervasive health monitoring. Nanosens. Biosens. Info-Tech Sens. Syst. 2012, 8344E. [Google Scholar]
- Rai, P.; Oh, S.; Shyamkumar, P.; Ramasamy, M.; Harbaugh, R.E.; Varadan, V.K. Nano-Bio-Textile Sensors with Mobile Wireless Platform for Wearable Health Monitoring of Neurological and Cardiovascular Disorders. J. Electrochem. Soc. 2014, 161, B3116–B3150. [Google Scholar] [CrossRef]
- Fuhrhop, S.; Lamparth, S.; Heuer, S. A textile integrated long-term ECG monitor with capacitively coupled electrodes. In Proceedings of the 2009 IEEE Biomedical Circuits and Systems Conference, Beijing, China, 26–28 Novemeber 2009; pp. 21–24.
- Ouwerkerk, M.; Pasveer, F.; Langereis, G. Unobtrusive Sensing of Psychophysiological Parameters. In Probing Experience; Springer: Dordrecht, The Netherlands, 2008; pp. 163–193. [Google Scholar]
- Merritt, C.; Nagle, H.; Grant, E. Fabric-Based Active Electrode Design and Fabrication for Health Monitoring Clothing. IEEE Trans. Inform. Technol. Biomed. 2009, 13, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, C.; Cunha, J.P.S.; Martins, R.E.; Ferreira, V.M.; Sa, J.P.M.D.; Barbosa, M.A.; Silva, A.M.D. A Novel Dry Active Electrode for EEG Recording. IEEE Trans. Biomed. Eng. 2007, 54, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Keller, T.; Kuhn, A. Electrodes for transcutaneous (surface) electrical stimulation. J. Autom. Control 2008, 18, 35–45. [Google Scholar] [CrossRef]
- Li, L.; Au, W.M.; Li, Y.; Wan, K.M.; Wan, S.H.; Wong, K.S. Design of Intelligent Garment with Transcutaneous Electrical Nerve Stimulation Function Based on the Intarsia Knitting Technique. Text. Res. J. 2009, 80, 279–286. [Google Scholar] [CrossRef]
- Malešević, N.M.; Maneski, L.Z.; Ilić, V.; Jorgovanović, N.; Bijelić, G.; Keller, T.; Popović, D.B. A multi-pad electrode based functional electrical stimulation system for restoration of grasp. J. Neuroeng. Rehabil. 2012, 9, 66. [Google Scholar] [CrossRef] [PubMed]
- Paul, G.; Torah, R.; Beeby, S.; Tudor, J. Novel active electrodes for ECG monitoring on woven textiles fabricated by screen and stencil printing. Sens. Actuators A Phys. 2015, 221, 60–66. [Google Scholar] [CrossRef]
- Lim, Z.; Chia, Z.; Kevin, M.; Wong, A.; Ho, G. A facile approach towards ZnO nanorods conductive textile for room temperature multifunctional sensors. Sens. Actuators B Chem. 2010, 151, 121–126. [Google Scholar] [CrossRef]
- Inoh, T.; Yoon, S.; Kim, T.E.; Wi, H.; Kim, K.J.; Woo, E.J.; Sadleir, R.J. Nanofiber Web Textile Dry Electrodes for Long-Term Biopotential Recording. IEEE Trans. Biomed. Circuits Syst. 2013, 7, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, T.N.; Chaudhari, S.; Patel, B.H.; Patel, M. Study of conductivity behavior of nano copper loaded nonwoven polypropylene based textile electrode for ECG. Int. J. Emerg. Sci. Eng. 2015, 3, 11–14. [Google Scholar]
- Rattfält, L.; Björefors, F.; Nilsson, D.; Wang, X.; Norberg, P.; Ask, P. Properties of screen printed electrocardiography smartware electrodes investigated in an electro-chemical cell. Biomed. Eng. Online 2013, 12, 64. [Google Scholar] [CrossRef] [PubMed]
- Cho, G.; Jeong, K.; Paik, M.J.; Kwun, Y.; Sung, M. Performance Evaluation of Textile-Based Electrodes and Motion Sensors for Smart Clothing. IEEE Sens. J. 2011, 11, 3183–3193. [Google Scholar] [CrossRef]
- Mestrovic, M.A.; Helmer, R.J.N.; Kyratzis, L.; Kumar, D. Preliminary study of dry knitted fabric electrodes for physiological monitoring. In Proceedings of the 2007 3rd International Conference on Intelligent Sensors, Sensor Networks and Information, Melbourne, Australia, 3–6 December 2007; pp. 601–606.
