Predicting Future European Breeding Distributions of British Seabird Species under Climate Change and Unlimited/No Dispersal Scenarios
Abstract
:1. Introduction
2. Experimental Section

| Species | Score | Prevalence * | AUC | European Southern Range Boundary | A1b | A2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R (%) | O (%) | Distance (km) | Direction | Forecast | R (%) | O (%) | Distance (km) | Direction | Forecast | |||||
| European Storm Petrel Hydrobates pelagicus | 10 | 84 | 0.82 | no | 101 | 15 | 1056 | NE | winner | 124 | 17 | 1200 | NE | winner |
| Leach’s Storm Petrel Oceanodroma leucorhoa | 10 | 12 | 0.83 | yes | 84 | 4 | 833 | N | loser | 96 | 0 | 1238 | N | loser |
| Northern Fulmar Fulmarus glacialis | 7 | 198 | 0.97 | yes | 141 | 69 | 826 | NE | winner | 107 | 56 | 826 | NE | winner |
| Manx shearwater Puffinus puffinus | 7 | 38 | 0.90 | no | 168 | 29 | 1055 | NE | winner | 169 | 28 | 1060 | NE | winner |
| Northern Gannet Morus bassanus | 5 | 33 | 0.82 | yes | 94 | 41 | 341 | N | loser | 84 | 29 | 331 | NW | loser |
| European Shag Phalacrocorax aristotelis | 8 | 280 | 0.88 | no | 134 | 75 | 312 | SE | winner | 136 | 75 | 321 | SE | winner |
| Great Cormorant Phalacrocorax carbo | 7 | 196 | 0.88 | no | 128 | 47 | 579 | NNW | winner | 128 | 43 | 578 | NNW | winner |
| Black-legged Kittiwake Rissa tridactyla | 16 | 225 | 0.94 | yes | 80 | 70 | 244 | NE | loser | 75 | 65 | 303 | N | loser |
| Mediterranean gull Ichthyaetus melanocephalus | 11 | 42 | 0.88 | yes | 195 | 18 | 1335 | NW | winner | 200 | 16 | 1366 | NW | winner |
| Great Black-backed Gul Larus marinus | 10 | 354 | 0.98 | yes | 67 | 60 | 294 | NW | loser | 63 | 58 | 330 | NW | loser |
| European Herring Gull Larus argentatus | 11 | 419 | 0.97 | yes | 72 | 69 | 304 | N | loser | 70 | 66 | 345 | N | loser |
| Lesser Black-backed Gull Larus fuscus | 11 | 350 | 0.93 | yes | 78 | 71 | 327 | N | loser | 77 | 70 | 345 | N | loser |
| Sandwich Tern Thalasseus sandvicensis | 19 | 124 | 0.83 | yes | 100 | 42 | 791 | N | loser | 113 | 47 | 774 | N | winner |
| Little Tern Sternula albifrons | 21 | 245 | 0.81 | no | 100 | 61 | 508 | N | loser | 97 | 57 | 554 | NNW | loser |
| Roseate Tern Sterna dougallii | 22 | 36 | 0.90 | no | 66 | 11 | 965 | NE | loser | 66 | 10 | 1265 | NE | loser |
| Common Tern Sterna hirundo | 20 | 455 | 0.89 | no | 79 | 71 | 391 | NNW | loser | 75 | 68 | 445 | NNW | loser |
| Arctic Tern Sterna paradisaea | 22 | 380 | 0.96 | yes | 52 | 50 | 353 | N | loser | 48 | 47 | 408 | N | loser |
