Biomedical Probes Based on Inorganic Nanoparticles for Electrochemical and Optical Spectroscopy Applications
Abstract
:1. Introduction
2. Types of Nanoparticles
2.1. Gold Nanoparticles
2.2. Magnetic Nanoparticles
2.3. Nanocomposites
2.4. Semiconductor Nanostructures
2.5. Silver Nanoparticles
2.6. Other Nanoparticles
3. Biomedical Applications
3.1. Amino Acids
3.1.1. Electrochemical Detection
3.1.2. Colorimetric and Spectrophotometric Detection
3.2. Antigen-Antibody
3.2.1. Electrochemical Detection
3.2.2. Colorimetric and Spectrophotometric Detection
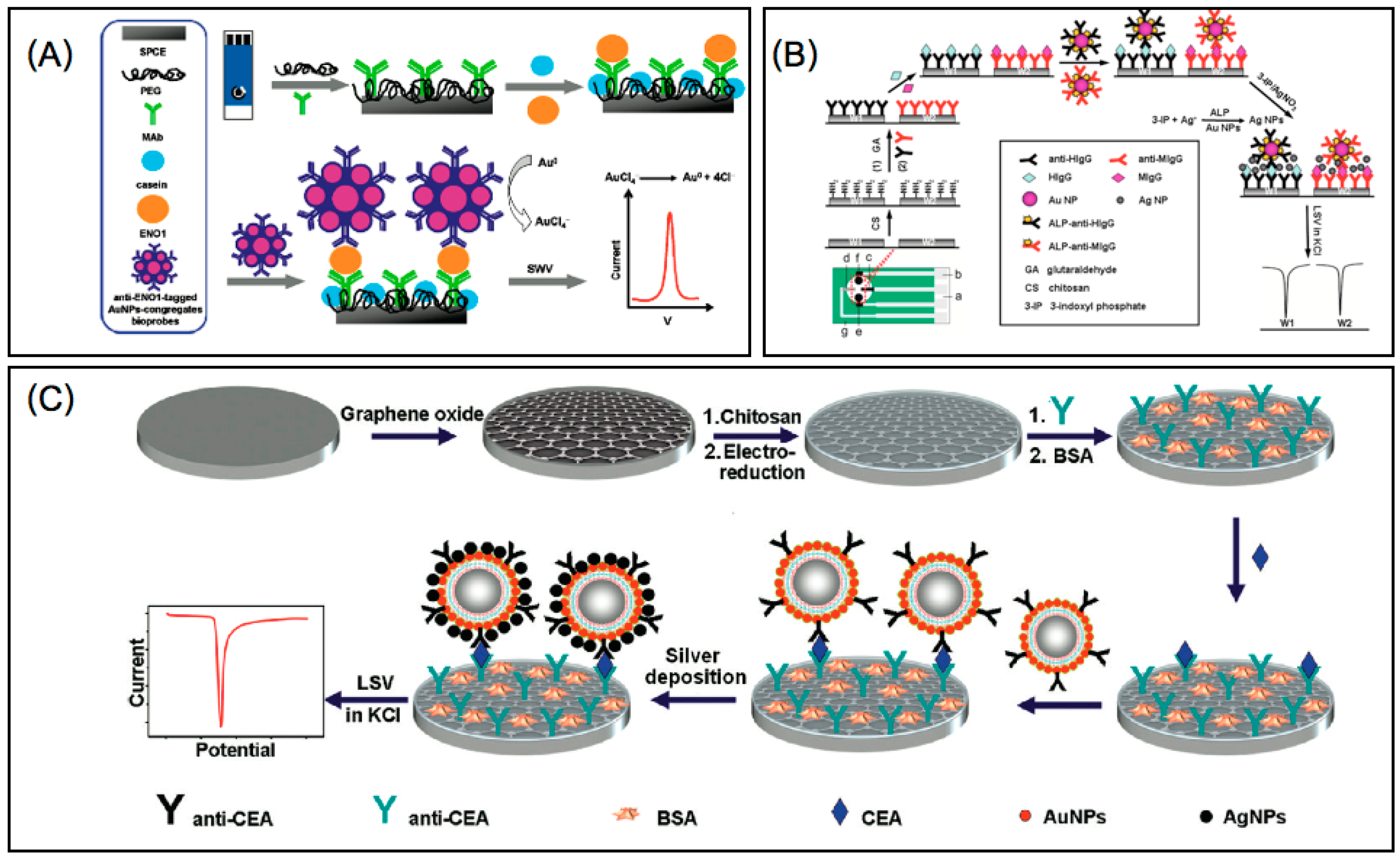
3.3. Antioxidants
3.3.1. Electrochemical Detection
3.3.2. Colorimetric and Spectrophotometric Detection
3.4. Cancer
3.4.1. Electrochemical Detection
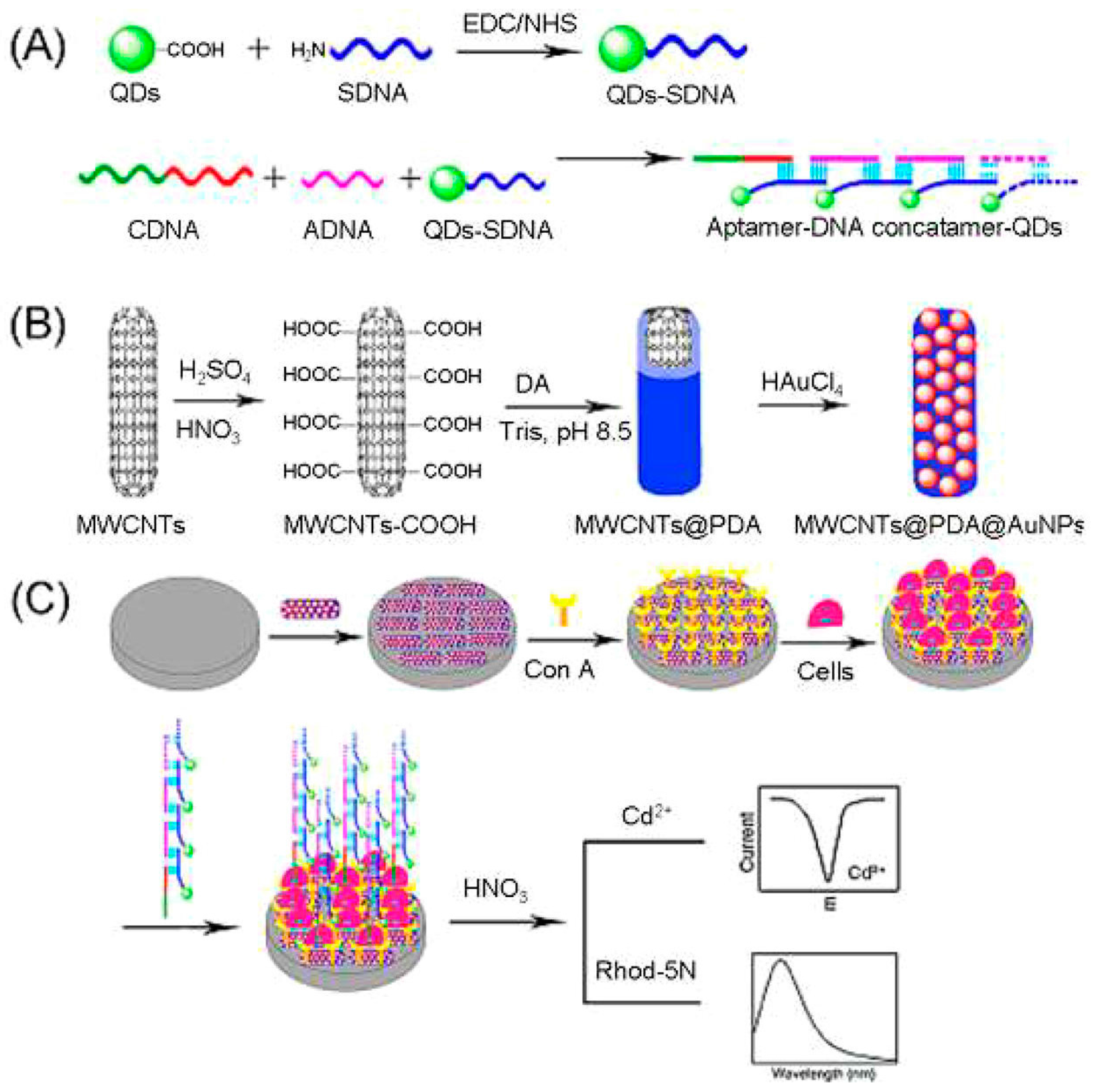
3.4.2. Colorimetric and Spectrophotometric Detection
3.5. Chemical Substances
3.5.1. Electrochemical Detection
3.5.2. Colorimetric and Spectrophotometric Detection
3.6. Hormones
3.6.1. Electrochemical Detection
3.6.2. Colorimetric and Spectrophotometric Detection
3.7. Lipids
3.7.1. Electrochemical Detection
3.7.2. Colorimetric and Spectrophotometric Detection
3.8. Microorganism
3.8.1. Electrochemical Detection
3.8.2. Colorimetric and Spectrophotometric Detection
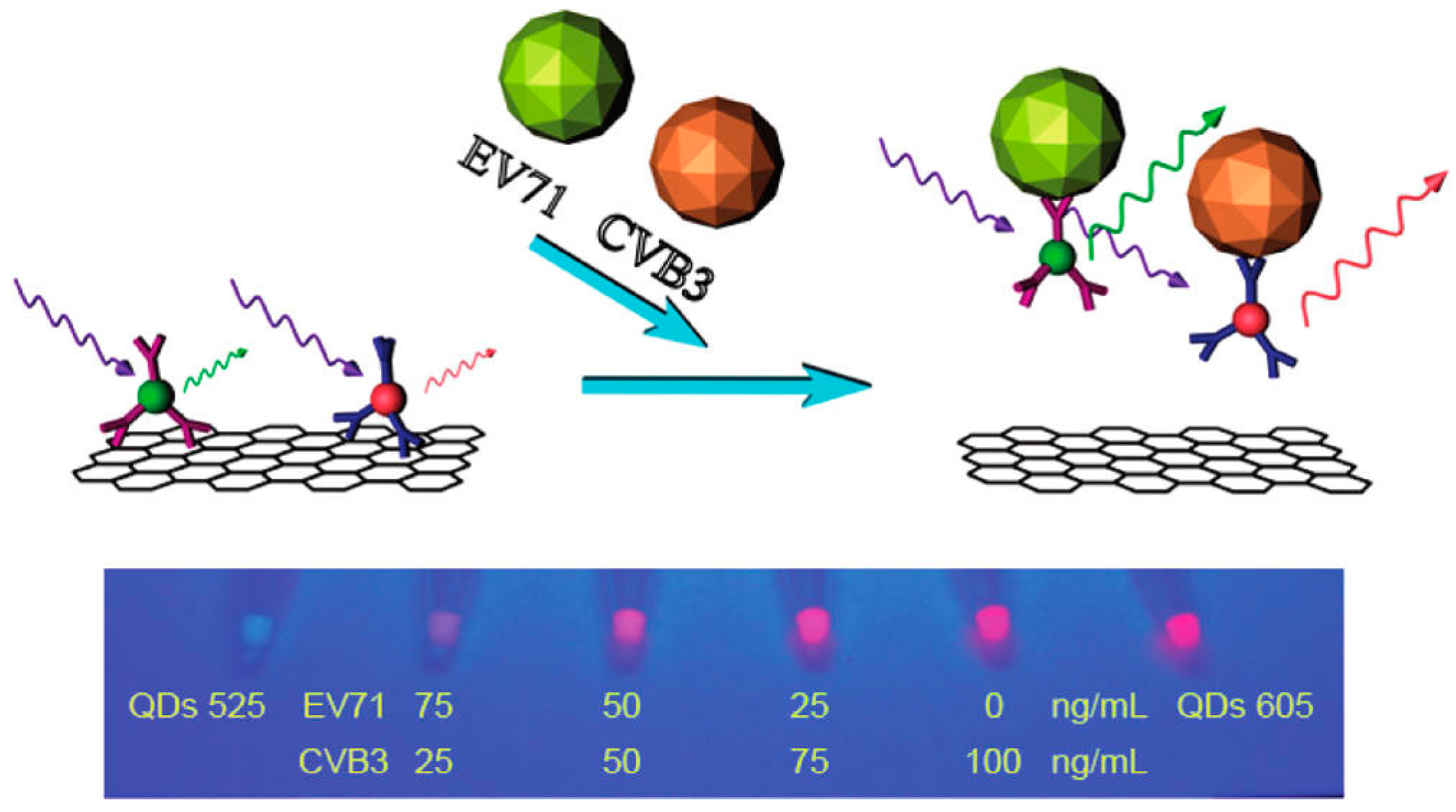
3.9. Neurotransmitters
3.9.1. Electrochemical Detection
3.9.2. Colorimetric and Spectrophotometric Detection
3.10. Nucleic Acids
3.10.1. Electrochemical Detection
3.10.2. Colorimetric and Spectrophotometric Detection
3.11. Proteins
3.11.1. Electrochemical Detection
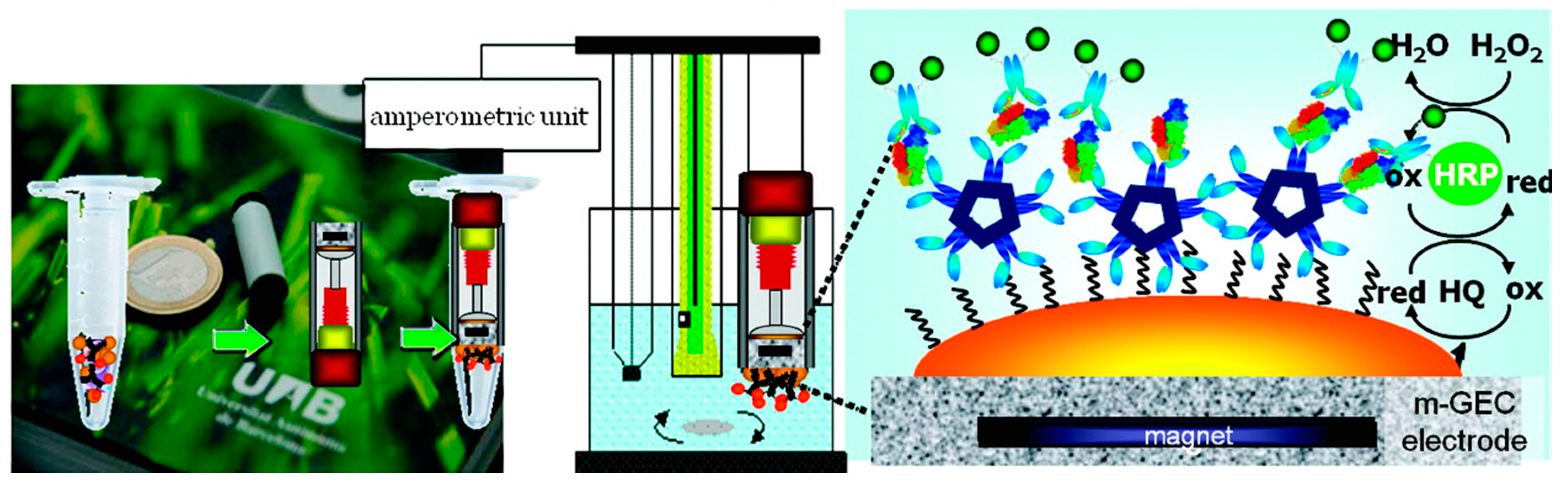
3.11.2. Colorimetric and Spectrophotometric Detection
3.12. Sugars
3.12.1. Electrochemical Detection
3.12.2. Colorimetric and Spectrophotometric Detection
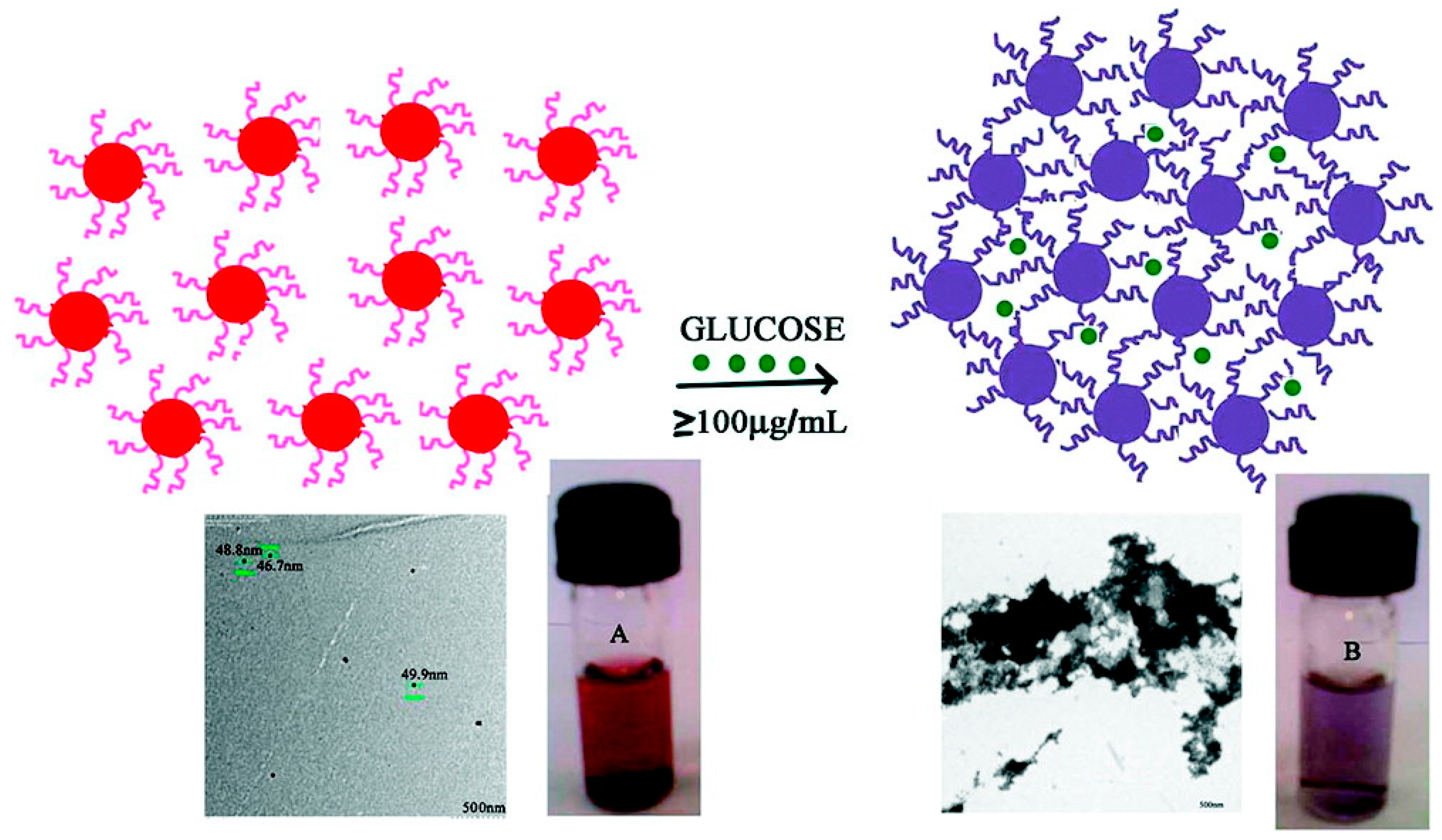
4. Analytical Performance
| Group of Analytes | Detection * | Materials | Analytes | Detection Limit | Linear Dynamic Range | Ref. |
|---|---|---|---|---|---|---|
| Amino acids | Amperometry | AuNP-CNT/GCE | Trpysin | 10 nM | 30 nM–2.5 μM | [23] |
| CV | Fe3O4-GO/GCE | Cysteine | 56 μM | 0.5–13.5 mM | [24] | |
| Acetaminophen | 25 μM | 0.12–13.3 mM | ||||
| DPV | AgNPs/GO/GCE | Trpysin | 2.0 nM | 0.01–50 μM | [25] | |
| 50.0–800.0 μM | ||||||
| CA | GC/DNA/NiOxNPs/Os(III)-complex electrode | Cysteine | 0.07 μM | up to 1000 μM | [21] | |
| Antigens-antibodies | SV | AuNPs | CEA | 0.12 pg·mL−1 | 0.5 pg·mL−1–0.5ng·mL−1 | [39] |
| Antioxidants | SWV | NiO/MWCNT | Glutathione | 0.006 μM | 0.01–200 μM | [49] |
| nanocomposite | Acetaminophen | 0.5 μM | 0.8–600 μM | |||
| CV | Fe2O3/RG nanocomposite | Ascorbic acid | 0.543 μM | 0.57–3.97 μM | [48] | |
| Amperometry | AgNPs/CMWCNT/PANI/Au electrode | Glutathoine | 0.3 μM | 0.3–3500 μM | [47] | |
| Cancer biomarkers | - | AuNP | Cancer-related gene sequence | 4 fM | 10 fM–1 nM | [58] |
| Voltammetry | Fe3O4 MNPs | Leukemia cells | 10 cells | - | [59] | |
| DPV | CdTe QDs/CGE | DNA sequences of bladder cancer cells | 6.435 × 10−13 M | 1 × 10−12–1 × 10−8 M | [61] | |
| Chemical substances | CV | Fe3O4-Au-NPs | Digoxin | 0.05 ng·mL−1 | 0.5–5 ng·mL−1 | [78] |
| CV/DPV/LSV | Fe3O4/GCE | Nimesulide | 1.3 × 10−7 M | 2.6 × 10−6–1.0 × 10−4 M | [10] | |
| DPV | AgNP/MWCNTs-COOH/GCE | Adriamycin | 1.7 × 10−9 M | 8.2 × 10−9–19.0 × 10−9 M | [76] | |
| Hormones | - | WS2/AuNPs/GCE | 17β-estradiol | 2 pM | 0.1 pM–5 nM | [93] |
| Amperometry | NiOPs/Nafion-MWCNTs/SPE | Insulin | 6.1 nM | 20–60 nM | [94] | |
| Lipids | Amperometry | AuE/dithiol/AuNPs/MUA/ ChOx | Cholesterol | 34.6 μM | 0.04–0.22 mM | [97] |
| CA | AgNPs/GCE | Cholesterol | 0.99 mg·dL−1 | 3.9–773.4 mg·dL−1 | [99] | |
| Microorganisms | EIS | EDC/NHS activated Au/1,6-HDT/AuNP/MUA | Type 5 adenovirus | 30 virus particle·mL−1 | 10–108 virus particle·mL−1 | [102] |
| Nurotransmitters | DPV | AuNP-CNTs/PGE | l-Dopa | 50 nM | 0.1–150 μM | [107] |
| DPV | NiFe2O4-MWCNT modified GCE | Dopamine | 0.02 μM | 0.05–6.0 μM 6.0–100 μM | [110] | |
| CV/LSV | RGO-Pd-NPs composite modified GCEs | Dopamine | 0.233 μM | 1–150 μM | [106] | |
| CV/DPA | PPyox-PTSA/Ag NP/Pt electrode | Dopamine | 0.58 nM | 1 × 10−9–1.2 × 10−7 M | [116] | |
| Nucleic acids | DPV | AuNPs-ATP-diazo-ATP/Au electrode | ssDNA | 9.10 × 10−11 M | 3.01× 10−10–1.32 × 10−8 M | [127] |
| Amperometry/CA/CV/DPV | Fe3O4/MWCNT/LDH/NAD+ modified GC electrode | NADH | 0.3 μM | Up to 300 μM | [129] | |
| Amperometry/CV | Au nanoparticle/rGO GCE | NADH | 1.13 nM | 50 nM–500 μM | [131] | |
| CV | CdSe | Adenine Guanine | 0.028 μM | 0.083–291 μM | [132] | |
| QDs-GO | 0.055 μM | 0.167–245 μM | ||||
| Nucleic acids | DPV | AgNPs-Pdop@Gr /GCE | DNA | 3.2 × 10−15 M | 1 × 10−13–1 × 10−8 M | [134] |
| CA | NiOxNPs/GC | NADH | 106 nM | Up to 1 mM | [136] | |
| Proteins | DPV | AuMNPs-Apt1/thrombin/Apt2-AuNPs-HRP modified Au electrode | Thrombin | 30 fM | 0.1–60 pM | [146] |
| DPV | MNPs/CS-MWCNTs | Bovine serum albumin | 2.8 × 10−11 g·mL−1 | 1.0 × 10−4–1.0 × 10−10 g·mL−1 | [7] | |
| Sugars | CA | AuNPs/GONRs/CS | Glucose | 0.5 μM | 2 μM–1.375 mM | [162] |
| 1.375–15 mM | ||||||
| Amperometry | MSENs/MGCE | Glucose | 0.2 μM | 0.5 μM–3.5 mM | [9] | |
| Amperometry | PVP-GNs-NiNPs-CS | Glucose | 30 nM | 0.1 μM–0.5 mM | [171] | |
| Amperometry | GOD-CS/AgNWs/ GCE | Glucose | 2.83 μM | 10 μM–0.8 mM | [177] | |
| Amperometry | CONM/GC | Glucose | 0.15 μM | 0.7–60 μM | [179] | |
| Amperometry | RGO-NiNPs/GCE | Glucose | 0.1 μM | 2 μM–2.1 mM | [184] | |
| Amperometry | Pd-MWCNTs | Glucose | 0.2 μM | 1–22 mM | [189] |
| Group of Analytes | Detections * | Materials | Analytes | Detection Limit | Linear Dynamic Range | Ref. |
|---|---|---|---|---|---|---|
| Amino acids | UV-Vis | CMC-AuNPs | Cysteine | ND | 10.0–100.0 μM | [27] |
| UV-Vis | Non-fluorosurfactant capped AgNPs | Cysteine | 0.05 μM | 1.5–6.0 μM | [16] | |
| SERS | AuNPs | Rabbit IgG | 1–10 ng·mL−1 | 0–100 ng·mL−1 | [40] | |
| Antigens-antibodies | SPR | Fe3O4/SiO2 and Fe3O4/Ag/SiO2 MNPs | Rabbit IgG | ND | 1.25–20 μg·mL−1 (for Fe3O4/SiO2) | [5] |
| 0.3–20 μg·mL−1 (for Fe3O4/Ag/SiO2) | ||||||
| ECL | Ag/graphene | CEA | 0.6 pg·mL−1 | 1 pg·mL−1–500 ng·mL−1 | [41] | |
| ECL | Si/CdTe/Ab2 | Rabbit IgG | 1.3 pg·mL−1 | 5 pg·mL−1–10 ng·mL−1 | [42] | |
| ECL | AgNPs-rGO-Ab2-GOD | CEA | 0.03 pg·mL−1 | 0.1 pg·mL−1–160 ng·mL−1 | [43] | |
| Fluorescence | CdS-2MPA | Rutin | 1.2 × 10−6 M | up to 4 × 10−5 M | [54] | |
| Antioxidants | Spectrophotometry | BSA-AgNCs | Ascorbic acid | 0.16 μM (3δ) | 2.0–50.0 μM | [55] |
| Colorimetric | Primer conjugated AuNPs | Human telomerase activity | 1 Hela cell·μL−1 | ND | [62] | |
| Cancer biomarkers | Colorimetric | Fe3O4 MNPs and PtNPs nanohybrids | Target cancer cells (breast cancer) | ND | ND | [67] |
| Wavelength-resolved imaging | Multiplexed QDs | Tumor cells in Hodgkin’s lymphoma | ND | ND | [69] | |
| Chemical substances | CL | l-cysteine capped CdS QDs | Baclofen | 0.0035 mg·L−1 | 0.012–24.0 mg·L−1 | [89] |
| Hormones | SPR | AuNPs-G4-OH SAM | Insulin | 0.5 pM | 2–43 pM | [95] |
| Fluorescence | Eu(III) chelated-bonded SiNPs | Human thyroid stimulating hormone | 0.0007 mL·UL−1 | 0.005–100 ml·UL−1 | [96] | |
| Lipids | ECL | AuNPs | Cholesterol | 5.7 nM | 0.17 nM–0.3 mM | [100] |
| ECL | Fe3O4@SiO2-Au@mpSiO2 | Cholesterol | 0.28 μM | 0.83–2.62 mM | [101] | |
| Microorganisms | CL | CMG-MNPs | HBV | 0.5 pM | ND | [103] |
| Fluorescence | Dual color QDs | Human enterovirus 71 | 0.42 ng·mL−1 (for EV71) | 1–14 ng·mL−1 (For EV 71) | [105] | |
