Wireless Power Transfer for Autonomous Wearable Neurotransmitter Sensors
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials and Apparatus
2.1.1. L-Glu Sensor Fabrication

2.1.2. Principle of Electrochemical L-Glu Sensors
2.2. Miniature Wireless Recording System

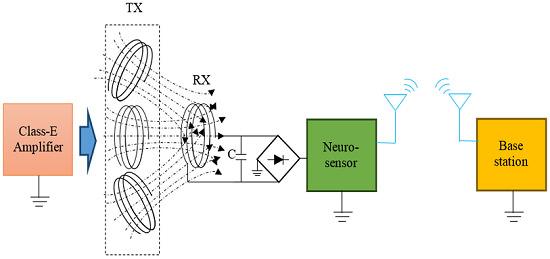
2.3. Wireless Power Transmission (WPT)
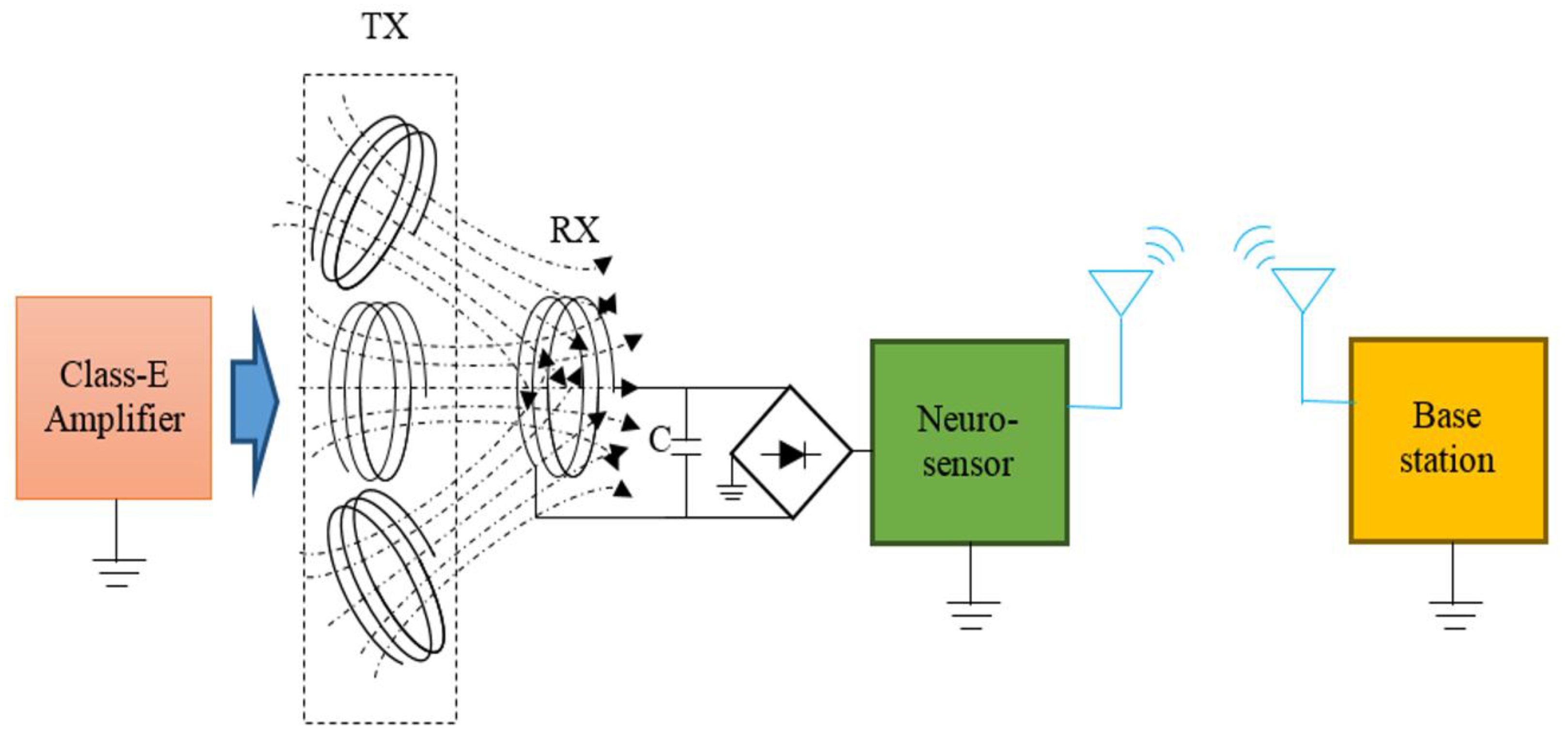
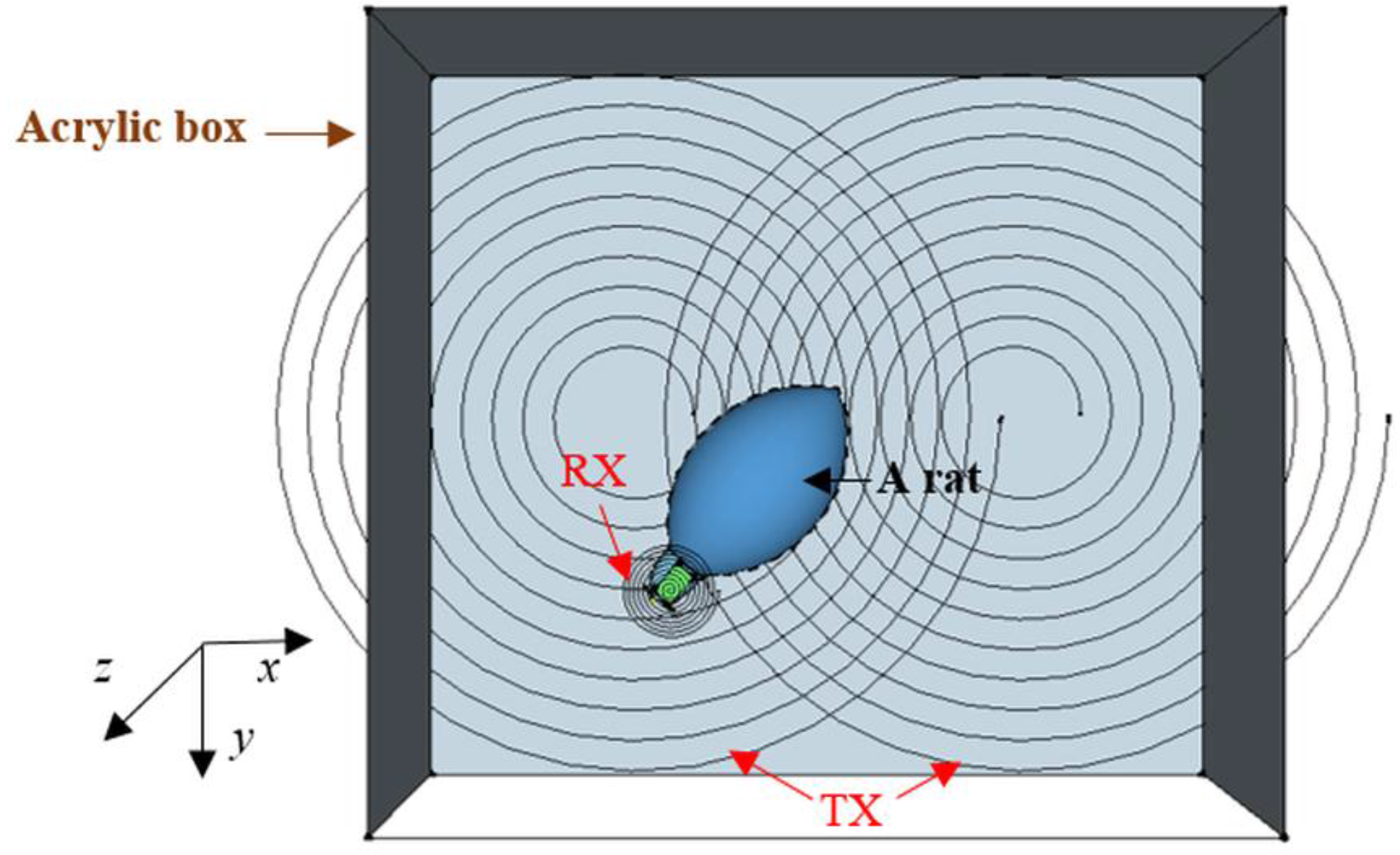
2.4. Experiment Setup
| Parameters | Transmitter | Receiver |
|---|---|---|
| Inner radius (cm) | 7 | 1.6 |
| Outer radius (cm) | 20 | – |
| Turn number | 20 | 60 |
| Turn spacing (cm) | 0.65 | – |
| Inductance (µH) | 142.3 | 239.3 |
| Resistance (Ω) | 20.5 | 212.2 |
| Quality factor | 56 | 8.3 |
2.5. In Vitro Experiment Protocol
3. Results and Discussion
3.1. One-Transmitter Antenna Configuration
3.1.1. Simulation
3.1.2. Lateral Misalignment
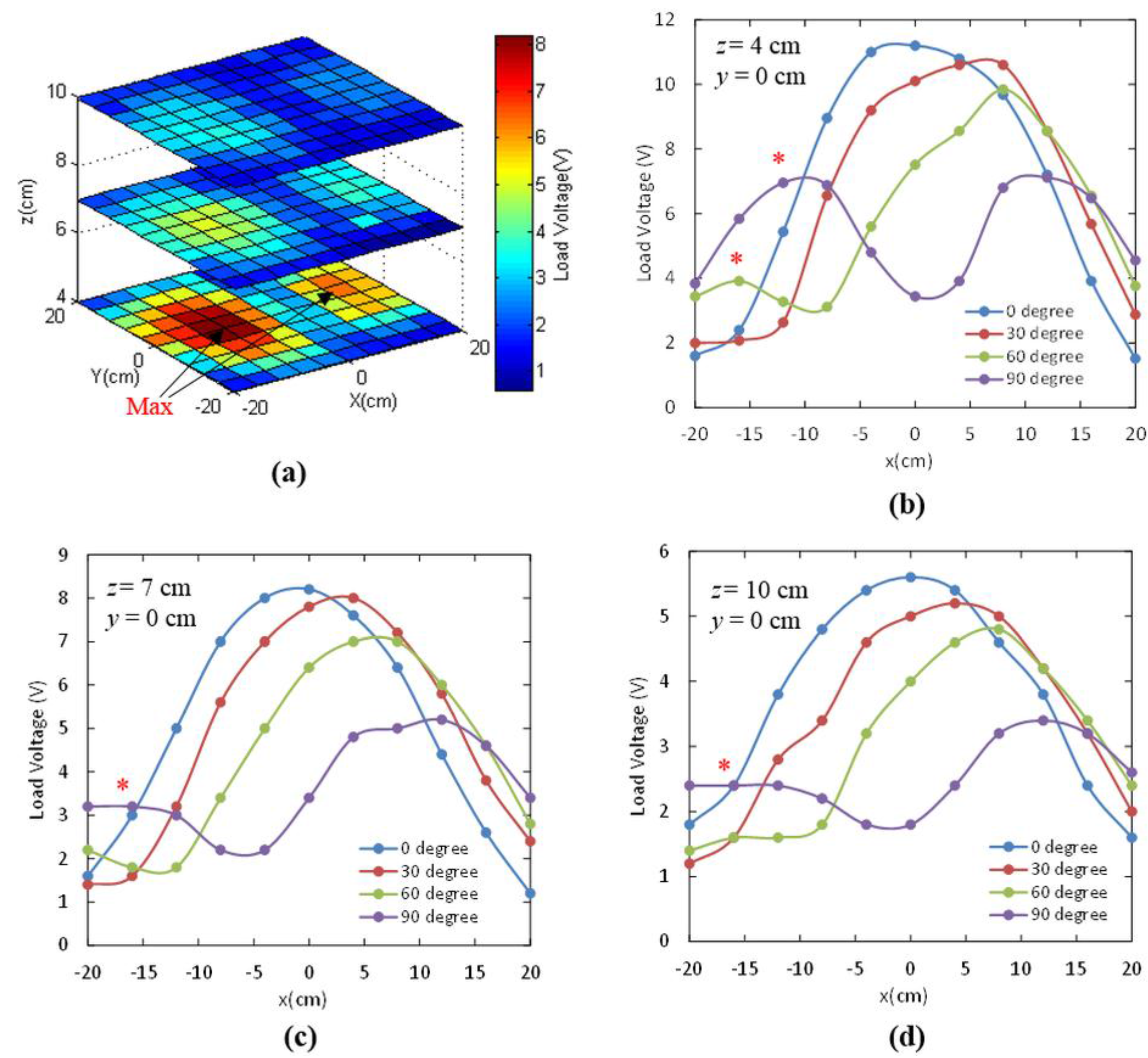
3.1.3. Angular Misalignment
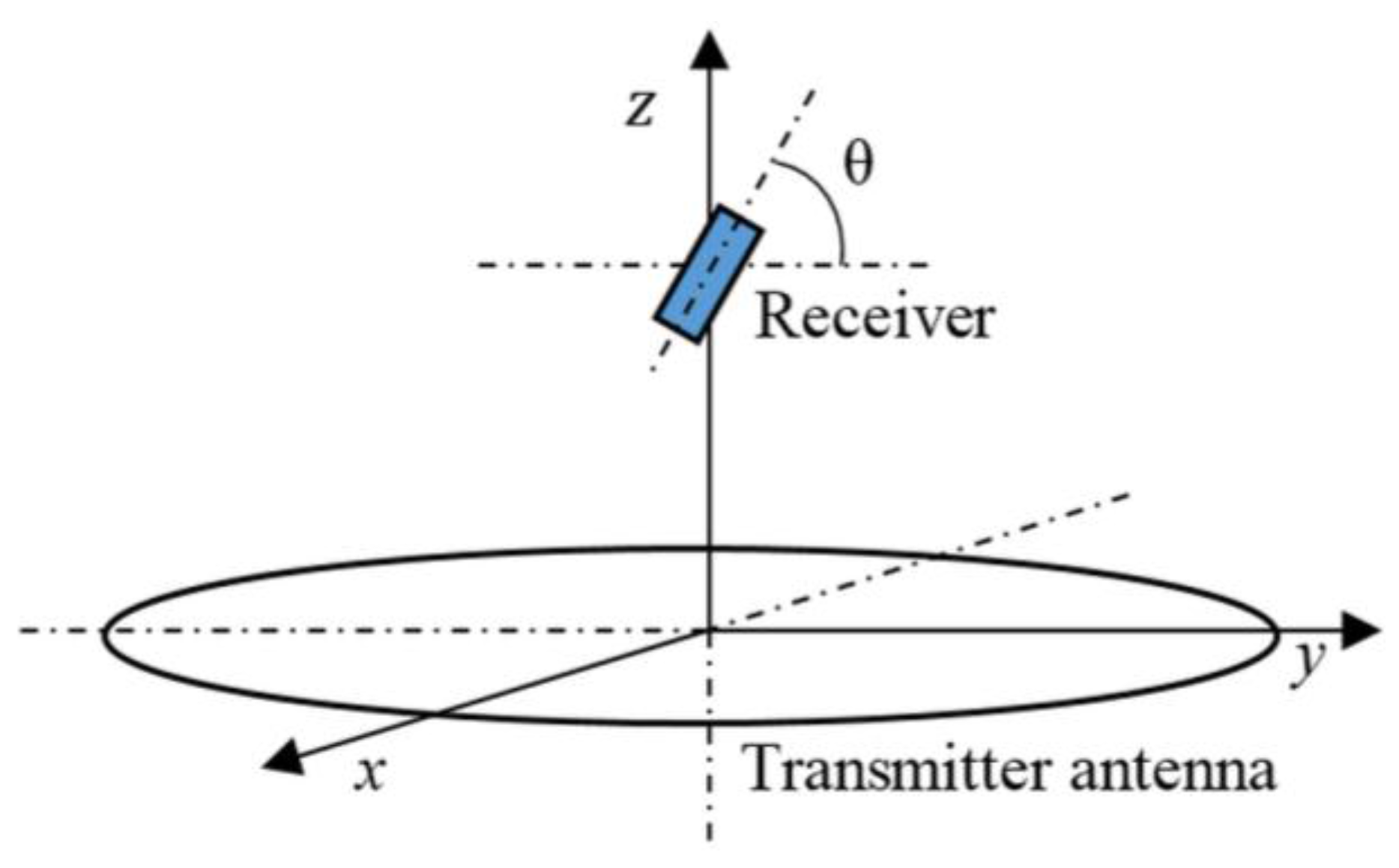
| Angular Misalignment θ | z = 4 cm | z = 7 cm | z = 10 cm |
|---|---|---|---|
| 0° | 54.5% | 56.2% | 55.3% |
| 30° | 63.6% | 54.1% | 54.1% |
| 60° | 73.9% | 52.1% | 45.8% |
| 90° | 78.1% | 63.6% | 46.3% |
