Synthesis and Sensing Applications of Fluorescent 3-Cinnamoyl Coumarins
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Instruments
2.3. Quantum Efficiency Measurement
2.4. TPF Measurement
2.5. TPA Cross Section Measurement
2.6. Stern-Volmer Constant (Ksv), Binding Constant (K) and Number of Binding Sites (n) Measurements
2.7. Chemistry
2.7.1. 3-Acetyl-4-hydroxy-7,8-dimethoxy-2H-chromen-2-one (6)
2.7.2. (E)-3-[3-(4-(Diethylaminophenyl)acryloyl]-4-hydroxy-7-methoxy-2H-chromen-2-one (7)
2.7.3. (E)-3-[3-(4-(Diethylaminophenyl)acryloyl]-4-hydroxy-7,8-dimethoxy-2H-chromen-2-one (8)
2.7.4. (E)-6-[3-(4-Diethylaminophenyl)acryloyl]-7-hydroxy-4-methyl-2H-chromen-2-one (12)
2.7.5. (E)-6-[3-(4-Diethylaminophenyl)acryloyl]-7-methoxy-4-methyl-2H-chromen-2-one (13)
3. Results and Discussion
3.1. Synthesis
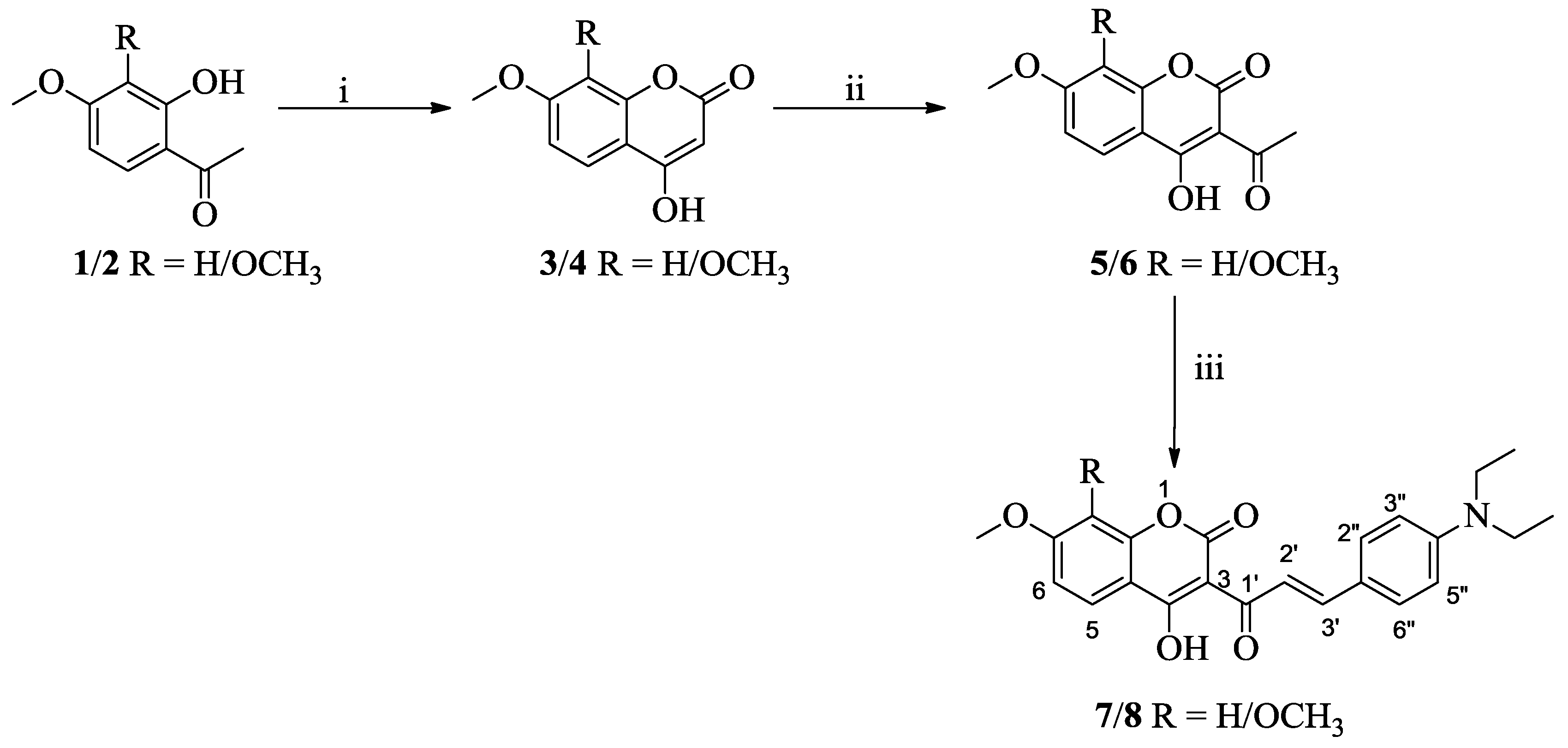
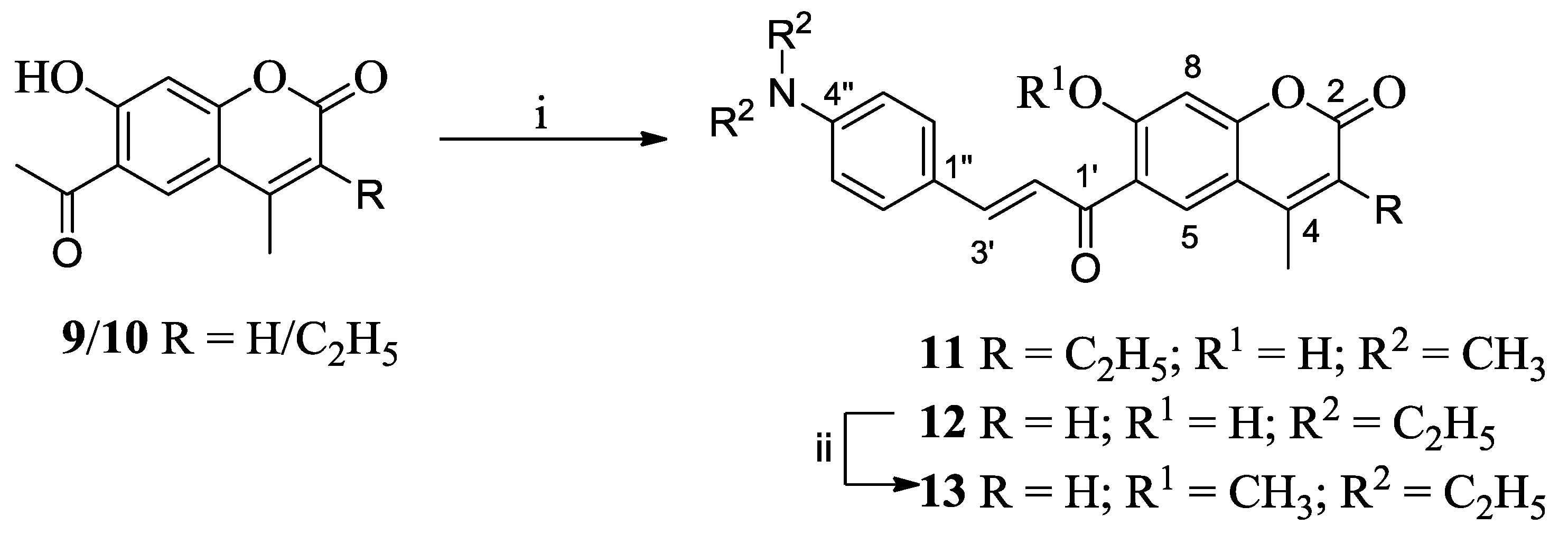
3.2. One-Photon and TP Activity of 3-Cinnamoylcoumarins
| Compd. | λAb. (nm) | λEm. (nm) | Stokes Shift (nm) | Ф (%) | Δ (GM) |
|---|---|---|---|---|---|
| 7 | 521 | 608 | 87 | 11.01 | 59 |
| 8 | 523 | 610 | 87 | 17.60 | 85 |
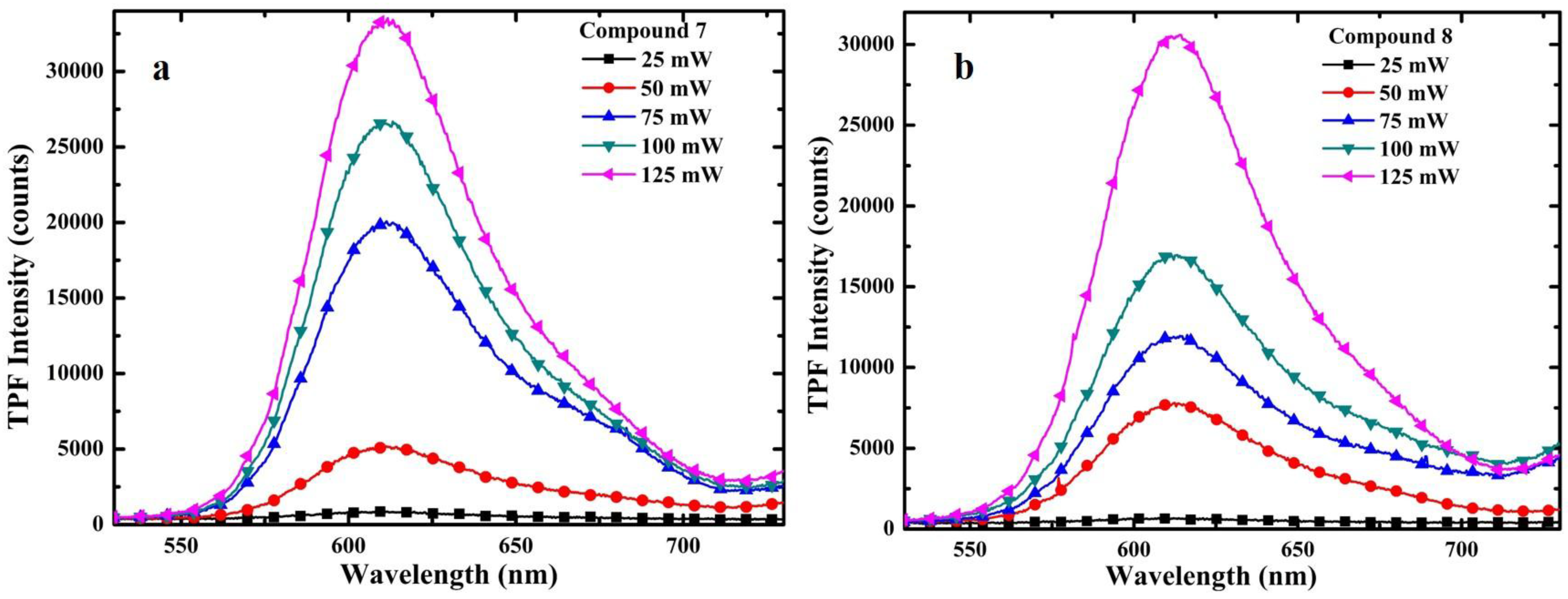
