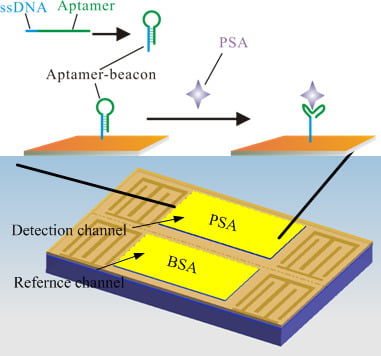
A Microfluidic Love-Wave Biosensing Device for PSA Detection Based on an Aptamer Beacon Probe
Abstract
:1. Introduction
2. Experimental Section
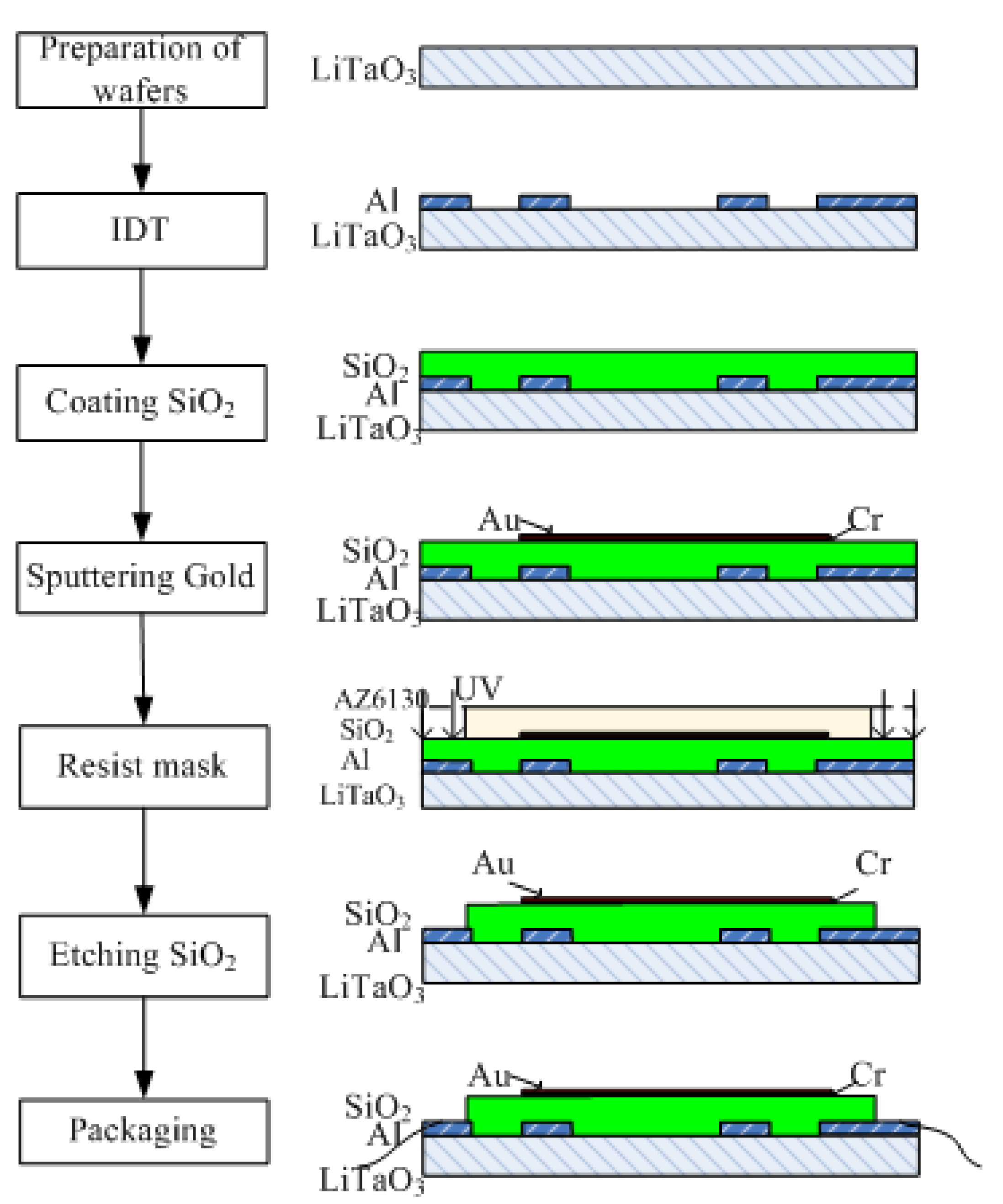
2.1. Fabrication of the Two-Channel Delay-Line Sensor Device
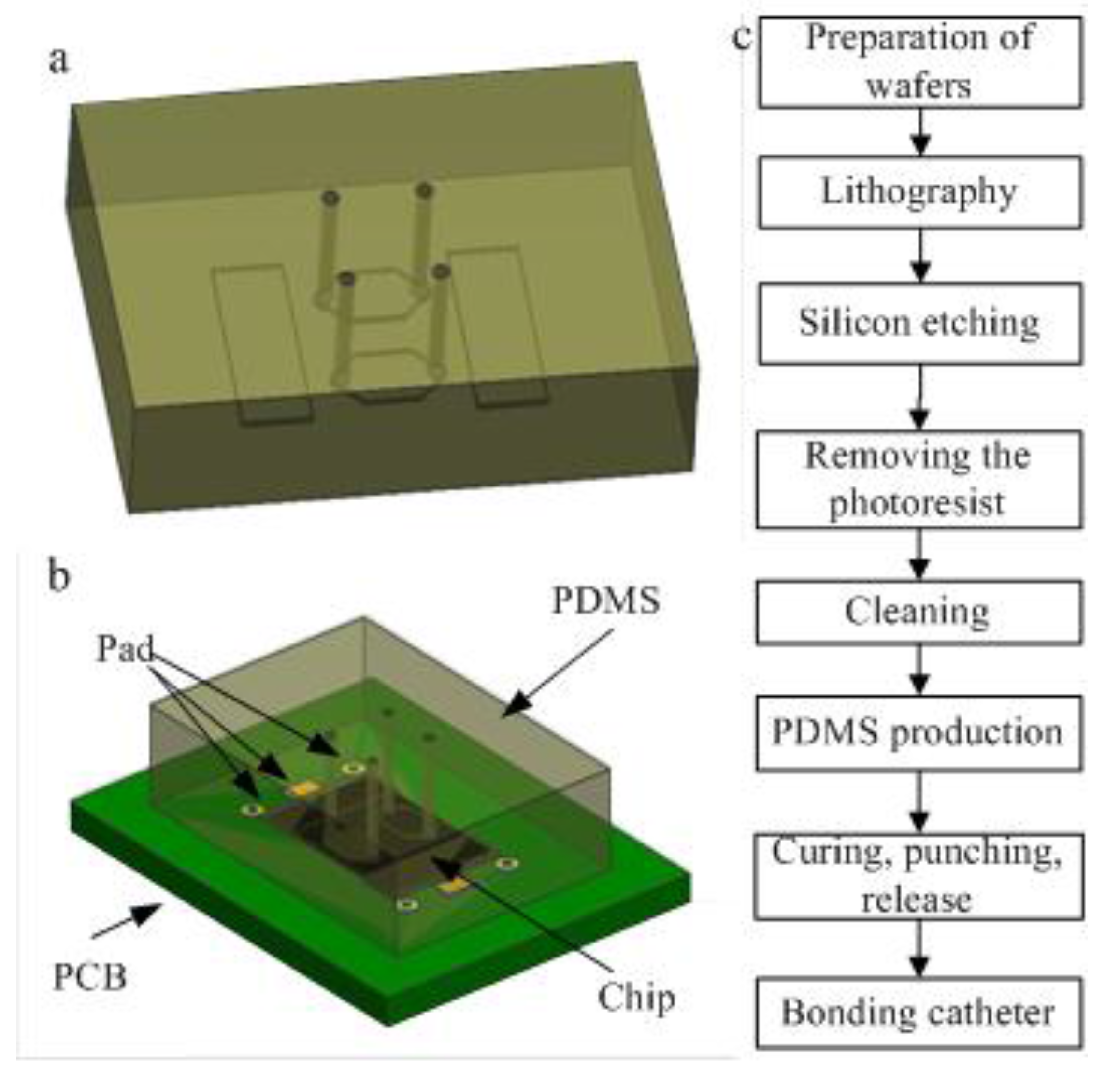
2.2. Fabrication of the PDMS Microfluidic Chip
2.3. Functionalization of the Sensor
2.4. PSA Detection Using the SAW Biosensor

3. Results and Discussion
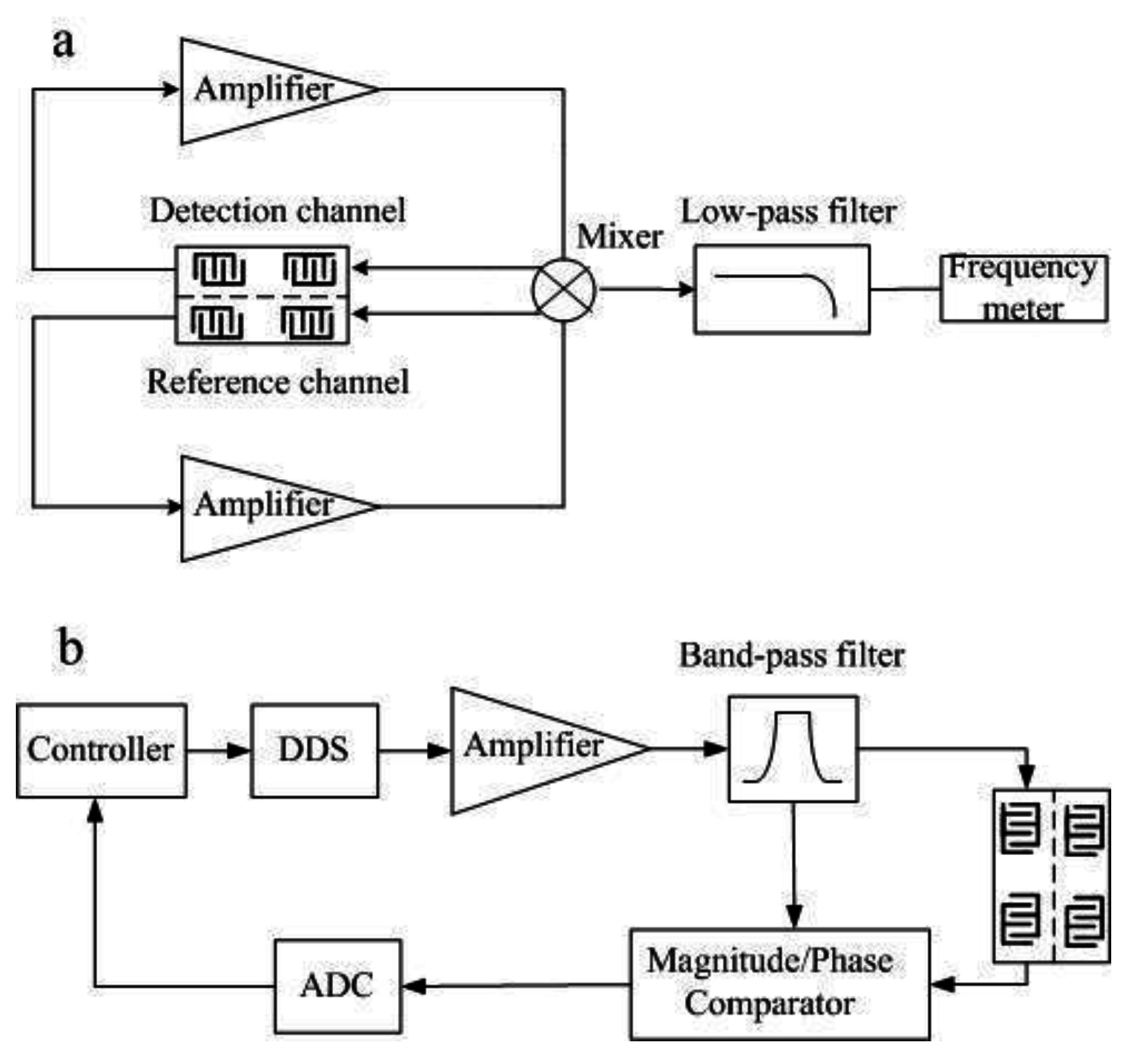
3.1. Two-Channel Delay-Line Sensor Device
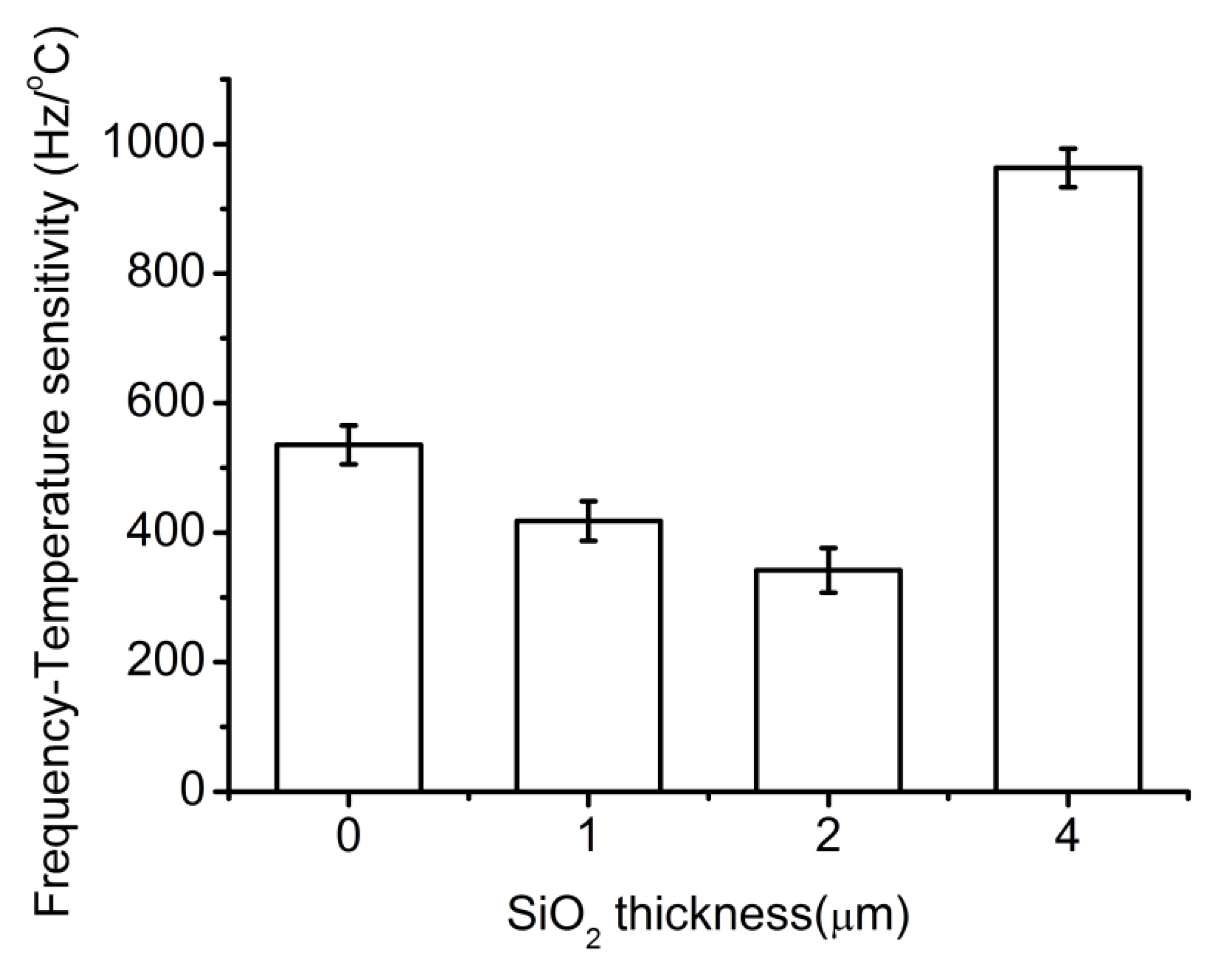
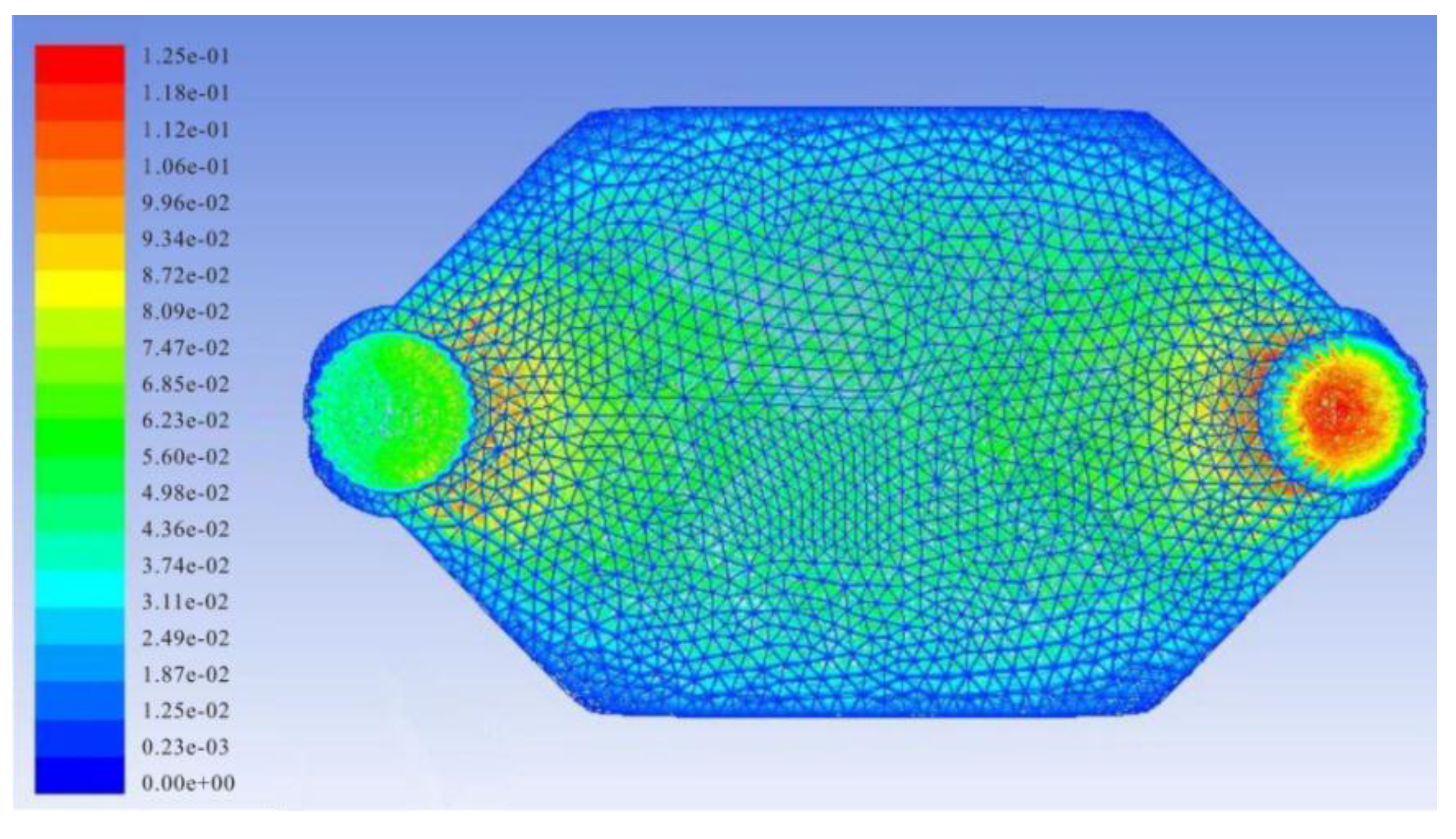
3.2. Simulation of Velocity Distribution of Fluid in PDMS Channel
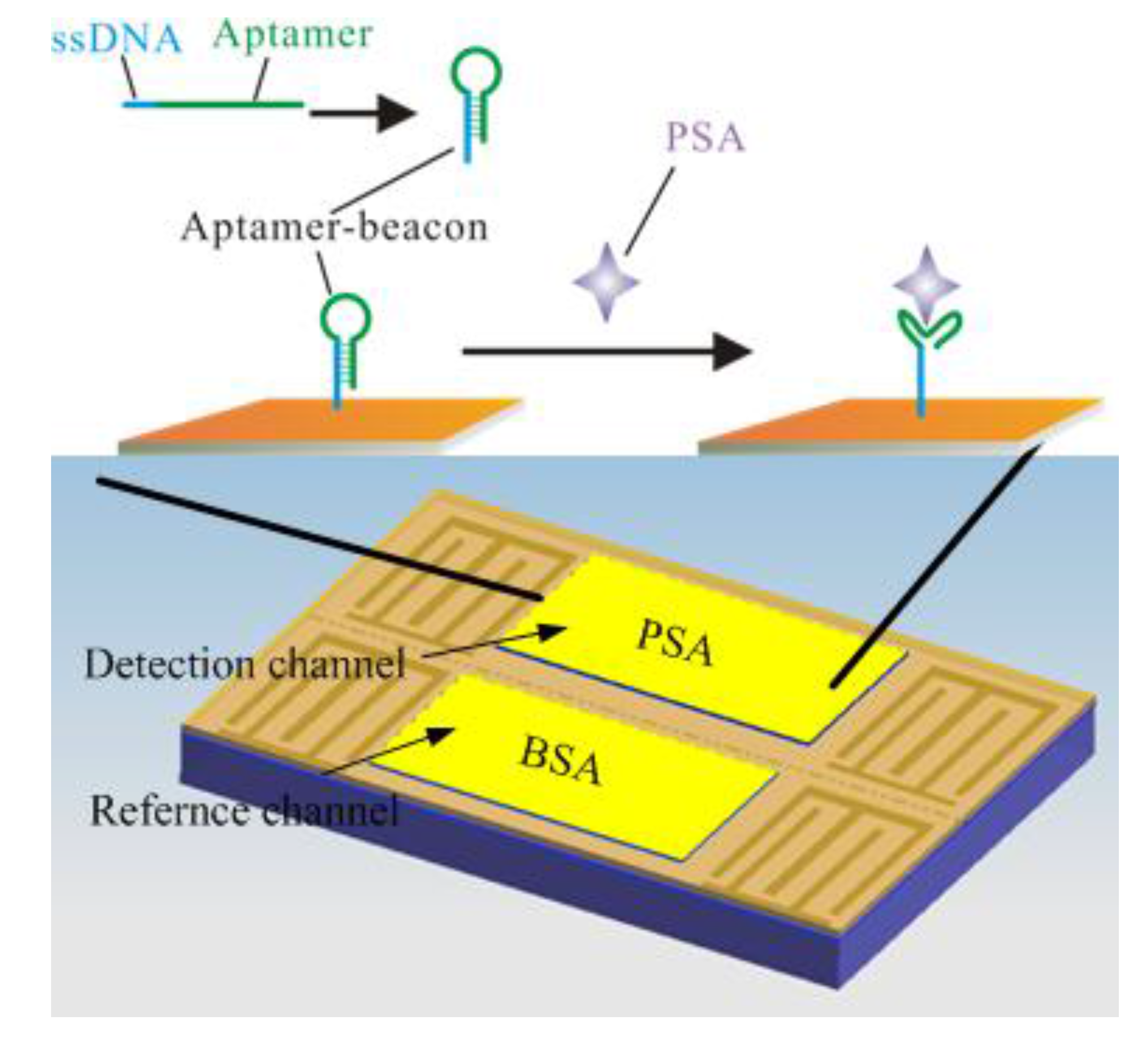
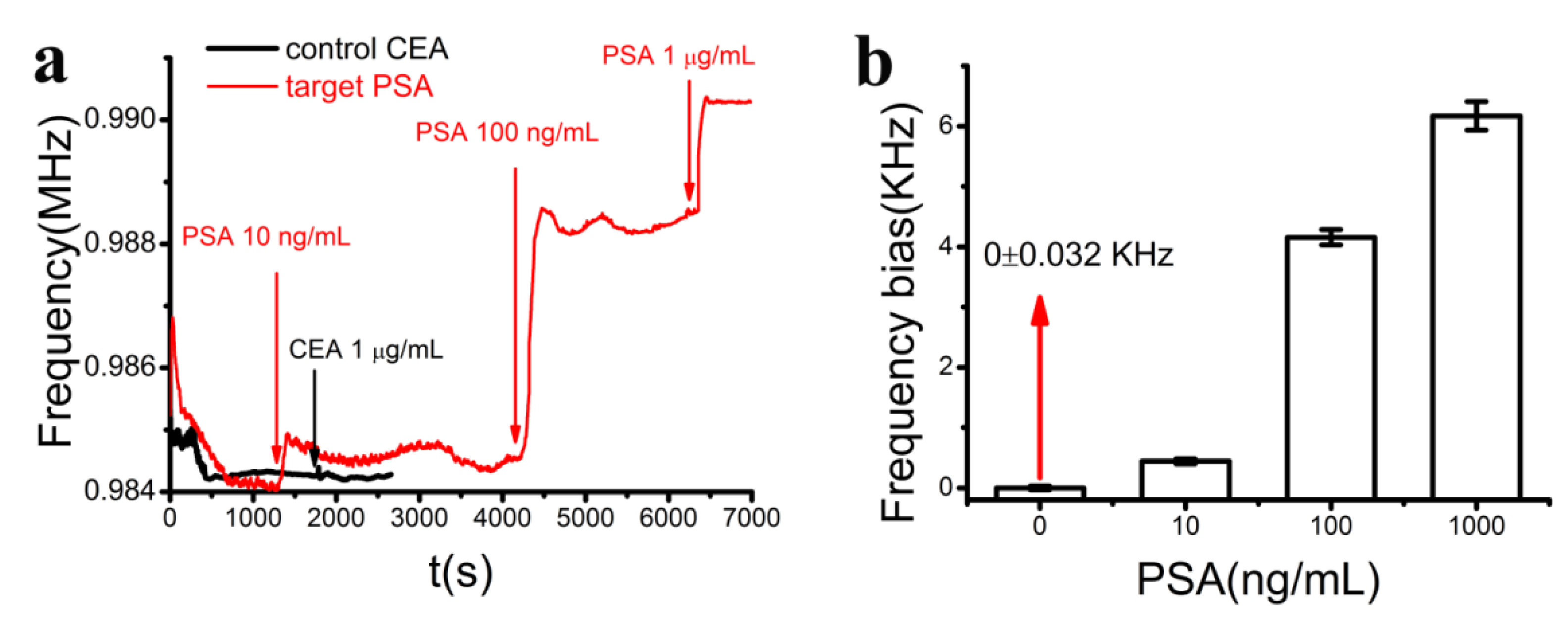
3.3. Detection of PSA
4. Conclusions/Outlook
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, Y.; Cheng, P.; Chan, D. A simple affinity spin tube filter method for removing high-abundant common proteins or enriching low-abundant biomarkers for serum proteomic analysis. Proteomics 2003, 3, 243–248. [Google Scholar]
- Butler, J. Enzyme-linked immunosorbent assay. J. Immunoassay. 2000, 21, 165–209. [Google Scholar] [PubMed]