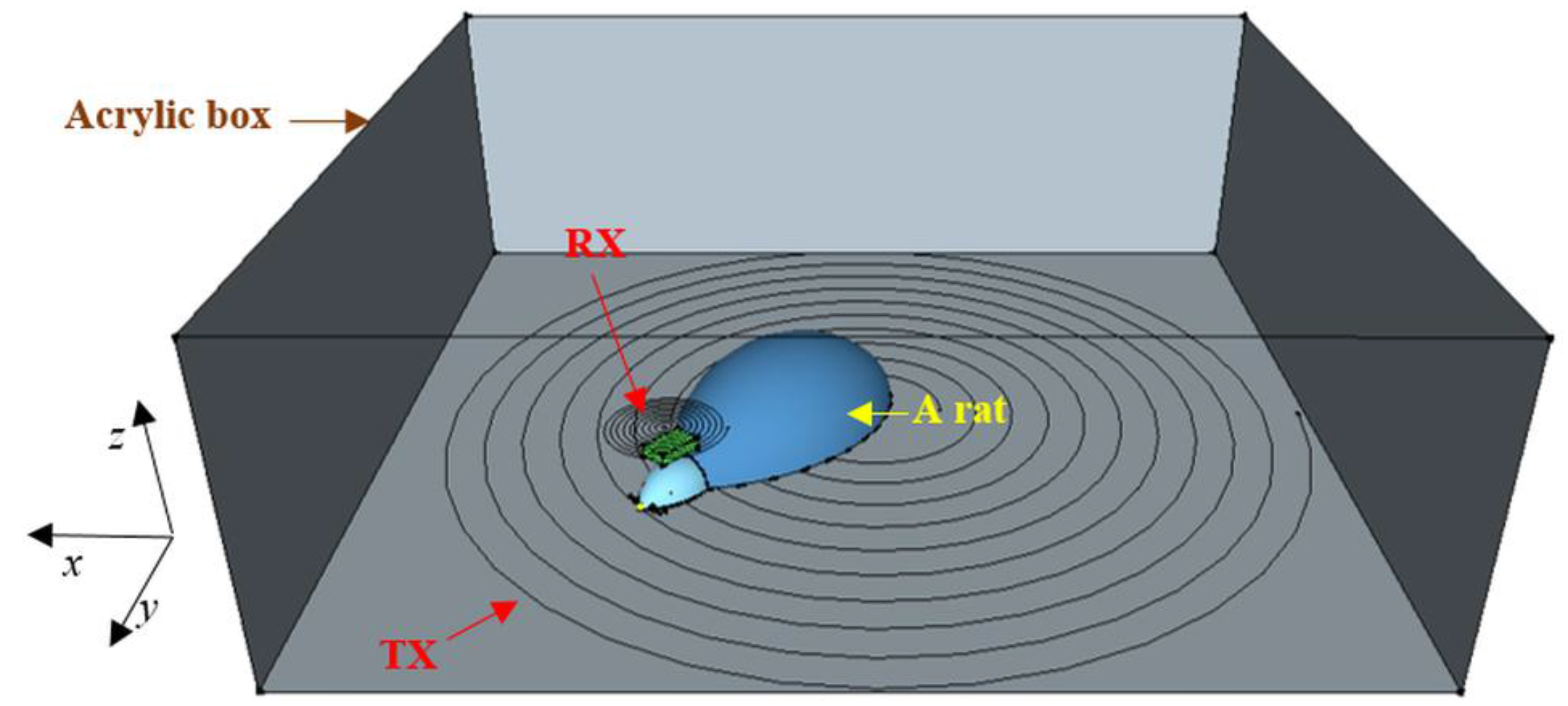
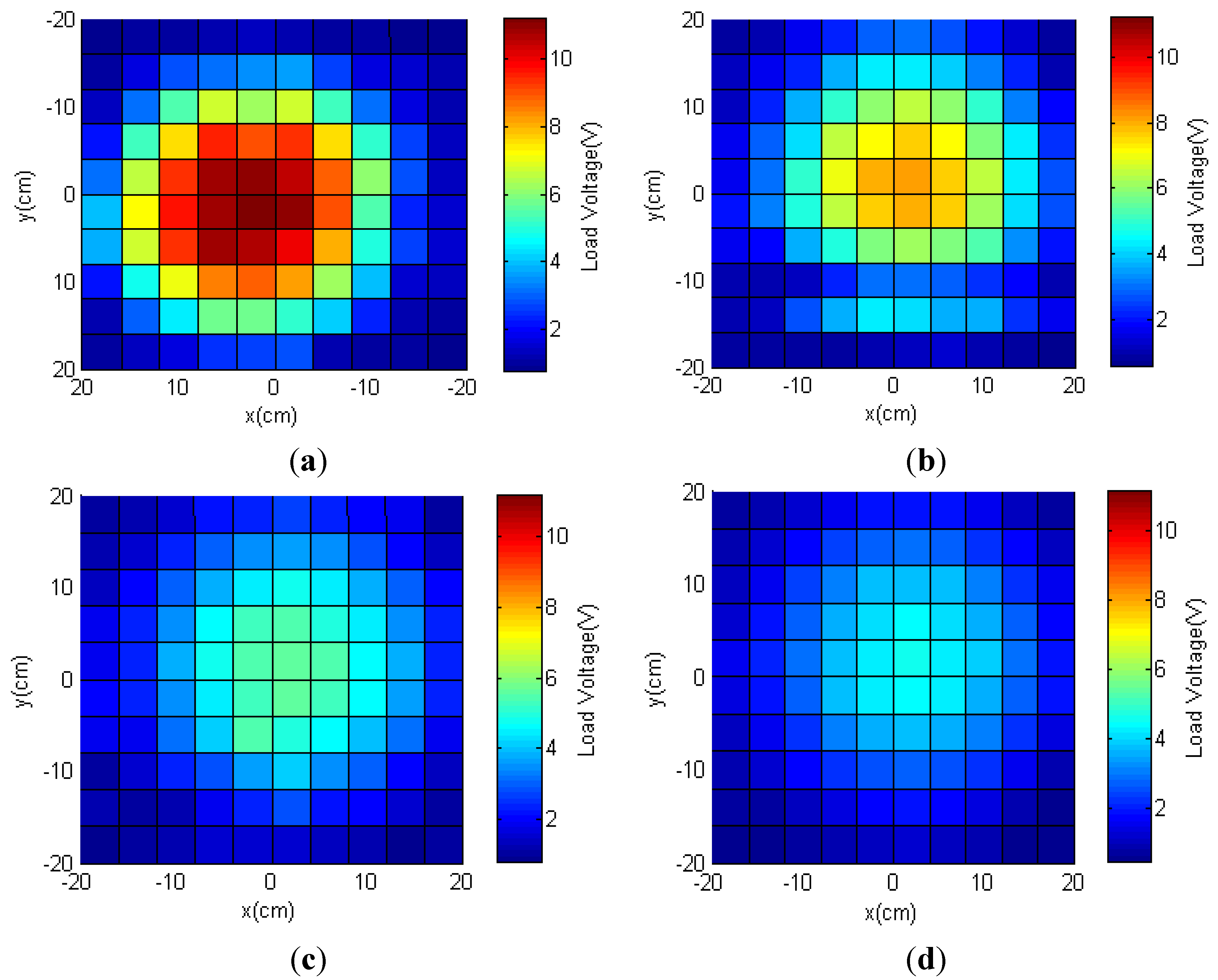
3.2. Two-Transmitter Antenna Configuration
3.2.1. Simulations
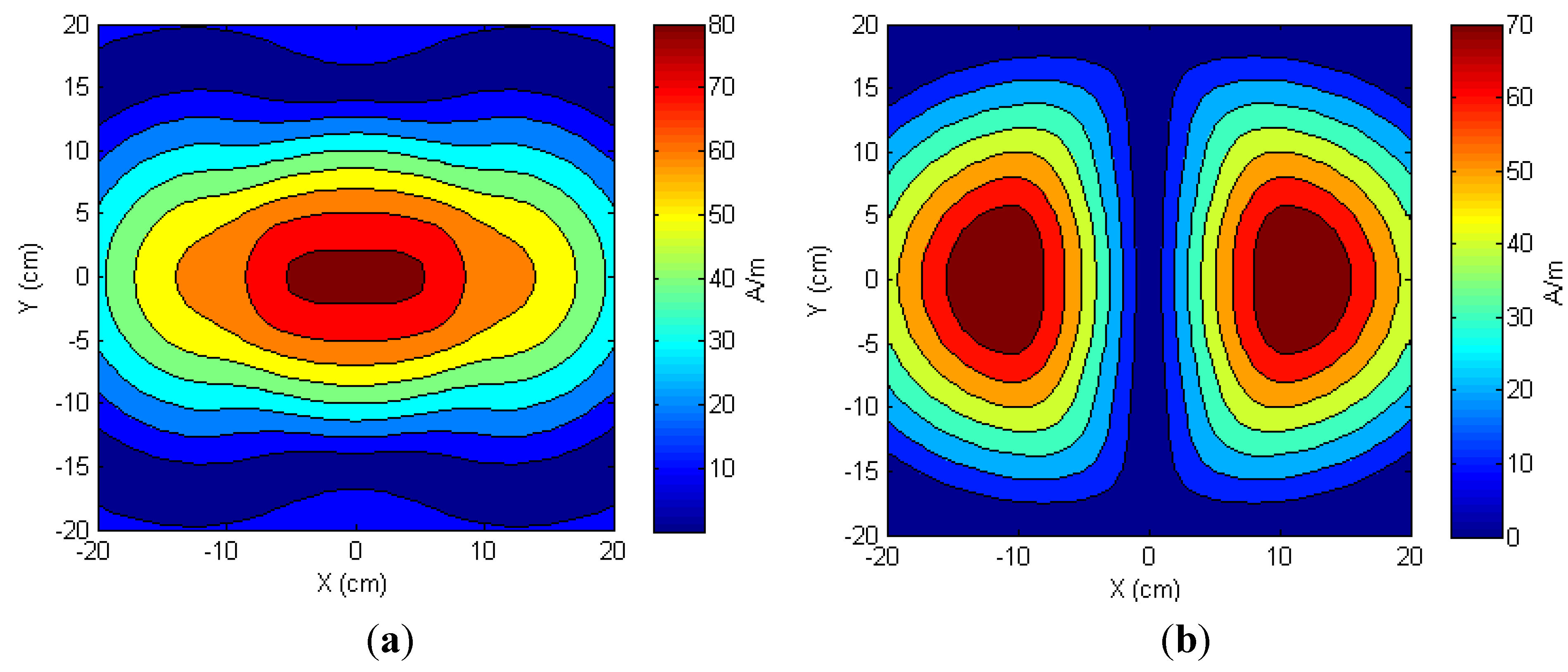
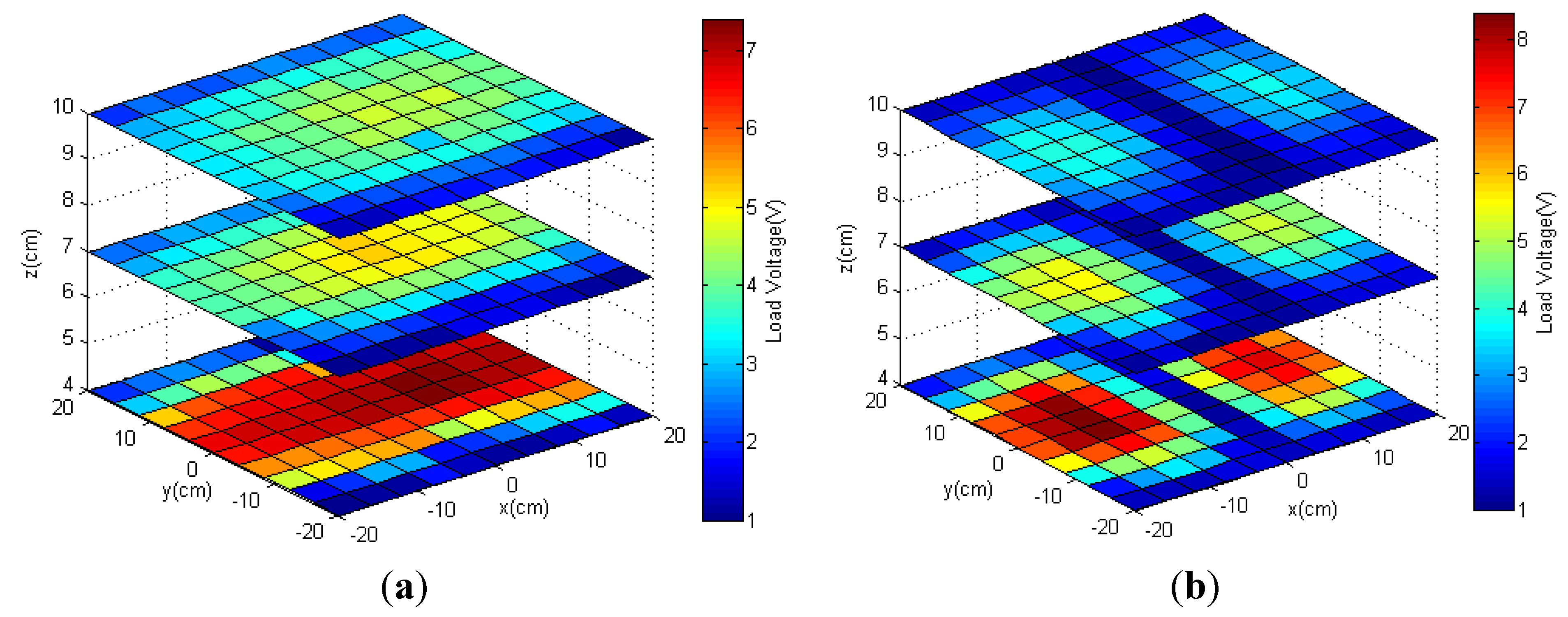
3.2.2. Measured Load Voltage
3.3. Multi-Transmitter Antenna Configuration
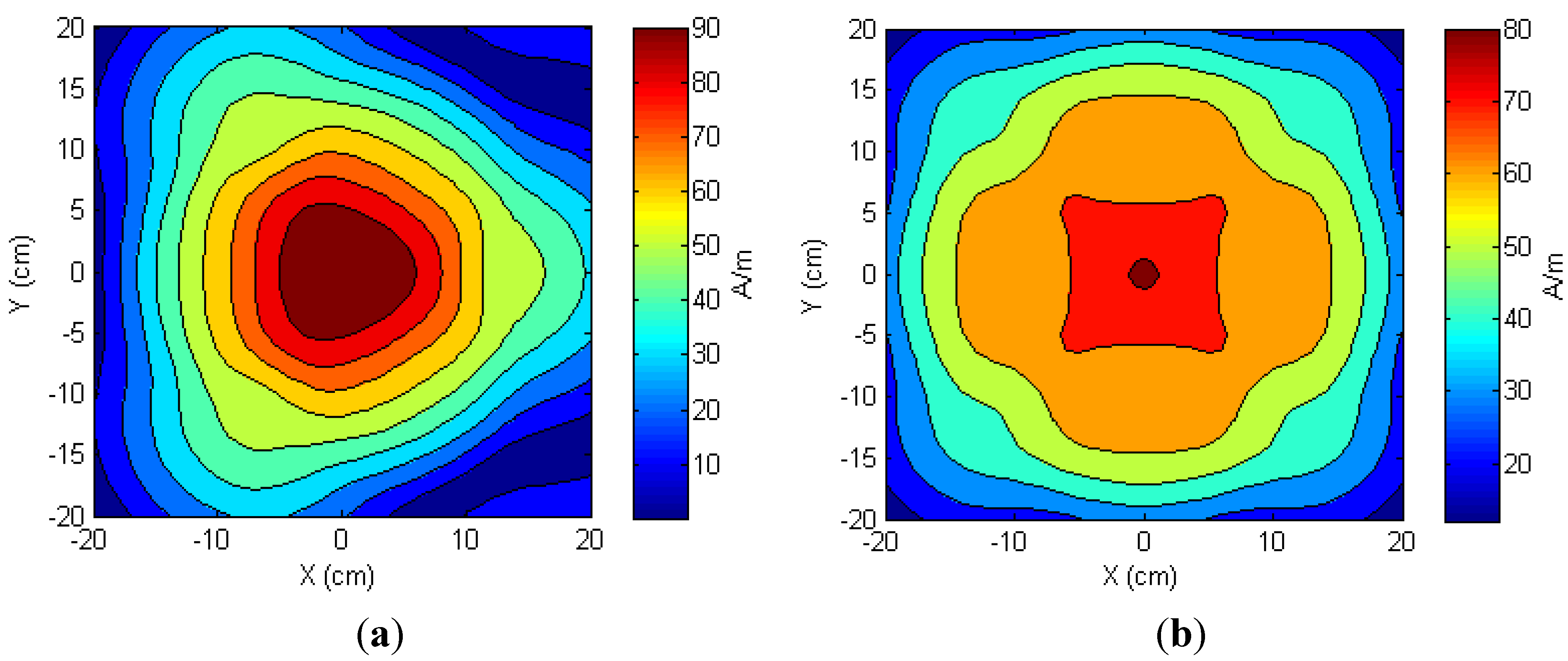
| Number of TX Antenna | Hz ≥ 30 A/m | Hz ≥ 20 A/m | Hz ≥ 15 A/m | Hz ≥ 10 A/m |
|---|---|---|---|---|
| One | 28.14% | 37.66% | 43.13% | 48.84% |
| Two | 48.70% | 58.60% | 65.73% | 76.80% |
| Three | 65.26% | 76.26% | 83.46% | 89.59% |
| Four | 88.10% | 98.97% | 99.29% | 100% |
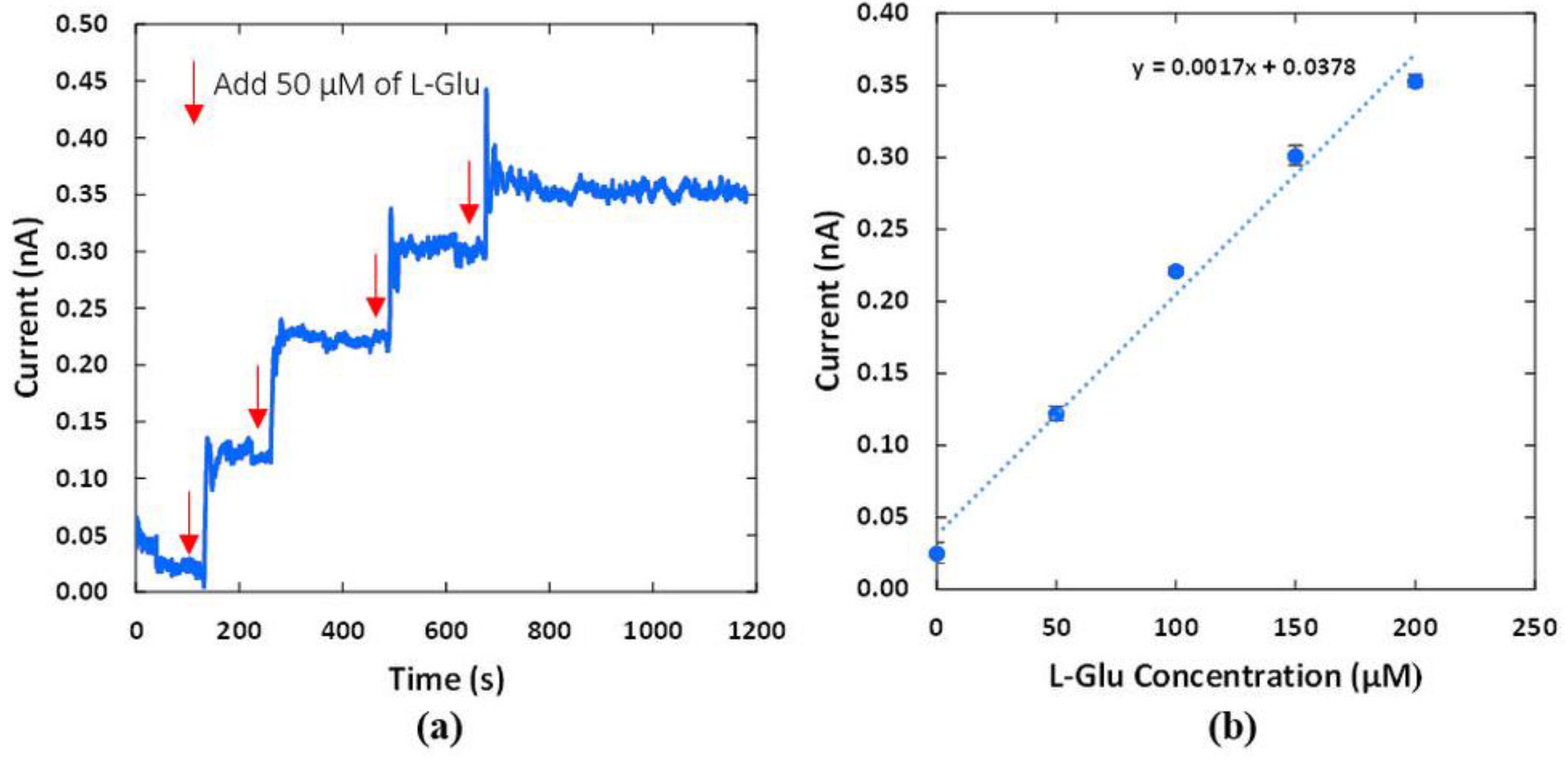
3.4. In Vitro Measurement of the L-Glu Sensors
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ryan, M.; Lowry, J.; O’Neill, R. Biosensor for neurotransmitter l-glutamic acid designed for efficient use of l-glutamate oxidase and effective rejection of interference. Analyst 1997, 122, 1419–1424. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Im, H.; Lee, D.; Kim, H.; Choi, Y. Ferrocene functionalized single-walled carbon nanotube bundles. Hybrid interdigitated construction film for L-Glutamate detection. J. Phys. Chem. C 2007, 111, 1200–1206. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, W.; Ma, C.; Li, Z. Fabrication of an electrochemical biosensor array for simultaneous detection of l-glutamate and acetylcholine. J. Biomed. Nanotechnol. 2013, 9, 1378–1382. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Li, A.; Nguyen, C.M.; Peng, Y.; Chiao, J.-C. An integrated flexible implantable micro-probe for sensing neurotransmitters. IEEE Sens. J. 2012, 12, 1618–1624. [Google Scholar] [CrossRef]
- Burmeister, J.J.; Pomerleau, F.; Palmer, M.; Day, B.K.; Huettl, P.; Gerhardt, G.A. Improved ceramic-based multisite microelectrode for rapid measurements of L-Glutamate in the CNS. J. Neurosci. Methods 2002, 119, 163–171. [Google Scholar] [CrossRef]
- Choi, D.W. Glutamate neurotoxicity and diseases of the nervous system. Neuron 1988, 1, 623–634. [Google Scholar] [CrossRef]
- Fonnum, F. Glutamate: A neurotransmitter in mammalian brain. J. Neurochem. 1984, 42, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Fagg, G.; Foster, A. Amino acid neurotransmitters and their pathways in the mammalian central nervous system. Neuroscience 1983, 9, 701–719. [Google Scholar] [CrossRef]
