3.1. Regression Analysis
The exponential relationships between INSEY and yield at V6 and V12 stages of corn where five wavelengths were used to compare the coefficient of determination (r
2) between corn yield and sensor reading are provided in
Table 4. The exponential relationships were roughly linear, but the r
2 values from the exponential models were nearly always superior to that of the linear model. In high clay sites at V6 using the CC, the red edge NDVI-based INSEY was significantly related to yield whereas both GS red edge NDVI-based INSEY was unrelated to yield. At V12 CC red NDVI, CC red edge NDVI, and GS red NDVI based INSEY were related to yield, while both wavelengths of GS red edge NDVI were not. In medium textured sites, the INSEY derived from wavelengths of both active-sensors at both growth stages were significantly related to yield (
Table 4).
In eastern North Dakota (ND) long-term no-till sites, there were no INSEY relationships between yield and sensor readings at V6 (
Table 5). In western ND long-term no-till sites, there were significant relationships between INSEY and yield using both sensors and all wavelengths at V6 (
Table 5). In previous research, there was no clear trend in the effect of crop residues on wavelength absorption and reflectance compared to soil; residue coverage of the soil can vary, with the effect of residue coverage influenced by residue moisture content and the origin of residue [
65,
66,
67]. More consistent identification and quantification of residue appears to be only possible using bands in wavelengths greater than 1110 nm. It is noteworthy that eastern no-till sites tended to have denser residue cover, partially due to the tendency of eastern site cooperators to utilize more fall cover crops, and the tendency at some eastern sites to follow