- Kennedy, J.; Chappel, S. Direct pituitary effects of testosterone and luteinizing hormone-releasing hormone upon follicle-stim ulating hormone: Analysis by radioimmuno-and radioreceptor assay. Endocrinology 1985, 116, 741–748. [Google Scholar] [PubMed]
- Li, T.; Jo, E.; Kim, M. A label-free fluorescence immunoassay system for the sensitive detection of the mycotoxin, ochratoxin a. Chem. Commun. 2012, 48, 2304–2306. [Google Scholar]
- Koutny, L.; Schmalzing, D.; Taylor, T.; Fuchs, M. Microchip electrophoretic immunoassay for serum cortisol. Anal. Chem. 1996, 68, 18–22. [Google Scholar] [PubMed]
- Sherma, N.; Borges, C.; Trenchevska, O.; Jarvis, J.; Rehder, D.; Oran, P.; Nelson, R.; Nedelkov, D. Mass spectrometric immunoassay for the qualitative and quantitative analysis of the cytokine macrophage migration inhibitory factor (MIF). Proteome Sci. 2014. [Google Scholar] [CrossRef]
- Zhang, J.; Vernes, J.; Ni, J.; Nelson, C.; Wong, A.; Chen, S.; Asundi, A.; Vandlen, R.; Meng, Y. Real-time immuno-polymerase chain reaction in a 384-well format: Detection of vascular endothelial growth factor and epidermal growth factor-like domain 7. Anal. Biochem. 2014, 463, 61–66. [Google Scholar] [PubMed]
- Schweitzer, B.; Wiltshire, S.; Lambert, J.; O’Malley, S.; Kukanskis, K.; Zhu, Z.; Kingsmore, S.F.; Lizardi, P.M.; Ward, D.C. Immunoassays with rolling circle DNA amplification: A versatile platform for ultrasensitive antigen detection. Proc. Natl. Acad. Sci. 2000, 97, 10113–10119. [Google Scholar] [CrossRef] [PubMed]