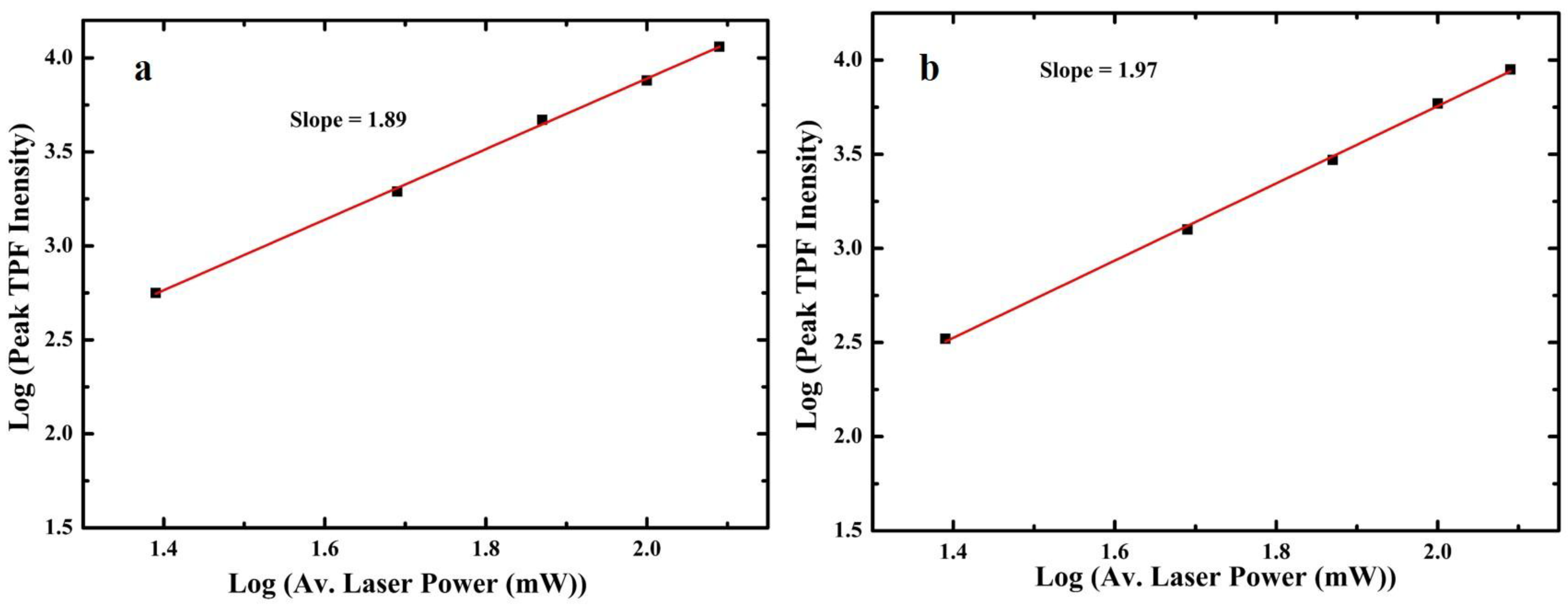
3.3. TPF Imaging

3.4. DCP Sensing Study

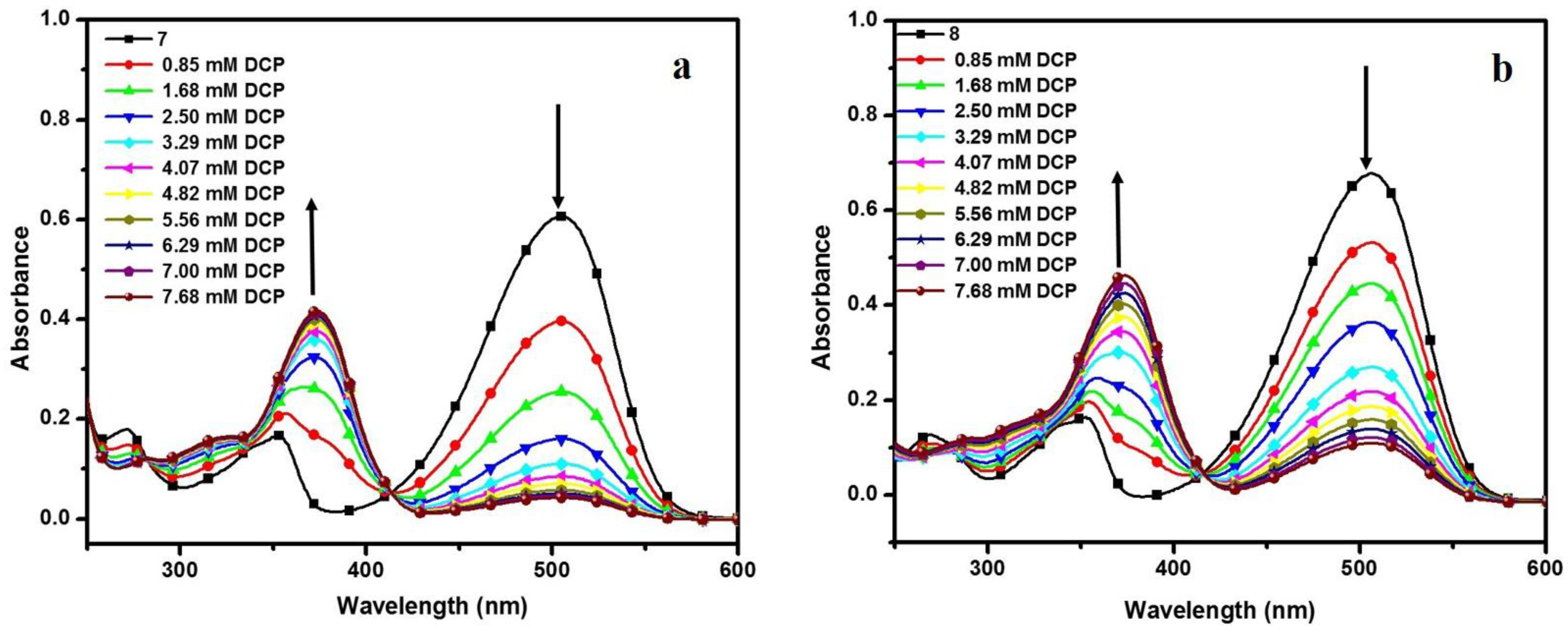

| Compd. | λAb. (nm) | λEm. (nm) | Ksv (M−1) | K (M−1) | n |
|---|---|---|---|---|---|
| 7 | 506 | 573 | 791 | 8.235 X 102 | 1.011 |
| 8 | 505 | 572 | 1070 | 1.410 X 103 | 1.054 |
3.5. NMR Study and the Sensing Mechanism
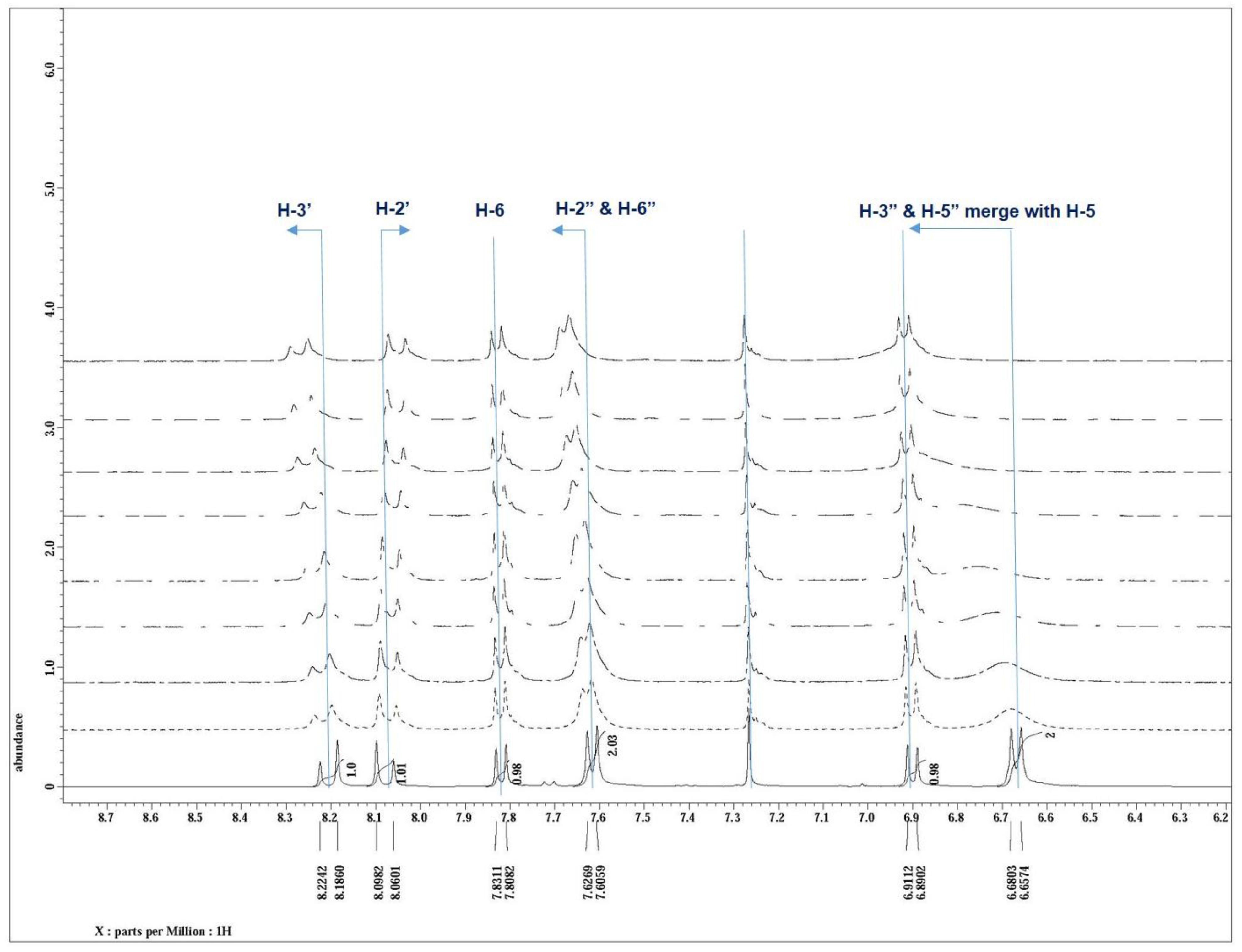
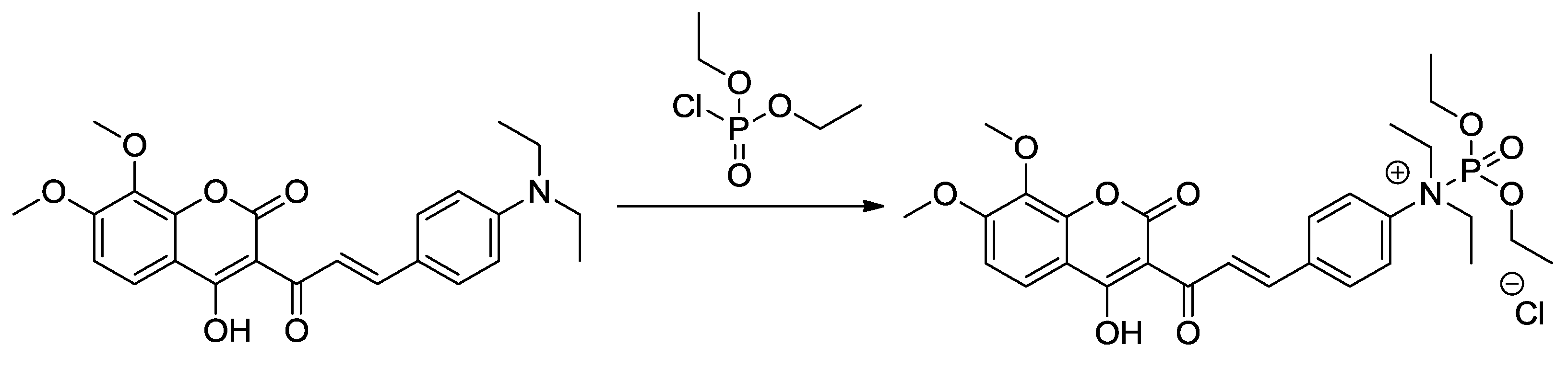
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Marrs, T.C. Organophosphate poisoning. Pharmacol. Ther. 1993, 58, 51–66. [Google Scholar]
- Sidell, F.R.; Borak, J. Chemical warfare agents: II. Nerve agents. Ann. Emerg. Med. 1992, 21, 865–871. [Google Scholar] [CrossRef]
- Gehauf, B.; Goldenson, J. Detection and estimation of nerve gases by fluorescence reaction. Anal. Chem. 1957, 29, 276–278. [Google Scholar] [CrossRef]
- Kim, K.; Tsay, O.G.; Atwood, D.A.; Churchill, D.G. Destruction and detection of chemical warfare agents. Chem. Rev. 2011, 111, 5345–5403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.W.; Swager, T.M. Fluorescent detection of chemical warfare agents: functional group specific ratiometric chemosensors. J. Am. Chem. Soc. 2003, 125, 3420–3421. [Google Scholar] [CrossRef] [PubMed]
