Surface Acoustic Wave (SAW) Resonators for Monitoring Conditioning Film Formation
Abstract
:1. Introduction

2. Experimental Section
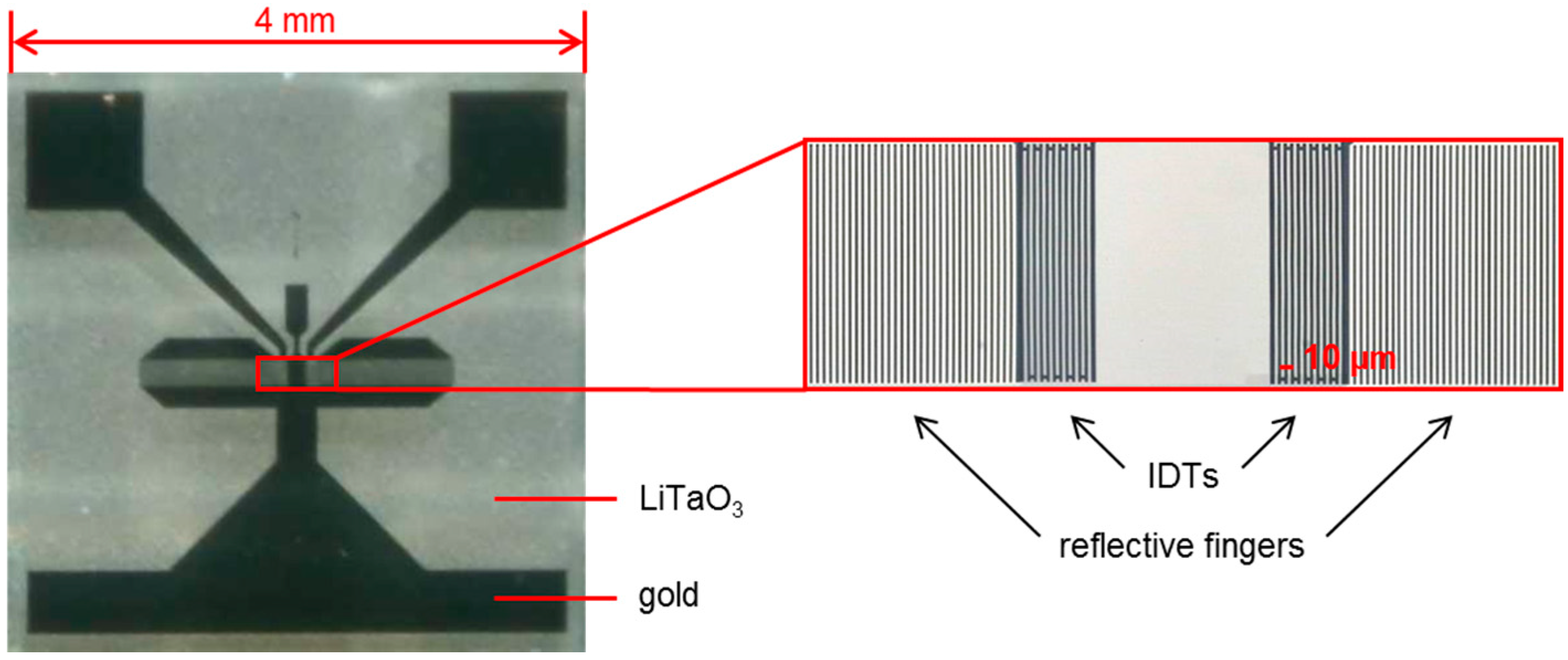
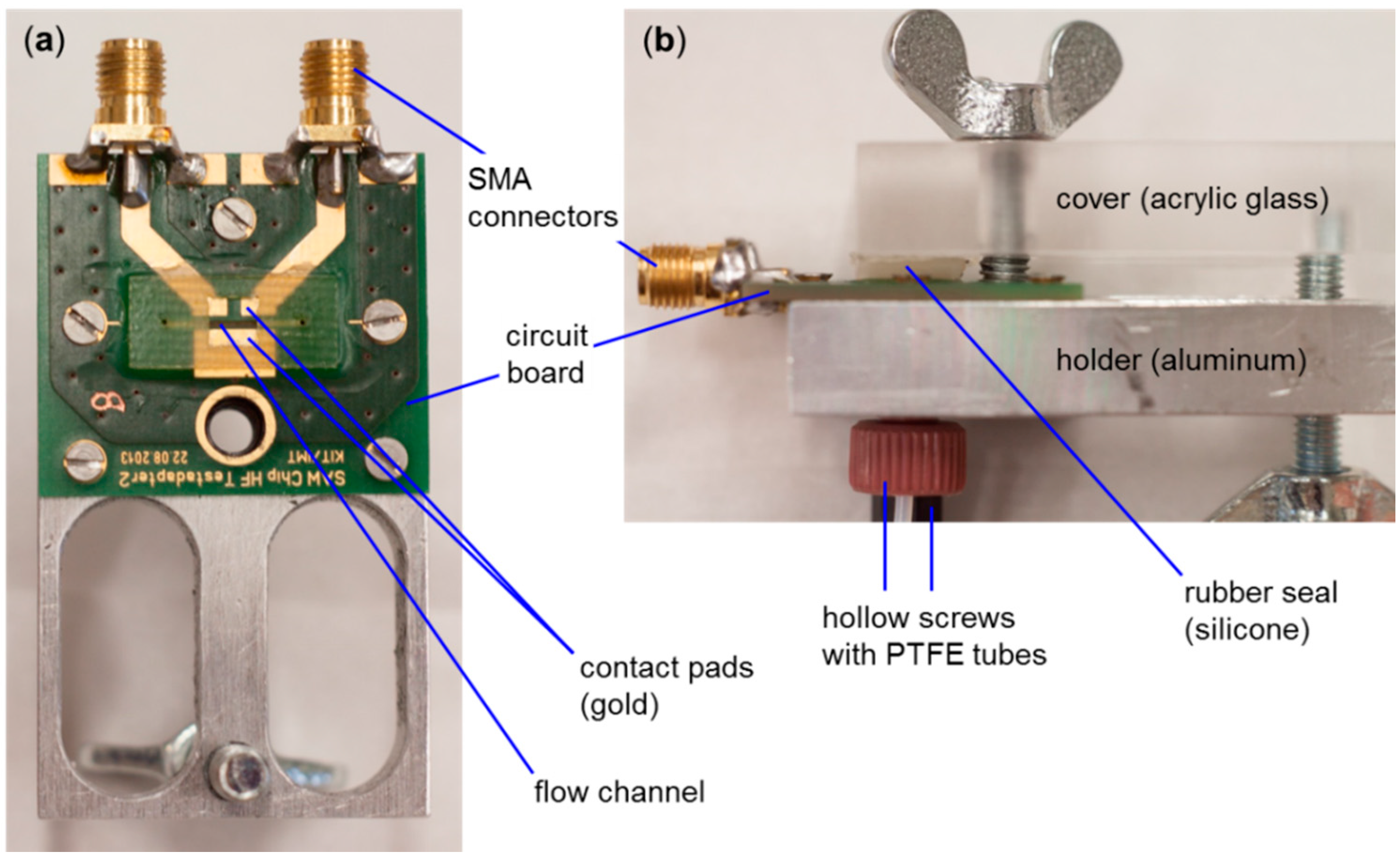
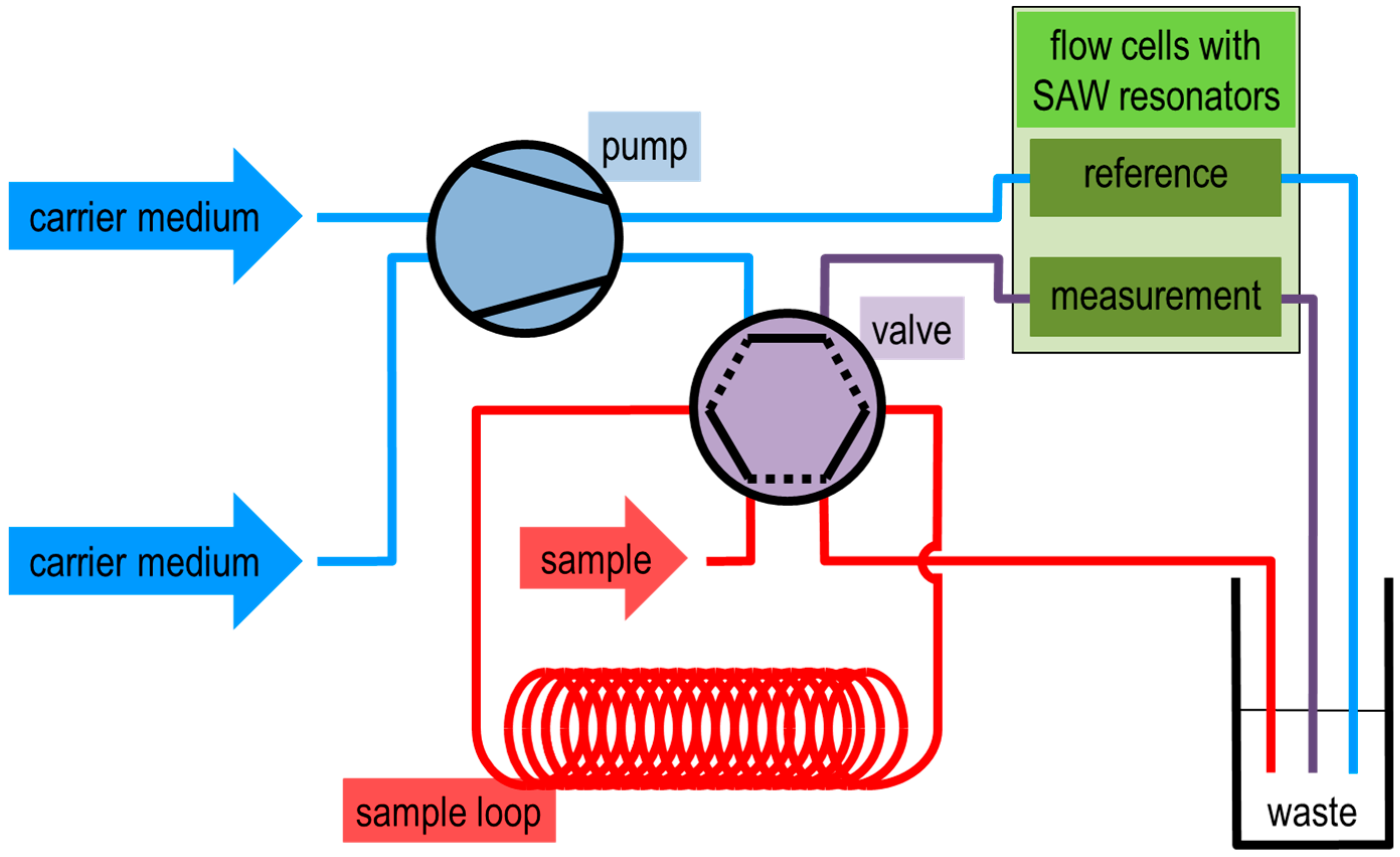
2.1. SAW Resonator (Sensor) Measurement Setup
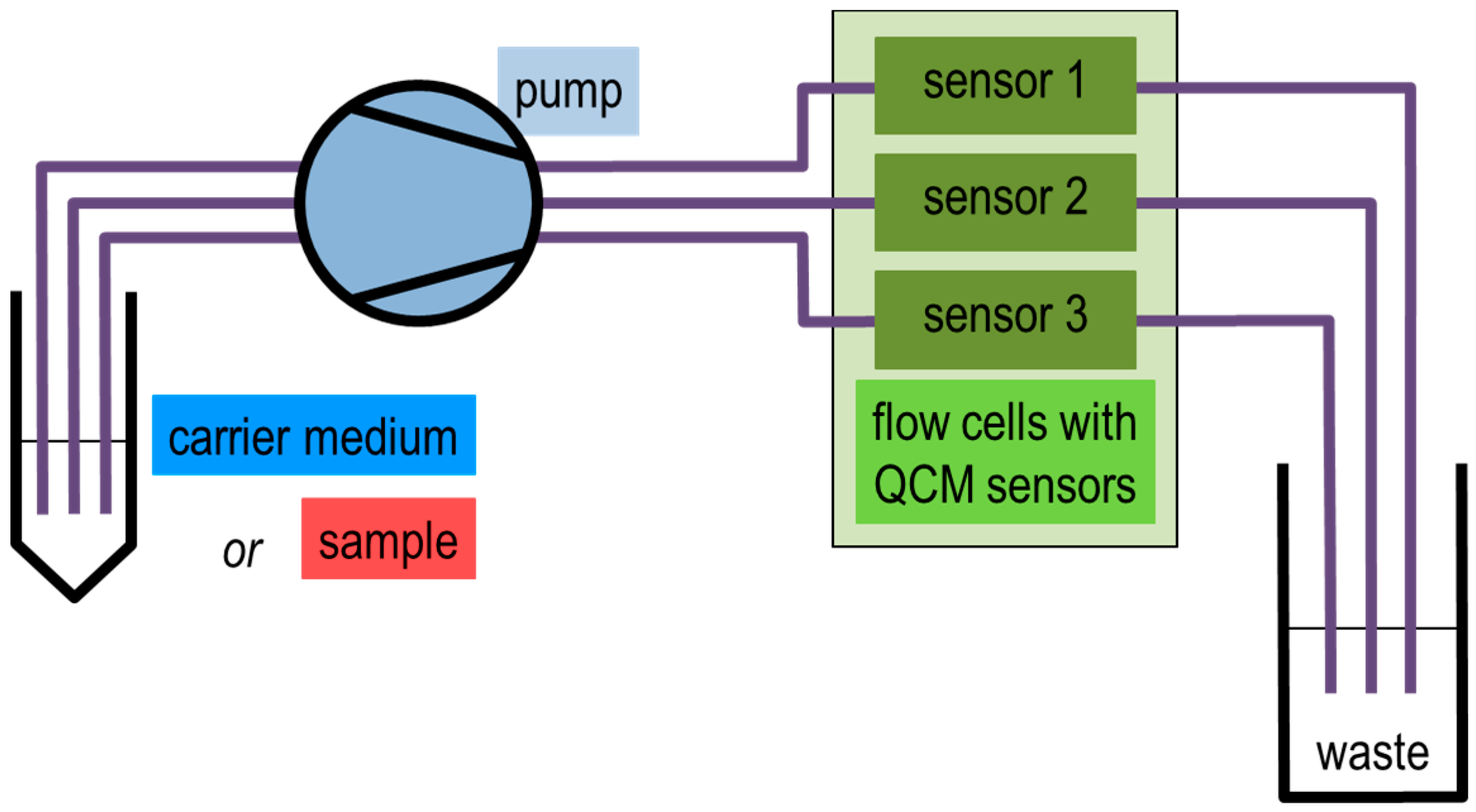
2.2. QCM Sensor Measurement Setup
2.3. Polymer Coatings and Contact Angle Measurements
| Spin Coating Parameter | PMMA | PS |
|---|---|---|
| Polymer concentration in toluene [mg/mL] | 0.1 | 10 |
| Rotation speed [rpm] | 5000 | 1500 |
| Rotation time [min] | 3 | 2 |
| Acceleration step [a. u.] | 10 (highest) | 10 (highest) |
2.4. Protein Adsorption Measurements for Conditioning Film Monitoring
3. Results and Discussion
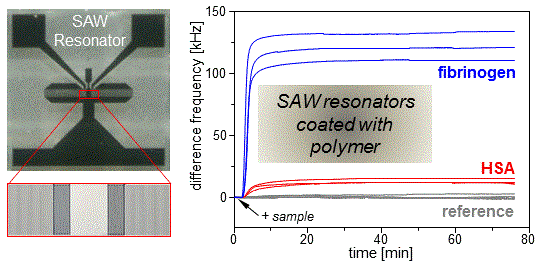
3.1. Conditioning Film Formation on Parylene C: Adsorption of Plasma Proteins
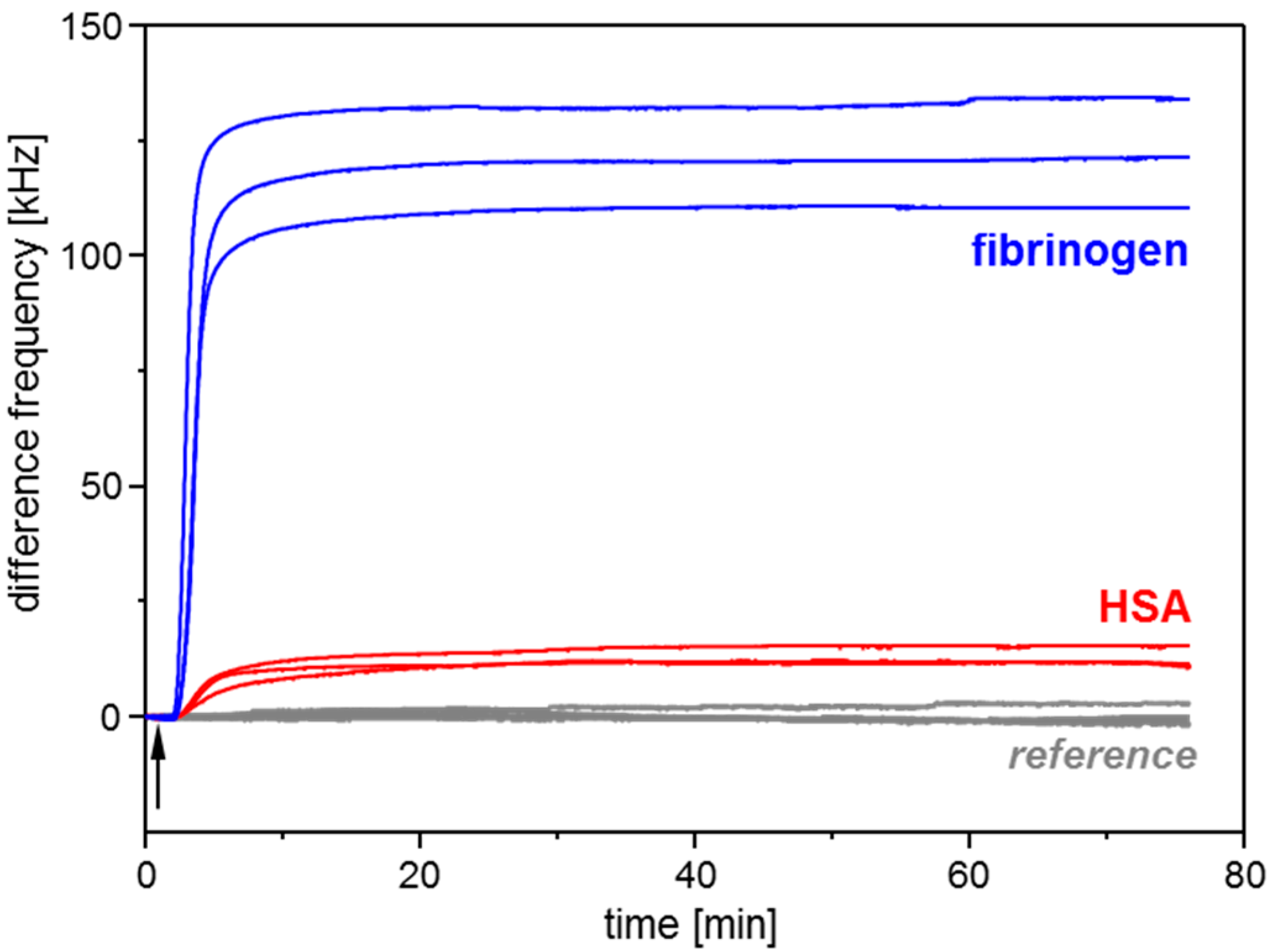
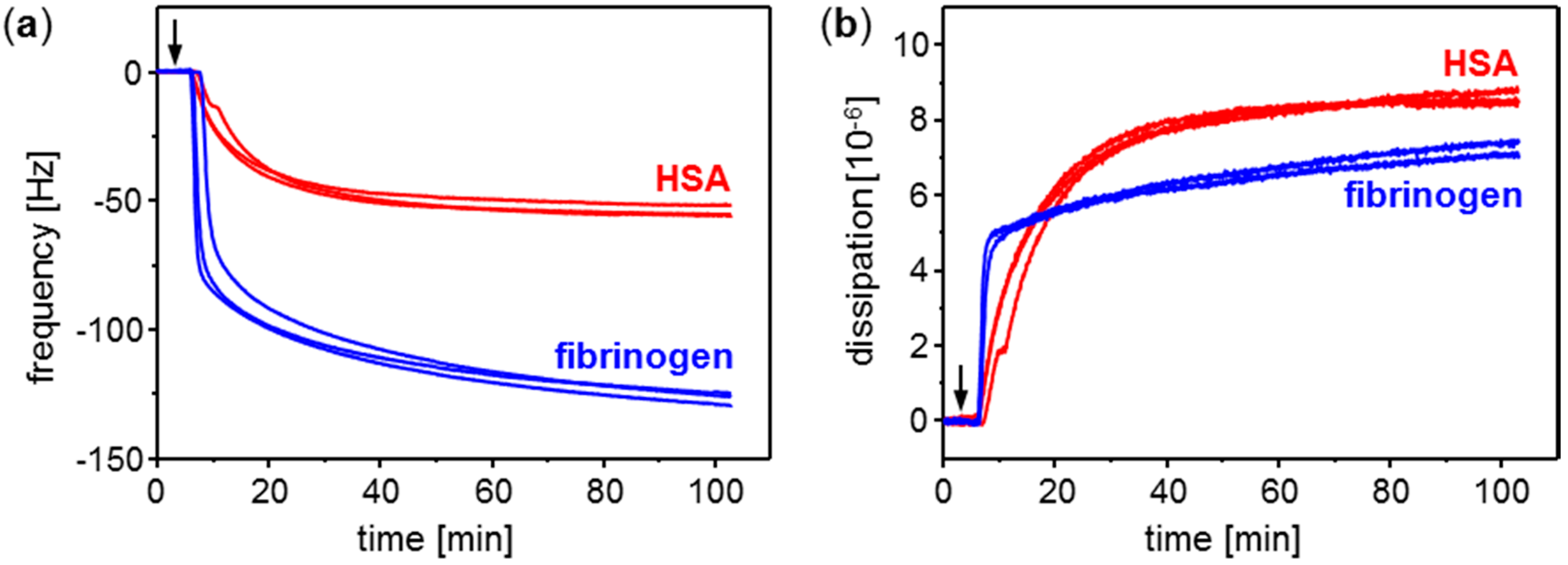
| Adsorbed Protein | SAW Resonator Difference Frequency Shift | QCM Sensor Frequency Shift | ||
|---|---|---|---|---|
| Δf [kHz] | Δf/f0 [ppm] | Δf [Hz] | Δf/f0 [ppm] | |
| HSA | 12.6 ± 2.3 | 29.5 ± 5.3 | −54.2 ± 2.3 | −10.9 ± 0.5 |
| Fibrinogen | 121.9 ± 11.9 | 285.9 ± 27.9 | −126.7 ± 2.4 | −25.6 ± 0.5 |
| None | Noise: 0.08 ± 0.02 | Noise: 0.19 ± 0.04 | Noise: 0.32 ± 0.06 | Noise: 0.06 ± 0.01 |
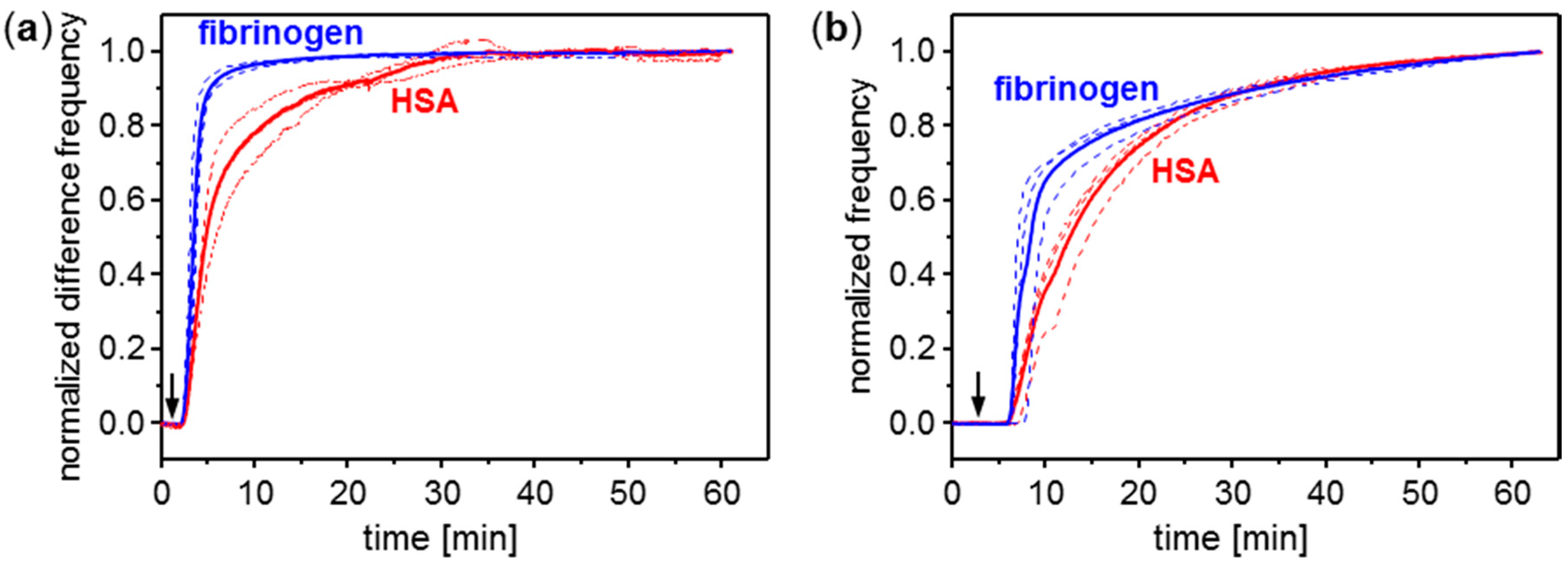
3.2. Conditioning Film Formation on Polymers: Adsorption of HSA on Parylene C, PMMA, and PS
| Polymer | Contact Angle [°] | Contact Angle [°] in Literature | Difference Frequency Shift by HSA Adsorption | |
|---|---|---|---|---|
| Δf [kHz] | Δf/f0 [ppm] | |||
| none | 77.9 ± 0.9 | n/a | 24.2 ± 5.2 | 57 ± 12 |
| Parylene C | 85.4 ± 2.2 | 85.1 ± 1.2 [50] | 23.5 ± 2.8 | 55 ± 7 |
| PMMA | 71.3 ± 1.4 | 67.8 ± 1.4 [51] | 22.3 ± 2.1 | 52 ± 5 |
