Recent Progresses in Nanobiosensing for Food Safety Analysis
Abstract
:1. Introduction
2. Different Functional Roles of Nanomaterials in Food Safety Analysis
Functionalization of Nanomaterials
3. Recent Development in Nanobiosensing for Food Safety Analysis
3.1. Pathogens
- (1)
- Recognized by complementary DNA (cDNA). One of the detection routes for microbial pathogens involves analyzing its genomic DNA (gDNA) [10,59,60,61,62] which can be specifically recognized by its cDNA. Since only a trace amount of target DNA is present in microbial pathogens, nanomaterials and amplification techniques (such as polymerase chain reaction (PCR, a non-isothermal and enzymatic process based on using DNA polymerase to synthesize new strands complementary to the offered template strand), rolling circle amplification (RCA, an isothermal and enzymatic process in which long single-stranded DNAs (ssDNA) are synthesized on a short circular ssDNA template by using a single DNA primer), DNAzyme) are concurrently recruited to amplify target DNA or signal. Recently, a metallic nanowire based electrical Escherichia coli (E. coli) genomic DNA detection method has been developed using RCA to generate long ssDNA with abundant repetitive sequences [59]. DNA modified AuNPs of 10 nm diameter is aligned along long ssDNA via DNA hybridization, followed by enhancing conductivity of AuNPs string using silver or gold solutions to form wide silver or gold nanowires, resulting a high signal-to-noise ratio and low limit of detection (LOD) towards E. coli gDNzA. In addition, GOx-HRP mimicking DNAzyme nanocomposites, AuNPs-magnetic Fe3O4 NPs, and DNA functionalized AuNPs-asymmetric PCR system have been employed for the detection of gDNA of microbial pathogens [10,60,61]. However, this strategy is hampered by cumbersome pretreatment of pathogen and extraction of gDNA.
- (2)
- Recognized by antibody. Antibodies with affinity towards the pathogens (immunologic approach) is a more convenient approach than analysis of gDNA [63,64,65,66,67]. A novel, sensitive, amplified detection of E. coli O157:H7 in food at real-time has been developed based on Pt–Au bimetal NPs with peroxidase activity using immunochromatographic assay (ICA) [27]; E. coli O157:H7 is one of the most notorious pathogens with low infectious dose commonly found in beef, raw milk, and vegetables. Indirect immunofluorescence assay, designed using FITC (fluorescein isothiocyanate)-doped silica NPs synthesized by W/O microemulsion method, demonstrated rapid detection of E. coli O157:H7 in beef [53]. In addition, polydiacetylene liposomes incorporated with antibody can be used for specific detection of Salmonella; the using of small liposomes can help in enhancing sensitivity [68]. Portable and automated paper-based detection methods are being rapidly developed in recently [69]. Merkoçi and co-workers have invented a lateral flow immunoassay for highly sensitive paper-based E. coli detection [70]. This design includes CdSe@ZnS QDs decorated with antibody (Ab-QDs) and GOx as photoluminescent probes and revealing-agent. The proposed device demonstrates highly specific and sensitive performance, detecting pathogen 10 CFU·mL−1 in standard buffer and 100 CFU·mL−1 in bottled water and milk. The similar portable and paper-based principle has been adopted using Pt–Au bimetal NPs and TMB as catalyst and colorimetric substrate, respectively [27], therefore, the pathogen detection can directly be observed by naked eyes. This proposed device exhibits a lower LOD of 100 cells/mL, which is 1000-fold lower than the AuNPs-based colorimetric method.
- (3)
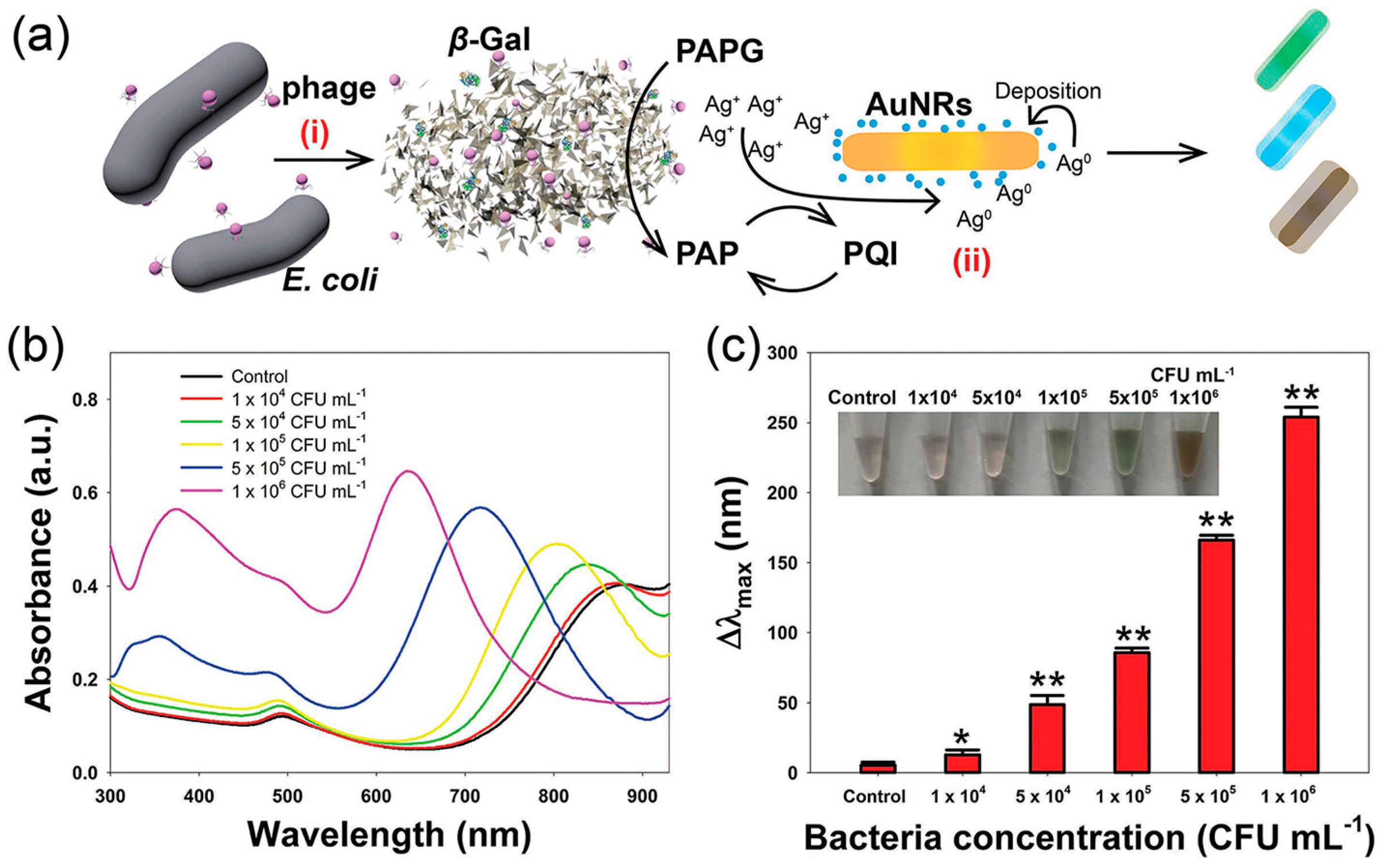
- Recognized by aptamer. Using antibodies as a part of a sensing system has some serious drawbacks such as rigorous production and purification processes and limited applicability (not work in harsh conditions, e.g., high temperature) [71]. These weaknesses can be neglected when using aptamer as recognition element. Many aptasensings based on nanomaterials (MNPs, silver NPs, nanorods, carbon quantum dots, and so on) have been designed for the quantification of microbial pathogen in various real samples [23,72,73,74,75]. Employing aptamer-conjugated fluorescent NPs and multicolor upconversion NPs as reporters, the LODs for Staphylococcus aureus, Vibrio parahemolyticus, and Salmonella typhimurium can lower to 25, 10, and 15 CFU·mL−1, respectively [76,77]. Alternatively, monitoring and measuring beta-galactosidase (β-gal) activity is another approach to detect E. coli. In the presence of β-gal released from E. coli, the substrate p-aminophenyl β-d-galactopyranoside is hydrolyzed to produce p-aminophenol. Reduction of Ag+ by p-aminophenol generates a silver shell on the surface of gold nanorods (AuNRs), resulting in the blue shift of the longitudinal localized surface plasmon resonance peak and multicolor change of the solution from light green to orange-red (Figure 1) [78].
