Suitability of Wheel Ruts: Characterization of Overlooked Breeding Habitats of Alpine Newts
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stuart, S.N.; Chanson, J.S.; Cox, N.A.; Young, B.E.; Rodrigues, A.S.L.; Fischman, D.L.; Waller, R.W. Status and trends of amphibian declines and extinctions worldwide. Science 2004, 306, 1783–1786. [Google Scholar] [CrossRef] [PubMed]
- Grant, E.H.C.; Miller, D.A.W.; Muths, E. A synthesis of evidence of drivers of amphibian declines. Herpetologica 2020, 76, 101–107. [Google Scholar] [CrossRef]
- Martel, A.; Blooi, M.; Adriaensen, C.; Van Rooij, P.; Beukema, W.; Fisher, M.C.; Farrer, R.A.; Schmidt, B.R.; Tobler, U.; Goka, K.; et al. Recent introduction of a chytrid fungus endangers Western Palearctic salamanders. Science 2014, 346, 630–631. [Google Scholar] [CrossRef] [PubMed]
- Arntzen, J.W.; Denoël, M.; Kuzmin, S.; Ishchenko, V.; Beja, P.; Andreone, F.; Jehle, R.; Nyström, P.; Miaud, C.; Anthony, B.; et al. Ichthyosaura alpestris. The IUCN Red List of Threatened Species. 2009. Available online: www.iucnredlist.org/species/59472/89702213 (accessed on 14 January 2023).
- Roček, Z.; Joly, P.; Grossenbacher, K. 2003: Bergmolch–Triturus alpestris (Laurenti, 1768). In Handbuch der Reptilien und Amphibian Europas; Böhme, W., Ed.; Aula Verlag: Wiebelsheim, Germany, 2003; pp. 607–656. [Google Scholar]
- Cirovic, R.; Radovi, D.; Vukov, T.D. Breeding site traits of European newts (Triturus macedonicus, Lissotriton vulgaris, and Mesotriton alpestris: Salamndridae) in the Montenegrin Karst region. Arch. Biol. Sci. Belgrade 2008, 60, 459–468. [Google Scholar] [CrossRef]
- Miaud, C. Identification of newt (Triturus alpestris and T. helveticus) eggs predators and protective role of oviposition behaviour. J. Zool. 1993, 231, 575–582. [Google Scholar] [CrossRef]
- Denoël, M. Effect of rival males on the courtship of paedomorphic and metamorphic Triturus alpestris (Amphibia: Salamandridae). Copeia 2003, 3, 618–623. [Google Scholar] [CrossRef]
- Denoël, M.; Mathieu, M.; Poncin, P. Effect of water temperature on the courtship behavior of the alpine newt Triturus alpestris. Behav. Ecol. Sociobiol. 2005, 58, 121–127. [Google Scholar] [CrossRef]
- Miaud, C.; Guyetant, R.; Faber, H. Age, size, and growth of the alpine newt, Triturus alpestris (Urodela: Salamandridae), at high altitude and a review of life-history trait variation throughout its range. Herpetologica 2000, 56, 135–144. [Google Scholar]
- Vogrin, M. Micro-habitat use within a guild of newt larvae (Trituridae) in an Alpine lake. Biol. Bratisl. 2006, 61, 579–584. [Google Scholar] [CrossRef]
- Denoël, M.; Schabetsberger, R.; Joly, P. Trophic specialisations in alternative heterochronic morphs. Naturwissenschaften 2004, 91, 81–84. [Google Scholar]
