New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria
Abstract
: The development of a rapid, sensitive, specific method for the foodborne pathogenic bacteria detection is of great importance to ensure food safety and security. In recent years impedimetric biosensors which integrate biological recognition technology and impedance have gained widespread application in the field of bacteria detection. This paper presents an overview on the progress and application of impedimetric biosensors for detection of foodborne pathogenic bacteria, particularly the new trends in the past few years, including the new specific bio-recognition elements such as bacteriophage and lectin, the use of nanomaterials and microfluidics techniques. The applications of these new materials or techniques have provided unprecedented opportunities for the development of high-performance impedance bacteria biosensors. The significant developments of impedimetric biosensors for bacteria detection in the last five years have been reviewed according to the classification of with or without specific bio-recognition element. In addition, some microfluidics systems, which were used in the construction of impedimetric biosensors to improve analytical performance, are introduced in this review.1. Introduction
In recent years, diseases and productivity losses caused by foodborne pathogenic bacteria have attracted considerable attention. Thousands of foodborne pathogenic bacteria have been found to affect the health and safety of the world’s populations of humans, animals and plants. Among these bacteria, Campylobacter, Salmonella, Listeria monocytogenes, Escherichia coli (E. coli) O157:H7, Staphylococcus aureus, and Bacillus cereus are the major foodborne pathogen bacteria, which are responsible for the majority of foodborne illness outbreaks [1–5]. Therefore, it is of great importance to develop methods for foodborne pathogenic bacteria detection.
Several methods have been explored for the bacteria determination, including the culture and colony counting method, polymerase chain reaction (PCR), and immunology-based method [6–10]. The traditional culture and colony counting method has been a practical for the detection and identification of pathogens in food, including microbiological culturing and isolation of the pathogen, followed by confirmation by biochemical and serological tests, which takes up to 5–7 days to get a result [11]. Although it can obtain reliable result, it is labor intensive and time consuming, which cannot satisfy the request for bacteria detection on-the-spot detection. The PCR and enzyme-linked immunosorbent assay (ELISA) are a lot less time-consuming than the traditional culture and colony counting method, which usually takes 30 mins or a few hours to achieve detection result [9,12]. However, there are still key issues that need to be considered in the development of rapid methods for the detection of foodborne pathogens, including differentiation of live and dead cells, automation, cost, simplicity, training, and accuracy.
Impedance technique, as one kind of the electrochemical biosensors, has been proved to be a promising method for foodborne pathogenic bacteria detection due to its portability, rapidity, sensitivity, and more importantly it could be used for on-the-spot detection [13–16]. Generally, the impedance detection techniques can be classified into two types depending on the presence or absence of specific bio-recognition elements. The first type works by measuring the impedance change caused by binding of targets to bioreceptors (antibodies and nucleic acids) immobilized onto the electrode surface, while the detection principle of the second type is based on metabolites produced by bacterial cells as a result of growth. The articles about impedance biosensors for bacteria detection before 2007 have been reviewed comprehensibly [11], however, in the last five years some new trends in this area have emerged, including the use of nanomaterials, microfluidics techniques and new specific bio-recognition elements such as bacteriophage and lectin. The applications of these new materials or techniques have provided unprecedented opportunities for the development of high-performance impedance bacteria biosensors. Nanomaterials in particular have exhibited unique advantages for constructing impedimetric biosensors and there are an abundance of research articles about that topic, so in this paper, we will focus on those new trends in the development of impedance bacteria biosensor. The significant developments of impedimetric biosensors for bacteria detection in the past five years have been reviewed according to the classification of with or without specific bio-recognition element. In addition, some microfluidics systems, which were used in the construction of impedimetric biosensors to improve analytical performance, have been covered in this review.
2. Principle of Impedance Technique
Electrical impedance (Z) is defined as the ratio V(t)/I(t) of an incremental change in voltage to the resulting change in current. From this definition, the impedance Z is the quotient of the voltage-time function V(t) and the resulting current−time function I(t):
Electrochemical impedance spectroscopy (EIS) is a method that describes the response of an electrochemical cell to a small amplitude sinusoidal voltage signal as function of frequency [18]. It is an ideal tool for observing the dynamics of biomolecule interactions [19]. The most popular formats for evaluating EIS data are the Nyquist and Bode plots. In the Nyquist plot, the imaginary impedance component (z″) is plotted against the real impedance component (z′). In the Bode plot, both the logarithm of the absolute impedance (|Z|) and the phase shift (φ) are plotted against the logarithm of the excitation frequency.
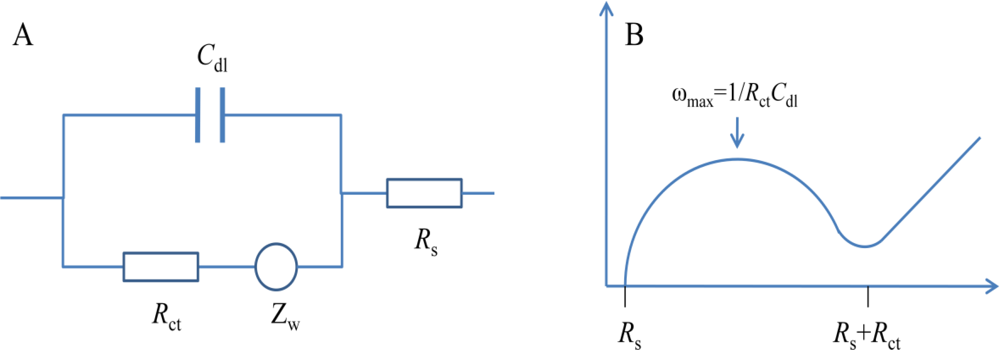
In order to express the characterization of surfaces, layers or membranes after the immobilization of biomolecules and bacteria binding, EIS is often analyzed using an equivalent circuit which is used to curve fit the experimental data and extract the necessary information about the electrical parameters responsible for the impedance change [17]. Since the electrochemical cell is a complex system, more than one circuit model can fit the experimental data [20]. The simplest, and in fact the most frequently used equivalent circuit for modelling the EIS experimental data is the so-called Randles circuit (Figure 1(A)), which comprises the uncompensated resistance of the electrolyte (Rs), in series with the capacitance of the dielectric layer (Cdl), the charge-transfer resistance (Rct) and the Warburg impedance (Zw) [18]. In the Nyquist plot shown in Figure 1(B), a typical shape of a Nyquist plot includes a semicircle region lying on the real axis followed by a straight line. The linear part (ψ = π/4), observed at the low frequency range, implies a mass-transfer limited process, whereas the semicircle portion, observed at high frequency range, implies a charge-transfer limited process. From the Nyquist plot, the values for Rs and Rct can be easily determined. The double layer capacitance can be calculated from the frequency at the maximum of the semicircle (ω = 2πf = 1/RctCdl). The charge-transfer resistance Rct and the double layer capacitance Cdl are the most important electrical parameters in analyzing the impedance signal change for detection of bacteria.
3. Types of Impedance Detection Techniques for Foodborne Pathogenic Bacteria Detection
3.1. Detection Based on the Use of Specific Bio-Recognition Element
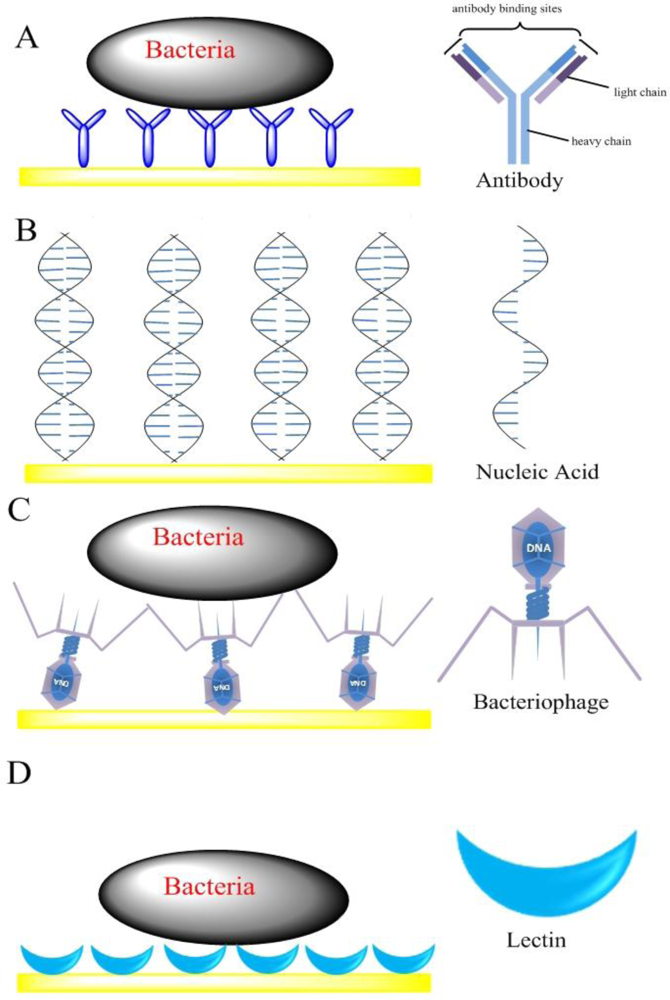
Impedimetric biosensors have been designed by immobilizing bioreceptors (such as antibodies, nucleic acids, bacteriophages and lectins) at the surface of a solid electrode. The binding ability of bacteria and the bioreceptors is then verified through the detection of either a shift in impedance, or change in capacitance or admittance at the bulk of the electrode interface due to the insulating properties [21]. The bacterial cell membrane consists of a lipid bilayer, where the lipid molecules are oriented with their polar groups facing outwards into the aqueous environment, and their hydrophobic hydrocarbon chains pointing inwards to form the membrane interior. Pethig reported that natural cell membranes (thickness 5–10 nm) show a membrane capacitance of 0.5–1.3 μF/cm2 and a membrane resistance of 102–105 Ω·cm2. If bacterial cells attach on an electrode surface, they would effectively reduce the electrode area that the current reaches and hence increases the interface impedance. Here, according to the types of bioreceptors, the impedimetric biosensors were classified into four different categories, including antibody-based sensors, nucleic acid-based sensors, bacteriophage-based sensors and lectin-based sensors (Figure 2).
