Assessing the Spatiotemporal Relationship between Coastal Habitats and Fish Assemblages at Two Neotropical Estuaries of the Mexican Pacific
Abstract
:1. Introduction
2. Materials and Methods
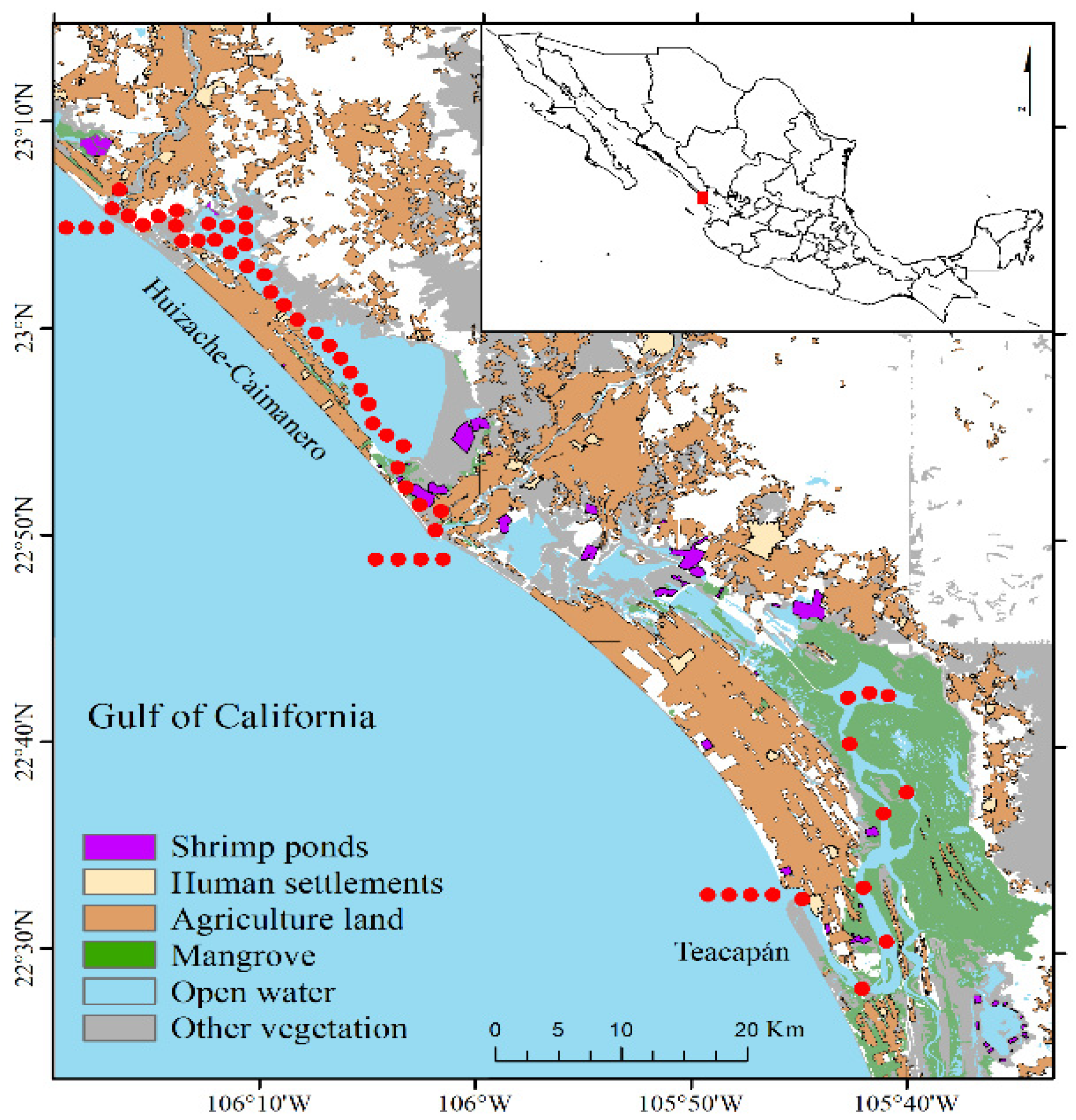
2.1. Study Area
2.2. Sampling Design and Procedures
2.3. Data Analysis Environmental Data and Habitat Characteristics
2.4. Fish Assemblages
3. Results
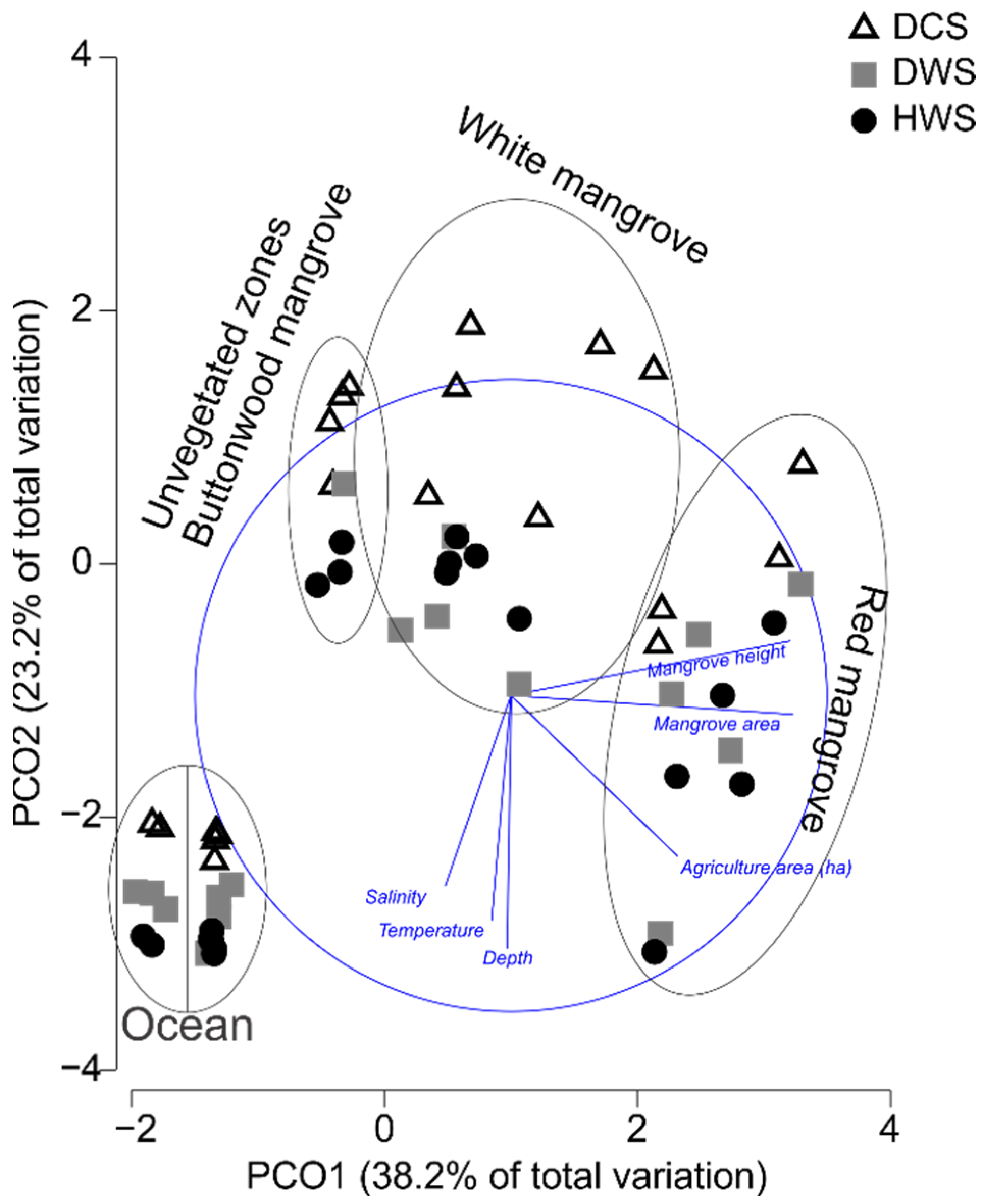
3.1. Environmental Data and Habitat Characteristics
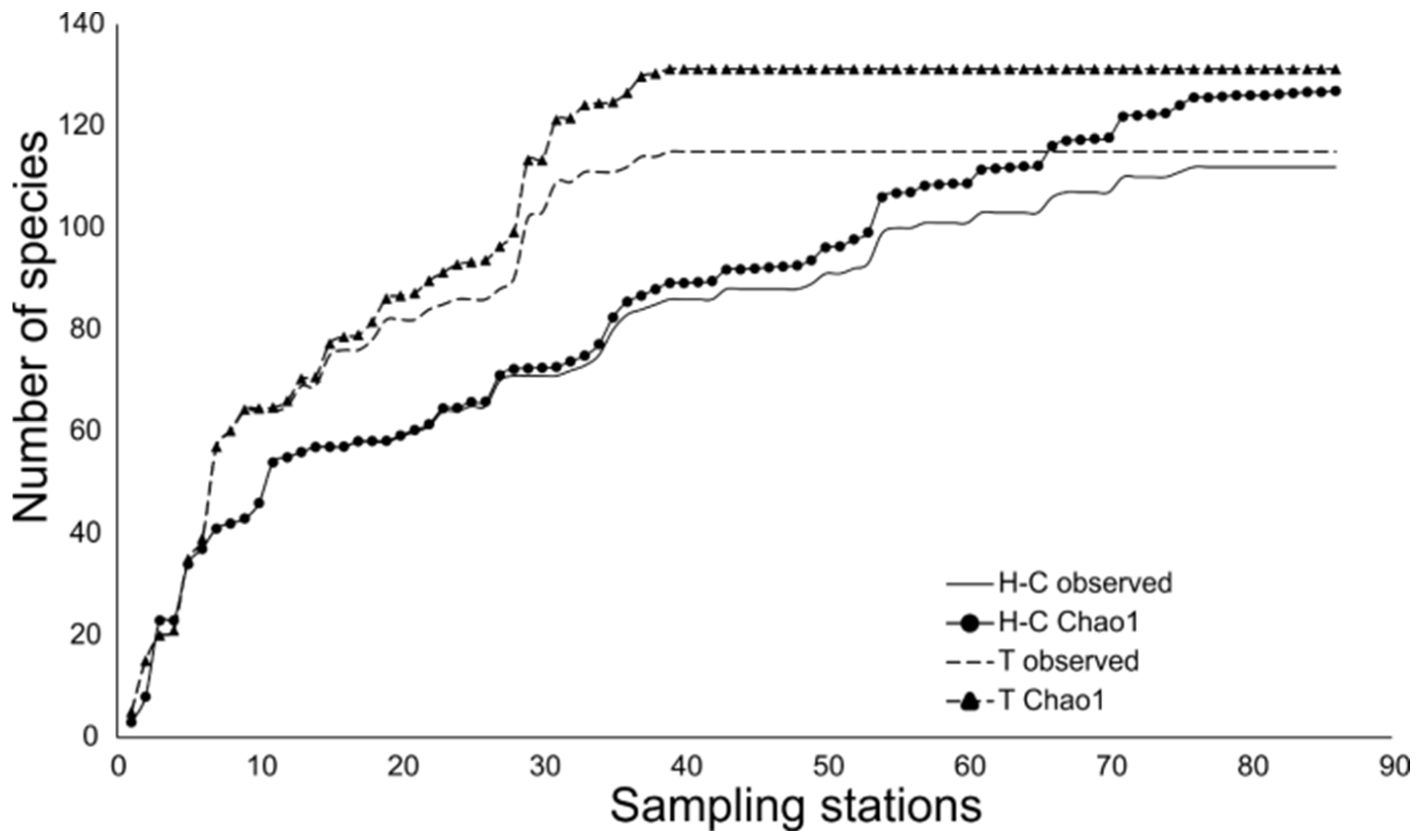
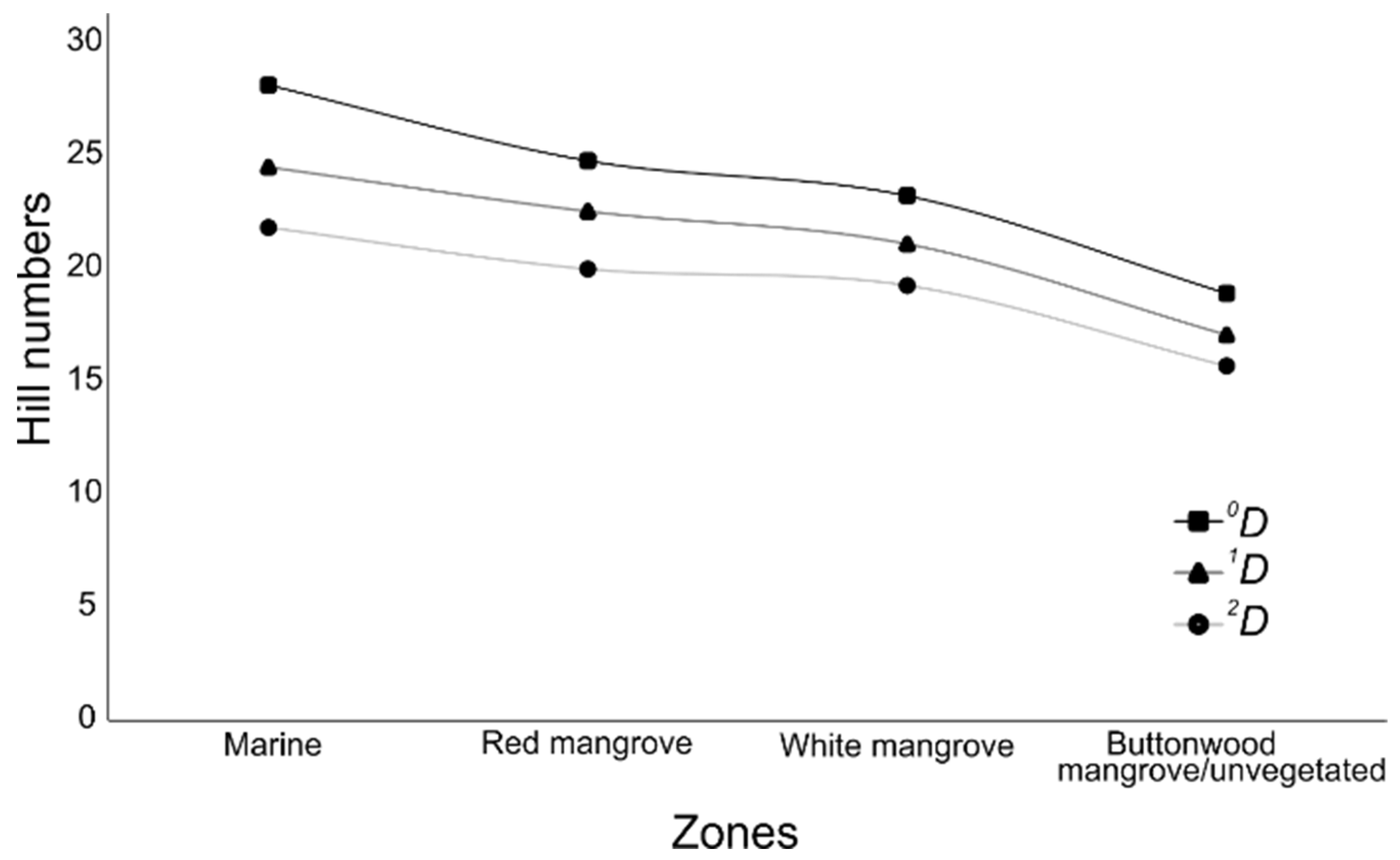
3.2. Fish Assemblages
4. Discussion
4.1. Environmental Data and Habitat Characteristics
4.2. Fish Assemblages
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kandasamy, K.; Bingham, B. Biology of Mangroves and Mangrove Ecosystems. Adv. Mar. Biol. 2001, 40, 81–251. [Google Scholar] [CrossRef]
- Crona, B.I.; Rönnbäck, P. Use of Replanted Mangroves as Nursery Grounds by Shrimp Communities in Gazi Bay, Kenya. Estuar. Coast. Shelf Sci. 2005, 65, 535–544. [Google Scholar] [CrossRef]
- Chowdhury, M.S.N.; Hossain, M.S.; Das, N.G.; Barua, P. Environmental Variables and Fisheries Diversity of the Naaf River Estuary, Bangladesh. J. Coast. Conserv. 2011, 15, 163–180. [Google Scholar] [CrossRef]
- Hossain, M.S.; Gopal Das, N.; Sarker, S.; Rahaman, M.Z. Fish Diversity and Habitat Relationship with Environmental Variables at Meghna River Estuary, Bangladesh. Egypt. J. Aquat. Res. 2012, 38, 213–226. [Google Scholar] [CrossRef] [Green Version]
- Larson, M. Coastal Lagoons. In Encyclopedia of Lakes and Reservoirs; Bengtsson, L., Herschy, R.W., Fairbridge, R.W., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2012; pp. 171–173. ISBN 978-1-4020-4410-6. [Google Scholar]
