Reconstructing the Biogeographic History of the Genus Aurelia Lamarck, 1816 (Cnidaria, Scyphozoa), and Reassessing the Nonindigenous Status of A. solida and A. coerulea in the Mediterranean Sea
Abstract
:1. Introduction
2. Material and Methods
2.1. Study Site and Genetic Identification of the Aurelia Species from the Mar Menor
2.2. Mediterranean Species of Aurelia Prior to and during the Construction of the Suez Canal
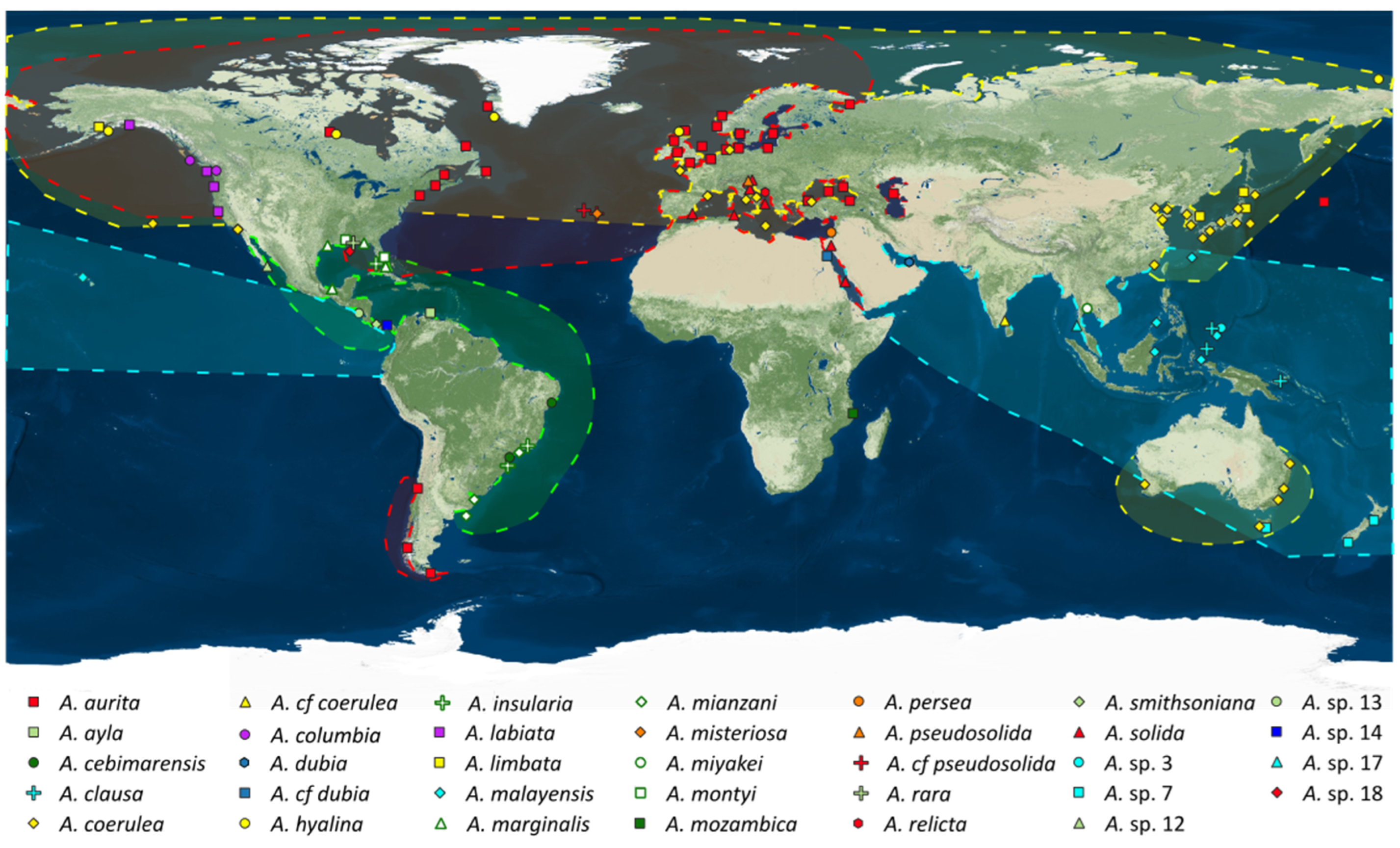
2.3. Present Biogeography of the Genus Aurelia
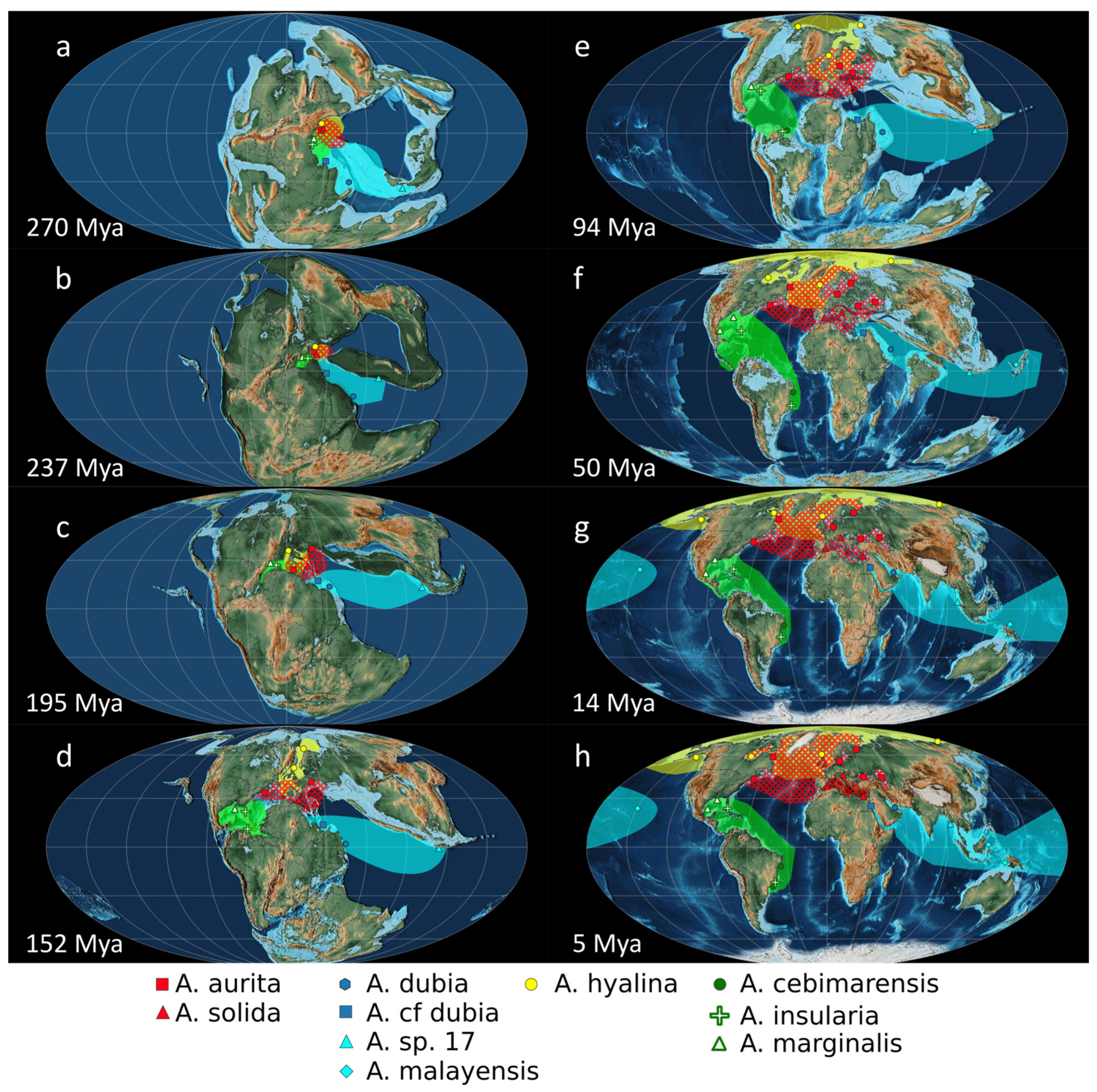
2.4. Paleogeography of the Genus Aurelia
3. Results
3.1. Determination of the Aurelia Species from the Mar Menor Coastal Lagoon
3.2. Mediterranean Species of Aurelia Prior to and during the Construction of the Suez Canal
3.3. Present Biogeography of the Genus Aurelia
3.4. Paleogeography of the Genus Aurelia
3.4.1. Hypothesis A: Disjunct Distributions Explained by Anthropic Translocations
3.4.2. Hypothesis B: Disjunct Distributions Explained by Paleogeographic Processes without Human Intervention
4. Discussion
4.1. Paleogeography of the Genus Aurelia
4.2. Limitations of the Hypotheses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lawley, J.W.; Gamero-Mora, E.; Maronna, M.M.; Chiaverano, L.M.; Stampar, S.N.; Hopcroft, R.R.; Collins, A.G.; Morandini, A.C. The importance of molecular characters when morphological variability hinders diagnosability: Systematics of the moon jellyfish genus Aurelia (Cnidaria: Scyphozoa). PeerJ 2021, 9, e11954. [Google Scholar] [CrossRef] [PubMed]
- Pitt, K.A.; Lucas, C.H.; Condon, R.H.; Duarte, C.M.; Stewart-Koster, B. Claims that anthropogenic stressors facilitate jellyfish blooms have been amplified beyond the available evidence: A systematic review. Front. Mar. Sci. 2018, 5, 451. [Google Scholar] [CrossRef]
- Linnaeus, C. Systema Naturae perR tria Naturæ, Secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis, 10th ed.; Impensis Direct Laurentii Salvii: Stockholm, Sweden, 1758; Volume 1, p. 824. [Google Scholar] [CrossRef]
- Péron, F.; Lesueur, C.A. Tableau des caractères génériques et spécifiques de toutes les espèces de méduses connues jusqu’à ce jour. Ann. Du Muséum Natl. D’histoire Nat. De Paris 1810, 14, 325–366. [Google Scholar]