small grain with corn, whereas western sites tended to follow a lower residue crop. At V12 in western ND there were no INSEY relationships with yield in eastern ND sites using red NDVI or red edge NDVI. Using red edge NDVI, the relationship between INSEY and yield was significant at eastern ND long-term no-till sites. At V12 in eastern ND, the relationships between INSEY and yield were significant with both sensors and all wavelengths. The r
2 values in eastern ND for V12 tended to be less than those at V6.
3.2. Wavelength Sensitivity and Absorption Spectra
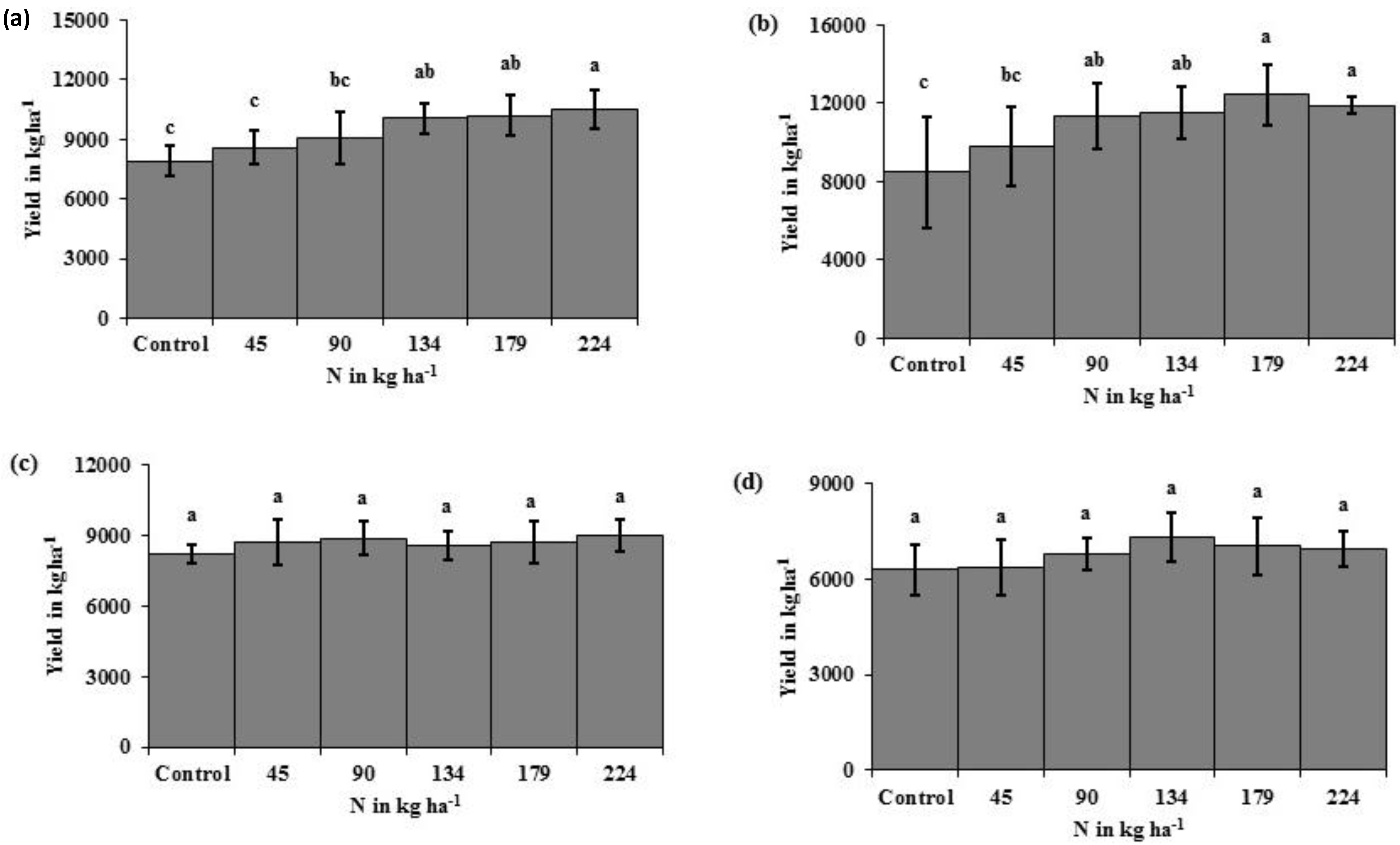
The N rate treatments resulted in significant yield differences, with increased yield as N rate increased in eastern ND high clay conventional-till sites (
Figure 1a) and eastern ND medium textured conventional-till sites (
Figure 1b). There were no yield responses due to N rate at eastern ND no-till sites (
Figure 1c) or western ND no-till sites (
Figure 1d). The INSEY varied similarly to yield. In
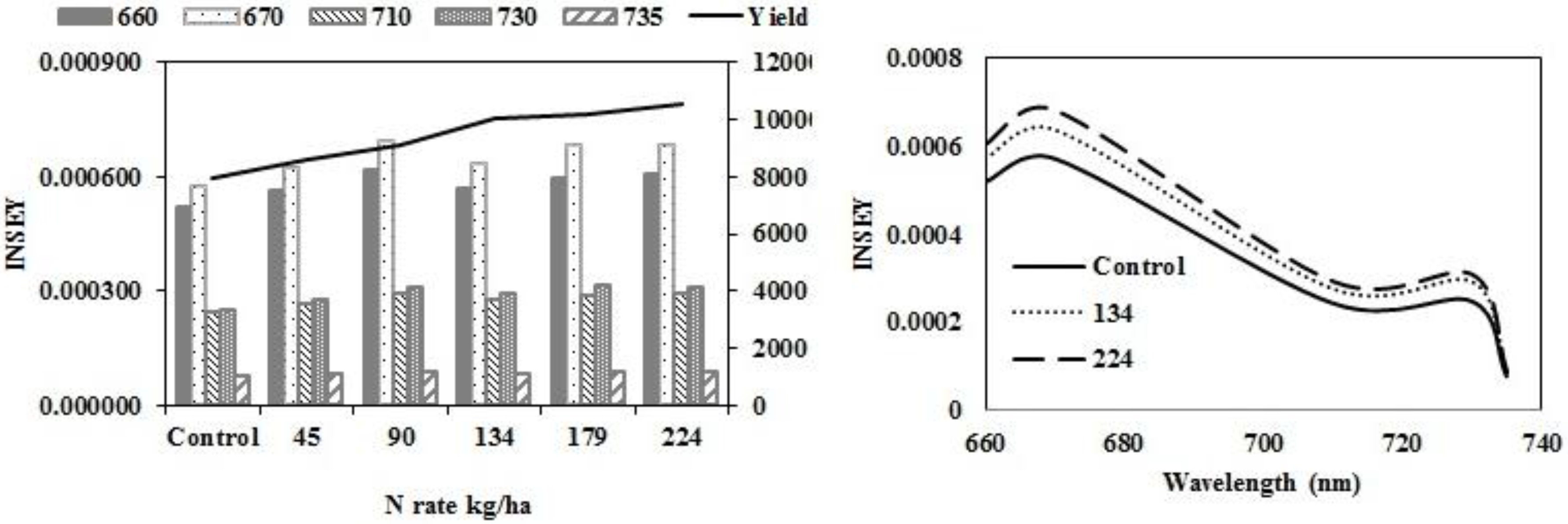
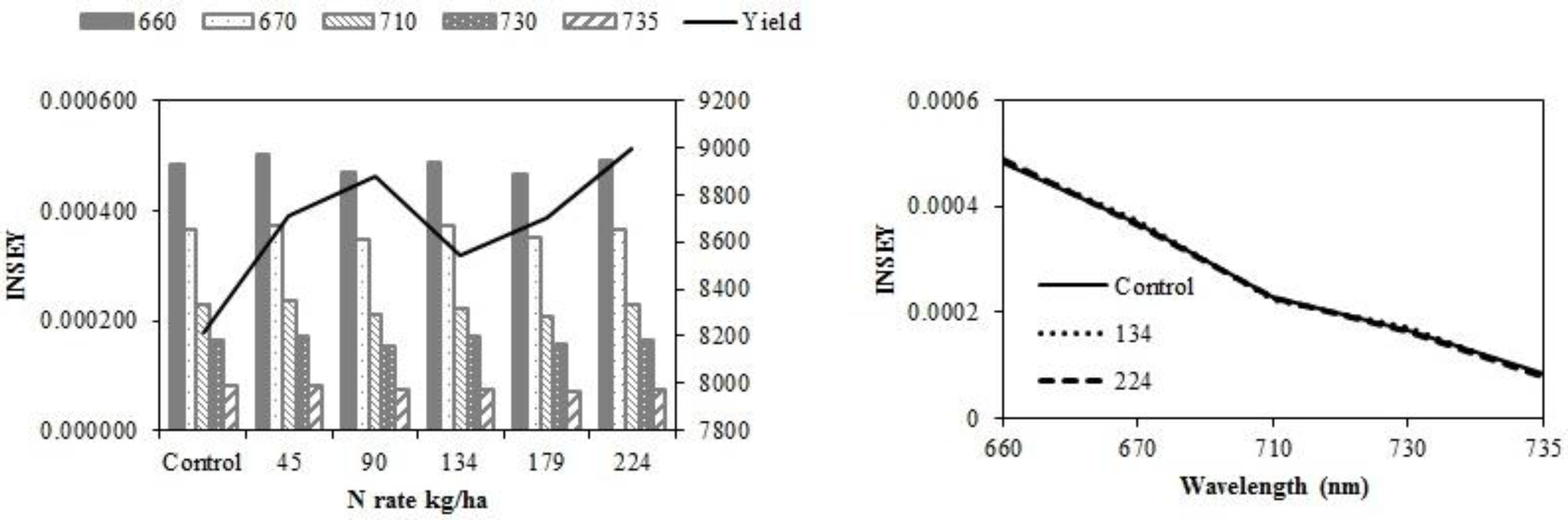
Figure 2a,c, and
Table 6, with yield and INSEY at eastern ND high clay sites, yield response and INSEY response to N rate were similar. The sensitivity, which is the ability of a measurement to relate to an outcome, was greater for the INSEY developed from red wavelengths of the GS (660) and CC (670) compared to red edge INSEYs (
Figure 2b,d,
Table 6), meaning that the separation of INSEY values with N rate was greater for INSEY developed under the red wavelengths than those developed under red-edge. At V6, although there appears to be a similar response of 710 nm and 730 nm (
Figure 2a) wavelengths with N rates the INSEY relationship using the 710 nm red edge NDVI was not significant, while the red edge INSEY relationship using 730 nm was significant (