- Barletta, M.; Amaral, C.S.; Corrêa, M.F.M.; Guebert, F.; Dantas, D.V.; Lorenzi, L.; Saint-Paul, U. Factors Affecting Seasonal Variations in Demersal Fish Assemblages at an Ecocline in a Tropical-Subtropical Estuary. J. Fish Biol. 2008, 73, 1314–1336. [Google Scholar] [CrossRef]
- Blaber, S.J.M. Tropical Estuarine Fishes: Ecology, Exploitation and Conservation; John Wiley & Sons: Hoboken, NJ, USA, 2008; ISBN 0470694874. [Google Scholar]
- Franco, A.; Pérez-Ruzafa, A.; Drouineau, H.; Franzoi, P.; Koutrakis, E.T.; Lepage, M.; Verdiell-Cubedo, D.; Bouchoucha, M.; López-Capel, A.; Riccato, F.; et al. Assessment of Fish Assemblages in Coastal Lagoon Habitats: Effect of Sampling Method. Estuar. Coast. Shelf Sci. 2012, 112, 115–125. [Google Scholar] [CrossRef]
- Rodríguez-Romero, J.; del Carmen López-González, L.; Galván-Magaña, F.; Sánchez-Gutiérrez, F.; Inohuye, R.; Pérez-Urbiola, J. Seasonal Changes in a Fish Assemblage Associated with Mangroves in a Coastal Lagoon of Baja California Sur, Mexico. Lat. Am. J. Aquat. Res. 2011, 39, 250–260. [Google Scholar] [CrossRef]
- Franco, A.; Franzoi, P.; Malavasi, S.; Riccato, F.; Torricelli, P.; Mainardi, D. Use of Shallow Water Habitats by Fish Assemblages in a Mediterranean Coastal Lagoon. Estuar. Coast. Shelf Sci. 2006, 66, 67–83. [Google Scholar] [CrossRef]
- Laegdsgaard, P.; Johnson, C. Why Do Juvenile Fish Utilise Mangrove Habitats? J. Exp. Mar. Biol. Ecol. 2001, 257, 229–253. [Google Scholar] [CrossRef] [Green Version]