- Lamarck, J.B.M. Histoire Naturelle des Animaux Sans Vertèbres; Smithsonian Institution: Paris, France, 1816; Volume 2, p. 568. [Google Scholar] [CrossRef]
- Mayer, A.G. Medusae of the World. Vol III, The Scyphomedusae; Carnegie Institution of Washington: Washington, DC, USA, 1910. [Google Scholar] [CrossRef]
- Lucas, C.H. Reproduction and life history strategies of the common jellyfish, Aurelia aurita, in relation to its ambient environment. Hydrobiologia 2001, 451, 229–246. [Google Scholar] [CrossRef]
- Gershwin, L.A. Jellyfish: A Natural History; University of Chicago Press: Chicago, IL, USA, 2016. [Google Scholar]
- Dawson, M.N.; Martin, L.E. Geographic variation and ecological adaptation in Aurelia (Scyphozoa, Semaeostomeae): Some implications from molecular phylogenetics. In Jellyfish Blooms: Ecological and Societal Importance; Purcell, J.E., Graham, W.M., Dumont, H.J., Eds.; Springer: Dordrecht, Netherlands, 2001; pp. 259–273. [Google Scholar]
- Dawson, M.N. Macro-morphological variation among cryptic species of the moon jellyfish, Aurelia (Cnidaria: Scyphozoa). Mar. Biol. 2003, 143, 369–379. [Google Scholar] [CrossRef]
- Scorrano, S.; Aglieri, G.; Boero, F.; Dawson, M.N.; Piraino, S. Unmasking Aurelia species in the Mediterranean Sea: An integrative morphometric and molecular approach. Zool. J. Linn. Soc. 2017, 180, 243–267. [Google Scholar] [CrossRef]
- Brown, M.; Scorrano, S.; Kuplik, Z.; Kuyper, D.; Ras, V.; Thibault, D.; Engelbrecht, A.; Gibbons, M.J. A new macromedusa from the coast of Mozambique: Aurelia mozambica sp. nov. (Scyphozoa: Ulmaridae). Zootaxa 2021, 4933, 263–276. [Google Scholar] [CrossRef]
- Garić, R.; Batistić, M. Description of Aurelia pseudosolida sp. nov. (Scyphozoa, Ulmaridae) from the Adriatic Sea. Water 2022, 14, 135. [Google Scholar] [CrossRef]
- Moura, C.J.; Magalhães, B.I.; Gonçalves, J.M. DNA Barcoding of Moon Jellyfish (Cnidaria, Scyphozoa, Ulmaridae, Aurelia): Two Cryptic Species from the Azores (NE Atlantic, Macaronesia), and Evaluation of the Non-Indigenous Species (NIS). Diversity 2023, 15, 323. [Google Scholar] [CrossRef]