| 0.39 ng·mL−1 (for CVB3) | 1–19 ng·mL−1 (for CVB3) | |||||
| Neurotransmitters | Fluorescence | AgNPs | 6-thioguanine | 9.7 nM | 1.5 × 10−8–7.5 × 10−7 M | [105] |
| Spectrophotometry | AuNPs | Dopamine | 33 nM | 33 nM–3.33 mM | [123] | |
| UV-Vis | CoxFe3-xO4 MNPs | Dopamine | 0.13 μM | 0.6–8.0 μM | [125] | |
| Nucleic acids | Colorimetric in flow strip | Oligonucleotides-modified AuNPs | miRNA | 1 fM (without silver enhancement) | ND | [140] |
| Fluorescence | NEase-amplified MNPs | p53 gene | 198 fM | ND | [142] | |
| CL | Oppy-PdNPs/Au | Sequence-specific DNA | 6.0 × 10−17 M | 1.0 × 10−16–1.0 × 10−15 M | [196] | |
| Proteins | Colorimetric | Fib-AuNPs | Thrombin | ND | 0.1–10 pM | [152] |
| UV-Vis | Fe3O4@AuNPs | Thrombin | 1.0 nM | 1.6–30.4 nM | [155] | |
| Proteins | Fluorescence | QD-apt nanoconjugates | Thrombin | 1 nM | nM–μM | [158] |
| Naked eyes | Apt-AgNPs | PDGF-BB | 1.56 ng·mL−1 | 1.56 ng·mL−1–100 ng·mL−1 | [29] | |
| Sugars | Colorimetric | AuNPs | Glucose | 10 μM (for pink assay) | Extended to 1.5 mM (for pink assay) | [191] |
| 5 μM (for blue assay) | Extended to 1.0 mM (for blue assay) | |||||
| Fluorescence | BiFeO3 MNPs | Glucose | 4.5 nM | 0.2 nM–0.2 μM | [193] | |
| Colorimetric | FPBA-AgNPs | Glucose | 89 μM | ND | [195] |
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Saha, K.; Agasti, S.S.; Kim, C.; Li, X.; Rotello, V.M. Gold nanoparticles in chemical and biological sensing. Chem. Rev. 2012, 112, 2739–2779. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Lei, C.; Nie, Z.; Guo, M.; Huang, Y.; Yao, S. Label-free fluorescent detection of thrombin activity based on a recombinant enhanced green fluorescence protein and nickel ions immobilized nitrilotriacetic acid-coated magnetic nanoparticles. Talanta 2013, 116, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Chen, L.; He, X.; Zhu, Y.; Zhang, X. Detection of polynucleotide kinase activity by using a gold electrode modified with magnetic microspheres coated with titanium dioxide nanoparticles and a DNA dendrimer. Analyst 2014, 139, 3895–3900. [Google Scholar] [CrossRef] [PubMed]
- Tang, L.; Casas, J.; Venkataramasubramani, M. Magnetic nanoparticle mediated enhancement of localized surface plasmon resonance for ultrasensitive bioanalytical assay in human blood plasma. Anal. Chem. 2013, 85, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sun, Y.; Wang, J.; Wang, J.; Yu, A.; Zhang, H.; Song, D. Preparation of surface plasmon resonance biosensor based on magnetic core/shell Fe3O4/SiO2 and Fe3O4/Ag/SiO2 nanoparticles. Coll. Surf. B Biointerfaces 2011, 84, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Cui, X.; Chen, J.; Zheng, X.; Liu, C.; Xue, T.; Wang, H.; Jin, Z.; Qiao, L.; Zheng, W. Well-dispersed palladium nanoparticles on graphene oxide as a non-enzymatic glucose sensor. RSC Adv. 2012, 2, 6245–6249. [Google Scholar] [CrossRef]
- Chen, H.J.; Zhang, Z.H.; Luo, L.J.; Yao, S.Z. Surface-imprinted chitosan-coated magnetic nanoparticles modified multi-walled carbon nanotubes biosensor for detection of bovine serum albumin. Sens. Actuators B Chem. 2012, 163, 76–83. [Google Scholar] [CrossRef]
- Rezaei, B.; Ensafi, A.A.; Haghighatnia, F.; Aalaye, S.E. Application of beta-cyclodextrin/MnFe2O4 magnetic nanoparticles as a catalyst for fast chemiluminescence determination of glutathione in human blood using luminol-diperiodatoargentate(III) system. J. Braz. Chem. Soc. 2012, 23, 2248–2257. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, C.; Zhang, J.; Huang, L. Development of magnetic single-enzyme nanoparticles as electrochemical sensor for glucose determination. Electrochim. Acta 2013, 111, 25–30. [Google Scholar] [CrossRef]
- Zhang, J.L.; Tan, X.C.; Zhao, D.D.; Tan, S.W.; Liu, L.; Wang, L.; Huang, Z.W. Fe3O4 magnetic nanoparticles modified electrode as a sensor for determination of nimesulide. Chem. Res. Chin. Univ. 2011, 27, 566–569. [Google Scholar]
- Wang, J.; Zhu, Z.; Munir, A.; Zhou, H.S. Fe3O4 nanoparticles-enhanced SPR sensing for ultrasensitive sandwich bio-assay. Talanta 2011, 84, 783–788. [Google Scholar] [CrossRef] [PubMed]
- Abu-Thabit, N.Y.; Makhlouf, A.S.H. Chapter 24—Recent Advances in Nanocomposite Coatings for Corrosion Protection Applications. In Handbook of Nanoceramic and Nanocomposite Coatings and Materials; Makhlouf, A.S.H., Scharnweber, D., Eds.; Elsevier: Boston, MA, USA, 2015; pp. 515–549. [Google Scholar]
- Camargo, P.H.C.; Satyanarayana, K.G.; Wypych, F. Nanocomposites: Synthesis, structure, properties and new application opportunities. Mater. Res. 2009, 12, 1–39. [Google Scholar] [CrossRef]
- Tomioka, K.; Fukui, T. 18—Growth of Semiconductor Nanocrystals. In Handbook of Crystal Growth (Second Edition); Nishinaga, T., Ed.; Elsevier: Boston, MA, USA, 2015; pp. 749–793. [Google Scholar]
- Rogach, A.L.; Talapin, D.V.; Weller, H. Semiconductor Nanoparticles. In Colloids and Colloid Assemblies; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2004; pp. 52–95. [Google Scholar]
- Chen, S.; Gao, H.; Shen, W.; Lu, C.; Yuan, Q. Colorimetric detection of cysteine using noncrosslinking aggregation of fluorosurfactant-capped silver nanoparticles. Sens. Actuatorsors B Chem. 2014, 190, 673–678. [Google Scholar] [CrossRef]
- Shahrokhian, S.; Nassab, N.H. Nanodiamond decorated with silver nanoparticles as a sensitive film modifier in a jeweled electrochemical sensor: Application to voltammetric determination of thioridazine. Electroanalysis 2013, 25, 417–425. [Google Scholar] [CrossRef]
- Ghosh, D.; Chattopadhyay, N. Gold and silver nanoparticles based superquenching of fluorescence: A review. J. Lumin. 2015, 160, 223–232. [Google Scholar] [CrossRef]
- Cesarino, I.; Galesco, H.V.; Machado, S.A.S. Determination of serotonin on platinum electrode modified with carbon nanotubes/polypyrrole/silver nanoparticles nanohybrid. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 40, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Parsons, B.J.; Spickett, C.M. Special issue on “Analytical methods for the detection of oxidized biomolecules and antioxidants”. Free Rad. Res. 2015, 49, 473–476. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, E.; Salimi, A.; Shams, E. DNA/nickel oxide nanoparticles/osmium(III)-complex modified electrode toward selective oxidation of 1-cysteine and simultaneous detection of 1-cysteine and homocysteine. Bioelectrochemistry 2012, 86, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Sattarahmady, N.; Heli, H. An electrocatalytic transducer for l-cysteine detection based on cobalt hexacyanoferrate nanoparticles with a core-shell structure. Anal. Biochem. 2011, 409, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Guo, S.; Fang, Y.; Dong, S. Gold nanoparticle/carbon nanotube hybrids as an enhanced material for sensitive amperometric determination of tryptophan. Electrochim. Acta 2010, 55, 3927–3931. [Google Scholar] [CrossRef]
- Song, Y.; He, Z.; Hou, H.; Wang, X.; Wang, L. Architecture of Fe3O4-graphene oxide nanocomposite and its application as a platform for amino acid biosensing. Electrochim. Acta 2012, 71, 58–65. [Google Scholar] [CrossRef]
- Li, J.; Kuang, D.; Feng, Y.; Zhang, F.; Xu, Z.; Liu, M.; Wang, D. Green synthesis of silver nanoparticles-graphene oxide nanocomposite and its application in electrochemical sensing of tryptophan. Biosens. Bioelectron. 2013, 42, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Karimi-Maleh, H.; Biparva, P.; Hatami, M. A novel modified carbon paste electrode based on NiO/CNTs nanocomposite and (9, 10-dihydro-9, 10-ethanoanthracene-11, 12-dicarboximido)-4-ethylbenzene-1, 2-diol as a mediator for simultaneous determination of cysteamine, nicotinamide adenine dinucleotide and folic acid. Biosens. Bioelectron. 2013, 48, 270–275. [Google Scholar] [PubMed]
