Impact of Dams on Riparian Frog Communities in the Southern Western Ghats, India
Abstract
:1. Introduction
2. Methods
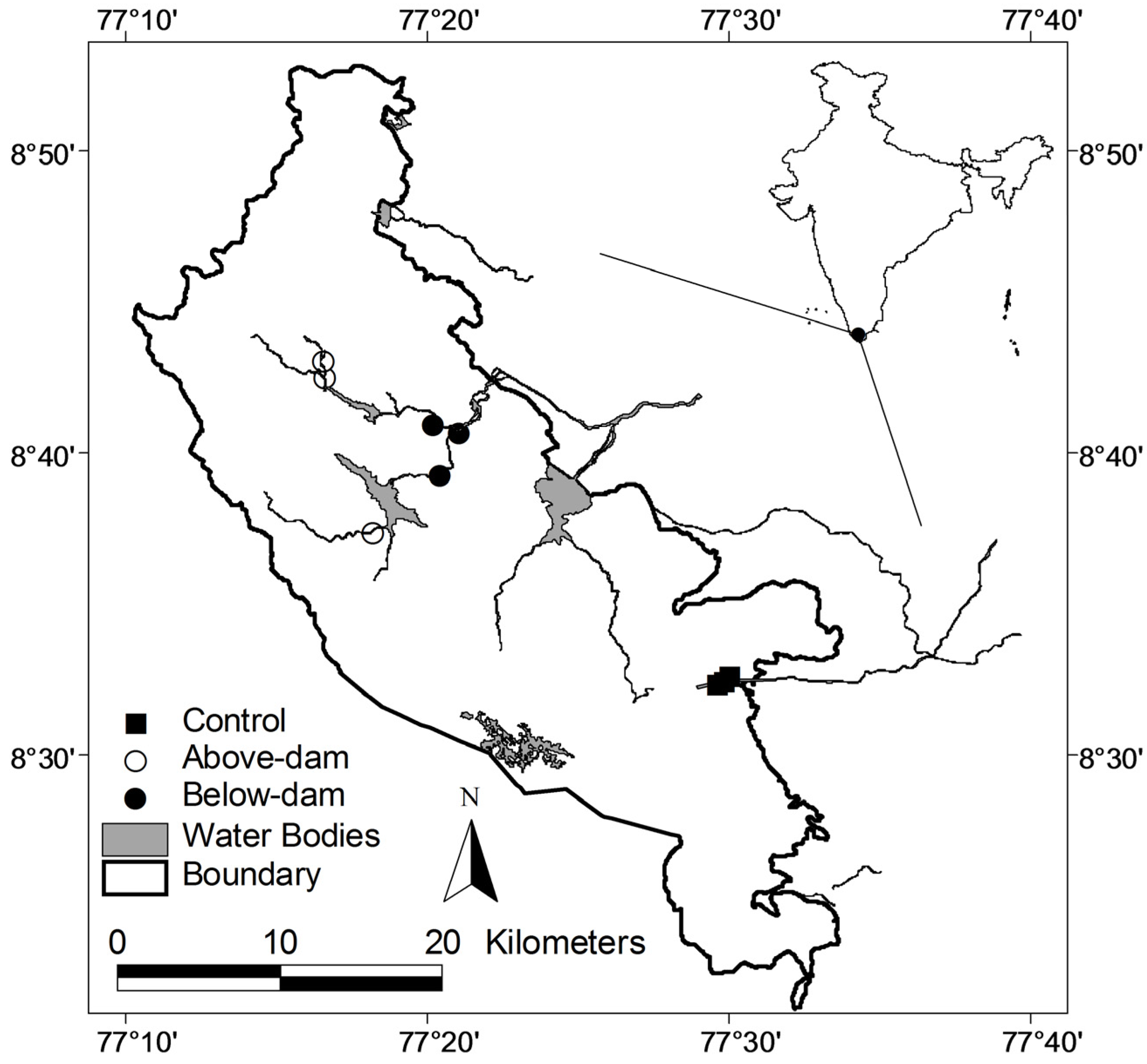
| Site Name | Sampling Time | Drainage | Location w.r.t. Dam | Elevation (m) | Observed Frog Species Richness | Estimated Frog Species Richness (±SE) |
|---|---|---|---|---|---|---|
| Karayar | March-05 | Banathirtam | Below | 180 | ||
| Mundanthurai | December-04 | Valayar | Below | 180 | ||
| Banathirtam | January-05 | Banathirtam | Above | 350 | 10 | 10.67 ± 0.37 |
| Kodamadi | December-04 | Valayar | Above | 240 | ||
| Madangal | December-04 and January-05 | Valayar | Above | 390 | ||
| Thalayanai 1 | April-05 | Pachayar | No Dam | 190 | 9 | 9.67 ± 0.39 |
| Thalayanai 2 | April-05 | Pachayar | No Dam | 200 | ||
| Thalayanai 3 | April-05 | Pachayar | No Dam | 210 |
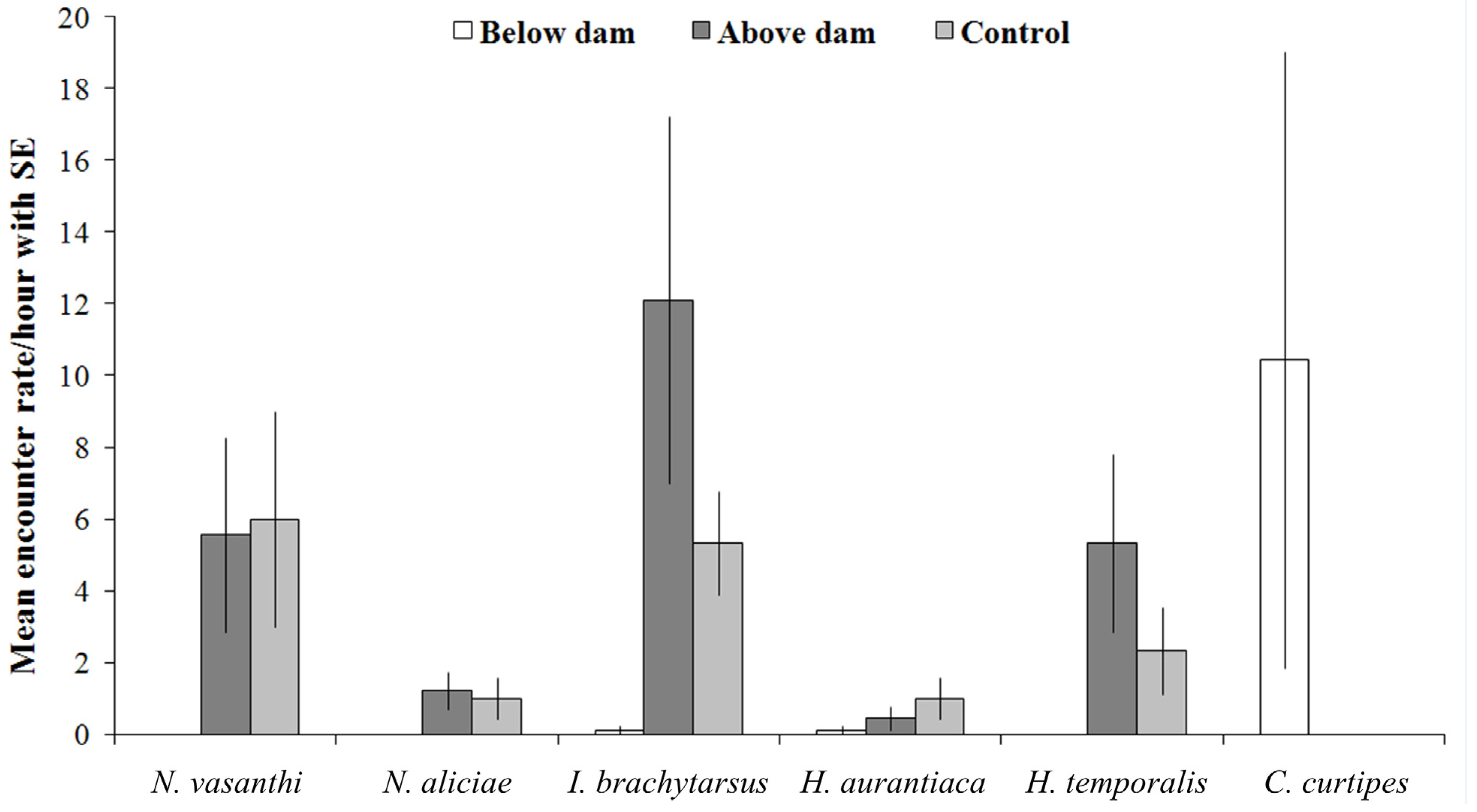
3. Results
| Species | Below-Dam | Above-Dam | Control | Distribution | Indicator Value % | Group | P |
|---|---|---|---|---|---|---|---|
| Clinotarsus curtipes | 94 | 0 | 0 | Endemic | 56 | Below-dam | 0.806 |
| Indirana brachytarsus | 1 | 109 | 16 | Endemic | 98 | Above-dam | 0.004 |
| Micrixalus fuscus | 0 | 55 | 0 | Endemic | |||