- Cho, H.; Lim, H.; Cho, S.; Lee, J.-W. Development of textile electrode for electrocardiogram measurement based on conductive electrode configuration. Fibers Polym. 2015, 16, 2148–2157. [Google Scholar] [CrossRef]
- Weder, M.; Hegemann, D.; Amberg, M.; Hess, M.; Boesel, L.; Abächerli, R.; Meyer, V.; Rossi, R. Embroidered Electrode with Silver/Titanium Coating for Long-Term ECG Monitoring. Sensors 2015, 15, 1750–1759. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.Q.; Newton, E.; Yuen, C.W.M.; Kan, C.W. Chemical silver plating on polyester/cotton blended fabric. J. Appl. Polym. Sci. 2006, 100, 4383–4387. [Google Scholar] [CrossRef]
- Xue, P.; Tao, X.M. Morphological and electromechanical studies of fibers coated with electrically conductive polymer. J. Appl. Polym. Sci. 2005, 98, 1844–1854. [Google Scholar] [CrossRef]
- Brevnov, D.A. Electrodeposition of Porous Silver Films on Blanket and Patterned Aluminum—Copper Films. J. Electrochem. Soc. 2006, 153, C249–C253. [Google Scholar] [CrossRef]
- Doezema, D.; Lunt, M.; Tandberg, D. Cerumen Occlusion Lowers Infrared Tympanic Membrane Temperature Measurement. Acad. Emerg. Med. 1995, 2, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Sund-Levander, M.; Grodzinsky, E. Time for a change to assess and evaluate body temperature in clinical practice. Int. J. Nurs. Pract. 2009, 15, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Togawa, T. Body temperature measurement. Clin. Phys. Physiol. Meas. 1985, 6, 83–108. [Google Scholar] [CrossRef] [PubMed]
- Niedermann, R.; Wyss, E.; Annaheim, S.; Psikuta, A.; Davey, S.; Rossi, R.M. Prediction of human core body temperature using non-invasive measurement methods. Int. J. Biometeorol. 2014, 58, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Imran, M.; Bhattacharyya, A. Thermal response of an on-chip assembly of RTD heaters, sputtered sample and microthermocouples. Sens. Actuators A Phys. 2005, 121, 306–320. [Google Scholar] [CrossRef]
- Jeon, J.; Lee, H.-B.-R.; Bao, Z. Flexible Wireless Temperature Sensors Based on Ni Microparticle-Filled Binary Polymer Composites. Adv. Mater. 2012, 25, 850–855. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, S.; Frydrysiak, M. Initial research into the structure and working conditions of textile thermocouples. Fibres Text. East. Eur. 2009, 17, 84–88. [Google Scholar]
- Houdas, Y.; Ring, E.F.J. Human Body Temperature: Its Measurement and Regulation; Springer Science & Business Media: New York, NY, USA, 1982. [Google Scholar]
- Cooper, K.; Veale, W.; Malkinson, T. Measurements of Body Temperature. Methods Psychobiol. Adv. Lab. Tech. Neuropsychol. 1977, 3, 149–187. [Google Scholar]
- Husain, M.D.; Kennon, R.; Dias, T. Design and fabrication of Temperature Sensing Fabric. J. Ind. Text. 2014, 44, 398–417. [Google Scholar] [CrossRef]
- Kinkeldei, T.; Zysset, C.; Cherenack, K.; Troester, G. Development and evaluation of temperature sensors for textile integration. In Proceedings of IEEE Sensors 2009 Conference, Christchurch, New Zealand, 25–28 October 2009; pp. 1580–1583.
- Soukup, R.; Hamacek, A.; Mracek, L.; Reboun, J. Textile based temperature and humidity sensor elements for healthcare applications. In Proceedings of the 2014 37th International Spring Seminar on Electronics Technology, Dresden, Germany, 7–11 May 2014; pp. 407–411.
- Bielska, S.; Sibinski, M.; Lukasik, A. Polymer temperature sensor for textronic applications. Mater. Sci. Eng. B 2009, 165, 50–52. [Google Scholar] [CrossRef]
- Dankoco, M.; Tesfay, G.; Benevent, E.; Bendahan, M. Temperature sensor realized by inkjet printing process on flexible substrate. Mater. Sci. Eng. B 2016, 205, 1–5. [Google Scholar] [CrossRef]
- Honda, W.; Harada, S.; Arie, T.; Akita, S.; Takei, K. Printed wearable temperature sensor for health monitoring. In Proceedings of the 2014 IEEE Sensors, Valencia, Spain, 2–5 November 2014; pp. 2227–2229.
- Yang, J.; Wei, D.; Tang, L.; Song, X.; Luo, W.; Chu, J.; Gao, T.; Shi, H.; Du, C. Wearable temperature sensor based on graphene nanowalls. RSC Adv. 2015, 5, 25609–25615. [Google Scholar] [CrossRef]
- Courbat, J.; Kim, Y.; Briand, D.; Rooij, N.D. Inkjet printing on paper for the realization of humidity and temperature sensors. In Proceedings of the 2011 16th International Solid-State Sensors, Actuators and Microsystems Conference, Beijing, China, 5–9 June 2011; pp. 1356–1359.
- Aliane, A.; Fischer, V.; Galliari, M.; Tournon, L.; Gwoziecki, R.; Serbutoviez, C.; Chartier, I.; Coppard, R. Enhanced printed temperature sensors on flexible substrate. Microelectron. J. 2014, 45, 1621–1626. [Google Scholar] [CrossRef]
- Li, H.; Yang, H.; Li, E.; Liu, Z.; Wei, K. Wearable sensors in intelligent clothing for measuring human body temperature based on optical fiber Bragg grating. Opt. Express 2012, 20, 11740–11752. [Google Scholar] [CrossRef] [PubMed]
- Rajdi, N.N.Z.M.; Bakira, A.A.; Saleh, S.M.; Wicaksono, D.H. Textile-based Micro Electro Mechanical System (MEMS) Accelerometer for Pelvic Tilt Mesurement. Procedia Eng. 2012, 41, 532–537. [Google Scholar] [CrossRef]
- Amjadi, M.; Pichitpajongkit, A.; Lee, S.; Ryu, S.; Park, I. Highly Stretchable and Sensitive Strain Sensor Based on Silver Nanowire–Elastomer Nanocomposite. ACS Nano 2014, 8, 5154–5163. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, S.; Lee, J.; Yang, D.; Park, B.C.; Ryu, S.; Park, I. A stretchable strain sensor based on a metal nanoparticle thin film for human motion detection. Nanoscale 2014, 6, 11932–11939. [Google Scholar] [CrossRef] [PubMed]