- Mani, V.; Chikkaveeraiah, B.V.; Patel, V.; Gutkind, J.S.; Rusling, J.F. Ultrasensitive immunosensor for cancer biomarker proteins using gold nanoparticle film electrodes and multienzyme-particle amplification. ACS nano 2009, 3, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Deng, W.; Su, Y.; Zhu, X.; Peng, C.; Hu, H.; Peng, H.; Song, S.; Fan, C. Carbon nanotube-based ultrasensitive multiplexing electrochemical immunosensor for cancer biomarkers. Biosens. Bioelectron. 2011, 30, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Yan, J.; He, Y.; Lu, H.; Weng, L.; Song, S.; Fan, C.; Wang, L. Ultrasensitive, multiplexed detection of cancer biomarkers directly in serum by using a quantum dot-based microfluidic protein chip. ACS Nano 2009, 4, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Song, S.; Li, B.; Zhang, Q.; Huang, Q.; Zhang, H.; Fan, C. An on-nanoparticle rolling-circle amplification platform for ultrasensitive protein detection in biological fluids. Small 2010, 6, 2520–2525. [Google Scholar] [CrossRef] [PubMed]
- Länge, K.; Rapp, B.E.; Rapp, M. Surface acoustic wave biosensors: A review. Anal. Bioanal. Chem. 2008, 391, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Pi, Y.; Lu, W.; Wang, F.; Pan, F.; Li, F.; Jia, S.; Shi, J.; Deng, S.; Chen, M. Label-free and high-sensitive detection of human breast cancer cells by aptamer-based leaky surface acoustic wave biosensor array. Biosens. Bioelectron. 2014, 60, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Onen, O.; Sisman, A.; Gallant, N.D.; Kruk, P.; Guldiken, R. A urinary BCL-2 surface acoustic wave biosensor for early ovarian cancer detection. Sensors 2012, 12, 7423–7437. [Google Scholar] [CrossRef] [PubMed]
- Branch, D.W.; Brozik, S.M. Low-level detection of a bacillus anthracis simulant using Love-wave biosensors on 36° YX LiTaO3. Biosens. Bioelectron. 2004, 19, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Gronewold, T.M. Surface acoustic wave sensors in the bioanalytical field: Recent trends and challenges. Anal. Chim. Acta 2007, 603, 119–128. [Google Scholar]
- Harding, G.; Du, J.; Dencher, P.; Barnett, D.; Howe, E. Love wave acoustic immunosensor operating in liquid. Sens. Actuators A Phys. 1997, 61, 279–286. [Google Scholar] [CrossRef]
- Matatagui, D.; Fontecha, J.L.; Fernández, M.J.; Gràcia, I.; Cané, C.; Santos, J.P.; Horrillo, M.C. Love-wave sensors combined with microfluidics for fast detection of biological warfare agents. Sensors 2014, 14, 12658–12669. [Google Scholar] [CrossRef] [PubMed]
