Habitat Provision and Erosion Are Influenced by Seagrass Meadow Complexity: A Seascape Perspective
Abstract
1. Introduction
2. Materials and Methods
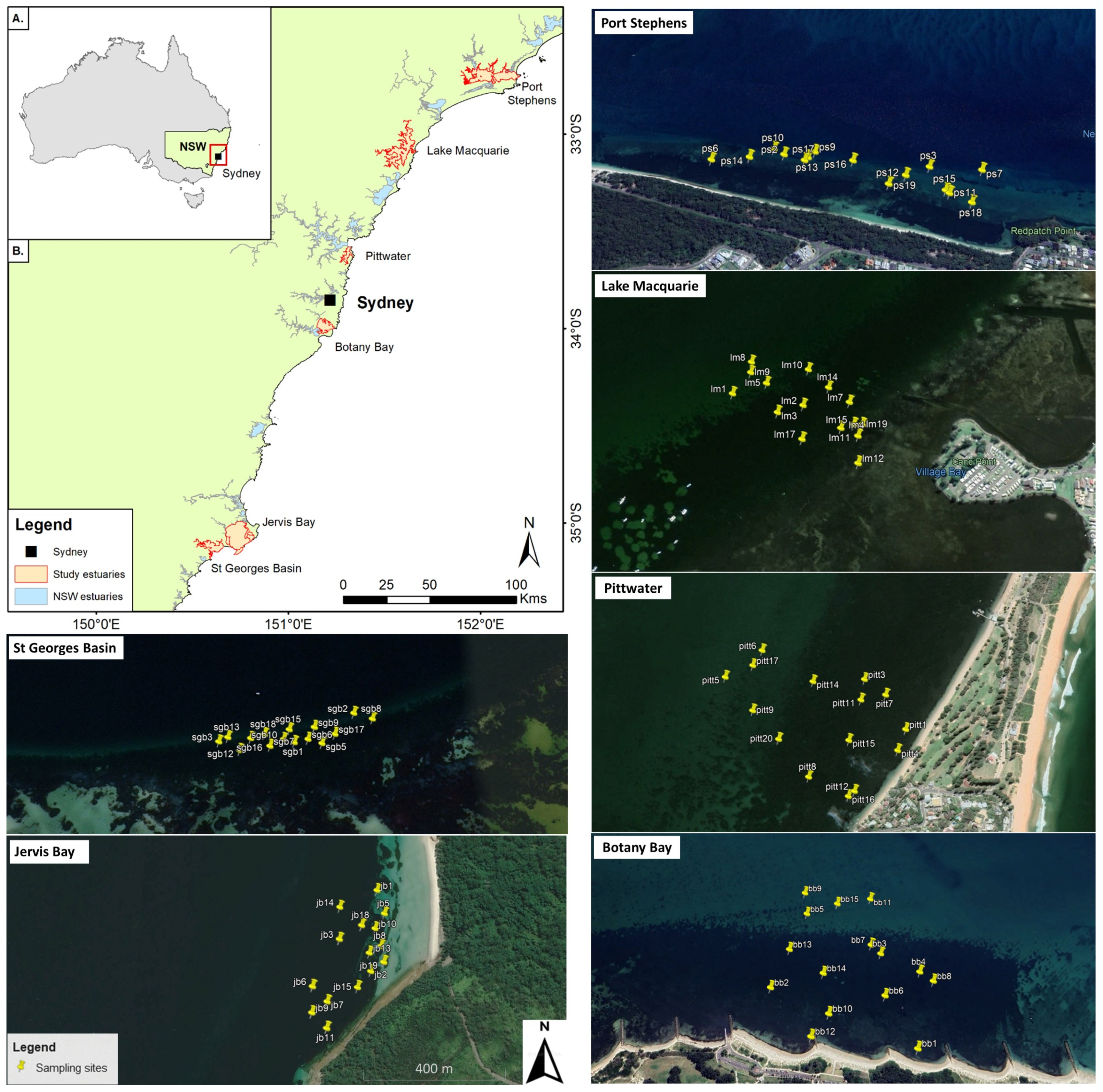
2.1. Study Estuaries and a-Priori Site Selection
2.2. Sampling
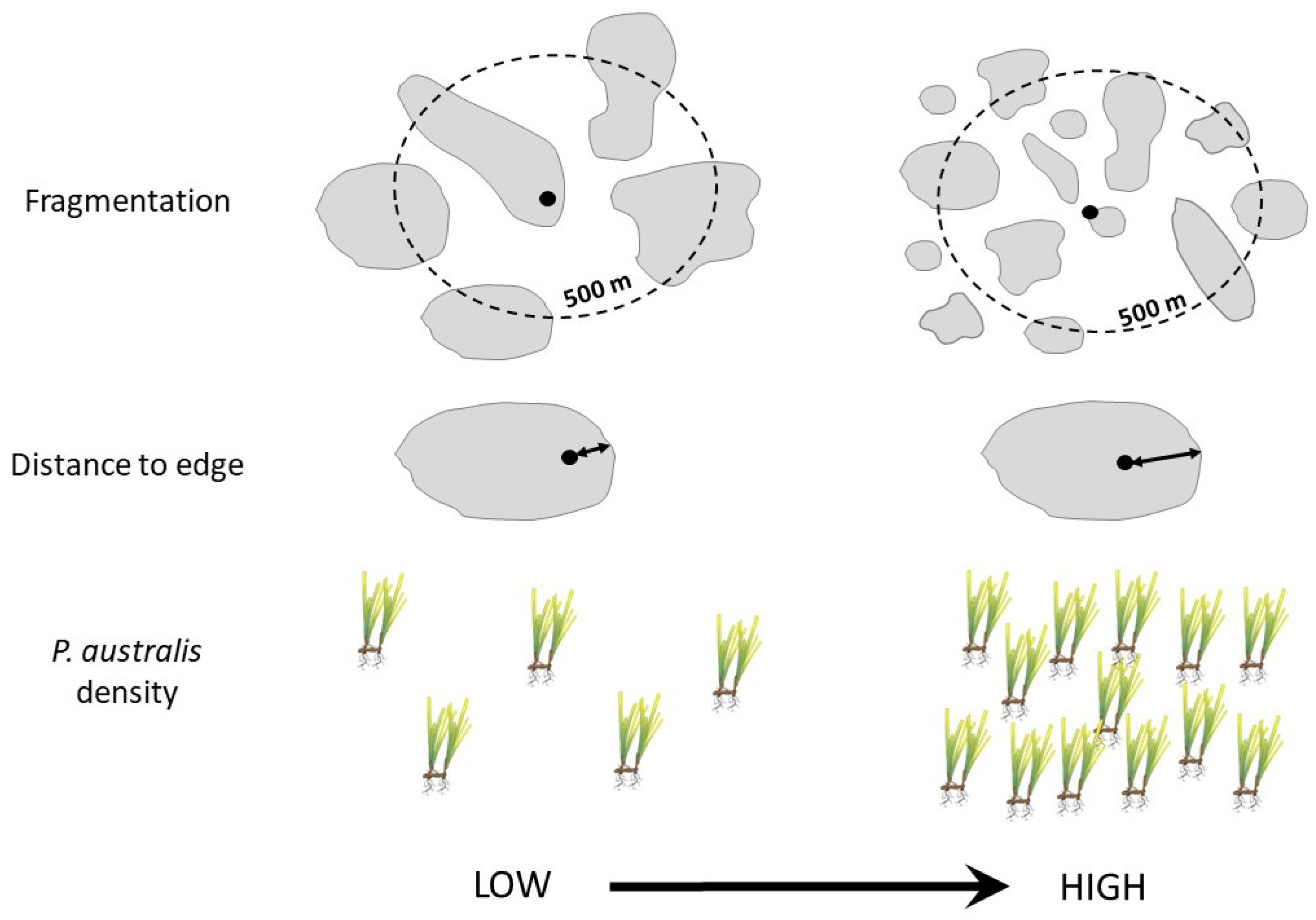
2.3. Variation in Abundance of Epifauna with Habitat Complexity
2.4. Variation in Fish Community with Habitat Complexity
2.5. Variation in Predation Rates with Habitat Complexity
2.6. Variation in Erosion with Habitat Complexity
2.7. Statistical Analysis
3. Results
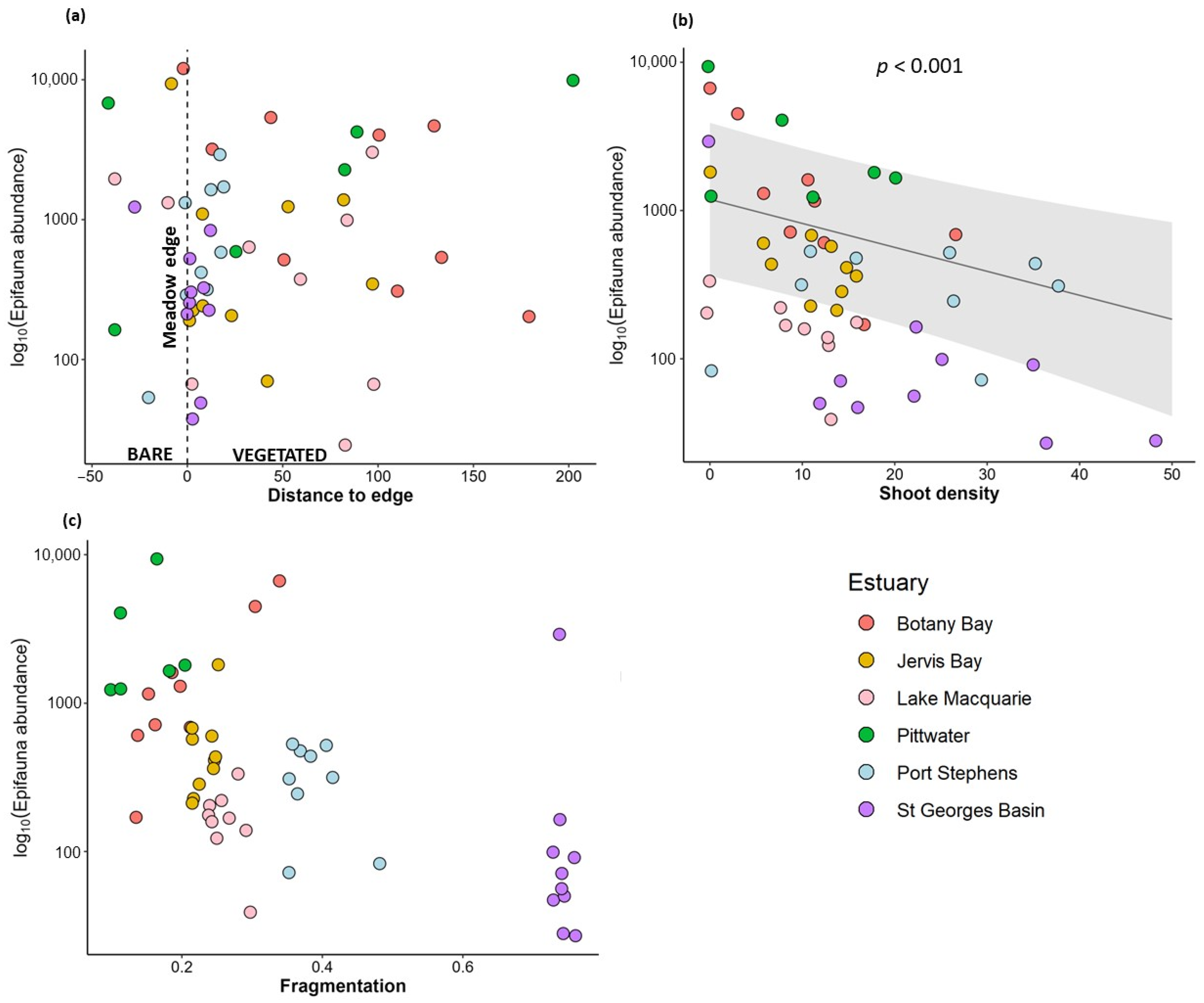
3.1. Variation in Abundance of Epifauna with Habitat Complexity
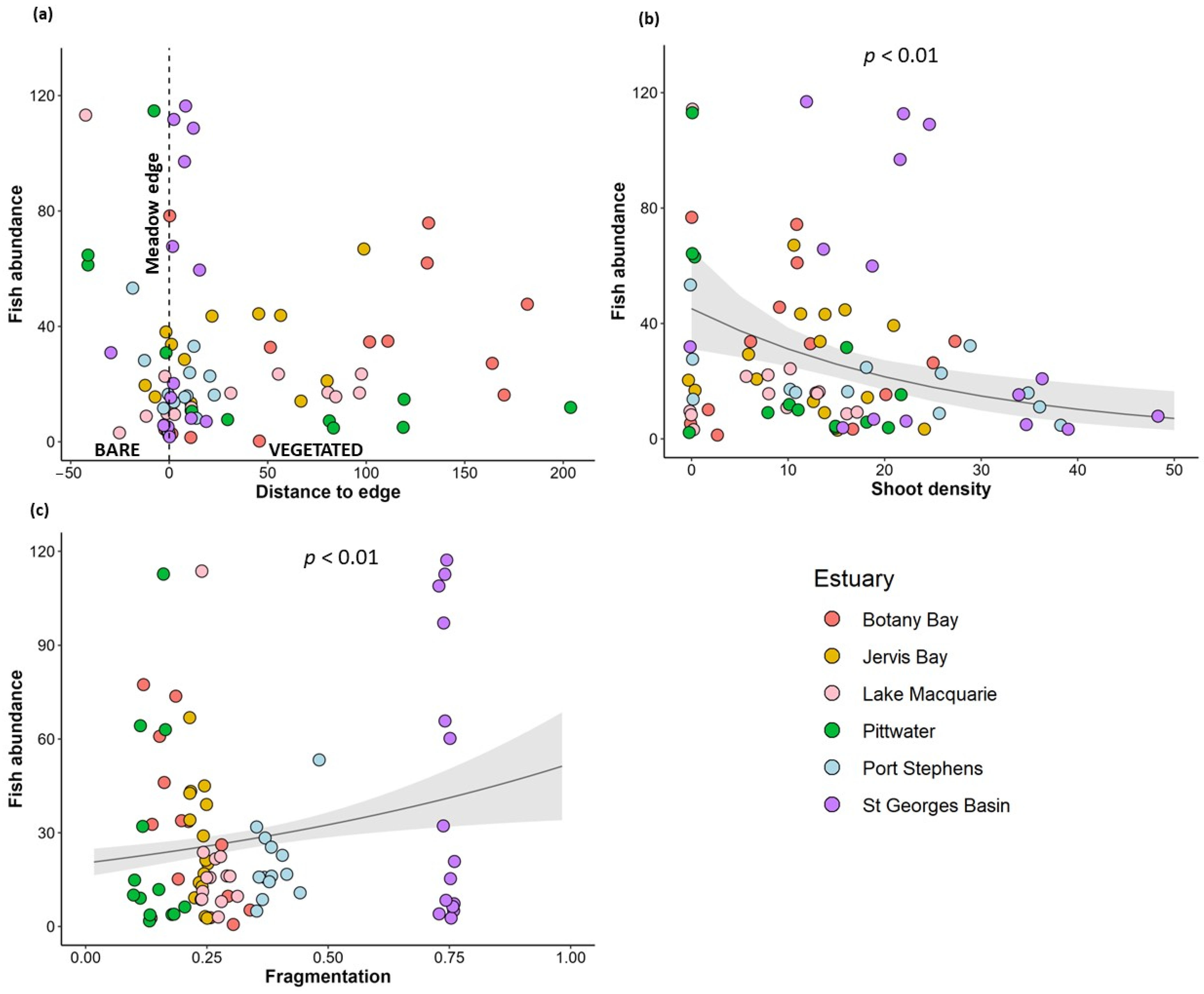
3.2. Variation in Fish Community with Habitat Complexity
3.3. Variation in Erosion with Habitat Complexity
3.4. Variation in Predation Rates with Habitat Complexity
4. Discussion
4.1. Habitat Use and Predation by Fish
4.2. Use of Seagrass Habitats by Invertebrate Epifauna
4.3. Variation in Erosion with Seagrass Density
4.4. Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kovalenko, K.E.; Thomaz, S.M.; Warfe, D.M. Habitat complexity: Approaches and future directions. Hydrobiologia 2012, 685, 1–17. [Google Scholar] [CrossRef]
- MacArthur, R.H.; MacArthur, J.W. On Bird Species Diversity. Ecology 1961, 42, 594–598. [Google Scholar] [CrossRef]
- Denno, R.F.; Finke, D.L.; Langellotto, G.A. Direct and indirect effects of vegetation structure and habitat complexity on predator-prey and predator-predator interactions. In Ecology of Predator-Prey Interactions; Oxford University: Oxford, UK, 2005; pp. 211–239. [Google Scholar]
- Warfe, D.M.; Barmuta, L. Habitat structural complexity mediates the foraging success of multiple predator species. Oecologia 2004, 141, 171–178. [Google Scholar] [CrossRef]
- Farina, S.; Arthur, R.; Pagès, J.; Prado, P.; Romero, J.; Verges, A.; Hyndes, G.; Heck, K.L.; Glenos, S.; Alcoverro, T. Differences in predator composition alter the direction of structure-mediated predation risk in macrophyte communities. Oikos 2014, 123, 1311–1322. [Google Scholar] [CrossRef]
- Boström, C.; Pittman, S.; Simenstad, C.; Kneib, R. Seascape ecology of coastal biogenic habitats: Advances, gaps, and challenges. Mar. Ecol. Prog. Ser. 2011, 427, 191–217. [Google Scholar] [CrossRef]
- Haddad, N.M.; Brudvig, L.A.; Clobert, J.; Davies, K.F.; Gonzalez, A.; Holt, R.D.; Lovejoy, T.E.; Sexton, J.O.; Austin, M.P.; Collins, C.D.; et al. Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci. Adv. 2015, 1, e1500052. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.C.; Chen, X.-Y.; Corlett, R.T.; Didham, R.K.; Ding, P.; Holt, R.D.; Holyoak, M.; Hu, G.; Hughes, A.C.; Jiang, L.; et al. Habitat fragmentation and biodiversity conservation: Key findings and future challenges. Landsc. Ecol. 2016, 33, 341–352. [Google Scholar]
- Bustamante, M.M.C.; Roitman, I.; Aide, T.M.; Alencar, A.; Anderson, L.O.; Aragão, L.; Asner, G.P.; Barlow, J.; Berenguer, E.; Chambers, J.; et al. Toward an integrated monitoring framework to assess the effects of tropical forest degradation and recovery on carbon stocks and biodiversity. Glob. Chang. Biol. 2016, 22, 92–109. [Google Scholar] [CrossRef] [PubMed]
