Species List and Temporal Trends of a Butterfly Community in an Urban Remnant in the Atlantic Forest
Abstract
1. Introduction
2. Methods
2.1. Study Area and Sampling
2.2. Butterfly Sampling
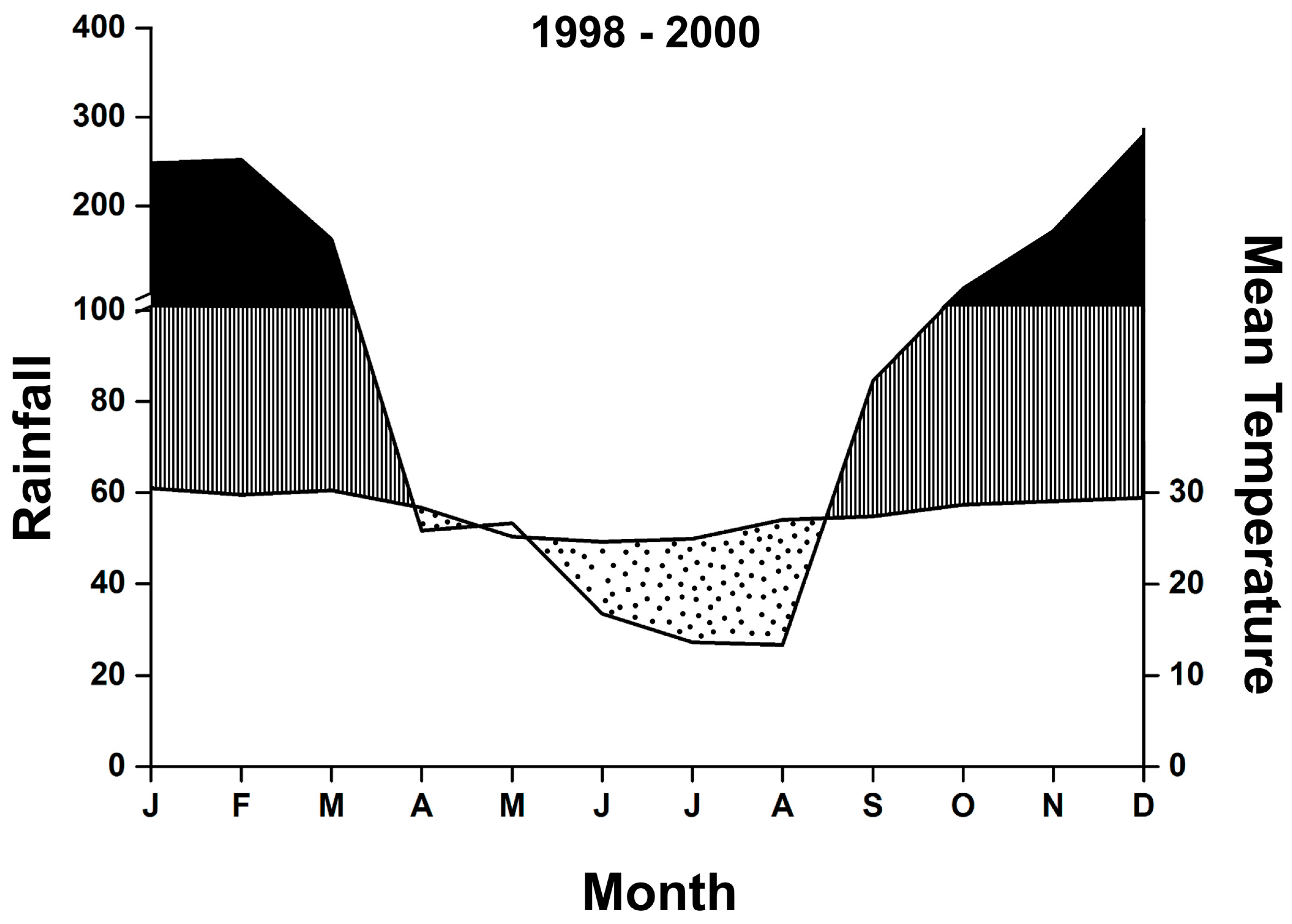
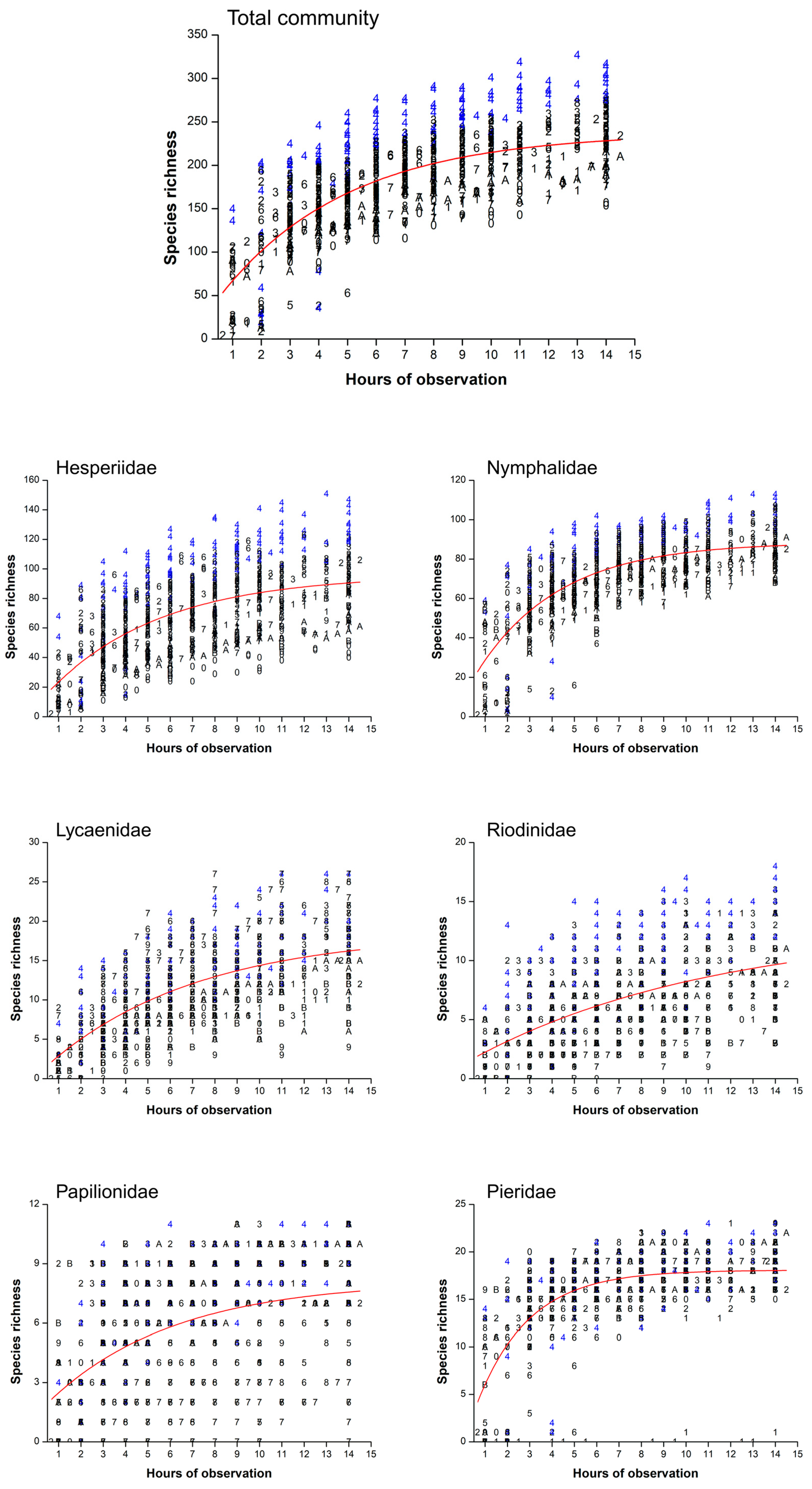
2.3. Temporal Analysis
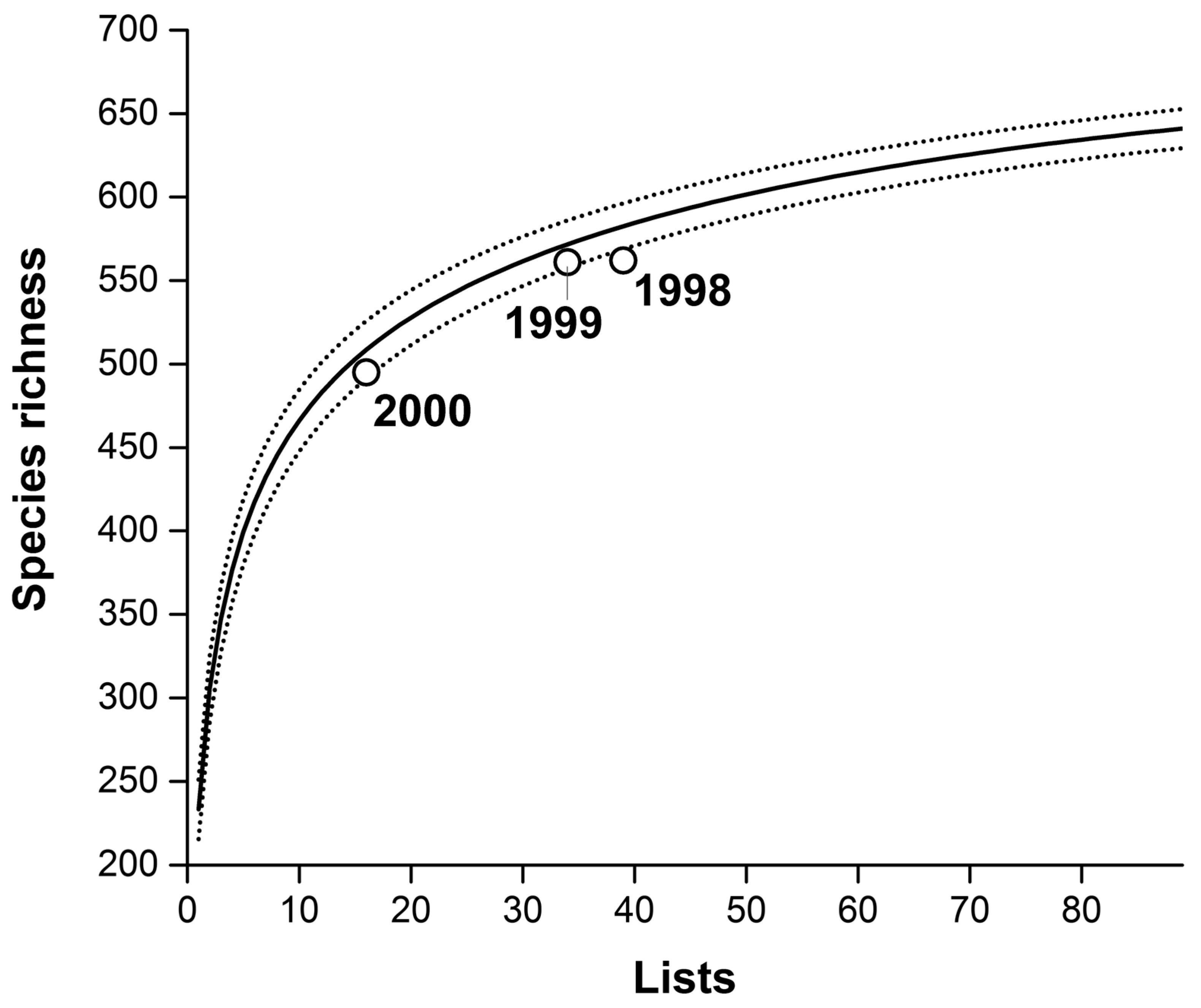
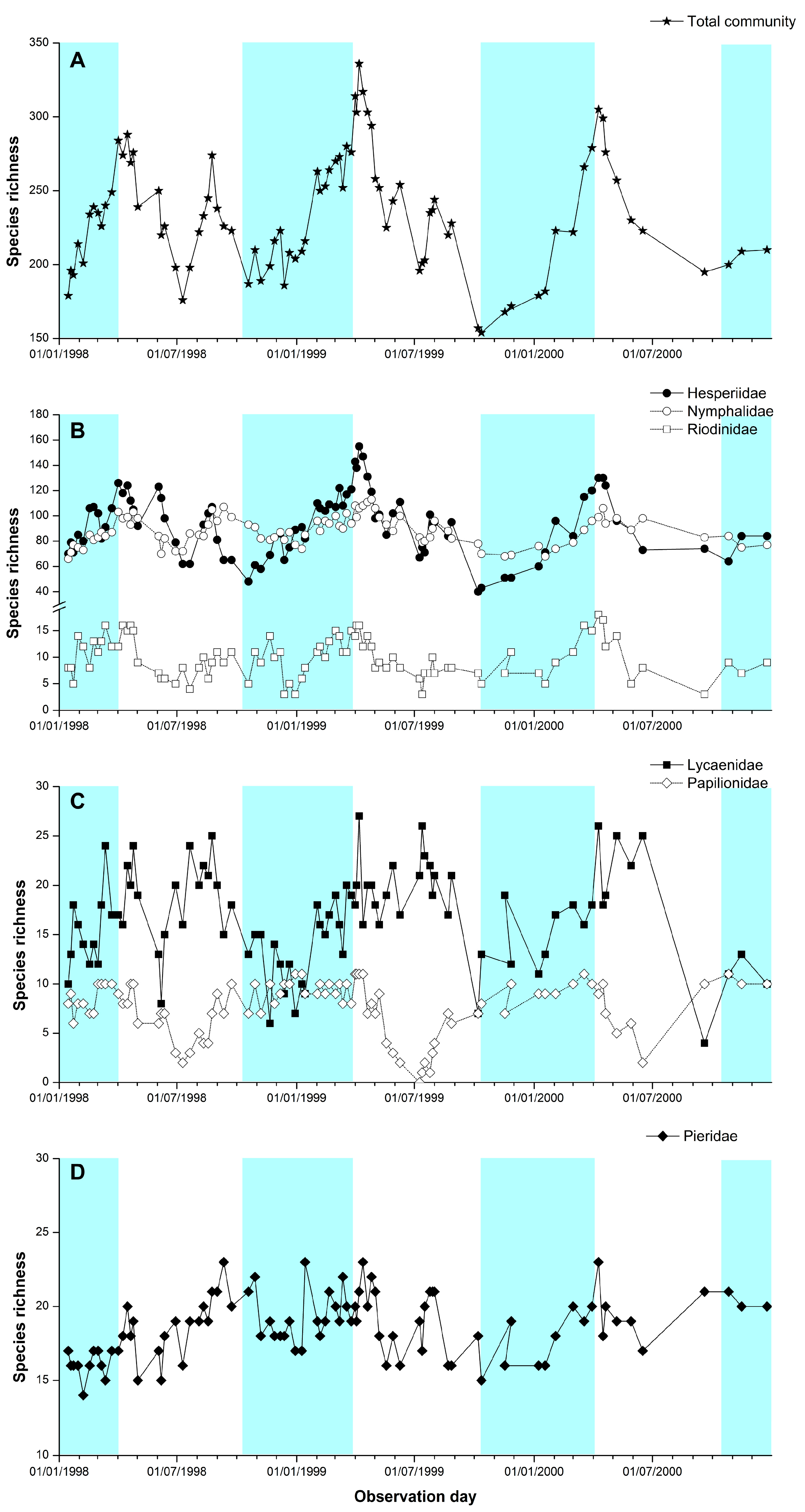
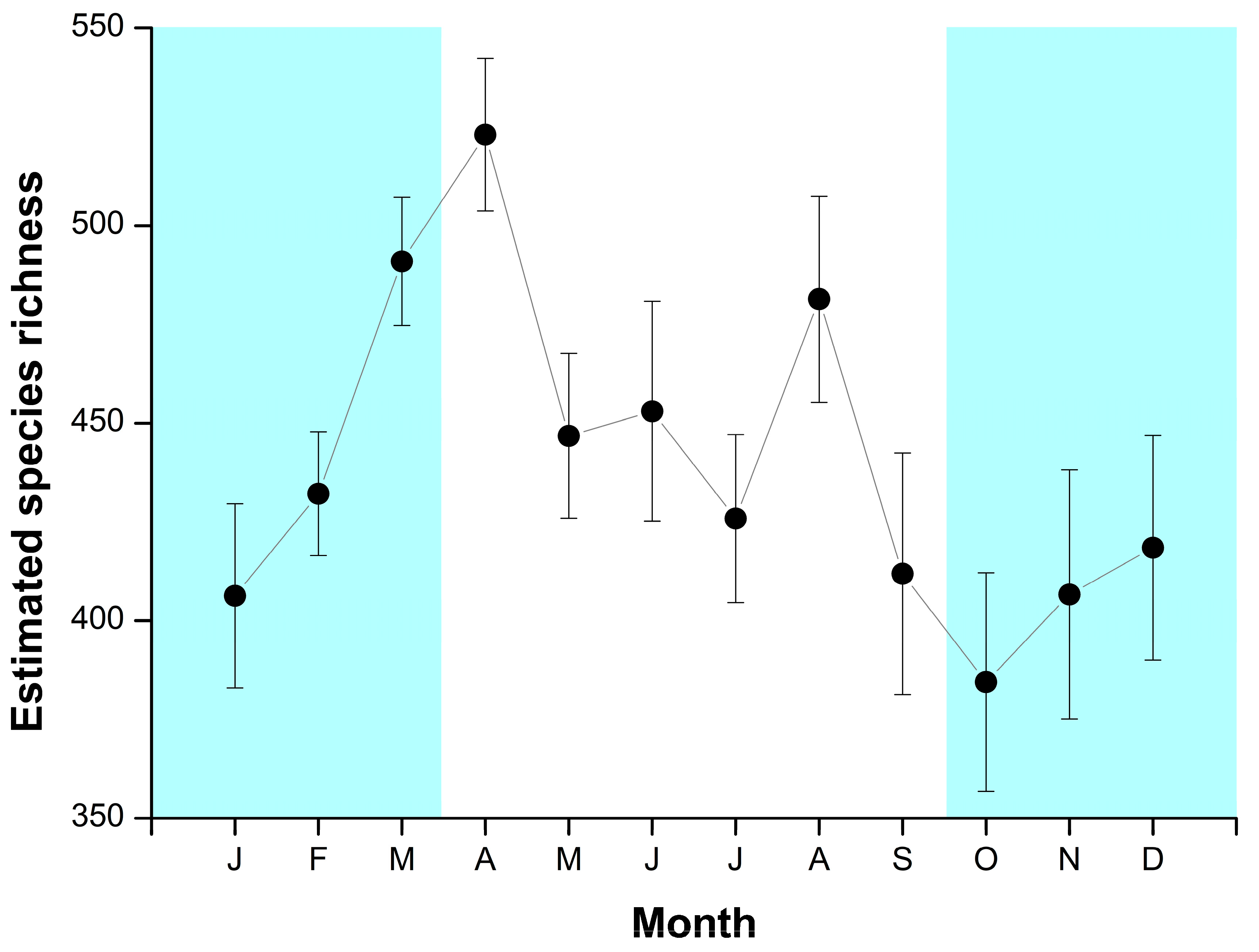
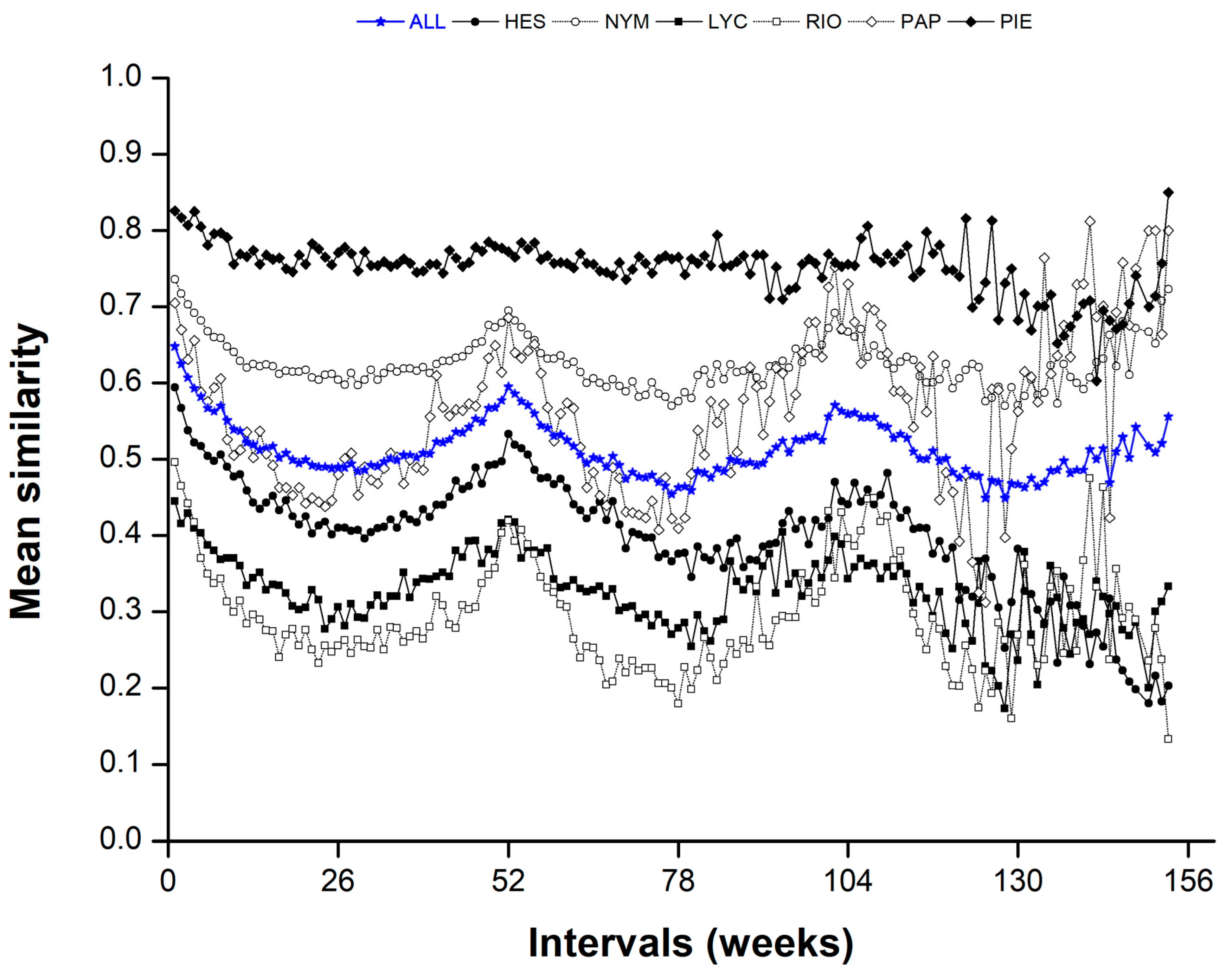
3. Results
Temporal Patterns
4. Discussion
4.1. Butterfly Diversity at MSG and Community Dynamics
4.2. Temporal Patterns
4.3. Implications for Inventories and Monitoring
4.4. Surrogates to Biodiversity Conservation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| PAPILIONOIDEA (706) | |
| Hesperiidae (314) | |
| Eudaminae (66) | |
| Entheini (2) | |
| Augiades vespasius vespasius (Fabricius, 1793) | |
| Phanus australis L. Miller, 1965 | |
| Eudamini (53) | |
| Aguna albistria albistria (Plötz, 1880) | |
| Aguna asander asander (Hewitson, 1867) | |
| Aguna cirrus Evans, 1952 | |
| Aguna megacles megacles (Mabille, 1888) | |
| Aguna metophis (Latreille [1824]) | |
| Aguna squamalba Austin and O. Mielke, 1998 | |
| Astraptes aulus (Plötz, 1881) | |
| Astraptes enotrus (Stoll, 1781) | |
| Astraptes janeira (Schaus, 1902) | |
| Autochton neis (Geyer, 1832) | |
| Autochton reflexus (Mabille and Boullet, 1912) | |
| Autochton sulfureolus (Mabille, 1883) | |
| Cecropterus dorantes dorantes (Stoll, 1790) | |
| Cecropterus doryssus albicuspis (Herrich-Schäffer, 1869) | |
| Cecropterus longipennis (Plötz, 1882) | |
| Cecropterus rica (Evans, 1952) | |
| Cecropterus virescens (Mabille, 1877) | |
| Cecropterus zarex (Hübner, 1818) | |
| Cephise cephise (Herrich-Schäffer, 1869) | |
| Chioides catillus catillus (Cramer, 1779) | |
| Codatractus aminias (Hewitson, 1867) | |
| Ectomis caunus (Herrich-Schäffer, 1869) | |
| Ectomis ceculus (Herrich-Schäffer, 1869) | |
| Ectomis octomaculata (Sepp [1844]) | |
| Epargyreus clavicornis (Herrich-Schäffer, 1869) | |
| Epargyreus exadeus exadeus (Cramer, 1779) | |
| Epargyreus socus socus Hübner [1825] | |
| Narcosius colossus granadensis (Möschler, 1879) | |
| Polygonus pallida (Röber, 1925) | |
| Polygonus savigny (Latreille [1824]) | |
| Proteides mercurius mercurius (Fabricius, 1787) | |
| Spathilepia clonius (Cramer, 1775) | |
| Spicauda procne (Plötz, 1880) | |
| Spicauda simplicius (Stoll, 1790) | |
| Spicauda teleus (Hübner, 1821) | |
| Telegonus alardus alardus (Stoll, 1790) | |
| Telegonus anaphus anaphus (Cramer, 1777) | |
| Telegonus chalco (Hübner, 1823) | |
| Telegonus creteus siges (Mabille, 1903) | |
| Telegonus elorus (Hewitson, 1867) | |
| Telegonus fulgerator fulgerator (Walch, 1775) | |
| Telegonus naxos (Hewitson, 1867) | |
| Telegonus talus (Cramer, 1777) | |
| Telemiades laogonus (Hewitson, 1876) | |
| Telemiades marpesus (Hewitson, 1876) | |
| Udranomia kikkawai (Weeks, 1906) | |
| Urbanus belli (Hayward, 1935) | |
| Urbanus esma Evans, 1952 | |
| Urbanus esmeraldus (Butler, 1877) | |
| Urbanus esta Evans, 1952 | |
| Urbanus pronta Evans, 1952 | |
| Urbanus proteus proteus (Linnaeus, 1758) | |
| Urbanus velinus (Plötz, 1880) | |
| Oileidini (5) | |
| Cogia calchas (Herrich-Schäffer, 1869) | |
| Cogia crameri (McHenry, 1960) | |
| Cogia undulatus (Hewitson, 1867) | |
| Marela tamyroides (C. Felder and R. Felder, 1867) | |
| Oechydrus evelinda (Butler, 1870) | |
| Phocidini (6) | |
| Dyscophellus doriscus (Hewitson, 1867) | |
| Nascus phocus (Cramer, 1777) | |
| Phocides charon (C. Felder and R. Felder, 1859) | |
| Phocides metrodorus metron Evans, 1952 | |
| Phocides pigmalion hewitsonius (Mabille, 1883) | |
| Phocides polybius phanias (Burmeister, 1880) | |
| Hesperiinae (158) | |
| Hesperiini (149) | |
| Anatrytone perfida (Möschler, 1879) | |
| Anthoptus epictetus (Fabricius, 1793) | |
| Anthoptus insignis (Plötz, 1882) | |
| Artines aquilina (Plötz, 1882) | |
| Artonia artona (Hewitson, 1868) | |
| Callimormus corades (C. Felder [1863]) | |
| Callimormus rivera (Plötz, 1882) | |
| Callimormus saturnus (Herrich-Schäffer, 1869) | |
| Calpodes ethlius (Stoll, 1782) | |
| Calpodes fusta Evans, 1955 | |
| Calpodes longirostris (Sepp [1840]) | |
| Calpodes mamurra (Plötz, 1886) | |
| Chitta chittara (Schaus, 1902) | |
| Cobalopsis nero (Herrich-Schäffer, 1869) | |
| Cobalopsis valerius (Möschler, 1879) | |
| Cobalus virbius hersilia (Plötz, 1882) | |
| Conga chydaea (Butler, 1877) | |
| Conga immaculata (Bell, 1930) | |
| Corticea corticea (Plötz, 1882) | |
| Corticea lysias potex Evans, 1955 | |
| Corticea mendica (Mabille, 1898) | |
| Corticea noctis (Plötz, 1882) | |
| Corticea obscura Mielke, 1979 | |
| Cumbre sp. | |
| Cymaenes alumna (Butler, 1877) | |
| Cymaenes gisca Evans, 1955 | |
| Cymaenes lepta (Hayward, 1939) | |
| Cymaenes loxa Evans, 1955 | |
| Cymaenes tripunctata tripunctata (Latreille [1824]) | |
| Cymaenes tripunctus theogenis (Capronnier 1874) | |
| Cynea cannae (Herrich-Schäffer, 1869) | |
| Cynea corisana (Plötz, 1882) | |
| Cynea irma (Möschler, 1879) | |
| Cynea sp. | |
| Dion uza (Hewitson, 1877) | |
| Dubia dubia (Bell, 1932) | |
| Duroca duroca (Plötz, 1882) | |
| Euphyes leptosema (Mabille, 1891) | |
| Eutocus vetulus matildae (Hayward, 1941) | |
| Eutocus vetulus vetulus (Mabille, 1883) | |
| Eutus mubevensis (Bell, 1932) | |
| Eutychide olympia (Plötz, 1882) | |
| Eutychide physcella (Hewitson, 1866) | |
| Flaccilla aecas (Stoll, 1781) | |
| Gallio garima massarus (Bell, 1940) | |
| Ginungagapus schmithi (Bell, 1930) | |
| Hedone catilina (Plötz, 1886) | |
| Hylephila phyleus phyleus (Drury, 1773) | |
| Justinia justinianus justinianus (Latreille [1824]) | |
| Justinia septa Evans, 1955 | |
| Koria kora (Hewitson, 1877) | |
| Lamponia elegantula (Herrich-Schäffer, 1869) | |
| Lamponia lamponia (Hewitson, 1876) | |
| Lento lento (Mabille, 1878) | |
| Lerema compta (Butler, 1877) | |
| Lerema geisa (Möschler, 1879) | |
| Lerema lenta (Evans, 1955) | |
| Lerodea erythrostictus (Prittwitz, 1868) | |
| Lerodea eufala eufala (W. H. Edwards, 1869) | |
| Lucida lucia (Capronnier, 1874) | |
| Ludens levina (Plötz, 1884) | |
| Lychnuchus celsus (Fabricius, 1793) | |
| Metrocles propertius (Fabricius, 1793) | |
| Metrocles santarus (Bell, 1940) | |
| Metron oropa (Hewitson, 1877) | |
| Mielkeus lucretius (Latreille [1824]) | |