- Shyr, T.-W.; Shie, J.-W.; Jiang, C.-H.; Li, J.-J. A Textile-Based Wearable Sensing Device Designed for Monitoring the Flexion Angle of Elbow and Knee Movements. Sensors 2014, 14, 4050–4059. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H. Flexible textile-based strain sensor induced by contacts. Meas. Sci. Technol. 2015, 26, 105102. [Google Scholar] [CrossRef]
- Atalay, O.; Kennon, W.; Husain, M. Textile-Based Weft Knitted Strain Sensors: Effect of Fabric Parameters on Sensor Properties. Sensors 2013, 13, 11114–11127. [Google Scholar] [CrossRef] [PubMed]
- Atalay, O.; Kennon, W. Knitted Strain Sensors: Impact of Design Parameters on Sensing Properties. Sensors 2014, 14, 4712–4730. [Google Scholar] [CrossRef] [PubMed]
- Seyedin, S.; Razal, J.M.; Innis, P.C.; Jeiranikhameneh, A.; Beirne, S.; Wallace, G.G. Knitted Strain Sensor Textiles of Highly Conductive All-Polymeric Fibers. ACS Appl. Mater. Interfaces 2015, 7, 21150–21158. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.F.D.; Pedro, R.; Paulo, J.; Higino, J. Photonic Sensors Based on Flexible Materials with FBGs for Use on Biomedical Applications. In Current Trends in Short- and Long-Period Fiber Gratings; InTech: Rijeka, Croatia, 2013; pp. 105–132. [Google Scholar]
- Krehel, M.; Rossi, R.; Bona, G.-L.; Scherer, L. Characterization of Flexible Copolymer Optical Fibers for Force Sensing Applications. Sensors 2013, 13, 11956–11968. [Google Scholar] [CrossRef] [PubMed]
- Halonen, T.; Romero, J.; Melero, J. GSM, GPRS and EDGE Performance: Evolution towards 3G/UMTS; John Wiley & Sons: Hoboken, NJ, USA, 2002. [Google Scholar]
- Salkintzis, K. Mobile Internet: Enabling Technologies and Services; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Dahlman, E.; Parkvall, S.; Skold, J.; Beming, P. 3G Evolution: HSPA and LTE for Mobile Broadband; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Ren, Y.; Werner, R.; Pazzi, N.; Boukerche, A. Monitoring patients via a secure and mobile healthcare system. IEEE Wirel. Commun. 2010, 17, 59–65. [Google Scholar] [CrossRef]
- Jang, C.S.; Lee, D.G.; Han, J.-W.; Park, J.H. Hybrid security protocol for wireless body area networks. Wirel. Commun. Mob. Comput. 2011, 11, 277–288. [Google Scholar] [CrossRef]
- Agrawal, V. Security and Privacy Issues in Wireless Sensor Networks for Healthcare. Internet Things User-Centric IoT 2015, 36, 223–228. [Google Scholar]
- Shoshani, B.; David, R.B. Vertical Conductive Textile Traces and Methods of Knitting Thereof. U.S. Patent No. US 14/646,971, 23 November 2013. [Google Scholar]
- Matsuhisa, N.; Kaltenbrunner, M.; Yokota, T.; Jinno, H.; Kuribara, K.; Sekitani, T.; Someya, T. Printable elastic conductors with a high conductivity for electronic textile applications. Nat. Commun. 2015, 6, 7461. [Google Scholar] [CrossRef] [PubMed]
- Parkova, I.; Vališevskis, A.; Ziemele, I.; Briedis, U.; Vilumsone, A. Improvements of Smart Garment Electronic Contact System. Adv. Sci. Technol. 2013, 80, 90–95. [Google Scholar] [CrossRef]
- Laine, T.H.; Lee, C.; Suk, H. Mobile Gateway for Ubiquitous Health Care System Using ZigBee and Bluetooth. In Proceedings of the 2014 Eighth International Conference on Innovative Mobile and Internet Services in Ubiquitous Computing, Birmingham, UK, 2–4 July 2014; pp. 139–145.
- Touati, F.; Tabish, R. U-Healthcare System: State-of-the-Art Review and Challenges. J. Med. Syst. 2013, 37, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.; Shen, W.; Xu, Y. An ANT Based Wireless Body Sensor Biofeedback Network for Medical E-Health Care. In Proceedings of the 2011 7th International Conference on Wireless Communications, Networking and Mobile Computing, Wuhan, China, 23–25 September 2011; pp. 1–5.
- Savci, H.; Sula, A.; Wang, Z.; Dogan, N.; Arvas, E. MICS Transceivers: Regulatory Standards and Applications. In IEEE SoutheastCon Proceedings; IEEE: Piscataway, NJ, USA, 2005; pp. 179–182. [Google Scholar]
- Fang, Q.; Lee, S.Y.; Permana, H.; Ghorbani, K.; Cosic, I. Developing a Wireless Implantable Body Sensor Network in MICS Band. IEEE Trans. Inform. Technol. Biomed. 2011, 15, 567–576. [Google Scholar] [CrossRef] [PubMed]
- Zhen, B.; Li, H.B.; Kohno, R. Networking issues in medical implant communications. Int. J. Multimed. Ubiquitous Eng. 2009, 4, 23–38. [Google Scholar]
- Lazaro, A.; Girbau, D.; Villarino, R. Analysis of Vital Signs Monitoring Using an Ir-Uwb Radar. Prog. Electromagn. Res. 2010, 100, 265–284. [Google Scholar] [CrossRef]
- Chu, Y.; Ganz, A. A UWB-based 3D location system for indoor environments. In Proceedings of the 2nd International Conference on Broadband Networks, Boston, MA, USA, 3–7 October 2005; pp. 1147–1155.
- Yoo, H.-J.; Cho, N. Body channel communication for low energy BSN/BAN. In Proceedings of the APCCAS 2008—2008 IEEE Asia Pacific Conference on Circuits and Systems, Macao, China, 30 November–3 December 2008; pp. 7–11.
- Sole, M.; Musu, C.; Boi, F.; Giusto, D.; Popescu, V. RFID sensor network for workplace safety management. In Proceedings of the 2013 IEEE 18th Conference on Emerging Technologies & Factory Automation (ETFA), Cagliari, Italy, 10–13 September 2013; pp. 1–4.
- Liu, Y. A Heart Rate Finger Ring and Its Smartphone APP through Customized NFC. Master’s Thesis, Rose-Hulman Institute of Technology, Terre Haute, IN, USA, 2015. [Google Scholar]
- Fontecha, J.; Hervas, R.; Bravo, J.; Villarreal, V. An NFC Approach for Nursing Care Training. In Proceedings of the 2011 Third International Workshop on Near Field Communication, Hagenberg, Austria, 22–23 February 2011; pp. 38–43.



