Enhancing Soil Quality and Plant Health Through Suppressive Organic Amendments
Abstract
:1. Organic Matter and Soil Quality
2. Organic Soil Amendments and Their Effect on Soil Quality
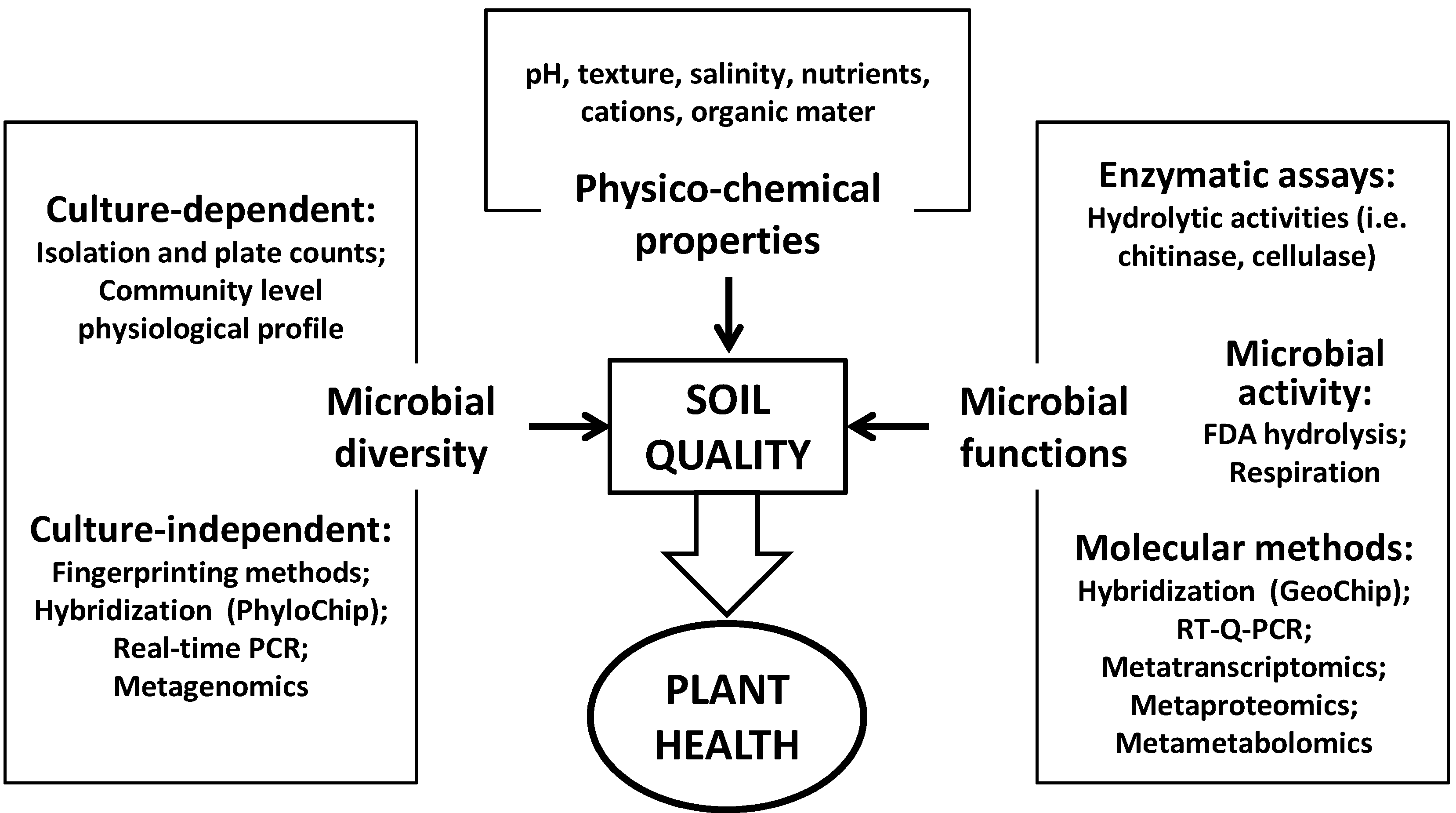
2.1. Microbial Populations and Community Composition
2.2. Microbial Activity and Soil Functioning
3. Organic Amendments and Their Effect on Plant Health
| Pathogen | Crop | Organic amendment | Reference |
|---|---|---|---|
| Fusarium spp. | Several hosts | Vegetal composts | Yogev et al. 2006 [36] |
| Phytophthora cinnamomi | White lupin | Fresh and composted chicken manure | Aryantha et al. 2000 [14] |
| Avocado | Sludge vermicompost | Bender et al. 1992 [43] | |
| Chipped eucaliptus trimmings | Downer et al. 2001 [37] | ||
| Pythium ultimum | Garden cress | Animal and vegetal composts | Pane et al. 2011 [41] |
| Bark compost | Erhart et al. 1999 [40] | ||
| Rhizoctonia solani | Garden cress | Viticulture waste compost Composted cow manure | Pane et al. 2011 [41] |
| Basil | Fresh farmyard manure | Tamm et al. 2010 [42] | |
| Rosellinia necatrix | Avocado | Vegetal composts | Bonilla et al. 2009 [38] |
| Sclerotinia minor | Garden cress | Composted municipal bio-waste Composted cow manure | Pane et al. 2011 [41] |
| Sclerotium rolfsii | Tomato | Vegetal compost Poultry manure Green manure (legumes) | Bulluck and Ristaino 2002 [13] |
| Verticillium dahliae | Eggplant | Horse manure Municipal green waste Wood shavings | Malandraki et al. 2008 [39] |
3.1. The Basis of Soil Suppressiveness
3.2. Looking for Indicators of Soil Suppressiveness
3.2.1. Physicochemical Soil Properties
3.2.2. Microbial Biomass, Diversity and Community Structure
3.2.3. Microbial Functions and Activities
4. Future Perspectives
5. Conclusions
Acknowledgements
References
- Fonte, S.J.; Yeboah, E.; Ofori, P.; Quansah, G.W.; Vanlauwe, B.; Six, J. Fertilizer and residue quality effects on organic matter stabilization in soil aggregates. Soil Sci. Soc. Am. J. 2009, 73, 961–966. [Google Scholar]
- Bailey, K.L.; Lazarovits, G. Suppressing soil-borne diseases with residue management and organic amendments. Soil Tillage Res. 2003, 72, 169–180. [Google Scholar] [CrossRef]
- Doran, J.W.; Zeiss, M.R. Soil health and sustainability: managing the biotic component of soil quality. Appl. Soil Ecol. 2000, 15, 3–11. [Google Scholar] [CrossRef]