| Mnasalcas ritans (Schaus, 1902) | |
| Mnaseas bicolor inca Bell, 1930 | |
| Mnaseas derasa derasa (Herrich-Schäffer, 1870) | |
| Mnasicles hicetaon Godman, 1901 | |
| Mnasicles remus (Fabricius, 1798) | |
| Mnasinous cinnamomea (Herrich-Schäffer, 1869) | |
| Mnasinous modestus (Plötz, 1882) | |
| Moeris striga (Geyer, 1832) | |
| Naevolus orius (Mabille, 1883) | |
| Nastra celeus vetus (Mielke, 1969) | |
| Niconiades merenda (Mabille, 1878) | |
| Niconiades xanthaphes Hübner [1821] | |
| Nyctelius nyctelius nyctelius (Latreille [1824]) | |
| Oligoria locutia (Hewitson, 1876) | |
| Oligoria lucifer (Hübner [1831]) | |
| Onophas columbaria distigma Bell, 1930 | |
| Orthos hyalinus (Bell, 1930) | |
| Oxynthes corusca (Herrich-Schäffer, 1869) | |
| Panoquina evadnes (Stoll, 1781) | |
| Panoquina fusina viola Evans, 1955 | |
| Panoquina hecebolus (Scudder, 1872) | |
| Panoquina lucas lucas (Fabricius, 1793) | |
| Panoquina lucas ocola (W. H. Edwards, 1863) | |
| Panoquina trix Evans, 1955 | |
| Papias allubita (Butler, 1870) | |
| Papias phainis Godman, 1900 | |
| Paracarystus hypargyra (Herrich-Schäffer, 1869) | |
| Paracarystus menestries menestries (Latreille [1824]) | |
| Phanes aletes (Geyer, 1832) | |
| Phanes rezia (Plötz, 1882) | |
| Phemiades anatolica (Plötz, 1883) | |
| Polites otho clavus (Erichson [1849]) | |
| Polites premnas (Wallengren 1860) | |
| Polites sapuca (Evans, 1955) | |
| Pompe postpuncta (Draudt, 1923) | |
| Pompeius amblyspila (Mabille, 1898) | |
| Pompeius pompeius (Latreille [1824]) | |
| Psoralis stacara (Schaus, 1902) | |
| Pyrrhopygopsis socrates socrates (Ménétriés, 1855) | |
| Quasimellana eulogius (Plötz, 1882) | |
| Quasimellana meridiani (Hayward, 1934) | |
| Saturnus conspicuus (E. Bell, 1941) | |
| Saturnus fartuga (Schaus, 1902) | |
| Sodalia argyrospila (Mabille, 1876) | |
| Sodalia coler (Schaus, 1902) | |
| Sodalia sodalis (Butler, 1877) | |
| Synapte malitiosa antistia (Plötz, 1882) | |
| Synapte silius (Latreille [1824]) | |
| Thargella caura occulta (Schaus, 1902) | |
| Thespieus aspernatus Draudt, 1923 | |
| Thespieus dalman (Latreille [1824]) | |
| Thespieus vividus (Mabille, 1891) | |
| Thespieus xarippe xarippe (Butler, 1870) | |
| Thoon sp. | |
| Thracides cleanthes cleanthes (Latreille [1824]) | |
| Tigasis arita (Schaus, 1902) | |
| Tigasis perloides (Plötz, 1882) | |
| Tigasis serra (Evans, 1955) | |
| Tirynthia conflua (Herrich-Schäffer, 1869) | |
| Tisias lesueur lesueur (Latreille [1824]) | |
| Tricrista advena advena (Draudt, 1923) | |
| Tricrista aethus (Hayward, 1951) | |
| Tricrista sp. | |
| Troyus diversa diversa (Herrich-Schäffer, 1869) | |
| Troyus fantasos (Cramer, 1780) | |
| Troyus phyllus (Fabricius, 1787) | |
| Vacerra bonfilius bonfilius (Latreille [1824]) | |
| Vacerra caniola elva Evans, 1955 | |
| Vacerra evansi Hayward, 1938 | |
| Vehilius inca (Scudder, 1872) | |
| Vehilius stictomenes stictomenes (Butler, 1877) | |
| Vertica verticalis verticalis (Plötz, 1882) | |
| Vettius lafrenaye lafrenaye (Latreille [1824]) | |
| Vidius sp. | |
| Vinius letis (Plötz, 1883) | |
| Vinius pulcherrimus Hayward, 1934 | |
| Vinius tryhana istria Evans, 1955 | |
| Virga austrinus (Hayward, 1934) | |
| Virga silvanus (Hayward, 1947) | |
| Xeniades chalestra chalestra (Hewitson, 1866) | |
| Xeniades orchamus (Cramer, 1777) | |
| Zariaspes mys (Hübner [1808]) | |
| Zenis jebus jebus (Plötz, 1882) | |
| Megathymini (1) | |
| Carystoides basoches (Latreille [1824]) | |
| Pericharini (7) | |
| Lycas devanes (Herrich-Schäffer, 1869) | |
| Orses cynisca (Swainson, 1821) | |
| Oz ozias ozias (Hewitson, 1878) | |
| Perichares aurina Evans, 1955 | |
| Perichares lotus (Butler, 1870) | |
| Perichares metallica (Riley, 1921) | |
| Perichares seneca seneca (Latreille [1824]) | |
| unknown (1) | |
| Hesperiinae sp. | |
| Heteropterinae (1) | |
| Heteropterini (1) | |
| Dardarina daridaeus (Godman, 1900) | |
| Pyrginae (78) | |
| Achlyodini (17) | |
| Achlyodes busirus rioja Evans, 1953 | |
| Aethilla echina coracina Butler, 1870 | |
| Eantis thraso (Hübner [1807]) | |
| Gindanes brebisson brebisson (Latreille [1824]) | |
| Grais stigmaticus (Mabille, 1883) | |
| Milanion clito (Fabricius, 1787) | |
| Milanion leucaspis (Mabille, 1878) | |
| Morvina fissimacula fissimacula (Mabille, 1878) | |
| Pythonides herennius lusorius Mabille, 1891 | |
| Pythonides jovianus fabricii Kirby, 1871 | |
| Pythonides limaea (Hewitson, 1868) | |
| Quadrus cerialis (Stoll, 1782) | |
| Quadrus fridericus riona (Evans, 1953) | |
| Quadrus hyacinthinus hyacinthinus (Mabille, 1877) | |
| Quadrus jacobus (Plötz, 1884) | |
| Quadrus u-lucida u-lucida (Plötz, 1884) | |
| Spioniades artemides (Stoll, 1782) | |
| Carcharodini (19) | |
| Bolla atahuallpai (Lindsey, 1925) | |
| Bolla catharina (Bell, 1937) | |
| Incisus incisus (Mabille, 1878) | |
| Nisoniades bipuncta (Schaus, 1902) | |
| Nisoniades brazia Evans, 1953 | |
| Nisoniades castolus (Hewitson, 1878) | |
| Nisoniades macarius (Herrich-Schäffer, 1870) | |
| Nisoniades maura (Mabille and Boullet, 1917) | |
| Pellicia costimacula costimacula Herrich-Schäffer, 1870 | |
| Pellicia sp. | |
| Pellicia theon Plötz, 1882 | |
| Perus minor (Schaus, 1902) | |
| Polyctor polyctor polyctor (Prittwitz, 1868) | |
| Sophista inops (Mabille, 1877) | |
| Staphylus ascalon (Staudinger, 1876) | |
| Staphylus melangon epicaste Mabille, 1903 | |
| Viola minor (Hayward, 1933) | |
| Viola violella (Mabille, 1898) | |
| Viuria herophile (Hayward, 1940) | |
| Erynnini (23) | |
| Anastrus sp. | |
| Anastrus ulpianus (Poey, 1832) | |
| Camptopleura auxo (Möschler, 1879) | |
| Camptopleura janthinus (Capronnier, 1874) | |
| Chiothion asychis autander (Mabille, 1891) | |
| Chiothion basigutta (Plötz, 1884) | |
| Cycloglypha caeruleonigra Mabille, 1903 | |
| Cycloglypha thrasibulus thrasibulus (Fabricius, 1793) | |
| Cycloglypha tisias (Godman and Salvin, 1896) | |
| Ebrietas anacreon anacreon (Staudinger, 1876) | |
| Ebrietas infanda (Butler, 1877) | |
| Echelatus sempiternus simplicior (Möschler, 1877) | |
| Festivia cronion (C. Felder and R. Felder, 1867) | |
| Gesta funeralis (Scudder and Burgess, 1870) | |
| Gesta gesta (Herrich-Schäffer, 1863) | |
| Gorgythion begga begga (Prittwitz, 1868) | |
| Gorgythion beggina escalaphoides (Hayward, 1941) | |
| Helias phalaenoides palpalis (Latreille [1824]) | |
| Hoodus jason (Ehrmann, 1907) | |