- Dale, T.J.; Rebek, J. Fluorescent sensors for organophosphorous nerve agent mimics. Jr. J. Am. Chem. Soc. 2006, 128, 4500–4501. [Google Scholar] [CrossRef] [PubMed]
- Wallace, K.J.; Fagbemi, R.I.; Folmer-Andersen, F.J.; Morey, J.; Lynch, V.M.; Anslyn, E.V. Detection of Chemical warfare simulants by phosphorylation of a coumarin oximate. Chem. Commun. 2006, 3886–3888. [Google Scholar] [CrossRef] [PubMed]
- Menzel, E.R.; Menzel, L.W.; Schwierking, J.R. Rapid fluorophosphates nerve agent detection with lanthanides. Talanta 2005, 67, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Dale, T.J.; Rebek, J. Hydroxy oximes as organophosphorus nerve agent sensor. Angew. Chem. Int. Ed. 2009, 48, 7850–7852. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Dong, J.; Wang, X.; Li, J.; Sui, S.; Chen, G.; Liu, J.; Zhang, M. Fluorogenic and chromogenic probe for rapid detection of a nerve agent simulant DCP. Analyst 2012, 137, 3224–3226. [Google Scholar] [CrossRef] [PubMed]
- Royo, S.; Costero, A.M.; Parra, M.; Gil, S.; Martínez-Mánez, R.; Sancenón, F. Chromogenic, specific detection of the Nerve-Agent Mimic DCNP (a Tabun Mimic). Chem. Eur. J. 2011, 17, 6931–6934. [Google Scholar] [CrossRef] [PubMed]
- Costero, A.M.; Gil, S.; Parra, M.; Mancini, P.M.E.; Martínez-Mánez, R.; Sancenón, F.; Royo, S. Chromogenic detection of nerve agent mimics. Chem. Commun. 2008. [Google Scholar] [CrossRef] [PubMed]
- Trenor, S.R.; Shultz, A.R.; Love, B.J.; Long, T.E. Coumarins in polymers: From light harvesting to photo-cross-linkable tissue scaffolds. Chem. Rev. 2004, 104, 3059–3077. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.-L.; Kuo, P.-Y.; Yang, D.-Y. Design and synthesis of a coumarin-based acidichromic colorant. Molecules 2007, 12, 1316–1324. [Google Scholar] [CrossRef] [PubMed]
- Suresh, M.; Das, A. New coumarin-based sensor molecule for magnesium and calcium ions. Tetrahedron Lett. 2009, 50, 5808–5812. [Google Scholar] [CrossRef]
- Komatsu, K.; Urano, Y.; Kojima, H.; Nagano, T. Development of an Iminocoumarin-based zinc sensor suitable for ratiometric fluorescence imaging of neuronal zinc. J. Am. Chem. Soc. 2007, 129, 13447–13454. [Google Scholar] [CrossRef] [PubMed]
