Direct Electrochemistry and Electrocatalysis of Hemoglobin at Mesoporous Carbon Modified Electrode
Abstract
:1. Introduction
2. Experimental
2.1. Reagents
2.2. Syntheses of mesoporous carbon FDU-15
2.3. Electrode modification
2.4. Apparatus and measurements
3. Results and Discussion
3.1. Characterization of FDU-15
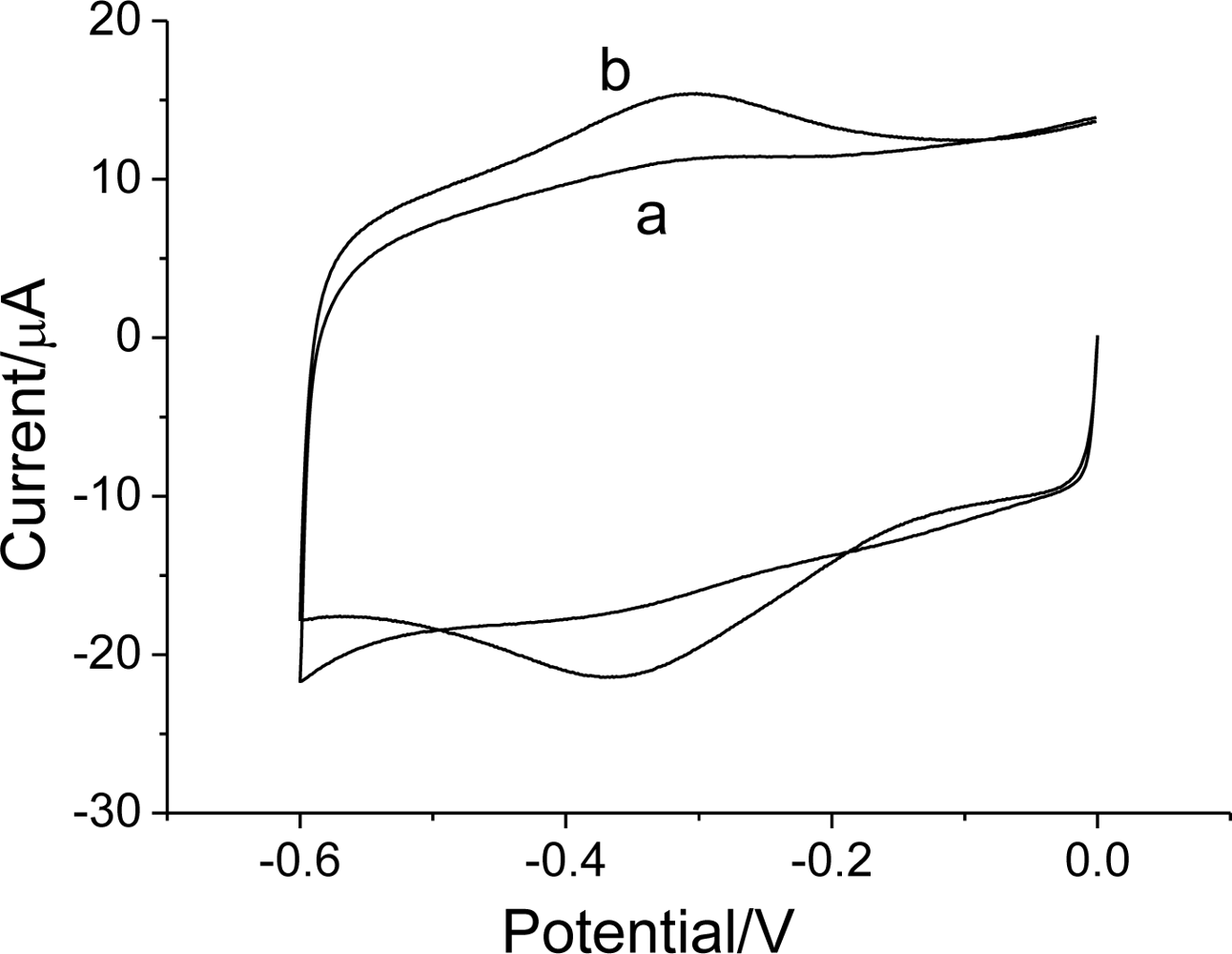
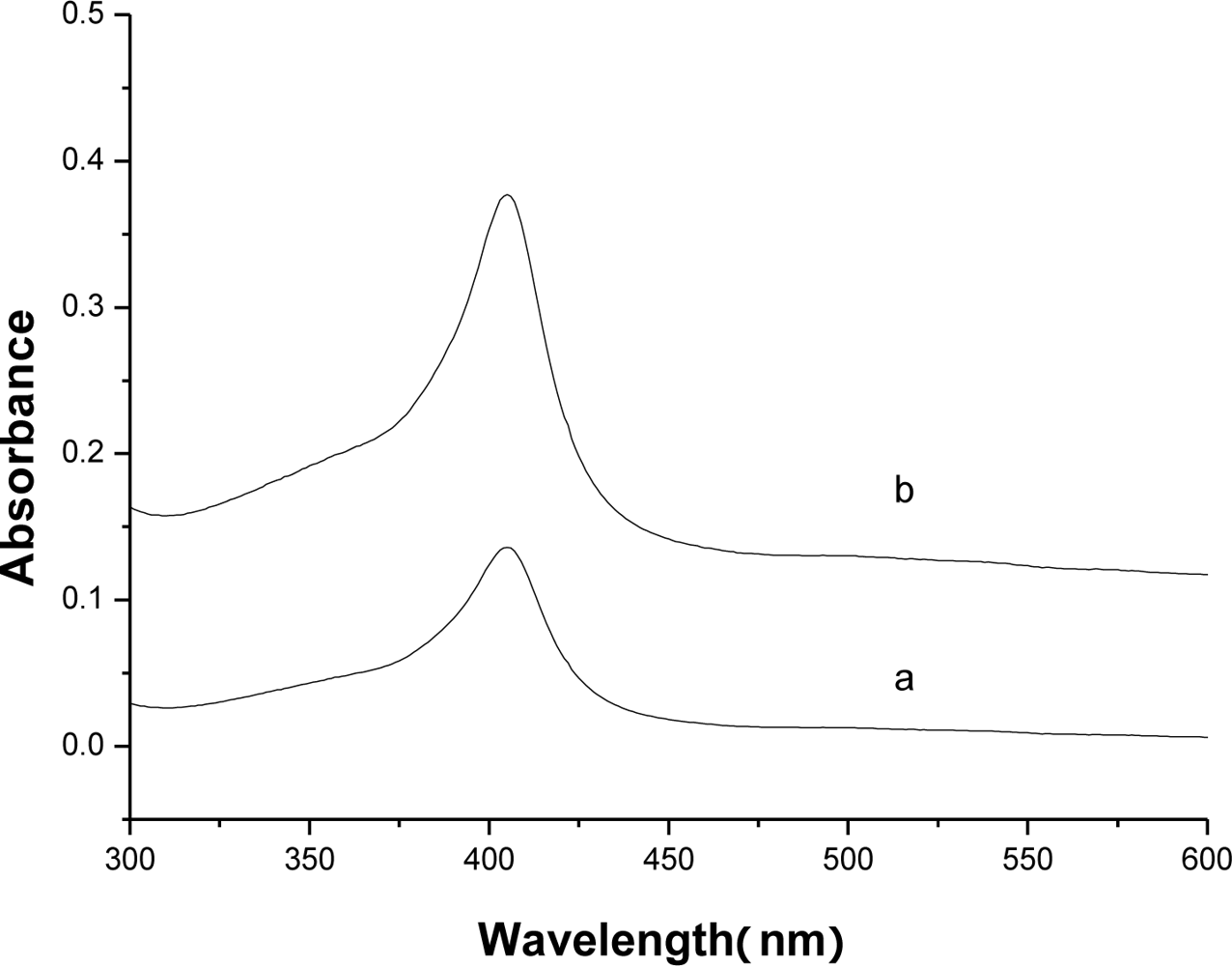
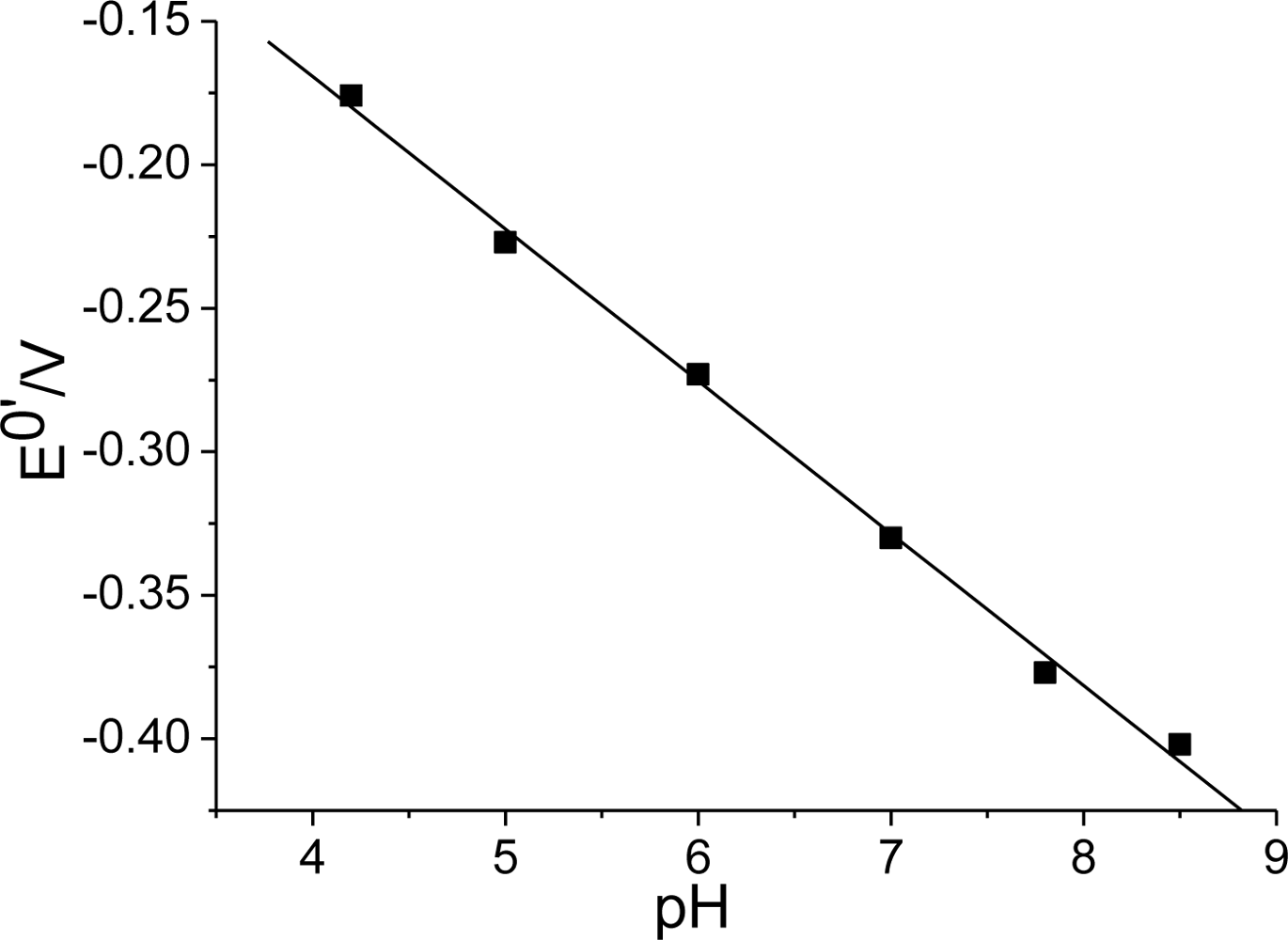
3.2. Direct electrochemistry of Hb on Hb/FDU-15 electrode
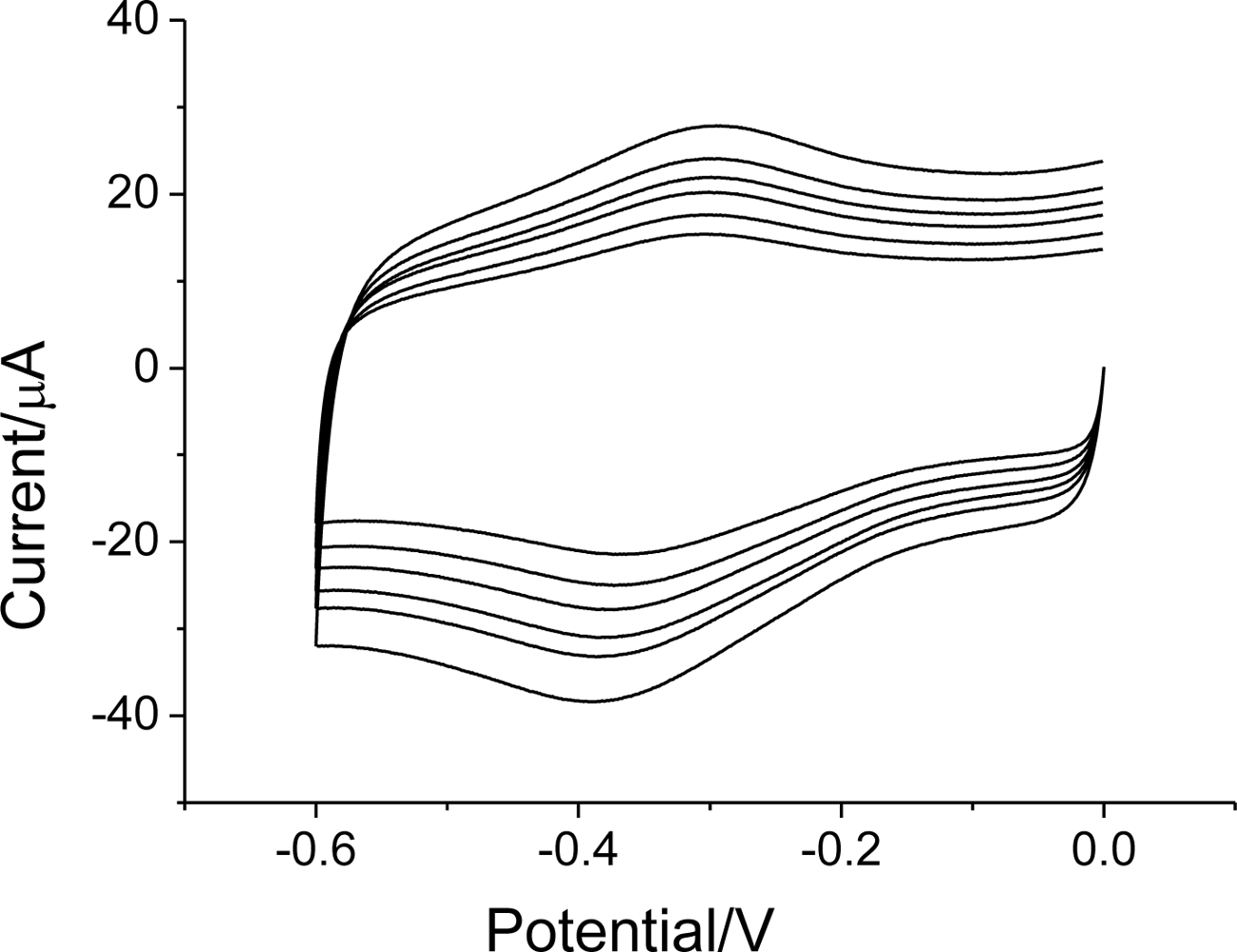
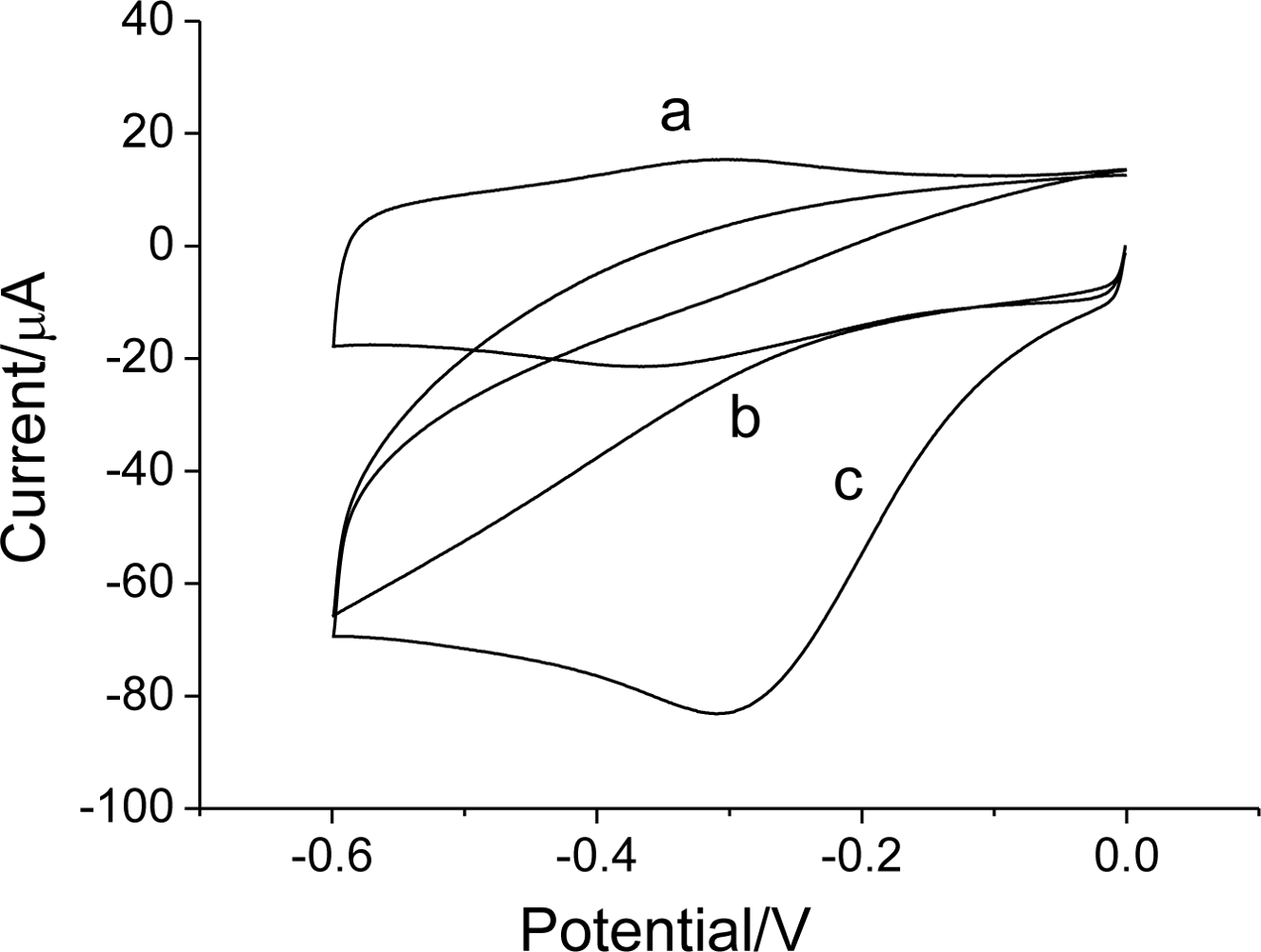
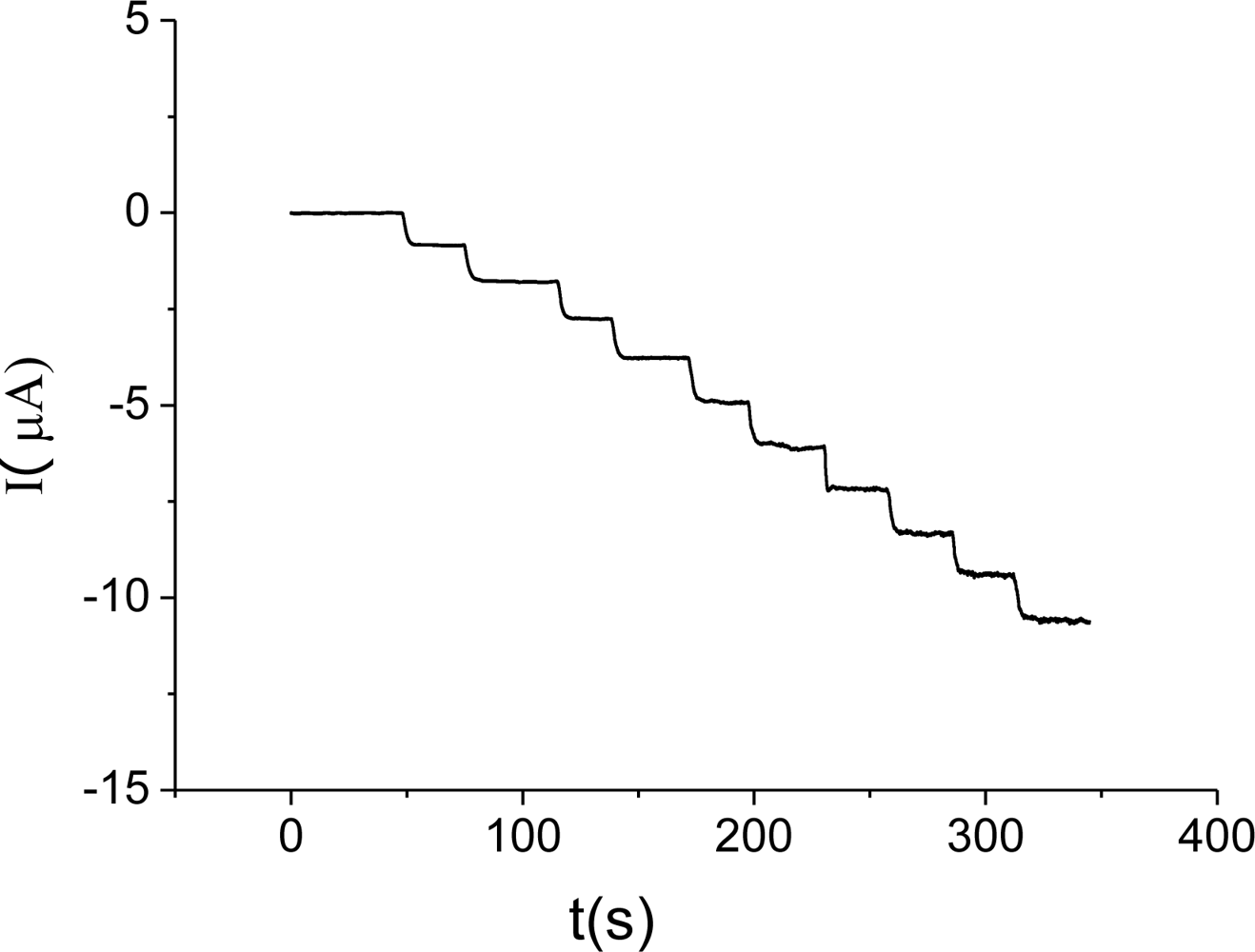
3.3. Electrocatalysis of H2O2 on Hb/FDU-15 electrode
4. Conclusions
Acknowledgments
References
- Kong, J.; Lu, Z.; Lvov, Y.M.; Desamero, R.Z.A.; Frank, H.A.; Rusling, J.F. Direct electrochemistry of cofactor redox sites in a bacterial photosynthetic reaction center protein. J. Am. Chem. Soc 1998, 120, 7371–7372. [Google Scholar]
- Willner, I.; Katz, E. Integration of layered redox proteins and conductive supports for bioelectronic applications. Angew. Chem. Int. Ed 2000, 39, 1180–1218. [Google Scholar]