3.2. Toxins
- (1)
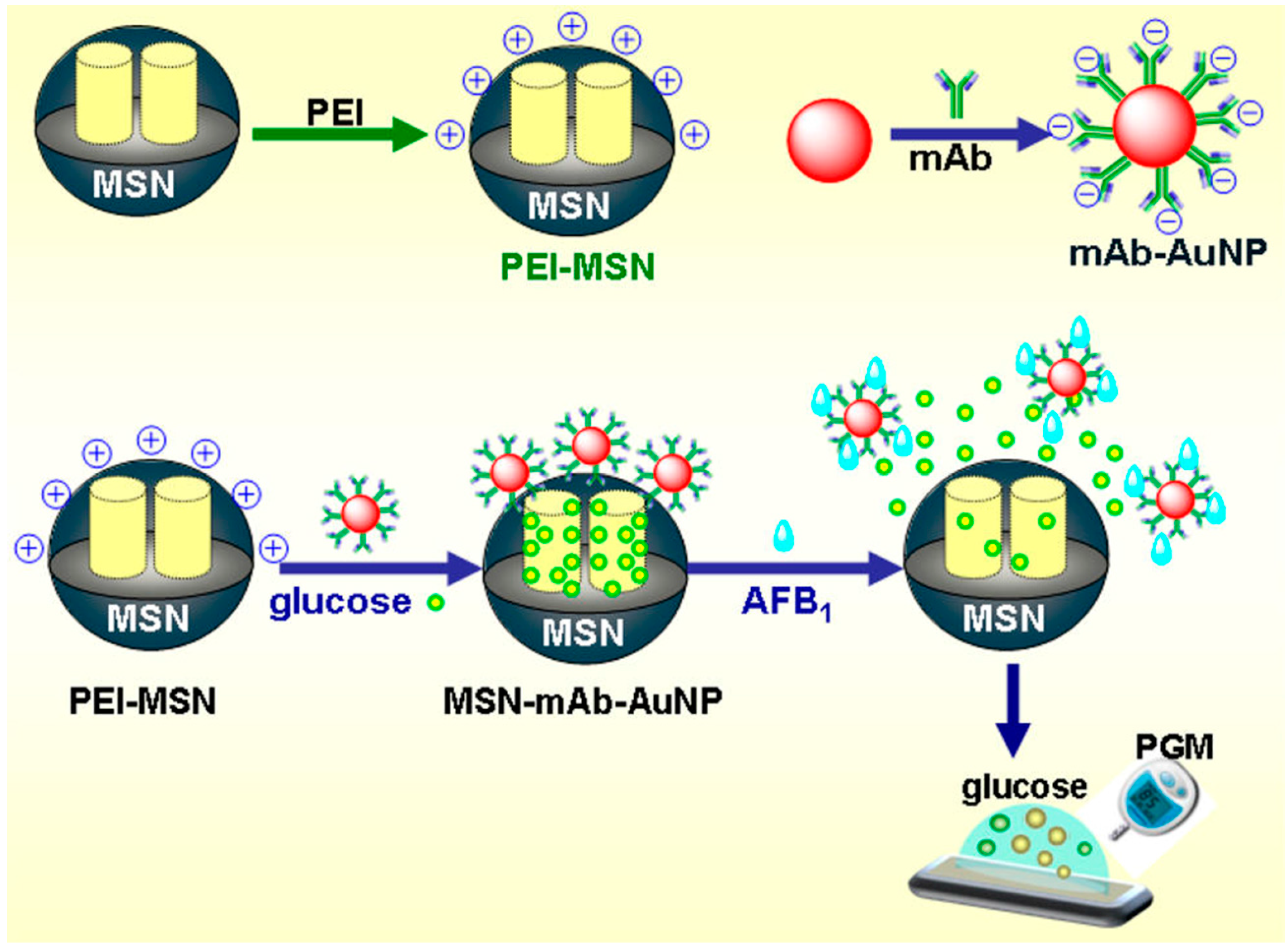
- Recognized by antibody. The majority of nanobiosensing techniques have been developed based on immunoassay. Tang et al. have developed an antibody-functionalized mesoporous carbon (MSC) NPs-based competitive-type biosensor for the detection of AFB1 (aflatoxin B1, classified as the first class carcinogen by WHO) [82] in peanuts. Recognition of AFB1 by antibody on MSC results in a departure of thionine—MSC from the electrode accompanying a decrease of current signal. Another competitive immunosensing strategy for the detection of AFB1 in peanut using mesoporous silica nanomaterial loaded with glucose and AuNPs as a lock (Figure 2) [8]. Interestingly, this low-cost, sensitive immunosensing platform can also be used with a portable personal glucometer (PGM) as the readout device [83]. The immune displacement reaction can open the lock and release glucose from the mesoporous silica to the solution, which can then be assayed by PGM. Other NPs, such as QDs, MNPs, and GOx, have also been used to develop nanobiosensors to detect toxins, including ochratoxins, aflatoxins, and deoxynivalenol (DON) in crops [52,84,85].
- (2)
- Recognized by aptamer. Another significant mechanism is the interaction of a toxin with its aptamer. Ochratoxin A (OTA) was the first mycotoxin targeted by aptamer-based assay in 2008. Since then, several nanomaterials and aptamer-based methods have been developed. Recently, a novel strategy based on fluorescent nitrogen-doped carbon dots (N,C-dots) on AuNPs have been proposed for the detection of AFB1 in peanut and corn samples [86]. The chemically-inert N,C-dots provides excellent resistance to photobleaching. This N,C-dots/AuNPs-based aptasensor shows high selectivity against other normally-coexisted mycotoxins, such as OTA, DON, fumonisin B1, and zearalenone. Various metal compound nanomaterials, involving iridium oxide NPs [87], AuNPs doped Fe3O4 NPs [28], CdTe QDs-GOx [47], nanoceria tagged GOx [88], silver nanoclusters (AgNCs) [89] and have also been used to assay toxins. Nonetheless, the association constants of small molecules with their aptamers are low in general; therefore, to obtain a lower LOD, various amplification methods have been employed. Wei et al. have used GOx and DNase I to achieve target recycling, resulting in high sensitivity in OTA detection with a LOD of 20 nM in real red wine samples [90]. Combining unique properties of QDs and MNPs with high efficiency of RCA amplification, an optimized detection for OTA can attain an ultra-low LOD of 0.13 ppt, a 10,000-fold improvement compared with the traditional methods [45].
- (3)
- Others. In addition to being recognized by antibodies and aptamers, many other nanomaterial-based mechanism were reported. (a) Nano-extraction with mass spectrometry (MS) [91]. Utilizing magnetic separation properties of MNPs, a magnetic solid phase extraction of aflatoxins from liquid samples has been developed using polydopamine-coated MNPs as the adsorbent. Coupled with HPLC-MS/MS quantification, LOD of 0.0012 ng/mL for AFB1, AFB2, and AFG1, and 0.0031 ng/mL for AFG2 can be achieved [92]; (b) NPs based molecular imprinting. An electrochemiluminescence sensor, based on Ru(bpy)32+-doped silica NPs combined with molecularly imprinted polymer, has exhibited efficient detection of OTA in corn with a LOD of 0.027 pg/mL [93].
3.3. Pesticides
- (1)
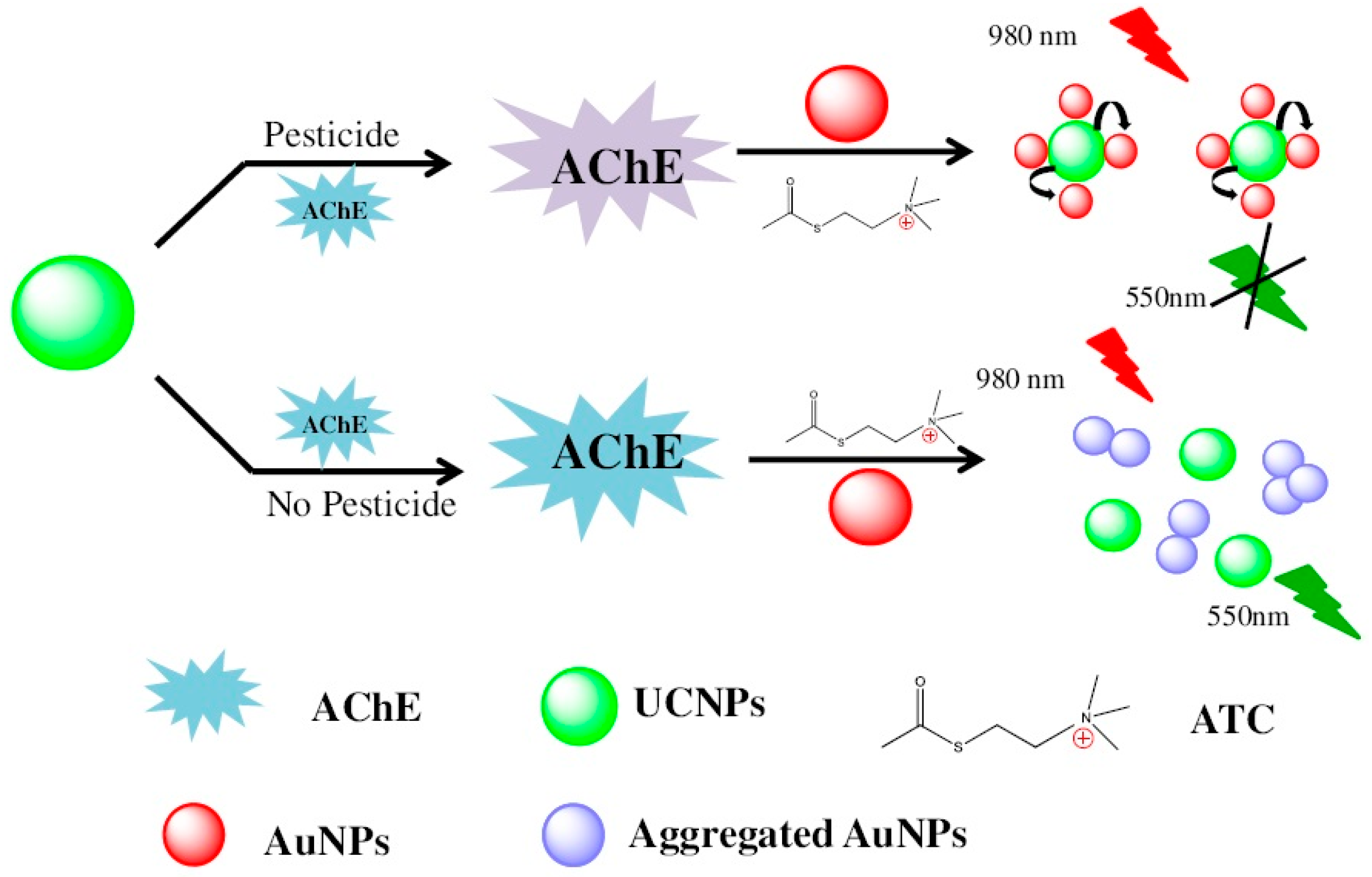
- Enzyme inhibition by pesticide is the most mature and widely used technology for the rapid detection of pesticide residues. Organophosphorus compounds and carbamates can specifically inhibit the activity of acetylcholine esterase (AChE). Zhang and coworkers developed a novel nanobiosensing for organophosphorus pesticides. Thiocholine generation by AChE catalysis leads to the aggregation of AuNPs, resulting in the recovery of fluorescence resonance energy transfer (FRET) between AuNPs and NaYF4:Yb, upconversion NPs (Figure 3) [38]. However, AChE is unstable in solution. Immobilization of AChE in fenugreek hydrogel-agarose matrix with AuNPs results in high enzyme retention efficiency of 92% and a significantly prolonged half-life of the AChE (55 days) [94]. Apart from AChE, pesticides can also inhibit other enzyme activity such as trypsin and tyrosinase [95,96]. Trypsin easily hydrolyzes protamine covered on the surface of AuNPs, leading to fluorescence quenching of QDs. Conversely, the fluorescence could be recovered by adding methyl parathion as it inhibits trypsin activity [96].