| Product Name | Monitored Parameters | Wireless Platform | Battery | |
|---|---|---|---|---|
| Type | Life | |||
| Hexoskin® Biometric® Shirt | Heart rate (HR), HR variability, respiratory rate, number of steps, distance traveled, pace, maximal oxygen consumption, and calories burned. | Bluetooth | 6–7 days (standalone) 14+ h (multi-training) | |
| Jawbone UP3™ Fitness Tracker | Sleep stages (REM, light and deep), HR, food and liquid intake, number of steps, distance traveled, running. | Bluetooth LE | Li-ion poly | 7 days |
| Striiv® Fusion Bio Fitness Tracker | HR, number of steps, distance traveled, calories burned, and sleep quality. | Bluetooth LE | Li-ion | 5 days |
| Microsoft® Band 2 | HR, calories burned, sleep quality, food, and liquid intake, number of steps, elevation, climbing, running, biking. | Bluetooth | Li-poly | 2 days |
| Fitbit Charge HR™ Fitness Tracker | HR, calories burned, sleep quality, food, and liquid intake, number of steps, elevation, climbing, running. | Bluetooth LE | Li-poly | 5–7 days |
| Garmin vivosmart® HR Fitness Tracker | HR, calories burned, sleep quality, number of steps, climbing, running, swimming. | Bluetooth LE, ANT+ | Li-ion | 5 days |
| Ref. | Proposition | Moni-Tored Signs | Electrode Type | Active Material | Electrode Size | Attachment Method | Wireless Connectivity | Accuracy | Signal Acquisistion Module | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Size | Freq. | A/D | Bat. Life, Power | |||||||||
| [28] | Sensorized T-shirt and textile belt | ECG, HR | Dry textile electrodes | Silver based conductive yarns | Snap buttons | Bluetooth LE | - | 512 Hz | 24 bit | - | ||
| [29] | Wearable mobile electro-cardiogram monitoring system | ECG, HR, location | Dry foam electrode | Ni/Cu coated compressed urethane polymer foam | 14 mm × 8 mm × 8 mm | Bluetooth v2.0, and GSM | 99.51% correlation with prerecorded ECG data, QRS detection accuracy ~98.14% | 4 cm × 2.5 cm × 0.6 cm | 512 Hz | 12 bit | 33 h, 1100 mAh Li-ion battery | |
| [10] | Wireless, portable capacitive ECG sensor | ECG, HR | Capacitive electrode with cotton insulator | 33 mm × 33 mm × 2 mm | Woven under a stretchable belt | ANT | 45 mm × 60 mm × 9 mm | 500 Hz | 10 bit | 15 h with 256 mAh 3 V Li battery | ||
| [30] | Use of flexible capacitive electrodes for reducing MA | ECG, HR | Flexible capacitive electrodes | Ni/Cu coated foam (polyolefincovered by polyurethane) | 300 mm × 20 mm × (1.1 ± 0.2 mm) | Integrated into a chest belt | Bluetooth | Upto 91.32% QRS detection at 7 km/h walking speed | - | 256 Hz | - | - |
| [31] | Common Electrode-FreeECG monitoring System | ECG, HR | Active capacitive electrodes | Copper layer | 5 cm × 3 cm | Adhesive tape | - | - | 2 kHz | 24 bit | - | |
| [32] | HR monitoring from pressure variance in ear canal | HR | Piezo-electric film sensor | 3.5 mm × 3.5 mm | Earpiece like device | 2.4 GHz RF | Sensitivity 97.25%, PPV 97.18%. | 15 mm × 17 mm | 100 Hz | 12 bit | Coin-cell battery | |
| [33] | Heart Rate Monitoring with pressure sensor | HR | Piezo-resistive pressure sensing | C black/silicone rubber nanocomposite encapsulated in conductive FCCL films | 15 mm × 30 mm | Embedded in elastic belt | - | Accuracy > 97% | - | - | - | - |
| Spatial Domain | Temporal Domain | Frequency Domain | Statistical Domain |
|---|---|---|---|
| Step length | Double support time | Spectral power | Correlation |
| Stride length | Stance time | Peak Frequency | Mean |
| Step width | Swing time | Maximum spectral amplitude | Standard deviation |
| RMS acceleration | Step time | Covariance | |
| Walking speed | Stride time | Skewness | |
| Cadence (steps/min) | Kurtosis | ||
| Energy |
| Ref. | Proposition | Feature Extraction | Classification Method | Sensors | Sensor Placement | Com. Tech. | Detection | Accuracy | Power Req. |
|---|---|---|---|---|---|---|---|---|---|
| [44] | Activity and gait recognition system on a smartphone | Fixed set of features | Support Vector Machine (SVM), Bayes network, and Random Tree | Accelerometer is embedded in smartphone | Different walking speed | >99%. | |||
| [45] | In-home, fine-grained activity recognition multimodal wearable sensors | Fixed feature set | Conditional random field (CRF) | Smartphones’ (Samsung Galaxy S4) onboard sensors (accelerometer, gyroscope, barometer, temperature and, humidity sensor), along with Gimbal Bluetooth beacons | Waist, lower back, thigh, and wrist | USB | Walk and run indoors, use refrigerator, clean utensil, cook, sit and eat, use bathroom sink, move from indoor to outdoor, move from outdoor to indoor, walk upstairs, and walk downstairs, stand, lie on the bed, sit on the bed, lie on the floor, sit on the floor, lie on the sofa, sit on the sofa, and sit on the toilet | 19 in-home activities with >80% accuracy | |
| [46] | Wearable device based on a 9-DOF IMU | Fixed set of features | Accelerometer, gyroscope, and magnetometer | Limb or trunk | Bluetooth | Balance hazards, balance monitoring for fall prediction | High correlation | Streaming ~6 h Logging > 16 h | |