- Fahrig, L. Ecological Responses to Habitat Fragmentation Per Se. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 1–23. [Google Scholar] [CrossRef]
- Halpern, B.S.; Walbridge, S.; Selkoe, K.A.; Kappel, C.V.; Micheli, F.; D’Agrosa, C.; Bruno, J.F.; Casey, K.S.; Ebert, C.; Fox, H.E.; et al. A global map of human impact on marine ecosystems. Science 2008, 319, 948–952. [Google Scholar] [CrossRef]
- Berger-Tal, O.; Saltz, D. Invisible barriers: Anthropogenic impacts on inter- and intra-specific interactions as drivers of landscape-independent fragmentation. Philos. Trans. R. Soc. B Biol. Sci. 2019, 374, 20180049. [Google Scholar] [CrossRef]
- Delarue, E.M.P.; Kerr, S.E.; Rymer, T.L. Habitat complexity, environmental change and personality: A tropical perspective. Behav. Process. 2015, 120, 101–110. [Google Scholar] [CrossRef]
- Hooper, D.U.; Adair, E.C.; Cardinale, B.J.; Byrnes, J.E.K.; Hungate, B.A.; Matulich, K.L.; Gonzalez, A.; Duffy, J.E.; Gamfeldt, L.; O’Connor, M.I. A global synthesis reveals biodiversity loss as a major driver of ecosystem change. Nature 2012, 486, 105–108. [Google Scholar] [CrossRef]
- Hooper, D.U.; Chapin, F.S., III; Ewel, J.J.; Hector, A.; Inchausti, P.; Lavorel, S.; Lawton, J.H.; Lodge, D.M.; Loreau, M.; Naeem, S.; et al. Effects of biodiversity on ecosystem functioning: A consensus of current knowledge. Ecol. Monogr. 2005, 75, 3–35. [Google Scholar] [CrossRef]
- Thompson, I.; Mackey, B.; McNulty, S.; Mosseler, A. Forest Resilience, Biodiversity, and Climate Change; Technical Series no. 43; Secretariat of the Convention on Biological Diversity: Montreal, QC, Canada, 2009; pp. 1–67. [Google Scholar]
- Duffy, J.E.; Godwin, C.M.; Cardinale, B.J. Biodiversity effects in the wild are common and as strong as key drivers of productivity. Nature 2017, 549, 261–264. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Duffy, J.E.; Gonzalez, A.; Hooper, D.U.; Perrings, C.; Venail, P.; Narwani, A.; Mace, G.M.; Tilman, D.; Wardle, D.A.; et al. Biodiversity loss and its impact on humanity. Nature 2012, 486, 59–67. [Google Scholar] [CrossRef]
- Sintayehu, D.W. Impact of climate change on biodiversity and associated key ecosystem services in Africa: A systematic review. Ecosyst. Health Sustain. 2018, 4, 225–239. [Google Scholar] [CrossRef]
- Ellison, A.M.; Bank, M.S.; Clinton, B.D.; Colburn, E.A.; Elliott, K.; Ford, C.R.; Foster, D.R.; Kloeppel, B.D.; Knoepp, J.D.; Lovett, G.M.; et al. Loss of foundation species: Consequences for the structure and dynamics of forested ecosystems. Front. Ecol. Environ. 2005, 3, 479–486. [Google Scholar] [CrossRef]
- Toenies, M.J.; Miller, D.A.W.; Marshall, M.R.; Stauffer, G.E. Shifts in vegetation and avian community structure following the decline of a foundational forest species, the eastern hemlock. Ornithol. Appl. 2018, 120, 489–506. [Google Scholar] [CrossRef]