3.1.1. Antibody Sensors
Impedimetric biosensors based on directly immobilizing antibodies on the surface of an electrode for the detection of bacteria, called impedimetric immunosensors, are constructed by immobilizing antibodies on the electrode surface, and then probing the attachment of the bacterial cells by measuring the change in electrical properties over a range of frequency due to the insulating properties of the cell membrane [11]. Antibodies have long been the most popular bio-recognition elements. The main advantage of the use of antibodies as bio-recognition elements is their sensitivity and selectivity. A wide variety of impedimetric immunosensors reported for different bacteria detection applications exists in the last five years [22,23].
There are some main means for improving impedimetric immunosensors efficiency: (I) improving immobilization methods of antibody on the electrode surface; (II) improving electrode performance to enhance sensitivity; (III) using enzyme-labeled and nanomaterials to amplify detection signal; (IV) optimal equivalent circuit for analyzing impedance change; (V) the dielectrophoresis technique for concentrating samples.
The immobilization method is the key process in the construction of impedimetric biosensors, since the efficiency of antibody immobilization on the electrode surface can profoundly affect the analytical performance of impedance biosensors. There are several methods for the immobilization of antibodies on the electrodes, including physical adsorption, self-assembled monolayer (SAM) and biotin-streptavidin system. Physical adsorption is the simplest and straightforward immobilization method that depends on the non-specific interactions of the biomolecules with the solid substrate. These non-specific interactions contain various non-covalent bridges, such as ionic and hydrogen bonds, hydrophobic interactions, and van der Waals forces. Yang et al. [24] developed a label-free electrochemical impedance immunosensor by physical adsorption method to immobilize anti-E. coli antibodies onto an indium-tin oxide interdigitated array microelectrode (IDAM) for detection of E. coli O157. The equivalent circuit consisted of an ohmic resistor of the electrolyte between two electrodes, double layer capacitor, an electron-transfer resistor, and Warburg impedance around each electrode. Experimental data fitting to the equivalent circuit showed that the electron transfer resistance and electrolyte resistance were responsible for the detection of E. coli O157:H7 cells. The detection range of the biosensor was from 4.3 × 105–4.36 × 108 cfu·mL−1 with the detection limit of 106 cfu mL−1. In spite of its convenience, this method is generally restricted to a limited number of applications due to low stability and the random orientation of the bio-recognition elements immobilizing on electrodes, resulted in the decrease of the binding bacteria. SAM is considered an ideal method to immobilize the antibodies in the construction of impedimetric immunosensors for bacteria detection [18,25–29], which provides a convenient and flexible route to generate ultrathin and ordered biological monomolecular films on a variety of substrates by organic molecules (both aliphatic and aromatic) containing free anchor groups such as thiols, disulphides, amines, silanes, or acids [29,30]. Geng et al. [27] used mercaptoacetic acid to form SAM for immobilizing the anti-E. coli antibodies on an Au electrode. The immobilization of antibodies on the SAM was carried out through a stable acyl amino ester intermediate generated by EDC and NHS, which can facilitate the formation of a suitable intermediate to condense antibodies on the SAM and enhance the stability sensitivity of the developed immunosensor. A linear relationship between the electron-transfer resistance and the logarithmic value of E. coli concentration was found in the range of E. coli cells from 3.0 × 103–3.0 × 107 cfu·mL−1 with the detection limit of 1.0 × 103 cfu·mL−1. However, the SAM immobilization method still suffers from some drawbacks, such as electric field induced and thermal desorption of monolayer and nonspecific adsorption due to high surface energy. Since the biotin-strept(avidin) system has high binding capacity for antibody immobilization due to the high affinity constant between streptavidin and biotin, it is also widely used to immobilize antibodies on solid support interface to construct impedimetric immunosensors. Barreiros et al. [25] compared the effect of two different antibody immobilization strategies: one is the use of chemical bond formation between antibody amino groups and a carboxylic acid-containing SAM molecule, and the other is based on linking a biotinylated anti-E. coli to avidin on a mixed-SAM. Very low concentrations of E. coli (10–100 cfu·mL−1) can be detected with the biosensors fabricated by the above design strategies. Though the biotin-strept(avidin) system is an effective method to immobilize the bio-recognition elements on the surface of solid supports, this method has some disadvantages such as the high cost of the reagents involved and the need for a suitable linker layer such as SAM in some cases to attach to the electrode.
Traditionally, three or four macro-sized metal electrodes system is used to measure impedance. With the development of minimization techniques, microelectrodes have been used in fabrication of impedimetric immunosensors due to the miniaturization of the sensor and improvement of the sensitivity [24,31]. Among these techniques, IDAM which has some advantages over the macro-sized electrode, including detecting small amounts of generated electrode products, eliminating the need for a reference electrode, providing simple means for obtaining a steady-state current response, and low response times, has been widely employed to fabricate impedimetric immunosensors [20]. According to the previous research, when the electrode bands become narrower, the biosensor becomes more sensitive. Stephen and coworkers [32] immobilized polyclonal antibodies onto an interdigitated gold electrode array. Each sensor chip had a total of 1,700 electrodes to form a large active area of 9.6 mm2. Each electrode finger had a length of 750 μm, a width of 3 μm and an in-between spacing of 4 μm. Each sensor was diced to a dimension of 12 mm × 8 mm. The biosensor was able to discriminate between cellular concentrations of 104–107 cfu·mL−1. At present, the commercial company producing IDAM for impedance detection is ABTECH Scientific, Inc.
In addition to minimizing the size of electrodes, some new electrode materials are used to construct impedimetric immunosensors, which enhance the performance of the biosensors for bacteria detection. It is reported that the electrode made from macroporous silicon (3D) structure could be used as the efficient trapping platform for bacteria detection, and the obtained sensitivity was found to be more sensitive than a planar (2D) sensor. Wan et al. [28] developed a 3D-immunosensor based on antibody-functionalized 3D-foam Ni substrate as the trapping platform for detection of sulfate-reducing bacteria using impedimetric technique, which can detect the sulfate-reducing bacteria concentration range of 2.1 × 101–2.1 × 107 cfu·mL−1.
In order to amplify the detection signal and achieve lower detection limits, the main interaction signal may be enhanced by case-specific amplification schemes, like enzyme-labeled amplification. Ruan et al. [33] reported an impedimetric immunosensor for bacteria detection using horseradish peroxide enzyme-labeled for signal amplification. After binding the bacterial cells, secondary antibodies with horseradish peroxide were used to produce precipitation of insoluble products on the electrode surface building thus a rather insulating layer in order to amplify impedance signal due to blocking the electron transfer. A linear response in the Rct for the concentration of E. coli cells was found between 6 × 104 and 6 × 107 cfu mL−1 and the detection limit was 6 × 103 cfu·mL−1.
The equivalent circuit used to curve fit the experimental data and extract the necessary information about the electrical parameters responsible for the impedance change is of great importance to analyze the EIS. Some efforts have been devoted into optimize the equivalent circuit. RoyChaudhuri et al. [34] developed a biomolecule compatible electrical model to establish a rapid and cost effective method for quantification of antibodies immobilized and bacteria captured which can be applied for the standardization of any new developing technique for improving immobilization and capture efficiency. The model had been applied to retrieve the information about actual number of antibodies immobilized on the electrode surface and the actual concentration range of E. coli K12 bacteria captured on the gold surface, which are 15.96 × 1010 and 106–103 cfu·mL−1, respectively.