- Sun, H.; Hu, N.; Ma, H. Direct electrochemistry of hemoglobin in polyacrylamide hydrogel films on pyrolytic graphite electrodes. Electroanalysis 2000, 12, 1064–1070. [Google Scholar]
- Li, Q.; Luo, G.; Feng, J. Direct electron transfer for heme proteins assembled on nanocrystalline TiO2 film. Electroanalysis 2001, 13, 359–363. [Google Scholar]
- Han, X.; Huang, W.; Jia, J.; Dong, S.; Wang, E. Direct electrochemistry of hemoglobin in egg-phosphatidylcholine films and its catalysis to H2O2. Biosens. Bioelectron 2002, 17, 741–746. [Google Scholar]
- Dai, Z.; Liu, S.Q.; Ju, H.X.; Chen, H.Y. Direct electron transfer and enzymatic activity of hemoglobin in a hexagonal mesoporous silica matrix. Biosens. Bioelectron 2004, 19, 861–867. [Google Scholar]
- Liu, J.Q.; Chou, A.; Rahmat, W.; Paddon-Row, M.N.; Gooding, J.J. Achieving direct electrical connection to glucose oxidase using aligned single walled carbon nanotube arrays. Electroanalysis 2005, 17, 38–46. [Google Scholar]
- Zhao, Y.D.; Bi, Y.H.; Zhang, W.D.; Luo, Q.M. The interface behavior of hemoglobin at carbon nanotube and the detection for H2O2. Talanta 2005, 65, 489–494. [Google Scholar]