| Nyctibatrachus aliciae | 0 | 11 | 3 | Endemic | 67 | Above-dam | 0.046 |
| Nyctibatrachus vasanthi | 0 | 50 | 18 | Endemic | 67 | Above-dam | 0.046 |
| Philautus sp.* | 0 | 1 | 0 | Endemic | |||
| Hylarana aurantiaca | 1 | 4 | 3 | Endemic | 60 | Above-dam | 0.089 |
| Duttaphrynus melanostictus | 6 | 3 | 0 | Non-endemic | 22 | Below-dam | 0.806 |
| Euphlyctis cyanophlyctis | 86 | 4 | 97 | Non-endemic | 77 | Above-dam | 0.114 |
| Fejervarya cf. limnocharis | 138 | 0 | 29 | Non-endemic | 48 | Below-dam | 0.779 |
| Hoplobatrachus crassus | 7 | 0 | 0 | Non-endemic | 44 | Below-dam | 0.446 |
| Microhyla rubra | 5 | 0 | 0 | Non-endemic | 22 | Below-dam | 1.000 |
| Polypedates maculatus | 2 | 0 | 3 | Non-endemic | 27 | Above-dam | 0.683 |
| Hylarana temporalis | 0 | 48 | 7 | Non-endemic | 67 | Above-dam | 0.046 |
| Fejervarya keralensis | 2 | 15 | 22 | Non-endemic | 97 | Above-dam | 0.009 |
| Total | 342 | 300 | 198 |
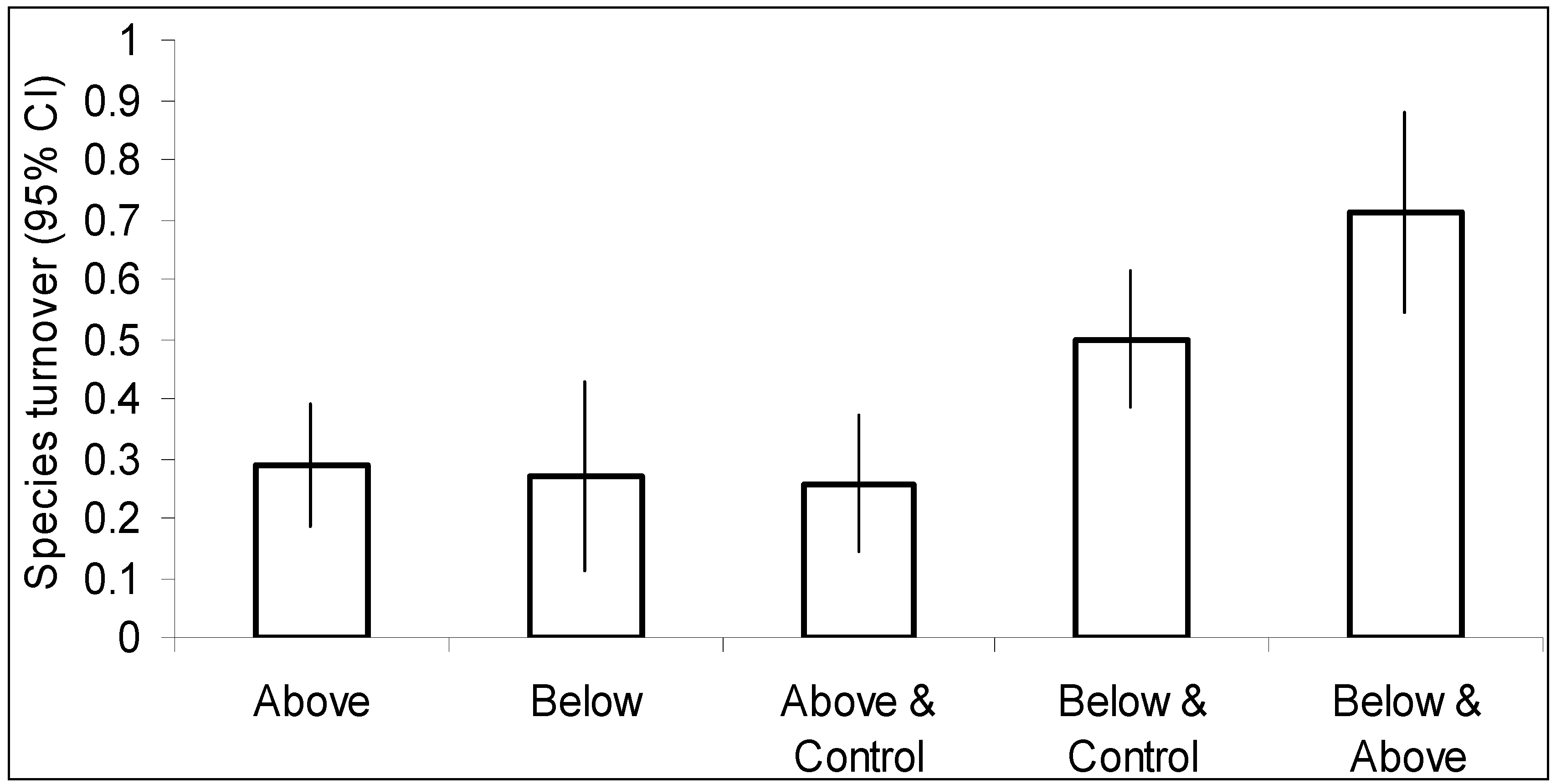
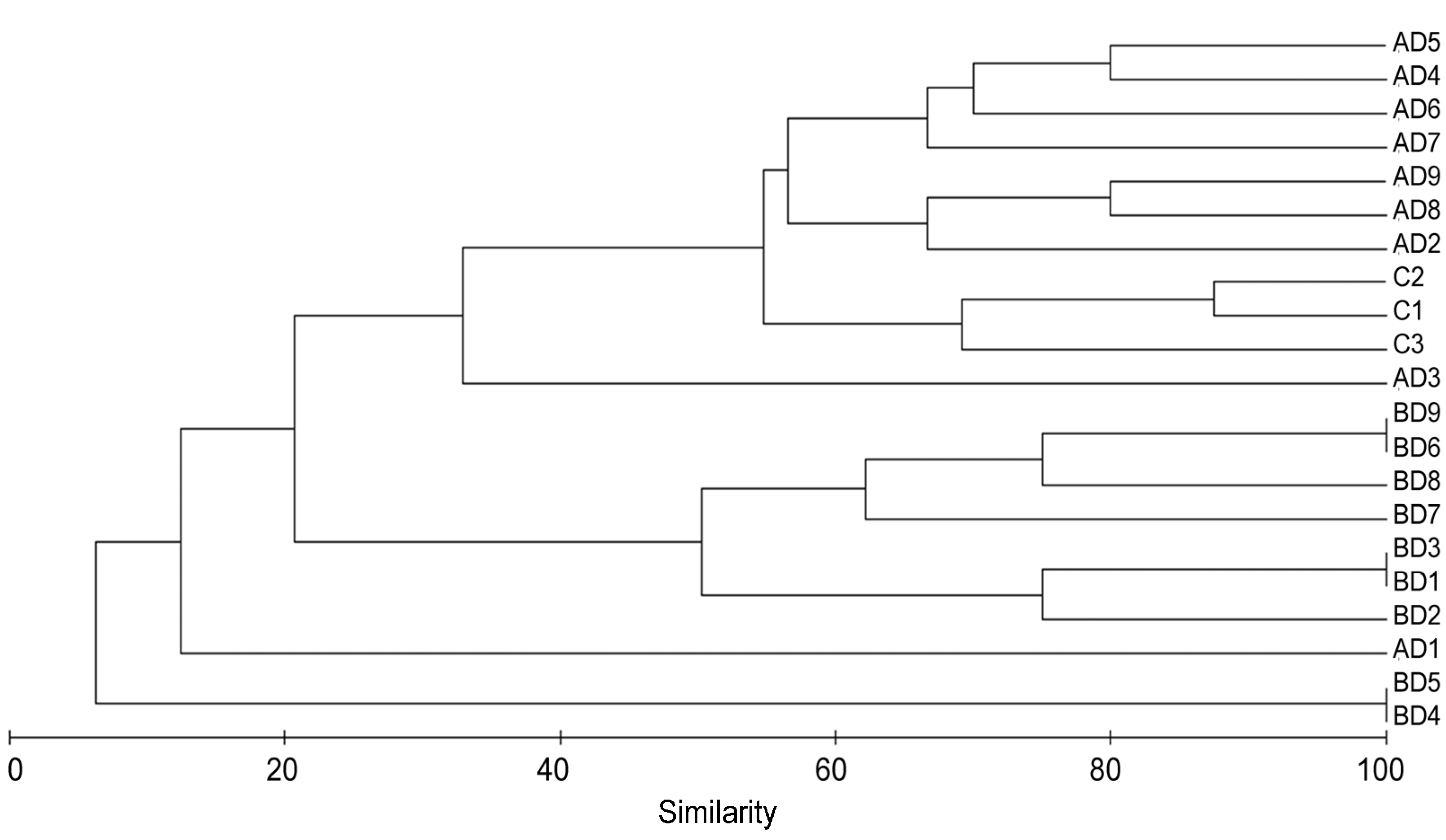
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Inger, R.F.; Dutta, S.K. An overview of the amphibian fauna of India. J. Bomb. Nat. Hist. Soc. 1986, 83, 135–146. [Google Scholar]