- Rutherford, E.C.; Pomerleau, F.; Huettl, P.; Strömberg, I.; Gerhardt, G.A. Chronic second-by-second measures of l-Glutamate in the central nervous system of freely moving rats. J. Neurochem. 2007, 102, 712–722. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, E. Molecular mechanism of neuronal plasticity: Induction and maintenance of long-term potentiation in the hippocampus. J. Pharmacol Sci. 2006, 100, 433–442. [Google Scholar] [CrossRef] [PubMed]
- McEntee, W.J.; Crook, T.H. Glutamate: Its role in learning, memory, and the aging brain. Psychopharmacology (Berl.) 1993, 111, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Neurotransmitters in the regulation of neuronal cytoarchitecture. Brain Res. Rev. 1988, 13, 179–212. [Google Scholar] [CrossRef]
- Erecińska, M.; Silver, I.A. Metabolism and role of glutamate in mammalian brain. Prog. Neurobiol. 1990, 35, 245–296. [Google Scholar] [CrossRef]
- Danbolt, N.C. Glutamate uptake. Prog. Neurobiol. 2001, 65, 1–105. [Google Scholar] [CrossRef]
- Sheldon, A.L.; Robinson, M.B. The role of glutamate transporters in neurodegenerative diseases and potential opportunities for intervention. Neurochem. Int. 2007, 51, 333–355. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Peng, Y.; Chiao, J.