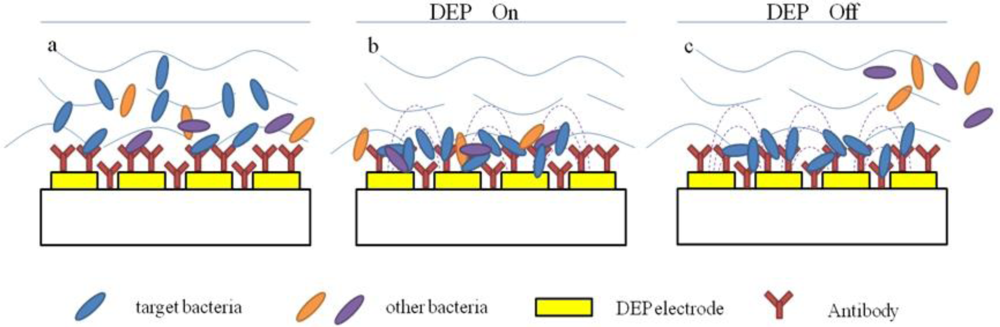
Recently, some researchers have combined the dielectrophoretic impedance measurement (DEPIM) technique with impedimetric biosensors for bacteria detection (Figure 3) [35,36]. The DEPIM utilizes the positive dielectrophoretic force to trap suspended biological cells onto the electrode in the form of pearl chains and then measured an impedance signal [37–39], which can detect bacteria in shorter time than traditional impedance methods due to the effect of dielectrophoretic force. Suehiro et al. [40] developed a selective detection method for specific bacteria by using a DEPIM method in conjunction with an antigen-antibody reaction. Antibodies were immobilized on the electrode chip before the preliminary bacteria trapping by positive dielectrophoresis (DEP). The bacteria were attracted to the electrode gap under the action of the positive DEP force and finally brought into contact with the glass surface to be bound with the immobilized antibodies. It was also confirmed that the proposed method realized selective detection of the target bacteria from a mixed suspension with non-target bacteria.
3.1.2. Nucleic Acid Sensors
In recent years, nucleic acid analysis has become an important tool for identification of disease-causing microorganisms in food and environment [41–45]. In the case of nucleic acid as bioreceptor for pathogen detection, the identification of a target analyte’s nucleic acid is achieved by matching the complementary base pairs that are often the genetic components of an organism. Since each organism has unique nucleic acid sequences, any self-replicating microorganism can be easily identified [4]. Compared to antibody, the biological recognition layers formed by nucleic acid have many advantages. First, nucleic acids can be chemically synthesized with high purity, avoiding batch-to-batch variation. Second, during synthesis they can be chemically modified with some functional groups, like −HS, −NH2, biotin, and so on, which can be easily immobilized onto the electrode surface. Third, the nucleic acid is highly stable and reusable after simple thermal melting of the DNA duplex, which is suitable for biosensor regeneration.
Due to their wide range of physical, chemical and biological activities, nucleic acid based biosensors have been reported by many researchers for the detection of food pathogens [4]. Commonly, nucleic acid based impedimetric biosensors contain immobilized nucleic acid probes that specifically hybridize to their complementary sequences in bacteria samples and an impedance transducer which transforms biomolecule recognition signal into an impedance signal (Figure 2(B)) [16]. Pinar et al. [46] developed nucleic acid based impedimetric biosensors for rapid and selective detection of Bacillus anthracis (B. anthracis). An alkanathiol-linked or unlabeled capture probe related to B. anthracis was immobilized onto gold or graphite electrode surface. The extent of hybridization between probe and target sequences was determined by using EIS. EIS analysis was based on Rct in the presence of [Fe(CN)6]3−/4− and Meldola’s Blue reduction signal as hybridization indicator. The method provided a highly sensitive detection of DNA of 1 × 104 copies (about 1.7 × 10−20 mol) of original genomic HBV DNA by combining a PCR procedure.
Although it is undeniable that nucleic acid based impedimetric biosensors have played an increasingly important role in the field of bacteria detection on site applications, they still suffer from some drawbacks. For example, EIS signals resulting from nucleic acid-based impedimetric biosensors are remarkably affected by repulsions between the negatively charged phosphate backbone and redox anions such as [Fe(CN)6]3−/4− that make the quantitation of DNA hybridization reactions rather difficult. Moreover, nucleic acid-based methods are unable to discriminate between viable and nonviable cells.
3.1.3. Bacteriophage Sensors
Bacteriophages are viruses, which are made of an outer protein coat that encases genetic material (DNA or RNA) [47]. They can recognize specific sites on the bacterium surface to which they bind and inject genetic material (Figure 2(C)). Since the recognition is highly specific, it can be used for the typing of bacteria [48–50]. Bacteriophages have several desirable advantages for the development of a real-time sensor to rapidly and selectively detect target bacteria in a variety of harsh conditions, such as under acidic or basic pH ranges, and even in the presence of nucleases or proteolytic enzymes. In addition, bacteriophages are not only more cost-effective than antibodies, but also more amenable than antibodies to manipulation at the molecular level to improve their interaction with bacteria [51].
Due to these advantages, the bacteriophages are ideal bioreceptors to make impedimetric biosensors for bacteria detection [52–54]. An example can be found in the determination of E. coli bacteria by covalently immobilization of T4 bacteriophages onto functionalized screen-printed carbon electrodes. The Rct undergoes a decrease with increasing bacteria concentration ranging from 102 to 108 cfu·mL−1, which is contrary to what is usually observed for simple attachment of intact bacteria cells to an electrode surface in impedimetric immunosensors (an increase of Rct with increasing concentration of intact bacteria at the surface). Since the lysis of bacteria resulting from the attack of bacteriophage could lead to the release of highly mobile ionic material (such as K+ and Na+), the conductivity of the media near the electrode surface was increased. Correspondingly, the values related to Rct show a clear decrease with increasing concentration of E. coli cells. The bacteriophage impedance biosensor showed excellent specificity for target bacteria E. coli with a detection limit of 104 cfu mL−1, and no significant change in impedance was observed in the presence of Salmonella [55]. Gervais et al. [56] developed an impedimetric biosensor for E. coli detection based bacteriophages immobilized on gold surfaces through biotin/streptavidin system. Such chemical attachment of bacteriophages onto sensor surfaces could in turn be leveraged in highly sensitive and more rapid transduction platforms such as surface plasmon resonance (SPR), quartz crystal microblance (QCM), and microcantilevers. Webster et al. [57] developed an impedimetric microelectrode array biosensor based bacteriophage for the detection of bacteria. The results indicated that reducing the width and gap of electrode and using the working solution with lower relative dielectric permittivity can increase the sensitivity of impedimetric biosensors for pathogenic bacteria.
3.1.4. Lectin Sensors
More recently, the use of lectin as the bioreceptor in biosensors has been proven to be very promising and effective. Lectins are plant or animal proteins or glycoproteins, which can bind selectively and reversibly with mono- and oligosaccharide components of polysaccharide structures that are major structural components of bacterial cells surfaces. Recognition of these carbohydrates on the surface of bacteria can be used for specific identification of target bacteria [58,59]. Such a recognition system is superior to antibody or nucleic acid based systems, since the latter systems always require a prior knowledge on the target and specific reagents, which become increasingly problematic when the identities of which are unknown [60]. Furthermore, the molecule size of lectins are much smaller than antibodies, thus they allow higher densities of carbohydrate-sensing elements leading to higher sensitivity and lower non-specific adsorption [61,62]. Finally, agglutination between the lectins and bacteria occurs quickly. Gamella et al. [61] reported the lectin modified screen-printed gold electrodes for the impedimetric label-free detection of E. coli bacteria. The biotinylated lectins were immobilized on the gold electrode, and then the selectively binding between bacteria and lectins was determined by EIS. The impedance biosensor showed a good performance with a detection range of 5.0 × 103 and 5.0 × 107 cfu·mL−1. A similar approach was used to detect sulfate-reducing bacteria by Wan et al. [63]. The lectin-concanavalin A as the bioreceptor was assembled on the gold electrode with 11-mercaptoundecanoic acid to bind sulfate-reducing bacteria. The lectin-based impedance biosensor exhibited good performance for sulfate-reducing bacteria detection with a concentration range of 1.8 to 1.8 × 107 cfu mL−1.
3.2. Detection Based on Metabolites Produced by Bacterial Cells as a Result of Growth
This method is based on the measurement of changes in electrical impedance of a culture medium or a reaction solution resulting from the bacterial growth. The impedance change in the medium is mainly produced by the release of ionic metabolites from the live cells, thus it could distinguish between viable and dead cells. Such a method has been developed as a rapid method that can detect bacteria within 24 h. Several commercial analytical instruments are based on this principle. These systems include the Bactometer (BioMerieux, Nuertingen, Germany), the Malthus system (Malthus Instruments Ltd., Crawley, UK), The Rapid Automated Bacterial Impedance Technique (RABIT; Don Whitley Scientific Ltd., Shipley, UK), and the Bac-Trac (Sy-Lab, Purkersdorf, Austria) [11]. However, these measurement systems are not suitable for on-the-spot applications, so many efforts have been made to minimize the instruments. Grossi et al. [64] developed an embedded portable biosensor system for the determination of bacterial concentration. This system is composed of an incubation chamber, containing the sample under test, and two electronic boards: one dedicated to measuring the sample electrical characteristics, the other controlling the sample temperature, fixed at a value suitable to enhance bacterial growth. Such a biosensor configuration could truly realize the miniaturization and portability. Kim et al. [65] proposed a plug-type, disposable electrode using a gold-coated silicon wafer, PDMS polymer, and a borosilicate glass tube to construct an impedimetric biosensor instrument. The developed biosensor could be used for in situ real-time monitoring of bacterial growth in a lab-scale fermentor by measuring impedance signals without the risk of introducing contamination.