- Daniels, R. Geographical distribution patterns of amphibians in the Western Ghats, India. J. Biogeog. 1992, 19, 521–529. [Google Scholar] [CrossRef]
- Inger, R.F. Distribution of amphibians in southern Asia and adjacent islands. In Patterns of Distributions of Amphibians: A Global Perspective; Duellman, W.E., Ed.; Johns Hopkins University Press: Baltimore, MD, USA & London, UK, 1999; pp. 445–482. [Google Scholar]
- Vasudevan, K.; Kumar, A.; Chellam, R. Species turnover: The case of stream amphibians of rainforests in the Western Ghats, Southern India. Biodiv. Conserv. 2006, 15, 3515–3525. [Google Scholar] [CrossRef]
- Naniwadekar, R.; Vasudevan, K. Patterns in diversity of anurans along an elevational gradient in the Western Ghats, South India. J. Biogeog. 2007, 34, 842–853. [Google Scholar] [CrossRef]
- Vasudevan, K.; Kumar, A.; Chellam, R.; Noon, B.R. Density and diversity of forest floor amphibians in the rainforests of the southern Western Ghats. Herpetologica 2008, 64, 207–215. [Google Scholar] [CrossRef]
- Poff, N.L.; Allan, J.D.; Bain, M.B.; Karr, J.R.; Prestegaard, K.L.; Richter, B.D.; Sparks, R.E.; Stromberg, J.C. The natural flow regime. A paradigm for river conservation and restoration. BioScience 1997, 47, 769–784. [Google Scholar]