- Wei, X.; Qi, L.; Tan, J.; Liu, R.; Wang, F. A colorimetric sensor for determination of cysteine by carboxymethyl cellulose-functionalized gold nanoparticles. Anal. Chim. Acta 2010, 671, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, G.; Yang, M.; Chen, L.C.; Xiong, X.L. Gold nanoparticle based signal enhancement liquid crystal biosensors for tyrosine assays. Sens. Actuators B Chem. 2015, 215, 152–158. [Google Scholar] [CrossRef]
- Hu, H.; Li, H.; Zhao, Y.; Dong, S.; Li, W.; Qiang, W.; Xu, D. Aptamer-functionalized silver nanoparticles for scanometric detection of platelet-derived growth factor-BB. Anal. Chim. Acta 2014, 812, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Cui, X.; Lv, Y.; Liu, Y.; Wu, B. Aqueous synthesis of near-infrared cdte quantum dots for biothiols detection in biological fluids. Acta Chim. Sin. 2014, 72, 75–82. [Google Scholar] [CrossRef]
- Wei, Y.; Li, H.; Hao, H.; Chen, Y.; Dong, C.; Wang, G. beta-Cyclodextrin functionalized Mn-doped ZnS quantum dots for the chiral sensing of tryptophan enantiomers. Polym. Chem. 2015, 6, 591–598. [Google Scholar] [CrossRef]
- Borase, H.P.; Patil, C.D.; Salunkhe, R.B.; Suryawanshi, R.K.; Kim, B.S.; Bapat, V.A.; Patil, S.V. Bio-functionalized silver nanoparticles: A novel colorimetric probe for cysteine detection. Appl. Biochem. Biotechnol. 2015, 175, 3479–3493. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Xu, K.; Liu, Q.; Liu, X.; Li, J. Colorimetric sensing of cysteine using label-free silver nanoparticles. Sens. Actuators B Chem. 2014, 202, 574–582. [Google Scholar] [CrossRef]
- Hajizadeh, S.; Farhadi, K.; Forough, M.; Molaei, R. Silver nanoparticles in the presence of Ca2+ as a selective and sensitive probe for the colorimetric detection of cysteine. Anal. Methods 2012, 4, 1747–1752. [Google Scholar] [CrossRef]
- Ravindran, A.; Mani, V.; Chandrasekaran, N.; Mukherjee, A. Selective colorimetric sensing of cysteine in aqueous solutions using silver nanoparticles in the presence of Cr3+. Talanta 2011, 85, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, F.; Han, C.; Cui, Z.; Xie, G.; Zhang, A. Highly sensitive and selective tryptophan colorimetric sensor based on 4,4-bipyridine-functionalized silver nanoparticles. Sens. Actuators B Chem. 2010, 145, 194–199. [Google Scholar] [CrossRef]
- Ho, J.A.A.; Chang, H.C.; Shih, N.Y.; Wu, L.C.; Chang, Y.F.; Chen, C.C.; Chou, C. Diagnostic detection of human lung cancer-associated antigen using a gold nanoparticle-based electrochemical immunosensor. Anal. Chem. 2010, 82, 5944–5950. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.; Yan, F.; Wu, J.; Leng, C.; Ju, H. Ultrasensitive multiplexed immunoassay with electrochemical stripping analysis of silver nanoparticles catalytically deposited by gold nanoparticles and enzymatic reaction. Anal. Chem. 2011, 83, 2726–2732. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Wu, J.; Wang, M.; Yan, F.; Ju, H. Triple signal amplification of graphene film, polybead carried gold nanoparticles as tracing tag and silver deposition for ultrasensitive electrochemical immunosensing. Anal. Chem. 2012, 84, 3662–3668. [Google Scholar] [CrossRef] [PubMed]
- Chon, H.; Lim, C.; Ha, S.M.; Ahn, Y.; Lee, E.K.; Chang, S.I.; Seong, G.H.; Choo, J. On-chip immunoassay using surface-enhanced raman scattering of hollow gold nanospheres. Anal. Chem. 2010, 82, 5290–5295. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liu, F.; Ge, S.; Yu, J.; Yan, M. Application of nanoporous Pd as catalytically promoted nanolabels for ultrasensitive electrochemiluminescence immunosensor based on Ag/graphene nanocomposite. Sens. Actuators B Chem. 2015, 210, 460–467. [Google Scholar] [CrossRef]
- Qian, J.; Zhang, C.; Cao, X.; Liu, S. Versatile immunosensor using a quantum dot coated silica nanosphere as a label for signal amplification. Anal. Chem. 2010, 82, 6422–6429. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Chai, Y.; Wang, H.; Yuan, R. Electrochemiluminescence of luminol enhanced by the synergetic catalysis of hemin and silver nanoparticles for sensitive protein detection. Biosens. Bioelectron. 2014, 54, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Lucas, J. Multienzymatic synthesis of nucleic acid derivatives: A general perspective. Appl. Microbiol. Biotechnol. 2015, 99, 4615–4627. [Google Scholar] [CrossRef] [PubMed]
- Matemadombo, F.; Apetrei, C.; Nyokong, T.; Rodriguez-Mendez, M.L.; de Saja, J.A. Comparison of carbon screen-printed and disk electrodes in the detection of antioxidants using CoPc derivatives. Sens. Actuators B Chem. 2012, 166, 457–466. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Tahernejad-Javazmi, F.; Ensafi, A.A.; Moradi, R.; Mallakpour, S.; Beitollahi, H. A high sensitive biosensor based on FePt/CNTs nanocomposite/N-(4-hydroxyphenyl)-3,5-dinitrobenzamide modified carbon paste electrode for simultaneous determination of glutathione and piroxicam. Biosens. Bioelectron. 2014, 60, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Narang, J.; Chauhan, N.; Jain, P.; Pundir, C.S. Silver nanoparticles/multiwalled carbon nanotube/polyaniline film for amperometric glutathione biosensor. Int. J. Biol. Macromol. 2012, 50, 672–678. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Li, H.; Lu, J.; Zhang, X.; Liu, N.; Zhang, X. Hydrothermal synthesis of Fe2O3/graphene nanocomposite for selective determination of ascorbic acid in the presence of uric acid. Electrochim. Acta 2015, 158, 264–270. [Google Scholar] [CrossRef]
- Shahmiri, M.R.; Bahari, A.; Karimi-Maleh, H.; Hosseinzadeh, R.; Mirnia, N. Ethynylferrocene-NiO/MWCNT nanocomposite modified carbon paste electrode as a novel voltammetric sensor for simultaneous determination of glutathione and acetaminophen. Sens. Actuators B Chem. 2013, 177, 70–77. [Google Scholar] [CrossRef]
- Li, F.; Chai, J.; Yang, H.; Han, D.; Niu, L. Synthesis of Pt/ionic liquid/graphene nanocomposite and its simultaneous determination of ascorbic acid and dopamine. Talanta 2010, 81, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, P.; Xu, H.; Zhang, H.; Zhong, X. Anti-aggregation of gold nanoparticle-based colorimetric sensor for glutathione with excellent selectivity and sensitivity. Analyst 2011, 136, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Tian, D.; Qian, Z.; Xia, Y.; Zhu, C. Gold nanocluster-based fluorescent probes for near-infrared and turn-on sensing of glutathione in living cells. Langmuir 2012, 28, 3945–3951. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Luo, F.; Yang, Y.; Lin, L.; Du, J.; Guo, Y.; Xiao, D.; Choi, M.M.F. CdS nanotubes thin film for electrochemiluminescence analysis of phenolic compounds. Anal. Methods 2012, 4, 1053–1059. [Google Scholar] [CrossRef]
- Carvalho, J.M.; Leandro, K.C.; da Silva, A.R.; Aucelio, R.Q. Selective determination of rutin by fluorescence attenuation of the cds-2-mercaptopropionic acid nanocrystal probe. Anal. Lett. 2013, 46, 207–224. [Google Scholar] [CrossRef]