- (2)
- Organophosphorus hydrolase-based strategies involve direct detection mechanism than enzymes inhibition strategies. Organophosphorus hydrolase is a homodimeric enzyme that catalyzes the hydrolysis of organophosphorus pesticides. As uniform porous channels, large surface area and well-defined pore topology, ordered mesoporous carbons was used to immobilize cell surface-displayed organophosphorus hydrolase on electrode for direct determination of organophosphates such as paraoxon, parathion, and methyl parathion [97]. Similar direct detection method has also been developed using single-walled CNTs as carrier to support recognition material [7].
- (3)
- Electrochemical and photochemical properties of pesticides themselves are commonly used to develop nanobiosensing. For example, omethoate, malathion, lindane, carbofuran, and carbaryl, etc. possess electrochemical properties. Therefore, nanobiosensors based on electrochemical analysis would be suitable for detecting those pesticides. Many such nanobiosensors, based on copper oxide nanowires-CNTs, AgNPs decorated polyaniline-nanocrystalline zeolite organic-inorganic hybrid material, cobalt oxide (CoO)-reduced GOx, zirconia-ordered macroporous polyaniline, and other nanosystems, have already been reported to improve the sensitivity [98,99,100,101,102]. In addition to electrochemical methods, a few NPs-enhanced SERS methods have been developed; however, low affinity limits the application of such methods. Such problems can be overcome by optimizing metal NPs, for example, the type, molecular linker, surface coverage, and laser excitation wavelength of NPs [103]. It is worth mentioning that, inspired by conductive ink pens for electronic devices on paper, Polavarapu et al. have developed a “pen-on-paper” approach for making SERS substrates [104]. The design involves employing an ordinary fountain pen filled with plasmonic inks comprising metal NPs with arbitrary size and shape; hence, no professional training is needed to manufacture SERS arrays on paper. This simple design lowers LOD of thiabendazole to 20 ppb. In spite of such progress in research, there is a limited translation of technology from laboratory to real life because of economic viability and operational simplicity.
- (4)
- Recognized by antibody. In addition, immunoassay based nanobiosensing are most common for detecting pesticides in food [105,106,107]. The application of nanometal organic framework and other materials can greatly reduce the LOD [55]. As pesticides are known to impede certain photophysical as well as photochemical functions of nanomaterial, through specific recognition of pesticides by antibodies decorated on nanomaterial, several excellent phenomena have been discovered: pentachlorophenol obstructs electrochemiluminescence of Au nanoclusters/graphene hybrid [108], acetamiprid decreases enhanced photocurrent produced by electron donor of quercetin in Co-doped ZnO diluted magnetic semiconductor, thiram quenches blue luminescence of Cu2+ decorated NaYF4:Yb/Tm upconversion NPs fixed on filter paper (monitored by the smartphone camera through a self-written Android program) [109].
3.4. Antibiotics
- (1)
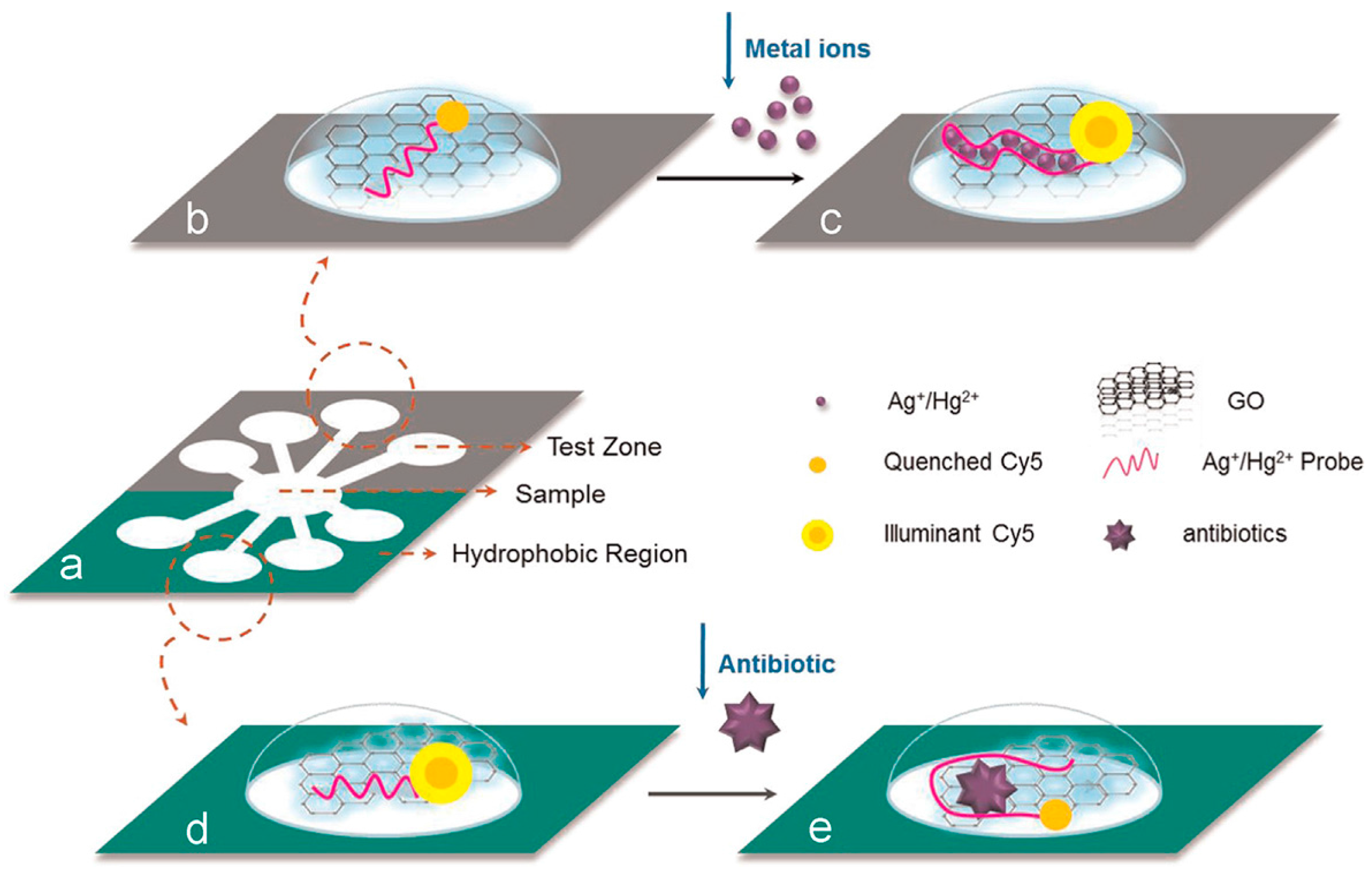
- Recognized by aptamer. Aptamer-based nanobiosensing methods are the most common used for the detection of antibiotics. The upconversion NPs (anti-Stokes)-based aptasensor has shown good specificity towards kanamycin without being disturbed by other antibiotics [110]. Nanomaterials, such as GOx and AuNPs, are used as quenchers in assays based on aptamers of targets and fluorescence-labeled single-stranded DNA to detect antibiotics [111,112]. Simultaneous detection of multiple chemical contaminants in a food sample is a challenging task since each one functions in different microenvironment. Using GOx as quencher, Zuo et al. developed a low-cost paper based microfluidic device for detecting multiple chemical contaminants (antibiotics and heavy metal ions) simultaneously in food samples (Figure 4) [111]. Interestingly, other functions of antibiotics, for example, protecting nature (protecting AgNPs against salt-induced aggregation [113]) of kanamycin, can also be utilized to develop new biosensing methods.