- Wernberg, T.; Krumhansl, K.; Filbee-Dexter, K.; Pedersen, M.F. Status and trends for the world’s kelp forests. In World Seas: An Environmental Evaluation; Elsevier: Amsterdam, The Netherlands, 2019; pp. 57–78. [Google Scholar]
- Burgos, E.; Montefalcone, M.; Ferrari, M.; Paoli, C.; Vassallo, P.; Morri, C.; Bianchi, C.N. Ecosystem functions and economic wealth: Trajectories of change in seagrass meadows. J. Clean. Prod. 2017, 168, 1108–1119. [Google Scholar] [CrossRef]
- Dunic, J.C.; Brown, C.J.; Connolly, R.M.; Turschwell, M.P.; Côté, I.M. Long-term declines and recovery of meadow area across the world’s seagrass bioregions. Glob. Change Biol. 2021, 27, 4096–4109. [Google Scholar] [CrossRef] [PubMed]
- Duffy, J. Biodiversity and the functioning of seagrass ecosystems. Mar. Ecol. Prog. Ser. 2006, 311, 233–250. [Google Scholar] [CrossRef]
- Lavery, P.S.; Mateo, M.; Serrano, O.; Rozaimi, M. Variability in the Carbon Storage of Seagrass Habitats and Its Implications for Global Estimates of Blue Carbon Ecosystem Service. PLoS ONE 2013, 8, e73748. [Google Scholar] [CrossRef] [PubMed]
- Ricart, A.M.; York, P.H.; Bryant, C.V.; Rasheed, M.A.; Ierodiaconou, D.; Macreadie, P.I. High variability of Blue Carbon storage in seagrass meadows at the estuary scale. Sci. Rep. 2020, 10, 5865. [Google Scholar] [CrossRef] [PubMed]
- Jackson, E.L.; Rowden, A.A.; Attrill, M.J.; Bossey, S.J.; Jones, M.B. The importance of seagrass beds as a habitat for fishery species. Oceanogr. Mar. Biol. 2001, 39, 269–304. [Google Scholar]
- Gillanders, B.M. Seagrasses, fish, and fisheries. In Seagrasses: Biology, Ecology and Conservation; Springer: Berlin/Heidelberg, Germany, 2007; pp. 503–505. [Google Scholar]
- Potouroglou, M.; Bull, J.C.; Krauss, K.W.; Kennedy, H.A.; Fusi, M.; Daffonchio, D.; Mangora, M.M.; Githaiga, M.N.; Diele, K.; Huxham, M. Measuring the role of seagrasses in regulating sediment surface elevation. Sci. Rep. 2017, 7, 11917. [Google Scholar] [CrossRef]
- Gray, C.; McElligott, D.; Chick, R.; Chick, R. Intra- and inter-estuary differences in assemblages of fishes associated with shallow seagrass and bare sand. Mar. Freshw. Res. 1996, 47, 723–735. [Google Scholar] [CrossRef]
- Ferrell, D.; Bell, J. Differences among assemblages of fish associated with Zostera capricorni and bare sand over a large spatial scale. Mar. Ecol. Prog. Ser. 1991, 72, 15–24. [Google Scholar] [CrossRef]
- Jackson, E.L.; Rowden, A.A.; Attrill, M.J.; Bossy, S.F.; Jones, M.B. Comparison of fish and mobile macroinvertebrates associated with seagrass and adjacent sand at St. Catherine Bay, Jersey (English Channel): Emphasis on commercial species. Bull. Mar. Sci. 2002, 71, 1333–1341. [Google Scholar]
- Boström, C.; Jackson, E.L.; Simenstad, C.A. Seagrass landscapes and their effects on associated fauna: A review. Estuar. Coast. Shelf Sci. 2006, 68, 383–403. [Google Scholar] [CrossRef]
- Curtis, J.; Vincent, A. Distribution of sympatric seahorse species along a gradient of habitat complexity in a seagrass-dominated community. Mar. Ecol. Prog. Ser. 2005, 291, 81–91. [Google Scholar] [CrossRef]