| Great Skua Stercorarius skua | 13 | 65 | 0.95 | yes | 85 | 22 | 618 | N | loser | 68 | 15 | 693 | N | loser |
| Parasitic Jaeger Stercorarius parasiticus | 15 | 230 | 0.97 | yes | 24 | 24 | 869 | WNW | loser | 20 | 19 | 914 | WNW | loser |
| Common Murre Uria aalge | 9 | 145 | 0.87 | yes | 111 | 62 | 667 | NE | winner | 103 | 54 | 718 | NE | winner |
| Razorbill Alca torda | 12 | 182 | 0.90 | yes | 88 | 48 | 485 | N | loser | 83 | 44 | 516 | N | loser |
| Black Guillemot Cepphus grylle | 14 | 287 | 0.97 | yes | 64 | 62 | 251 | NW | loser | 59 | 57 | 294 | NW | loser |
| Atlantic Puffin Fratercula arctica | 13 | 142 | 0.93 | yes | 72 | 43 | 365 | NE | loser | 61 | 35 | 435 | N | loser |
| mean | 95 | 46 | 599 | 92 | 42 | 663 | ||||||||
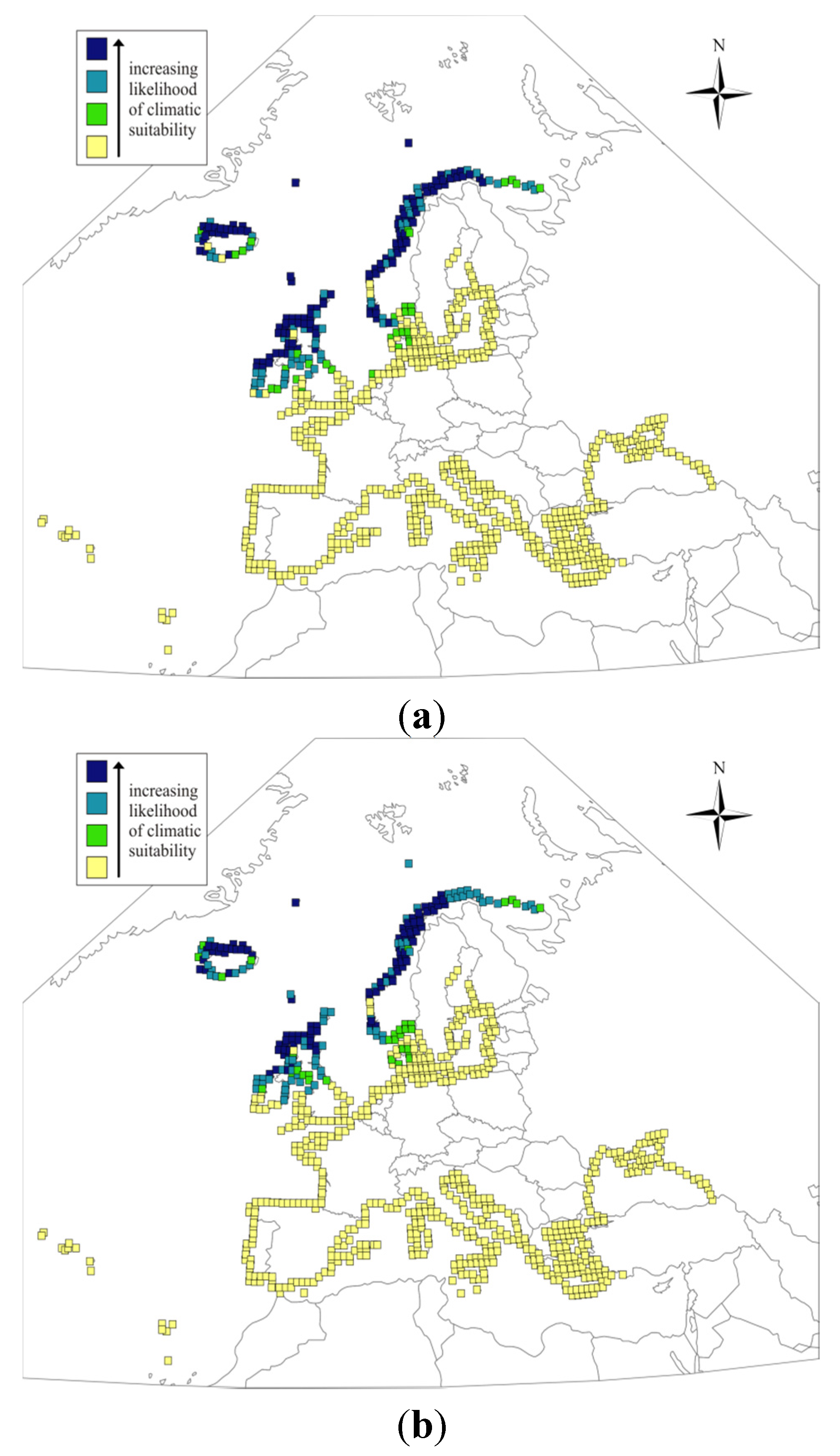
3. Results and Discussion
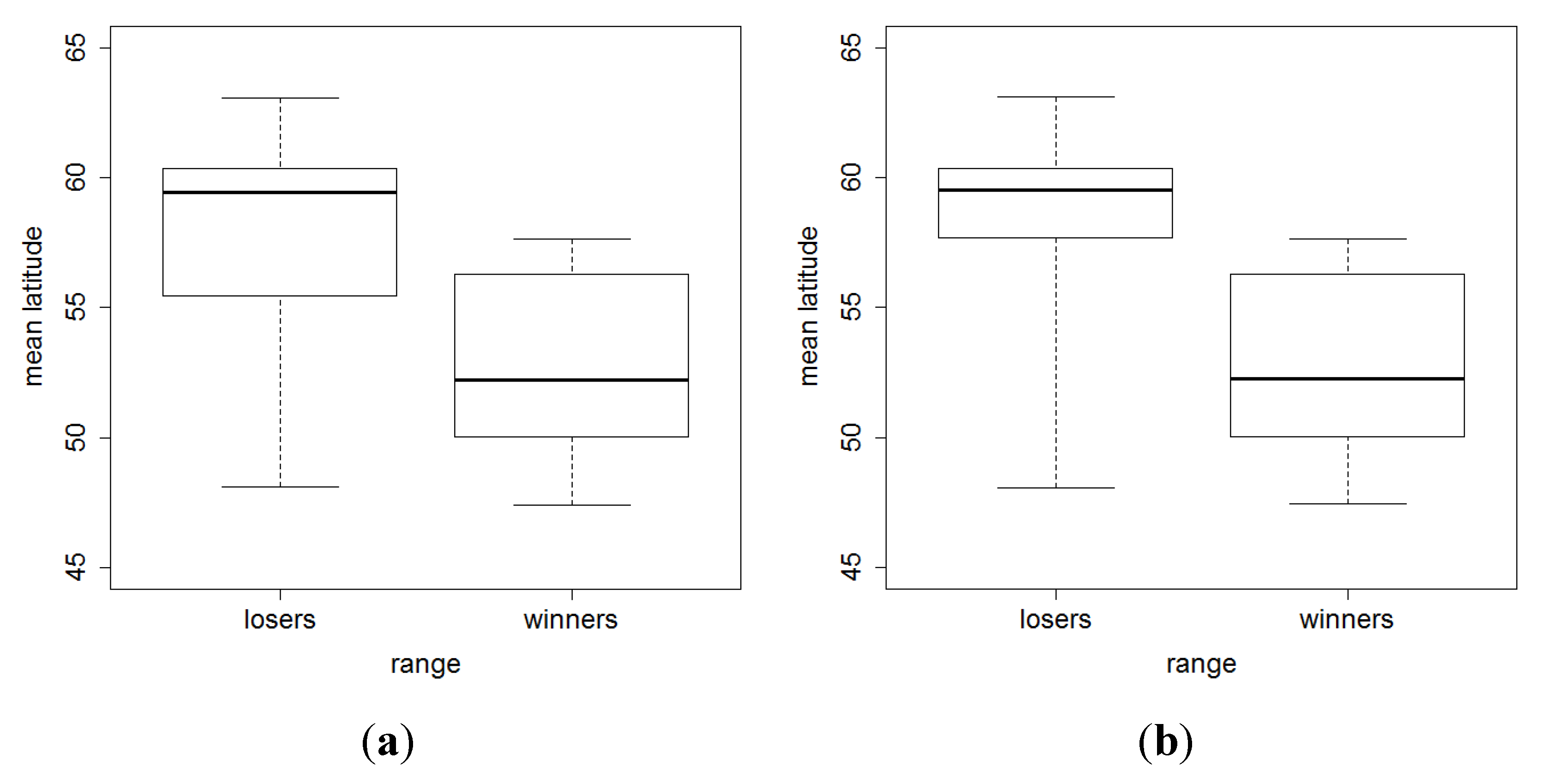
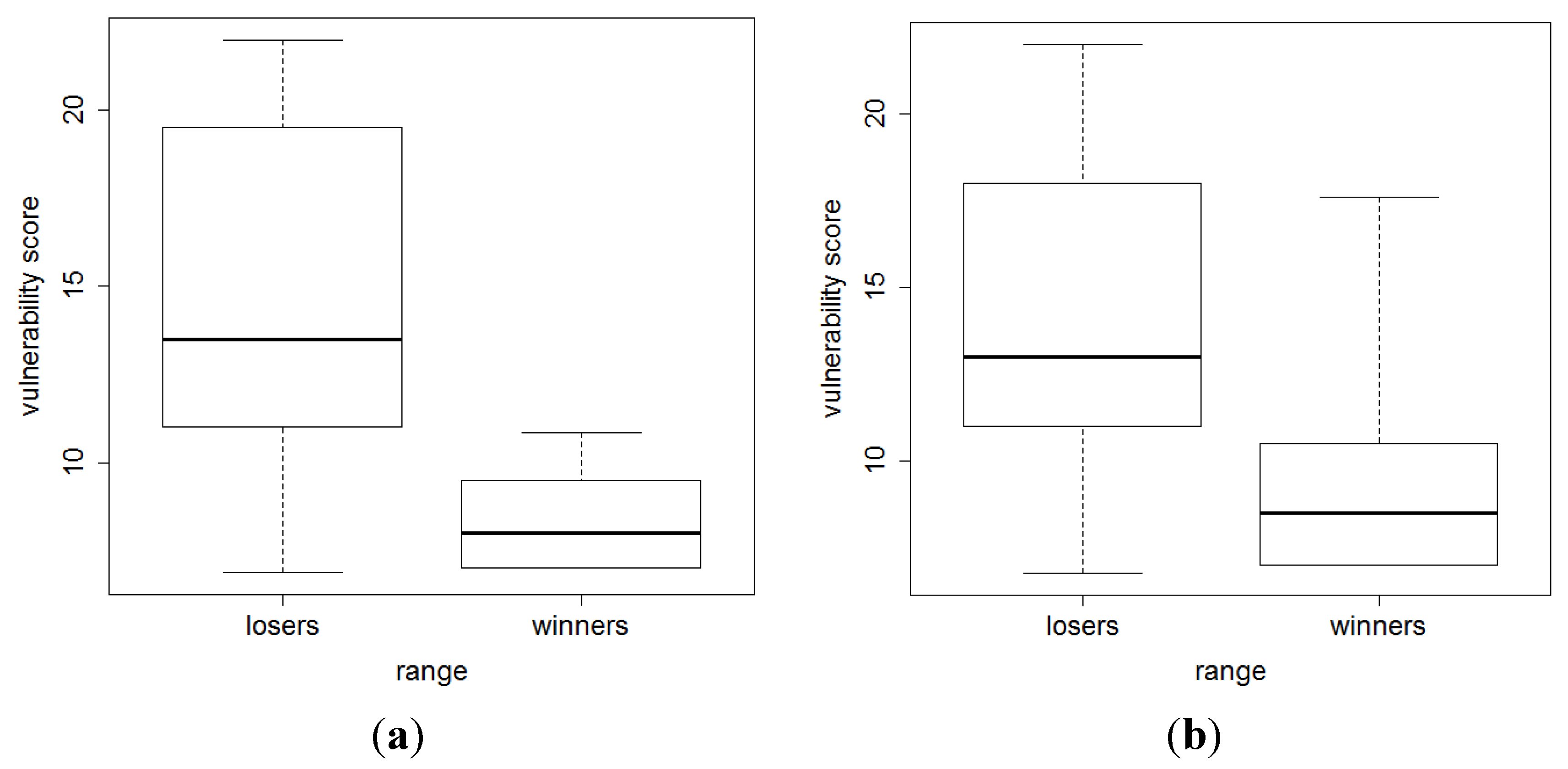
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hughes, L. Biological consequences of global warming: Is the signal already. Trends Ecol. Evol. 2000, 15, 56–61. [Google Scholar] [CrossRef]
- McCarty, J.P. Ecological consequences of recent climate change. Conserv. Biol. 2001, 15, 320–331. [Google Scholar] [CrossRef]
- Walther, G.-R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.C.; Fromentin, J.-M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.-C.; Shiu, H.-J.; Benedick, S.; Holloway, J.D.; Chey, V.K.; Barlow, H.S.; Hill, J.K.; Thomas, C.D. Elevation increases in moth assemblages over 42 years on a tropical mountain. Proc. Natl. Acad. Sci. USA 2009, 106, 1479–1483. [Google Scholar] [CrossRef] [PubMed]
- Lehikoinen, A.; Jaatinen, K.; Vähätalo, A.V.; Clausen, P.; Crowe, O.; Deceuninck, B.; Hearn, R.; Holt, C.A.; Hornman, M.; Keller, V.; et al. Rapid climate driven shifts in wintering distributions of three common waterbird species. Global Chang. Biol. 2013, 19, 2071–2081. [Google Scholar] [CrossRef] [PubMed]
- Berry, P.M.; Dawson, T.P.; Harrison, P.A.; Pearson, R.G. Modelling potential impacts of climate change on the bioclimatic envelope of species in Britain and Ireland. Global Ecol. Biogeogr. 2002, 11, 453–462. [Google Scholar] [CrossRef]
- Araújo, M.B.; Cabeza, M.; Thuiller, W.; Hannah, L.; Williams, P.H. Would climate change drive species out of reserves? An assessment of existing reserve-selection methods. Global Chang. Biol. 2004, 10, 1618–1626. [Google Scholar] [CrossRef]