Table 4). At V12, the sensitivity of both the 710 nm and 730 nm derived INSEY was similar (
Figure 2b) and the relationship to yield was also similar (
Table 4 and
Table 6). The INSEY values at high clay sites increased from V6 to V12, which is commonly observed due to greater biomass with growth (
Figure 2a,b). The absorption spectrum trends of INSEY with wavelength at three different N rates were similar at V6 and V12 (
Figure 2b,d).
Figure 1.
Corn yield difference in response to N fertilizer rates under high clay conventional-till (a); medium textured conventional-till (b); eastern ND no-till (c); and western ND no-till (d) sites. Bars with different letters indicate significance at p < 0.05. Bars represent the range of yields within each N rate.
Figure 1.
Corn yield difference in response to N fertilizer rates under high clay conventional-till (a); medium textured conventional-till (b); eastern ND no-till (c); and western ND no-till (d) sites. Bars with different letters indicate significance at p < 0.05. Bars represent the range of yields within each N rate.
Figure 2.
Variation of INSEY for corn at high clay conventional-till sites calculated from CC active-sensor spectra (670 nm and 730 nm) and GS spectra (660 nm, 710 nm and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134∙kg∙ha−1 and 224∙kg∙ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 in figures at (upper right) and at V12 in (lower right).
Figure 2.
Variation of INSEY for corn at high clay conventional-till sites calculated from CC active-sensor spectra (670 nm and 730 nm) and GS spectra (660 nm, 710 nm and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134∙kg∙ha−1 and 224∙kg∙ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 in figures at (upper right) and at V12 in (lower right).