- (2)
- Recognized by antibody. Alternatively, immunization is another strategy to detect antibiotics, though it is not popular than the aptamer method. Metallic nanomaterials (gold nanoflower, AuNPs)-based electrochemical immunosensing methods have frequently been employed to assess chloramphenicol, ofloxacin, and tetracycline in multifarious foods, including milk, honey, and other samples [48,50]. In addition to electrochemistry, a competitive chemiluminescent immunoassay based on new luminol functionalized silver NPs was reported to determine chloramphenicol in milk and honey [114].
- (3)
- Recognized by liposome. Liposomes were often used in molecular biology and pharmaceutics, but rarely used in other fields. Phospholipid liposomes containing R6G dyes on their surface have been utilized to develop a self-signaling sensing platform to detect neomycin—selective recognition of the target by phospholipid displaces R6G dyes from the surface and turns on fluorescence [115].
3.5. Metal Contaminants
3.6. Other Analytes
4. Conclusions and Future Perspectives
Acknowledgments
Conflicts of Interest
References
- Warriner, K.; Reddy, S.M.; Namvar, A.; Neethirajan, S. Developments in nanoparticles for use in biosensors to assess food safety and quality. Trends Food Sci. Tech. 2014, 40, 183–199. [Google Scholar] [CrossRef]
- Sharma, R.; Ragavan, K.V.; Thakur, M.S.; Raghavarao, K. Recent advances in nanoparticle based aptasensors for food contaminants. Biosens. Bioelectron. 2015, 74, 612–627. [Google Scholar] [CrossRef] [PubMed]
- Bulbul, G.; Hayat, A.; Andreescu, S. Portable nanoparticle-based sensors for food safety assessment. Sensors 2015, 15, 30736–30758. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Ratinac, K.R.; Ringer, S.P.; Thordarson, P.; Gooding, J.J.; Braet, F. Carbon nanomaterials in biosensors: Should you use nanotubes or graphene? Angew. Chem. Int. Ed. 2010, 49, 2114–2138. [Google Scholar] [CrossRef] [PubMed]
- Verma, M.S.; Rogowski, J.L.; Jones, L.; Gu, F.X. Colorimetric biosensing of pathogens using gold nanoparticles. Biotechnol. Adv. 2015, 33, 666–680. [Google Scholar] [CrossRef] [PubMed]
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.N.; Rotello, V.M. Gold nanoparticles in chemical and biological sensing. Chem. Rev. 2012, 112, 2739–2779. [Google Scholar] [CrossRef] [PubMed]
- Intae, K.; Geon Hwee, K.; Chang Sup, K.; Hyung Joon, C.; Geunbae, L. Optical detection of paraoxon using single-walled carbon nanotube films with attached organophosphorus hydrolase-expressed Escherichia coli. Sensors 2015, 15, 12513–12525. [Google Scholar]
- Tang, D.P.; Lin, Y.X.; Zhou, Q.; Lin, Y.P.; Li, P.W.; Niessner, R.; Knopp, D. Low-cost and highly sensitive lmmunosensing platform for aflatoxins using one-step competitive displacement reaction mode and portable glucometer-based detection. Anal. Chem. 2014, 86, 11451–11458. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Xiao, Y.; Yin, Y.Q.; Hu, W.L.; Liu, W.; Yang, H.H. Facile synthesis of enzyme-inorganic hybrid nanoflowers and its application as a colorimetric platform for visual detection of hydrogen peroxide and phenol. ACS Appl. Mater. Interfaces 2014, 6, 10775–10782. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Deng, J.; Fang, L.; Yu, K.; Huang, H.; Jiang, L.; Liang, W.; Zheng, J. A novel electrochemical DNA biosensor based on HRP-mimicking hemin/G-quadruplex wrapped GOx nanocomposites as tag for detection of Escherichia coli O157:H7. Biosens. Bioelectron. 2015, 63, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, G.M.; Tang, L.; Chen, J.; Zhu, Y.; He, X.X.; He, Y. Electrochemical sensor based on electrodeposited graphene-Au modified electrode and nanoau carrier amplified signal strategy for attomolar mercury detection. Anal. Chem. 2015, 87, 989–996. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.N.; Yan, B. Highly sensitive and selective fluorescent probe for Ag+ based on a Eu3+ post-functionalized metal-organic framework in aqueous media. J. Mater. Chem. A 2014, 2, 18018–18025. [Google Scholar] [CrossRef]
- Kalita, P.; Dasgupta, A.; Sritharan, V.; Gupta, S. Nanoparticle-drug bioconjugate as dual functional affinity ligand for rapid point-of-care detection of endotoxin in water and serum. Anal. Chem. 2015, 87, 11007–11012. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yu, J.; Gui, R.; Jin, H.; Xia, Y. Carbon nanomaterials-based electrochemical aptasensors. Biosens. Bioelectron. 2016, 79, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Yin, Y.; Yu, H.; Guo, W.; Pei, M. A novel signal amplification strategy of an electrochemical aptasensor for kanamycin, based on thionine functionalized graphene and hierarchical nanoporous PtCu. Biosens. Bioelectron. 2016, 77, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.J.; Liu, Y.J.; Liu, Y.M.; Wang, L.L. Molybdenum disulfide nanoflower-chitosan-Au nanoparticles composites based electrochemical sensing platform for bisphenol A determination. J. Hazard. Mater. 2014, 276, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Masdor, N.A.; Altintas, Z.; Tothill, I.E. Sensitive detection of Campylobacter jejuni using nanoparticles enhanced QCM sensor. Biosens. Bioelectron. 2016, 78, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wu, T.-H.; Zhang, Y. Novel silver nanoparticle-enhanced fluorometric determination of trace tetracyclines in aqueous solutions. Talanta 2016, 146, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Abadian, P.N.; Kelley, C.P.; Goluch, E.D. Cellular analysis and detection using surface plasmon resonance techniques. Anal. Chem. 2014, 86, 2799–2812. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Zhang, L.; Jin, H.; Wang, X.L.; Zhou, J. In situ controlled sputtering deposition of gold nanoparticles on MnO2 nanorods as surface-enhanced Raman scattering substrates for molecular detection. Dalton Trans. 2015, 44, 7606–7612. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Wang, Z.Y.; Zong, S.F.; Chen, H.; Zhu, D.; Wu, L.; Hu, G.H.; Cui, Y.P. SERS detection and removal of mercury(II)/silver(I) using oligonucleotide-functionalized core/shell magnetic silica Sphere@Au nanoparticles. ACS Appl. Mater. Interfaces 2014, 6, 7371–7379. [Google Scholar] [CrossRef] [PubMed]
- Marks, H.L.; Pishko, M.V.; Jackson, G.W.; Cote, G.L. Rational design of a bisphenol A aptamer selective surface-enhanced Raman scattering nanoprobe. Anal. Chem. 2014, 86, 11614–11619. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Zhou, Y.F.; Jiang, X.X.; Sun, B.; Zhu, Y.; Wang, H.; Su, Y.Y.; He, Y. Simultaneous capture, detection, and inactivation of bacteria as enabled by a surface-enhanced Raman scattering multifunctional chip. Angew. Chem. Int. Ed. 2015, 54, 5132–5136. [Google Scholar] [CrossRef] [PubMed]
- Lien, C.W.; Tseng, Y.T.; Huang, C.C.; Chang, H.T. Logic control of enzyme-like gold nanoparticles for selective detection of lead and mercury ions. Anal. Chem. 2014, 86, 2065–2072. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.W.; He, S.B.; Peng, H.P.; Deng, H.H.; Liu, A.L.; Lin, X.H.; Xia, X.H.; Chen, W. Citrate-capped platinum nanoparticle as a smart probe for ultrasensitive mercury sensing. Anal. Chem. 2014, 86, 10955–10960. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Chen, C.X.; Lu, L.X.; Yang, F.; Yang, X.R. A label-free colorimetric sensor for sulfate based on the inhibition of peroxidase-like activity of cysteamine-modified gold nanoparticles. Sens. Actuators B Chem. 2015, 215, 437–444. [Google Scholar] [CrossRef]