- Barry, S.; Elith, J. Error and uncertainty in habitat models. J. Appl. Ecol. 2006, 43, 413–423. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Graham, C.H. The ability of climate envelope models to predict the effect of climate change on species distributions. Global Chang. Biol. 2006, 12, 2272–2281. [Google Scholar] [CrossRef]
- Brooker, R.W.; Travis, J.M.J.; Clark, E.J.; Dytham, C. Modelling species’ range shifts in a changing climate: The impacts of biotic interactions, dispersal distance and the rate of climate change. J. Theor. Biol. 2007, 245, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Willis, K.J.; Araújo, M.B.; Bennett, K.D.; Figueroa-Rangel, B.; Froyd, C.A.; Myers, N. How can a knowledge of the past help to conserve the future? Biodiversity conservation and the relevance of long-term ecological studies. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2007, 362, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Huntley, B.; Berry, P.M.; Cramert, W.; Environmental, A.P.M. Modelling present and potential future ranges of some European higher plants using climate response surfaces. J. Biogeogr. 1995, 22, 967–1001. [Google Scholar] [CrossRef]
- Araújo, M.B.; Thuiller, W.; Pearson, R.G. Climate warming and the decline of amphibians and reptiles in Europe. J. Biogeogr. 2006, 33, 1712–1728. [Google Scholar] [CrossRef]
- Levinsky, I.; Skov, F.; Svenning, J.-C. Potential impact of climate change on the distribuions and civersity patterns of European mammals. Biodivers. Conserv. 2007, 16, 3803–3816. [Google Scholar] [CrossRef]
- Thomas, C.D.; Cameron, A.; Green, R.E.; Bakkenes, M.; Beaumont, L.J.; Collingham, Y.C.; Erasmus, B.F.N.; de Siqueira, M.F.; Grainger, A.; Hannah, L.; et al. Extinction risk from climate change. Nature 2004, 427, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.K.; Thomas, C.D.; Fox, R.; Telfer, M.G.; Willis, S.G.; Asher, J.; Huntley, B. Responses of butterflies to twentieth century climate warming: Implications for future ranges. Proc. Biol. Sci. 2002, 269, 2163–2171. [Google Scholar] [CrossRef] [PubMed]
- Pearson, R.G.; Dawson, T.P. Predicting the impacts of climate change on the distribution of speces: Are bioclimate envelope models useful? Global Ecol. Biogeogr. 2003, 12, 361–371. [Google Scholar] [CrossRef]
- Thuiller, W. Climate change and the ecologist. Nature 2007, 448, 550–552. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, M.C.; Weltzin, J.F.; Sanders, N.J.; Dunn, R.R. The biogeography of prediction error: Why does the introduced range of the fire ant over-predict its native range? Global Ecol. Biogeogr. 2007, 16, 24–33. [Google Scholar] [CrossRef]