- Yang, X.H.; Ling, J.; Peng, J.; Cao, Q.E.; Wang, L.; Ding, Z.T.; Xiong, J. Catalytic formation of silver nanoparticles by bovine serum albumin protected-silver nanoclusters and its application for colorimetric detection of ascorbic acid. Spectrochim. Acta Part. Mol. Biomol. Spectrosc. 2013, 106, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, L.; Zhu, L.; Jiang, J.; Tang, H. A surface-enhanced Raman scattering method for detection of trace glutathione on the basis of immobilized silver nanoparticles and crystal violet probe. Anal. Chim. Acta 2014, 816, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Jensen, G.C.; Krause, C.E.; Sotzing, G.A.; Rusling, J.F. Inkjet-printed gold nanoparticle electrochemical arrays on plastic. Application to immunodetection of a cancer biomarker protein. Phys. Chem. Chem. Phys. 2011, 13, 4888–4894. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Qiu, L.; Zhou, H.; Wu, Z.; Shen, G.; Yu, R. Sensitive and selective electrochemical DNA sensor for the analysis of cancer-related single nucleotide polymorphism. New J. Chem. 2014, 38, 4711–4715. [Google Scholar] [CrossRef]
- Zhang, K.; Tan, T.; Fu, J.J.; Zheng, T.; Zhu, J.J. A novel aptamer-based competition strategy for ultrasensitive electrochemical detection of leukemia cells. Analyst 2013, 138, 6323–6330. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Xu, S.; He, Z.; Deng, A.; Zhu, J.J. Supersandwich cytosensor for selective and ultrasensitive detection of cancer cells using aptamer-DNA concatamer-quantum dots probes. Anal. Chem. 2013, 85, 3385–3392. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.Y.; Hu, J. Single quantum dot-based nanosensor for multiple DNA detection. Anal. Chem. 2010, 82, 1921–1927. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wu, L.; Ren, J.; Qu, X. Visualizing human telomerase activity with primer-modified Au nanoparticles. Small 2012, 8, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.H.; Sim, S.J. Nanoplasmonic biosensor: Detection and amplification of dual bio-signatures of circulating tumor DNA. Biosens. Bioelectron. 2015, 67, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.Y.; Lai, L.J.; Ko, F.H. Rapid and sensitive detection of rare cancer cells by the coupling of immunomagnetic nanoparticle separation with ELISA analysis. Int. J. Nanomed. 2012, 7, 2967–2973. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, M.Z.; Chen, H.Y.; Wu, S.H.; Peng, S.W.; Lee, K.L.; Wei, P.K.; Cheng, J.Y. Magnetic nanoparticle-enhanced SPR on gold nanoslits for ultra-sensitive, label-free detection of nucleic acid biomarkers. Analyst 2013, 138, 2740–2748. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Wang, C.; Xiang, J.; Cheng, L.; Song, X.; Xu, L.; Peng, R.; Liu, Z. Aptamer-conjugated upconversion nanoprobes assisted by magnetic separation for effective isolation and sensitive detection of circulating tumor cells. Nano Res. 2014, 7, 1327–1336. [Google Scholar] [CrossRef]
- Kim, M.I.; Kim, M.S.; Woo, M.A.; Ye, Y.; Kang, K.S.; Lee, J.; Park, H.G. Highly efficient colorimetric detection of target cancer cells utilizing superior catalytic activity of graphene oxide-magnetic-platinum nanohybrids. Nanoscale 2014, 6, 1529–1536. [Google Scholar] [CrossRef] [PubMed]
- Jie, G.F.; Liu, P.; Zhang, S.S. Highly enhanced electrochemiluminescence of novel gold/silica/CdSe-CdS nanostructures for ultrasensitive immunoassay of protein tumor marker. Chem. Commun. 2010, 46, 1323–1325. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lau, S.K.; Varma, V.A.; Kairdolf, B.A.; Nie, S. Multiplexed detection and characterization of rare tumor cells in hodgkin’s lymphoma with multicolor quantum dots. Anal. Chem. 2010, 82, 6237–6243. [Google Scholar] [CrossRef] [PubMed]
- Jie, G.; Wang, L.; Yuan, J.; Zhang, S. Versatile electrochemiluminescence assays for cancer cells based on dendrimer/cdse-zns-quantum dot nanoclusters. Anal. Chem. 2011, 83, 3873–3880. [Google Scholar] [CrossRef] [PubMed]
- Karadas, N.; Bozal-Palabiyik, B.; Uslu, B.; Ozkan, S.A. Functionalized carbon nanotubes-with silver nanoparticles to fabricate a sensor for the determination of zolmitriptan in its dosage forms and biological samples. Sens. Actuators B Chem. 2013, 186, 486–494. [Google Scholar] [CrossRef]
- Kumary, V.A.; Divya, J.; Nancy, T.E.M.; Sreevalsan, K. Voltammetric detection of paracetamol at cobalt ferrite nanoparticles modified glassy carbon electrode. Int. J. Electrochem. Sci. 2013, 8, 6610–6619. [Google Scholar]
- Liu, G.T.; Chen, H.F.; Lin, G.M.; Ye, P.P.; Wang, X.P.; Jiao, Y.Z.; Guo, X.Y.; Wen, Y.; Yang, H.F. One-step electrodeposition of graphene loaded nickel oxides nanoparticles for acetaminophen detection. Biosens. Bioelectron. 2014, 56, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Razmi, H.; Habibi, E. Amperometric detection of acetaminophen by an electrochemical sensor based on cobalt oxide nanoparticles in a flow injection system. Electrochim. Acta 2010, 55, 8731–8737. [Google Scholar] [CrossRef]
- Liu, M.; Chen, Q.; Lai, C.; Zhang, Y.; Deng, J.; Li, H.; Yao, S. A double signal amplification platform for ultrasensitive and simultaneous detection of ascorbic acid, dopamine, uric acid and acetaminophen based on a nanocomposite of ferrocene thiolate stabilized Fe3O4@Au nanoparticles with graphene sheet. Biosens. Bioelectron. 2013, 48, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Zhang, Y. Electrochemical behavior of adriamycin at an electrode modified with silver nanoparticles and multi-walled carbon nanotubes, and its application. Microchim. Acta 2010, 169, 161–165. [Google Scholar] [CrossRef]
- Hasanzadeh, M.; Pournaghi-Azar, M.H.; Shadjou, N.; Jouyban, A. Magnetic nanoparticles incorporated on functionalized mesoporous silica: An advanced electrochemical sensor for simultaneous determination of amiodarone and atenolol. RSC Adv. 2014, 4, 4710–4717. [Google Scholar] [CrossRef]
- Ahmadi, A.; Shirazi, H.; Pourbagher, N.; Akbarzadeh, A.; Omidfar, K. An electrochemical immunosensor for digoxin using core-shell gold coated magnetic nanoparticles as labels. Mol. Biol. Rep. 2014, 41, 1659–1668. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Zhang, Z.; Zhang, H.; Luo, L.; Yao, S. A sensitive and selective sensor-coated molecularly imprinted sol-gel film incorporating beta-cyclodextrin-multi-walled carbon nanotubes and cobalt nanoparticles-chitosan for oxacillin determination. Surf. Interface Anal. 2012, 44, 334–341. [Google Scholar] [CrossRef]
- Rastgar, S.; Shahrokhian, S. Nickel hydroxide nanoparticles-reduced graphene oxide nanosheets film: Layer-by-layer electrochemical preparation, characterization and rifampicin sensory application. Talanta 2014, 119, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Parvin, M.H.; Golivand, M.B.; Najafi, M.; Shariaty, S.M. Carbon paste electrode modified with cobalt nanoparticles and its application to the electrocatalytic determination of chlorpromazine. J. Electroanal. Chem. 2012, 683, 31–36. [Google Scholar] [CrossRef]
- De Oliveira, P.R.; Oliveira, M.M.; Zarbin, A.J.G.; Marcolino-Junior, L.H.; Bergamini, M.F. Flow injection amperometric determination of isoniazid using a screen-printed carbon electrode modified with silver hexacyanoferrates nanoparticles. Sens. Actuators B Chem. 2012, 171, 795–802. [Google Scholar] [CrossRef]