| Hoodus pelopidas (Fabricius, 1793) | |
| Mylon maimon (Fabricius, 1775) | |
| Sostrata bifasciata bifasciata (Ménétriés, 1829) | |
| Theagenes dichrous (Mabille, 1878) | |
| Pyrgini (19) | |
| Anisochoria pedaliodina extincta Hayward, 1933 | |
| Anisochoria sublimbata Mabille, 1883 | |
| Anisochoria superior Mabille, 1898 | |
| Antigonus erosus (Hübner [1812]) | |
| Antigonus nearchus (Latreille, 1817) | |
| Burnsius orcus (Stoll, 1780) | |
| Burnsius orcynoides (Giacomelli, 1928) | |
| Canesia pallida (Röber, 1925) | |
| Diaeus lacaena (Hewitson, 1869) | |
| Heliopetes alana (Reakirt, 1868) | |
| Heliopetes arsalte (Linnaeus, 1758) | |
| Heliopetes libra Evans, 1944 | |
| Heliopetes omrina (Butler, 1870) | |
| Heliopetes orbigera (Mabille, 1888) | |
| Heliopetes willi (Plötz, 1884) | |
| Paches liborius areta (Evans, 1953) | |
| Trina geometrina geometrina (C. Felder and R. Felder, 1867) | |
| Xenophanes tryxus (Stoll, 1780) | |
| Zopyrion evenor evenor Godman, 1901 | |
| Pyrrhopyginae (10) | |
| Pyrrhopygini (10) | |
| Microceris blanda Evans, 1951 | |
| Microceris intersecta rufitegula (Mielke, 1995) | |
| Microceris lamprus (Hopffer, 1874) | |
| Mimoniades versicolor (Latreille [1824]) | |
| Myscelus epigona Herrich-Schäffer, 1869 | |
| Mysoria barcastus barta Evans, 1951 | |
| Mysoria sejanus ssp. | |
| Parelbella ahira extrema (Röber, 1925) | |
| Passova passova practa Evans, 1951 | |
| Pyrrhopyge pelota Plötz, 1879 | |
| Tagiadinae (1) | |
| Celaenorrhinini (1) | |
| Celaenorrhinus similis Hayward, 1933 | |
| Lycaenidae (89) | |
| Polyommatinae (4) | |
| Polyommatini (4) | |
| Elkalyce cogina (Schaus, 1902) | |
| Hemiargus hanno hanno (Stoll, 1790) | |
| Leptotes cassius cassius (Cramer, 1775) | |
| Zizula cyna (W. H. Edwards, 1881) | |
| Theclinae (85) | |
| Theclini (85) | |
| Arawacus aetolus (Sulzer, 1776) | |
| Arawacus ellida (Hewitson, 1867) | |
| Arawacus meliboeus (Fabricius, 1793) | |
| Arawacus tadita (Hewitson, 1877) | |
| Arzecla arza (Hewitson, 1874) | |
| Arzecla nubilum (H. Druce, 1907) | |
| Atlides cosa (Hewitson, 1867) | |
| Atlides polybe (Linnaeus, 1763) | |
| Aubergina hesychia (Godman and Salvin, 1887) | |
| Aubergina vanessoides (Prittwitz, 1865) | |
| Badecla badaca (Hewitson, 1868) | |
| Brangas ca. neora | |
| Brangas silumena (Hewitson, 1867) | |
| Calycopis caulonia (Hewitson, 1877) | |
| Calycopis sp. | |
| Calycopis ca. xeneta | |
| Celmia celmus (Cramer, 1775) | |
| Celmia sp. | |
| Celmia uzza (Hewitson, 1873) | |
| Chalybs hassan (Stoll, 1790) | |
| Contrafacia imma (Prittwitz, 1865) | |
| Contrafacia muattina (Schaus, 1902) | |
| Cyanophrys acaste (Prittwitz, 1865) | |
| Cyanophrys herodotus (Fabricius, 1793) | |
| Dicya carnica (Hewitson, 1873) | |
| Electrostrymon endymion (Fabricius, 1775) | |
| Electrostrymon sp. | |
| Erora biblia (Hewitson, 1868) | |
| Erora sp. (opisena) (H. Druce, 1912) | |
| Evenus latreillii (Hewitson, 1865) | |
| Evenus regalis (Cramer, 1775) | |
| Gargina emessa (Hewitson, 1867) | |
| Iaspis ca. temesa | |
| Ignata brasiliensis (Talbot, 1928) | |
| Ignata norax (Godman and Salvin, 1887) | |
| Kolana ergina (Hewitson, 1867) | |
| Lamprospilus orcidia (Hewitson, 1874) | |
| Magnastigma hirsuta (Prittwitz, 1865) | |
| Michaelus ira (Hewitson, 1867) | |
| Michaelus jebus (Godart [1824]) | |
| Michaelus thordesa (Hewitson, 1867) | |
| Ministrymon azia (Hewitson, 1873) | |
| Ministrymon ca. cruenta | |
| Ministrymon cleon (Fabricius, 1775) | |
| Ministrymon cruenta (Gosse, 1880) | |
| Ocaria ocrisia (Hewitson, 1868) | |
| Ocaria thales (Fabricius, 1793) | |
| Oenomaus ortygnus (Cramer, 1779) | |
| Olynthus fancia (E. Jones, 1912) | |
| Ostrinotes ca. sophocles | |
| Ostrinotes empusa (Hewitson, 1867) | |
| Ostrinotes sophocles (Fabricius, 1793) | |
| Paiwarria venulius (Cramer, 1779) | |
| Panthiades hebraeus (Hewitson, 1867) | |
| Panthiades phaleros (Linnaeus, 1767) | |
| Parrhasius orgia (Hewitson, 1867) | |
| Parrhasius polibetes (Stoll, 1781) | |
| Pseudolycaena marsyas (Linnaeus, 1758) | |
| Rekoa malina (Hewitson, 1867) | |
| Rekoa marius (Lucas, 1857) | |
| Rekoa meton (Cramer, 1779) | |
| Rekoa palegon (Cramer, 1780) | |
| Rekoa stagira (Hewitson, 1867) | |
| Rubroserrata ecbatana (Hewitson, 1868) | |
| Siderus giapor (Schaus, 1902) | |
| Siderus philinna (Hewitson, 1868) | |
| Strephonota sphinx (Fabricius, 1775) | |
| Strephonota tephraeus (Geyer, 1837) | |
| Strymon astiocha (Prittwitz, 1865) | |
| Strymon bazochii bazochii (Godart [1824]) | |
| Strymon bubastus bubastus (Stoll, 1780) | |
| Strymon ca. bazochii | |
| Strymon cardus (Hewitson, 1874) | |
| Strymon cestri (Reakirt [1867]) | |
| Strymon eurytulus (Hübner [1819]) | |
| Strymon megarus (Godart [1824]) | |
| Strymon mulucha (Hewitson, 1867) | |
| Strymon yojoa (Reakirt [1867]) | |
| Strymon ziba (Hewitson, 1868) | |
| Symbiopsis lenitas (H. Druce, 1907) | |
| Theclopsis sp. | |
| Theritas hemon (Cramer, 1775) | |
| Theritas triquetra (Hewitson, 1865) | |
| Tmolus echion (Linnaeus, 1767) | |
| Ziegleria hesperitis (A. Butler and H. Druce, 1872) | |
| Nymphalidae (215) | |
| Apaturinae (5) | |
| Doxocopa agathina vacuna (Godart [1824]) | |
| Doxocopa kallina (Staudinger, 1886) | |
| Doxocopa laurentia laurentia (Godart [1824]) | |
| Doxocopa linda mileta (Boisduval, 1870) | |
| Doxocopa zunilda zunilda (Godart [1824]) | |
| Biblidinae (43) | |
| Ageroniini (9) | |
| Ectima thecla thecla (Fabricius, 1796) | |
| Hamadryas amphinome amphinome (Linnaeus, 1767) | |
| Hamadryas arete (E. Doubleday, 1847) | |
| Hamadryas epinome (C. Felder and R. Felder, 1867) | |
| Hamadryas februa februa (Hübner [1823]) | |
| Hamadryas feronia feronia (Linnaeus, 1758) | |
| Hamadryas iphthime iphthime (H. Bates, 1864) | |
| Hamadryas laodamia laodamia (Cramer, 1777) | |
| Pyrrhogyra neaerea ophni A. Butler, 1870 | |
| Biblidini (2) | |
| Biblis hyperia nectanabis (Fruhstorfer, 1909) | |
| Mestra hersilia hypermestra Hübner [1825] | |
| Callicorini (9) | |
| Callicore astarte selima (Guenée, 1872) | |
| Callicore hydaspes (Drury, 1782) | |
| Callicore sorana sorana (Godart [1824]) | |
| Diaethria candrena candrena (Godart [1824]) | |
| Diaethria clymena janeira (C. Felder, 1862) | |
| Diaethria eluina eluina (Hewitson [1855]) | |
| Haematera pyrame pyrame (Hübner [1819]) | |
| Paulogramma pygas thamyras Ménétriés, 1857 | |