- Beale, C.M.; Lennon, J.J.; Gimona, A. Opening the climate envelope reveals no macroscale associations with climate in European birds. Proc. Natl. Acad. Sci. USA 2008, 105, 14908–14912. [Google Scholar] [CrossRef] [PubMed]
- Guisan, A.; Thuiller, W. Predicting species distribution: Offering more than simple habitat models. Ecol. Lett. 2005, 8, 993–1009. [Google Scholar] [CrossRef]
- Fitzpatrick, M.C.; Hargrove, W.W. The projection of species distribution models and the problem of non-analog climate. Biodivers. Conserv. 2009, 18, 2255–2261. [Google Scholar] [CrossRef]
- Wisz, M.S.; Pottier, J.; Kissling, W.D.; Pellissier, L.; Lenoir, J.; Damgaard, C.F.; Dormann, C.F.; Forchhammer, M.C.; Grytnes, J.A.; Guisan, A.; et al. The role of biotic interactions in shaping distributions and realised assemblages of species: Implications for species distribution modelling. Biol. Rev. 2013, 88, 15–30. [Google Scholar] [CrossRef] [PubMed]
- De Araújo, C.B.; Marcondes-Machado, L.O.; Costa, G.C. The importance of biotic interactions in species distribution models: A test of the Eltonian noise hypothesis using parrots. J. Biogeogr. 2014, 41, 513–523. [Google Scholar] [CrossRef]
- Cahill, A.E.; Aiello-Lammens, M.E.; Fisher-Reid, M.C.; Hua, X.; Karanewsky, C.J.; Yeong Ryu, H.; Sbeglia, G.C.; Spagnolo, F.; Waldron, J.B.; Warsi, O.; et al. How does climate change cause extinction? Proc. R. Soc. B Biol. Sci. 2013, 280, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Graham, R.W.; Grimm, E.C. Effects of global climate change on the patterns of terrestrial biological communities. Trends Ecol. Evol. 1990, 5, 289–292. [Google Scholar] [CrossRef]
- Yvonne, C.; Mark, O.; Collingham, Y.C.; HIll, M.O.; Huntley, B. The migration of sessile organisms: A simulation model with measurable parameters. J. Veg. Sci. 1996, 7, 831–846. [Google Scholar]
- Barlow, E.J.; Daunt, F.; Wanless, S.; Reid, J.M. Estimating dispersal distributions at multiple scales: Within-colony and among-colony dispersal rates, distances and directions in European Shags Phalacrocorax aristotelis. Ibis 2013, 155, 762–778. [Google Scholar] [CrossRef]
- Harris, M.P.; Wanless, S. The Puffin; T & AD Poyser: London, UK, 2011. [Google Scholar]
- Danchin, E.; Boulinier, T.; Massot, M. Conspecific reproductive success and breeding habitat selection: implications for the study of coloniality. Ecology 1998, 79, 2415–2428. [Google Scholar] [CrossRef]