| [47] | Algorithm development | Time-Frequncy domain analysis | Hidden Markov Model | 3-axis accelerometer, 3-axis gyroscope | Chest | USB | Walking, running, ascending upstairs, descending downstairs and standing | ~95% | |
| [48] | A real-time, adaptive algorithm for gait-event detection | Two inertial and magnetic sensors ( 1 IMU = 1 accelerometer, 1 gyroscope) | External part of both shanks | Gait events: Initial Contact (IC), End Contact (EC) and Mid-Swing for both right and left leg while walking at three different speed | F1-scores 1(IC, EC), 0.998 (IC) and 0.944 (EC) for stroke subjects | ||||
| [49] | Recognition method for similar gait action | Inter-class relation Ship | Support vector machine, K-nearest neighbor | 3 IMUs (each IMU: 1 tri-axis accelerometer,1 tri-axis gyro) | Fixed at the back, left, and right waist | Walking on flat ground, up/down stairs, and up/down slope | ~93% average | ||
| [50] | Stochastic approximation framework | Fixed set of features | K–means and Gaussian Mixture Models | Accelerometer | Belt-like strap around the waist | 3 intensity level of walking: 93.8%; 3 intensity level of running 95.6% | |||
| [51] | Power-aware feature selection for minimum processing energy | Minimum cost feature selection by using a redundancy graph | K-nearest neighbor | 6 IMUs (each IMU has one three-axis accelerometer and a two-axis gyroscope) | Waist, right wrist, left wrist, right arm, left thigh, right ankle | BSN | Switching between stand and sit, sit and lie, bend to grasp, rising from bending, kneeling right, rising from kneeling, look back and return, turn clockwise, step forward and backward, jumping | 30% energy savings with 96.7% accuracy | |
| [52] | Parameter optimization strategy for phase-dependent locomotion mode recognition | Fixed set of features | 2 IMUs, 2 pressure insoles (each having 4 pressure sensors) | IMUs on the shank and the shoe, pressure sensors insole | Walking, up/down stairs, and up/down slope, passive mode | 88%–98% | |||
| [53] | Electronic insole for wireless monitoring of motor activities and shoe comfort | Fixed set of features | Humidity and temperature sensors, accelerometer and 4 pressure sensors | Insole | ZigBee | Foot accelerations, orientation in space, temperature and moisture data | 10 h of data logging | ||
| [54] | Shoe-based activity monitoringsystem (smartshoe) | Fixed set of features | Support vector machine, multilayer perception (MLP) | Five pressure sensors (PS) and one 3-D accelerometer | PS on insole and accelerometer on heel of shoe | Sit, stand, walk, ascend stairs, descend stairs and cycling | 99.8% ± 0.1% with MLP | ||
| [55] | A wearable device for monitoring daily use of the wrist and fingers | Fixed set of features | K-means | 2 tri-axial magnetometers | Watch-like enclosure worn on the wrist and a small neodymium ring worn on the index finger | Finger and wrist movement | 92%–98% with a 19%–28% STD | 20.5 mA at 3.3 V | |
| [56] | Combined kinematic models to estimate human joint angles | Unscented Kalman filter | 3 IMUs | Upper arm, forearm, and wrist | Shoulder internal/external rotation; flexion/extension of shoulder, elbow, and wrist, supination/pronation of forearm, wrist twist | Average RMS angle error ~3° | |||
| [58] | Wearable device with automatic gait and balance analyzing algorithms for Alzheimer patients (AP) | Fixed set of features | 3 IMUs (each IMU has a 3-d accelerometer, a uni-axial gyroscope, and a biaxial gyroscope | On feet for gait analysis on waist for balance analysis | Gait parameters and balance | 30 mA at 3.7 V | |||
| [59] | IMU based fall Detection system | Madgwick orientation filter | Accelerometer, gyroscope, and magnetometer | Waist | Bluetooth | Backward fall, forward fall, lateral left fall, lateral right fall, syncope | Accuracy: 90.37%–100% Sensitivity: 80.74%–100% | 15 mA–34 mA using 3.7 V | |
| Ref. | Proposed Device | Principle | Measured Parameters | Used Device for Measurement | Location | Wireless Connectivity | Performance Evaluation | Accuracy |
|---|---|---|---|---|---|---|---|---|
| [66] | Kalman filter based body temp. estimation model | Temperature variation with HR | HR, skin temperature | Ag/AgCl gel electrodes | Chest | - | Compared with data from ingestible temperature capsule | RMSE: 0.40 °C |
| [68] | Wireless, dual channel body temp measurement system | Mean of measurements from two ear canals | Core body temp | Digital temp sensor DS18B20 | Ear canal | Bluetooth | ±0.1 °C | |
| [69] | Wearable wireless temperature monitoring | Two-point calibration | Circadian rhythms, Skin temp | MF51E NTC thermistor | Skin | RF (Tyndall node) over Body sensor network (BSN) | Compared with data from a thermometer | 0.02 °C |
| [71] | Embedded NTC temperature sensor and conductive textile wires in a belt made with soft bamboo | ECG, skin temperate | NTC Mon-A-Therm 90045 and Shieldex® Silver Plated Nylon yarn | Skin | - | Compared with data from the NICU sensor connected to the Solar® 8000M patient monitor | ±0.1 °C | |
| [72] | Wireless body temperature monitoring | Skin temperature | LM35 | Hand | ZigBbee and WLAN | ±0.25 °C | ||