| Paulogramma pyracmon pyracmon (Godart [1824]) | |
| Cybdelini (1) | |
| Cybdelis phaesyla (Hübner [1831]) | |
| Epicaliini (3) | |
| Myscelia orsis (Drury, 1782) | |
| Catonephele acontius acontius (Linnaeus, 1771) | |
| Catonephele numilia penthia (Hewitson, 1852) | |
| Eunicini (5) | |
| Eunica eburnea Fruhstorfer, 1907 | |
| Eunica maja maja (Fabricius, 1775) | |
| Eunica margarita (Godart [1824]) | |
| Eunica tatila bellaria Fruhstorfer, 1908 | |
| Eunica volumna volumna (Godart [1824]) | |
| Epiphilini (4) | |
| Epiphile hubneri Hewitson, 1861 | |
| Epiphile orea orea (Hübner [1823]) | |
| Nica flavilla flavilla (Godart [1824]) | |
| Temenis laothoe meridionalis Ebert, 1965 | |
| Eubagini (10) | |
| Dynamine aerata (A. Butler, 1877) | |
| Dynamine agacles agacles (Dalman, 1823) | |
| Dynamine artemisia artemisia (Fabricius, 1793) | |
| Dynamine athemon athemaena (Hübner [1824]) | |
| Dynamine coenus coenus (Fabricius, 1793) | |
| Dynamine meridionalis Röber, 1915 | |
| Dynamine myrrhina (E. Doubleday, 1849) | |
| Dynamine postverta postverta (Cramer, 1779) | |
| Dynamine sideria Rosa and Freitas, 2021 | |
| Dynamine tithia tithia (Hübner [1823]) | |
| Charaxinae (18) | |
| Anaeini (11) | |
| Consul fabius drurii (A. Butler, 1874) | |
| Fountainea glycerium cratais (Hewitson, 1874) | |
| Fountainea ryphea phidile (Geyer, 1837) | |
| Hypna clytemnestra huebneri A. Butler, 1866 | |
| Memphis acidalia victoria (H. Druce, 1877) | |
| Memphis appias (Hübner [1825]) | |
| Memphis arginussa arginussa (Geyer, 1832) | |
| Memphis moruus stheno (Prittwitz, 1865) | |
| Memphis otrere (Hübner [1825]) | |
| Memphis polyxo (H. Druce, 1874) | |
| Zaretis strigosus (Gmelin [1790]) | |
| Preponiini (7) | |
| Archaeoprepona amphimachus pseudomeander (Fruhstorfer, 1906) | |
| Archaeoprepona chalciope (Hübner [1823]) | |
| Archaeoprepona demophon thalpius (Hübner [1814]) | |
| Archaeoprepona demophoon antimache (Hübner [1819]) | |
| Prepona eugenes laertides Staudinger, 1898 | |
| Prepona laertes demodice (Godart [1824]) | |
| Prepona pylene Hewitson [1854] | |
| Cyrestinae (2) | |
| Cyrestini (2) | |
| Marpesia chiron marius (Cramer, 1779) | |
| Marpesia petreus petreus (Cramer, 1776) | |
| Danainae (33) | |
| Danaini (5) | |
| Danaus eresimus plexaure (Godart, 1819) | |
| Danaus erippus (Cramer, 1775) | |
| Danaus gilippus gilippus (Cramer, 1775) | |
| Lycorea halia discreta Haensch, 1909 | |
| Lycorea ilione ilione (Cramer, 1775) | |
| Ithomiini (28) | |
| Aeria olena olena Weymer, 1875 | |
| Brevioleria aelia plisthenes (R.F. d’Almeida, 1958) | |
| Brevioleria seba emyra (Haensch, 1905) | |
| Dircenna dero rhoeo x celtina C. Felder and R. Felder, 1860 | |
| Episcada hymenaea hymenaea (Prittwitz, 1865) | |
| Episcada philoclea (Hewitson [1855]) | |
| Episcada striposis Haensch, 1909 | |
| Episcada sylvo (Geyer, 1832) | |
| Epityches eupompe (Geyer, 1832) | |
| Heterosais edessa (Hewitson [1855]) | |
| Hypoleria adasa adasa (Hewitson [1855]) | |
| Hypoleria alema proxima Weymer, 1899 | |
| Hypothyris euclea laphria x nina | |
| Hypothyris ninonia daeta (Boisduval, 1836) | |
| Ithomia agnosia zikani R.F. d’Almeida, 1940 | |
| Ithomia drymo Hübner, 1816 | |
| Mcclungia cymo salonina (Hewitson, 1855) | |
| Mechanitis lysimnia lysimnia (Fabricius, 1793) | |
| Mechanitis polymnia casabranca Haensch, 1905 | |
| Melinaea ludovica paraiya Reakirt, 1866 | |
| Methona themisto (Hübner, 1818) | |
| Oleria aquata (Weymer, 1875) | |
| Placidina euryanassa (C. Felder and R. Felder, 1860) | |
| Pseudoscada acilla quadrifasciata Talbot, 1928 | |
| Pseudoscada erruca (Hewitson, 1855) | |
| Pteronymia carlia Schaus, 1902 | |
| Thyridia psidii hippodamia x pallida | |
| Tithorea harmonia pseudethra A. Butler, 1873 | |
| Heliconiinae (25) | |
| Acraeini (12) | |
| Actinote canutia (Hopffer, 1874) | |
| Actinote carycina Jordan, 1913 | |
| Actinote discrepans R.F. d’Almeida, 1958 | |
| Actinote genitrix genitrix R.F. d’Almeida, 1922 | |
| Actinote mamita mitama (Schaus, 1902) | |
| Actinote melanisans Oberthür, 1917 | |
| Actinote parapheles Jordan, 1913 | |
| Actinote pellenea pellenea Hübner [1821] | |
| Actinote pratensis Francini, Freitas and Penz, 2004 | |
| Actinote pyrrha pyrrha (Fabricius, 1775) | |
| Actinote rhodope R.F. d’Almeida, 1923 | |
| Actinote surima surima (Schaus, 1902) | |
| Heliconiini (13) | |
| Agraulis maculosa (Stichel [1908]) | |
| Dione juno juno (Cramer, 1779) | |
| Dione moneta moneta Hübner [1825] | |
| Dryadula phaetusa (Linnaeus, 1758) | |
| Dryas iulia alcionea (Cramer, 1779) | |
| Eueides aliphera aliphera (Godart, 1819) | |
| Eueides isabella dianasa (Hübner [1806]) | |
| Euptoieta hegesia meridiania Stichel, 1938 | |
| Heliconius besckei (Ménétriés, 1857) | |
| Heliconius erato phyllis (Fabricius, 1775) | |
| Heliconius ethilla narcaea x polychrous | |
| Heliconius sara apseudes (Hübner [1813]) | |
| Philaethria wernickei (Röber, 1906) | |
| Libytheinae (1) | |
| Libytheana carinenta carinenta (Cramer, 1777) | |
| Limenitidinae (17) | |
| Limenitidini (17) | |
| Adelpha abia (Hewitson, 1850) | |
| Adelpha calliphane Fruhstorfer, 1915 | |
| Adelpha capucinus velia (C. Felder and R. Felder, 1867) | |
| Adelpha cocala didia Fruhstorfer, 1915 | |
| Adelpha cytherea aea (C. Felder and R. Felder, 1867) | |
| Adelpha epizygis epizygis Fruhstorfer, 1915 | |
| Adelpha gavina Fruhstorfer, 1915 | |
| Adelpha iphiclus ephesa (Ménétriés, 1857) | |
| Adelpha lycorias lycorias (Godart [1824]) | |
| Adelpha malea goyama Schaus, 1902 | |
| Adelpha mythra (Godart [1824]) | |
| Adelpha plesaure plesaure Hübner, 1823 | |
| Adelpha poltius A. Hall, 1938 | |
| Adelpha serpa serpa (Boisduval, 1836) | |
| Adelpha syma (Godart [1824]) | |
| Adelpha thesprotia (C. Felder and R. Felder, 1867) | |
| Adelpha thessalia indefecta Fruhstorfer, 1913 | |
| Nymphalinae (20) | |
| Coeini (2) | |
| Historis acheronta acheronta (Fabricius, 1775) | |
| Historis odius dious Lamas, 1995 | |
| Junoniini (1) | |
| Junonia evarete evarete (Cramer, 1779) | |
| Melitaeini (7) | |
| Anthanassa hermas hermas (Hewitson, 1864) | |
| Chlosyne lacinia saundersi (E. Doubleday [1847]) | |
| Eresia lansdorfi (Godart, 1819) | |
| Ithra ithra (W. F. Kirby, 1900) | |
| Notilia velica velica (Hewitson, 1864) | |
| Tegosa claudina (Eschscholtz, 1821) | |
| Telenassa teletusa teletusa (Godart [1824]) | |
| Nymphalini (7) | |