- Denoël, M. Amphibiens et Reptiles de Wallonie, Le Triton alpestre, Tri Tur Us Alpestris (Laurenti, 1768); Aves-Raînne & Région Wallonne: Namur, Belgium, 2007; pp. 62–71. [Google Scholar]
- Joger, U. Wassergefullte Wangenspuren Auf Forstwegen: Synokologische Untersuchungen an Einem Kurzlebigen Okosystem; Chimaira: Frankfurt am Main, Germany, 2000. [Google Scholar]
- Kopecký, O.; Vojar, J.; Denoël, M. Movements of Alpine newts (Mesotriton alpestris) between small aquatic habitats (ruts) during the breeding season. Amphibia-Reptilia 2010, 31, 109–116. [Google Scholar] [CrossRef]
- Kopecký, O.; Vojar, J.; Denoël, M. Sex-specific effect of pool desiccation on the movement of Alpine Newts, Mesotriton alpestris (Laurenti, 1768), among breeding sites. Herpetozoa 2012, 24, 127–134. [Google Scholar]
- Babik, W.; Rafinski, J. Amphibian breeding site characteristics in the Western Carpathians, Poland. Herpetol. J. 2001, 11, 41–51. [Google Scholar]
- Denoël, M.; Demars, B. The benefits of heterospecific oophagy in a top predator. Acta Oecol. 2008, 34, 74–79. [Google Scholar] [CrossRef]
- Semlitsch, R.D. Critical elements for biologically based recovery plans of aquatic-breeding Amphibians. Conserv. Biol. 2002, 16, 619–629. [Google Scholar] [CrossRef]
- Kopecký, O.; Vojar, J. Které vlastnosti vodních biotopů ovlivňují početnost čolků horských (Mesotriton alpestris) za nestálých podmínek prostředí. Sborník Sev. Muz. Lib. 2007, 25, 131–139. [Google Scholar]
- Crawley, M.J. The R Book; John Wiley and Sons: Chichester, UK, 2007. [Google Scholar]
- Pekár, S.; Brabec, M. Moderní Analýza Biologických Dat 1; Scientia: Brno, Czech Republic, 2009. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Baber, M.J.; Fleishman, E.; Babbitt, K.J.; Tarr, T.L. The relationship between wetland hydroperiod and nestedness patterns in assemblages of larval amphibians and predatory macroinvertebrates. Oikos 2004, 107, 16–27. [Google Scholar] [CrossRef]
- Semlitsch, R.D. Density-dependent growth and fecundity in the paedomorphic salamander Ambystoma talpoideum. Ecology 1987, 68, 1003–1008. [Google Scholar] [CrossRef]
- Denoël, M.; Lehmann, A. Multi-scale effect of landscape processes and habitat quality on newt abundance: Implications for conservation. Biol. Conserv. 2006, 130, 495–504. [Google Scholar] [CrossRef]
- Hartel, T.; Nemes, S.; Cogalniceanu, D.; Ollerer, K.; Schweiger, O.; Moga, C.-I.; Demeter, L. The effect of fish and aquatic habitat complexity on amphibians. Hydrobiologia 2007, 583, 173–178. [Google Scholar] [CrossRef]
- DeTroyer, N.; Forio, M.A.E.; Roels, K.; DeMeester, L.; Lemmens, P.; Declerck, S.A.J.; Martens, K.; Goethals, P. Key management rules for agricultural newt breeding ponds based on habitat suitability models. Glob. Ecol. Conserv. 2020, 23, e01086. [Google Scholar] [CrossRef]