- Jiang, T.; Song, Y.; Wei, T.X.; Li, H.; Du, D.; Zhu, M.J.; Lin, Y.H. Sensitive detection of Escherichia coli O157:H7 using Pt-Au bimetal nanoparticles with peroxidase-like amplification. Biosens. Bioelectron. 2016, 77, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Q.; Qian, J.; Wang, K.; Yang, X.W.; Liu, Q.; Hao, N.; Wang, C.K.; Dong, X.Y.; Huang, X.Y. Colorimetric aptasensing of ochratoxin A using Au@Fe3O4 nanoparticles as signal indicator and magnetic separator. Biosens. Bioelectron. 2016, 77, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Abbaspour, A.; Norouz-Sarvestani, F.; Noon, A.; Soltani, N. Aptamer-conjugated silver nanoparticles for electrochemical dual-aptamer-based sandwich detection of Staphylococcus aureus. Biosens. Bioelectron. 2015, 68, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Li, W.J.; Jiang, T.J.; Liu, Z.G.; Chen, X.; Cong, H.P.; Liu, J.H.; Huang, Y.Y.; Li, L.N.; Huang, X.J. Iron oxide with different crystal phases (alpha- and gamma-Fe2O3) in electroanalysis and uftrasensitive and selective detection of lead(II): An advancing approach using XPS and EXAFS. Anal. Chem. 2016, 88, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Li, S.S.; Guo, Z.; Chen, X.; Liu, J.H.; Huang, X.J. Adsorbent assisted in situ electrocatalysis: An ultra-sensitive detection of As(III) in water at Fe3O4 nanosphere densely decorated with Au nanoparticles. Anal. Chem. 2016, 88, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Vijian, D.; Chinni, S.V.; Yin, L.S.; Lertanantawong, B.; Surareungchai, W. Non-protein coding RNA-based genosensor with quantum dots as electrochemical labels for attomolar detection of multiple pathogens. Biosens. Bioelectron. 2016, 77, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yuan, Z.; Liu, X.P.; Liu, Q.; Mao, C.J.; Niu, H.L.; Jin, B.K.; Zhang, S.Y. Electrochemical biosensor for Ni2+ detection based on a DNAzyme-CdSe nanocomposite. Biosens. Bioelectron. 2016, 77, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Li, R.P.; Xu, P.P.; Fan, J.; Di, J.W.; Tu, Y.F.; Yan, J.L. Sensitive iodate sensor based on fluorescence quenching of gold nanocluster. Anal. Chim. Acta 2014, 827, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Dai, H.C.; Shi, Y.; Wang, Y.L.; Sun, Y.J.; Hu, J.T.; Ni, P.J.; Li, Z. Label-free turn-on fluorescent detection of melamine based on the anti-quenching ability of Hg2+ to gold nanoclusters. Biosens. Bioelectron. 2014, 53, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Brennan, M.B.; Wilton, R.; Rowland, C.E.; Rozhkova, E.A.; Forrester, S.; Hannah, D.C.; Carlson, J.; Shevchenko, E.V.; Schabacker, D.S.; et al. Fast, ratiometric FRET from quantum dot conjugated stabilized single chain variable fragments for quantitative botulinum neurotoxin sensing. Nano Lett. 2015, 15, 7161–7167. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.Y.; Shen, W.; Gao, Z.Q. Carbon quantum dots and their applications. Chem. Soc. Rev. 2015, 44, 362–381. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.; Li, H.T.; Zhang, Y.Y.; Yao, S.Z. Upconversion nanoparticle-based fluorescence resonance energy transfer assay for organophosphorus pesticides. Biosens. Bioelectron. 2015, 68, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Si, Y.; Grazon, C.; Clavier, G.; Rieger, J.; Audibert, J.F.; Sclavi, B.; Meallet-Renault, R. Rapid and accurate detection of Escherichia coli growth by fluorescent pH-sensitive organic nanoparticles for high-throughput screening applications. Biosens. Bioelectron. 2016, 75, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.L.; Yin, H.Z.; Wei, R.R.; Wang, W.W. Facile colorimetric detection of Hg2+ based on anti-aggregation of silver nanoparticles. Biosens. Bioelectron. 2014, 57, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.H.; Wang, Y.; Chen, P.; Wang, Y.S.; Mang, J.L.; Aili, D.; Liedberg, B. Biofunctionalized gold nanoparticles for colorimetric sensing of botulinum neurotoxin A light chain. Anal. Chem. 2014, 86, 2345–2352. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.C.; Ho, J.A.A. Gold nanocluster-assisted fluorescent detection for hydrogen peroxide and cholesterol based on the inner filter effect of gold nanoparticles. Anal. Chem. 2015, 87, 10362–10367. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.-L.; Wang, Z.; Zhao, S.-N.; Meng, X.; Song, X.-Z.; Feng, J.; Song, S.-Y.; Zhang, H.-J. A Metal-Organic Framework/DNA Hybrid System as a Novel Fluorescent Biosensor For Mercury(II) Ion Detection. Chem. Eur. J. 2016, 22, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Dulkeith, E.; Ringler, M.; Klar, T.A.; Feldmann, J.; Muñoz Javier, A.; Parak, W.J. Gold nanoparticles quench fluorescence by phase induced radiative rate suppression. Nano Lett. 2005, 5, 585–589. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Chen, Y.J.; Teng, J.; Zheng, W.L.; Wu, J.J.; Adeloju, S.B.; Pan, D.D.; Chen, W. Integrated platform with magnetic purification and rolling circular amplification for sensitive fluorescent detection of ochratoxin A. Biosens. Bioelectron. 2015, 74, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Ranjbari, E.; Hadjmohammadi, M.R.; Kiekens, F.; de Wael, K. Mixed hemi/Ad-micelle sodium dodecyl sulfate-coated magnetic iron oxide nanoparticles for the efficient removal and trace determination of rhodamine-B and rhodamine-6G. Anal. Chem. 2015, 87, 7894–7901. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.S.; Chen, X.J.; Wang, Y.; Zheng, X.T.; Li, C.M. Aptamer based fluorescence recovery assay for aflatoxin B1 using a quencher system composed of quantum dots and graphene oxide. Microchim. Acta 2015, 182, 571–578. [Google Scholar] [CrossRef]
- He, Z.; Zang, S.; Liu, Y.; He, Y.; Lei, H. A multi-walled carbon nanotubes-poly(l-lysine) modified enantioselective immunosensor for ofloxacin by using multi-enzyme-labeled gold nanoflower as signal enhancer. Biosens. Bioelectron. 2015, 73, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Li, J.R.; Zhang, G.N.; Wang, L.H.; Shen, A.G.; Hu, J.M. Simultaneous enzymatic and SERS properties of bifunctional chitosan-modified popcorn-like Au-Ag nanoparticles for high sensitive detection of melamine in milk powder. Talanta 2015, 140, 204–211. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.Q.; Zhang, B.; Chen, G.N.; Tang, D.P. Biotin-avidin-conjugated metal sulfide nanoclusters for simultaneous electrochemical immunoassay of tetracycline and chloramphenicol. Microchim. Acta 2014, 181, 257–262. [Google Scholar] [CrossRef]
- Zhang, L.L.; Wong, J.X.H.; Li, X.C.; Li, Y.C.; Yi, H.Z. Detection and quantitation of heavy metal ions on bona fide DVDs using DNA molecular beacon probes. Anal. Chem. 2015, 87, 5062–5067. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Gao, P.; Liu, Y.; Li, R.; Ma, H.; Du, B.; Wei, Q. Label-free photoelectrochemical immunosensor for sensitive detection of Ochratoxin A. Biosens. Bioelectron. 2015, 64, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.Z.; Cai, L.; Chen, M.Y.; Lin, Y.; Pang, D.W.; Tang, H.W. Indirect immunofluorescence detection of E. coli O157:H7 with fluorescent silica nanoparticles. Biosens. Bioelectron. 2015, 66, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Tedsana, W.; Tuntulani, T.; Ngeontae, W. A circular dichroism sensor for Ni2+ and Co2+ based on l-cysteine capped cadmium sulfide quantum dots. Anal. Chim. Acta 2015, 867, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Deep, A.; Bhardwaj, S.K.; Paul, A.K.; Kim, K.H.; Kumar, P. Surface assembly of nano-metal organic framework on amine functionalized indium tin oxide substrate for impedimetric sensing of parathion. Biosens. Bioelectron. 2015, 65, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.L.; Patel, K.; Schexnider, A.; Banu, S.; Radadia, A.D. Nanostructuring of biosensing electrodes with nanodiamonds for antibody immobilization. ACS Nano 2014, 8, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Dinda, D.; Shaw, B.K.; Saha, S.K. Thymine functionalized graphene oxide for fluorescence “turn-off-on” sensing of Hg2+ and I- in aqueous medium. ACS Appl. Mater. Interfaces 2015, 7, 14743–14749. [Google Scholar] [CrossRef] [PubMed]
- Yoo, K.H.; Lee, S.H.; Kim, H.J.; Sung, K.W.; Jung, H.L.; Cho, E.J.; Park, H.K.; Kim, H.A.; Koo, H.H. The impact of post-thaw colony-forming units-granulocyte//macrophage on engraftment following unrelated cord blood transplantation in pediatric recipients. Bone Marrow Transplant. 2007, 39, 515–521. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.; Welch, K.; Jarvius, J.; Cai, Y.X.; Brucas, R.; Nikolajeff, F.; Svedlindh, P.; Nilsson, M. Gold nanowire based electrical DNA detection using rolling circle amplification. ACS Nano 2014, 8, 1147–1153. [Google Scholar] [CrossRef] [PubMed]
- Khunrattanaporn, N.; Rijiravanich, P.; Somasundrum, M.; Surareungchai, W. Highly sensitive electrochemical detection of genomic DNA based on stem loop probes structured for magnetic collection and measurement via metalised hollow polyelectrolyte shells. Biosens. Bioelectron. 2015, 73, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Quintela, I.A.; de los Reyes, B.G.; Lin, C.S.; Wu, V.C.H. Simultaneous direct detection of Shiga-toxin producing Escherichia coli (STEC) strains by optical biosensing with oligonucleotide-functionalized gold nanoparticles. Nanoscale 2015, 7, 2417–2426. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Lee, J.; Lee, H.S.; Chang, J.H. Rapid pathogen detection with bacterial-assembled magnetic mesoporous silica. Biosens. Bioelectron. 2014, 53, 123–128. [Google Scholar] [CrossRef] [PubMed]