Table 6.
Treatment differences between INSEY developed from five wavelengths utilizing two ground-based active-optical sensors in eastern high clay (n = 143) and eastern medium-textured (n = 71) categories under conventional tillage at two corn growth stages from nitrogen rate treatments.
Table 6.
Treatment differences between INSEY developed from five wavelengths utilizing two ground-based active-optical sensors in eastern high clay (n = 143) and eastern medium-textured (n = 71) categories under conventional tillage at two corn growth stages from nitrogen rate treatments.
| Soil Type/Tillage System | Growth Stage | N Treatment kg·N·ha−1 | INSEY † X 10,000 |
|---|
| Holland Crop Circle | GreenSeeker |
|---|
| Red Edge 730 * | Red 670 | Red 660 | Red Edge 710 | Red Edge 735 |
|---|
| High Clay | V6 | 0 | 0.000249 b | 0.000572 a | 0.000519 a | 0.000244 a | 0.000078 a |
| 45 | 0.000275 ab | 0.000623 a | 0.000564 a | 0.000265 a | 0.000082 a |
| 90 | 0.000311 ab | 0.000694 a | 0.000618 a | 0.000296 a | 0.000088 a |
| 135 | 0.000293 ab | 0.000636 a | 0.000571 a | 0.000277 a | 0.000085 a |
| 179 | 0.000315 a | 0.000685 a | 0.000598 a | 0.000291 a | 0.000089 a |
| 224 | 0.000310 ab | 0.000682 a | 0.000605 a | 0.000295 a | 0.000090 a |
| V12 | 0 | 0.000311 d | 0.000650 c | 0.000647 b | 0.000362 b | 0.000110 c |
| 45 | 0.000338 cd | 0.000684 bc | 0.000668 b | 0.000378 b | 0.000114 c |
| 90 | 0.000350 bc | 0.000703 ab | 0.000694 ab | 0.000393 ab | 0.000122 bc |
| 135 | 0.000365 abc | 0.000711 ab | 0.000693 ab | 0.000397 ab | 0.000120 c |
| 179 | 0.000376 ab | 0.000720 ab | 0.000720 a | 0.000423 a | 0.000135 ab |
| 224 | 0.000382 a | 0.000737 a | 0.000727 a | 0.000426 a | 0.000137 a |
| Medium- textured | V6 | 0 | 0.000160 a | 0.000360 a | 0.000423 a | 0.000190 a | 0.000062 a |