- Sparks, R.E.; Nelson, J.C.; Yin, Y. Naturalization of the flood regime in regulated rivers. BioScience 1998, 48, 706–720. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Mooney, H.A.; Lubchenco, J.; Melillo, J.M. Human domination of earth’s ecosystems. Science 1997, 277, 494–499. [Google Scholar] [CrossRef]
- Magilligan, F.J.; Nislow, K.H. Changes in hydrologic regime by dams. Geomorphology 2005, 71, 61–78. [Google Scholar] [CrossRef]
- Scheidegger, K.J.; Bain, M.B. Larval fish distribution and microhabitat use in free-flowing and regulated rivers. Copeia 1995, 125–135. [Google Scholar] [CrossRef]
- Nislow, K.H.; Magilligan, F.J.; Fassnacht, H.; Bechtel, D.; Ruesink, A. Effects of dam impoundment on the flood regime of natural floodplain communities in the upper Connecticut River. J. Am. Water Res. Assoc. 2002, 38, 1533–1548. [Google Scholar] [CrossRef]
- Rood, S.; Mahoney, J.M. Collapse of riparian poplar forests downstream from dams in western prairies: Probable causes and prospects for mitigation. Environ. Manag. 1990, 14, 451–464. [Google Scholar] [CrossRef]
- Ligon, F.K.; Dietrich, W.E.; Trush, W.J. Downstream ecological effects of dams. BioScience 1995, 45, 183–192. [Google Scholar] [CrossRef]