- El Ichi, S.; Leon, F.; Vossier, L.; Marchandin, H.; Errachid, A.; Coste, J.; Jaffrezic-Renault, N.; Fournier-Wirth, C. Microconductometric immunosensor for label-free and sensitive detection of gram-negative bacteria. Biosens. Bioelectron. 2014, 54, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Lee, G.; Song, X.; Park, S.; Kim, M. Immunoliposome-based immunomagnetic concentration and separation assay for rapid detection of Cronobacter sakazakii. Biosens. Bioelectron. 2016, 77, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Meng, K.; Sun, W.; Zhao, P.; Zhang, L.; Cai, D.; Cheng, Z.; Guo, H.; Liu, J.; Yang, D.; Wang, S.; Chai, T. Development of colloidal gold-based immunochromatographic assay for rapid detection of Mycoplasma suis in porcine plasma. Biosens. Bioelectron. 2014, 55, 396–399. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Huang, Y.J.; Wang, J.Y.; Rong, Y.; Lai, W.H.; Zhang, J.W.; Chen, T. Hierarchical flowerlike gold nanoparticles labeled immunochromatography test strip for highly sensitive detection of Escherichia coli O157:H7. Langmuir 2015, 31, 5537–5544. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.N.; Zhang, F.; Zhang, H.Y.; Shen, J.Z.; Han, E.; Dong, X.Y. Functionalized gold nanorod-based labels for amplified electrochemical immunoassay of E. coli as indicator bacteria relevant to the quality of dairy product. Talanta 2015, 132, 600–605. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, T.V.; Soares, N.D.F.; Coimbra, J.S.D.; de Andrade, N.J.; Moura, L.G.; Medeiros, E.A.A.; de Medeiros, H.S. Stability and sensitivity of polydiacetylene vesicles to detect Salmonella. Sens. Actuators B Chem. 2015, 221, 653–658. [Google Scholar] [CrossRef]
- Park, J.; Shin, J.H.; Park, J.-K. Pressed paper-based bipstick for detection of foodborne pathogens with multistep reactions. Anal. Chem. 2016, 88, 3781–3788. [Google Scholar] [CrossRef] [PubMed]
- Morales-Narvaez, E.; Naghdi, T.; Zor, E.; Merkoci, A. Photo luminescent lateral-flow immunoassay revealed by graphene oxide: Highly sensitive paper-based pathogen detection. Anal. Chem. 2015, 87, 8573–8577. [Google Scholar] [CrossRef] [PubMed]
- Jayasena, S.D. Aptamers: An emerging class of molecules that rival antibodies in diagnostics. Clin. Chem. 1999, 45, 1628–1650. [Google Scholar] [PubMed]
- Chen, J.H.; Alcaine, S.D.; Jiang, Z.W.; Rotello, V.M.; Nugen, S.R. Detection of Escherichia coli in drinking water using T7 bacteriophage-conjugated magnetic probe. Anal. Chem. 2015, 87, 8977–8984. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Bejhed, R.S.; Svedlindh, P.; Stromberg, M. Blu-ray optomagnetic measurement based competitive immunoassay for Salmonella detection. Biosens. Bioelectron. 2016, 77, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.I.; Chang, H.T.; Lin, C.H.; Shen, Y.W.; Unnikrishnan, B.; Li, Y.J.; Huang, C.C. One-step synthesis of biofunctional carbon quantum dots for bacterial labeling. Biosens. Bioelectron. 2015, 68, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wu, X.M.; Huang, Y.W.; Zhao, Y.P. Detection of E. coli using SERS active filters with silver nanorod array. Sens. Actuators B Chem. 2014, 191, 485–490. [Google Scholar] [CrossRef]
- Wu, S.J.; Duan, N.; Shi, Z.; Fang, C.C.; Wang, Z.P. Simultaneous aptasensor for multiplex pathogenic bacteria detection based on multicolor upconversion nanoparticles labels. Anal. Chem. 2014, 86, 3100–3107. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.; Kang, J.S.; Jurng, J.S.; Jung, J.H.; Kim, B.C. Fast and continuous microorganism detection using aptamer-conjugated fluorescent nanoparticles on an optofluidic platform. Biosens. Bioelectron. 2015, 67, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Jackson, A.A.; Rotello, V.M.; Nugen, S.R. Colorimetric detection of Escherichia coli based on the enzyme-induced metallization of gold nanorods. Small 2016, 12, 2469–2475. [Google Scholar] [CrossRef] [PubMed]
- Hussein, H.S.; Brasel, J.M. Toxicity, metabolism, and impact of mycotoxins on human and animals. Toxicology 2001, 167, 101–134. [Google Scholar] [CrossRef]
- Turner, N.W.; Subrahmanyam, S.; Piletsky, S.A. Analytical methods for determination of mycotoxins: A review. Anal. Chim. Acta 2009, 632, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Das, A.P.; Kumar, P.S.; Swain, S. Recent advances in biosensor based endotoxin detection. Biosens. Bioelectron. 2014, 51, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhou, Q.; Lin, Y.; Tang, D.; Chen, G.; Tang, D. Simple and sensitive detection of aflatoxin B1 within five minute using a non-conventional competitive immunosensing mode. Biosens. Bioelectron. 2015, 74, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Zhu, Z.; Zou, Y.; Huang, Y.; Liu, D.; Jia, S.; Xu, D.; Wu, M.; Zhou, Y.; Zhou, S.; et al. Target-responsive “sweet” hydrogel with glucometer readout for portable and quantitative detection of non-glucose targets. J. Am. Chem. Soc. 2013, 135, 3748–3751. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.L.; Xu, H.Y.; Huang, X.L.; Kuang, M.; Xiong, Y.H.; Xu, H.; Xu, Y.; Chen, H.Y.; Wang, A. Immunochromatographic assay for ultrasensitive detection of aflatoxin B-1 in maize by highly luminescent quantum dot beads. ACS Appl. Mater. Interfaces 2014, 6, 14215–14222. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Lim, H.B. Chemiluminescence immunoassay using magnetic nanoparticles with targeted inhibition for the determination of ochratoxin A. Talanta 2015, 140, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Chen, Y.; Wu, Y.; Weng, B.; Liu, Y.; Lu, Z.; Li, C.M.; Yu, C. Aptamer induced assembly of fluorescent nitrogen-doped carbon dots on gold nanoparticles for sensitive detection of AFB 1. Biosens. Bioelectron. 2016, 78, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Rivas, L.; Mayorga-Martinez, C.C.; Quesada-Gonzalez, D.; Zamora-Galvez, A.; de la Escosura-Muniz, A.; Merkoci, A. Label-free impedimetric aptasensor for ochratoxin-A detection using iridium oxide nanoparticles. Anal. Chem. 2015, 87, 5167–5172. [Google Scholar] [CrossRef] [PubMed]
- Bulbul, G.; Hayat, A.; Andreescu, S. A generic amplification strategy for electrochemical aptasensors using a non-enzymatic nanoceria tag. Nanoscale 2015, 7, 13230–13238. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Zhang, X.; Cai, S.X.; Wu, D.Z.; Chen, M.; Wang, S.H.; Zhang, J. A fluorescent aptasensor based on DNA-scaffolded silver-nanocluster for ochratoxin A detection. Biosens. Bioelectron. 2014, 57, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhang, J.; Wang, X.; Duan, Y.X. Amplified fluorescent aptasensor through catalytic recycling for highly sensitive detection of ochratoxin A. Biosens. Bioelectron. 2015, 65, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Kuo, F.Y.; Chang, B.Y.; Wu, C.Y.; Mong, K.K.T.; Chen, Y.C. Magnetic nanoparticle-based platform for characterization of shiga-like Toxin 1 from complex samples. Anal. Chem. 2015, 87, 10513–10520. [Google Scholar] [CrossRef] [PubMed]
- McCullum, C.; Tchounwou, P.; Ding, L.-S.; Liao, X.; Liu, Y.-M. Extraction of aflatoxins from liquid foodstuff samples with polydopamine-coated superparamagnetic nanoparticles for HPLC-MS/MS analysis. J. Agric. Food Chem. 2014, 62, 4261–4267. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.L.; Chen, M.M.; Zhang, H.Q.; Wen, W.; Zhang, X.H.; Wang, S.F. Solid-state electrochemiluminescence sensor based on RuSi nanoparticles combined with molecularly imprinted polymer for the determination of ochratoxin A. Sens. Actuators B Chem. 2016, 222, 264–269. [Google Scholar] [CrossRef]
- Kestwal, R.M.; Bagal-Kestwal, D.; Chiang, B.H. Fenugreek hydrogel-agarose composite entrapped gold nanoparticles for acetylcholinesterase based biosensor for carbamates detection. Anal. Chim. Acta 2015, 886, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Haddaoui, M.; Raouafi, N. Chlortoluron-induced enzymatic activity inhibition in tyrosinase/ZnO NPs/SPCE biosensor for the detection of ppb levels of herbicide. Sens. Actuators B Chem. 2015, 219, 171–178. [Google Scholar] [CrossRef]