Over time, much work has been done in the field of medium engineering, since the direct impedance microbiology is based on the monitoring of impedance change in the medium. The ideal medium should not only support the selective growth of the target bacteria, but also provide optimal impedance signals. For instance, one can predict that the weakly buffered media would allow a greater conductance change than the strongly buffered media. Banada et al. [66] used a low conductive growth medium for growth and detection of Listeria monocytogenes with an impedance-based microfluidics biochip detection platform. This kind of medium was suitable for growth of Listeria monocytogenes and the low conductive characteristic was suitable for getting greater impedance signal change due to low threshold in the variation of the impedance signal. Choi et al. [67] firstly attempt to use solid medium and two plane electrodes attached on two facing sides of an acryl well to fabricate an impedimetric biosensor for real-time monitoring of microorganisms. Compared to liquid medium, solid medium has advantages in that it is easy to handle and portable.
4. Nanomaterials
Nanomaterials, an emerging subdiscipline in chemistry have been used in impedimetric biosensors to amplify detection signal and achieve lower detection limit due to their high surface area, favorable electronic properties and electrocatalytic activity as well as good biocompatibility induced by the nanometer size and specific physicochemical characteristics [68,69]. Until now, nanomaterials [70], including metal nanoparticles, nanowires, nanorods, carbon nanotubes, and graphene, have been successfully used for constructing impedimetric biosensors for bacteria determination with enhanced analytical performance (Table 1).
In the published work, gold (Au) nanoparticles have received extensive attention in view of their easy synthesis and good stability in aqueous solution. Many efforts have been made to explore Au nanomaterials-based impedimetric biosensors. Yang et al. [71] reported a capacitive immunosensor for the detection of Salmonella spp. which was fabricated by immobilizing a Au nanoparticles monolayer onto a glassy carbon electrode and then the Salmonella monoclonal antibodies through physical adsorption. It was found that the Au nanoparticles can effectively improve the sensitivity and stability of the immunosensors, which can detect the Salmonella spp. concentrations in the range of 1.0 × 102 to 1.0 × 105 cfu·mL−1 (R = 0.991) with the detection limit of 1.0 × 102 cfu·mL−1. The stability of immunosensor remained almost the same after two months storage.
In addition to Au nanoparticles, metal-oxide nanoparticles which possess high surface area and thermally stable, chemically inert, non-toxic inorganic oxide, have been also used in the development of bacteria biosensors. Huang et al. [69] used Fe3O4 nanoparticles to immobilize monoclonal antibodies in the construction of electrochemical impedimetric immunosensors for the rapid detection of Campylobacter jejuni. The Fe3O4 nanoparticles-based immunosensor showed good performance with respect to simplicity of use, fast response, wide linear range, acceptable reproducibility and long stability.
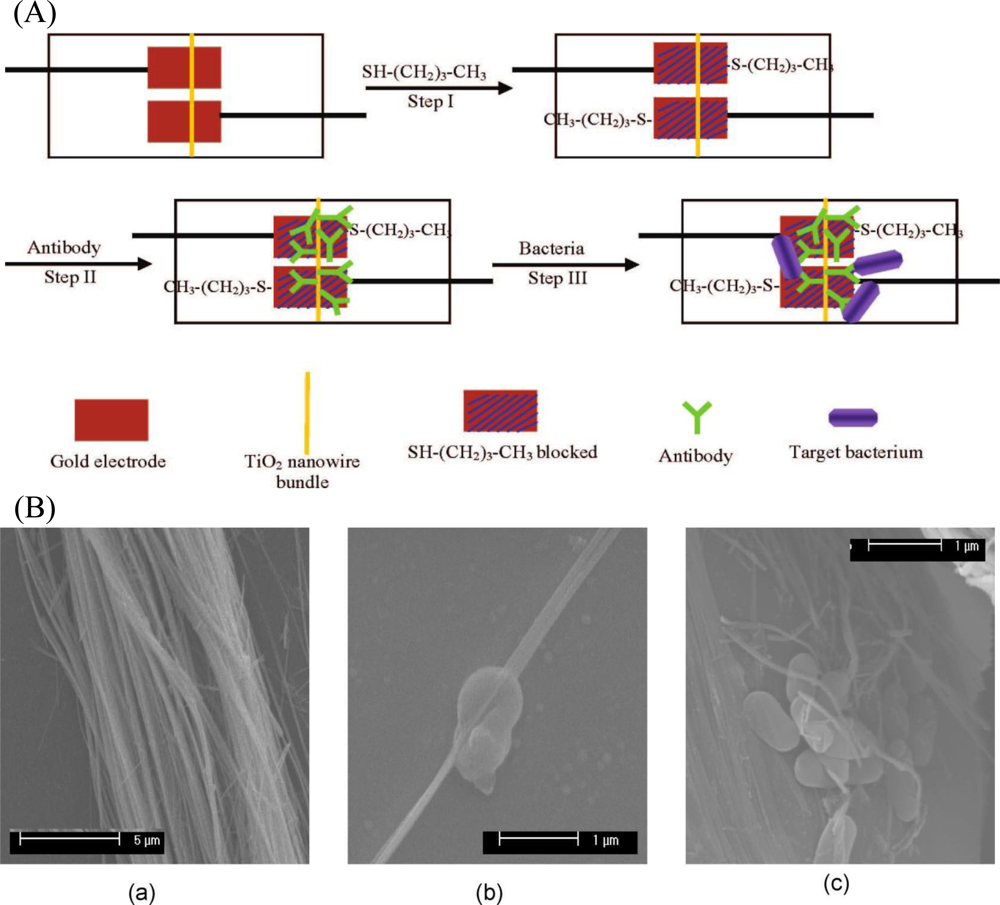
In addition to nanoparticles, nanowires have been attracted much scientific interest in analytical chemistry, especially in biosensing technologies. This is due to their unique semiconductive properties associated with the nanostructures, and they are believed to be ultrasensitive in performing single molecule sensing. Wang et al. [72] developed a TiO2 nanowire bundle microelectrode based impedimetric immunosensor for rapid and sensitive detection of Listeria monocytogenes. TiO2 nanowire bundle was connected to gold microelectrodes using mask welding and then monoclonal antibodies were immobilized on the surface of a TiO2 nanowire bundle to specifically capture bacteria (Figure 4). Impedance changes caused by the nanowire-antibody-bacteria complex were measured and correlated to bacterial number. Since the TiO2 nanowires can be highly oriented on substrates or form free-standing membranes, the fabricated electrode showed a large specific surface area, good biocompatibility, good chemical and photochemical stabilities, and negligible protein denaturation. This nanowire bundle based immunosensor also exhibited a good performance that can detect as low as 102 cfu·mL−1 of Listeria monocytogenes in 1 h without significant interference from other foodborne pathogens.
In recent years, reduced graphene sheets (RGSs), which are monolayers of carbon atoms packed into a dense honeycomb crystal structure, have been drawn tremendous attention from both the experimental and theoretical scientific communities. This unique nanostructure exhibits excellent electrical conductivity, mechanical strength, and chemical stability, which make it quite promising for the design of high sensitive and selective biosensors. Wan et al. [73] developed a RGSs-doped impedimetric immunosensor through a controllable electrodeposition method using soluble RGSs-doped CS solution for the facile and rapid detection of sulfate-reducing bacteria. They used RGSs as electron conductors to obtain good analytical performance, namely, sensitivity, selectivity, and stability, of the biosensor towards the detection of pathogen. The RGSs based immunosensor can detect the sulfate-reducing bacteria at the concentration range of 1.8 × 101 to 1.8 × 107 cfu·mL−1 and give a distinct response to sulfate-reducing bacteria without obvious response to Vibrio angillarum.
In addition, nanopore membrane materials such as aluminum anodized oxide nanopore membranes were used for immobilizing bioreceptors to construct impedance biosensors. Wang et al. [74] developed an impedimetric biosensor based on dynamic polymerase-extending hybridization for E. coli O157:H7 DNA detection. They immobilized ssDNA probe onto functional aluminum anodized oxide nanopore membranes. The probe strand would be extended as long as the target DNA strand, then the capability to block the ionic flow in the pores could be prominently enhanced by the double strand complex. This approach provides much lower detection limit for DNA (a few hundreds of pmol), rapid label-free and easy-to-use bacteria detection, which holds the potential for future use in various ssDNA analyses by integrated into a self-contained biochip.