- McCloskey, R.M.; Unsworth, R.K. Decreasing seagrass density negatively influences associated fauna. PeerJ 2015, 3, e1053. [Google Scholar] [CrossRef] [PubMed]
- Staveley, T.A.B.; Perry, D.; Lindborg, R.; Gullström, M. Seascape structure and complexity influence temperate seagrass fish assemblage composition. Ecography 2016, 40, 936–946. [Google Scholar] [CrossRef]
- Bologna, P.A.X.; Heck, K.L. Impact of habitat edges on density and secondary production of seagrass-associated fauna. Estuaries 2002, 25, 1033–1044. [Google Scholar] [CrossRef]
- Smith, T.; Hindell, J.S.; Jenkins, G.P.; Connolly, R.M. Seagrass patch size affects fish responses to edges. J. Anim. Ecol. 2009, 79, 275–281. [Google Scholar] [CrossRef]
- Tanner, J.E. Edge effects on fauna in fragmented seagrass meadows. Austral Ecol. 2005, 30, 210–218. [Google Scholar] [CrossRef]
- Gilby, B.; Olds, A.; Connolly, R.; Maxwell, P.; Henderson, C.; Schlacher, T. Seagrass meadows shape fish assemblages across estuarine seascapes. Mar. Ecol. Prog. Ser. 2018, 588, 179–189. [Google Scholar] [CrossRef]
- Ricart, A.M. Insights into Seascape Ecology: Landscape Patterns as Drivers in Coastal Marine Ecosystems. Doctoral Dissertation, Universitat de Barcelona, Barcelona, Spain, 2016. [Google Scholar]
- González-Ortiz, V.; Egea, L.G.; Ramos, R.J.; Moreno-Marín, F.; Perez-Llorens, J.L.; Bouma, T.J.; Brun, F.G. Interactions between Seagrass Complexity, Hydrodynamic Flow and Biomixing Alter Food Availability for Associated Filter-Feeding Organisms. PLoS ONE 2014, 9, e104949. [Google Scholar] [CrossRef]
- Folkard, A.M. Hydrodynamics of model Posidonia oceanica patches in shallow water. Limnol. Oceanogr. 2005, 50, 1592–1600. [Google Scholar] [CrossRef]
- Middleton, M.; Bell, J.; Burchmore, J.; Pollard, D.; Pease, B. Structural differences in the fish communities of Zostera capricorni and Posidonia australis seagrass meadows in Botany Bay, New South Wales. Aquat. Bot. 1984, 18, 89–109. [Google Scholar] [CrossRef]
- Burchmore, J.; Pollard, D.; Bell, J. Community structure and trophic relationships of the fish fauna of an estuarine Posidonia Australis seagrass habitat in port hacking, new South Wales. Aquat. Bot. 1984, 18, 71–87. [Google Scholar] [CrossRef]
- Bell, J.D.; Westoby, M. Variation in seagrass height and density over a wide spatial scale: Effects on common fish and decapods. J. Exp. Mar. Biol. Ecol. 1986, 104, 275–295. [Google Scholar] [CrossRef]
- Bell, J.D.; Westoby, M. Abundance of macrofauna in dense seagrass is due to habitat preference, not predation. Oecologia 1986, 68, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Evans, S.M.; Griffin, K.J.; Blick, R.A.J.; Poore, A.; Vergés, A. Seagrass on the brink: Decline of threatened seagrass Posidonia australis continues following protection. PLoS ONE 2018, 13, e0190370. [Google Scholar] [CrossRef] [PubMed]
- West, G.J.; Glasby, T.M. Interpreting Long-Term Patterns of Seagrasses Abundance: How Seagrass Variability Is Dependent on Genus and Estuary Type. Estuaries Coasts 2022, 45, 1393–1408. [Google Scholar] [CrossRef]