- Dawson, M.N.; Gupta, A.S.; England, M.H. Coupled biophysical global ocean model and molecular genetic analyses identify multiple introductions of cryptogenic species. Proc. Natl. Acad. Sci. USA 2005, 102, 11968–11973. [Google Scholar] [CrossRef]
- Browne, E.T. Scyphomedusae. In The Fauna and Geography of the Maldive and Laccadive Archipelagoes; Gardiner, I.S., Ed.; University Press: Cambridge, UK, 1905; Volume 2, (Suppl. S1), pp. 958–971. [Google Scholar] [CrossRef]
- Agassiz, L. Contributions to the Natural History of the United States of America; Little, Brown and Company: Boston, MA, USA, 1862. [Google Scholar] [CrossRef]
- Mizrahi, G.A. Phylogenetic Analysis of Gelatinous Marine Fauna in the Eastern Mediterranean Basin—An Ecosystem under Anthropogenic Stress. Ph.D. Thesis, University of Haifa, Haifa, Israel, 2014. [Google Scholar]
- Pérez-Ruzafa, A. Estudio Ecológico y Bionómico de los Poblamientos Bentónicos del Mar Menor (Murcia, SE de España). Ph.D. Thesis, University of Murcia, Murcia, Spain, 1989. [Google Scholar]
- Pérez-Ruzafa, A.; Campillo, S.; Fernández-Palacios, J.M.; Garcia-Lacunza, A.; Garcia-Oliva, M.; Ibañez, H.; Navarro-Martínez, P.C.; Pérez-Marcos, M.; Pérez-Ruzafa, I.M.; Quispe-Becerra, J.I.; et al. Long-term dynamic in nutrients, chlorophyll a, and water quality parameters in a coastal lagoon during a process of eutrophication for decades, a sudden break and a relatively rapid recovery. Front. Mar. Sci. 2019, 6, 26. [Google Scholar] [CrossRef]
- Fernández-Alías, A.; Montaño-Barroso, T.; Conde-Caño, M.R.; Manchado-Pérez, S.; López-Galindo, C.; Quispe-Becerra, J.I.; Marcos, C.; Pérez-Ruzafa, A. Nutrient overload promotes the transition from top-down to bottom-up control and triggers dystrophic crises in a Mediterranean coastal lagoon. Sci. Total Environ. 2022, 846, 157388. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Ruzafa, A.; Gilabert, J.; Gutiérrez, J.M.; Fernández, A.I.; Marcos, C.; Sabah, S. Evidence of a planktonic food web response to changes in nutrient input dynamics in the Mar Menor coastal lagoon, Spain. Hydrobiologia 2002, 475/476, 359–369. [Google Scholar] [CrossRef]