- Krajewska, A.; Radecki, J.; Radecka, H. A voltammetric biosensor based on glassy carbon electrodes modified with single-walled carbon nanotubes/hemoglobin for detection of acrylamide in water extracts from potato crisps. Sensors 2008, 8, 5832–5844. [Google Scholar]
- Li, H.H.; Liu, S.Q.; Dai, Z.H.; Bao, J.C.; Yang, X.D. Applications of nanomaterials in electrochemical enzyme biosensors. Sensors 2009, 9, 8547–8561. [Google Scholar]
- Zhang, L.; Zhang, Q.; Li, J.H. Direct electrochemistry and electrocatalysis of hemoglobin immobilized in bimodal mesoporous silica and chitosan inorganic-organic hybrid film. Electrochem. Commun 2007, 9, 1530–1535. [Google Scholar]
- Chen, C.C.; Do, J.S.; Gu, Y.S. Immobilization of HRP in mesoporous silica and its application for the construction of polyaniline modified hydrogen peroxide biosensor. Sensors 2009, 9, 4635–4648. [Google Scholar]
- Lei, C.H.; Valenta, M.M.; Sparipalli, K.P.; Ackerman, E.J. Biosensing paraoxon in simulated environmental samples by immobilized organophosphorus hydrolase in functionalized mesoporous silica. J. Environ. Qual 2007, 36, 233–238. [Google Scholar]