- Verweij, R.; Verrelst, J.; Loth, P.E.; Heitkonig, I.; Brunsting, A.M.H. Grazing Lawns Contribute to the Subsistence of Mesoherbivores on Dystrophic Savannas. Oikos 2006, 114, 108–116. [Google Scholar] [CrossRef]
- Lugendo, B.; Nagelkerken, I.; Kruitwagen, G.; Van der Velde, G.; Mgaya, Y. Relative Importance of Mangroves as Feeding Habitats for Fishes: A Comparison between Mangrove Habitats with Different Settings. Bull. Mar. Sci. 2007, 80, 497–512. [Google Scholar]
- Robertson, A.I.; Duke, N.C. Mangrove Fish-Communities in Tropical Queensland, Australia: Spatial and Temporal Patterns in Densities, Biomass and Community Structure. Mar. Biol. 1990, 104, 369–379. [Google Scholar] [CrossRef]
- Krumme, U.; Saint-Paul, U. Observations of Fish Migration in a Macrotidal Mangrove Channel in Northern Brazil Using a 200 kHz Split-Beam Sonar. Aquat. Living Resour. 2003, 16, 175–184. [Google Scholar] [CrossRef]
- Matheson, R.; Gilmore, R.G. Mojarras (Pisces: Gerreidae) of the Indian River Lagoon. Bull. Mar. Sci. 1995, 57, 281–285. [Google Scholar]
- Ikejima, K.; Tongnunui, P.; Medej, T.; Taniuchi, T. Juvenile and Small Fishes in a Mangrove Estuary in Trang Province, Thailand: Seasonal and Habitat Differences. Estuar. Coast. Shelf Sci. 2003, 56, 447–457. [Google Scholar] [CrossRef]
- Shervette, V.R.; Aguirre, W.E.; Blacio, E.; Cevallos, R.; Gonzalez, M.; Pozo, F.; Gelwick, F. Fish Communities of a Disturbed Mangrove Wetland and an Adjacent Tidal River in Palmar, Ecuador. Estuar. Coast. Shelf Sci. 2007, 72, 115–128. [Google Scholar] [CrossRef]
- Sheridan, P.; Hays, C. Are Mangroves Nursery Habitat for Transient Fishes and Decapods? Wetlands 2003, 23, 449–458. [Google Scholar] [CrossRef]