- Fernández-Alías, A.; Molinero, J.C.; Quispe-Becerra, J.I.; Bonnet, D.; Marcos, C.; Pérez-Ruzafa, A. Phenology of scyphozoan jellyfish species in a eutrophication and climate change context. Mar. Pollut. Bull. 2023, 194, 115286. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Alías, A.; Marcos, C.; Quispe, J.I.; Sabah, S.; Pérez-Ruzafa, A. Population dynamics and growth in three scyphozoan jellyfishes, and their relationship with environmental conditions in a coastal lagoon. Estuar. Coast. Shelf Sci. 2020, 243, 106901. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, A.R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Piraino, S.; Aglieri, G.; Martell, L.; Mazzoldi, C.; Melli, V.; Milisenda, G.; Scorrano, S.; Boero, F. Pelagia benovici sp.nov. (Cnidaria, Scyphozoa): A new jellyfish in the Mediterranean Sea. Zootaxa 2014, 3794, 455–468. [Google Scholar] [CrossRef] [PubMed]
- Bayha, K.M.; Dawson, M.N.; Collins, A.G.; Barbeitos, M.S.; Haddock, S.H. Evolutionary relationships among scyphozoan jellyfish families based on complete taxon sampling and phylogenetic analyses of 18S and 28S ribosomal DNA. Integr. Comp. Biol. 2010, 50, 436–455. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: More models, new heuristics and high-performance computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, T.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. The PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Forsskål, P. Descriptiones Animalium, Avium, Amphibiorum; Mölleri: Copenhagen, Denmark, 1775. [Google Scholar] [CrossRef]
- Gmelin, J.F. Caroli a Linne. Systema Naturae Per Regna Tria Naturae, Secundum Classes, Ordines, Genera, Speciescum Characteribus, Differentiis, Synonymis, Locis; Apud J.B. Delamolliere: Lyon, France, 1789. [Google Scholar] [CrossRef]
- Chamisso, A.; Eysenhardt, C.G. De Animalibus Quibusdam e Classe Vermium Linneana, in Circumnavigatione Terrae, Auspicante Comite N. Romanoff, duce Ottone di Kotzebue, Annis 1815–1818 Peracta, Observatis Fasciculus Secundus, Reliquos Vermes Continens. Nova Acta physico-medica Academiae Cesareae Leopoldino-Carolinae 10. 1821. Available online: https://www.biodiversitylibrary.org/page/37020829#page/479/mode/1up (accessed on 27 November 2023).