- Subramoney, S. Novel nanocarbons-structure, properties, and potential applications. Adv. Mater 1998, 10, 1157–1171. [Google Scholar]
- Shiflett, M.B.; Foley, H.C. Ultrasonic Deposition of high-selectivity nanoporous carbon membranes. Science 1999, 285, 1902–1905. [Google Scholar]
- Flandrois, S.; Simon, B. Carbon materials for lithium ion rechargeable batteries. Carbon 1999, 37, 165–180. [Google Scholar]
- Chai, G.S.; Shin, I.S.; Yu, J.S. Synthesis of ordered, uniform, macroporous carbons with mesoporous walls templated by aggregates of polystyrene spheres and silica particles for use as catalyst supports in direct methanol fuel cells. Adv. Mater 2004, 16, 2057–2061. [Google Scholar]
- Zhou, M.; Shang, L.; Li, B.L.; Huang, L.J.; Dong, S.J. Highly ordered mesoporous carbons as electrode material for the construction of electmchemical dehydrogenase- and oxidase-based biosensors. Biosens. Bioelectron 2008, 24, 442–447. [Google Scholar]
- Zhu, L.D.; Tian, C.Y.; Zhu, D.X.; Yang, R.L. Ordered mesopomus carbon paste electrodes for electrochemical sensing and biosensing. Electroanalysis 2008, 20, 1128–1134. [Google Scholar]