- Lazarovits, G. Management of soil-borne plant pathogens with organic soil amendments: a disease control strategy salvaged from the past. Can. J. Plant Pathol. 2001, 23, 1–7. [Google Scholar] [CrossRef]
- Doran, J.W.; Parkin, T.B. Defining and assessing soil quality. In Defining soil quality for a sustainable environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; Volume Special Publication 35, pp. 3–21. [Google Scholar]
- Saison, C.; Degrange, V.; Oliver, R.; Millard, P.; Commeaux, C.; Montange, D.; Le Roux, X. Alteration and resilience of the soil microbial community following compost amendment: effects of compost level and compost-borne microbial community. Environ. Microbiol. 2006, 8, 247–257. [Google Scholar] [CrossRef]
- Bonilla, N.; Cazorla, F.M.; Martínez-Alonso, M.; Hermoso, J.M.; González-Fernández, J.; Gaju, N.; Landa, B.B.; de Vicente, A. Organic amendments and land management affect bacterial community composition, diversity and biomass in avocado crop soils. Plant Soil 2012, 357, 215–226. [Google Scholar] [CrossRef]
- Pérez-Piqueres, A.; Edel-Hermann, V.; Alabouvette, C.; Steinberg, C. Response of soil microbial communities to compost amendments. Soil Biol. Biochem. 2006, 38, 460–470. [Google Scholar] [CrossRef]
- Albiach, R.; Canet, R.; Pomares, F.; Ingelmo, F. Microbial biomass content and enzymatic activities after the application of organic amendments to a horticultural soil. Bioresour. Technol. 2000, 75, 43–48. [Google Scholar] [CrossRef]
- Janvier, C.; Villeneuve, F.; Alabouvette, C.; Edel-Hermann, V.; Mateille, T.; Steinberg, C. Soil health through soil disease suppression: Which strategy from descriptors to indicators? Soil Biol. Biochem. 2007, 39, 1–23. [Google Scholar] [CrossRef]
- Tiquia, S.M.; Lloyd, J.; Herms, D.A.; Hoitink, H.A.J.; Michel, F.C., Jr. Effects of mulching and fertilization on soil nutrients, microbial activity and rhizosphere bacterial community structure determined by analysis of TRFLPs of PCR-amplified 16S rRNA genes. Appl. Soil Ecol. 2002, 21, 31–48. [Google Scholar] [CrossRef]
- Peacock, A.D.; Mullen, M.D.; Ringelberg, D.B.; Tyler, D.D.; Hedrick, D.B.; Gale, P.M.; White, D.C. Soil microbial community responses to dairy manure or ammonium nitrate applications. Soil Biol. Biochem. 2001, 33, 1011–1019. [Google Scholar] [CrossRef]