- Glasby, T.M.; West, G. Dragging the chain: Quantifying continued losses of seagrasses from boat moorings. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 28, 383–394. [Google Scholar] [CrossRef]
- EPBC Act. Environment Protection and Biodiversity Conservation Act 1999 (EPBC Act) (s266B). Approved Conservation Advice (Including Listing Advice) for Posidonia Australis Seagrass Meadows of the Manning-Hawkesbury Ecoregion Ecological Community; Office of Legislative Drafting and Publishing: Canberra, Australia, 2015. [Google Scholar]
- Meehan, A.J.; West, R.J. Recovery times for a damaged Posidonia australis bed in south eastern Australia. Aquat. Bot. 2000, 67, 161–167. [Google Scholar] [CrossRef]
- Ferretto, G.; Glasby, T.M.; Poore, A.G.; Callaghan, C.T.; Housefield, G.P.; Langley, M.; Sinclair, E.A.; Statton, J.; Kendrick, G.A.; Vergés, A. Naturally-detached fragments of the endangered seagrass Posidonia australis collected by citizen scientists can be used to successfully restore fragmented meadows. Biol. Conserv. 2021, 262, 109308. [Google Scholar] [CrossRef]
- Waltham, N.J.; Elliott, M.; Lee, S.Y.; Lovelock, C.; Duarte, C.M.; Buelow, C.; Simenstad, C.; Nagelkerken, I.; Claassens, L.; Wen, C.K.-C.; et al. UN Decade on Ecosystem Restoration 2021–2030—What chance for success in restoring coastal ecosystems? Front. Mar. Sci. 2020, 7, 71. [Google Scholar] [CrossRef]
- Jackson, E.L.; Attrill, M.J.; Jones, M.B. Habitat characteristics and spatial arrangement affecting the diversity of fish and decapod assemblages of seagrass (Zostera marina) beds around the coast of Jersey (English Channel). Estuar. Coast. Shelf Sci. 2006, 68, 421–432. [Google Scholar] [CrossRef]
- Boström, C.; Bonsdorff, E. Zoobenthic community establishment and habitat complexity the importance of seagrass shoot-density, morphology and physical disturbance for faunal recruitment. Mar. Ecol. Prog. Ser. 2000, 205, 123–138. [Google Scholar] [CrossRef]
- Swadling, D.S.; Knott, N.A.; Rees, M.J.; Davis, A.R. Temperate zone coastal seascapes: Seascape patterning and adjacent seagrass habitat shape the distribution of rocky reef fish assemblages. Landsc. Ecol. 2019, 34, 2337–2352. [Google Scholar] [CrossRef]
- Pittman, S.J. Seascape Ecology; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Pittman, S.; Yates, K.; Bouchet, P.; Alvarez-Berastegui, D.; Andréfouët, S.; Bell, S.; Berkström, C.; Boström, C.; Brown, C.; Connolly, R.; et al. Seascape ecology: Identifying research priorities for an emerging ocean sustainability science. Mar. Ecol. Prog. Ser. 2021, 663, 1–29. [Google Scholar] [CrossRef]
- Roy, P.; Williams, R.; Jones, A.; Yassini, I.; Gibbs, P.; Coates, B.; West, R.; Scanes, P.; Hudson, J.; Nichol, S. Structure and Function of South-east Australian Estuaries. Estuar. Coast. Shelf Sci. 2001, 53, 351–384. [Google Scholar] [CrossRef]
- NSW Department of Primary Industries. Fisheries NSW Spatial Data Portal. Available online: https://webmap.industry.nsw.gov.au/Html5Viewer/index.html?viewer=Fisheries_Data_Portal (accessed on 11 February 2019).