- Lu, X.B.; Xiao, Y.; Lei, Z.B.; Chen, J.P.; Zhang, H.J.; Ni, Y.W.; Zhang, Q. A promising electrochemical biosensing platform based on graphitized ordered mesoporous carbon. J. Mater. Chem 2009, 19, 4707–4714. [Google Scholar]
- Ryoo, R.; Joo, S.H.; Jun, S. Synthesis of highly ordered carbonmolecular sieves via template-mediated structuraltransformation. J. Phys. Chem. B 1999, 103, 7743–7746. [Google Scholar]
- Meng, Y.; Gu, D.; Zhang, F.Q.; Shi, Y.F.; Yang, H.F.; Li, Z.; Yu, C.Z.; Tu, B.; Zhao, D.Y. Ordered mesoporous polymers and homologous carbon frameworks: amphiphilic surfactant templating and direct transformation. Angew. Chem. Int. Edit 2005, 44, 7053–7059. [Google Scholar]
- Zhang, F.Q.; Meng, Y.; Gu, D.; Yan, Y.; Yu, C.Z.; Tu, B.; Zhao, D.Y. A facile aqueous route to synthesize highly ordered mesoporous polymer and carbon frameworks with Ia-3d bicontinuous cubic structure. J. Am. Chem. Soc 2005, 127, 13508–13509. [Google Scholar]
- Tanaka, S.; Nishiyama, N.; Egashira, Y.; Ueyama, K. Synthesis of ordered mesoporous carbons with channel structure from an organic-organic nanocomposite. Chem. Commun 2005, 16, 2125–2127. [Google Scholar]