- Lesson, R.P. Zoologie. Voyage Autour du Monde: Exécuté Par Ordre du roi, sur la Corvette de Sa Majesté, la Coquille, Pendant les Années 1822, 1823, 1824, et 1825; Duperrey, M.L.I.: Paris, France, 1830. [Google Scholar] [CrossRef]
- Brandt, J.F. Prodromus descriptionis animalium ab H. Mertensio observatorum: Fascic. I. Polypos, Acalephas Discophoras et Siphonophoras, nec non Echinodermata continens. In Recueil des Actes de la Séance Publique de l’Académie Impériale des Sciences de St. Pétersbourg; Académie Impériale des Sciences: Saint Petersburg, Russia, 1835. [Google Scholar] [CrossRef]
- Haeckel, E. System der Ascrapeden. In Monographie der Medusen; Fisher, J.G., Ed.; Jena Publisher: Germany, 1879. [Google Scholar] [CrossRef]
- Von Lendenfeld, R. The scyphomedusae of the southern hemisphere. Part I-III. Proc. Linn. Soc. New South Wales 1884, 9, 259–306. [Google Scholar]
- Vanhöffen, E. Untersuchungen über Semäostome und Rhizostome Medusen. In Bibliotheca Zoologica; Wentworth Press: London, UK, 1888. [Google Scholar] [CrossRef]
- Agassiz, A.; Mayer, A.G. Acalephs from the Fiji Islands. Bull. Mus. Comp. Zool. 1899, 32. Available online: https://www.biodiversitylibrary.org/page/28879983 (accessed on 27 November 2023).
- Bigelow, H.B. Medusae from the Maldive Islands. Bull. Mus. Comp. Zool. 1904, 39. Available online: https://ia802502.us.archive.org/3/items/biostor-57858/biostor-57858.pdf (accessed on 27 November 2023).
- WORMS. World Register of Marine Species. 2023. Available online: https://www.marinespecies.org (accessed on 20 October 2023).
- Khalturin, K.; Shinzato, C.; Khalturina, M.; Hamada, M.; Fujie, M.; Koyanagi, R.; Kanda, M.; Goto, H.; Anton-Erxleben, F.; Toyokawa, M.; et al. Medusozoan genomes inform the evolution of the jellyfish body plan. Nat. Ecol. Evol. 2019, 3, 811–822. [Google Scholar] [CrossRef]
- Scotese, C.R.; McKerrow, W.S. Revised world maps and introduction. Geol. Soc. Lond. Mem. 1990, 12, 1–21. [Google Scholar] [CrossRef]
- Scotese, C.R. Atlas of Earth History; PALEOMAP Project; University of Texas at Arlington, Department of Geology: Arlington, TX, USA, 2001. [Google Scholar]
- Young, G.A.; Hagadorn, J.W. Evolving preservation and facies distribution of fossil jellyfish: A slowly closing taphonomic window. Boll. Della Soc. Paleontol. Ital. 2020, 59, 186. [Google Scholar] [CrossRef]
- Fu, D.; Tong, G.; Dai, T.; Liu, W.; Yang, Y.; Zhang, Y.; Cui, L.; Li, L.; Yun, H.; Wu, Y.; et al. The Qingjiang biota—A Burgess Shale–type fossil Lagerstätte from the early Cambrian of South China. Science 2019, 363, 1338–1342. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, C.; Goy, J.; Bernier, P.; Bourseau, J.P.; Gall, J.C.; Barale, G.; Buffetaut, E.; Wenz, S. New jellyfish taxa from the Upper Jurassic lithographic limestones of Cerin (France): Taphonomy and ecology. Palaeontology 2006, 49, 1287–1302. [Google Scholar] [CrossRef]
- Bruton, D.L. Beach and laboratory experiment with the jellyfish Aurelia and remarks on some fossil medusoid traces. In The Early Evolution of Metazoa and the Significance of Problematic Taxa; Simonetta, A.M., Conway Morris, S., Eds.; University Press: Cambridge, UK, 1991; pp. 125–129. [Google Scholar]