- Bulluck, L.R.; Ristaino, J.B. Effect of synthetic and organic soil fertility amendments on southern blight, soil microbial communities, and yield of processing tomatoes. Phytopathology 2002, 92, 181–189. [Google Scholar] [CrossRef]
- Aryantha, I.P.; Cross, R.; Guest, D.J. Suppression of Phytophthora cinnamomi in potting mixes amended with uncomposted and composted animal manures. Phytopathology 2000, 90, 775–782. [Google Scholar] [CrossRef]
- Yang, Y.J.; Dungan, R.S.; Ibekwe, A.M.; Valenzuela-Solano, C.; Crohn, D.M.; Crowley, D.E. Effect of organic mulches on soil bacterial communities one year after application. Biol. Fertil. Soils 2003, 38, 273–281. [Google Scholar] [CrossRef]
- Crecchio, C.; Curci, M.; Mininni, R.; Ricciuti, P.; Ruggiero, P. Short-term effects of municipal solid waste compost amendments on soil carbon and nitrogen content, some enzyme activities and genetic diversity. Biol. Fertil. Soils 2001, 34, 311–318. [Google Scholar] [CrossRef]
- Edel-Hermann, V.; Dreumont, C.; Pérez-Piqueres, A.; Steinberg, C. Terminal restriction fragment length polymorphism analysis of ribosomal RNA genes to assess changes in fungal community structure in soils. FEMS Microbiol. Ecol. 2004, 47, 397–404. [Google Scholar] [CrossRef]
- Van Elsas, J.D.; Costa, R. Molecular assessment of soil microbial communities with potential for plant disease suppression. In Biotechnology and Plant Disease Management; Punja, Z.K., Boer, S.H., Sanfaçon, H., Eds.; CAB International: King's Lynn, UK, 2007; p. 498. [Google Scholar]
- Mazzola, M. Assessment and menagement of soil microbial community structure for disease suppression. Annu. Rev. Phytopathol. 2004, 42, 35–59. [Google Scholar] [CrossRef]
- Wertz, S.; Degrange, V.; Prosser, J.I.; Poly, F.; Commeaux, C.; Freitag, T.; Guillaumaud, N.; Roux, X.L. Maintenance of soil functioning following erosion of microbial diversity. Environ. Microbiol. 2006, 8, 2162–2169. [Google Scholar] [CrossRef]
- Peter, H.; Beier, S.; Bertilsson, S.; Lindstrom, E.S.; Langenheder, S.; Tranvik, L.J. Function-specific response to depletion of microbial diversity. ISME J. 2011, 5, 351–361. [Google Scholar] [CrossRef]
- Frossard, A.; Gerull, L.; Mutz, M.; Gessner, M.O. Disconnect of microbial structure and function: enzyme activities and bacterial communities in nascent stream corridors. ISME J. 2012, 6, 680–691. [Google Scholar] [CrossRef]