| PS | 80.4 ± 3.1 | 80 [52] | 22.0 ± 2.6 | 52 ± 6 |
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Characklis, W.G. Bioengineering report. Fouling biofilm development: A process analysis. Biotechnol Bioeng., Vol. XXIII, pp. 1923–60 (1981) Introduced by Bryers, J.D. Biotechnol. Bioeng. 2009, 102, 309–347. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C. Microbial biofouling: Unsolved problems, insufficient approaches, and possible solutions. Springer Ser. Biofilms 2011, 5, 81–109. [Google Scholar]
- Muffler, K.; Lakatos, M.; Schlegel, C.; Strieth, D.; Kuhne, S.; Ulber, R. Application of biofilm bioreactors in white biotechnology. Adv. Biochem. Eng. Biotechnol. 2014, 146, 123–161. [Google Scholar] [PubMed]
- Baier, R.E.; Meyer, A.E.; Natiella, J.R.; Natiella, R.R.; Carter, J.M. Surface properties determine bioadhesive outcomes: Methods and results. J. Biomed. Mater. Res. 1984, 18, 337–355. [Google Scholar] [CrossRef] [PubMed]
- Gristina, A.G. Biomaterial-centered infection: Microbial adhesion versus tissue integration. Science 1987, 237, 1588–1595. [Google Scholar] [CrossRef] [PubMed]
- Lappin-Scott, H.M.; Bass, C. Biofilm formation: Attachment, growth, and detachment of microbes from surfaces. Am. J. Infection Control 2001, 29, 250–251. [Google Scholar] [CrossRef]
- Gottenbos, B.; Busscher, H.J.; van der Mei, H.C. Pathogenesis and prevention of biomaterial centered infections. J. Mater. Sci. Mater. Med. 2002, 13, 717–722. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.Y.; Chen, M.J.; Lee, P.F.; Cheng, L.H.; Huang, L.J.; Lai, C.H.; Huang, K.H. Towards real-time observation of conditioning film and early biofilm formation under laminar flow conditions using a quartz crystal microbalance. Biochem. Eng. J. 2010, 53, 121–130. [Google Scholar] [CrossRef]