- Giusberti, L.; Fornasiero, M.; Zorzin, R. The Pesciara-Monte Postale fossil-Lagerstätte: 4. the “minor fauna” of the laminites. In The Bolca Fossil-Lagerstätte: A window into the Eocene World; Papazzoni, C.A., Giusberti, L., Carnevale, G., Roghi, G., Bassi, D., Zorzin, R., Eds.; Società Paleontologica Italiana: Milan, Italy, 2014; pp. 73–87. [Google Scholar]
- Kishinouye, K. Aurelia japonica, nov. sp. Zoological Magazine (Dobutsugaku zasshi). Zootaxa 1891, 3, 289–291. [Google Scholar]
- Chatterjee, S.; Goswami, A.; Scotese, C.R. The longest voyage: Tectonic, magmatic, and paleoclimatic evolution of the Indian plate during its northward flight from Gondwana to Asia. Gondwana Res. 2013, 23, 238–267. [Google Scholar] [CrossRef]
- Gibbons, M.J.; Morandini, A.C.; Straehler-Pohl, I.; Bezio, N. Identification Guide to Macro Jellyfishes of West Africa; FAO: Rome, Italy, 2022. [Google Scholar] [CrossRef]
- Kramp, P.L. Medusae, mainly from the west coast of Africa. Inst. R. Des Sci. Nat. De Belg. 1959, 3, 1–33. [Google Scholar]
- Lynam, C.P.; Gibbons, M.J.; Axelsen, B.E.; Sparks, C.A.J.; Coetzee, J.; Heywood, B.G.; Brierley, A.S. Jellyfish overtake fish in a heavily fished ecosystem. Curr. Biol. 2006, 16, 492–493. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, S.; Gueroun, S.K.; Andrade, C.; Canning-Clode, J. Combined effects of temperature and salinity on polyps and ephyrae of Aurelia solida (Cnidaria: Scyphozoa). Diversity 2021, 13, 573. [Google Scholar] [CrossRef]
- Gueroun, S.M.; Molinero, J.C.; Piraino, S.; Dali Yahia, M.N. Population dynamics and predatory impact of the alien jellyfish Aurelia solida (Cnidaria, Scyphozoa) in the Bizerte Lagoon (southwestern Mediterranean Sea). Mediterr. Mar. Sci. 2020, 21, 22–35. [Google Scholar] [CrossRef]




| Sample | Date | Latitude | Longitude | COI | 28S |
|---|---|---|---|---|---|
| I00 | June 1987 | 37°41.159′ N | 0°47.836′ W | OR724094 | |
| I01 | 28 April 2020 | 37°39.374′ N | 0°47.291′ W | OR727336 | OR724077 |
| I02 | 28 April 2020 | 37°39.374′ N | 0°47.291′ W | OR727337 | OR724078 |
| I03 | 28 April 2020 | 37°39.374′ N | 0°47.291′ W | OR724079 | |
| I04 | 28 April 2020 | 37°39.374′ N | 0°47.291′ W | OR727338 | OR724080 |
| I05 | 28 April 2020 | 37°39.374′ N | 0°47.291′ W | OR727339 | OR724081 |
| I06 | 28 April 2020 | 37°43.405′ N | 0°49.813′ W | OR727340 | OR724082 |
| I07 | 28 April 2020 | 37°43.405′ N | 0°49.813′ W | OR727341 | OR724083 |
| I08 | 28 April 2020 | 37°44.691′ N | 0°47.292′ W | OR724084 | |
| I37 | 15 June 2020 | 37°47.777′ N | 0°47.531′ W | OR727326 | OR724085 |
| I40 | 15 June 2020 | 37°47.777′ N | 0°47.531′ W | OR727325 | OR724086 |
| I83 | 15 June 2020 | 37°47.777′ N | 0°47.531′ W | OR727324 | OR724087 |
| I84 | 15 June 2020 | 37°47.777′ N | 0°47.531′ W | OR727323 | OR724088 |
| I88 | 4 February 2021 | 37°46.193′ N | 0°47.655′ W | OR727335 | OR724070 |
| I89 | 4 February 2021 | 37°46.193′ N | 0°47.655′ W | OR727334 | OR724071 |
| I90 | 4 February 2021 | 37°44.691′ N | 0°47.292′ W | OR727333 | OR724072 |