| Colobura dirce dirce (Linnaeus, 1758) | |
| Hypanartia bella (Fabricius, 1793) | |
| Hypanartia lethe lethe (Fabricius, 1793) | |
| Smyrna blomfildia blomfildia (Fabricius, 1781) | |
| Vanessa braziliensis (Moore, 1883) | |
| Vanessa carye (Hübner [1812]) | |
| Vanessa myrinna (E. Doubleday, 1849) | |
| Victorinini (4) | |
| Anartia amathea roeselia (Eschscholtz, 1821) | |
| Anartia jatrophae jatrophae (Linnaeus, 1763) | |
| Siproeta epaphus trayja Hübner [1823] | |
| Siproeta stelenes meridionalis (Fruhstorfer, 1909) | |
| Satyrinae (50) | |
| Brassolini (13) | |
| Blepolenis batea didymaon (C. Felder and R. Felder, 1867) | |
| Brassolis sophorae (Linnaeus, 1758) | |
| Caligo brasiliensis brasiliensis (C. Felder, 1862) | |
| Caligo illioneus (Cramer, 1775) | |
| Catoblepia berecynthia unditaenia Fruhstorfer, 1907 | |
| Dynastor darius darius (Fabricius, 1775) | |
| Eryphanis automedon amphimedon (C. Felder and R. Felder, 1867) | |
| Eryphanis reevesii reevesii (E. Doubleday [1849]) | |
| Narope cyllastros E. Doubleday [1849] | |
| Opoptera syme (Hübner [1821]) | |
| Opsiphanes cassiae cassiae (Linnaeus, 1758) | |
| Opsiphanes invirae invirae (Hübner [1808]) | |
| Opsiphanes quiteria meridionalis Staudinger, 1887 | |
| Haeteriini (1) | |
| Pierella nereis (Drury, 1782) | |
| Morphini (5) | |
| Antirrhea archaea Hübner [1822] | |
| Morpho aega aega (Hübner [1822]) | |
| Morpho epistrophus catenaria Perry, 1811 | |
| Morpho helenor achillides C. Felder and R. Felder, 1867 | |
| Morpho menelaus coeruleus (Perry, 1810) | |
| Satyrini (31) | |
| Argentaria libitina (Butler, 1870) | |
| Argentaria sp. | |
| Capronnieria galesus (Godart [1824]) | |
| Carminda paeon (Godart [1824]) | |
| Cissia eous (A. Butler, 1867) | |
| Erichthodes julia (Weymer, 1911) | |
| Eteona tisiphone (Boisduval, 1836) | |
| Forsterinaria necys (Godart [1824]) | |
| Forsterinaria quantius (Godart [1824]) | |
| Godartiana muscosa (A. Butler, 1870) | |
| Hermeuptychia atalanta (A. Butler, 1867) | |
| Hermeuptychia sp. | |
| Malaveria affinis (Butler, 1867) | |
| Malaveria sp. | |
| Moneuptychia soter (Butler, 1877) | |
| Nhambikuara doxes (Godart [1824]) | |
| Pareuptychia ocirrhoe interjecta (d’Almeida, 1952) | |
| Paryphthimoides poltys poltys (Prittwitz, 1865) | |
| Pharneuptychia ca. innocentia | |
| Pharneuptychia sp.1 | |
| Praepedaliodes phanias (Hewitson, 1862) | |
| Taguaiba ypthima (Hübner [1821]) | |
| Taygetina kerea (Butler, 1869) | |
| Taygetis laches marginata Staudinger [1887] | |
| Taygetis tripunctata Weymer, 1907 | |
| Taygetis virgilia (Cramer, 1776) | |
| Yphthimoides celmis (Godart [1824]) | |
| Yphthimoides ochracea (Butler, 1867) | |
| Yphthimoides ordinaria Freitas, Kaminski and Mielke, 2012 | |
| Yphthimoides renata (Stoll, 1780) | |
| Yphthimoides yphthima (C. Felder and R. Felder, 1867) | |
| Papilionidae (17) | |
| Papilioninae (17) | |
| Leptocircini (3) | |
| Eurytides asius (Fabricius, 1781) | |
| Eurytides helios (Rothschild and Jordan, 1906) | |
| Eurytides protesilaus nigricornis (Staudinger, 1884) | |
| Papilionini (7) | |
| Heraclides anchisiades capys (Hübner [1809]) | |
| Heraclides astyalus astyalus (Godart, 1819) | |
| Heraclides hectorides (Esper, 1794) | |
| Heraclides thoas brasiliensis (Rothschild and Jordan, 1906) | |
| Heraclides torquatus polybius (Swainson, 1823) | |
| Pterourus cleotas (G. Gray, 1832) | |
| Pterourus scamander grayi (Boisduval, 1836) | |
| Troidini (7) | |
| Battus polydamas polydamas (Linnaeus, 1758) | |
| Battus polystictus polystictus (A. Butler, 1874) | |
| Parides agavus (Drury, 1782) | |
| Parides anchises nephalion (Godart, 1819) | |
| Parides bunichus bunichus (Hübner [1821]) | |
| Parides neophilus eurybates (Geyer, 1837) | |
| Parides proneus (Hübner [1831]) | |
| Pieridae (28) | |
| Coliadinae (17) | |
| Coliadini (8) | |
| Anteos clorinde (Godart [1824]) | |
| Anteos menippe (Hübner [1818]) | |
| Aphrissa statira statira (Cramer, 1777) | |
| Phoebis argante argante (Fabricius, 1775) | |
| Phoebis neocypris neocypris (Hübner [1823]) | |
| Phoebis philea philea (Linnaeus, 1763) | |
| Phoebis sennae marcellina (Cramer, 1777) | |
| Phoebis trite banksi (Breyer, 1939) | |
| Euremini (9) | |
| Eurema agave pallida (Chavannes, 1850) | |
| Eurema albula sinoe (Godart, 1819) | |
| Eurema arbela arbela (Geyer, 1832) | |
| Eurema deva deva (E. Doubleday, 1847) | |
| Eurema elvina (Godart, 1819) | |
| Eurema flavescens flavescens (Chavannes, 1850) | |
| Eurema leuce leuce (Boisduval, 1836) | |
| Eurema nise tenella (Boisduval, 1836) | |
| Eurema phiale paula (Röber, 1909) | |
| Dismorphiinae (6) | |
| Dismorphia amphione astynome (Dalman, 1823) | |
| Dismorphia astyocha (Hübner [1831]) | |
| Dismorphia thermesia thermesia (Godart, 1819) | |
| Enantia clarissa (Weymer, 1895) | |
| Enantia lina psamathe (Fabricius, 1793) | |
| Pseudopieris nehemia nehemia (Boisduval, 1836) | |
| Pierinae (5) | |
| Anthocharidini (1) | |
| Hesperocharis anguitia anguitia (Godart, 1819) | |
| Pierini (4) | |
| Archonias brassolis tereas (Godart, 1819) | |
| Ascia monuste orseis (Godart, 1819) | |
| Glutophrissa drusilla drusilla (Cramer, 1777) | |
| Melete lycimnia paulista Fruhstorfer, 1908 | |
| Riodinidae (43) | |
| Euselasiinae (3) | |
| Euselasiini (3) | |
| Erythia thucydides thucydides (Fabricius, 1793) | |
| Methone eucerus (Hewitson, 1872) | |
| Myselasia hygenius occulta (Stichel, 1919) | |
| Riodiniinae (40) | |
| Emesidini (7) | |
| Emesis diogenia Prittwitz, 1865 | |
| Emesis fastidiosa Ménétriés, 1855 | |
| Emesis fatimella fatimella Westwood, 1851 | |
| Emesis mandana (Cramer, 1780) | |
| Emesis ocypore zelotes Hewitson, 1872 | |
| Emesis russula Stichel, 1910 | |
| Emesis satema (Schaus, 1902) | |
| Eurybiini (2) | |
| Eurybia pergaea (Geyer, 1832) | |
| Leucochimona icare matatha (Hewitson, 1873) | |
| Nymphidiini (12) | |
| Ariconias glaphyra (Westwood, 1851) | |
| Aricoris signata (Stichel, 1910) | |
| Aricoris tutana (Godart [1824]) | |
| Catocyclotis malca (Schaus, 1902) | |
| Lemonias zygia zygia Hübner [1807] | |
| Nymphidium lisimon (Stoll, 1790) | |
| Synargis calyce (C. Felder and R. Felder, 1862) | |
| Synargis paulistina (Stichel, 1910) | |
| Theope lycaenina H. Bates, 1868 | |
| Theope nycteis (Westwood, 1851) | |
| Thisbe irenea (Stoll, 1780) | |
| Zabuella castanea (Prittwitz, 1865) | |
| Riodiniini (16) | |