Nanoscale magnetic materials have shown unique advantages that provide many exciting opportunities in bacteria detection applications. First, they can enhance the efficiency of immobilization of biofunctional molecules (e.g., antibodies, or ligands) due to their high specific surface area. Second, the nanoparticles can be manipulated by an external magnetic force, therefore, they can separate and concentrate bacteria from crude samples before impedance detection, which can detect bacteria at ultralow concentrations without time-consuming procedures and reduce the background noise ratio caused by the non-target components in the sample. Third, they improve the utilization of electrode as there is no bio-recognition biomolecule directly immobilized on the electrode surface. Due to the above advantages, many researchers have used the bio-recognition elements immobilizing on magnetic beads to separate and concentrate bacteria in samples, and then the combination complexes of the biofunctional magnetic nanoparticles and bacteria were measured as an impedance signal [75–79].
An example can be found in the determination of Salmonella typhimurium. Anti-Salmonella antibodies were coated with immunomagnetic beads to separate Salmonella typhimurium from samples. Then the concentrated sample was spread on the surface of electrodes to detect impedance signal over a range of frequency. A linear relationship between the detection time and the logarithmic value of the initial cell number was found in the Salmonella cell number ranging from 101 to 106 cfu mL−1 [80]. Madhukar et al. [81] developed an impedance biosensor based on IDAM coupled with magnetic nanoparticles-antibody conjugates (MNAC) for rapid and specific detection of E. coli O157:H7 in ground beef samples. MNAC were prepared by immobilizing biotin-labeled polyclonal goat anti-E. coli antibodies onto streptavidin-coated magnetic nanoparticles, which were used to separate and concentrate E. coli O157:H7 from ground beef samples. Magnitude of impedance and phase angle were measured in a frequency range of 10 Hz to 1 MHz in the presence of 0.1 M mannitol solution. The equivalent circuit analysis showed that bulk resistance and double layer capacitance were responsible for the impedance change caused by the presence of E. coli O157:H7 on the surface of IDAM. The lowest detection limits of this biosensor for detection of E. coli O157:H7 in pure culture and ground beef samples were 7.4 × 104 and 8.0 × 105 cfu·mL−1.
5. Microfluidics Techniques
Besides nanomaterials, microfluidics techniques are a good strategy for improving the performance of impedimetric bacteria biosensors [86–94]. Microfluidics techniques in general seek to improve analytical performance by reducing the consumption of reagents, decreasing the analysis time, increasing reliability and sensitivity through automation, and integrating multiple processes in a single device. These features are particularly suitable for hand-held impedance biosensors for bacteria detection [95]. Varshney et al. [84] integrated a microfluidics flow cell with embedded gold IDAM into an impedance biosensor to rapidly detect pathogenic bacteria in ground beef samples. The flow cell consisting of a detection microchamber and inlet and outlet microchannels was fabricated by binding an IDAM chip to a poly (dimethylsiloxane) (PDMS) microchannel (Figure 5). The detection microchamber with a dimension of 6 mm × 0.5 mm × 0.02 mm and a volume of 60 nL was used to collect bacterial cells in the active layer above the microelectrode for sensitive impedance change. Antibody coated magnetic nanoparticles were used to specifically separate and concentrate the target bacteria and then the biomolecule functional magnetic nanoparticles-bacteria complexes were injected into microfluidic cell to detect the impedance change. Using the microfluidic system, the limit of detection has been improved an order of magnitude as low as 1.6 × 102 and 1.2 × 103 cfu·mL−1 of E. coli O157:H7 cells present in pure culture and ground beef sample, respectively. Tan et al. [82] devised a PDMS microfluidic immunosensor integrated with specific antibody immobilized alumina nanoporous membrane for rapid detection of foodborne pathogens E. coli O157:H7 and Staphylococcus aureus with EIS. When the target bacteria were injected into the chamber to bind antibody, the electrolyte current will be blocked which can be monitored by the impedance spectrum. This microfluidic immunosensor based on nanoporous membrane impedance spectrum could achieve rapid bacteria detection within 2 h with a high sensitivity of 102 cfu mL−1.
The microfluidic biochip system has been used to effectively improve the detection limit and reduce detection time of impedance biosensor for bacteria detection by confining a few live bacterial cells into a small volume on the order of nano-to pico-liters. Gomez and coworkers [96] were the first to fabricate integrated silicon-based microfluidic biochips for impedance detection of microbial metabolism. The impedance microbiology-on-a chip contained two sets of interdigitated microelectrodes. One set was for dielectrophoretically capturing bacterial cells from the flow into the small chamber, and the other set was for monitoring the impedance change when bacterial cells grew in the chamber. The design concept was to use DEP to deviate the bacterial cells from a main channel into a small channel that led the cells into a measurement chamber which had a volume of 400 pL. This on-chip impedance microbiology has achieved a detection time of 1 h for a sample with a starting concentration of 104 cfu·mL−1. A similar microfluidic system has been developed to concentrate bacteria. Yang et al. [36] developed a microfluidic system with multiple functions, including concentration of bacteria using DEP and selective capture using antibody recognition, resulting in a high capture efficiency of bacterial cells. The device consisted of an array of oxide covered interdigitated electrodes on a flat silicon substrate and a ∼16 mm high and ∼260 mm wide micro-channel within a PDMS cover. The impedimetric biosensor that combined DEPIM offered advantages inherited from both DEP and antibody recognition, including increasing antibody capture efficiency and decreasing binding time of bacteria and antibody.
In addition, low cost and disposability is another trend for microfluidics biochip development. Gottschamel et al. [88] developed a disposable microfluidics biochip for multiparameter Candida albicans population measurements, which can monitor Candida albicans growth rates and metabolic activities by simultaneous bioimpedance spectroscopy and amperometric measurements. Zhu et al. [92] used fluidic electrodes to fabricate a microfluidics device for detecting bacterial cells in deionized water suspensions with a detection limit of 103 cfu·mL−1. KCl solution was utilized as both sheath flow and fluidic electrodes, and the bacterial suspension was squeezed to form three flowing layers with different conductivities on a microfluidics chip. An impedance analyzer was connected with the KCl solution through two Ag/AgCl wires to apply an AC voltage to fluidic layers within a certain frequency for impedance measurements. Compared with traditional metal electrode, the use of fluidic electrodes can effective decrease the cost for fabrication of a microfluidics biochip.
6. Conclusions
Impedimetric biosensors have been used to monitor foodborne pathogenic bacteria for many years. Compared to the other methods, it has several main advantages as follows:
– They are label-free, which simplifies the assembly process and lowers the cost.
– They are rapid and the detection time is generally less than 30 min.
– Realization of the impedance device miniaturization, which have been proved to be very successful in maximizing the impedance signal, minimizing the volume of testing sample, increasing sensitivity, and saving assay time.
– They can reach detection limits as low as those of SPR and ELISA. After combination of nanoparticles or with microfluidic techniques, they can achieve a lower detection limit than standard immunoassays.
– They are reproducible when the bio-recognition elements are immobilized on the electrode using strong chemical bonds such as SAM immobilization mehod, which reduces the cost of use.
Although the impedimetric biosensors have many advantages, they still have some limits. After two decades of research efforts and hundreds of publications, no product based on impedance-based biosensors has enjoyed widespread commercial success. Therefore, further efforts should be devoted to developing commercial products in the area of impedimetric biosensors for foodborne pathogenic bacteria detection, which will require improved stability, reduced volume, increased sensitivity and lowered costs.
Acknowledgments
The authors gratefully acknowledge the financial support provided by National Natural Science Foundation of China (No. 30825027) and the Key Scientific Project of China (No. 2009ZX08012-004B).