- Anderson, J.M.; Rodriguez, A.; Chang, D.T. Foreign body reaction to biomaterials. Semin. Immunol. 2008, 20, 86–100. [Google Scholar] [CrossRef] [PubMed]
- Kaminska, M.; Okroj, W.; Szymanski, W.; Jakubowski, W.; Komorowski, P.; Nosal, A.; Szymanowski, H.; Gazicki-Lipman, M.; Jerczynska, H.; Pawlowska, Z.; et al. Interaction of parylene C with biological objects. Acta Bioeng. Biomech. 2009, 11, 19–25. [Google Scholar] [PubMed]
- Veerachamy, S.; Yarlagadda, T.; Manivasagam, G.; Yarlagadda, P. Bacterial adherence and biofilm formation on medical implants: A review. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2014, 228, 1083–1099. [Google Scholar] [CrossRef]
- Dörner, K. Klinische Chemie und Hämatologie, 7th ed.; Thieme: Stuttgart, Germany, 2009. [Google Scholar]
- Green, R.J.; Davies, J.; Davies, M.C.; Roberts, C.J.; Tendler, S.J.B. Surface plasmon resonance for real time in situ analysis of protein adsorption to polymer surfaces. Biomaterials 1997, 18, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Vörös, J. The density and refractive index of adsorbing protein layers. Biophys. J. 2004, 87, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Roach, P.; Farrar, D.; Perry, C.C. Interpretation of protein adsorption: Surface-induced conformational changes. J. Am. Chem. Soc. 2005, 127, 8168–8173. [Google Scholar] [CrossRef] [PubMed]
- Kao, P.; Parhi, P.; Krishnan, A.; Noh, H.; Haider, W.; Tadigadapa, S.; Allara, D.L.; Vogler, E.A. Volumetric interpretation of protein adsorption: Interfacial packing of protein adsorbed to hydrophobic surfaces from surface-saturating solution concentrations. Biomaterials 2011, 32, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, Z.; Bratek-Skicki, A.; Zeliszewska, P.; Wasilewska, M. Mechanisms of Fibrinogen Adsorption at Solid Substrates. Curr. Top. Med. Chem. 2014, 14, 702–729. [Google Scholar] [CrossRef] [PubMed]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding protein adsorption phenomena at solid surfaces. Adv. Colloid Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef] [PubMed]