- Ray, D.; Bharadwaj, P.K. A coumarin-derived fluorescence probe selective for magnesium. Inorg. Chem. 2008, 47, 2252–2254. [Google Scholar] [CrossRef] [PubMed]
- Lim, N.C.; Bruckner, C. DPA-substituted coumarins as chemosensors for Zinc (II): Modulation of the chemosensory characteristics by variation of the position of the chelate on the coumarin. Chem. Commun. 2004, 1094–1095. [Google Scholar] [CrossRef] [PubMed]
- Hanshaw, R.G.; Hilkert, S.M.; Jiang, H.; Smith, B.D. An indicator displacement system for fluorescent detection of phosphate oxyanions under physiological conditions. Tetrahedron Lett. 2004, 45, 8721–8724. [Google Scholar] [CrossRef]
- Denk, W.; Strickler, J.H.; Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 1990, 248, 73–76. [Google Scholar] [CrossRef] [PubMed]
- So, P.T.; Dong, C.Y.; Masters, B.R.; Berland, K.M. Two-photon excitation fluorescence microscopy. Annu. Rev. Biomed. Eng. 2000, 2, 399–429. [Google Scholar] [CrossRef] [PubMed]
- Diaspro, A.; Chirico, G.; Collini, M. Two-photon fluorescence excitation and related techniques in biological microscopy. Q. Rev. Biophys. 2005, 38, 97–166. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, S.J.; MacVicar, B.A. Two-photon fluorescence microscopy: Basic principles, advantages and risks. Mod. Res. Educ. Top. Microsc. 2007, 2, 881–889. [Google Scholar]