- Zhao, D.Y.; Feng, J.L.; Huo, Q.S.; Melosh, N.; Fredirckson, G.H.; Chemlka, B.F.; Stucky, G.D. Triblock Copolymer Syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 1998, 279, 548–552. [Google Scholar]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscow, L.; Pierotti, R.A.; Rouquerol, T.; Siemienewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar]
- Wang, J.X.; Li, M.X.; Shi, Z.J.; Li, N.Q.; Gu, Z.N. Direct electrochemistry of cytochrome c at a glassy carbon electrode modified with single-wall carbon nanotubes. Anal. Chem 2002, 74, 1993–1997. [Google Scholar]
- Liu, S.; Chen, A. Coadsorption of horseradish peroxidase with thionine on TiO2 nanotubes for biosensing. Langmuir 2005, 21, 8409–8413. [Google Scholar]
- Zhao, G.; Feng, J.; Xu, J.; Chen, H.Y. Direct electrochemistry and electrocatalysis of heme proteins immobilized on self-assembled ZrO2 film. Electrochem. Commun 2005, 7, 724–729. [Google Scholar]
- Xian, Y.Z.; Xian, Y.; Zhou, L.H.; Wu, F.H.; Ling, Y.; Jin, L.T. Encapsulation hemoglobin in ordered mesoporous silicas: Influence factors for immobilization and bioelectrochemistry. Electrochem. Commun 2007, 9, 142–148. [Google Scholar]