- Vogler, E.A. Protein adsorption in three dimensions. Biomaterials 2012, 33, 1201–1237. [Google Scholar] [CrossRef] [PubMed]
- Gruhl, F.J.; Rapp, B.E.; Länge, K. Biosensors for Diagnostic Applications. Adv. Biochem. Eng. Biotechnol. 2013, 133, 115–148. [Google Scholar] [PubMed]
- Perumal, V.; Hashim, U. Advances in biosensors: Principle, architecture and applications. J. Appl. Biomed. 2014, 12, 1–15. [Google Scholar] [CrossRef]
- Contreras, A.E.; Steiner, Z.; Miao, J.; Kasher, R.; Li, Q.L. Studying the role of common membrane surface functionalities on adsorption and cleaning of organic foulants using QCM-D. Environ. Sci. Technol. 2011, 45, 6309–6315. [Google Scholar] [CrossRef] [PubMed]
- Kirschhöfer, F.; Rieder, A.; Prechtl, C.; Kühl, B.; Sabljo, K.; Wöll, C.; Obst, U.; Brenner-Weiß, G. Quartz crystal microbalance with dissipation coupled to on-chip MALDI-ToF mass spectrometry as a tool for characterising proteinaceous conditioning films on functionalised surfaces. Anal. Chim. Acta 2013, 802, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Höök, F.; Vörös, J.; Rodahl, M.; Kurrat, R.; Böni, P.; Ramsden, J.J.; Textor, M.; Spencer, N.D.; Tengvall, P.; Gold, J.; et al. A comparative study of protein adsorption on titanium oxide surfaces using in situ ellipsometry, optical waveguide lightmode spectroscopy, and quartz crystal microbalance/dissipation. Colloids Surfaces B Biointerfaces 2002, 24, 155–170. [Google Scholar] [CrossRef]