- Wellborn, G.A.; Skelly, D.K.; Werner, E.E. Mechanisms creating community structure across a freshwater habitat gradient. Annu. Rev. Ecol. Syst. 1996, 27, 337–363. [Google Scholar] [CrossRef]
- D’Amen, M.; Vignoli, L.; Bologna, M.A. The effects of temperature and pH on the embryonic development of two species of Triturus (Caudata: Salamandridae). Amphib. -Reptil. 2007, 28, 295–300. [Google Scholar]
- Griffiths, R.A.; DeWijer, P.; Brady, L. The effect of pH on embryonic and larval development in smooth and palmate newts, Triturus vulgaris and Triturus helveticus. J. Zool. 1993, 230, 401–409. [Google Scholar] [CrossRef]
- Griffiths, R.A.; DeWijer, P. Differential effects of pH and temperature on embryonic development in the British newts (Triturus). J. Zool. 1994, 234, 613–622. [Google Scholar] [CrossRef]
- Strijbosch, H. Habitat selection of amphibians during their aquatic phase. Oikos 1979, 33, 363–372. [Google Scholar] [CrossRef]
- Pavignano, I.; Giacoma, C.; Castellano, S. A multivariate analysis of amphibian habitat determinants in north western Italy. Amphibia-Reptilia 1990, 11, 311–324. [Google Scholar] [CrossRef]
- Orizaola, G.; Braňa, F. Oviposition behaviour and vulnerability of eggs to predation in four newt species (genus Triturus). Herpetol. J. 2003, 13, 121–124. [Google Scholar]
- Marco, A.; Lizana, M.; Alvarez, A.; Blaustein, A.R. Egg-wrapping behaviour protects newt embryos from UV radiation. Anim. Behav. 2001, 61, 639–644. [Google Scholar] [CrossRef]
- Dvořák, J.; Gvoždík, L. Oviposition preferences in newts: Does temperature matter? Ethology 2009, 115, 533–539. [Google Scholar] [CrossRef]
- Hof, C.; Araújo, M.B.; Jetz, W.; Rahbek, C. Additive threats from pathogens, climate and land-use change for global amphibian diversity. Nature 2011, 480, 516–519. [Google Scholar] [CrossRef]

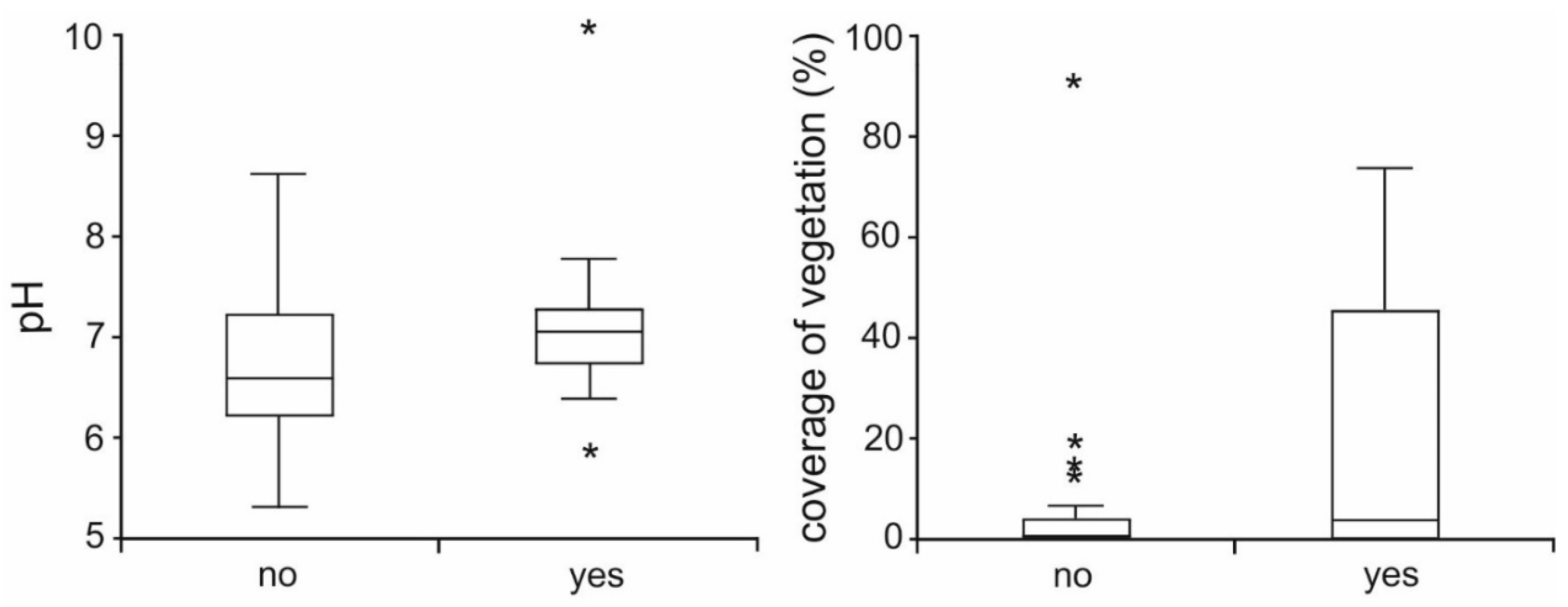
| df | F | p | |
|---|---|---|---|
| coverage of plants | 1 | 4.03 | 0.045 |
| pH | 1 | 4.75 | 0.029 |
| presence of prey | 1 | 6.76 | 0.009 |
| hydroperiod | 1 | 9.13 | 0.003 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopecký, O. Suitability of Wheel Ruts: Characterization of Overlooked Breeding Habitats of Alpine Newts. Diversity 2023, 15, 762. https://doi.org/10.3390/d15060762
Kopecký O. Suitability of Wheel Ruts: Characterization of Overlooked Breeding Habitats of Alpine Newts. Diversity. 2023; 15(6):762. https://doi.org/10.3390/d15060762
Chicago/Turabian StyleKopecký, Oldřich. 2023. "Suitability of Wheel Ruts: Characterization of Overlooked Breeding Habitats of Alpine Newts" Diversity 15, no. 6: 762. https://doi.org/10.3390/d15060762
APA StyleKopecký, O. (2023). Suitability of Wheel Ruts: Characterization of Overlooked Breeding Habitats of Alpine Newts. Diversity, 15(6), 762. https://doi.org/10.3390/d15060762