- Laviron, E. The use of linear potential sweep voltammetry and of a.c. voltammetry for the study of the surface electrochemical reaction of strongly adsorbed systems and of redox modified electrodes. J. Electroanal. Chem 1979, 100, 263–270. [Google Scholar]
- Laviron, E. General expression of the linear potential sweep voltammogram for a surface redox reaction with interactions between the adsorbed molecules: Applications to modified electrodes. J. Electroanal. Chem 1980, 115, 65–74. [Google Scholar]
- Zhao, Y.D.; Bi, Y.H.; Zhang, W.D.; Luo, Q.M. The interface behavior of hemoglobin at carbon nanotube and the detection for H2O2. Talanta 2005, 65, 489–494. [Google Scholar]
- Zhao, G.C.; Yin, Z.Z.; Zhang, L.; Wei, X.W. Direct electrochemistry of cytochrome c on a multi-walled carbon nanotubes modified electrode and its electrocatalytic activity for the reduction of H2O2. Electrochem. Commun 2005, 7, 256–260. [Google Scholar]
- Kamin, R.A.; Wilson, G.S. Rotating ring-disk enzyme electrode for biocatalysis kinetic studies and characterization of the immobilized enzyme layer. Anal. Chem 1980, 52, 1198–1205. [Google Scholar]