Reference
- Park, S.S.; Worobo, R.W.; Durst, R.A. Escherichia coli O157: H7 as an emerging foodborne pathogen: A literature review. Crit. Rev. Biotechnol 2001, 21, 27–48. [Google Scholar]
- Gandhi, M.; Chikindas, M.L. Listeria: A foodborne pathogen that knows how to survive. Int. J. Food Microbiol 2007, 113, 1–15. [Google Scholar]
- Murphy, C.; Carroll, C.; Jordan, K.N. Environmental survival mechanisms of the foodborne pathogen Campylobacter jejuni. J. Appl. Microbiol 2006, 100, 623–632. [Google Scholar]
- Velusamy, V.; Arshak, K.; Korostynska, O.; Oliwa, K.; Adley, C. An overview of foodborne pathogen detection: In the perspective of biosensors. Biotechnol. Adv 2010, 28, 232–254. [Google Scholar]
- Wang, Y.X.; Ye, Z.Z.; Si, C.Y.; Ying, Y.B. Subtractive inhibition assay for the detection of E. coli O157:H7 using surface plasmon resonance. Sensors 2011, 11, 2728–2739. [Google Scholar]
- de Boer, E.; Beumer, R.R. Methodology for detection and typing of foodborne microorganisms. Int. J. Food Microbiol 1999, 50, 119–130. [Google Scholar]
- Gracias, K.S.; McKillip, J.L. A review of conventional detection and enumeration methods for pathogenic bacteria in food. Can. J. Microbiol 2004, 50, 883–890. [Google Scholar]
- Hall, R.H. Biosensor technologies for detecting microbiological foodborne hazards. Microbes Infect 2002, 4, 425–432. [Google Scholar]
- Lazcka, O.; Del Campo, F.J.; Munoz, F.X. Pathogen detection: A perspective of traditional methods and biosensors. Biosens. Bioelectron 2007, 22, 1205–1217. [Google Scholar]
- Swaminathan, B.; Feng, P. Rapid detection of food-borne pathogenic bacteria. Annu. Rev. Microbiol 1994, 48, 401–426. [Google Scholar]
- Yang, L.; Bashir, R. Electrical/electrochemical impedance for rapid detection of foodborne pathogenic bacteria. Biotechnol. Adv 2008, 26, 135–150. [Google Scholar]
- Sapsford, K.E.; Bradburne, C.; Detehanty, J.B.; Medintz, I.L. Sensors for detecting biological agents. Mater. Today 2008, 11, 38–49. [Google Scholar]
- Katz, E.; Willner, I. Probing biomolecular interactions at conductive and semiconductive surfaces by impedance spectroscopy: Routes to impedimetric immunosensors, DNA-Sensors, and enzyme biosensors. Electroanalysis 2003, 15, 913–947. [Google Scholar]
- Ivnitski, D.; Abdel-Hamid, I.; Atanasov, P.; Wilkins, E. Biosensors for detection of pathogenic bacteria. Biosens. Bioelectron 1999, 14, 599–624. [Google Scholar]
- Ivnitski, D.; Abdel-Hamid, I.; Atanasov, P.; Wilkins, E.; Stricker, S. Application of electrochemical biosensors for detection of food pathogenic bacteria. Electroanalysis 2000, 12, 317–325. [Google Scholar]
- K’Owino, I.O.; Sadik, O.A. Impedance spectroscopy: A powerful tool for rapid biomolecular screening and cell culture monitoring. Electroanalysis 2005, 17, 2101–2113. [Google Scholar]
- Lisdat, F.; Schafer, D. The use of electrochemical impedance spectroscopy for biosensing. Anal. Bioanal. Chem 2008, 391, 1555–1567. [Google Scholar]
- Prodromidis, M.I. Impedimetric immunosensors—A review. Electrochim. Acta 2010, 55, 4227–4233. [Google Scholar]
- Pejcic, B.; De Marco, R. Impedance spectroscopy: Over 35 years of electrochemical sensor optimization. Electrochim. Acta 2006, 51, 6217–6229. [Google Scholar]
- Varshney, M.; Li, Y.B. Interdigitated array microelectrodes based impedance biosensors for detection of bacterial cells. Biosens. Bioelectron 2009, 24, 2951–2960. [Google Scholar]
- Sadik, O.A.; Aluoch, A.O.; Zhou, A.L. Status of biomolecular recognition using electrochemical techniques. Biosens. Bioelectron 2009, 24, 2749–2765. [Google Scholar]
- Li, D.J.; Feng, Y.Y.; Zhou, L.; Ye, Z.Z.; Wang, J.P.; Ying, Y.B.; Ruan, C.M.; Wang, R.H.; Li, Y.B. Label-free capacitive immunosensor based on quartz crystal Au electrode for rapid and sensitive detection of Escherichia coli O157:H7. Anal. Chim. Acta 2011, 687, 89–96. [Google Scholar]
- Li, D.; Xu, K.; Wang, J.; Ye, Z.; Ying, Y.; Li, Y. Quartz crystal Au electrode-based electrochemical immunosensor for rapid detection of Escherichia coli O157:H7. Trans. ASABE 2008, 51, 1847–1852. [Google Scholar]
- Yang, L.J.; Li, Y.B.; Erf, G.F. Interdigitated array microelectrode-based electrochemical impedance immunosensor for detection of Escherichia coli O157:H7. Anal. Chem 2004, 76, 1107–1113. [Google Scholar]
- dos Santos, M.B.; Sporer, C.; Sanvicens, N.; Pascual, N.; Errachid, A.; Martinez, E.; Marco, M.P.; Teixeira, V.; Samiter, J. Detection of pathogenic Bacteria by Electrochemical Impedance Spectroscopy: Influence of the immobilization strategies on the sensor performance. Procedia Chem 2009, 1, 1291–1294. [Google Scholar]
- Escamilla-Gomez, V.; Campuzano, S.; Pedrero, M.; Pingarron, J.M. Gold screen-printed-based impedimetric immunobiosensors for direct and sensitive Escherichia coli quantisation. Biosens. Bioelectron 2009, 24, 3365–3371. [Google Scholar]
- Geng, P.; Zhang, X.N.; Meng, W.W.; Wang, Q.J.; Zhang, W.; Jin, L.T.; Feng, Z.; Wu, Z.R. Self-assembled monolayers-based immunosensor for detection of Escherichia coli using electrochemical impedance spectroscopy. Electrochim. Acta 2008, 53, 4663–4668. [Google Scholar]
- Wan, Y.; Zhang, D.; Wang, Y.; Hou, B. A 3D-impedimetric immunosensor based on foam Ni for detection of sulfate-reducing bacteria. Electrochem. Commun 2010, 12, 288–291. [Google Scholar]
- Yang, L.J.; Li, Y.B. AFM and impedance spectroscopy characterization of the immobilization of antibodies on indium-tin oxide electrode through self-assembled monolayer of epoxysilane and their capture of Escherichia coli O157:H7. Biosens. Bioelectron 2005, 20, 1407–1416. [Google Scholar]
- Arya, S.K.; Solanki, P.R.; Datta, M.; Malhotra, B.D. Recent advances in self-assembled monolayers based biomolecular electronic devices. Biosens. Bioelectron 2009, 24, 2810–2817. [Google Scholar]
- Laczka, O.; Baldrich, E.; Munoz, F.X.; del Campo, F.J. Detection of Escherichia coli and Salmonella typhimurium using interdigitated microelectrode capacitive immunosensors: The importance of transducer geometry. Anal. Chem 2008, 80, 7239–7247. [Google Scholar]
- Radke, S.A.; Alocilja, E.C. A high density microelectrode array biosensor for detection of E. coli O157:H7. Biosens. Bioelectron 2005, 20, 1662–1667. [Google Scholar]
- Ruan, C.M.; Yang, L.J.; Li, Y.B. Immunobiosensor chips for detection of Escherichia coli O157:H7 using electrochemical impedance spectroscopy. Anal. Chem 2002, 74, 4814–4820. [Google Scholar]
- Roychaudhuri, C.; Das, R.D. A biomolecule compatible electrical model of microimpedance affinity biosensor for sensitivity improvement in cell detection. Sens. Actuat. A 2010, 157, 280–289. [Google Scholar]
- Yang, L. Dielectrophoresis assisted immuno-capture and detection of foodborne pathogenic bacteria in biochips. Talanta 2009, 80, 551–558. [Google Scholar]
- Yang, L.J.; Banada, P.P.; Chatni, M.R.; Lim, K.S.; Bhunia, A.K.; Ladisch, M.; Bashir, R. A multifunctional micro-fluidic system for dielectrophoretic concentration coupled with immuno-capture of low numbers of Listeria monocytogenes. Lab Chip 2006, 6, 896–905. [Google Scholar]
- Suehiro, J.; Hamada, R.; Noutomi, D.; Shutou, M.; Hara, M. Selective detection of viable bacteria using dielectrophoretic impedance measurement method. J. Electrostat 2003, 57, 157–168. [Google Scholar]
- Suehiro, J.; Shutou, M.; Hatano, T.; Hara, M. High sensitive detection of biological cells using dielectrophoretic impedance measurement method combined with electropermeabilization. Sens. Actuat. B 2003, 96, 144–151. [Google Scholar]
- Suehiro, J.; Hatano, T.; Shutou, M.; Hara, M. Improvement of electric pulse shape for electropermeabilization-assisted dielectrophoretic impedance measurement for high sensitive bacteria detection. Sens. Actuat. B 2005, 109, 209–215. [Google Scholar]