-C. An integrated flexible implantable L-Glutamate sensor. In Proceedings of the 2010 IEEE Sensors, Big Island, HI, USA, 1–4 November 2010; pp. 2346–2350.
- Hirsch, S.R.; Das, I.; Garey, L.J.; de Belleroche, J. A pivotal role for glutamate in the pathogenesis of schizophrenia, and its cognitive dysfunction. Pharmacol. Biochem. Behav. 1997, 56, 797–802. [Google Scholar] [CrossRef]
- Greenamyre, J.T. The role of glutamate in neurotransmission and in neurologic disease. Arch. Neurol. 1986, 43, 1058–1063. [Google Scholar] [CrossRef] [PubMed]
- Pines, G.; Danbolt, N.C.; Bjørås, M.; Zhang, Y.; Bendahan, A.; Eide, L.; Koepsell, H.; Storm-Mathisen, J.; Seeberg, E.; Kanner, B.I. Cloning and expression of a rat brain L-Glutamate transporter. Nature 1992, 360, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Michael, A.C.; Borland, L.M.; Hascup, K.N.; Rutherford, E.C.; Quintero, J.E.; Day, B.K.; Nickell, J.R.; Pomerleau, F.; Huettl, P.; Burmeister, J.J. Second-by-second measures of l-glutamate and other neurotransmitters using enzyme-based microelectrode arrays. In Electrochemical Methods for Neuroscience; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Tseng, T.T.; Monbouquette, H.G. Implantable