- Yan, X.; Li, H.X.; Han, X.S.; Su, X.G. A ratiometric fluorescent quantum dots based biosensor for organophosphorus pesticides detection by inner-filter effect. Biosens. Bioelectron. 2015, 74, 277–283. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.J.; Zhang, T.T.; Liang, B.; Han, D.F.; Zeng, L.X.; Zheng, C.; Li, T.; Wei, M.D.; Liu, A.H. Sensitive electrochemical microbial biosensor for p-nitrophenylorganophosphates based on electrode modified with cell surface-displayed organophosphorus hydrolase and ordered mesopore carbons. Biosens. Bioelectron. 2014, 60, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Huo, D.Q.; Li, Q.; Zhang, Y.C.; Hou, C.J.; Lei, Y. A highly efficient organophosphorus pesticides sensor based on CuO nanowires-SWCNTs hybrid nanocomposite. Sens. Actuators B Chem. 2014, 199, 410–417. [Google Scholar] [CrossRef]
- Kaur, B.; Srivastava, R.; Satpati, B. Silver nanoparticle decorated polyaniline-zeolite nanocomposite material based non-enzymatic electrochemical sensor for nanomolar detection of lindane. RSC Adv. 2015, 5, 57657–57665. [Google Scholar] [CrossRef]
- Wang, M.Y.; Huang, J.R.; Wang, M.; Zhang, D.E.; Chen, J. Electrochemical nonenzymatic sensor based on CoO decorated reduced graphene oxide for the simultaneous determination of carbofuran and carbaryl in fruits and vegetables. Food Chem. 2014, 151, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Jin, J.; Yuan, C.X.; Zhang, F.; Ma, L.L.; Qin, D.D.; Shan, D.L.; Lu, X.Q. A novel electrochemical sensor based on zirconia/ordered macroporous polyaniline for ultrasensitive detection of pesticides. Analyst 2015, 140, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.W.; Hou, L.J.; Du, M.; Zhang, T.T.; Wang, Z.H.; Xue, Z.H.; Lu, X.Q. A molecularly imprinted electrochemical enzymeless sensor based on functionalized gold nanoparticle decorated carbon nanotubes for methyl-parathion detection. RSC Adv. 2014, 4, 53701–53710. [Google Scholar] [CrossRef]
- Kubackova, J.; Fabriciova, G.; Miskovsky, P.; Jancura, D.; Sanchez-Cortes, S. Sensitive surface-enhanced Raman spectroscopy (SERS) detection of organochlorine hesticides by alkyl dithiol-functionalized metal nanoparticles-induced plasmonic hot spots. Anal. Chem. 2015, 87, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Polavarapu, L.; la Porta, A.; Novikov, S.M.; Coronado-Puchau, M.; Liz-Marzan, L.M. Pen-on-paper approach toward the design of universal surface enhanced Raman scattering substrates. Small 2014, 10, 3065–3071. [Google Scholar] [CrossRef] [PubMed]
- Belkhamssa, N.; Justino, C.I.L.; Santos, P.S.M.; Cardoso, S.; Lopes, I.; Duarte, A.C.; Rocha-Santos, T.; Ksibi, M. Label-free disposable immunosensor for detection of atrazine. Talanta 2016, 146, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.H.; Wang, W.H.; Wen, H.B.; Gan, C.F.; Lei, H.T.; Liu, Y.J. Sensitive electrochemical immunoassay for chlorpyrifos by using flake-like Fe3O4 modified carbon nanotubes as the enhanced multienzyme label. Anal. Chim. Acta 2015, 899, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.T.; Shi, X.Z.; Jiao, H.F.; Sun, A.L.; Ding, H.; Zhang, R.R.; Pan, D.D.; Li, D.X.; Chen, J. Selective and sensitive determination of cypermethrin in fish via enzyme-linked immunosorbent assay-like method based on molecularly imprinted artificial antibody-quantum dot optosensing materials. Biosens. Bioelectron. 2016, 75, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Xiao, H.; Yang, S.; Liu, C.; Liang, J.; Tang, Y. Ultrasensitive detection of pentachlorophenol based on enhanced electrochemiluminescence of Au nanoclusters/graphene hybrids. Sens. Actuators B Chem. 2014, 194, 325–331. [Google Scholar] [CrossRef]
- Mei, Q.S.; Jing, H.R.; Li, Y.; Yisibashaer, W.; Chen, J.; Li, B.N.; Zhang, Y. Smartphone based visual and quantitative assays on upconversional paper sensor. Biosens. Bioelectron. 2016, 75, 427–432. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Sun, D.E.; Liu, Y.J.; Liu, Z.H. An ultrasensitive homogeneous aptasensor for kanamycin based on upconversion fluorescence resonance energy transfer. Biosens. Bioelectron. 2014, 55, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zuo, P.; Ye, B.C. A low-cost and simple paper-based microfluidic device for simultaneous multiplex determination of different types of chemical contaminants in food. Biosens. Bioelectron. 2015, 68, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Emrani, A.S.; Danesh, N.M.; Lavaee, P.; Ramezani, M.; Abnous, K.; Taghdisi, S.M. Colorimetric and fluorescence quenching aptasensors for detection of streptomycin in blood serum and milk based on double-stranded DNA and gold nanoparticles. Food Chem. 2016, 190, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.Y.; Han, T.; Li, X.Q.; Sun, L.H.; Zhang, Y.J.; Zhang, Y.S. Colorimetric detection of kanamycin based on analyte-protected silver nanoparticles and aptamer-selective sensing mechanism. Anal. Chim. Acta 2015, 891, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.X.; He, Y.; Jiang, J.; Cui, H. A competitive immunoassay for sensitive detection of small molecules chloramphenicol based on luminol functionalized silver nanoprobe. Anal. Chim. Acta 2014, 812, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Kwon, M.S.; Phillips, A.W.; Seo, D.; Kim, J. Highly sensitive turn-on biosensors by regulating fluorescent dye assembly on liposome surfaces. Chem. Commun. 2015, 51, 10229–10232. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.Z.; Guo, Z.; Yuan, Q.H.; Liu, Z.G.; Liu, J.H.; Huang, X.J. Exploiting differential electrochemical stripping behaviors of Fe3O4 nanocrystals toward heavy metal ions by crystal cutting. ACS Appl. Mater. Interfaces 2014, 6, 12203–12213. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Yu, H.M.; Wu, L.; Hou, X.D.; Yang, L.; Zheng, C.B. Three birds with one Fe3O4 nanoparticle: Integration of microwave digestion, solid phase extraction, and magnetic separation for sensitive determination of arsenic and antimony in fish. Anal. Chem. 2015, 87, 5866–5871. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, N.; Mohapatra, M.; Leung, K.T. Bimetallic nanoparticles for arsenic detection. Anal. Chem. 2015, 87, 5546–5552. [Google Scholar] [CrossRef] [PubMed]
- Rong, M.; Lin, L.; Song, X.; Wang, Y.; Zhong, Y.; Yan, J.; Feng, Y.; Zeng, X.; Chen, X. Fluorescence sensing of chromium (VI) and ascorbic acid using graphitic carbon nitride nanosheets as a fluorescent ldquoswitchrdquo. Biosens. Bioelectron. 2015, 68, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Sun, J.F.; Cao, D.; Zhang, L.Q.; Liu, J.F.; Jiang, G.B. Fabrication of highly-specific SERS substrates by co-precipitation of functional nanomaterials during the self-sedimentation of silver nanowires into a nanoporous film. Chem. Commun. 2015, 51, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.W.; Cao, F.J.; Zheng, W.S.; Tian, Y.; Xianyu, Y.L.; Xu, P.; Zhang, W.; Wang, Z.; Deng, K.; Jiang, X.Y. Detection of the nanomolar level of total Cr (III) and (VI) by functionalized gold nanoparticles and a smartphone with the assistance of theoretical calculation models. Nanoscale 2015, 7, 2042–2049. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, P.; Singh, D.K.; Sadhu, S.; Poddar, P. Study of the nucleation and growth of antibiotic labeled Au NPs and blue luminescent Au-8 quantum clusters for Hg2+ ion sensing, cellular imaging and antibacterial applications. Nanoscale 2015, 7, 19985–20002. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.N.; Ren, W.X.; Kim, J.S.; Yoon, J. Fluorescent and colorimetric sensors for detection of lead, cadmium, and mercury ions. Chem. Soc. Rev. 2012, 41, 3210–3244. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.S.; Nagi, R.; Sadeghi, K.; Feng, S.; Yan, E.; Ki, S.J.; Caire, R.; Tseng, D.; Ozcan, A. Detection and spatial mapping of mercury contamination in water samples using a smart-phone. ACS Nano 2014, 8, 1121–1129. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Wang, X.; Xue, F.; Zheng, L.; Liu, J.; Yan, F.; Xia, F.; Chen, W. Ultrasensitive and rapid screening of mercury(II) ions by dual labeling colorimetric method in aqueous samples and applications in mercury-poisoned animal tissues. Anal. Chim. Acta 2015, 868, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Li, Y.; Zhang, C.-Y. Simultaneous detection of mercury(ii) and silver(i) ions with picomolar sensitivity. Chem. Commun. 2014, 50, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xu, L.G.; Ma, W.; Kuang, H.; Wang, L.B.; Xu, C.L. Triple Raman label-encoded gold nanoparticle trimers for simultaneous heavy metal ion detection. Small 2015, 11, 3435–3439. [Google Scholar] [CrossRef] [PubMed]