- García, C.; Hernández, T.; Costa, F.; Ceccanti, B. Biochemical parameters in soils regenerated by the addition of organic wastes. Waste Manag. Res. 1994, 12, 457–466. [Google Scholar]
- Ros, M.; Hernandez, M.T.; Garca, C. Soil microbial activity after restoration of a semiarid soil by organic amendments. Soil Biol. Biochem. 2003, 35, 463–469. [Google Scholar] [CrossRef]
- Tejada, M.; Garcia, C.; Gonzalez, J.L.; Hernandez, M.T. Use of organic amendment as a strategy for saline soil remediation: Influence on the physical, chemical and biological properties of soil. Soil Biol. Biochem. 2006, 38, 1413–1421. [Google Scholar] [CrossRef]
- Goyal, S.; Chander, K.; Mundra, M.; Kapoor, K. Influence of inorganic fertilizers and organic amendments on soil organic matter and soil microbial properties under tropical conditions. Biol. Fertil. Soils 1999, 29, 196–200. [Google Scholar] [CrossRef]
- Nannipieri, P.; Ascher, J.; Ceccherini, M.T.; Landi, L.; Pietramellara, G.; Renella, G. Microbial diversity and soil functions. Eur. J. Soil Sci. 2003, 54, 655–670. [Google Scholar]
- Tiquia, S.M. Evolution of extracellular enzyme activities during manure composting. J. Appl. Microbiol. 2002, 92, 764–775. [Google Scholar] [CrossRef]
- Van Elsas, J.D.; Postma, J. Suppression of soil-borne phytopathogens by compost. In Compost Science and Technology; Diaz, L.F., de Bertoldi, M., Bidlingmaier, W., Stentiford, E., Eds.; Elsevier: Amsterdam, The Netherlands, 2007; Volume 8, pp. 201–214. [Google Scholar]
- Noble, R.; Coventry, E. Suppression of soil-borne plant diseases with composts: a review. Biocontrol Sci. Technol. 2005, 15, 3–20. [Google Scholar] [CrossRef]
- Roy, S.; Arunachalam, K.; Dutta, B.K.; Arunachalam, A. Effect of organic amendments of soil on growth and productivity of three common crops viz. Zea mays, Phaseolus vulgaris and Abelmoschus esculentus. Appl. Soil Ecol. 2010, 45, 78–84. [Google Scholar] [CrossRef]
- Yan, X.; Gong, W. The role of chemical and organic fertilizers on yield, yield variability and carbon sequestration: results of a 19-year experiment. Plant Soil 2010, 331, 471–480. [Google Scholar] [CrossRef]
- Hermoso, J.M.; Torres, M.D.; Farré, J.M. Effects of organic materials on adult Hass trees. In Proceedings of the VII World Avocado Congress 2011, Cairns, Australia, 5–9 September 2011.