- Puiu, M.; Gurban, A.; Rotariu, L.; Brajnicov, S.; Viespe, C.; Bala, C. Enhanced sensitive Love wave surface acoustic wave sensor designed for immunoassay formats. Sensors 2015, 15, 10511–10525. [Google Scholar] [CrossRef] [PubMed]
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Farokhzad, O.; Jon, S.; Khademhosseini, A.; Tran, T.; LaVan, D.A.; Langer, R. Nanoparticle-aptamer bioconjugates a new approach for targeting prostate cancer cells. Cancer Res. 2004, 64, 7668–7672. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Frieder, J.; Teply, B.A.; Farokhzad, O. Aptamer conjugates: Emerging delivery platforms for targeted cancer therapy. In Drug Delivery Oncology: From Basic Research Cancer Therapy; Wiley: Weinheim, Germany, 2012; pp. 1263–1281. [Google Scholar]
- Liu, J.; Lu, Y. Fast colorimetric sensing of adenosine and cocaine based on a general sensor design involving aptamers and nanoparticles. Angew. Chem. 2006, 118, 96–100. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, L.; Pan, D.; Song, S.; Boey, F.Y.; Zhang, H.; Fan, C. Visual cocaine detection with gold nanoparticles and rationally engineered aptamer structures. Small 2008, 4, 1196–1200. [Google Scholar] [CrossRef] [PubMed]