- Stevens, D.L., Jr.; Olsen, A.R. Spatially balanced sampling of natural resources. J. Am. Stat. Assoc. 2004, 99, 262–278. [Google Scholar] [CrossRef]
- Rees, M.; Knott, N.A.; Hing, M.L.; Hammond, M.; Williams, J.; Neilson, J.; Swadling, D.S.; Jordan, A. Habitat and humans predict the distribution of juvenile and adult snapper (Sparidae: Chrysophrys auratus) along Australia’s most populated coastline. Estuar. Coast. Shelf Sci. 2021, 257, 107397. [Google Scholar] [CrossRef]
- Sleeman, J.C.; Kendrick, G.; Boggs, G.; Hegge, B. Measuring fragmentation of seagrass landscapes: Which indices are most appropriate for detecting change? Mar. Freshw. Res. 2005, 56, 851–864. [Google Scholar] [CrossRef]
- Santos, R.O.; Lirman, D.; Pittman, S.J. Long-term spatial dynamics in vegetated seascapes: Fragmentation and habitat loss in a human-impacted subtropical lagoon. Mar. Ecol. 2015, 37, 200–214. [Google Scholar] [CrossRef]
- Bivand, R.; Keitt, T.; Rowlingson, B.; Pebesma, E.; Sumner, M.; Hijmans, R.; Rouault, E.l. Package ‘Rgdal’. Bindings for the Geospatial Data Abstraction Library 2015. Available online: https://CRAN.R-project.org/package=rgdal (accessed on 11 February 2019).
- Hijmans, R.J.; van Etten, J.; Sumner, M.; Cheng, J.; Baston, D.; Bevan, A.; Bivand, R.; Busetto, L.; Canty, M.; Fasoli, B.; et al. Package ‘raster’: Geographic Data Analysis and Modeling. 2015. Available online: https://CRAN.R-project.org/package=raster (accessed on 11 February 2019).
- Hijmans, R.J.; Karney, C.; Williams, E.; Vennes, C.; Hijmans, R.J. Package ‘geosphere’: Spherical Trigonometry 2017. Available online: https://CRAN.R-project.org/package=geosphere (accessed on 11 February 2019).
- Martin-Smith, K.M. Abundance of mobile epifauna: The role of habitat complexity and predation by fishes. J. Exp. Mar. Biol. Ecol. 1993, 174, 243–260. [Google Scholar] [CrossRef]
- Cappo, M.; Harvey, E.; Malcolm, H.; Speare, P. Potential of Video Techniques to Monitor Diversity, Abundance and Size of Fish in Studies of Marine Protected Areas; Aquatic Protected Areas-what works best and how do we know; University of Queensland: Brisbane, Australia, 2003; pp. 455–464. [Google Scholar]
- Willis, T.J.; Millar, R.B.; Babcock, R.C. Detection of spatial variability in relative density of fishes: Comparison of visual census, angling, and baited underwater video. Mar. Ecol. Prog. Serles 2000, 198, 249–260. [Google Scholar] [CrossRef]
- Froese, R.; Pauly, D. FishBase 2000: Concepts, Design and Data Sources; ICLARM: Los Baños, Laguna, Philippines, 2000; 344p. [Google Scholar]
- Bray, D.J.; Gomon, M.F.; Fishes of Australia. Museums Victoria and OzFishNet. Available online: https://fishesofaustralia.net.au/ (accessed on 28 April 2022).
- McGrouther, M. Australian Museum Website. Available online: https://australian.museum/learn/animals/fishes/ (accessed on 28 April 2022).
- Duffy, J.E.; Ziegler, S.; Campbell, J.E.; Bippus, P.M.; Lefcheck, J. Squidpops: A Simple Tool to Crowdsource a Global Map of Marine Predation Intensity. PLoS ONE 2015, 10, e0142994. [Google Scholar] [CrossRef] [PubMed]
- Vila-Concejo, A.; Harris, D.L.; Power, H.E.; Shannon, A.M.; Webster, J.M. Sediment transport and mixing depth on a coral reef sand apron. Geomorphology 2014, 222, 143–150. [Google Scholar] [CrossRef]
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Maechler, M.; Bolker, B.M. GlmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. The R Journal 2017, 9, 378–400. [Google Scholar] [CrossRef]
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; D’Agostino McGowan, L.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Heck, K.L., Jr.; Orth, R.J. Seagrass habitats: The roles of habitat complexity, competition and predation in structuring associated fish and motile macroinvertebrate assemblages. In Estuarine Perspectives; Elsevier: Amsterdam, The Netherlands, 1980; pp. 449–464. [Google Scholar]
- Worthington, D.; Ferrell, D.J.; McNeill, S.E.; Bell, J.D. Effects of the shoot density of seagrass on fish and decapods: Are correlation evident over larger spatial scales? Mar. Biol. 1992, 112, 139–146. [Google Scholar] [CrossRef]
- Bell, J.D.; Westoby, M. Importance of local changes in leaf height and density to fish and decapods associated with seagrasses. J. Exp. Mar. Biol. Ecol. 1986, 104, 249–274. [Google Scholar] [CrossRef]
- Kiggins, R.S.; Knott, N.A.; Davis, A.R. Miniature baited remote underwater video (mini-BRUV) reveals the response of cryptic fishes to seagrass cover. J. Appl. Phycol. 2018, 101, 1717–1722. [Google Scholar] [CrossRef]
- Swadling, D.S.; Knott, N.A.; Rees, M.J.; Pederson, H.; Adams, K.R.; Taylor, M.D.; Davis, A.R. Seagrass canopies and the performance of acoustic telemetry: Implications for the interpretation of fish movements. Anim. Biotelem. 2020, 8, 8. [Google Scholar] [CrossRef]
- French, B.; Wilson, S.; Holmes, T.; Kendrick, A.; Rule, M.; Ryan, N. Comparing five methods for quantifying abundance and diversity of fish assemblages in seagrass habitat. Ecol. Indic. 2021, 124, 107415. [Google Scholar] [CrossRef]
- Henderson, C.J.; Gilby, B.L.; Lee, S.Y.; Stevens, T. Contrasting effects of habitat complexity and connectivity on biodiversity in seagrass meadows. Mar. Biol. 2017, 164, 117. [Google Scholar] [CrossRef]