- Zhou, C.; Friedt, J.M.; Angelova, A.; Choi, K.H.; Laureyn, W.; Frederix, F.; Francis, L.A.; Campitelli, A.; Engelborghs, Y.; Borghs, G. Human immunoglobulin adsorption investigated by means of quartz crystal microbalance dissipation, atomic force microscopy, surface acoustic wave, and surface plasmon resonance techniques. Langmuir 2004, 20, 5870–5878. [Google Scholar] [CrossRef] [PubMed]
- Rodahl, M.; Höök, F.; Fredriksson, C.; Keller, C.A.; Krozer, A.; Brzezinski, P.; Voinova, M.; Kasemo, B. Simultaneous frequency and dissipation factor QCM measurements of biomolecular adsorption and cell adhesion. Faraday Discuss. 1997, 107, 229–246. [Google Scholar] [CrossRef] [PubMed]
- Dixon, M.C. Quartz crystal microbalance with dissipation monitoring: Enabling real-time characterization of biological materials and their interactions. J. Biomol. Tech. 2008, 19, 151–158. [Google Scholar] [PubMed]
- Grate, J.W.; Frye, G.C. Acoustic Wave Sensors. Sens. Update 1996, 2, 37–83. [Google Scholar] [CrossRef]
- Gizeli, E. Study of the sensitivity of the acoustic waveguide sensor. Anal. Chem. 2000, 72, 5967–5972. [Google Scholar] [CrossRef] [PubMed]
- Klumpers, F.; Götz, U.; Kurtz, T.; Herrmann, C.; Gronewold, T.M.A. Conformational changes at protein-protein interaction followed with an SAW biosensor. Sens. Actuators B Chem. 2014, 203, 904–908. [Google Scholar] [CrossRef]
- Länge, K.; Rapp, B.E.; Rapp, M. Surface acoustic wave biosensors: A review. Anal. Bioanal. Chem. 2008, 391, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Rodahl, M.; Höök, F.; Kasemo, B. QCM operation in liquids: An explanation of measured variations in frequency and Q factor with liquid conductivity. Anal. Chem. 1996, 68, 2219–2227. [Google Scholar] [CrossRef] [PubMed]
- Parker, T.E.; Wichansky, H. Temperature-compensated surface-acoustic-wave devices with SiO2 film overlays. J. Appl. Phys. 1979, 50, 1360–1369. [Google Scholar] [CrossRef]