- Bagalkot, V.; Zhang, L.; Levy-Nissenbaum, E.; Jon, S.; Kantoff, P.W.; Langer, R.; Farokhzad, O.C. Quantum dot-aptamer conjugates for synchronous cancer imaging, therapy, and sensing of drug delivery based on bi-fluorescence resonance energy transfer. Nano Lett. 2007, 7, 3065–3070. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, M.N.; de Prada, P.; Landry, D.W. Aptamer-based folding fluorescent sensor for cocaine. J. Am. Chem. Soc. 2001, 123, 4928–4931. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Zhu, T.; Qi, Y.; Zhao, Y.; Xia, H.; Fu, W. Development of a quartz crystal microbalance biosensor with aptamers as bio-recognition element. Sensors 2010, 10, 5859–5871. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.; Song, S.; Zhang, J.; Pan, D.; Wang, L.; Fan, C. A target-responsive electrochemical aptamer switch (TREAS) for reagentless detection of nanomolar ATP. J. Am. Chem. Soc. 2007, 129, 1042–1043. [Google Scholar] [CrossRef] [PubMed]
- Shu, H.; Wen, W.; Xiong, H.; Zhang, X.; Wang, S. Novel electrochemical aptamer biosensor based on gold nanoparticles signal amplification for the detection of carcinoembryonic antigen. Electrochem. Commun. 2013, 37, 15–19. [Google Scholar] [CrossRef]