microprobe with arrayed microsensors for combined amperometric monitoring of the neurotransmitters, glutamate and dopamine. J. Electroanal. Chem. 2012, 682, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Tseng, T.T.; Chang, C.; Chan, W. Fabrication of implantable, enzyme-immobilized glutamate sensors for the monitoring of glutamate concentration changes in vitro and in vivo. Molecules 2014, 19, 7341–7355. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Mays, J.; Cao, H.; Allard, H.; Rao, S.; Chiao, J.-C. A Wearable system for highly selective l-Glutamate neurotransmitter sensing. In Proceedings of the 2015 IEEE Topical Conference on Biomedical Wireless Technologies, Networks, and Sensing Systems (BioWireleSS), San Diego, CA, USA, 25–28 January 2015; pp. 1–3.
- Agnesi, F.; Tye, S.J.; Bledsoe, J.M.; Griessenauer, C.J.; Kimble, C.J.; Sieck, G.C.; Bennet, K.E.; Garris, P.A.; Blaha, C.D.; Lee, K.H. Wireless instantaneous neurotransmitter concentration system-based amperometric detection of dopamine, adenosine, and glutamate for intraoperative neurochemical monitoring. J. Neurosurg. 2009, 111, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Stanacevic, M.; Murari, K.; Rege, A.; Cauwenberghs, G.; Thakor, N.V. VLSI potentiostat array with oversampling gain modulation for wide-range neurotransmitter sensing. IEEE Trans. Biomed. Circuits Syst. 2007, 1, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Range, W. Integrated potentiostat for neurotransmitter sensing. IEEE Eng. Med. Biol. Mag. 2005, 24, 23–29. [Google Scholar]
- Beeby, S.P.; Tudor, M.J.; White, N. Energy harvesting vibration sources for microsystems applications. Meas. Sci. Technol. 2006, 17, R175–R195. [Google Scholar] [CrossRef]
- Anton, S.R.; Sodano, H.A. A review of power harvesting using piezoelectric materials (2003–2006). Smart Mater. Struct. 2007, 16, R1–R21. [Google Scholar] [CrossRef]
- Xu, S.; Hansen, B.J.; Wang, Z.L. Piezoelectric-nanowire-enabled power source for driving wireless microelectronics. Nat. Commun. 2010, 1. [Google Scholar] [CrossRef] [PubMed]
- Venkatasubramanian, R.; Watkins, C.; Stokes, D.; Posthill, J.; Caylor, C. Energy harvesting for electronics with thermoelectric devices using nanoscale materials. In Proceedings of the IEEE International Electron Devices Meeting, Washington, DC, USA, 10–12 December 2007; pp. 367–370.