- Mumby, P.J.; Edwards, A.J.; Arias-González, J.E.; Lindeman, K.C.; Blackwell, P.G.; Gall, A.; Gorczynska, M.I.; Harborne, A.R.; Pescod, C.L.; Renken, H.; et al. Mangroves Enhance the Biomass of Coral Reef Fish Communities in the Caribbean. Nature 2004, 427, 533–536. [Google Scholar] [CrossRef] [Green Version]
- Barletta, M.; Barletta-Bergan, A.; Saint-Paul, U.; Hubold, G. The Role of Salinity in Structuring the Fish Assemblages in a Tropical Estuary. J. Fish Biol. 2005, 66, 45–72. [Google Scholar] [CrossRef]
- Whitfield, A. Abundance of Larval and 0+juvenile Marine Fishes in Three Southern African Estuaries with Differing Freshwater Inputs. Mar. Ecol. Ser. Mar. Ecol. Prog. Ser. 1994, 105, 257–267. [Google Scholar] [CrossRef]
- Huxham, M.; Kimani, E.; Augley, J. Mangrove Fish: A Comparison of Community Structure between Forested and Cleared Habitats. Estuar. Coast. Shelf Sci. 2004, 60, 637–647. [Google Scholar] [CrossRef]
- Blaber, S.; Brewer, D.; Harris, A.N. Distribution, Biomass and Community Structure of Demersal Fishes of the Gulf of Carpentaria, Australia. Mar. Freshw. Res. 1994, 45, 375–396. [Google Scholar] [CrossRef]
- Nagelkerken, I.; Faunce, C. Colonisation of Artificial Mangrove Shorelinies by Reef Fishes in a Marine Landscape. Estuar. Coast. Shelf Sci. 2007, 75, 417–422. [Google Scholar] [CrossRef]
- Barletta, M.; Barletta-Bergan, A.; Saint-Paul, U.; Hubold, G. Seasonal Changes in Density, Biomass, and Diversity of Estuarine Fishes in Tidal Mangrove Creeks of the Lower Caeté Estuary (Northern Brazilian Coast, East Amazon). Mar. Ecol. Prog. Ser. 2003, 256, 217–228. [Google Scholar] [CrossRef] [Green Version]