- Horinouchi, M.; Tongnunui, P.; Nanjyo, K.; Nakamura, Y.; Sano, M.; Ogawa, H. Differences in fish assemblage structures between fragmented and continuous seagrass beds in Trang, southern Thailand. Fish. Sci. 2009, 75, 1409–1416. [Google Scholar] [CrossRef]
- Smith, T.; Hindell, J.S.; Jenkins, G.P.; Connolly, R.M. Edge effects on fish associated with seagrass and sand patches. Mar. Ecol. Prog. Ser. 2008, 359, 203–213. [Google Scholar] [CrossRef]
- Macreadie, P.I.; Hindell, J.S.; Jenkins, G.P.; Connolly, R.M.; Keough, M.J. Fish Responses to Experimental Fragmentation of Seagrass Habitat. Conserv. Biol. 2009, 23, 644–652. [Google Scholar] [CrossRef] [PubMed]
- Macreadie, P.I.; Hindell, J.S.; Keough, M.J.; Jenkins, G.P.; Connolly, R.M. Resource distribution influences positive edge effects in a seagrass fish. Ecology 2010, 91, 2013–2021. [Google Scholar] [PubMed]
- Bender, D.J.; Contreras, T.A.; Fahrig, L. Habitat loss and population decline: A meta-analysis of the patch size effect. Ecology 1998, 79, 517–533. [Google Scholar] [CrossRef]
- Heck, K., Jr.; Hays, G.; Orth, R.J. Critical evaluation of the nursery role hypothesis for seagrass meadows. Mar. Ecol. Prog. Ser. 2003, 253, 123–136. [Google Scholar] [CrossRef]
- Yarnall, A.H.; Fodrie, F.J. Predation patterns across states of landscape fragmentation can shift with seasonal transitions. Oecologia 2020, 193, 403–413. [Google Scholar] [CrossRef]
- Irlandi, E.A. Large- and small-scale effects of habitat structure on rates of predation: How percent coverage of seagrass affects rates of predation and siphon nipping on an infaunal bivalve. Oecologia 1994, 98, 176–183. [Google Scholar] [CrossRef]
- Smith, T.M.; Hindell, J.S.; Jenkins, G.P.; Connolly, R.; Keough, M.J. Edge effects in patchy seagrass landscapes: The role of predation in determining fish distribution. J. Exp. Mar. Biol. Ecol. 2011, 399, 8–16. [Google Scholar] [CrossRef]
- Hovel, K.A.; Regan, H.M. Using an individual-based model to examine the roles of habitat fragmentation and behavior on predator–prey relationships in seagrass landscapes. Landsc. Ecol. 2007, 23, 75–89. [Google Scholar] [CrossRef]
- Lester, E.K.; Langlois, T.J.; Simpson, S.D.; McCormick, M.I.; Meekan, M.G. Reef-wide evidence that the presence of sharks modifies behaviors of teleost mesopredators. Ecosphere 2021, 12, e03301. [Google Scholar] [CrossRef]
- Lubbers, L.; Boynton, W.; Kemp, W. Variations in structure of estuarine fish communities in relation to abundance of submersed vascular plants. Mar. Ecol. Prog. Ser. 1990, 65, 1–14. [Google Scholar] [CrossRef]
- Olney, J.; Boehlert, G. Nearshore ichthyoplankton associated with seagrass beds in the lower Chesapeake Bay. Mar. Ecol. Prog. Ser. 1988, 45, 33–43. [Google Scholar] [CrossRef]
- Orth, R.J.; Heck, K.L.; van Montfrans, J. Faunal Communities in Seagrass Beds: A Review of the Influence of Plant Structure and Prey Characteristics on Predator: Prey Relationships. Estuaries 1984, 7, 339–350. [Google Scholar] [CrossRef]
- James, P.L.; Heck, K.L. The effects of habitat complexity and light intensity on ambush predation within a simulated seagrass habitat. J. Exp. Mar. Biol. Ecol. 1994, 176, 187–200. [Google Scholar] [CrossRef]
- Heck, K.L., Jr.; Orth, R.J. Predation in Seagrass Beds. In Seagrasses: Biology, Ecology and Conservation; Springer: Berlin/Heidelberg, Germany, 2006; pp. 537–550. [Google Scholar]
- Edgar, G.J. Artificial algae as habitats for mobile epifauna: Factors affecting colonization in a Japanese Sargassum bed. Hydrobiologia 1991, 226, 111–118. [Google Scholar] [CrossRef]
- Costa, V.; Chemello, R.; Iaciofano, D.; Brutto, S.L.; Rossi, F. Small-scale patches of detritus as habitat for invertebrates within a Zostera noltei meadow. Mar. Environ. Res. 2021, 171, 105474. [Google Scholar] [CrossRef]
- Macreadie, P.I.; Connolly, R.M.; Jenkins, G.P.; Hindell, J.S.; Keough, M.J. Edge patterns in aquatic invertebrates explained by predictive models. Mar. Freshw. Res. 2010, 61, 214–218. [Google Scholar] [CrossRef]
- Lanham, B.S.; Poore, A.G.; Gribben, P.E. Fine-scale responses of mobile invertebrates and mesopredatory fish to habitat configuration. Mar. Environ. Res. 2021, 168, 105319. [Google Scholar] [CrossRef]
- Moore, E.C.; Hovel, K.A. Relative influence of habitat complexity and proximity to patch edges on seagrass epifaunal communities. Oikos 2010, 119, 1299–1311. [Google Scholar] [CrossRef]
- Paul, M. The protection of sandy shores—Can we afford to ignore the contribution of seagrass? Mar. Pollut. Bull. 2018, 134, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, P.S.; Eklöf, J.S.; van Katwijk, M.M.; O’Brien, K.R.; de la Torre-Castro, M.; Boström, C.; Bouma, T.J.; Krause-Jensen, D.; Unsworth, R.K.F.; van Tussenbroek, B.I.; et al. The fundamental role of ecological feedback mechanisms for the adaptive management of seagrass ecosystems—A review. Biol. Rev. 2017, 92, 1521–1538. [Google Scholar] [CrossRef] [PubMed]
- van Rijn, L. Coastal erosion and control. Ocean Coast. Manag. 2011, 54, 867–887. [Google Scholar] [CrossRef]
- James, R.K.; Silva, R.; Van Tussenbroek, B.I.; Escudero-Castillo, M.; Mariño-Tapia, I.; Dijkstra, H.A.; Van Westen, R.M.; Pietrzak, J.D.; Candy, A.; Katsman, C.A.; et al. Maintaining Tropical Beaches with Seagrass and Algae: A Promising Alternative to Engineering Solutions. Bioscience 2019, 69, 136–142. [Google Scholar] [CrossRef]