- Li, X.; Zhao, Y.X.; Wang, T.; Shi, M.Q.; Wu, F.P. Coumarin derivatives with enhanced Two-photon absorption cross sections. Dyes Pigm. 2007, 74, 108–112. [Google Scholar] [CrossRef]
- Zou, Q.; Fang, Y.; Zhao, Y.; Zhao, H.; Wang, Y.; Gu, Y. Synthesis and in vitro photocytotoxicity of coumarin derivatives for one- and two-photon excited photodynamic therapy. J. Med. Chem. 2013, 56, 5288–5294. [Google Scholar] [CrossRef] [PubMed]
- Chand, K.; Shirazi, A.N.; Yadav, P.; Tiwari, R.K.; Kumari, M.; Parang, K.; Sharma, S.K. Synthesis and antiproliferative and c-Src kinase inhibitory activities of cinnamoyl- and pyranochromen-2-one derivatives. Can. J. Chem. 2013, 91, 741–754. [Google Scholar] [CrossRef]
- Yadav, P.; Satapathi, S.; Kumari, M.; Chaturvedi, A.; Li, L.; Samuelson, L.A.; Kumar, J.; Sharma, S.K. Synthesis of two-photon active cinnamoyl coumarins for high-contrast imaging of cancer cells and their photophysical characterization. J. Photochem. Photobiol. A Chem. 2014, 280, 39–45. [Google Scholar] [CrossRef]