| 45 | 0.000164 a | 0.000365 a | 0.000452 a | 0.000206 a | 0.000063 a |
| 90 | 0.000190 a | 0.000426 a | 0.000502 a | 0.000225 a | 0.000065 a |
| 135 | 0.000186 a | 0.000399 a | 0.000483 a | 0.000223 a | 0.000066 a |
| 179 | 0.000212 a | 0.000460 a | 0.000515 a | 0.000242 a | 0.000072 a |
| 224 | 0.000196 a | 0.000429 a | 0.000503 a | 0.000235 a | 0.000069 a |
| V12 | 0 | 0.000306 c | 0.000646 b | 0.000651 b | 0.000359 c | 0.000104 c |
| 45 | 0.000337 bc | 0.000675 ab | 0.00068 ab | 0.000388 bc | 0.000114 bc |
| 90 | 0.000357 ab | 0.000702 a | 0.000706 a | 0.000406 ab | 0.000126 ab |
| 135 | 0.000362 ab | 0.000704 a | 0.000696 ab | 0.000403 ab | 0.000125 ab |
| 179 | 0.000374 a | 0.000713 a | 0.000711 a | 0.00042 a | 0.000135 a |
| 224 | 0.000365 ab | 0.000704 a | 0.000701 a | 0.000414 ab | 0.00013 a |
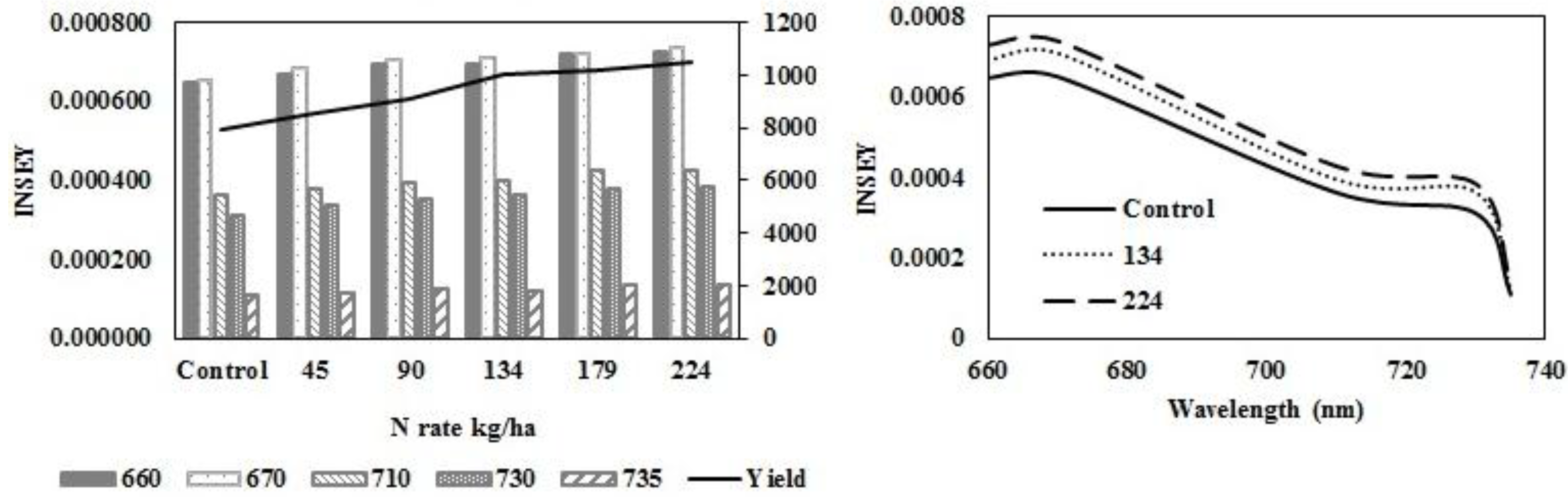
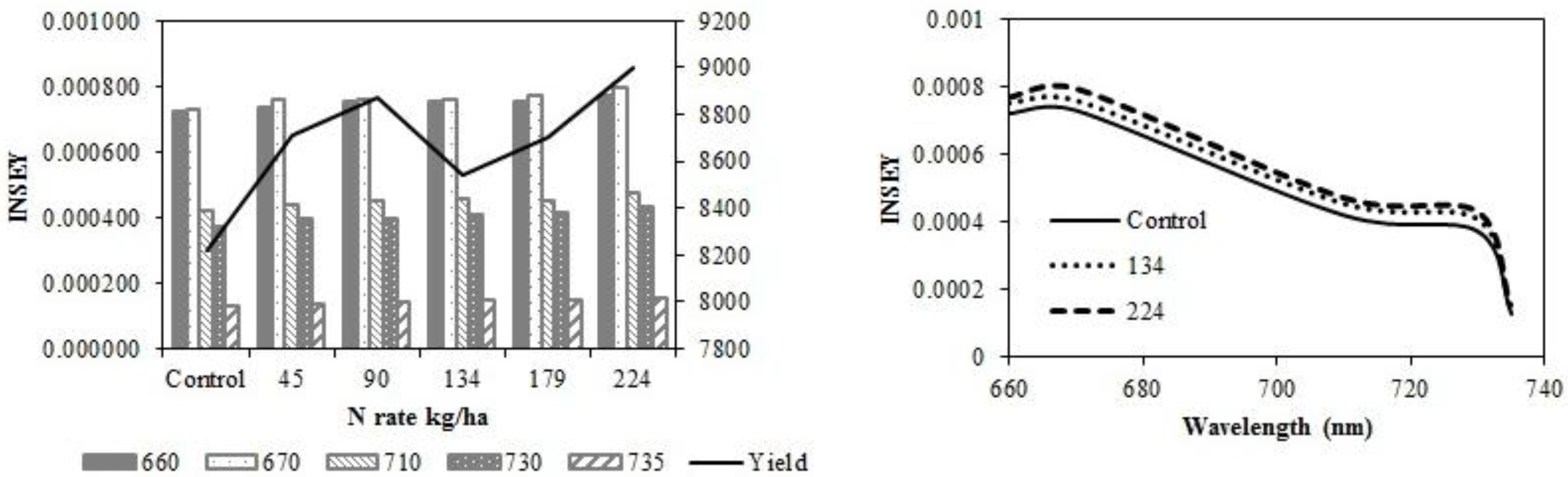
In eastern ND medium textured sites (
Figure 3a–d), wavelength sensitivity of active-optical sensor INSEY to yield was similar at V6 (
Figure 3a) and V12 (
Figure 3c) with sensitivity greater with red NDVI derived INSEY ranges at V6 and V12. At V6 and V12, the sensitivity of red NDVI INSEY in both sensors to N rates was similar and both were similarly related to yield at both growth stages (
Table 4). The trend for greater INSEY with N rate was less clear at V6 (
Table 6). At V6, there were no significant relationships between INSEY and yield for all wavelengths. At V12, the relationships between INSEY and yield were significant for all wavelengths. Yield is influenced by factors other than N rate [
68,
69,
70,
71], which is why an N non-limiting area should be established within a field where active-optical sensors will be used to help determine in-season supplemental N fertilization [
72].