- Miao, P.; Han, K.; Sun, H.; Yin, J.; Zhao, J.; Wang, B.; Tang, Y. Melamine functionalized silver nanoparticles as the probe for electrochemical sensing of clenbuterol. ACS Appl. Mater. Interfaces 2014, 6, 8667–8672. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Lin, R.; Li, L.; Jiang, L.; Ye, B.; He, H.; Qiu, L. Label-free silver nanoparticles for the naked eye detection of entecavir. Spectrochim. Acta Part Mol. Biomol. Spectrosc. 2014, 126, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.J.; Dong, C.X.; Chen, J.; Zhang, T.Y.; Zhang, Y.P.; Bai, L.Y. Preparation of a chemical sensor based on modified silver nanoparticles for quick recognition of 5-fluorocytosine. Asian J. Chem. 2012, 24, 3695–3698. [Google Scholar]
- Rastegarzadeh, S.; Hashemi, F. A surface plasmon resonance sensing method for determining captopril based on in situ formation of silver nanoparticles using ascorbic acid. Spectrochim. Acta Part Mol. Biomol. Spectrosc. 2014, 122, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Laliwala, S.K.; Mehta, V.N.; Rohit, J.V.; Kailasa, S.K. Citrate-modified silver nanoparticles as a colorimetric probe for simultaneous detection of four triptan-family drugs. Sens. Actuators B Chem. 2014, 197, 254–263. [Google Scholar] [CrossRef]
- Algarra, M.; Campos, B.B.; Aguiar, F.R.; Rodriguez-Borges, J.E.; Esteves da Silva, J.C.G. Novel beta-cyclodextrin modified CdTe quantum dots as fluorescence nanosensor for acetylsalicylic acid and metabolites. Mater. Sci. Eng. C Mater. Biol. Appl. 2012, 32, 799–803. [Google Scholar] [CrossRef]
- Khataee, A.; Hasanzadeh, A.; Iranifam, M.; Joo, S.W. A novel flow-injection chemiluminescence method for determination of baclofen using l-cysteine capped CdS quantum dots. Sens. Actuators B Chem. 2015, 215, 272–282. [Google Scholar] [CrossRef]
- Krishna, A.S.; Radhakumary, C.; Sreenivasan, K. In vitro detection of calcium in bone by modified carbon dots. Analyst 2013, 138, 7107–7111. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; Zhang, Y.; Huang, H.; Zhang, Y.; Du, B.; Chen, F.; Zheng, Q.; He, X.; Wang, K. Conjugated polyelectrolyte-stabilized silver nanoparticles coupled with pyrene derivative for ultrasensitive fluorescent detection of iodide. Talanta 2015, 131, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Olowu, R.A.; Arotiba, O.; Mailu, S.N.; Waryo, T.T.; Baker, P.; Iwuoha, E. Electrochemical aptasensor for endocrine disrupting 17 beta-estradiol based on a poly(3,4-ethylenedioxylthiopene)-Gold nanocomposite platform. Sensors 2010, 10, 9872–9890. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.J.; Liu, Y.J.; Zhang, J.Z.; Liu, Y.M. A novel aptamer sensor based on layered tungsten disulfide nanosheets and Au nanoparticles amplification for 17 beta-estradiol detection. Anal. Methods 2014, 6, 8011–8017. [Google Scholar] [CrossRef]
- Rafiee, B.; Fakhari, A.R. Electrocatalytic oxidation and determination of insulin at nickel oxide nanoparticles-multiwalled carbon nanotube modified screen printed electrode. Biosens. Bioelectron. 2013, 46, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Frasconi, M.; Tortolini, C.; Botre, F.; Mazzei, F. Multifunctional Au nanoparticle dendrimer-based surface plasmon resonance biosensor and its application for improved insulin detection. Anal. Chem. 2010, 82, 7335–7342. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xia, X.; Xu, Y.; Ke, W.; Yang, W.; Li, Q. Application of europium(III) chelates-bonded silica nanoparticle in time-resolved immunofluorometric detection assay for human thyroid stimulating hormone. Anal. Chim. Acta 2012, 722, 95–99. [Google Scholar] [CrossRef] [PubMed]
- Saxena, U.; Chakraborty, M.; Goswami, P. Covalent immobilization of cholesterol oxidase on self-assembled gold nanoparticles for highly sensitive amperometric detection of cholesterol in real samples. Biosens. Bioelectron. 2011, 26, 3037–3043. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Bai, H.; Liu, Q.; Bao, J.; Han, M.; Dai, Z. A nonenzymatic cholesterol sensor constructed by using porous tubular silver nanoparticles. Biosens. Bioelectron. 2010, 25, 2356–2360. [Google Scholar] [CrossRef] [PubMed]
- Nantaphol, S.; Chailapakul, O.; Siangproh, W. Sensitive and selective electrochemical sensor using silver nanoparticles modified glassy carbon electrode for determination of cholesterol in bovine serum. Sens. Actuators B Chem. 2015, 207, 193–198. [Google Scholar] [CrossRef]
- Ou, X.; Tan, X.; Wei, S.; Chen, S.; Zhang, J.; Liu, X. Electrochemiluminescence biosensor for cholesterol detection based on AuNPs/l-cys-C-60 nanocomposites. Anal. Methods 2014, 6, 3804–3810. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, S.; Tan, X.; Zhong, X.; Yuan, D.; Cheng, Y. Highly sensitive electrochemiluminescence biosensors for cholesterol detection based on mesoporous magnetic core-shell microspheres. Biotechnol. Lett. 2014, 36, 1835–1841. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Tang, T.; Harrison, D.J.; Lee, W.E.; Jemere, A.B. A regenerating ultrasensitive electrochemical impedance immunosensor for the detection of adenovirus. Biosens. Bioelectron. 2015, 68, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Liang, W.; Si, J.; Li, Z.; He, N. Long spacer arm-functionalized magnetic nanoparticle platform for enhanced chemiluminescent detection of hepatitis B virus. J. Biomed. Nanotechnol. 2014, 10, 3610–3619. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.; Liang, W.; Jin, L.; Tang, Y.; Mou, X.; Shah, M.A.A.; Yang, H.; Deng, Y.; He, N.; Li, Z. Development of magnetic nanoparticles based nucleic acid extraction method and application in hepatitis c virus chemiluminescent detection. Sci. Adv. Mater. 2015, 7, 1233–1240. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, X.; Zhou, G.; Xiang, X.; Ji, X.; Zheng, Z.; He, Z.; Wang, H. Simultaneous determination of human enterovirus 71 and coxsackievirus b3 by dual-color quantum dots and homogeneous immunoassay. Anal. Chem. 2012, 84, 3200–3207. [Google Scholar] [CrossRef] [PubMed]
- Palanisamy, S.; Ku, S.; Chen, S.M. Dopamine sensor based on a glassy carbon electrode modified with a reduced graphene oxide and palladium nanoparticles composite. Microchim. Acta 2013, 180, 1037–1042. [Google Scholar] [CrossRef]
- Hu, G.; Chen, L.; Guo, Y.; Wang, X.; Shao, S. Selective determination of l-dopa in the presence of uric acid and ascorbic acid at a gold nanoparticle self-assembled carbon nanotube-modified pyrolytic graphite electrode. Electrochim. Acta 2010, 55, 4711–4716. [Google Scholar] [CrossRef]
- Wang, C.; Yuan, R.; Chai, Y.; Zhang, Y.; Hu, F.; Zhang, M. Au-nanoclusters incorporated 3-amino-5-mercapto-1,2,4-triazole film modified electrode for the simultaneous determination of ascorbic acid, dopamine, uric acid and nitrite. Biosens. Bioelectron. 2011, 30, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Arora, K.; Bahadur, D. Impedimetric biosensor based on magnetic nanoparticles for electrochemical detection of dopamine. Mater. Sci. Eng. B-Adv. Funct. Solid-State Mater. 2012, 177, 1531–1537. [Google Scholar] [CrossRef]
- Ensafi, A.A.; Arashpour, B.; Rezaei, B.; Allafchian, A.R. Voltammetric behavior of dopamine at a glassy carbon electrode modified with NiFe2O4 magnetic nanoparticles decorated with multiwall carbon nanotubes. Mater. Sci. Eng. C Mater. Biol. Appl. 2014, 39, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Wang, H.; Wang, X.; Miao, Z.; Chen, L.; Zhao, W.; Shan, M.; Chen, Q. Amperometric biosensors based on gold nanoparticles-decorated multiwalled carbon nanotubes-poly(diallyldimethylammonium chloride) biocomposite for the determination of choline. Sens. Actuators B Chem. 2010, 147, 593–598. [Google Scholar] [CrossRef]