- Boulinier, T.; McCoy, K.D.; Yoccoz, N.G.; Gasparini, J.; Tveraa, T. Public information affects breeding dispersal in a colonial bird: Kittiwakes cue on neighbours. Biol. Lett. 2008, 4, 538–540. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, E.A.; Burger, J. Biology of Marine Birds; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Mitchell, P.I.; Newton, S.F.; Ratcliffe, N.; Dunn, T.E. Seabird Populations of Britain and Ireland: Results of the Seabird 2000 Census; T. & A.D. Poyser: London, UK, 2004. [Google Scholar]
- Thaxter, C.B.; Lascelles, B.; Sugar, K.; Cook, A.S.C.P.; Roos, S.; Bolton, M.; Langston, R.H.W.; Burton, N.H.K. Seabird foraging ranges as a tool for identifying Marine Protected Areas. Biol. Conserv. 2012, 156, 53–61. [Google Scholar] [CrossRef]
- Barbraud, C.; Weimerskirch, H. Antarctic birds breed later in response to climate change. Proc. Natl. Acad. Sci. USA 2006, 103, 6248–6251. [Google Scholar] [CrossRef] [PubMed]
- Frederiksen, M.; Harris, M.P.; Daunt, F.; Rothery, P.; Wanless, S. Scale-dependent climate signals drive breeding phenology of three seabird species. Global Chang. Biol. 2004, 10, 1214–1221. [Google Scholar] [CrossRef]
- Frederiksen, M.; Wanless, S.; Harris, M.P.; Rothery, P.; Wilson, L.J. The role of industrial fisheries and oceanographic change in the decline of North Sea black-legged kittiwakes. J. Appl. Ecol. 2004, 41, 1129–1139. [Google Scholar] [CrossRef]
- Russell, D.J.F.; Wanless, S.; Collingham, Y.C.; Anderson, B.J.; Beale, C.; Reid, J.B.; Huntley, B.; Hamer, K.C. Beyond climate envelopes: Bio-climate modelling accords with observed 25-year changes in seabird populations of the British Isles. Divers. Distrib. 2015, 21, 211–222. [Google Scholar] [CrossRef]
- Furness, R.W.; Tasker, M. Seabird-fishery interactions: Quantifying the sensitivity of seabirds to reductions in sandeel abundance, and identification of key areas for sensitive seabirds in the North Sea. Mar. Ecol. Prog. Ser. 2000, 202, 253–264. [Google Scholar] [CrossRef]
- Arnott, S.; Ruxton, G. Sandeel recruitment in the North Sea: Demographic, climatic and trophic effects. Mar. Ecol. Prog. Ser. 2002, 238, 199–210. [Google Scholar]
- Oswald, S.A.; Bearhop, S.; Furness, R.W.; Huntley, B.; Hamer, K.C. Heat stress in a high-latitude seabird: Effects of temperature and food supply on bathing and nest attendance of great skuas Catharacta skua. J. Avian Biol. 2008, 39, 163–169. [Google Scholar] [CrossRef]