- Brunelli, D.; Moser, C.; Thiele, L.; Benini, L. Design of a solar-harvesting circuit for batteryless embedded systems. IEEE Trans. Circuits Syst. Regul. Pap. 2009, 56, 2519–2528. [Google Scholar] [CrossRef]
- Priya, S.; Inman, D.J. Electromagnetic energy harvesting. In Energy Harvesting Technologies; Springer: New York, NY, USA, 2009. [Google Scholar]
- Chalasani, S.; Conrad, J.M. A survey of energy harvesting sources for embedded systems. In Proceedings of the IEEE Southeastcon 2008, Huntsville, AL, USA, 3–6 April 2018; pp. 442–447.
- Fujii, T.; Ishida, S. Cardiac Pacemaker Using Wireless Transmission. U.S. Patent 5,411,535, 2 May 1995. [Google Scholar]
- Kim, S.; Ho, J.S.; Chen, L.Y.; Poon, A.S. Wireless power transfer to a cardiac implant. Appl. Phys. Lett. 2012, 101. [Google Scholar] [CrossRef]
- Jow, U.; Ghovanloo, M. Design and optimization of printed spiral coils for efficient transcutaneous inductive power transmission. IEEE Trans. Biomed. Circuits Syst. 2007, 1, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Landge, V.; Tata, U.; Seo, Y.; Rao, S.; Tang, S.; Tibbals, H.; Spechler, S.; Chiao, J.-C. An implantable, batteryless, and wireless capsule with integrated impedance and pH sensors for gastroesophageal reflux monitoring. IEEE Trans. Biomed. Eng. 2012, 59, 3131–3139. [Google Scholar] [PubMed]
- Seo, Y.; Nguyen, M.Q.; Hughes, Z.; Rao, S.; Chiao, J.-C. Wireless power transfer by inductive coupling for implantable batteryless stimulators. In Proceedings of the 2012 IEEE MTT-S International Microwave Symposium, Montreal, QC, Canada, 17–22 Jun 2012; pp. 1–3.
- Terasawa, Y.; Uehara, A.; Yonezawa, E.; Saitoh, T.; Shodo, K.; Ozawa, M.; Tano, Y.; Ohta, J. A visual prosthesis with 100 electrodes featuring wireless signals and wireless power transmission. IEICE Electron. Express 2008, 5, 574–580. [Google Scholar] [CrossRef]
- Lee, S.Y.; Lee, S.C. An implantable wireless bidirectional communication microstimulator for neuromuscular stimulation. IEEE Trans. Circuits Syst. 2005, 52, 2526–2538. [Google Scholar]
- Bagotsky, V.S. Fundamentals of Electrochemistry; John Wiley & Sons: Hoboken, NJ, USA, 2005. [Google Scholar]
- Villarta, R.L.; Cunningham, D.D.; Guilbault, G.G. Amperometric enzyme electrodes for the determination of L-Glutamate. Talanta 1991, 38, 49–55. [Google Scholar] [CrossRef]
- Turner, A.; Karube, I.; Wilson, G.S. Fundamentals of amperometric sensor. In Biosensors: Fundamentals and Applications; Oxford University Press: Oxford, UK, 1987. [Google Scholar]
- Burmeister, J.J.; Gerhardt, G.A. Self-referencing ceramic-based multisite microelectrodes for the detection and elimination of interferences from the measurement of L-Glutamate and other analytes. Anal. Chem. 2001, 73, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.Q.; Hughes, Z.; Woods, P.; Seo, Y.; Rao, S.; Chiao, J.-C. Field distribution models of spiral coil for misalignment analysis in wireless power transfer systems. IEEE Trans. Microw. Theory Tech. 2014, 62, 920–930. [Google Scholar] [CrossRef]
- Wu, W.; Fang, Q. Design and simulation of printed spiral coil used in wireless power transmission systems for implant medical devices. In Proceedings of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 4018–4021.
- Sample, A.P.; Meyer, D.; Smith, J.R. Analysis, experimental results, and range adaptation of magnetically coupled resonators for wireless power transfer. IEEE Trans. Ind. Electron. 2011, 58, 544–554. [Google Scholar] [CrossRef]
- Nguyen, M.Q.; Plesa, D.; Rao, S.; Chiao, J.-C. A multi-input and multi-output wireless energy transfer system. In Proceedings of the 2014 IEEE MTT-S International Microwave Symposium, Tampa Bay, FL, USA, 1–6 June 2014; pp. 1–3.
- Basar, M.R.; Ahmad, M.Y.; Cho, J.; Ibrahim, F. Application of wireless power transmission systems in wireless capsule endoscopy: An overview. Sensors 2014, 14, 10929–10951. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.M.; Mays, J.; Plesa, D.; Rao, S.; Nguyen, M.; Chiao, J.-C. Wireless sensor nodes for environmental monitoring in internet of things. In Proceedings of the 2015 IEEE MTT-S International Microwave Symposium, Phoenix, AZ, USA, 17–22 May 2015; pp. 1–3.
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, C.M.; Kota, P.K.; Nguyen, M.Q.; Dubey, S.; Rao, S.; Mays, J.; Chiao, J.-C. Wireless Power Transfer for Autonomous Wearable Neurotransmitter Sensors. Sensors 2015, 15, 24553-24572. https://doi.org/10.3390/s150924553
Nguyen CM, Kota PK, Nguyen MQ, Dubey S, Rao S, Mays J, Chiao J-C. Wireless Power Transfer for Autonomous Wearable Neurotransmitter Sensors. Sensors. 2015; 15(9):24553-24572. https://doi.org/10.3390/s150924553
Chicago/Turabian StyleNguyen, Cuong M., Pavan Kumar Kota, Minh Q. Nguyen, Souvik Dubey, Smitha Rao, Jeffrey Mays, and J.-C. Chiao. 2015. "Wireless Power Transfer for Autonomous Wearable Neurotransmitter Sensors" Sensors 15, no. 9: 24553-24572. https://doi.org/10.3390/s150924553
APA StyleNguyen, C. M., Kota, P. K., Nguyen, M. Q., Dubey, S., Rao, S., Mays, J., & Chiao, J.-C. (2015). Wireless Power Transfer for Autonomous Wearable Neurotransmitter Sensors. Sensors, 15(9), 24553-24572. https://doi.org/10.3390/s150924553