- Zhang, F.; Li, Y.; Gu, Y.E.; Wang, Z.; Wang, C. One-pot solvothermal synthesis of a Cu2O/Graphene nanocomposite and its application in an electrochemical sensor for dopamine. Microchim. Acta 2011, 173, 103–109. [Google Scholar] [CrossRef]
- Li, J.; Yang, J.; Yang, Z.; Li, Y.; Yu, S.; Xu, Q.; Hu, X. Graphene-Au nanoparticles nanocomposite film for selective electrochemical determination of dopamine. Anal. Methods 2012, 4, 1725–1728. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, M.; Li, Y.; Cheng, S.; Chen, X.; Wang, J. Evaluation of electrochemically reduced gold nanoparticle-graphene nanocomposites for the determination of dopamine. Anal. Lett. 2015, 48, 1437–1453. [Google Scholar] [CrossRef]
- Wang, W.; Wang, W.; Davis, J.J.; Luo, X. Ultrasensitive and selective voltammetric aptasensor for dopamine based on a conducting polymer nanocomposite doped with graphene oxide. Microchim. Acta 2015, 182, 1123–1129. [Google Scholar] [CrossRef]
- Saha, S.; Sarkar, P.; Turner, A.P.F. Interference-free electrochemical detection of nanomolar dopamine using doped polypyrrole and silver nanoparticles. Electroanalysis 2014, 26, 2197–2206. [Google Scholar] [CrossRef]
- Kaur, B.; Pandiyan, T.; Satpati, B.; Srivastava, R. Simultaneous and sensitive determination of ascorbic acid, dopamine, uric acid, and tryptophan with silver nanoparticles-decorated reduced graphene oxide modified electrode. Coll. Surfaces B Biointerfaces 2013, 111, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Noroozifar, M.; Khorasani-Motlagh, M.; Taheri, A. Preparation of silver hexacyanoferrate nanoparticles and its application for the simultaneous determination of ascorbic acid, dopamine and uric acid. Talanta 2010, 80, 1657–1664. [Google Scholar] [CrossRef] [PubMed]
- Rafati, A.A.; Afraz, A.; Hajian, A.; Assari, P. Simultaneous determination of ascorbic acid, dopamine, and uric acid using a carbon paste electrode modified with multiwalled carbon nanotubes, ionic liquid, and palladium nanoparticles. Microchim. Acta 2014, 181, 1999–2008. [Google Scholar] [CrossRef]
- Figueiredo-Filho, L.C.S.; Silva, T.A.; Vicentini, F.C.; Fatibello-Filho, O. Simultaneous voltammetric determination of dopamine and epinephrine in human body fluid samples using a glassy carbon electrode modified with nickel oxide nanoparticles and carbon nanotubes within a dihexadecylphosphate film. Analyst 2014, 139, 2842–2849. [Google Scholar] [CrossRef] [PubMed]
- Babaei, A.; Taheri, A.R.; Farahani, I.K. Nanomolar simultaneous determination of levodopa and melatonin at a new cobalt hydroxide nanoparticles and multi-walled carbon nanotubes composite modified carbon ionic liquid electrode. Sens. Actuators B Chem. 2013, 183, 265–272. [Google Scholar] [CrossRef]
- Tao, Y.; Lin, Y.; Ren, J.; Qu, X. A dual fluorometric and colorimetric sensor for dopamine based on BSA-stabilized Au nanoclusters. Biosens. Bioelectron. 2013, 42, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, C.; Zhou, T.; Ma, H. Gold nanoparticle based colorimetric probe for dopamine detection based on the interaction between dopamine and melamine. Microchim. Acta 2015, 182, 1003–1008. [Google Scholar] [CrossRef]
- Leng, Y.; Xie, K.; Ye, L.; Li, G.; Lu, Z.; He, J. Gold-nanoparticle-based colorimetric array for detection of dopamine in urine and serum. Talanta 2015, 139, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Xu, Y.; Dong, Y.; Qi, L.; Qi, S.; Chen, H.; Chen, X. Visual and quantitative determination of dopamine based on CoxFe3-xO4 magnetic nanoparticles as peroxidase mimetics. J. Alloys Compd. 2014, 587, 74–81. [Google Scholar] [CrossRef]
- Li, H.; Wu, X. Silver nanoparticles-enhanced rare earth co-luminescence effect of Tb (III)-Y(III)-dopamine system. Talanta 2015, 138, 203–208. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Feng, Y.; Dong, P.; Tang, B. Gold nanoparticles modified electrode via a mercapto-diazoaminobenzene monolayer and its development in DNA electrochemical biosensor. Biosens. Bioelectron. 2010, 25, 2084–2088. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Zhao, W.; Xu, J.; Chen, H. Magnetic particles and cadmium sulfide nanoparticles tagging for signal-amplifying detection of nucleic acids. Sci. China Chem. 2011, 54, 1304–1310. [Google Scholar] [CrossRef]
- Teymourian, H.; Salimi, A.; Hallaj, R. Low potential detection of NADH based on Fe3O4 nanoparticles/multiwalled carbon nanotubes composite: Fabrication of integrated dehydrogenase-based lactate biosensor. Biosens. Bioelectron. 2012, 33, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Huang, K.J.; Niu, D.J.; Yang, C.P.; Jing, Q.S. TiO2-graphene nanocomposite for electrochemical sensing of adenine and guanine. Electrochim. Acta 2011, 56, 4685–4690. [Google Scholar] [CrossRef]
- Govindhan, M.; Amiri, M.; Chen, A. Au nanoparticle/graphene nanocomposite as a platform for the sensitive detection of NADH in human urine. Biosens. Bioelectron. 2015, 66, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Kalaivani, A.; Narayanan, S.S. Simultaneous determination of adenine and guanine using cadmium selenide quantum dots-graphene oxide nanocomposite modified electrode. J. Nanosci. Nanotechnol. 2015, 15, 4697–4705. [Google Scholar] [CrossRef]
- Dong, H.; Yan, F.; Ji, H.; Wong, D.K.Y.; Ju, H. Quantum-dot-functionalized poly(styrene-co-acrylic acid) microbeads: step-wise self-assembly, characterization, and applications for sub-femtomolar electrochemical detection of DNA hybridization. Adv. Funct. Mater. 2010, 20, 1173–1179. [Google Scholar] [CrossRef]
- Huang, K.J.; Liu, Y.J.; Wang, H.B.; Wang, Y.Y. A sensitive electrochemical DNA biosensor based on silver nanoparticles-polydopamine@graphene composite. Electrochim. Acta 2014, 118, 130–137. [Google Scholar] [CrossRef]
- Huang, K.J.; Wang, L.; Wang, H.B.; Gan, T.; Wu, Y.Y.; Li, J.; Liu, Y.M. Electrochemical biosensor based on silver nanoparticles-polydopamine-graphene nanocomposite for sensitive determination of adenine and guanine. Talanta 2013, 114, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, E.; Salimi, A.; Shams, E. Electrocatalytic activity of nickel oxide nanoparticles as mediatorless system for NADH and ethanol sensing at physiological pH solution. Biosens. Bioelectron. 2013, 45, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Hassan, K.M.; Hathoot, A.A.; Ashour, W.F.D.; Abdel-Azzem, M. Electrochemical and analytical applications for NADH detection at glassy carbon electrode modified with nickel nanoparticles dispersed on poly 1,5-diaminonaphthalene. J. Solid State Electrochem. 2015, 19, 1063–1072. [Google Scholar] [CrossRef]
- Noorbakhsh, A.; Salimi, A. Development of DNA electrochemical biosensor based on immobilization of ssDNA on the surface of nickel oxide nanoparticles modified glassy carbon electrode. Biosens. Bioelectron. 2011, 30, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Jyoti, A.; Pandey, P.; Singh, S.P.; Jain, S.K.; Shanker, R. Colorimetric detection of nucleic acid signature of shiga toxin producing escherichia coli using gold nanoparticles. J. Nanosci. Nanotechnol. 2010, 10, 4154–4158. [Google Scholar] [CrossRef] [PubMed]
- Hou, S.Y.; Hsiao, Y.L.; Lin, M.S.; Yen, C.C.; Chang, C.S. MicroRNA detection using lateral flow nucleic acid strips with gold nanoparticles. Talanta 2012, 99, 375–379. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.Y.; Chen, Y.C. Functional magnetic nanoparticle-based trapping and sensing approaches for label-free fluorescence detection of DNA. Talanta 2011, 86, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Gao, Z.F.; Kang, B.H.; Li, N.B.; Luo, H.Q. Sensitive mutant DNA biomarker detection based on magnetic nanoparticles and nicking endonuclease assisted fluorescence signal amplification. RSC. Adv. 2015, 5, 20020–20024. [Google Scholar] [CrossRef]