- Thompson, K.R.; Furness, R.W. The influence of rainfall and nest-site quality on the population-dynamics of the Manx Shearwater Puffinus puffinus on Rhum. J. Zool. 1991, 225, 427–437. [Google Scholar] [CrossRef]
- Gray, C.; Phillips, R.; Hamer, K. Non-random nestling mortality in northern fulmars: Implications for monitoring marine environments. J. Zool. 2003, 259, 109–113. [Google Scholar] [CrossRef]
- Hagemeijer, W.J.M.; Blair, M.J. The EBCC Atlas of European Breeding Birds. Their Distribution and Abundance; T. & A.D. Poyser: London, UK, 1997. [Google Scholar]
- Collins, M.; Knutti, R.; Arblaster, J.; Dufresne, J.-L.; Fichefet, T.; Friedlingstein, P.; Gao, X.; Gutowski, W.; Johns, T.; Krinner, G.; et al. Long-term Climate Change: Projections, Commitments and Irreversibility. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocke, T., Qin, D., Plattner, G., Tignor, M., Allen, S., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P., Eds.; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M.; Averyt, K.; Tignor, M.; Miller, H.L. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Manning, M., Chen, Z., Marquis, M., Averyt, K., Tignor, M., Miller, H.L., Eds.; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Beaumont, L.J.; Pitman, A.J.; Poulsen, M.; Hughes, L. Where will species go? Incorporating new advances in climate modelling into projections of species distributions. Global Chang. Biol. 2007, 13, 1368–1385. [Google Scholar] [CrossRef]
- World Data Center for Climate. Lowe IPCC DDC AR4 UKMO-HadGEM SRESA1B run1. CERA-DB “UKMO_HadGEM_SRESA1B_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- World Data Center for Climate. Lowe IPCC DDC AR4 UKMO-HadGEM SRESA2 run1. CERA-DB “UKMO_HadGEM_SRESA2_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- World Data Center for Climate. Roeckner IPCC DDC AR4 ECHAM5/MPI-OM SRESA1B run1. CERA-DB “EH5_MPI_OM_SRESA1B_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- World Data Center for Climate. Roeckner IPCC DDC AR4 ECHAM5/MPI-OM SRESA2 run1. CERA-DB “EH5_MPI_OM_SRESA2_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- World Data Center for Climate. GFDL IPCC DDC AR4 GFDL-CM2.1 SRESA1B run1. CERA-DB “GFDL_CM2.1_SRESA1B_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- World Data Center for Climate. GFDL IPCC DDC AR4 GFDL-CM2.1 SRESA2 run1. CERA-DB “GFDL_CM2.1_SRESA2_1” 2005. Available online: http://cera-www.dkrz.de/WDCC/ui/ (accessed on 5 December 2008).