| I91 | 4 February 2021 | 37°44.691′ N | 0°47.292′ W | OR727332 | OR724092 |
| I92 | 4 February 2021 | 37°43.405′ N | 0°49.813′ W | OR727331 | OR724093 |
| I93 | 4 February 2021 | 37°43.405′ N | 0°49.813′ W | OR727330 | OR724073 |
| I94 | 4 February 2021 | 37°42.269′ N | 0°47.202′ W | OR727329 | OR724074 |
| I95 | 4 February 2021 | 37°42.269′ N | 0°47.202′ W | OR727328 | OR724075 |
| I96 | 4 February 2021 | 37°42.269′ N | 0°47.202′ W | OR727327 | OR724076 |
| I117 | 31 May 2022 | 37°41.141′ N | 0°47.977′ W | OR724089 | |
| I118 | 31 May 2022 | 37°41.155′ N | 0°48.730′ W | OR727322 | OR724090 |
| I119 | 31 May 2022 | 37°44.691′ N | 0°47.292′ W | OR727321 | OR724091 |
| References (Chronological Order) |
|---|
| Linnaeus, C. Systema Naturae per tria Naturæ, Secundum Classes, Ordines, Genera, Species, Cum Characteribus, Differentiis, Synonymis, Locis, 10th ed.; Impensis Direct Laurentii Salvii: Stockholm, Sweden, 1758; Volume 1, p. 824. [3] |
| Forsskål, P. Descriptiones Animalium, Avium, Amphibiorum; Mölleri: Copenhagen, Denmark, 1775. [34] |
| Gmelin, J.F. Caroli a Linne. Systema Naturae Per Regna Tria Naturae, Secundum Classes, Ordines, Genera, Speciescum Characteribus, Differentiis, Synonymis, Locis; Delamolliere, J.B., Ed.; Lyon, France, 1789. [35] |
| Péron, F.; Lesueur, C.A. Tableau des caractères génériques et spécifiques de toutes les espèces de méduses connues jusqu’à ce jour. Ann. Du Muséum Natl. D’histoire Nat. De Paris 1810, 14, 325–366. [4] |
| Lamarck, J.B.M. Histoire Naturelle des Animaux Sans Vertèbres; Paris, France, 1816. Volume 2, p. 568. [5] |
| Chamisso, A.; Eysenhardt, C.G. De animalibus quibusdam e classe vermium Linneana, in circumnavigatione Terrae, auspicante Comite N. Romanoff, duce Ottone di Kotzebue, annis 1815–1818 peracta, observatis Fasciculus secundus, reliquos vermes continens. Nova Acta physico-medica Academiae Cesareae Leopoldino-Carolinae 10, 1821. [36] |
| Lesson, R.P. Zoologie. In Voyage Autour du Monde: Exécuté Par Ordre du roi, sur la Corvette de Sa Majesté, la Coquille, Pendant les Années 1822, 1823, 1824, et 1825; Duperrey, M.L.I. Paris, France, 1830. [37] |
| Brandt, J.F. Prodromus descriptionis animalium ab H. Mertensio observatorum: Fascic. I. Polypos, Acalephas Discophoras et Siphonophoras, nec non Echinodermata continens. In Recueil des Actes de la Séance Publique de l’Académie Impériale des Sciences de St. Pétersbourg; Académie Impériale des Sciences: Saint Petersburg, Russia, 1835. [38] |
| Agassiz, L. Contributions to the Natural History of the United States of America; Little, Brown and Company: Boston, MA, USA, 1862. [17] |
| Haeckel, E. System der Ascrapeden. Monographie der Medusen; Fisher, J.G., Ed.; Germany, 1879. [39] |
| von Lendenfeld, R. The scyphomedusae of the southern hemisphere. Part I-III. Proc. Linn. Soc. New South Wales 1884, 9, 259–306. [40] |
| Vanhöffen, E. Untersuchungen über Semäostome und Rhizostome Medusen; Bibliotheca Zoologica, 1888. [41] |
| Agassiz, A.; Mayer, A.G. Acalephs from the Fiji Islands. Bull. Mus. Comp. Zool., 32, 1899. [42] |
| Bigelow, H.B. Medusae from the Maldive Islands. Bull. Mus. Comp. Zool, 39. 1904. [43] |