| [75] | RFID sensor chip in 0.35-μm CMOS standard process | Temperature dependence of the frequency of ring oscillator | Tag and reader communicate at 868 MHz | Measurement was performed in a climate chamber | ~±0.1°C Resolution: 0.035°C | |||
| [76] | Epidermal-like RFID tag made on a Poli-caprolactone membrane | Re-tunable epidermal tag | Skin temperature | EM4325 | Abdomen | Tag and reader communicate within a band of 780–950 MHz | Compared with data from PT104 thermocouple | ±0.25 °C |
| [77] | Deep body temperature measurement system embedded in a neck pillow | Embedding 1 Dual-heat-flux, 2 double-sensor in neck pillow | Core body temperature | Around neck | - | Compared with data from infrared thermometer (thermoscan IRT 4520) | - | |
| [79] | Heater-less deep body temperature probe | Dual-heat-flux method | Core body temperature | Forehead | - | Compared with data from zero-heat-flow thermometers | Correlation: 97% |
| Ref. | Proposition | Electrode Type/Device | Measurement Location | Wireless Connectivity | Size | Sampling Rate | A/D | Battery Life/Power Req. | Evaluation | Accuracy |
|---|---|---|---|---|---|---|---|---|---|---|
| [85] | A small wristband for unobtrusive and continuous EDA measurements during everyday activities | Ag/AgCl electrodes | Dorsal forearms | 2.4 GHz transceiver module (nRF2401) | 70 mm × 70 mm × 20 mm | 32 Hz | 12 bit | 1199 mAh, 3.7 V LiPo | Measurement compared with commerecial system. | overall correlation: 93%–99% |
| [86] | An ambulatory device for measuring HR, GSR, and skin temperature | Arduino based e-textile lilypad platform (SHT15 for T measurement) | Not implemented | Supply voltage: 2 V to 5 V | ||||||
| [87] | Highly wearable and reliable galvanic skin response (GSR) sensor | flexible dry polymer foam Ni/Cu | Back | Bluetooth | 42.5 mm × 38.5 mm | 10 bit | compared thesignal with a finger reference GSR | average Correlation: 76.8% | ||
| [88] | Wearable multi-sensor device for real-time biofeedback and data acquisition | Ag electrodes | Bluetooth LE | 4 cm × 4 cm | 4 Hz | 38 h of operation | resolution 900 pS between 0.01 µS and 100 µS | |||
| [89] | A pervasive and unobtrusive system for sensing human emotions | Commercial Shimmer GSR sensor | Finger | Bluetooth | 65 mm × 32 mm × 12 mm | 10 Hz | 450 mAh Li-ion battery | Classification of 4 emotions with ~80% of accuracy (amusement, fear, sadness, and relaxation) | ||
| [90] | Distinguishing stress from cognitive load in an office environment by EDA | Dry Ag/AgCl electrodes | left index and middle fingers | Bluetooth | 41 mm × 67 mm | 16 Hz | Power consumption: 182 mW | Investigated 6 classifiers to discriminate cognitive load from stress | Accuracy 82.8% (max), achieved by LDA | |
| [91] | Use of wearable sensors and wireless technology to measure the autonomic function and stress level in the ambulatory setting | Ag/AgCl electrodes in Shimmer Platform | Palm of non-dominant hand | Bluetooth | 30 Hz | GSR preconditioning circuit consumes 60 µA | ||||
| [93] | A wearable device for predicting blood pressure (BP) and cardiovascular dynamics | Ag/AgCl electrodes | Fingers or opposite sides of palm | Bluetooth | 1280 Hz, averaged over 32 samples: results 40 Hz | 10 bit | 10 h with 9 V battery, 220 mA with Bluetooth | correlation with pulse pressure with GSR | R2 value for PP: 0.923, SBP: 0.801 | |
| Ref. | Proposition | Principle | Measured Parameters | Sampling Rate | Size | Power/Current Req. | Wireless Connectivity | Performance Evaluation | |
|---|---|---|---|---|---|---|---|---|---|
| [97] | Ring shaped backside silicon p-n photodiode | Transmittance oximetry | Temperature, Pulse, SpO2 | 8 kHz | Radius = 3.68 mm width = 0.78 mm | <10 mA | Quantum eff. = 62% Reverse current density = 55 nA/cm2 Forward saturation current = 0.14 nA/cm2 | ||
| [98] | Sensors embedded in soft fabrics | Reflectance oximetry | HR, SpO2 | Measurement compared graphically with commercial oximeter measurements | |||||
| [99] | Wireless oximeter | Reflectance oximetry | HR, RR, SpO2, PPT | 240 Hz | 41mm × 36 mm | <150 mA | ZigBee | SNR of IR = 8 SNR of red =3 | |
| [100] | Micro-machined Pt electrodes | Transmittance oximetry | ECG, HR, SpO2 and SBP | 200 Hz | <35 mA | ZigBee | |||
| [102] | Ring probe, novel distribution of optical sensors around the phalanx | Transmittance oximetry | HR, SpO2 | Diameter of the finger | Measurement compared graphically with commercial oximeter measurements | ||||
| [103] | Wrist band Sensor | Reflectance oximetry | HR, SpO2 | CC2500 RF TRX | Ratio of change rates of reflected light intensity in two wavelengths (660 nm and 900 nm) | ||||
| [104] | Ring-type pulse oximeters | Reflectance oximetry | HR, RR, SpO2, PPT | Bluetooth | Correlation between SpO2 values measured by the proposed and commercial oximeter | 98.26% | |||
| [107] | Analog single-chip pulse oximeter | SpO2 | 2.2 mm × 2.2 mm | 4.8 mW | Measurement compared with commercial oximeter measurements | Mean diff. ~−1.2% SD = 1.5% | |||