| Baeotis johannae johannae Sharpe, 1890 | |
| Barbicornis basilis mona Westwood, 1851 | |
| Calephelis braziliensis McAlpine, 1971 | |
| Caria marsyas Godman, 1903 | |
| Caria plutargus plutargus (Fabricius, 1793) | |
| Chadia cadytis (Hewitson, 1866) | |
| Chalodeta theodora (C. Felder and R. Felder, 1862) | |
| Chamaelimnas briola doryphora Stichel, 1910 | |
| Dachetola azora (Godart [1824]) | |
| Detritivora zama (H. Bates, 1868) | |
| Lasaia agesilas (Latreille [1809]) | |
| Melanis electron auriferax (Stichel, 1910) | |
| Melanis smithiae smithiae (Westwood, 1851) | |
| Notheme erota hemicosmeta Seitz, 1917 | |
| Parcella amarynthina (C. Felder and R. Felder, 1865) | |
| Riodina lycisca lycisca (Hewitson [1853]) | |
| Symmachiini (3) | |
| Esthemopsis teras teras (Stichel, 1910) | |
| Mesene pyrippe sanguilenta Stichel, 1910 | |
| Pirascca sagaris satnius (Dalman, 1823) | |
References
- Myers, N.; Mittermeier, R.; Mittermeier, C.; Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Ribeiro, M.C.; Metzger, J.P.; Martensen, A.C.; Ponzoni, F.J.; Hirota, M.M. The Brazilian Atlantic Forest: How much is left, and how is the remaining forest distributed? Implications for conservation. Biol. Conserv. 2009, 142, 1141–1153. [Google Scholar] [CrossRef]
- Vancine, M.H.; Muylaert, R.L.; Niebuhr, B.B.; Oshima, J.E.F.; Tonetti, V.; Bernardo, R.; De Angelo, C.; Rosa, M.R.; Grohmann, C.H.; Ribeiro, M.C. The Atlantic Forest of South America: Spatiotemporal dynamics of vegetation and implications for conservation. Biol. Conserv. 2024, 291, 110499. [Google Scholar] [CrossRef]
- Harper, K.A.; MacDonald, S.E.; Burton, P.J.; Chen, J.; Brosofske, K.D.; Saunders, S.C.; Euskirchen, E.S.; Roberts, D.; Jaiteh, M.S.; Esseen, P. Edge Influence on Forest Structure and Composition in Fragmented Landscapes. Conserv. Biol. 2005, 19, 768–782. [Google Scholar] [CrossRef]
- Lourenço, G.M.; Soares, G.R.; Santos, T.P.; Dáttilo, W.; Freitas, A.V.L.; Ribeiro, S.P. Equal but different: Natural ecotones are dissimilar to anthropic edges. PLoS ONE 2019, 14, e0213008. [Google Scholar] [CrossRef]
- Galindo-Leal, C.; Camara, I.B. (Eds.) The Atlantic Forest of South America: Biodiversity Status, Threats, and Outlook; Center for Applied Biodiversity Science at Conservation International State of the Hotspots Series; Island Press: Washington, DC, USA, 2003; p. 488. [Google Scholar]
- Uehara-Prado, M.; Brown Jr., K.S.; Freitas, A.V.L. Species richness, composition and abundance of fruit-feeding butterflies in the Brazilian Atlantic Forest: Comparison between a fragmented and a continuous landscape. Glob. Ecol. Biogeogr. 2007, 16, 43–54. [Google Scholar] [CrossRef]
- Melo, D.H.A.; Filgueiras, B.K.C.; Iserhard, C.A.; Iannuzzi, L.; Freitas, A.V.L.; Leal, I.R. Effect of habitat loss and fragmentation on fruit-feeding butterflies in the Brazilian Atlantic Forest. Can. J. Zool. 2019, 97, 588–596. [Google Scholar] [CrossRef]
- Moris, R.J. Anthropogenic impacts on tropical forest biodiversity: A network structure and ecosystem functioning perspective. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 3709–3718. [Google Scholar] [CrossRef]
- Oliver, T.H.; Isaac, N.J.B.; August, T.A.; Woodcock, B.A.; Roy, D.B.; Bullock, J.M. Declining resilience of ecosystem functions under biodiversity loss. Nat. Commun. 2015, 6, 10122. [Google Scholar] [CrossRef] [PubMed]
- Hatfield, J.H.; Orme, C.D.L.; Banks-Leite, C. Using functional connectivity to predict potential meta-population sizes in the Brazilian Atlantic Forest. Perspect. Ecol. Conserv. 2018, 16, 215–220. [Google Scholar] [CrossRef]
- Amaya-Espinel, J.D.; Hostetler, M.E. The value of small forest fragments and urban tree canopy for Neotropical migrant birds during winter and migration seasons in Latin American countries: A systematic review. Landsc. Urban Plan. 2019, 190, 103592. [Google Scholar] [CrossRef]
- Brown Jr., K.S.; Freitas, A.V.L. Butterfly communities of urban forest fragments in Campinas, São Paulo, Brazil: Structure, instability, environmental correlates, and conservation. J. Insect Conserv. 2002, 6, 217–231. [Google Scholar] [CrossRef]
- Gathof, A.K.; Grossmann, A.J.; Herrmann, J.; Buchholz, S. Who can pass the urban filter? A multi-taxon approach to disentangle pollinator trait–environmental relationships. Oecologia 2022, 199, 165–179. [Google Scholar] [CrossRef]
- Collins, C.M.; Audusseau, H.; Hassall, C.; Keyghobadi, N.; Sinu, P.A.; Saunders, M.E. Insect ecology and conservation in urban areas: An overview of knowledge and needs. Insect Conserv. Divers. 2024, 17, 169–181. [Google Scholar] [CrossRef]
- Lewinsohn, T.M.; Agostini, K.; Freitas, A.V.L.; Melo, A.S. Insect decline in Brazil: An appraisal of current evidence. Biol. Lett. 2022, 18, 20220219. [Google Scholar] [CrossRef]
- Brown Jr., K.S.; Freitas, A.V.L. Atlantic Forest Butterflies: Indicators for Landscape Conservation. Biotropica 2000, 32, 934–956. [Google Scholar] [CrossRef]
- Bonebrake, T.; Ponisio, L.; Boggs, C.L.; Ehrlich, P.R. More than just indicators: A review of tropical butterfly ecology and conservation. Biol. Conserv. 2010, 143, 1831–1841. [Google Scholar] [CrossRef]
- Freitas, A.V.L.; Santos, J.P.; Rosa, A.H.B.; Iserhard, C.A.; Richter, A.; Siewert, R.R.; Gueratto, P.E.; Carreira, J.Y.O.; Lourenço, G.M. Sampling Methods for Butterflies (Lepidoptera). In Measuring Arthropod Biodiversity; Santos, J.C., Fernandes, G.W., Eds.; Springer: Cham, Switzerland, 2021; pp. 101–123. [Google Scholar]
- Morellato, L.P.C.; Leitão Filho, H.F. (Eds.) Ecologia e Preservação de Uma Floresta Tropical Urbana—Reserva de Santa Genebra, Campinas; Editora da Unicamp: Campinas, Brazil, 1995. [Google Scholar]
- Guarantini, M.T.G.; Gomes, E.P.C.; Tamashiro, J.Y.; Rodrigues, R.R. Composição florística da Reserva Municipal de Santa Genebra, Campinas, SP. Rev. Bras. Bot. 2008, 31, 323–337. [Google Scholar] [CrossRef]
- Morellato, L.P.C. As estações do ano na floresta. In Ecologia e preservação de Uma Floresta Tropical Urbana—Reserva de Santa Genebra; Leitão-Filho, H.F., Morellato, L.P.C., Eds.; Editora da Unicamp: Campinas, Brazil, 1995; pp. 37–41. [Google Scholar]
- Heikkilä, M.; Kaila, L.; Mutanen, M.; Peña, C.; Wahlberg, N. Cretaceous origin and repeated tertiary diversification of the redefined butterflies. Proc. R. Soc. B Biol. Sci. 2012, 279, 1093–1099. [Google Scholar] [CrossRef] [PubMed]