- Huntley, B.; Green, R.E.; Collingham, Y.C.; Willis, S.G. Climate Atlas of Breeding Birds in Europe; Lynx Edicions: Barcelona, Spain, 2007. [Google Scholar]
- Burnham, K.; Anderson, D. Model Selection and Multimodel Inference: A Practical Information Theoretic Approach; Springer-Verlag: New York, NY, USA, 2002. [Google Scholar]
- R Core Team Team. R: A Language and Environment for Statistical Computing; Vienna, Austria, 2014. [Google Scholar]
- Gill, F.; Donsker, D. IOC World Bird List 2014, v 4.2. Available online: http://www.worldbirdnames.org/ (accessed on 1 August 2015).
- Manel, S.; Ceri Williams, H.; Ormerod, S.J. Evaluating presence-absence models in ecology: The need to account for prevalence. J. Appl. Ecol. 2001, 38, 921–931. [Google Scholar] [CrossRef]
- Thuiller, W.; Brotons, L.; Araújo, M.B.; Lavorel, S. Effects of restricting environmental range of data to project current and future species distributions. Ecography 2004, 27, 165–172. [Google Scholar] [CrossRef]
- Wisz, M.S.; Hijmans, R.J.; Li, J.; Peterson, A.T.; Graham, C.H.; Guisan, A.; Elith, J.; Dudík, M.; Ferrier, S.; Huettmann, F.; et al. Effects of sample size on the performance of species distribution models. Divers. Distrib. 2008, 14, 763–773. [Google Scholar] [CrossRef]
- Thomas, C.D.; Lennon, J.J. Birds extend their ranges northwards. Nature 1999. [Google Scholar] [CrossRef]
- Parmesan, C.; Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 2003, 421, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Hickling, R.; Roy, D.B.; Hill, J.K.; Thomas, C.D. A northward shift of range margins in British Odonata. Global Chang. Biol. 2005, 11, 502–506. [Google Scholar] [CrossRef]
- Dunnet, G.M.; Ollason, J.C. The estimation of survival rate in the fulmar Fulmarus glacialis. J. Anim. Ecol. 1978, 47, 507–520. [Google Scholar] [CrossRef]
- Coulson, J.C. The population dynamics of culling Herring Gulls Larus argentatus and Lesser Black-backed Gulls Larus fuscus. In Bird Population Studies: Relevance to Conservation and Management; Perrins, C., Lebreton, J., Hirons, G., Eds.; Oxford University Press: Oxford, UK, 1991. [Google Scholar]
- Reynolds, T.J.; King, R.; Harwood, J.; Frederiksen, M.; Harris, M.P.; Wanless, S. Integrated data analysis in the presence of emigration and mark loss. J. Agric. Biol. Environ. Stat. 2009, 14, 411–431. [Google Scholar] [CrossRef]
- Lavers, J.L.; Jones, I.L.; Diamond, A.W. Natal and Breeding Dispersal of Razorbills (Alca torda) in Eastern North America. Waterbirds 2007, 30, 588–594. [Google Scholar] [CrossRef]
- Frederiksen, M.; Petersen, A. The Importance of Natal Dispersal in a Colonial Seabird, the Black Guillemot Cepphus grylle. Ibis 2000, 142, 48–57. [Google Scholar] [CrossRef]
- Becker, P.H.; Ezard, T.H.G.; Ludwigs, J.D.; Sauer-Gürth, H.; Wink, M. Population sex ratio shift from fledging to recruitment: Consequences for demography in a philopatric seabird. Oikos 2008, 117, 60–68. [Google Scholar] [CrossRef]
- Coulson, J.C.; Coulson, B.A. Measuring immigration and philopatry in seabirds; Recruitment to Black-legged Kittiwake colonies. Ibis 2008, 150, 288–299. [Google Scholar] [CrossRef]
- Kim, S.Y.; Torres, R.; Rodríguez, C.; Drummond, H. Effects of breeding success, mate fidelity and senescence on breeding dispersal of male and female blue-footed boobies. J. Anim. Ecol. 2007, 76, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Devlin, C.M.; Diamond, A.W.; Kress, S.W.; Hall, C.S.; Welch, L. Breeding dispersal and survival of arctic terns (Sterna paradisaea) nesting in the Gulf of Maine. Auk 2008, 125, 850–858. [Google Scholar] [CrossRef]