- Baker, K.; Cook, R.J. Biological control of plant pathogens; WH Freeman and Company: San Francisco, CA, USA, 1974. [Google Scholar]
- Hoitink, H.A.L.; Boehm, M.J. Biocontrol within the context of soil microbial communities: a substrate-dependent phenomenon. Annu. Rev. Phytopathol. 1999, 37, 427–446. [Google Scholar] [CrossRef]
- Yogev, A.; Raviv, M.; Hadar, Y.; Cohen, R.; Katan, J. Plant waste-based composts suppressive to diseases caused by pathogenic Fusarium oxysporum. Eur. J. Plant Pathol. 2006, 116, 267–278. [Google Scholar] [CrossRef]
- Downer, A.J.; Menge, J.A.; Pond, E. Association of cellulytic enzyme activities in eucalyptus mulches with biological control of Phytophthora cinnamomi. Phytopathology 2001, 91, 847–855. [Google Scholar] [CrossRef]
- Bonilla, N.; Torés, J.A.; Hermoso, J.M.; González, J.; Cazorla, F.M.; de Vicente, A. Biological control of avocado root rots by suppressive organic amendments. In IOBC/wprs Bulletin: Biological control of fungal and bacterial plant pathogens; Elad, Y., Maurhofer, M., Keel, C., Gessler, C., Duffy, B., Eds.; Interlaken, Switzerland, 2009; Volume 43, pp. 231–234. [Google Scholar]
- Malandraki, I.; Tjamos, S.E.; Pantelides, I.S.; Paplomatas, E.J. Thermal inactivation of compost suppressiveness implicates possible biological factors in disease management. Biol. Control 2008, 44, 180–187. [Google Scholar] [CrossRef]
- Erhart, E.; Burian, K.; Hartl, W.; Stich, K. Suppression of Pythium ultimum by biowaste composts in relation to compost microbial miomass, activity and content of phenolic compounds. J. Phytopathol. 1999, 147, 299–305. [Google Scholar]
- Pane, C.; Spaccini, R.; Piccolo, A.; Scala, F.; Bonanomi, G. Compost amendments enhance peat suppressiveness to Pythium ultimum, Rhizoctonia solani and Sclerotinia minor. Biol. Control 2011, 56, 115–124. [Google Scholar] [CrossRef]
- Tamm, L.; Thürig, B.; Bruns, C.; Fuchs, J.; Köpke, U.; Laustela, M.; Leifert, C.; Mahlberg, N.; Nietlispach, B.; Schmidt, C.; et al. Soil type, management history, and soil amendments influence the development of soil-borne (Rhizoctonia solani, Pythium ultimum) and air-borne (Phytophthora infestans, Hyaloperonospora parasitica) diseases. Eur. J. Plant Pathol. 2010, 127, 465–481. [Google Scholar] [CrossRef]
- Bender, G.S.; Casale, W.L.; Rahimian, M. Use of worm-composted sludge as a soil amendment for avocados in Phytophthora-infested soil. In Proceeding of Second World Avocado Congress, Orange, CA, USA, 1992; p. 143.
- Pérez-Jiménez, R.M. Significant avocado diseases caused by fungi and oomycetes. Eur. J. Plant Sci. Biotechnol. 2008, 2, 1–24. [Google Scholar]
- Bonanomi, G.; Antignani, V.; Pane, C.; Scala, F. Suppression of soilborne fungal diseases with organic amendments. J. Plant Pathol. 2007, 89, 311–324. [Google Scholar]
- Delgado, M.M.; Martin, J.V.; Miralles, R.; León-Cófreces, C.; García, M.C. Phytotoxicity of uncomposted and composted poultry manure. Afr. J. Plant Sci. 2010, 4, 154–162. [Google Scholar]
- Wolstenholme, B.N. Prospects for integrated and biological control of avocado root rot-some overseas impressions. Available online: www.avocadosource.com/Journals/.../SAAGA_1979_PG_17-20.pdf (accessed on 14 December 2012).