- Herrmann, F.; Weihnacht, M.; Büttgenbach, S. Properties of sensors based on shear-horizontal surface acoustic waves in LiTaO3/SiO2 and quartz/SiO2 structures. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2001, 48, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Mitsakakis, K.; Gizeli, E. Detection of multiple cardiac markers with an integrated acoustic platform for cardiovascular risk assessment. Anal. Chim. Acta 2011, 699, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Rapp, B.E.; Schickling, B.; Prokop, J.; Piotter, V.; Rapp, M.; Länge, K. Design and integration of a generic disposable array-compatible sensor housing into an integrated disposable indirect microfluidic flow injection analysis system. Biomed. Microdev. 2011, 13, 909–922. [Google Scholar] [CrossRef]
- Chang, K.; Wang, F.; Ding, Y.; Pan, F.; Li, F.K.; Jia, S.R.; Lu, W.P.; Deng, S.L.; Shi, J.F.; Chen, M. Development and validation of a novel leaky surface acoustic wave immunosensor array for label-free and high-sensitive detection of cyclosporin A in whole-blood samples. Biosens. Bioelectron. 2014, 54, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Afzal, A.; Iqbal, N.; Mujahid, A.; Schirhagl, R. Advanced vapor recognition materials for selective and fast responsive surface acoustic wave sensors: A review. Anal. Chim. Acta 2013, 787, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.P.; Craighead, H.G. Surface Engineering and Patterning Using Parylene for Biological Applications. Materials 2010, 3, 1803–1832. [Google Scholar] [CrossRef]
- Hassler, C.; von Metzen, R.P.; Ruther, P.; Stieglitz, T. Characterization of parylene C as an encapsulation material for implanted neural prostheses. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 93B, 266–274. [Google Scholar] [PubMed]