| Browne, E.T. Scyphomedusae. In The Fauna and Geography of the Maldive and Laccadive Archipelagoes; Gardiner, I.S., Ed.; University Press: Cambridge, UK, 1905; Volume 2 (Suppl. 1) [16] |
| Mayer, A.G. Medusae of the World. Vol III, The Scyphomedusae; Carnegie Institution of Washington: Washington, DC, USA, 1910. [6] |
| Mar Menor | A. solida | A. aurita | A. relicta | A. coerulea | Outgroup | |
|---|---|---|---|---|---|---|
| Mar Menor | 0.1 ± 0 | 2.7 ± 0.6 | 2.4 ± 0.5 | 2.1 ± 0.5 | 19.0 ± 1.7 | |
| A. solida | 2.1 ± 0.5 | 2.7 ± 0.6 | 2.4 ± 0.5 | 2.1 ± 0.5 | 19.0 ± 1.7 | |
| A. aurita | 22.3 ± 2.3 | 22.0 ± 2.3 | 4.1 ± 0.7 | 3.7 ± 0.7 | 19.1 ± 1.7 | |
| A. relicta | 22.2 ± 2.4 | 21.7 ± 2.4 | 20.5 ± 2.2 | 2.9 ± 0.5 | 20.3 ± 1.7 | |
| A. coerulea | 20.2 ± 2.1 | 20.2 ± 2.1 | 21.0 ± 2.3 | 21.9 ± 2.4 | 19.7 ± 1.7 | |
| Outgroup | 28.4 ± 2.8 | 28.4 ± 2.8 | 28.0 ± 2.7 | 27.5 ± 2.8 | 27.7 ± 2.7 |
| Reference | Species (as Indicated) | Location |
|---|---|---|
| Forsskål, 1775 [34] | Medusa persea | Mediterranean Sea |
| Medusa cruciata | Mediterranean Sea | |
| Gmelin, 1789 [35] | Medusa tyrrhena | Thyrrenian Sea |
| Medusa persea | ||
| Medusa crucigera | ||
| Péron & Lesueur, 1810 [4] | Aurellia phosphorica | Strait of Messina |
| Aurellia amaranthea | Naples | |
| Aurellia rufescens | Mediterranean Sea | |
| Lamarck, 1816 [5] | Aurelia phosphorea | Strait of Messina |
| Aurelia tyrrhena | Naples | |
| Aurelia crucigera | Mediterranean Sea | |
| Agassiz, 1862 [17] | Aurelia aurita | Mediterranean Sea |
| Haeckel, 1879 [39] | Aurelia aurita | Atlantic coast of Europe and Mediterranean Sea |
| Mayer, 1910 [6] | Aurelia aurita | Atlantic coast of Europe and Mediterranean Sea |
| Aurelia cruciata (a variety of A. aurita) | Atlantic coast of Spain and Mediterranean Sea |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Alías, A.; Marcos, C.; Pérez-Ruzafa, A. Reconstructing the Biogeographic History of the Genus Aurelia Lamarck, 1816 (Cnidaria, Scyphozoa), and Reassessing the Nonindigenous Status of A. solida and A. coerulea in the Mediterranean Sea. Diversity 2023, 15, 1181. https://doi.org/10.3390/d15121181
Fernández-Alías A, Marcos C, Pérez-Ruzafa A. Reconstructing the Biogeographic History of the Genus Aurelia Lamarck, 1816 (Cnidaria, Scyphozoa), and Reassessing the Nonindigenous Status of A. solida and A. coerulea in the Mediterranean Sea. Diversity. 2023; 15(12):1181. https://doi.org/10.3390/d15121181
Chicago/Turabian StyleFernández-Alías, Alfredo, Concepción Marcos, and Angel Pérez-Ruzafa. 2023. "Reconstructing the Biogeographic History of the Genus Aurelia Lamarck, 1816 (Cnidaria, Scyphozoa), and Reassessing the Nonindigenous Status of A. solida and A. coerulea in the Mediterranean Sea" Diversity 15, no. 12: 1181. https://doi.org/10.3390/d15121181
APA StyleFernández-Alías, A., Marcos, C., & Pérez-Ruzafa, A. (2023). Reconstructing the Biogeographic History of the Genus Aurelia Lamarck, 1816 (Cnidaria, Scyphozoa), and Reassessing the Nonindigenous Status of A. solida and A. coerulea in the Mediterranean Sea. Diversity, 15(12), 1181. https://doi.org/10.3390/d15121181