- Serafy, J.; Faunce, C.; Lorenz, J. (Jerry) Mangrove Shoreline Fishes of Biscayne Bay, Florida. Bull. Mar. Sci. 2003, 72, 161–180. [Google Scholar]
- Pittman, S.; Mcalpine, C.; Pittman, K. Linking Fish and Prawns to Their Environment: A Hierarchical Landscape Approach. Mar. Ecol. Prog. Ser. 2004, 283, 233–254. [Google Scholar] [CrossRef]
- Blaber, S. Mangroves and Fishes: Issues of Diversity, Dependence, and Dogma. Bull. Mar. Sci. 2007, 80, 457–472. [Google Scholar]
- Gullström, M.; Bodin, M.; Nilsson, P.; Öhman, M. Seagrass Structural Complexity and Landscape Configuration as Determinants of Tropical Fish Assemblage Composition. Mar. Ecol. Prog. Ser. 2008, 363, 241–255. [Google Scholar] [CrossRef] [Green Version]
- Amezcua, F.; Ramirez, M.; Flores-Verdugo, F. Classification and Comparison of Five Estuaries in the Southeast Gulf of California Based on Environmental Variables and Fish Assemblages. Bull. Mar. Sci. 2019, 95, 139–159. [Google Scholar] [CrossRef]
- Rönnbäck, P.; Troell, M.; Kautsky, N.; Primavera, J.H. Distribution Pattern of Shrimps and Fish AmongAvicenniaandRhizophoraMicrohabitats in the Pagbilao Mangroves, Philippines. Estuar. Coast. Shelf Sci. 1999, 48, 223–234. [Google Scholar] [CrossRef]
- Attrill, M.; Rundle, S. Ecotone or Ecocline: Ecological Boundaries in Estuaries. Estuar. Coast. Shelf Sci. 2002, 55, 929–936. [Google Scholar] [CrossRef]
- Rubio-Alvarez, M.; Amezcua-Linares, F.; Yáñez-Arancibia, A. Ecología y Estructura de Las Comunidades de Peces En El Sistema Lagunar Teacapán-Agua Brava, Nayarit, México. An. Inst. Cienc. Mar Limnol. 1986, 13, 185–242. [Google Scholar]