- Lamas, G. Checklist: Part 4A. Hesperioidea—Papilionoidea. In Atlas of Neotropical Lepidoptera; Heppner, J.B., Ed.; Scientific Publishers, Association for Tropical Lepidoptera: Gainsville, FL, USA, 2004; Volume 5A. [Google Scholar]
- Wahlberg, N.; Leneveu, J.; Kodandaramaiah, U.; Peña, C.; Nylin, S.; Freitas, A.V.L.; Brower, A.V.Z. Nymphalidae butterflies diversify following near demise at the cretaceous/tertiary boundary. Proc. R. Soc. B Biol. Sci. 2009, 276, 4295–4302. [Google Scholar] [CrossRef] [PubMed]
- Tyler, H.A.; Brown Jr., K.S.; Wilson, K.H. Swallowtail Butterflies of the Americas: A Study in Biological Dynamics, Ecological Diversity, Biosystematics, and Conservation; Scientific Publishers: Gainesville, FL, USA, 1994. [Google Scholar]
- Seraphim, N. Riodinidae Species Checklist: A Preliminary Species Checklist for the Riodinidae. 2025. Available online: https://labbor.ib.unicamp.br/?page_id=805 (accessed on 3 March 2025).
- Zhang, J.; Cong, Q.; Shen, J.; Opler, P.A.; Grishin, N.V. Genomics-guided refinement of butterfly taxonomy. Taxon. Rep. Int. Lepid. Surv. 2021, 9, 3. [Google Scholar]
- Robbins, R.K.; Lamas, G.; Mielke, O.H.H.; Harvey, D.J.; Casagrande, M.M. Taxonomic composition and ecological structure of the species-rich butterfly community at Pakitza, Parque Nacional del Manu, Perú. In Manu. The Biodiversity of Southeastern Peru. La Biodiversidad del Sureste del Perú; Wilson, D.E., Sandoval, A., Eds.; Editorial Horizonte: Lima, Peru, 1996; pp. 217–252. [Google Scholar]
- Zhang, J.; Cong, Q.; Shen, J.; Song, L.; Grishin, N.V. Taxonomic advances driven by the genomic analysis of butterflies. Taxon. Rep. Int. Lepid. Surv. 2024, 11, 90. [Google Scholar]
- Leong, J.V.; Matos-Maraví, P.; Núñez, R.; Nunes, R.; Liang, W.; Braby, M.F.; Doleck, T.; Aduse-Poku, K.; Inayoshi, Y.; Hsu, Y.-F.; et al. Around the World in 26 Million Years: Diversification and Biogeography of Pantropical Grass-Yellow Eurema Butterflies (Pieridae: Coliadinae). J. Biogeogr. 2025, 52, e15107. [Google Scholar] [CrossRef]
- Zacca, T.; Barbosa, E.P.; Freitas, A.V.L. Euptychiina Species Checklist. 2018. Available online: https://labbor.ib.unicamp.br/?page_id=1020 (accessed on 3 March 2025).
- Paluch, M. Revisão das Espécies de Actinote Hübner, [1819] (Lepidoptera, Nymphalidae, Heliconiinae, Acraeini). Ph.D. Thesis, Universidade Federal do Paraná, Curitiba, Brazil, 2006. [Google Scholar]
- Núñez, R.; Willmott, K.R.; Álvarez, Y.; Genaro, J.A.; Pérez-Asso, A.R.; Quejereta, M.; Turner, T.; Miller, J.Y.; Brévignon, C.; Lamas, G.; et al. Integrative taxonomy clarifies species limits in the hitherto monotypic passion-vine butterfly genera Agraulis and Dryas (Lepidoptera, Nymphalidae, Heliconiinae). Syst. Entomol. 2021, 47, 152–178. [Google Scholar] [CrossRef]
- Penz, C.M. Reinstated status of the butterfly genus Agraulis (Lepidoptera, Nymphalidae, Heliconiinae). Zootaxa 2022, 5209, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cong, Q.; Shen, J.; Brockmann, E.; Grishin, N.V. Three new subfamilies of skipper butterflies (Lepidoptera, Hesperiidae). Zookeys 2019, 861, 91–105. [Google Scholar] [CrossRef] [PubMed]
- Mielke, O.H.H. Catalogue of the American Hesperioidea: Hesperiidae (Lepidoptera); Sociedade Brasileira de Zoologia: Curitiba, Brazil, 2005. [Google Scholar]
- Cong, Q.; Zhang, J.; Shen, J.; Grishin, N.V. Fifty new genera of Hesperiidae (Lepidoptera). Insecta Mundi 2019, 2019, 0731. [Google Scholar]
- Li, W.; Cong, Q.; Shen, J.; Zhang, J.; Hallwachs, W.; Janzen, D.H.; Grishin, N.V. Genomes of skipper butterflies reveal extensive convergence of wing patterns. Proc. Natl. Acad. Sci. USA 2019, 116, 6232–6237. [Google Scholar] [CrossRef]
- Zhang, J.; Cong, Q.; Shen, J.; Brockmann, E.; Grishin, N.V. Genomes reveal drastic and recurrent phenotypic divergence in firetip skipper butterflies (Hesperiidae: Pyrrhopyginae). Proc. R. Soc. B Biol. Sci. 2019, 286, 20190609. [Google Scholar] [CrossRef]
- Zhang, J.; Cong, Q.; Shen, J.; Opler, P.A.; Grishin, N.V. Changes to North American butterfly names. Taxon. Rep. Int. Lepid. Surv. 2019, 8, 1. [Google Scholar]
- Zhang, J.; Cong, Q.; Shen, J.; Grishin, N.V. Taxonomic changes suggested by the genomic analysis of Hesperiidae (Lepidoptera). Insecta Mundi 2022, 921, 1409. [Google Scholar]
- Zhang, J.; Cong, Q.; Shen, J.; Song, L.; Gott, R.J.; Boyer, P.; Guppy, C.S.; Kohler, S.; Lamas, G.; Opler, P.A.; et al. Taxonomic discoveries enabled by genomic analysis of butterflies. Taxon. Rep. Int. Lepid. Surv. 2022, 10, 1. [Google Scholar]
- Zhang, J.; Cong, Q.; Burns, J.M.; Grishin, N.V. Checking the checkered taxonomy of Plötz’s checkered skippers (Hesperiidae: Pyrgini). Taxon. Rep. Int. Lepid. Surv. 2022, 10, 5. [Google Scholar]
- Zhang, J.; Cong, Q.; Song, L.; Shen, J.; Léger, T.; Lamas, G.; Mielke, O.H.H.; Grishin, N.V. Resolving inconsistencies between Plötz’s descriptions and presumed type specimens of some Hesperiidae (Lepidoptera). Mitteilungen aus dem Museum fur Naturkunde in Berlin. Deutsche Entomologische Zeitschrift 2023, 70, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Dolibaina, D.; Cong, Q.; Shen, J.; Song, L.; Mielke, C.G.C.; Casagrande, M.M.; Mielke, O.H.H.; Grishin, N.V. Taxonomic notes on Neotropical Hesperiidae (Lepidoptera). Zootaxa 2023, 5271, 91–114. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cong, Q.; Shen, J.; Song, L.; Opler, P.A.; Grishin, N.V. Additional taxonomic refinements suggested by genomic analysis of butterflies. Taxon. Rep. Int. Lepid. Surv. 2023, 11, 1. [Google Scholar]
- Brown Jr., K.S. Maximizing daily butterfly counts. J. Lepid. Soc. 1972, 26, 183–196. [Google Scholar]
- Iserhard, C.A.; Brown Jr., K.S.; Freitas, A.V.L. Maximized sampling of butterflies to detect temporal changes in tropical communities. J. Insect Conserv. 2013, 17, 615–622. [Google Scholar] [CrossRef]
- DeVries, P.J.; Walla, T.R. Species diversity and community structure in neotropical fruit-feeding butterflies. Biol. J. Linn. Soc. 2001, 74, 1–15. [Google Scholar] [CrossRef]
- Chao, A.; Chiu, C.H. Species richness: Estimation and comparison. Wiley StatsRef Stat. Ref. Online 2016, 1, 26. [Google Scholar]
- Colwell, R.K. EstimateS: Statistical Estimation of Species Richness and Shared Species from Samples, Version 9; User’s Guide and Application. 2013. Available online: http://www.robertkcolwell.org/pages/estimates (accessed on 30 May 2025).