- Brooker, M.P. The impact of impoundments on the downstream fisheries and general ecology of rivers. In Advances of Applied Ecology; Coaker, T.H., Ed.; Academic Press: London, UK, 1981; pp. 91–152. [Google Scholar]
- Ward, J.V.; Stanford, J.A. Ecological connectivity in alluvial river ecosystems and its disruption by flow regulation. Regul. Rivers: Res. Manag. 1995, 11, 105–119. [Google Scholar]
- Nilsson, C.; Svedmark, M. Basic principles and ecological consequences of changing water regimes: Riparian plant communities. Environ. Manag. 2002, 30, 468–480. [Google Scholar] [CrossRef]
- Merritt, D.M.; Wohl, E.E. Plant dispersal along rivers fragmented by dams. River Res. Appl. 2006, 22, 1–26. [Google Scholar] [CrossRef]
- Molles, M.C., Jr.; Crawford, C.S.; Ellis, L.M.; Valett, H.M.; Dahm, C.N. Managed flooding for riparian ecosystem restoration. BioScience 1998, 48, 749–756. [Google Scholar]
- Toth, L.A.; Melvin, S.L.; Arrington, D.A.; Chamberlain, J. Hydrologic manipulations of the channelized Kissimmee River. BioScience 1998, 48, 757–764. [Google Scholar] [CrossRef]
- Bodie, J.R. Stream and riparian management for freshwater turtles. J. Environ. Manag. 2001, 62, 443–455. [Google Scholar] [CrossRef]