- Saran, R.; Liu, J. A silver DNAzyme. Anal. Chem. 2016, 88, 4014–4020. [Google Scholar] [CrossRef] [PubMed]
- Gong, L.; Kuai, H.L.; Ren, S.L.; Zhao, X.H.; Huan, S.Y.; Zhang, X.B.; Tan, W.H. Ag nanocluster-based label-free catalytic and molecular beacons for amplified biosensing. Chem. Commun. 2015, 51, 12095–12098. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.S.; Li, C.H.; Sun, C.; Yang, X.D. Simultaneously determination of trace Cd2+ and Pb2+ based on l-cysteine/graphene modified glassy carbon electrode. Food Chem. 2016, 192, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.Q.; Liu, H.W.L.; Wu, Z.; Liu, A.; Yao, C.Z.; Li, X.Q.; Xiao, W.; Yu, S.T.; Luo, Z.; Tang, Y. Rough surface Au@Ag core-shell nanoparticles to fabricating high sensitivity SERS immunochromatographic sensors. J. Nanobiotechnol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.W.; Liu, J.W. DNA adsorption by magnetic iron oxide nanoparticles and its application for arsenate detection. Chem. Commun. 2014, 50, 8568–8570. [Google Scholar] [CrossRef] [PubMed]
- Niu, C.X.; Liu, Q.L.; Shang, Z.H.; Zhao, L.; Ouyang, J. Dual-emission fluorescent sensor based on AIE organic nanoparticles and Au nanoclusters for the detection of mercury and melamine. Nanoscale 2015, 7, 8457–8465. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, Q.Q.; Zhu, L.L.; Zhang, J.Y.; Wang, F.Y.; Lu, L.L.; Yu, H.J.; Xu, Z.A.; Zhang, W. Triplex molecular beacons for sensitive recognition of melamine based on abasic-site-containing DNA and fluorescent silver nanoclusters. Chem. Commun. 2015, 51, 7958–7961. [Google Scholar] [CrossRef] [PubMed]
- Mani, V.; Dinesh, B.; Chen, S.M.; Saraswathi, R. Direct electrochemistry of myoglobin at reduced graphene oxide-multiwalled carbon nanotubes-platinum nanoparticles nanocomposite and biosensing towards hydrogen peroxide and nitrite. Biosens. Bioelectron. 2014, 53, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Khafilzadeh, M.A.; Karimi-Maleh, H. A new strategy for determination of bisphenol A in the presence of Sudan I using a ZnO/CNTs/ionic liquid paste electrode in food samples. Food Chem. 2014, 158, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhao, S.Q.; Zhang, K.; Zhou, J.Q. Cd-doped ZnO quantum dots-based immunoassay for the quantitative determination of bisphenol A. Chemosphere 2014, 95, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Wang, J.; Zhou, G.; Wang, J.; Wu, N.; Lu, J.; Gao, J.; Chen, X.; Shi, J.; Zuo, X.; Fan, C. Programmable engineering of a biosensing interface with tetrahedral DNA nanostructures for ultrasensitive DNA detection. Angew. Chem. 2015, 127, 2179–2183. [Google Scholar] [CrossRef]
- Thacker, V.V.; Herrmann, L.O.; Sigle, D.O.; Zhang, T.; Liedl, T.; Baumberg, J.J.; Keyser, U.F. DNA origami based assembly of gold nanoparticle dimers for surface-enhanced Raman scattering. Nat. Commun. 2014. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hong, C.Y.; Wu, S.X.; Liang, H.; Wang, L.P.; Huang, G.M.; Chen, X.; Yang, H.H.; Shangguan, D.H.; Tan, W.H. Facile phase transfer and surface biofunctionalization of hydrophobic nanoparticles using Janus DNA tetrahedron nanostructures. J. Am. Chem. Soc. 2015, 137, 11210–11213. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, H.; Wang, L.M.; Yang, S.J.; Wang, W.W.; Wang, L.; Liu, F.Q.; Fu, Z.F. Chemiluminescence reaction kinetics-resolved multianalyte immunoassay strategy using a bispecific monoclonal antibody as the unique recognition reagent. Anal. Chem. 2015, 87, 2952–2958. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Mizzotti, C.; Masiero, S.; Kater, M.M.; Pesaresi, P. Peptide aptamers: The versatile role of specific protein function inhibitors in plant biotechnology. J. Integr. Plant Biol. 2015, 57, 892–901. [Google Scholar] [CrossRef] [PubMed]
| Category | Nanomaterial | Size * (Shape) | Main Function |
|---|---|---|---|
| Metallic nanomaterial | AuNPs | <100 nm (sphere) | Carrier, enhancer, reporter, quencher |
| Silver NPs (AgNPs) | <100 nm (sphere) | Enhancer, reporter | |
| Platinum NPs (PtNPs) | <100 nm (sphere) | Catalyst | |
| Metal nanoclusters | <10 nm (sphere) | Reporter | |
| Metal compound nanomaterials | Quantum dots (QDs) | 1–10 nm (sphere) | Carrier, reporter |
| Upconversion NPs | <100 nm (sphere) | Reporter | |
| Fe3O4 NPs | 5–500 nm (sphere) | Separator | |
| CuO NPs | <100 nm (sphere) | Enhancer, catalyst | |
| Non-metallic nanomaterials | SiO2 nanomaterials | Dozens of nm (sphere) | Carrier |
| Polyaniline NPs | <100 nm (sphere) | Enhancer | |
| Carbon materials | Graphene | Various (sheet) | Carrier, quencher |
| Carbon nanotube (CNTs) | Various (tube) | Carrier, enhancer, quencher | |
| Carbon dots (C dots) | <10 nm (sphere) | Reporter | |
| Nanostructures | DNA nanostructures | Various (polyhedron) | Carrier |
| Type of Contaminant | Contaminant | Recognition Biomolecule | Nanomaterials Used | Functions of Nanomaterials | Detection Format | LOD | Ref. |
|---|---|---|---|---|---|---|---|
| Pathogens | E. coli O157:H7 | cDNA | GOx, Au@SiO2 | Carrier, enhancer | Electrochemical | 0.01 nM | [10] |
| E. coli | cDNA | AuNPs, Fe3O4 | Reporter, seperator | Electrochemical | 1.8 aM | [60] | |
| C. sakazakii | Antibody | Fe3O4, liposomes | Carrier, seperator | Fluorescent | 103 CFU/mL | [64] | |
| Mycoplasma suis | Antibody | AuNPs | Carrier, reporter | Colorimetric | 100 ng/mL | [65] | |
| S. aureus, V. parahemolyticus, S. typhimurium | Aptamer | Upconversion NPs | Reporter | Fluorescent | 25, 10, 15 CFU/mL | [76] | |
| E. coli BL21 | β-galactosidase | Ag-AuNRs | Reporter | Colorimetric | 104 CFU/mL | [78] | |
| Toxins | Aflatoxin B1 | Antibody | AuNPs, SiO2 | Carrier | Electrochemical | 5 ppt | [8] |
| Shiga-like toxin 1 | Antibody | Al2O3-Fe3O4 | Carrier, seperator | Mass spectrometry | 44 pM | [91] | |
| Ochratoxin A | Aptamer | Au doped Fe3O4 | Carrier, catalyst, seperator | Colorimetric | 30 pg/mL | [28] | |
| Aflatoxin B1 | Aptamer | N-doped C dots, AuNPs | Carrier, reporter | Fluorescent | 16 pM | [86] | |
| Ochratoxin A | Aptamer | Nanoceria, GOx | Carrier, catalyst | Electrochemical | 0.1 nM | [88] | |
| Pesticides | Methyl parathion, monocrotophos, dimethoate | AChE inhibition | Upconversion NPs, AuNPs | Reporter, quencher | Fluorescent | 0.67, 23, 67 ng/L | [38] |
| Carbofuran, oxamyl, methomyl, carbaryl | AChE inhibition | AuNPs | Enhancer | Colorimetric | 2, 21, 113, 236 nM | [94] | |
| Methyl parathion | Trypsin inhibition | QDs, AuNPs | Reporter, quencher | Fluorescent | 18 ng/L | [96] | |
| Paraoxon, parathion methyl parathion | Organophosphorus hydrolase | Mesoporous carbon | Carrier | Electrochemical | 9.0, 10, 15 nM | [97] | |
| Parathion | Antibody | nanoMOF | Carrier, enhancer | Electrochemical | 0.1 ng/mL | [55] | |
| Antibiotics | Kanamycin | Aptamer | Upconversion NPs, GOx | Reporter, quencher | Fluorescent | 18 pM | [110] |
| Streptomycin | Aptamer | AuNPs | Quencher | Colorimetric and fluorescence | 73.1 nM, 47.6 nM | [112] | |
| Chloramphenicol | Antibody | AgNPs | Carrier, enhancer | Electrochemical | 7.6 ng/mL−1 | [114] | |
| Neomycin | Receptor | Liposome | Carrier | Fluorescent | 2.3 nM | [115] | |
| Metal ions | Hg2+, Ag+ | Nucleotide | AuNPs | Carrier, reporter | SERS | 8.4, 16.8 × 10−12 M | [127] |
| Pb2+ | DNAzyme | DNA-stabilized AgNCs | Reporter | Fluorescent | 17 μM | [129] | |
| Cd2+, Pb2+ | Amino acid | Graphene | Carrier | Electrochemical | 0.45, 0.12 μg/L | [130] | |
| Ni2+ | Antibody | Au@Ag core-shell NPs | Carrier, reporter | SERS | 0.05 ng/mL | [131] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, T.; Huang, H.; Zhu, F.; Lin, Q.; Zhang, L.; Liu, J. Recent Progresses in Nanobiosensing for Food Safety Analysis. Sensors 2016, 16, 1118. https://doi.org/10.3390/s16071118
Yang T, Huang H, Zhu F, Lin Q, Zhang L, Liu J. Recent Progresses in Nanobiosensing for Food Safety Analysis. Sensors. 2016; 16(7):1118. https://doi.org/10.3390/s16071118
Chicago/Turabian StyleYang, Tao, Huifen Huang, Fang Zhu, Qinlu Lin, Lin Zhang, and Junwen Liu. 2016. "Recent Progresses in Nanobiosensing for Food Safety Analysis" Sensors 16, no. 7: 1118. https://doi.org/10.3390/s16071118
APA StyleYang, T., Huang, H., Zhu, F., Lin, Q., Zhang, L., & Liu, J. (2016). Recent Progresses in Nanobiosensing for Food Safety Analysis. Sensors, 16(7), 1118. https://doi.org/10.3390/s16071118