- Suehiro, J.; Ohtsubo, A.; Hatano, T.; Hara, M. Selective detection of bacteria by a dielectrophoretic impedance measurement method using an antibody-immobilized electrode chip. Sens. Actuat. B 2006, 119, 319–326. [Google Scholar]
- Farabullini, F.; Lucarelli, F.; Palchetti, I.; Marrazza, G.; Mascini, M. Disposable electrochemical genosensor for the simultaneous analysis of different bacterial food contaminants. Biosens. Bioelectron 2007, 22, 1544–1549. [Google Scholar]
- LaGier, M.J.; Scholin, C.A.; Fell, J.W.; Wang, J.; Goodwin, K.D. An electrochemical RNA hybridization assay for detection of the fecal indicator bacterium Escherichia coli. Mar. Pollut. Bull 2005, 50, 1251–1261. [Google Scholar]
- Liao, J.C.; Mastali, M.; Gau, V.; Suchard, M.A.; Moller, A.K.; Bruckner, D.A.; Babbitt, J.T.; Li, Y.; Gornbein, J.; Landaw, E.M.; McCabe, E.R.B.; Churchill, B.M.; Haake, D.A. Use of electrochemical DNA biosensors for rapid molecular identification of uropathogens in clinical urine specimens. J. Clin. Microbiol 2006, 44, 561–570. [Google Scholar]
- Miranda-Castro, R.; de-los-Santos-Alvarez, N.; Lobo-Castanon, M.J.; Miranda-Ordieres, A.J.; Tunon-Blanco, P. Structured nucleic acid probes for electrochemical devices. Electroanalysis 2009, 21, 2077–2090. [Google Scholar]
- Yeung, S.W.; Lee, T.M.H.; Cai, H.; Hsing, I.M. A DNA biochip for on-the-spot multiplexed pathogen identification. Nucl. Acids Res 2006, 34, e118. [Google Scholar]
- Kara, P.; Meric, B.; Ozsoz, M. Application of impedimetric and voltammetric genosensor for detection of a biological warfare: Anthrax. Electroanalysis 2008, 20, 2629–2634. [Google Scholar]
- Mao, C.B.; Liu, A.H.; Cao, B.R. Virus-based chemical and biological sensing. Angew. Chem. Int. Ed 2009, 48, 6790–6810. [Google Scholar]
- Hagens, S.; Loessner, M.J. Application of bacteriophages for detection and control of foodborne pathogens. Appl. Microbiol. Biotechnol 2007, 76, 513–519. [Google Scholar]
- Iqbal, S.S.; Mayo, M.W.; Bruno, J.G.; Bronk, B.V.; Batt, C.A.; Chambers, J.P. A review of molecular recognition technologies for detection of biological threat agents. Biosens. Bioelectron 2000, 15, 549–578. [Google Scholar]
- Petrenko, V.A.; Vodyanoy, V.J. Phage display for detection of biological threat agents. J. Microbiol. Methods 2003, 53, 253–262. [Google Scholar]
- Dover, J.E.; Hwang, G.M.; Mullen, E.H.; Prorok, B.C.; Suh, S.J. Recent advances in peptide probe-based biosensors for detection of infectious agents. J. Microbiol. Methods 2009, 78, 10–19. [Google Scholar]
- Balasubramanian, S.; Sorokulova, I.B.; Vodyanoy, V.J.; Simonian, A.L. Lytic phage as a specific and selective probe for detection of Staphylococcus aureus—A surface plasmon resonance spectroscopic study. Biosens. Bioelectron 2007, 22, 948–955. [Google Scholar]
- Nanduri, V.; Sorokulova, I.B.; Samoylov, A.M.; Simonian, A.L.; Petrenko, V.A.; Vodyanoy, V. Phage as a molecular recognition element in biosensors immobilized by physical adsorption. Biosens. Bioelectron 2007, 22, 986–992. [Google Scholar]
- Neufeld, T.; Schwartz-Mittelmann, A.; Biran, D.; Ron, E.Z.; Rishpon, J. Combined phage typing and amperometric detection of released enzymatic activity for the specific identification and quantification of bacteria. Anal. Chem 2003, 75, 580–585. [Google Scholar]
- Shabani, A.; Zourob, M.; Allain, B.; Marquette, C.A.; Lawrence, M.F.; Mandeville, R. Bacteriophage-modified microarrays for the direct impedimetric detection of bacteria. Anal. Chem 2008, 80, 9475–9482. [Google Scholar]
- Gervals, L.; Gel, M.; Allain, B.; Tolba, M.; Brovko, L.; Zourob, M.; Mandeville, R.; Griffiths, M.; Evoy, S. Immobilization of biotinylated bacteriophages on biosensor surfaces. Sens. Actuat. B 2007, 125, 615–621. [Google Scholar]
- Webster, M.S.; Timoshkin, I.V.; MacGregor, S.J.; Mattey, M. Computer aided modelling of an interdigitated microelectrode array impedance biosensor for the detection of bacteria. IEEE Trans. Dielectr. Electr. Insul 2009, 16, 1356–1363. [Google Scholar]
- Safina, G.; van Lier, M.; Danielsson, B. Flow-injection assay of the pathogenic bacteria using lectin-based quartz crystal microbalance biosensor. Talanta 2008, 77, 468–472. [Google Scholar]
- Xi, F.; Gao, J.; Wang, J.; Wang, Z. Discrimination and detection of bacteria with a label-free impedimetric biosensor based on self-assembled lectin monolayer. J. Electroanal. Chem 2011, 656, 252–257. [Google Scholar]
- Ngundi, M.M.; Kulagina, N.V.; Anderson, G.P.; Taitt, C.R. Nonantibody-based recognition: Alternative molecules for detection of pathogens. Expert Rev. Proteom 2006, 3, 511–524. [Google Scholar]
- Gamella, M.; Campuzano, S.; Parrado, C.; Reviejo, A.J.; Pingarron, J.M. Microorganisms recognition and quantification by lectin adsorptive affinity impedance. Talanta 2009, 78, 1303–1309. [Google Scholar]
- Mandenius, C.F.; Wang, R.H.; Alden, A.; Bergstrom, G.; Thebault, S.; Lutsch, C.; Ohlson, S. Monitoring of influenza virus hemagglutinin in process samples using weak affinity ligands and surface plasmon resonance. Anal. Chim. Acta 2008, 623, 66–75. [Google Scholar]
- Wan, Y.; Zhang, D.; Hou, B. Monitoring microbial populations of sulfate-reducing bacteria using an impedimetric immunosensor based on agglutination assay. Talanta 2009, 80, 218–223. [Google Scholar]
- Grossi, M.; Lanzoni, M.; Pompei, A.; Lazzarini, R.; Matteuzzi, D.; Ricco, B. An embedded portable biosensor system for bacterial concentration detection. Biosens. Bioelectron 2010, 26, 983–990. [Google Scholar]
- Kim, Y.H.; Park, J.S.; Jung, H.I. An impedimetric biosensor for real-time monitoring of bacterial growth in a microbial fermentor. Sens. Actuat. B 2009, 138, 270–277. [Google Scholar]
- Banada, P.P.; Liu, Y.S.; Yang, L.J.; Bashir, R.; Bhunia, A.K. Performance evaluation of a low conductive growth medium (LCGM) for growth of healthy and stressed Listeria monocytogenes and other common bacterial species. Int. J. Food Microbiol 2006, 111, 12–20. [Google Scholar]
- Choi, A.; Park, J.S.; Jung, H.I. Solid-medium-integrated impedimetric biosensor for real-time monitoring of microorganisms. Sens. Actuat. B 2009, 137, 357–362. [Google Scholar]
- Chai, R.; Yuan, R.; Chai, Y.Q.; Ou, C.F.; Cao, S.R.; Li, X.L. Amperometric immunosensors based on layer-by-layer assembly of gold nanoparticles and methylene blue on thiourea modified glassy carbon electrode for determination of human chorionic gonadotrophin. Talanta 2008, 74, 1330–1336. [Google Scholar]
- Huang, J.L.; Yang, G.J.; Meng, W.J.; Wu, L.P.; Zhu, A.P.; Jiao, X.A. An electrochemical impedimetric immunosensor for label-free detection of Campylobacter jejuni in diarrhea patients’ stool based on O-carboxymethylchitosan surface modified Fe3O4 nanoparticles. Biosens. Bioelectron 2010, 25, 1204–1211. [Google Scholar]
- Jiang, X.S.; Wang, R.H.; Wang, Y.; Su, X.L.; Ying, Y.B.; Wang, J.P.; Li, Y.B. Evaluation of different micro/nanobeads used as amplifiers in QCM immunosensor for more sensitive detection of E. coli O157:H7. Biosens. Bioelectron 2011, 29, 23–28. [Google Scholar]
- Yang, G.J.; Huang, J.L.; Meng, W.J.; Shen, M.; Jiao, X.A. A reusable capacitive immunosensor for detection of Salmonella spp. based on grafted ethylene diamine and self-assembled gold nanoparticle monolayers. Anal. Chim. Acta 2009, 647, 159–166. [Google Scholar]
- Wang, R.; Dong, W.; Ruan, C.; Kanayeva, D.; Lassiter, K.; Tian, R.; Li, Y. TiO2 nanowire bundle microelectrode based impedance immunosensor for rapid and sensitive detection of Listeria monocytogenes. Nano Lett 2009, 9, 4570. [Google Scholar]