- Coulson, J.C.; Nève de Mévergnies, G. Where do young kittiwakes Rissa tridactyla breed? Philopatry or dispersal? Ardea 1992, 80, 187–197. [Google Scholar]
- Phillips, B.L.; Brown, G.P.; Travis, J.M.J.; Shine, R. Reid’s paradox revisited: The evolution of dispersal kernels during range expansion. Am. Nat. 2008, 172, S34–S48. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P. Identifying drivers of change; did fisheries play a role in the spread of North Atlantic fulmars? In Conservation Biology Series; Boyd, I., Wanless, S., Camphuysen, C.J., Eds.; Cambridge University Press: Cambridge, UK, 2006; pp. 143–156. [Google Scholar]
- Fisher, J. The Fulmar Population. Nature 1952, 170, 725–725. [Google Scholar]
- Wynne-Edwards, V. Animal Dispersion in Relation to Social Behaviour. Can. Med. Assoc. J. 1963, 88, 1255–1256. [Google Scholar]
- Salomonsen, F. The geographical variation of the fulmar Fulmarus glacialis and the zones of marine environment in the North Atlantic. Auk 1965, 82, 327–355. [Google Scholar] [CrossRef] [Green Version]
- Blois, J.L.; Zarnetske, P.L.; Fitzpatrick, M.C.; Finnegan, S. Climate change and the past, present, and future of biotic interactions. Science 2013, 341, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Molinos, J.G.; Halpern, B.S.; Schoeman, D.S.; Brown, C.J.; Kiessling, W.; Moore, P.J.; Pandolfi, J.M.; Poloczanska, E.S.; Richardson, A.J.; Burrows, M.T. Climate velocity and the future global redistribution of marine biodiversity. Nat. Clim. Chang. 2015, 4–11. [Google Scholar]
- Petitgas, P.; Alheit, J.; Peck, M.; Raab, K.; Irigoien, X.; Huret, M.; van der Kooij, J.; Pohlmann, T.; Wagner, C.; Zarraonaindia, I.; et al. Anchovy population expansion in the North Sea. Mar. Ecol. Prog. Ser. 2012, 444, 1–13. [Google Scholar] [CrossRef]
- Heath, M.R.; Neat, F.C.; Pinnegar, J.K.; Reid, D.G.; Sims, D.W.; Wright, P.J. Review of climate change impacts on marine fish and shellfish around the UK and Ireland. Aquat. Conserv. Mar. Freshw. Ecosyst. 2012, 22, 337–367. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Russell, D.J.F.; Wanless, S.; Collingham, Y.C.; Huntley, B.; Hamer, K.C. Predicting Future European Breeding Distributions of British Seabird Species under Climate Change and Unlimited/No Dispersal Scenarios. Diversity 2015, 7, 342-359. https://doi.org/10.3390/d7040342
Russell DJF, Wanless S, Collingham YC, Huntley B, Hamer KC. Predicting Future European Breeding Distributions of British Seabird Species under Climate Change and Unlimited/No Dispersal Scenarios. Diversity. 2015; 7(4):342-359. https://doi.org/10.3390/d7040342
Chicago/Turabian StyleRussell, Deborah J.F., Sarah Wanless, Yvonne C. Collingham, Brian Huntley, and Keith C. Hamer. 2015. "Predicting Future European Breeding Distributions of British Seabird Species under Climate Change and Unlimited/No Dispersal Scenarios" Diversity 7, no. 4: 342-359. https://doi.org/10.3390/d7040342
APA StyleRussell, D. J. F., Wanless, S., Collingham, Y. C., Huntley, B., & Hamer, K. C. (2015). Predicting Future European Breeding Distributions of British Seabird Species under Climate Change and Unlimited/No Dispersal Scenarios. Diversity, 7(4), 342-359. https://doi.org/10.3390/d7040342