- Cieslik, M.; Zimowski, S.; Golda, M.; Engvall, K.; Pan, J.S.; Rakowski, W.; Kotarba, A. Engineering of bone fixation metal implants biointerface—Application of parylene C as versatile protective coating. Mater. Sci. Eng. C: Mater. Biol. Appl. 2012, 32, 2431–2435. [Google Scholar] [CrossRef]
- Magnan, B.; Bondi, M.; Maluta, T.; Samaila, E.; Schirru, L.; Dall'Oca, C. Acrylic bone cement: current concept review. Musculoskelet. Surg. 2013, 97, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Nowak, J.; Watala, C.; Boncler, M. Antibody binding, platelet adhesion, and protein adsorption on various polymer surfaces. Blood Coagul. Fibrinolysis 2014, 25, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Länge, K.; Blaess, G.; Voigt, A.; Götzen, R.; Rapp, M. Integration of a surface acoustic wave biosensor in a microfluidic polymer chip. Biosens. Bioelectron. 2006, 22, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Bender, F.; Länge, K.; Barié, N.; Kondoh, J.; Rapp, M. On-line monitoring of polymer deposition for tailoring the waveguide characteristics of love-wave biosensors. Langmuir 2004, 20, 2315–2319. [Google Scholar] [CrossRef] [PubMed]
- Rapp, B.E.; Voigt, A.; Dirschka, M.; Länge, K. Deposition of ultrathin parylene C films in the range of 18 nm to 142 nm: Controlling the layer thickness and assessing the closeness of the deposited films. Thin Solid Films 2012, 520, 4884–4888. [Google Scholar] [CrossRef]
- Nomura, T.; Saitoh, A.; Horikoshi, Y. Measurement of acoustic properties of liquid using liquid flow SH-SAW sensor system. Sens. Actuators B Chem. 2001, 76, 69–73. [Google Scholar] [CrossRef]
- Doweiko, J.P.; Nompleggi, D.J. Role of albumin in human physiology and pathophysiology. J. Parenter. Enteral Nutr. 1991, 15, 207–211. [Google Scholar] [CrossRef]