- Alula, M.T.; Yang, J. Photochemical decoration of silver nanoparticles on magnetic microspheres as substrates for the detection of adenine by surface-enhanced Raman scattering. Anal. Chim. Acta 2014, 812, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, L.; Qiu, T.; Cao, W. Chemiluminescent detection of DNA hybridization based on signal DNA probe modified with gold and cobalt nanoparticles. Int. J. Electrochem. Sci. 2011, 6, 5325–5336. [Google Scholar]
- Li, L.D.; Zhao, H.T.; Chen, Z.B.; Mu, X.J.; Guo, L. Aptamer biosensor for label-free impedance spectroscopy detection of thrombin based on gold nanoparticles. Sens. Actuators B Chem. 2011, 157, 189–194. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Y.; Li, H.; Wen, Y.; Fan, X.; Lin, F.; Tan, L.; Yao, S. Ultrasensitive electrochemical aptasensor for thrombin based on the amplification of aptamer-AuNPs-HRP conjugates. Biosens. Bioelectron. 2011, 26, 2297–2303. [Google Scholar] [CrossRef] [PubMed]
- Wen, T.; Zhu, W.; Xue, C.; Wu, J.; Han, Q.; Wang, X.; Zhou, X.; Jiang, H. Novel electrochemical sensing platform based on magnetic field-induced self-assembly of Fe3O4@Polyaniline nanoparticles for clinical detection of creatinine. Biosens. Bioelectron. 2014, 56, 180–185. [Google Scholar] [CrossRef] [PubMed]
- de Souza Castilho, M.; Laube, T.; Yamanaka, H.; Alegret, S.; Pividori, M.I. Magneto immunoassays for plasmodium falciparum histidine-rich protein 2 related to malaria based on magnetic nanoparticles. Anal. Chem. 2011, 83, 5570–5577. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Gou, X.; Yuan, R.; Chai, Y.; Zhuo, Y.; Ye, X.; Gan, X. Graphene-promoted 3,4,9,10-perylenetetracarboxylic acid nanocomposite as redox probe in label-free electrochemical aptasensor. Biosens. Bioelectron. 2011, 30, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; He, J.; Xu, W.; Gao, L.; Guo, Y.; Li, W.; Yu, C. A novel immunosensor for detection of beta-galactoside alpha-2, 6-sialyltransferase in serum based on gold nanoparticles loaded on Prussian blue-based hybrid nanocomposite film. Electrochim. Acta 2015, 156, 45–52. [Google Scholar] [CrossRef]
- Ibupoto, Z.H.; Jamal, N.; Khun, K.; Liu, X.; Willander, M. A potentiometric immunosensor based on silver nanoparticles decorated ZnO nanotubes, for the selective detection of D-dimer. Sens. Actuators B Chem. 2013, 182, 104–111. [Google Scholar] [CrossRef]
- Chen, C.K.; Huang, C.C.; Chang, H.T. Label-free colorimetric detection of picomolar thrombin in blood plasma using a gold nanoparticle-based assay. Biosens. Bioelectron. 2010, 25, 1922–1927. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Wang, P.; Zhao, Y.; Fan, A. Turn-on colorimetric sensor for ultrasensitive detection of thrombin using fibrinogen-gold nanoparticle conjugate. Analyst 2013, 138, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Li, B. A sensitive, label-free, aptamer-based biosensor using a gold nanoparticle-initiated chemiluminescence system. Chem. Eur. J. 2011, 17, 1642–1648. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.; Cai, S.; Zhang, P.; Peng, Y.; Chen, H.; Zhang, S.; Kong, J. Magnetic relaxation switch and colorimetric detection of thrombin using aptamer-functionalized gold-coated iron oxide nanoparticles. Anal. Chim. Acta 2011, 689, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Qi, F.; Zhou, H.; Jia, S.; Gao, Y.; Koh, K.; Yin, Y. Fe3O4@Au nanoparticles as a means of signal enhancement in surface plasmon resonance spectroscopy for thrombin detection. Sens. Actuators B Chem. 2015, 212, 505–511. [Google Scholar] [CrossRef]
- Yang, J.Y.; Lin, Y.J.; Su, M.Y.; Li, W.J.; Liu, M.Y. Use of magnetic nanoparticles and a microplate reader with fluorescence detection to detect C-reactive protein. J. Chin. Chem. Soc. 2014, 61, 221–226. [Google Scholar] [CrossRef]
- Chi, C.W.; Lao, Y.H.; Li, Y.S.; Chen, L.C. A quantum dot-aptamer beacon using a DNA intercalating dye as the FRET reporter: Application to label-free thrombin detection. Biosens. Bioelectron. 2011, 26, 3346–3352. [Google Scholar] [CrossRef] [PubMed]
- Tennico, Y.H.; Hutanu, D.; Koesdjojo, M.T.; Bartel, C.M.; Remcho, V.T. On-chip aptamer-based sandwich assay for thrombin detection employing magnetic beads and quantum dots. Anal. Chem. 2010, 82, 5591–5597. [Google Scholar] [CrossRef] [PubMed]
- Miao, P.; Liu, T.; Li, X.; Ning, L.; Yin, J.; Han, K. Highly sensitive, label-free colorimetric assay of trypsin using silver nanoparticles. Biosens. Bioelectron. 2013, 49, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Ismail, N.S.; Le, Q.H.; Yoshikawa, H.; Saito, M.; Tamiya, E. Development of non-enzymatic electrochemical glucose sensor based on graphene oxide nanoribbon—Gold nanoparticle hybrid. Electrochim. Acta 2014, 146, 98–105. [Google Scholar] [CrossRef]
- Zhong, G.X.; Zhang, W.X.; Sun, Y.M.; Wei, Y.Q.; Lei, Y.; Peng, H.P.; Liu, A.L.; Chen, Y.Z.; Lin, X.H. A nonenzymatic amperometric glucose sensor based on three dimensional nanostructure gold electrode. Sens. Actuators B Chem. 2015, 212, 72–77. [Google Scholar] [CrossRef]
- Kim, M.I.; Ye, Y.; Won, B.Y.; Shin, S.; Lee, J.; Park, H.G. A highly efficient electrochemical biosensing platform by employing conductive nanocomposite entrapping magnetic nanoparticles and oxidase in mesoporous carbon foam. Adv. Funct. Mater. 2011, 21, 2868–2875. [Google Scholar] [CrossRef]
- Krishna, R.; Titus, E.; Chandra, S.; Bardhan, N.K.; Krishna, R.; Bahadur, D.; Gracio, J. Fabrication of a glucose biosensor based on citric acid assisted cobalt ferrite magnetic nanoparticles. J. Nanosci. Nanotechnol. 2012, 12, 6631–6638. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.J.; Li, Y.F.; Li, Z.H.; Feng, J.J.; Sun, Y.L.; Chen, J.R. Amperometric glucose sensor based on enhanced catalytic reduction of oxygen using glucose oxidase adsorbed onto core-shell Fe3O4@silica@Au magnetic nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2012, 32, 1640–1647. [Google Scholar] [CrossRef] [PubMed]
- Bo, X.; Bai, J.; Yang, L.; Guo, L. The nanocomposite of PtPd nanoparticles/onion-like mesoporous carbon vesicle for nonenzymatic amperometric sensing of glucose. Sens. Actuators B Chem. 2011, 157, 662–668. [Google Scholar] [CrossRef]
- Jiang, X.; Wu, Y.; Mao, X.; Cui, X.; Zhu, L. Amperometric glucose biosensor based on integration of glucose oxidase with platinum nanoparticles/ordered mesoporous carbon nanocomposite. Sens. Actuators B Chem. 2011, 153, 158–163. [Google Scholar] [CrossRef]
- Luo, L.; Zhu, L.; Wang, Z. Nonenzymatic amperometric determination of glucose by CuO nanocubes-graphene nanocomposite modified electrode. Bioelectrochemistry 2012, 88, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Qiao, N.; Zheng, J. Nonenzymatic glucose sensor based on glassy carbon electrode modified with a nanocomposite composed of nickel hydroxide and graphene. Microchim. Acta 2012, 177, 103–109. [Google Scholar] [CrossRef]
- Xiao, X.; Zhou, B.; Zhu, L.; Xu, L.; Tan, L.; Tang, H.; Zhang, Y.; Xie, Q.; Yao, S. An reagentless glucose biosensor based on direct electrochemistry of glucose oxidase immobilized on poly(methylene blue) doped silica nanocomposites. Sens. Actuators B Chem. 2012, 165, 126–132. [Google Scholar] [CrossRef]