- Wan, Y.; Lin, Z.F.; Zhang, D.; Wang, Y.; Hou, B.R. Impedimetric immunosensor doped with reduced graphene sheets fabricated by controllable electrodeposition for the non-labelled detection of bacteria. Biosens. Bioelectron 2011, 26, 1959–1964. [Google Scholar]
- Wang, L.J.; Liu, Q.J.; Hu, Z.Y.; Zhang, Y.F.; Wu, C.S.; Yang, M.; Wang, P. A novel electrochemical biosensor based on dynamic polymerase-extending hybridization for E. coli O157:H7 DNA detection. Talanta 2009, 78, 647–652. [Google Scholar]
- Berti, F.; Laschi, S.; Palchetti, I.; Rossier, J.S.; Reymond, F.; Mascini, M.; Marrazza, G. Microfluidic-based electrochemical genosensor coupled to magnetic beads for hybridization detection. Talanta 2009, 77, 971–978. [Google Scholar]
- Kwon, Y.; Hara, C.A.; Knize, M.G.; Hwang, M.H.; Venkateswaran, K.S.; Wheeler, E.K.; Bell, P.M.; Renzi, R.F.; Fruetel, J.A.; Bailey, C.G. Magnetic bead based immunoassay for autonomous detection of toxins. Anal. Chem 2008, 80, 8416–8423. [Google Scholar]
- Liu, Y.J.; Yao, D.J.; Chang, H.Y.; Liu, C.M.; Chen, C. Magnetic bead-based DNA detection with multi-layers quantum dots labeling for rapid detection of Escherichia coli O157:H7. Biosens. Bioelectron 2008, 24, 558–565. [Google Scholar]
- Soelberg, S.D.; Stevens, R.C.; Limaye, A.P.; Furlong, C.E. Surface plasmon resonance detection using antibody-linked magnetic nanoparticles for analyte capture, purification, concentration, and signal amplification. Anal. Chem 2009, 81, 2357–2363. [Google Scholar]
- Sun, Y.; Bi, N.; Song, D.Q.; Bai, Y.; Wang, L.Y.; Zhang, H.Q. Preparation of Titania sol-gel matrix for the immunoassay by SPR biosensor with magnetic beads. Sens. Actuat. B 2008, 134, 566–572. [Google Scholar]
- Yang, L.J.; Li, Y.B. Detection of viable Salmonella using microelectrode-based capacitance measurement coupled with immunomagnetic separation. J. Microbiol. Methods 2006, 64, 9–16. [Google Scholar]
- Varshney, M.; Li, Y.B. Interdigitated array microelectrode based impedance biosensor coupled with magnetic nanoparticle-antibody conjugates for detection of Escherichia coli O157:H7 in food samples. Biosens. Bioelectron 2007, 22, 2408–2414. [Google Scholar]
- Tan, F.; Leung, P.H.M.; Liu, Z.B.; Zhang, Y.; Xiao, L.D.; Ye, W.W.; Zhang, X.; Yi, L.; Yang, M. A PDMS microfluidic impedance immunosensor for E. coli O157:H7 and Staphylococcus aureus detection via antibody-immobilized nanoporous membrane. Sens. Actuat. B 2011, 159, 328–335. [Google Scholar]
- Syed, L.U.; Liu, J.W.; Price, A.K.; Li, Y.F.; Culbertson, C.T.; Li, J. Dielectrophoretic capture of E. coli cells at micropatterned nanoelectrode arrays. Electrophoresis 2011, 32, 2358–2365. [Google Scholar]
- Varshney, M.; Li, Y.B.; Srinivasan, B.; Tung, S. A label-free, microfluidics and interdigitated array microelectrode-based impedance biosensor in combination with nanoparticles immunoseparation for detection of Escherichia coli O157:H7 in food samples. Sens. Actuat. B 2007, 128, 99–107. [Google Scholar]
- Maalouf, R.; Hassen, W.M.; Fournier-Wirth, C.; Coste, J.; Jaffrezic-Renault, N. Comparison of two innovatives approaches for bacterial detection: Paramagnetic nanoparticles and self-assembled multilayer processes. Microchim. Acta 2008, 163, 157–161. [Google Scholar]
- Mernier, G.; Hasenkamp, W.; Piacentini, N.; Renaud, P. Multiple-frequency impedance measurements in continuous flow for the evaluation of electrical lysis of yeast cells. Sens. Actuat. B 2010, 5, 37–40. [Google Scholar]
- Boehm, D.A.; Gottlieb, P.A.; Hua, S.Z. On-chip microfluidic biosensor for bacterial detection and identification. Sens. Actuat. B 2007, 126, 508–514. [Google Scholar]
- Gottschamel, J.; Richter, L.; Mak, A.; Jungreuthmayer, C.; Birnbaumer, G.; Milnera, M.; Bruckl, H.; Ertl, P. Development of a disposable microfluidic biochip for multiparameter cell population measurements. Anal. Chem 2009, 81, 8503–8512. [Google Scholar]
- Richter, L.; Stepper, C.; Mak, A.; Reinthaler, A.; Heer, R.; Kast, M.; Bruckl, H.; Ertl, P. Development of a microfluidic biochip for online monitoring of fungal biofilm dynamics. Lab Chip 2007, 7, 1723–1731. [Google Scholar]
- Yu, J.J.; Liu, Z.B.; Liu, Q.J.; Yuen, K.T.; Mak, A.F.T.; Yang, M.; Leung, P. A polyethylene glycol (PEG) microfluidic chip with nanostructures for bacteria rapid patterning and detection. Sens. Actuat. A 2009, 154, 288–294. [Google Scholar]
- Zaytseva, N.V.; Goral, V.N.; Montagna, R.A.; Baeumner, A.J. Development of a microfluidic biosensor module for pathogen detection. Lab Chip 2005, 5, 805–811. [Google Scholar]
- Zhu, T.; Pei, Z.H.; Huang, J.Y.; Xiong, C.Y.; Shi, S.G.; Fang, J. Detection of bacterial cells by impedance spectra via fluidic electrodes in a microfluidic device. Lab Chip 2010, 10, 1557–1560. [Google Scholar]
- Bayoudh, S.; Othmane, A.; Ponsonnet, L.; Ben Ouada, H. Electrical detection and characterization of bacterial adhesion using electrochemical impedance spectroscopy-based flow chamber. Colloids Surf. A 2008, 318, 291–300. [Google Scholar]
- Liu, Y.S.; Walter, T.M.; Chang, W.J.; Lim, K.S.; Yang, L.J.; Lee, S.W.; Aronson, A.; Bashir, R. Electrical detection of germination of viable model Bacillus anthracis spores in microfluidic biochips. Lab Chip 2007, 7, 603–610. [Google Scholar]
- Varshney, M.; Li, Y.; Srinivasan, B.; Tung, S.; Erf, G.; Slavik, M.F.; Ying, Y.; Fang, W. A microfluidic filter biochip-based chemiluminescence biosensing method for detection of Escherichia coli O157:H7. Trans. ASABE 2006, 49, 2061–2068. [Google Scholar]
- Gomez-Sjoberg, R.; Morisette, D.T.; Bashir, R. Impedance microbiology-on-a-chip: Microfluidic bioprocessor for rapid detection of bacterial metabolism. J. Microelectromech. Syst 2005, 14, 829–838. [Google Scholar]

| Nanomaterials | Microorganism | Electrode | Detection range (cfu·mL−1) | Reference |
|---|---|---|---|---|
| Au NPs | Sulfate-reducing bacteria | foam Ni electrode | 2.1 × 101–2.1 × 107 | [28] |
| Fe3O4 NPs | Campylobacter jejuni | GCE | 1.0 × 103–1.0 × 107 | [69] |
| Au NPs | Salmonella Spp. | GCE | 1.0 × 102–1.0 × 105 | [71] |
| TiO2 nanowire bundle | Listeria monocytogenes | Au microelectrodes | 102–107 | [72] |
| reduced graphene sheets | Sulfate-reducing bacteria | GCE | 1.8 × 101–1.8 × 107 | [73] |
| aluminum anodized oxide (AAO) nanopore membranes | E. coli O157:H7 | Au electrode | – | [74] |
| alumina nanoporous membrane | E. coli O157:H7 | Platinum electrode | 102–107 | [82] |
| carbon nanofiber (CNF) nanoelectrode array (NEA) | E. coli | ITO | – | [83] |
| magnetic nanoparticles | E. coli O157:H7 | IDAM | pure culture 7.4 × 104–7.4 × 107 beef sample 8.0 × 105–8.0 × 107 | [81] |
| magnetic nanoparticles | E. coli O157:H7 | IDAM with microfluidic flow cell | pure culture 1.6 × 102–1.6 × 107 beef sample 1.2 × 103–1.2 × 107 | [84] |
| magnetic nanoparticles | E. coli | Pt plate electrode | 10–104 | [85] |
ITO: indium-tin oxide; GCE: glassy carbon electrode; Au NPs: gold nanoparticles.
© 2012 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, Y.; Ye, Z.; Ying, Y. New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria. Sensors 2012, 12, 3449-3471. https://doi.org/10.3390/s120303449
Wang Y, Ye Z, Ying Y. New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria. Sensors. 2012; 12(3):3449-3471. https://doi.org/10.3390/s120303449
Chicago/Turabian StyleWang, Yixian, Zunzhong Ye, and Yibin Ying. 2012. "New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria" Sensors 12, no. 3: 3449-3471. https://doi.org/10.3390/s120303449
APA StyleWang, Y., Ye, Z., & Ying, Y. (2012). New Trends in Impedimetric Biosensors for the Detection of Foodborne Pathogenic Bacteria. Sensors, 12(3), 3449-3471. https://doi.org/10.3390/s120303449