- Tedonkeng Pamo, E.; Tchamba, M.N. Elephants and vegetation change in the Sahelo-Soudanian region of Cameroon. J. Arid Environ. 2001, 48, 243–253. [Google Scholar]
- Lind, A.J.; Welsh, H.H., Jr.; Wilson, R.A. The effects of a dam on breeding habitat and egg survival of the foothill yellow-legged frog (Rana boylii) in northwestern California. Herpet. Rev. 1996, 27, 62–67. [Google Scholar]
- Ramesh, B.R.; Menon, S.; Bawa, K.S. A vegetation based approach to biodiversity gap analysis in the Agasthyamalai region, Western Ghats, India. Ambio 1997, 26, 529–536. [Google Scholar]
- Vasudevan, K.; Kumar, A.; Chellam, R. Structure and composition of rainforest floor amphibian communities in Kalakad-Mundanthurai Tiger Reserve. Curr. Sci. 2001, 80, 406–412. [Google Scholar]
- Crump, M.L.; Scott, N.J., Jr. Visual encounter surveys. In Measuring and Monitoring Biological Diversity: Standard Methods for Amphibians; Heyer, W.R., Donnelly, M.A., McDiarmid, R.W., Hayek, L.C., Foster, M.S., Eds.; Smithsonian Institution: Washington, DC, USA, 1994; pp. 84–92. [Google Scholar]
- Colwell, R.K. EstimateS: Statistical estimation of species richness and shared species from samples, 2005 Version 7.5 . Available online: http://viceroy.eeb.uconn.edu/estimates/index.html (accessed on 27 August 2014).