- Merck Millipore: 126658 Albumin, Human Serum, Fraction V, High Purity (Calbiochem). Available online: http://www.merckmillipore.com/DE/en/product/Albumin%2C-Human-Serum%2C-Fraction-V%2C-High-Purity,EMD_BIO-126658 (accessed on 5 May 2015).
- Trantidou, T.; Prodromakis, T.; Toumazou, C. Oxygen plasma induced hydrophilicity of Parylene-C thin films. Appl. Surface Sci. 2012, 261, 43–51. [Google Scholar] [CrossRef]
- Ma, Y.; Cao, X.; Feng, X.; Ma, Y.; Zou, H. Fabrication of super-hydrophobic film from PMMA with intrinsic water contact angle below 90 degrees. Polymer 2007, 48, 7455–7460. [Google Scholar] [CrossRef]
- Wang, Y.; Balowski, J.; Phillips, C.; Phillips, R.; Sims, C.E.; Allbritton, N.L. Benchtop micromolding of polystyrene by soft lithography. Lab Chip 2011, 11, 3089–3097. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hohmann, S.; Kögel, S.; Brunner, Y.; Schmieg, B.; Ewald, C.; Kirschhöfer, F.; Brenner-Weiß, G.; Länge, K. Surface Acoustic Wave (SAW) Resonators for Monitoring Conditioning Film Formation. Sensors 2015, 15, 11873-11888. https://doi.org/10.3390/s150511873
Hohmann S, Kögel S, Brunner Y, Schmieg B, Ewald C, Kirschhöfer F, Brenner-Weiß G, Länge K. Surface Acoustic Wave (SAW) Resonators for Monitoring Conditioning Film Formation. Sensors. 2015; 15(5):11873-11888. https://doi.org/10.3390/s150511873
Chicago/Turabian StyleHohmann, Siegfried, Svea Kögel, Yvonne Brunner, Barbara Schmieg, Christina Ewald, Frank Kirschhöfer, Gerald Brenner-Weiß, and Kerstin Länge. 2015. "Surface Acoustic Wave (SAW) Resonators for Monitoring Conditioning Film Formation" Sensors 15, no. 5: 11873-11888. https://doi.org/10.3390/s150511873
APA StyleHohmann, S., Kögel, S., Brunner, Y., Schmieg, B., Ewald, C., Kirschhöfer, F., Brenner-Weiß, G., & Länge, K. (2015). Surface Acoustic Wave (SAW) Resonators for Monitoring Conditioning Film Formation. Sensors, 15(5), 11873-11888. https://doi.org/10.3390/s150511873