- R Development Team. R: A Language and Environment for Statistical Computing, R Version 3.5.1; R Foundation for Statistical Computing: Austria, Vienna, 2018. [Google Scholar]
- Brown Jr., K.S. Geologic, Evolutionary, and Ecological Bases of the Diversification of Neotropical Butterflies. In Tropical Rainforests: Past, Present and Future; Bermingham, E., Dick, C.W., Moritz, C., Eds.; University of Chicago Press: Chicago, IL, USA, 2005; pp. 166–201. [Google Scholar]
- Beccaloni, G.W.; Gaston, K.J. Predicting the species richness of Neotropical forest butterflies: Ithomiinae (Lepidoptera: Nymphalidae) as indicators. Biol. Conserv. 1995, 71, 77–86. [Google Scholar] [CrossRef]
- Uehara-Prado, M.; Freitas, A.V.L. The effect of rainforest fragmentation on species diversity and mimicry ring composition of ithomiine butterflies. Insect Conserv. Divers. 2009, 2, 23–28. [Google Scholar] [CrossRef]
- Freitas, A.V.L.; Iserhard, C.A.; Santos, J.P.; Carreira, J.Y.; Ribeiro, D.B.; Melo, D.H.A.; Rosa, A.H.B.; Marini-Filho, O.J.; Accacio, G.M.; Uehara-Prado, M. Studies with butterfly bait traps: An overview. Rev. Colomb. Entomol. 2014, 40, 209–218. [Google Scholar]
- Wolda, H. Insect Seasonality: Why? Annu. Rev. Ecol. Syst. 1988, 19, 1–18. [Google Scholar] [CrossRef]
- Kishimoto-Yamada, K.; Itioka, T. How much have we learned about seasonality in tropical insect abundance since Wolda (1988)? Entomol. Sci. 2015, 18, 407–419. [Google Scholar] [CrossRef]
- Francini, R.B.; Duarte, M.; Mielke, O.H.H.; Caldas, A.; Freitas, A.V.L. Butterflies (Lepidoptera, Papilionoidea and Hesperioidea) of the “Baixada Santista” region, coastal São Paulo, Southeastern Brazil. Rev. Bras. Entomol. 2011, 55, 55–68. [Google Scholar] [CrossRef]
- Brown Jr., K.S. Borboletas da Serra do Japi: Diversidade, habitats, recursos alimentares e variação temporal. In História Natural da Serra do Japi: Ecologia e Preservação de uma Área Florestal no Sudeste do Brasil; Morellato, L.P.C., Ed.; Editora Unicamp/FAPESP: Campinas, Brazil, 1995; pp. 142–181. [Google Scholar]
- Ebert, H. On the frequency of butterflies in eastern Brazil, with a list of the butterfly fauna of Poços de Caldas, Minas Gerais. J. Lepid. Soc. 1969, 23, 1–48. [Google Scholar]
- Brown Jr., K.S.; Brown, G.G. Habitat alteration and species loss in Brazilian forests. In Tropical Deforestation and Species Extinction; Whitmore, T.C., Sayer, J., Eds.; Chapman and Hall: London, UK, 1992; pp. 119–142. [Google Scholar]
- Santos, J.P.; Carreira, J.Y.O.; Uehara-Prado, M.; Duarte, L.; Freitas, A.V.L. Outer bounds: Forest edges emulate vertical strata as a habitat filter for butterfly assemblages. Insect Conserv. Divers. 2025; online version. [Google Scholar]
- Ribeiro, D.B.; Prado, P.I.; Brown Jr., K.S.; Freitas, A.V.L. Temporal diversity patterns and phenology in fruit-feeding butterflies in the Atlantic Forest. Biotropica 2010, 42, 710–716. [Google Scholar] [CrossRef]
- Santos, J.P.; Iserhard, C.A.; Carreira, J.Y.O.; Freitas, A.V.L. Monitoring fruit-feeding butterfly assemblages in two vertical strata in seasonal Atlantic Forest: Temporal species turnover is lower in the canopy. J. Trop. Ecol. 2017, 33, 345–355. [Google Scholar] [CrossRef]
- Lourenço, G.M.; Luna, P.; Guevara, R.; Dáttilo, W.; Freitas, A.V.L.; Ribeiro, S.P. Temporal shifts in butterfly diversity: Responses to natural and anthropic forest transitions. J. Insect Conserv. 2020, 24, 353–363. [Google Scholar] [CrossRef]
- Freitas, A.V.L.; Gueratto, P.E.; Carreira, J.Y.O.; Lourenço, G.M.; Shirai, L.T.; Santos, J.P.; Rosa, A.H.B.; Evora, G.B.; Ramos, R.R.; Marín, M.A. Fruit-Feeding Butterfly Assemblages: Trends, Changes, and the Importance of Monitoring Schemes in Neotropical Environments. In Insect Decline and Conservation in the Neotropics; León-Cortés, J.L., Córdoba-Aguilar, A., Eds.; Springer: Cham, Switzerland, 2024; pp. 205–233. [Google Scholar]
- Pozo, C.; Luis-Martinez, A.; Llorente-Bousquets, J.; Salas-Suárez, N.; Maya-Martínez, A.; Vargas-Fernández, I.; Warren, A.D. Seasonality and Phenology of the Butterflies (Lepidoptera: Papilionoidea and Hesperioidea) of Mexico’s Calakmul Region. Fla. Entomol. 2008, 91, 407–422. [Google Scholar] [CrossRef]
- Kishimoto-Yamada, K.; Itioka, T. Seasonality in phytophagous scarabaeid (Melolonthinae and Rutelinae) abundances in an ‘aseasonal’ Bornean rainforest. Insect Conserv. Divers. 2013, 6, 179–188. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Wright, J.P.; Cadotte, M.W.; Carroll, I.T.; Hector, A.; Srivastava, D.S.; Loreau, M.; Weis, J.J. Impacts of plant diversity on biomass production increase through time because of species complementarity. Proc. Natl. Acad. Sci. USA 2007, 104, 18123–18128. [Google Scholar] [CrossRef] [PubMed]
- Caldas, A.; Robbins, R.K. Modified Pollard Transects for Assessing Tropical Butterfly Abundance and Diversity. Biol. Conserv. 2003, 110, 211–219. [Google Scholar] [CrossRef]
- McKinley, D.C.; Miller-Rushing, A.J.; Ballard, H.L.; Bonney, R.; Brown, H.; Cook-Patton, S.C.; Evans, D.M.; French, R.A.; Parris, J.K.; Phillips, T.B.; et al. Citizen science can improve conservation science, natural resource management, and environmental protection. Biol. Conserv. 2017, 208, 15–28. [Google Scholar] [CrossRef]
- Costa-Pereira, R.; Roque, F.O.; Constantino, P.A.L.; Sabino, J.; Uehara-Prado, M. Monitoramento In Situ da Biodiversidade: Proposta para um Sistema Brasileiro de Monitoramento da Biodiversidade; ICMBio: Brasília, Brazil, 2013; pp. 1–61.


| Common | Uncommon | Erratic | Visitors | Total | |
|---|---|---|---|---|---|
| All butterflies | 228 (35.6%) | 114 (17.8%) | 118 (18.4%) | 181 (28.2%) | 641 |
| Hesperiidae | 88 (30.7%) | 55 (19.2%) | 59 (20.6%) | 85 (29.6%) | 287 |
| Nymphalidae | 89 (45.9%) | 28 (14.4%) | 28 (14.4%) | 49 (25.3%) | 194 |
| Lycaenidae | 12 (15.6%) | 18 (23.4%) | 18 (23.4%) | 29 (37.7%) | 77 |
| Riodinidae | 10 (25.6%) | 9 (23.1%) | 6 (15.4%) | 14 (35.9%) | 39 |
| Pieridae | 20 (71.4%) | 2 (7.1%) | 5 (17.9%) | 1 (3.6%) | 28 |
| Papilionidae | 9 (56.3%) | 2 (12.5%) | 2 (12.5%) | 3 (18.8%) | 16 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carreira, J.Y.O.; Brown Jr., K.S.; Freitas, A.V.L. Species List and Temporal Trends of a Butterfly Community in an Urban Remnant in the Atlantic Forest. Diversity 2025, 17, 604. https://doi.org/10.3390/d17090604
Carreira JYO, Brown Jr. KS, Freitas AVL. Species List and Temporal Trends of a Butterfly Community in an Urban Remnant in the Atlantic Forest. Diversity. 2025; 17(9):604. https://doi.org/10.3390/d17090604
Chicago/Turabian StyleCarreira, Junia Y. O., Keith S. Brown Jr., and André V. L. Freitas. 2025. "Species List and Temporal Trends of a Butterfly Community in an Urban Remnant in the Atlantic Forest" Diversity 17, no. 9: 604. https://doi.org/10.3390/d17090604
APA StyleCarreira, J. Y. O., Brown Jr., K. S., & Freitas, A. V. L. (2025). Species List and Temporal Trends of a Butterfly Community in an Urban Remnant in the Atlantic Forest. Diversity, 17(9), 604. https://doi.org/10.3390/d17090604