- Wolda, H. Similarity indices, sample size and diversity. Oecologia 1981, 50, 296–302. [Google Scholar] [CrossRef]
- Dufrene, M.; Legendre, P. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecol. Monogr. 1997, 67, 345–366. [Google Scholar]
- McCune, B.; Mefford, M.J. PC-ORD: Multivariate analysis of ecological data, Version 4 for Windows; MjM Software Design: Edinburgh, UK, 1999. [Google Scholar]
- Brandao, R.A.; Araujo, A.F.B. Changes in anuran species richness and abundance resulting from hydroelectric dam flooding in central Brazil. Biotropica 2008, 40, 263–266. [Google Scholar] [CrossRef]
- Eskew, E.A.; Price, S.J.; Dorcas, M.E. Effects of river-flow Regulation on anuran occupancy and abundance in riparian zones. Conserv. Biol. 2012, 26, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Kupferberg, S.J.; Palen, W.J.; Lind, A.J.; Bobzien, S.; Catenazzi, A.; Drennan, J.O.E.; Power, M.E. Effects of flow regimes altered by dams on survival, population declines, and range-wide Losses of California river-breeding frogs. Conserv. Biol. 2012, 26, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Kunte, K. Natural history and reproductive behaviour of Nyctibatrachus cf. humayuni (Anura: Ranidae). Herpet. Rev. 2004, 35, 137–140. [Google Scholar]
- Jansson, R.; Nilsson, C.; Renöfält, B. Fragmentation of riparian floras in rivers with multiple dams. Ecology 2000, 81, 899–903. [Google Scholar] [CrossRef]
- Peek, R.A. Landscape Genetics of Foothill Yellow-Legged Frogs (Rana boylii) in Regulated and Unregulated Rivers: Assessing Connectivity and Genetic Fragmentation. Master’s Thesis, University of San Francisco, San Francisco, CA, USA, 2011. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Naniwadekar, R.; Vasudevan, K. Impact of Dams on Riparian Frog Communities in the Southern Western Ghats, India. Diversity 2014, 6, 567-578. https://doi.org/10.3390/d6030567
Naniwadekar R, Vasudevan K. Impact of Dams on Riparian Frog Communities in the Southern Western Ghats, India. Diversity. 2014; 6(3):567-578. https://doi.org/10.3390/d6030567
Chicago/Turabian StyleNaniwadekar, Rohit, and Karthikeyan Vasudevan. 2014. "Impact of Dams on Riparian Frog Communities in the Southern Western Ghats, India" Diversity 6, no. 3: 567-578. https://doi.org/10.3390/d6030567
APA StyleNaniwadekar, R., & Vasudevan, K. (2014). Impact of Dams on Riparian Frog Communities in the Southern Western Ghats, India. Diversity, 6(3), 567-578. https://doi.org/10.3390/d6030567