- Albota, M.A.; Xu, C.; Webb, W.W. Two-Photon Fluorescence Excitation Cross Sections of Biomolecular Probes from 690 to 960 nm. Appl. Opt. 1998, 37, 7352–7356. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.J.; Sethna, S. Synthesis of Some 4-hydroxycoumarin derivatives. J. Org. Chem. 1957, 22, 388–390. [Google Scholar] [CrossRef]
- Dholakia, V.N.; Parekh, M.G.; Trivedi, K.N. Studies in 4-hydroxy coumarins. Aust. J. Chem. 1968, 21, 2345–2347. [Google Scholar] [CrossRef]
- Kubin, R.F.; Fletcher, A.N. Fluorescence quantum yields of some rhodamine dyes. J. Lumin. 1982, 27, 455–462. [Google Scholar] [CrossRef]
- Nag, A.; Goswami, D. Solvent effect on two-photon absorption and fluorescence of rhodamine dyes. J. Photochem. Photobiol. A Chem. 2009, 206, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.C.; Wang, Y.; Agren, H. Two-photon absorption cross-sections of reference dyes: A critical examination. Chem. Phys. Chem. 2008, 9, 111–116. [Google Scholar]
- Jo, S.; Kim, D.; Son, S.-H.; Kim, Y.; Lee, T.S. Conjugated poly(fluorene-quinoxaline) for fluorescence imaging and chemical detection of nerve agents with its paper-based strip. Appl. Mater. Interfac. 2014, 6, 1330–1336. [Google Scholar] [CrossRef] [PubMed]