- Robbins, B.D.; Bell, S.S. Seagrass landscapes: A terrestrial approach to the marine subtidal environment. Trends Ecol. Evol. 1994, 9, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Hemminga, M.A.; Duarte, C.M. Seagrass Ecology; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Waycott, M.; Duarte, C.M.; Carruthers, T.J.B.; Orth, R.J.; Dennison, W.C.; Olyarnik, S.; Calladine, A.; Fourqurean, J.W.; Heck, K.L., Jr.; Hughes, A.R.; et al. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proc. Natl. Acad. Sci. USA 2009, 106, 12377–12381. [Google Scholar] [CrossRef]
- Gilby, B.L.; Olds, A.D.; Connolly, R.M.; Henderson, C.J.; Schlacher, T.A. Spatial Restoration Ecology: Placing Restoration in a Landscape Context. Bioscience 2018, 68, 1007–1019. [Google Scholar] [CrossRef]
- Unsworth, R.; De León, P.; Garrard, S.; Jompa, J.; Smith, D.; Bell, J. High connectivity of Indo-Pacific seagrass fish assemblages with mangrove and coral reef habitats. Mar. Ecol. Prog. Ser. 2008, 353, 213–224. [Google Scholar] [CrossRef]
- Vargas-Fonseca, E.; Olds, A.D.; Gilby, B.L.; Connolly, R.M.; Schoeman, D.S.; Huijbers, C.M.; Hyndes, G.A.; Schlacher, T.A. Combined effects of urbanization and connectivity on iconic coastal fishes. Divers. Distrib. 2016, 22, 1328–1341. [Google Scholar] [CrossRef]
- Tanner, J.E. Landscape ecology of interactions between seagrass and mobile epifauna: The matrix matters. Estuar. Coast. Shelf Sci. 2006, 68, 404–412. [Google Scholar] [CrossRef]
- Olds, A.; Connolly, R.; Pitt, K.; Maxwell, P. Primacy of seascape connectivity effects in structuring coral reef fish assemblages. Mar. Ecol. Prog. Ser. 2012, 462, 191–203. [Google Scholar] [CrossRef]
- Whippo, R.; Knight, N.S.; Prentice, C.; Cristiani, J.; Siegle, M.R.; O’Connor, M.I. Epifaunal diversity patterns within and among seagrass meadows suggest landscape-scale biodiversity processes. Ecosphere 2018, 9, e02490. [Google Scholar] [CrossRef]
- Roper, T.; Creese, B.; Scanes, P.; Stephens, K.; Williams, R.; Dela-Cruz, J.; Coade, G.; Coates, B.; Fraser, M. Assessing the condition of estuaries and coastal lake ecosystems in NSW, Monitoring, evaluation and reporting program. In Estuaries and Coastal Lakes; Technical Report Series; Office of Environment and Heritage: Sydney, Australia, 2011. [Google Scholar]
- Creese, R.G.; Glasby, T.M.; West, G.; Gallen, C. Mapping the Habitats of NSW Estuaries; Fisheries Final Report Series No. 113; Industry & Investment NSW: Nelson Bay, NSW, Australia, 2009. [Google Scholar]
- Truong, L.; Suthers, I.M.; Cruz, D.O.; Smith, J.A. Plankton supports the majority of fish biomass on temperate rocky reefs. Mar. Biol. 2017, 164, 1–12. [Google Scholar] [CrossRef]
- Manjakasy, J.M.; Day, R.D.; Kemp, A.; Tibbetts, I.R. Functional morphology of digestion in the stomachless, piscivorous needlefishes Tylosurus gavialoides and Strongylura leiura ferox (Teleostei: Beloniformes). J. Morphol. 2009, 270, 1155. [Google Scholar] [CrossRef]
- Champion, C.; Suthers, I.M.; Smith, J.A. Zooplanktivory is a key process for fish production on a coastal artificial reef. Mar. Ecol. Prog. Ser. 2015, 541, 1–14. [Google Scholar] [CrossRef]

| Response Variables | Predictor Variables | Estimate | p-Value |
|---|---|---|---|
| Relative abundance of fish | Distance to meadow edge | 0.002 | 0.32 |
| Fragmentation | 1.61 | 0.004 ** | |
| Shoot density | −0.037 | 0.001 ** | |
| Fish richness | Distance to meadow edge | 0.004 | <0.001 *** |
| Fragmentation | 0.44 | 0.34 | |
| Shoot density | −0.01 | 0.03 * | |
| Epifauna abundance | Distance to meadow edge | −0.003 | 0.17 |
| Fragmentation | −0.63 | 0.75 | |
| Shoot density | −0.06 | 0.0004 *** | |
| Predation after 1 h | Distance to meadow edge | −0.01 | 0.08 |
| Fragmentation | −4.65 | 0.05 | |
| Shoot density | 0.02 | 0.55 | |
| Predation after 24 h | Distance to meadow edge | −0.009 | 0.31 |
| Fragmentation | 0.29 | 0.94 | |
| Shoot density | 0.01 | 0.72 | |
| Sediment erosion | Distance to meadow edge | 0.005 | 0.07 |
| Fragmentation | −0.89 | 0.15 | |
| Shoot density | 0.037 | 0.004 ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferretto, G.; Vergés, A.; Poore, A.G.B.; Glasby, T.M.; Griffin, K.J. Habitat Provision and Erosion Are Influenced by Seagrass Meadow Complexity: A Seascape Perspective. Diversity 2023, 15, 125. https://doi.org/10.3390/d15020125
Ferretto G, Vergés A, Poore AGB, Glasby TM, Griffin KJ. Habitat Provision and Erosion Are Influenced by Seagrass Meadow Complexity: A Seascape Perspective. Diversity. 2023; 15(2):125. https://doi.org/10.3390/d15020125
Chicago/Turabian StyleFerretto, Giulia, Adriana Vergés, Alistair G. B. Poore, Tim M. Glasby, and Kingsley J. Griffin. 2023. "Habitat Provision and Erosion Are Influenced by Seagrass Meadow Complexity: A Seascape Perspective" Diversity 15, no. 2: 125. https://doi.org/10.3390/d15020125
APA StyleFerretto, G., Vergés, A., Poore, A. G. B., Glasby, T. M., & Griffin, K. J. (2023). Habitat Provision and Erosion Are Influenced by Seagrass Meadow Complexity: A Seascape Perspective. Diversity, 15(2), 125. https://doi.org/10.3390/d15020125