| [108] | Forehead mounted sensor | Reflectance oximetry | HR, SpO2 | WiFi | Measurement compared with commercial oximeter measurements | ||||
| [109] | Electronic Patch with an optical biomedical sensor | Reflectance oximetry | PPG, HR, RR | 125 Hz | 88 mm × 60 mm (× 5 mm) | I < 33 mA P < 99 mW | PPG is measured using Datex pulse oximeter. SpO2 is calculated and plotted against optical ratio for calibration, MSE ~ 2.6% | ||
| Ref. | Proposition | Electrode Type | Size | Base Material | Conductive Material | Technology | Performance | Contact Resistance |
|---|---|---|---|---|---|---|---|---|
| [131] | Direct attach and Interposer electrode | Active electrode | 20 × 13 mm2 (direct-attach) 11.6 × 11.6 mm2 (Interposer) | Nonwoven Evolon fabrics | Conductive ink (CMI 112-15) | Screen printing, stenciling, curing, and encapsulation | *PSDs for sitting and jogging are close to Ag/AgCl electrodes*Durable upto 5 washing cycles | |
| [136] | Active electrodes on woven textiles | Active electrode | 28 mm × 23 mm (skin contact area) | Woven textile composed of cotton, polyester and Lycra fibers | Silver polymer paste (Fabinks TC-C-4001) | Screen and stencil printing | The printed active and Ag/AgCl electrodes had very similar rms levels after filtering | |
| [138] | 2 textile nanofiber web electrodes | Dry electrode | 9 mm diameter | PVDF Nanofiber Web | Poly (3,4-ethylene-dioxythiophene) (PEDOT) | Electrospinning-vapor phase polymerization | Tested ECG is 95% similar to Ag/AgCl electrodes | ~1000 Ω |
| PVDF Nano fiber Web | Silver | Silver mirror reaction | Tested ECG is ~92% similar to Ag/AgCl electrodes | ~100 Ω | ||||
| [139] | Nano copper loaded poly-propylene based textile electrode | Dry electrode | 4 cm × 6 cm | Polypropylene nonwoven fabric | Copper nanoparticles on fabric | Multiple dip chemical processes | Max conductivity: 142.8 kΩ·m | |
| [143] | 8 types of electro-thread | Dry fabric electrode | 2 × 2 cm2, 2 × 5 cm2 | Polyester 75 denier | Silver thread | Inclusion of one strand or two strands of 50 μm silver thread | 32 kΩ at 120 Hz (for 2 Ag strand based 1300TM polyester fabric) | |
| [141] | Several textile-based electrodes | Dry fabric electrode | 1.5 cm × 3 cm | PU laminated or dry- coated nylon | Copper coating | Sputtering | 5.7 Ω (PU laminated nylon), 10.26 Ω (PU dry-coated nylon). | |
| Ripstop, Mesh fabric | Cu/Ni coating | Electroless Plating | 0.23 Ω/sq (Ripstop), 0.29 Ω/sq (Mesh) | |||||
| 5 cm × 5 cm | Cotton, Steel/cotton | Stainless Steel Filament Yarn | Embroidering or Knitting | R peak detection accuracy: 58.8% and 64.2% | 32.55 Ω/m (linear resistance) | |||
| [142] | Knitted fabric electrodes | Dry electrodes | 20 mm × 20 mm | Wool and polyester | Silver coated nylon, stainless steel yarn, and silver coated copper | Knitting | FFT response of the multifilament electrodes retains ECG spectralcomponents | |
| [144] | Embroidered textile electrode | Wet, moisturized by water vapor using the polyester wetting pad. | 2 cm × 7 cm | Polyethylene terephthalate yarn of 50 μm diameter | Silver and ultra-thin titanium | Coating by plasma sputtering | Similar signal quality and signal strength after 1 h as after 72 h of use |
| Ref. | Proposition | Type | Fabrication Method | Temperature Range | Sensing Material | Sensitivity | Size | Substrate/Embedding Platform | Performance | Nominal Resistance |
|---|---|---|---|---|---|---|---|---|---|---|
| [160] | Polymer sensor | Thermistor | Screen printing | Carbon polymer paste | Polyamide foil Kapton | High flexibility, linear characteristic, high thermal resistance change | ||||
| [166] | Optical fiber Bragg grating based sensor | Optical | Grinding, polishing | 33 °C to 42 °C | Fiber Bragg grating | 0.15 nm/°C | Encapsulated with polymer (copolymerization of unsaturated Methyl Ethyl Ketone Peroxide (MEKP) and cobalt naphthenate) filled strip. | Accuracy ~± 0.18 °C | ||
| [165] | Printed sensors on flexible substrate | RTD | Screen printing | 20 °C to 80 °C | PTC and NTC resistive pastes | 0.025 V/°C at 37 °C | 320 mm × 380 mm | Poly Ethylene Naphtalate (PEN) | ||
| [161] | Inkjet printed flexible sensor | Thermistor | Inkjet printing | 20 °C to 60 °C | Silver | 4.5 Ω/°C at 38.5 °C | 2.85 cm × 2.26 cm | Polyimide substrate (Kapton HN) | Good linearity (coefficient of linearity ~ 0.9998) Hysteresis less than 5% | 2.032 kΩ at 38.5 °C |
| [158] | Arrays of single sensors on a flexible substrate | RTD | Electron beam evaporation followed by photolitho-graphy | 25°C to 90°C | Meander shaped structures of platinum | 1.52 Ω/°C | 67.5 mm × 67.5mm | Kapton E foils, Integrated into textile using weaving | The sensors damage at strong bending of around 11% due to cracking of the sensing lines | |
| [164] | Sensors on paper substrate | RTD | Inkjet printing | −20 °C to 60 °C | Silver nano-particles | 16 mm × 16 mm | Nano-porous oxide film coated paper | Good linearity with a TCR of 0.0011/°C, with perylene coating linearity, is 0.9999, resistivity 30 µΩ·cm | 740 Ω with perylene coating | |