- Weerasinghe, A.J.; Schmiesing, C.; Sinn, E. Synthesis, characterization, and evaluation of rhodamine based sensors for nerve gas mimics. Tetrahedron 2011, 67, 2833–2838. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, P.; Gill, H.S.; Chand, K.; Li, L.; Kumar, J.; Sharma, S.K. Synthesis and Sensing Applications of Fluorescent 3-Cinnamoyl Coumarins. Sensors 2015, 15, 31987-31998. https://doi.org/10.3390/s151229902
Yadav P, Gill HS, Chand K, Li L, Kumar J, Sharma SK. Synthesis and Sensing Applications of Fluorescent 3-Cinnamoyl Coumarins. Sensors. 2015; 15(12):31987-31998. https://doi.org/10.3390/s151229902
Chicago/Turabian StyleYadav, Preeti, Hardeep Singh Gill, Karam Chand, Lian Li, Jayant Kumar, and Sunil K. Sharma. 2015. "Synthesis and Sensing Applications of Fluorescent 3-Cinnamoyl Coumarins" Sensors 15, no. 12: 31987-31998. https://doi.org/10.3390/s151229902
APA StyleYadav, P., Gill, H. S., Chand, K., Li, L., Kumar, J., & Sharma, S. K. (2015). Synthesis and Sensing Applications of Fluorescent 3-Cinnamoyl Coumarins. Sensors, 15(12), 31987-31998. https://doi.org/10.3390/s151229902