- Amezcua-Linares, F. Generalidades Ictiológicas Del Sistema Lagunar Costero de Huizache-Caimanero, Sinaloa, México. Anales 1977, 4, 1–26. [Google Scholar]
- Amezcua-Linares, F.; Álvarez-Rubio, M.; Yáñez-Arancibia, A. Dinámica y Estructura de La Comunidad de Peces En Un Sistema Ecológico de Manglares de La Costa Del Pacífico de México, Nayarit. An. Inst. Cienc. Mar Limnol. 1987, 14, 221–248. [Google Scholar]
- Flores-Verdugo, F.; González-Farías, F.; Ramírez-Flores, O.; Amezcua-Linares, F.; Yáñez-Arancibia, A.; Alvarez-Rubio, M.; Day, J.W. Mangrove Ecology, Aquatic Primary Productivity, and Fish Community Dynamics in the Teacapán-Agua Brava Lagoon-Estuarine System (Mexican Pacific). Estuaries 1990, 13, 219–230. [Google Scholar] [CrossRef]
- Fischer, W.; Krupp, F.; Schneider, W.; Sommer, C.; Carpenter, K.; Niem, V. Guía FAO Para la Identificación de Especies Para Los Fines de la Pesca, Pacífico Centro-Oriental; FAO: Rome, Italy, 1995; ISBN 92-5-303408-4. [Google Scholar]
- Amezcua-Linares, F. Peces Demersales del Pacífico de México; Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México: Ciudad de México, México, 2009; ISBN 978-970-764-558-5. [Google Scholar]
- Anderson, M.J.; Gorley, R.N.; Clarke, K.R. PRIMER+ for PERMANOVA: Guide to Software and Statistical Methods. 214; Primer-e: Plymouth, UK, 2008. [Google Scholar]
- Anderson, M.J. Permutational Multivariate Analysis of Variance (PERMANOVA). Wiley StatsRef Stat. Ref. Online 2017, 1, 1–15. [Google Scholar] [CrossRef]