- Liu, Z.; Guo, Y.; Dong, C. A high performance nonenzymatic electrochemical glucose sensor based on polyvinylpyrrolidone-graphene nanosheets-nickel nanoparticles-chitosan nanocomposite. Talanta 2015, 137, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Senel, M. Simple method for preparing glucose biosensor based on in-situ polypyrrole cross-linked chitosan/glucose oxidase/gold bionanocomposite film. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 48, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Shahnavaz, Z.; Lorestani, F.; Meng, W.P.; Alias, Y. Core-shell-CuFe2O4/PPy nanocomposite enzyme-free sensor for detection of glucose. J. Solid State Electrochem. 2015, 19, 1223–1233. [Google Scholar] [CrossRef]
- Xu, W.; He, J.; Gao, L.; Zhang, J.; Hui, J.; Guo, Y.; Li, W.; Yu, C. A sensitive glucose biosensor based on the abundant immobilization of glucose oxidase on hollow Pt nanospheres assembled on graphene oxide-Prussian Blue-PTC-NH2 nanocomposite film. J. Electroanal. Chem. 2015, 741, 8–13. [Google Scholar]
- Wang, L.; Tang, Y.; Wang, L.; Zhu, H.; Meng, X.; Chen, Y.; Sun, Y.; Yang, X.J.; Wan, P. Fast conversion of redox couple on Ni(OH)2/C nanocomposite electrode for high-performance nonenzymatic glucose sensor. J. Solid State Electrochem. 2015, 19, 851–860. [Google Scholar] [CrossRef]
- Chen, L.; Xie, H.; Li, J. Electrochemical glucose biosensor based on silver nanoparticles/multiwalled carbon nanotubes modified electrode. J. Solid State Electrochem. 2012, 16, 3323–3329. [Google Scholar] [CrossRef]
- Wang, L.; Gao, X.; Jin, L.; Wu, Q.; Chen, Z.; Lin, X. Amperometric glucose biosensor based on silver nanowires and glucose oxidase. Sens. Actuators B Chem. 2013, 176, 9–14. [Google Scholar] [CrossRef]
- Joshi, A.C.; Markad, G.B.; Haram, S.K. Rudimentary simple method for the decoration of graphene oxide with silver nanoparticles: Their application for the amperometric detection of glucose in the human blood samples. Electrochim. Acta 2015, 161, 108–114. [Google Scholar] [CrossRef]
- Sattarahmady, N.; Heli, H. A non-enzymatic amperometric sensor for glucose based on cobalt oxide nanoparticles. J. Exp. Nanosci. 2012, 7, 529–546. [Google Scholar] [CrossRef]
- Li, S.J.; Du, J.M.; Chen, J.; Mao, N.N.; Zhang, M.J.; Pang, H. Electrodeposition of cobalt oxide nanoparticles on reduced graphene oxide: A two-dimensional hybrid for enzyme-free glucose sensing. J. Solid State Electrochem. 2014, 18, 1049–1056. [Google Scholar] [CrossRef]
- Wang, T.; Yu, Y.; Tian, H.; Hu, J. A Novel non-enzymatic glucose sensor based on cobalt nanoparticles implantation-modified indium tin oxide electrode. Electroanalysis 2014, 26, 2693–2700. [Google Scholar] [CrossRef]
- Nie, H.; Yao, Z.; Zhou, X.; Yang, Z.; Huang, S. Nonenzymatic electrochemical detection of glucose using well-distributed nickel nanoparticles on straight multi-walled carbon nanotubes. Biosens. Bioelectron. 2011, 30, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.J.; Zhang, Y.H.; Xiao, P.; Li, X.L.; Meng, X.Q.; Yang, Y.N. An amperometric non-enzymatic glucose sensor by nickel hexacyanoferrate nanoparticles modified TiO2 nanotube arrays. Chin. J. Anal. Chem. 2012, 40, 313–316. [Google Scholar]
- Zhang, Y.; Xiao, X.; Sun, Y.; Shi, Y.; Dai, H.; Ni, P.; Hu, J.; Li, Z.; Song, Y.; Wang, L. Electrochemical deposition of nickel nanoparticles on reduced graphene oxide film for nonenzymatic glucose sensing. Electroanalysis 2013, 25, 959–966. [Google Scholar] [CrossRef]
- Kung, C.W.; Cheng, Y.H.; Ho, K.C. Single layer of nickel hydroxide nanoparticles covered on a porous Ni foam and its application for highly sensitive non-enzymatic glucose sensor. Sens. Actuators B Chem. 2014, 204, 159–166. [Google Scholar] [CrossRef]
- Chen, X.M.; Lin, Z.J.; Chen, D.J.; Jia, T.T.; Cai, Z.M.; Wang, X.R.; Chen, X.; Chen, G.N.; Oyama, M. Nonenzymatic amperometric sensing of glucose by using palladium nanoparticles supported on functional carbon nanotubes. Biosens. Bioelectron. 2010, 25, 1803–1808. [Google Scholar] [CrossRef] [PubMed]
- Gutes, A.; Carraro, C.; Maboudian, R. Nonenzymatic glucose sensing based on deposited palladium nanoparticles on epoxy-silver electrodes. Electrochim. Acta 2011, 56, 5855–5859. [Google Scholar] [CrossRef]
- Cai, Z.X.; Liu, C.C.; Wu, G.H.; Chen, X.M.; Chen, X. Palladium nanoparticles deposit on multi-walled carbon nanotubes and their catalytic applications for electrooxidation of ethanol and glucose. Electrochim. Acta 2013, 112, 756–762. [Google Scholar] [CrossRef]
- Singh, B.; Bhardwaj, N.; Jain, V.K.; Bhatia, V. Palladium nanoparticles decorated electrostatically functionalized MWCNTs as a non enzymatic glucose sensor. Sens. Actuators A Phys. 2014, 220, 126–133. [Google Scholar] [CrossRef]
- Radhakumary, C.; Sreenivasan, K. Naked eye detection of glucose in urine using glucose oxidase immobilized gold nanoparticles. Anal. Chem. 2011, 83, 2829–2833. [Google Scholar] [CrossRef] [PubMed]
- Palazzo, G.; Facchini, L.; Mallardi, A. Colorimetric detection of sugars based on gold nanoparticle formation. Sens. Actuatorsors B Chem. 2012, 161, 366–371. [Google Scholar] [CrossRef]
- Luo, W.; Li, Y.S.; Yuan, J.; Zhu, L.; Liu, Z.; Tang, H.; Liu, S. Ultrasensitive fluorometric determination of hydrogen peroxide and glucose by using multiferroic BiFeO3 nanoparticles as a catalyst. Talanta 2010, 81, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Z.G.; Li, J.P.; Tang, L.; Cheng, Z.Q. A novel electrochemiluminescence biosensor based on glucose oxidase immobized on magnetic nanoparticles. Chin. J. Anal. Chem. 2010, 38, 800–804. [Google Scholar] [CrossRef]
- Su, L.; Qin, W.; Zhang, H.; Rahman, Z.U.; Ren, C.; Ma, S.; Chen, X. The peroxidase/catalase-like activities of MFe2O4 (M = Mg, Ni, Cu) MNPs and their application in colorimetric biosensing of glucose. Biosens. Bioelectron. 2015, 63, 384–391. [Google Scholar] [CrossRef] [PubMed]
- Cao, K.; Jiang, X.; Yan, S.; Zhang, L.; Wu, W. Phenylboronic acid modified silver nanoparticles for colorimetric dynamic analysis of glucose. Biosens. Bioelectron. 2014, 52, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Yang, Q.; Wu, T.; Liu, L.; Ding, Y.; Yu, B. Fluorescence enhancement of cadmium selenide quantum dots assembled on silver nanoparticles and its application to glucose detection. Langmuir 2014, 30, 6324–6330. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakoh, A.; Pinyorospathum, C.; Siangproh, W.; Chailapakul, O. Biomedical Probes Based on Inorganic Nanoparticles for Electrochemical and Optical Spectroscopy Applications. Sensors 2015, 15, 21427-21477. https://doi.org/10.3390/s150921427
Yakoh A, Pinyorospathum C, Siangproh W, Chailapakul O. Biomedical Probes Based on Inorganic Nanoparticles for Electrochemical and Optical Spectroscopy Applications. Sensors. 2015; 15(9):21427-21477. https://doi.org/10.3390/s150921427
Chicago/Turabian StyleYakoh, Abdulhadee, Chanika Pinyorospathum, Weena Siangproh, and Orawon Chailapakul. 2015. "Biomedical Probes Based on Inorganic Nanoparticles for Electrochemical and Optical Spectroscopy Applications" Sensors 15, no. 9: 21427-21477. https://doi.org/10.3390/s150921427
APA StyleYakoh, A., Pinyorospathum, C., Siangproh, W., & Chailapakul, O. (2015). Biomedical Probes Based on Inorganic Nanoparticles for Electrochemical and Optical Spectroscopy Applications. Sensors, 15(9), 21427-21477. https://doi.org/10.3390/s150921427