- Tenuta, M.; Lazarovits, G. Ammonia and nitrous acid from nitrogenous amendments kill microsclerotia of Verticillium dahliae. Phytopathology 2002, 92, 255–264. [Google Scholar] [CrossRef]
- Steinberg, C.; Edel-Hermann, V.; Alabouvette, C.; Lemanceau, P. Soil suppressiveness to plant diseases. In Modern Soil Microbiology, 2nd; van Elsas, J.D., Jansson, J.C., Trevors, J.T., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 455–478. [Google Scholar]
- Weller, D.M.; Raaijmakers, J.M.; McSpadden, B.B.; Thomashow, L.S. Microbial populations responsible for specific soil suppressiveness to plant pathogens. Annu. Rev. Phytopathol. 2002, 40, 309–348. [Google Scholar] [CrossRef]
- Bonanomi, G.; Antignani, V.; Capodilupo, M.; Scala, F. Identifying the characteristics of organic soil amendments that suppress soilborne plant diseases. Soil Biol. Biochem. 2010, 42, 136–144. [Google Scholar] [CrossRef]
- Garbeva, P.; Postma, J.; van Veen, J.A.; van Elsas, J.D. Effect of above-ground plant species on soil microbial community structure and its impact on suppression of Rhizoctonia solani AG3. Environ. Microbiol. 2006, 8, 233–246. [Google Scholar] [CrossRef]
- Cohen, M.F.; Yamasaki, H.; Mazzola, M. Brassica napus seed meal soil amendment modifies microbial community structure, nitric oxide production and incidence of Rhizoctonia root rot. Soil Biol. Biochem. 2005, 37, 1215–1227. [Google Scholar] [CrossRef]
- Borrero, C.; Trillas, M.I.; Ordovás, J.; Tello, J.C.; Avilés, M. Predictive factors for the suppression of Fusarium wilt of tomato in plant growth media. Phytopathology 2004, 94, 1094–1101. [Google Scholar] [CrossRef]
- Van Os, G.J.; van Ginkel, J.H. Suppression of Pythium root rot in bulbous Iris in relation to biomass and activity of the soil microflora. Soil Biol. Biochem. 2001, 33, 1447–1454. [Google Scholar] [CrossRef]
- Heyman, F.; Lindahl, B.; Persson, L.; Wikström, M.; Stenlid, J. Calcium concentrations of soil affect suppressiveness against Aphanomyces root rot of pea. Soil Biol. Biochem. 2007, 39, 2222–2229. [Google Scholar] [CrossRef]
- Larkin, R.P.; Honeycutt, C.W. Effects of different 3-year cropping systems on soil microbial communities and rhizoctonia diseases of potato. Phytopathology 2006, 96, 68–79. [Google Scholar] [CrossRef]
- Grünwald, N.J.; Hu, S.; van Bruggen, A.H.C. Short-term cover crop decomposition in organic and conventional soils: characterization of soil C, N, microbial and plant pathogen dynamics. Eur. J. Plant Pathol. 2000, 106, 37–50. [Google Scholar] [CrossRef]
- Van Elsas, J.D.; Garbeva, P.; Salles, J. Effects of agronomical measures on the microbial diversity of soils as related to the suppression of soil-borne plant pathogens. Biodegradation 2002, 13, 29–40. [Google Scholar] [CrossRef]
- Jin, H.; Sun, O.J.; Liu, J. Changes in soil microbial biomass and community structure with addition of contrasting types of plant litter in a semiarid grassland ecosystem. J. Plant Ecol. 2010, 3, 209–217. [Google Scholar] [CrossRef]
- Larkin, R.P.; Larkin, R.P.; Hopkins, D.L.; Martin, F.N. Suppression of Fusarium wilt of watermelon by nonpathogenic Fusarium oxysporum and other microorganisms recovered from disease-suppressive soil. Phytopathology 1996, 86, 812–819. [Google Scholar] [CrossRef]