Figure 3.
Variation of INSEY for corn at medium-textured conventional-till sites calculated from CC active-sensor spectra (670 nm and 730 nm) and GS spectra (660 nm, 710 nm and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg∙ha−1 and 224 kg∙ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).
Figure 3.
Variation of INSEY for corn at medium-textured conventional-till sites calculated from CC active-sensor spectra (670 nm and 730 nm) and GS spectra (660 nm, 710 nm and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg∙ha−1 and 224 kg∙ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).
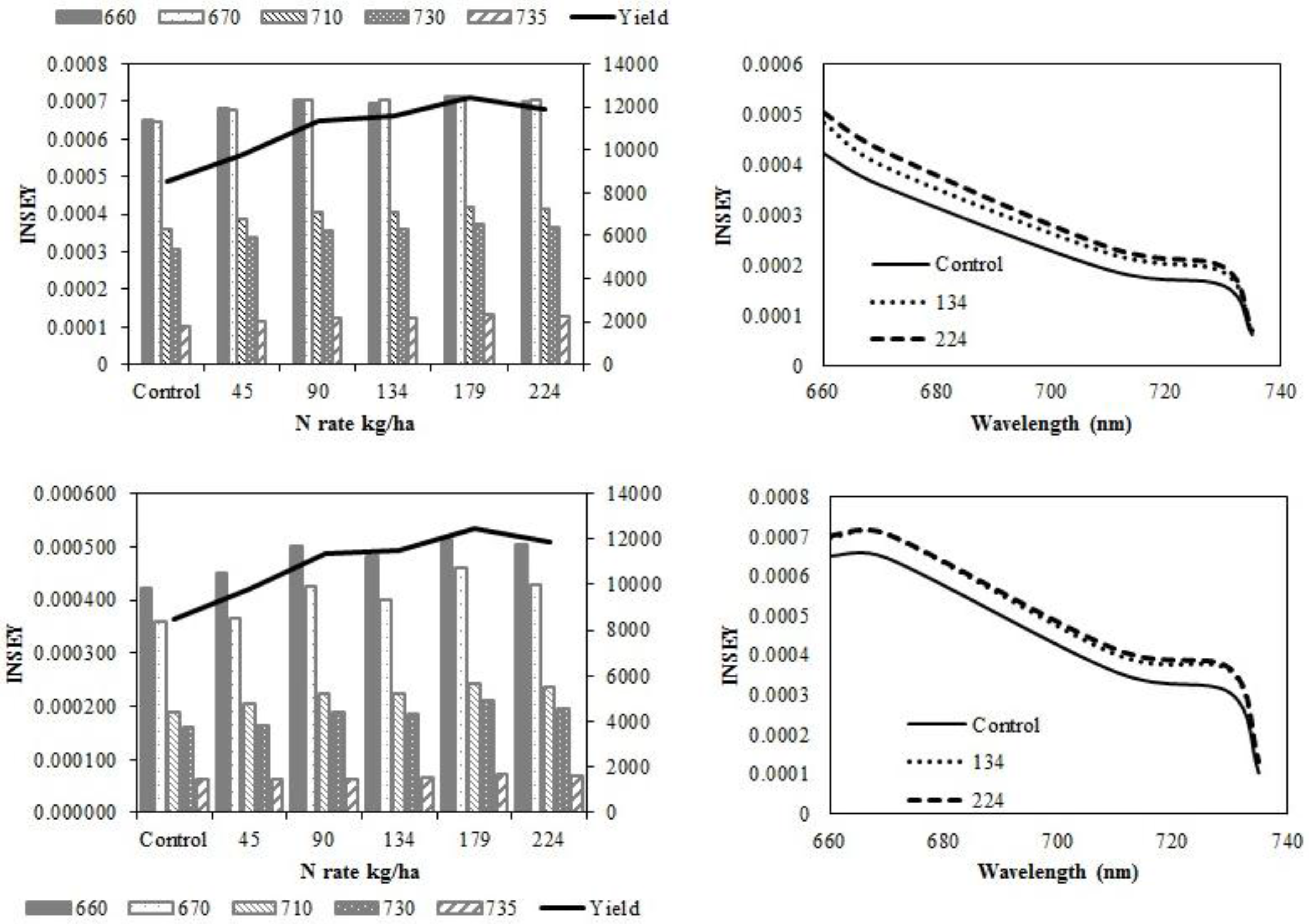
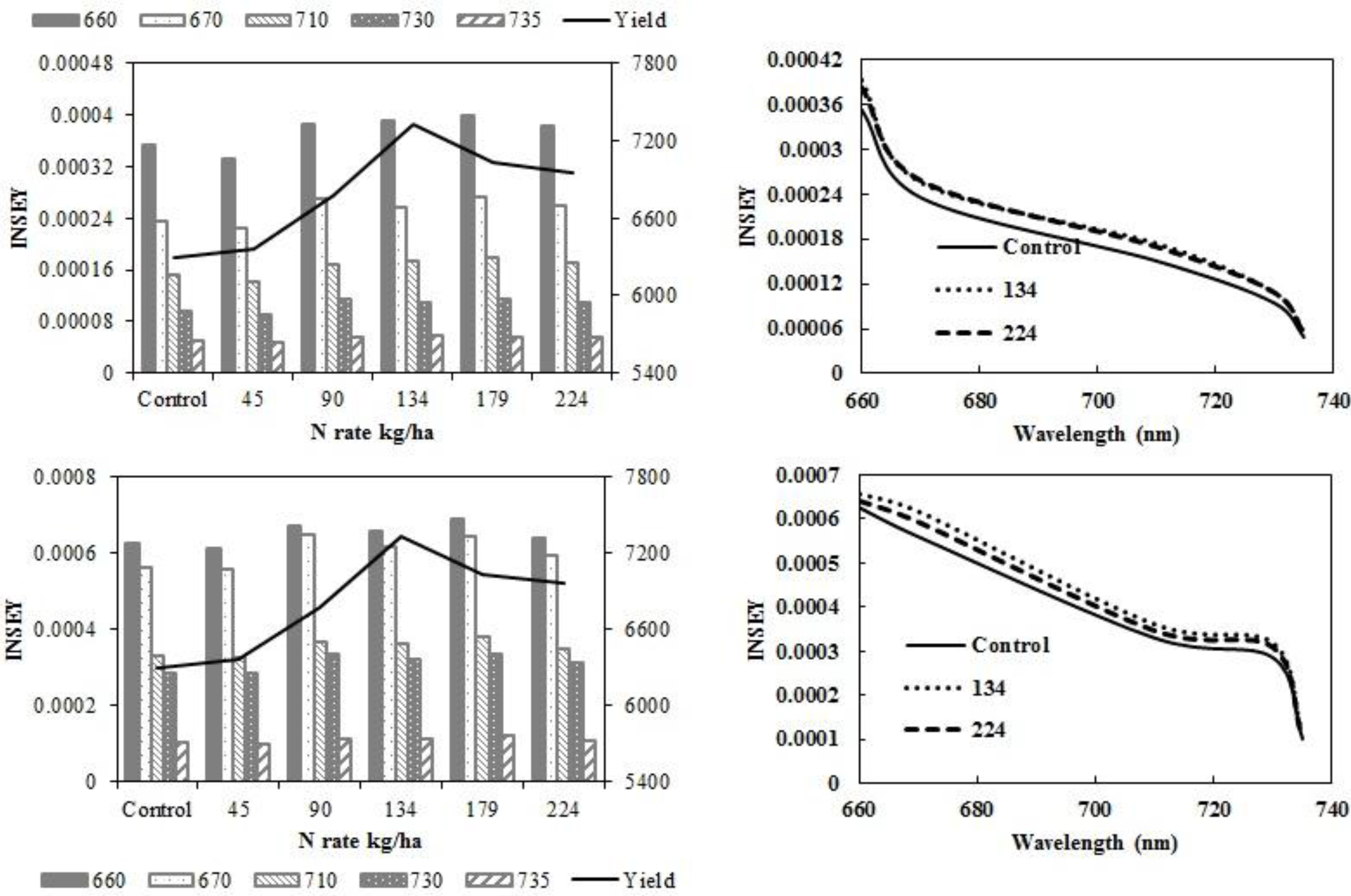
In eastern ND no-till sites, yield did not increase with N rate. At V6, there was no relationship between INSEY and yield (
Table 5) or INSEY or N rate (
Table 7). Yield trends depicted in
Figure 4a,c are not significant. At V12, the r
2 between CC 730 nm NDVI derived INSEY and yield and the GS 735 nm NDVI derived INSEY and yield were significant. The INSEY from all wavelengths of both sensors at V12 was also related to N rate (
Table 7).
Table 7.