- Cai, X.; Chen, J. Direct electron transfer and bioelectrocatalysis of hemoglobin at a carbon nanotube electrode. Anal. Biochem 2004, 325, 285–292. [Google Scholar]



© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pei, S.; Qu, S.; Zhang, Y. Direct Electrochemistry and Electrocatalysis of Hemoglobin at Mesoporous Carbon Modified Electrode. Sensors 2010, 10, 1279-1290. https://doi.org/10.3390/s100201279
Pei S, Qu S, Zhang Y. Direct Electrochemistry and Electrocatalysis of Hemoglobin at Mesoporous Carbon Modified Electrode. Sensors. 2010; 10(2):1279-1290. https://doi.org/10.3390/s100201279
Chicago/Turabian StylePei, Supeng, Song Qu, and Yongming Zhang. 2010. "Direct Electrochemistry and Electrocatalysis of Hemoglobin at Mesoporous Carbon Modified Electrode" Sensors 10, no. 2: 1279-1290. https://doi.org/10.3390/s100201279
APA StylePei, S., Qu, S., & Zhang, Y. (2010). Direct Electrochemistry and Electrocatalysis of Hemoglobin at Mesoporous Carbon Modified Electrode. Sensors, 10(2), 1279-1290. https://doi.org/10.3390/s100201279