- Sneh, B.; Dupler, M.; Elad, Y.; Baker, R. Chlamydospore germination of Fusarium oxysporum f.sp.cucumerinum as affected by fluorescent and lytic bacteria from a Fusarium -suppressive soil. Phytopathology 1984, 74, 1115–1124. [Google Scholar] [CrossRef]
- Weller, D.M.; Cook, R.J. Suppression of take-all of wheat by seed treatments with fluorescent pseudomonads. Phytopathology 1983, 73, 463–469. [Google Scholar] [CrossRef]
- Simon, A.; Sivasithamparam, K. Pathogen-suppression: A case study in biological suppression of Gaeumannomyces graminis var. tritici in soil. Soil Biol. Biochem. 1989, 21, 331–337. [Google Scholar] [CrossRef]
- Duffy, B.K.; Ownley, B.H.; Weller, D.M. Soil chemical and physical properties associated with suppression of take-all of wheat by Trichoderma koningii. Phytopathology 1997, 87, 1118–1124. [Google Scholar] [CrossRef]
- Duffy, B.K.; Simon, A.; Weller, D.M. Combination of Trichoderma koningii with fluorescent pseudomonads for control of talk-all on wheat. Phytopathology 1996, 86, 188–194. [Google Scholar] [CrossRef]
- Alabouvette, C.; Backhouse, D.; Steinberg, C.; Donovan, N.J.; Edel-Hermann, V.; Burgess, L.W. Microbial diversity in soil: effects on crop health. In Managing Soil Quality: Challenges in Modern Agriculture; Schjonning, P., Elmholt, S., Christensen, B.T., Eds.; CAB International: Wallingford, UK, 2004; pp. 121–138. [Google Scholar]
- Gomez, E.; Ferreras, L.; Toresani, S. Soil bacterial functional diversity as influenced by organic amendment application. Bioresour. Technol. 2006, 97, 1484–1489. [Google Scholar] [CrossRef]
- Thomashow, L.S.; Weller, D.M.; Bonsall, R.F.; Pierson, L.S. Production of the antibiotic phenazine-1-carboxylic acid by fluorescent Pseudomonas species in the rhizosphere of wheat. Appl. Environ. Microbiol. 1990, 56, 908–912. [Google Scholar]
- Raaijmakers, J.M.; Weller, D.M. Natural plant protection by 2,4-diacetylphloroglucinol-producing Pseudomonas spp. in take-all decline soils. Mol. Plant-Microbe Interact. 1998, 11, 144–152. [Google Scholar] [CrossRef]
- Raaijmakers, J.M.; Weller, D.M.; Thomashow, L.S. Frequency of antibiotic-producing Pseudomonas spp. in natural environments. Appl. Environ. Microbiol. 1997, 63, 881–887. [Google Scholar]
- Simon, A.; Dunlop, R.W.; Ghisalberti, E.L.; Sivasithamparam, K. Trichoderma koningii produces a pyrone compound with antibiotic properties. Soil Biol. Biochem. 1988, 20, 263–264. [Google Scholar] [CrossRef]
- Ghisalberti, E.L.; Narbey, M.J.; Dewan, M.M.; Sivasithamparam, K. Variability among strains of Trichoderma harzianum in their ability to reduce take-all and to produce pyrones. Plant Soil 1990, 121, 287–291. [Google Scholar] [CrossRef]
- Weller, D.M. Pseudomonas biocontrol agents of soilborne pathogens: looking back over 30 years. Phytopathology 2007, 97, 250–256. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-Growth-Promoting Rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Ahmed, A.S.; Ezziyyani, M.; Pérez-Sánchez, C.; Candela, M.E. Effect of chitin on biological control activity of Bacillus spp. and Trichoderma harzianum against root rot disease in pepper (Capsicum annuum) plants. Eur. J. Plant Pathol. 2003, 109, 633–637. [Google Scholar] [CrossRef]
- Viterbo, A.; Ramot, O.; Chernin, L.; Chet, I. Significance of lytic enzymes from Trichoderma spp. in the biocontrol of fungal plant pathogens. Antonie Leeuwenhoek 2002, 81, 549–556. [Google Scholar] [CrossRef]