Treatment differences between INSEY developed from five wavelengths utilizing two ground-based active-optical sensors in eastern North Dakota (ND) (n = 47) and western ND no-till (n = 95) systems at two corn growth stages from nitrogen rate treatments.
Table 7.
Treatment differences between INSEY developed from five wavelengths utilizing two ground-based active-optical sensors in eastern North Dakota (ND) (n = 47) and western ND no-till (n = 95) systems at two corn growth stages from nitrogen rate treatments.
| Soil Category | Growth Stage | N Treatment kg·N·ha−1 | INSEY † X 10,000 |
|---|
| Holland Crop Circle | GreenSeeker |
|---|
| Red Edge 730 | Red 670 | Red 660 | Red Edge 710 | Red Edge 735 |
|---|
| Eastern ND | V6 | 0 | 1.65 a | 3.65 a | 4.83 a | 2.28 a | 0.83 a |
| 45 | 1.72 a | 3.73 a | 5.03 a | 2.35 a | 0.83 a |
| 90 | 1.52 a | 3.46 a | 4.67 a | 2.12 a | 0.76 a |
| 135 | 1.70 a | 3.72 a | 4.85 a | 2.23 a | 0.76 a |
| 179 | 1.58 a | 3.52a | 4.65 a | 2.09 a | 0.73 a |
| 224 | 1.65 a | 3.66 a | 4.89 a | 2.28 a | 0.76 a |
| V12 | 0 | 3.74 c | 7.31 b | 7.22 b | 4.21 b | 1.29 c |
| 45 | 3.97 bc | 7.57 ab | 7.38 ab | 4.37 ab | 1.36 bc |
| 90 | 3.99 bc | 7.61 ab | 7.55 ab | 4.52 ab | 1.42 abc |
| 135 | 4.09 ab | 7.60 ab | 7.52 ab | 4.54 ab | 1.47 ab |
| 179 | 4.16 ab | 7.74 ab | 7.55 ab | 4.54 ab | 1.46 abc |
| 224 | 4.31 a | 7.96 a | 7.70 a | 4.73 a | 1.54 a |
| Western ND | V6 | 0 | 9.45 a | 2.35 a | 3.53 a | 1.51 a | 4.88 a |
| 45 | 9.07 a | 2.26 a | 3.33 a | 1.41 a | 4.74 a |
| 90 | 1.15 a | 2.69 a | 3.85 a | 1.68 a | 5.40 a |
| 135 | 1.09 a | 2.56 a | 3.92 a | 1.74 a | 5.68 a |
| 179 | 1.14 a | 2.73 a | 3.98 a | 1.79 a | 5.63 a |
| 224 | 1.08 a | 2.58 a | 3.83 a | 1.69 a | 5.40 a |
| V12 | 0 | 2.85 a | 5.61 a | 6.27 a | 3.30 a | 1.01 a |
| 45 | 2.85 a | 5.58 a | 6.12 a | 3.26 a | 1.00a |
| 90 | 3.33 a | 6.48 a | 6.70 a | 3.64 a | 1.12 a |
| 135 | 3.19 a | 6.16 a | 6.56 a | 3.61 a | 1.14 a |
| 179 | 3.36 a | 6.45 a | 6.87 a | 3.80 a | 1.20 a |
| 224 | 3.10 a | 5.92 a | 6.40 a | 3.47 a | 1.08 a |
Figure 4.
Variation of INSEY for corn at eastern no-till sites calculated from CC active-sensor spectra (670 and 730 nm) and GS spectra (660, 710 and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg·ha−1 and 224 kg·ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).
Figure 4.
Variation of INSEY for corn at eastern no-till sites calculated from CC active-sensor spectra (670 and 730 nm) and GS spectra (660, 710 and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg·ha−1 and 224 kg·ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).
At western ND no-till sites, there was no response to N. However, there were significant relationships between all wavelengths of both sensors and yield (
Table 4), and N rate at V12 (
Table 7). The sensitivity of INSEY to yield was low at V6 and V12 in western ND no-till sites. The lack of relationship was particularly evident at the lower N rates (
Figure 5).
Figure 5.
Variation of INSEY for corn at western ND no-till sites calculated from CC active-sensor spectra (670 and 730 nm) and GS spectra (660, 710 and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg·ha−1 and 224 kg·ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).
Figure 5.
Variation of INSEY for corn at western ND no-till sites calculated from CC active-sensor spectra (670 and 730 nm) and GS spectra (660, 710 and 735 nm) with N rate compared to yield variation with N rate at V6 (upper left) and V12 (lower left). Trend for INSEY at three N rates (control, 134 kg·ha−1 and 224 kg·ha−1) over the five wavelengths represented as a continuous spectrum is displayed at V6 (upper right) and at V12 (lower right).