| [159] | Embroidered sensors | RTD | Embroidery | 20 °C to 100 °C | Conductive yarn made of austenitic Cr-Ni stainless steel wires | 2.68 Ω/°C | 90 mm × 90 mm | Embroidered on a textile substrate | Good resistance against washing cycles | |
| [162] | Printed wearable sensor | RTD | Shadow mask printing | 22 °C to 50 °C | Mixer of carbon nanotube and PEDOT:PSS | 0.6 %/°C | SiO2-coated Kapton | Good stability, highly sensitive | ||
| [163] | Ultrasensitive wearable sensor | RTD | PECVD and polymer-assisted transfer method | 35°C to 45°C | Grapheme nanowalls | 20 mm × 10 mm | Polydimethylsiloxane (PDMS) | TCR = 0.214/°C, response time 1.6 s and recovery time 8.52 s | 706.2 Ω at 25 °C | |
| [153] | Flexible wireless sensors | RTD with integrated passive RFID antenna | 35 °C to 42 °C | Ni microparticle- filled binary polymer (polyethylene (PE) and polyethylene oxide (PEO)) composites | 0.1 to 0.3 V/°C | Accuracy ~± 2.7 °C | ||||
| [157] | Temperature sensing fabric | RTD | Metal wire inlaid in the middle of a rib knitted structure | 20 °C to 50 °C | Platinum wire, Diameter < 25 mm | 8 cm × 8 cm | Polyester fabric | Coefficient linearity in the range of 0.99–0.999 | 3 Ω to 130 Ω | |
| Ref. | Proposition | Sensing Mechanism | Structure/Base | Sensing Material | Gauge Factor | Stable Strain Range | Demonstrated/Potential Applications |
|---|---|---|---|---|---|---|---|
| [171] | Textile-structured flexible strain sensor | Contact resistance of fiber/yarn/fabric | Single warp fabric | Carbon fiber | 10–200 depending on fiber length | Max 200% | Wearable strain sensor |
| [172] | Textile-based strain sensor | Contact resistance of conductive fiber loops | Fabric with elastomeric yarns | Silver coated polymeric yarn made loops | 0.75 | 40% | Wearable strain sensor |
| [168] | Stretchable and Sensitive Strain Sensor | Piezoresistive | PDMS | Ag nano-walls thin film | 2 to 14 | 70% | Finger movements |
| [170] | Textile-based strain sensor for monitoring the elbow and knee movements | Piezoresistive | Elastic yarns made from Lycra fiber wrapped with two polyester yarns. | Carbon particles coated polyamide fiber twisted with polyester yarn | ~0.3 | 30% | Flexion angle of elbow and knee movements |
| [169] | Stretchable strain sensor based on a metal nanoparticle thin film for human motion detection | Piezoresistive | PDMS | Silver nanoparticle | 2.5 | 20% | Finger movements |
| [175] | Knee’s kinematic monitoring using single optical FBG sensor | Fiber Bragg grating | Optical Fiber | Polymer encapsulated FBG sensor | ~0.8 | 0.04% | Knee, finger movements, HR, RR |
| [176] | Force sensors based on light pipes in the form of multimode optical fibers made of copolymers. | Loss of light due to deflection of the fiber with force | Multimodal optical fiber | Copolymers containing silicon and polyurethane | Force sensing | ||
| [167] | Textile-based MEMS accelerometer | Piezoresistive | Cotton fiber | Silver nanoparticles | 7.796 ± 2.835 | Motion sensing | |
| [174] | All-polymeric knitted textile strain sensor | Piezoresistive | Commercial Spandex yarn | PU/PEDOT:PSS fibers | 0.2 to 1 | 160% | Knee bending movements |
| Wireless Technology | Frequency Band | Range | Data Rate | Power Consumption | Maximum Number of Nodes Supported | Supported Network Topologies | Security | Modulation | Reference |
|---|---|---|---|---|---|---|---|---|---|
| RFID | 13.56 MHz 860–960 MHz | 0-3 m | 640 kbps | 200 mW | 1 at a time | P2P (passive) | N/A | ASK, PSK, FSK | [73,74,196] |
| Bluetooth | 2.4–2.5 GHz | 1–100 m | 1–3 Mbps | 2.5–100 mW | 1 master + 7 slave | P2P, star | 56–128 bit key | GFSK | [29,30,45,59,66,87,90,91,92,93,104] |
| BLE | 2.4–2.5 GHz | 1–100 m | 1 Mbps | 10 mW | 1 master + 7 slave | P2P, star | 128-bit AES | GFSK | [28,88] |
| ZigBee | 2.4–2.5 GHz | 10–100 m | 250 kbps | 35 mW | 65,533 | P2P, star, tree and mesh | 128-bit AES | OQPSK, BPSK | [70,99,100] |
| WiFi | 2.4–2.5 GHz | 150–200 m | 54 Mbps | 1 W | 255 | P2P, star | WEP, WPA, WPA2 | BPSK, QPSK, QAM | [108] |
| UWB | 3.1–10.6 GHz | 3–10 m | 53–480 Mbps | 250 mW | 1 master + 7 slave | P2P, star | BPPM, FSK | ||
| ANT | 2.4–2.5 GHz | 30 m | 20–60 kbps | 0.01–1 mW | 65,533 in one channel | P2P, star, tree and mesh | 64-bit key | GFSK | [10,19,188] |
| MICS | 402–405 MHz | 2 m | 200–800 kbps | 25 µW | P2P, star | FSK | |||
| IrDA | 38 kHz | 10 cm | 1 Gbps | 1 at a time | P2P | ||||
| NFC | 13.56 MHz | 5 cm | 424 kbps | 15 mW | 1 at a time | P2P | AES | ASK |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majumder, S.; Mondal, T.; Deen, M.J. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. https://doi.org/10.3390/s17010130
Majumder S, Mondal T, Deen MJ. Wearable Sensors for Remote Health Monitoring. Sensors. 2017; 17(1):130. https://doi.org/10.3390/s17010130
Chicago/Turabian StyleMajumder, Sumit, Tapas Mondal, and M. Jamal Deen. 2017. "Wearable Sensors for Remote Health Monitoring" Sensors 17, no. 1: 130. https://doi.org/10.3390/s17010130
APA StyleMajumder, S., Mondal, T., & Deen, M. J. (2017). Wearable Sensors for Remote Health Monitoring. Sensors, 17(1), 130. https://doi.org/10.3390/s17010130