- Chae, D.H.; Jin, R.D.; Hwangbo, H.; Kim, Y.H.; Kim, Y.W.; Park, R.D.; Krishnan, H.B.; Kim, K.Y. Control of late blight (Phytophthora capsici ) in pepper plant with a compost containing multitude of chitinase-producing bacteria. Biocontrol Sci. Technol. 2006, 51, 339–351. [Google Scholar]
- Torsvik, V.; Øvreås, L. Microbial diversity and function in soil: from genes to ecosystems. Curr. Opin. Microbiol. 2002, 5, 240–245. [Google Scholar] [CrossRef]
- Mendes, R.; Kruijt, M.; de Bruijn, I.; Dekkers, E.; van der Voort, M.; Schneider, J.H.M.; Piceno, Y.M.; DeSantis, T.Z.; Andersen, G.L.; Bakker, P.A.H.M. Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 2011, 332, 1097–1100. [Google Scholar]
- He, Z.; Gentry, T.J.; Schadt, C.W.; Wu, L.; Liebich, J.; Chong, S.C.; Huang, Z.; Wu, W.; Gu, B.; Jardine, P.; et al. GeoChip: a comprehensive microarray for investigating biogeochemical, ecological and environmental processes. ISME J. 2007, 1, 67–77. [Google Scholar] [CrossRef]
- Jansson, J.K.; Neufeld, J.D.; Moran, M.A.; Gilbert, J.A. Omics for understanding microbial functional dynamics. Environ. Microbiol. 2012, 14, 1–3. [Google Scholar] [CrossRef]
- Van Elsas, J.D.; Costa, R.; Jansson, J.; Sjöling, S.; Bailey, M.; Nalin, R.; Vogel, T.M.; van Overbeek, L. The metagenomics of disease-suppressive soils–experiences from the METACONTROL project. Trends Biotechnol. 2008, 26, 591–601. [Google Scholar] [CrossRef]
- Poulsen, P.H.B.; Al-Soud, W.A.; Bergmark, L.; Magid, J.; Hansen, L.H.; Sørensen, S.J. Effects of fertilization with urban and agricultural organic wastes in a field trial – Prokaryotic diversity investigated by pyrosequencing. Soil Biol. Biochem. 2012. [Google Scholar] [CrossRef]
- Hollister, E.B.; Hu, P.; Wang, A.S.; Hons, F.M.; Gentry, T.J. Differential impacts of brassicaceous and nonbrassicaceous oilseed meals on soil bacterial and fungal communities. FEMS Microbiol. Ecol. 2012. [Google Scholar] [CrossRef]
- Yogev, A.; Raviv, M.; Hadar, Y.; Cohen, R.; Wolf, S.; Gil, L.; Katan, J. Induced resistance as a putative component of compost suppressiveness. Biol. Control 2010, 54, 46–51. [Google Scholar] [CrossRef]
- Hadar, Y. Suppressive compost: when plant pathology met microbial ecology. Phytoparasitica 2011, 39, 311–314. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bonilla, N.; Gutiérrez-Barranquero, J.A.; Vicente, A.D.; Cazorla, F.M. Enhancing Soil Quality and Plant Health Through Suppressive Organic Amendments. Diversity 2012, 4, 475-491. https://doi.org/10.3390/d4040475
Bonilla N, Gutiérrez-Barranquero JA, Vicente AD, Cazorla FM. Enhancing Soil Quality and Plant Health Through Suppressive Organic Amendments. Diversity. 2012; 4(4):475-491. https://doi.org/10.3390/d4040475
Chicago/Turabian StyleBonilla, Nuria, José A. Gutiérrez-Barranquero, Antonio De Vicente, and Francisco M. Cazorla. 2012. "Enhancing Soil Quality and Plant Health Through Suppressive Organic Amendments" Diversity 4, no. 4: 475-491. https://doi.org/10.3390/d4040475
APA StyleBonilla, N., Gutiérrez-Barranquero, J. A., Vicente, A. D., & Cazorla, F. M. (2012). Enhancing Soil Quality and Plant Health Through Suppressive Organic Amendments. Diversity, 4(4), 475-491. https://doi.org/10.3390/d4040475