- Schlensog, M.D.; Gronewold, T.M.; Tewes, M.; Famulok, M.; Quandt, E. A Love-wave biosensor using nucleic acids as ligands. Sens. Actuators B Chem. 2004, 101, 308–315. [Google Scholar] [CrossRef]
- Herrmann, F.; Weihnacht, M.; Buttgenbach, S. Properties of sensors based on shear-horizontal surface acoustic waves in LiTaO3/SiO2 and quartz/SiO2 structures. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2001, 48, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Lei, Y.; Chen, X.; Wang, Z.; Liu, J. An aptamer based resonance light scattering assay of prostate specific antigen. Biosens. Bioelectron. 2012, 36, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Flewitt, A.J. On-chip temperature-compensated love mode surface acoustic wave device for gravimetric sensing. Appl. Phys. Lett. 2014, 105, 213511–213514. [Google Scholar] [CrossRef]
- Tomar, M.; Gupta, V.; Mansingh, A.; Sreenivas, K. Temperature stability of c-axis oriented LiNbO3/SiO2/Si thin film layered structures. J. Phys. D Appl. Phys. 2001, 34, 2267–2273. [Google Scholar] [CrossRef]
- Hamaguchi, N.; Ellington, A.; Stanton, M. Aptamer beacons for the direct detection of proteins. Anal. Biochem. 2001, 294, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Song, S.; Fan, C. Target-responsive structural switching for nucleic acid-based sensors. Acc. Chem. Res. 2010, 43, 631–641. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Su, Y.; Zhu, X.; Yang, S.; Lu, J.; Gao, J.; Fan, C.; Huang, Q. Ultrasensitive electrochemical DNA sensor based on the target induced structural switching and surface-initiated enzymatic polymerization. Biosens. Bioelectron. 2014, 55, 231–236. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, F.; Li, S.; Cao, K.; Wang, P.; Su, Y.; Zhu, X.; Wan, Y. A Microfluidic Love-Wave Biosensing Device for PSA Detection Based on an Aptamer Beacon Probe. Sensors 2015, 15, 13839-13850. https://doi.org/10.3390/s150613839
Zhang F, Li S, Cao K, Wang P, Su Y, Zhu X, Wan Y. A Microfluidic Love-Wave Biosensing Device for PSA Detection Based on an Aptamer Beacon Probe. Sensors. 2015; 15(6):13839-13850. https://doi.org/10.3390/s150613839
Chicago/Turabian StyleZhang, Feng, Shuangming Li, Kang Cao, Pengjuan Wang, Yan Su, Xinhua Zhu, and Ying Wan. 2015. "A Microfluidic Love-Wave Biosensing Device for PSA Detection Based on an Aptamer Beacon Probe" Sensors 15, no. 6: 13839-13850. https://doi.org/10.3390/s150613839
APA StyleZhang, F., Li, S., Cao, K., Wang, P., Su, Y., Zhu, X., & Wan, Y. (2015). A Microfluidic Love-Wave Biosensing Device for PSA Detection Based on an Aptamer Beacon Probe. Sensors, 15(6), 13839-13850. https://doi.org/10.3390/s150613839