- Carson, H.S.; Ulrich, M.; Lowry, D.; Pacunski, R.E.; Sizemore, R. Status of the California Sea Cucumber (Parastichopus Californicus) and Red Sea Urchin (Mesocentrotus Franciscanus) Commercial Dive Fisheries in the San Juan Islands, Washington State, USA. Fish. Res. 2016, 179, 179–190. [Google Scholar] [CrossRef]
- Chao, A. Nonparametric Estimation of the Number of Classes in a Population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Magurran, A.E. Measuring Biological Diversity, 1st ed.; Blakwell Publishing: Oxford, UK, 2004; ISBN 0-632-05633-9. [Google Scholar]
- McArdle, B.H.; Anderson, M.J. Fitting Multivariate Models to Community Data: A Comment on Distance-based Redundancy Analysis. Ecology 2001, 82, 290–297. [Google Scholar] [CrossRef]
- Legendre, P.; Anderson, M.J. Distance-Based Redundancy Analysis: Testing Multispecies Responses in Multifactorial Ecological Experiments. Ecol. Monogr. 1999, 69, 1–24. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; Plymouth Marine Laboratory: Plymouth, UK, 2001; ISBN 1855311402. [Google Scholar]
- Jost, L. Entropy and Diversity. Oikos 2006, 113, 363–375. [Google Scholar] [CrossRef]
- Jost, L. Partitioning Diversity into Independent Alpha and Beta Components. Ecology 2007, 88, 2427–2439. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, A.; Chiu, C.-H.; Hsieh, T.C. Proposing a Resolution to Debates on Diversity Partitioning. Ecology 2012, 93, 2037–2051. [Google Scholar] [CrossRef] [Green Version]
- Hastie, T.; Tibshirani, R. Generalized Additive Models. Stat. Sci. 1986, 1, 297–310. [Google Scholar] [CrossRef]
- Wood, S.N. Package “Mgcv”. R Packag. Version 2015, 1, 729. [Google Scholar]
- Dray, S.; Blanchet, G.; Borcard, D.; Clappe, S.; Guenard, G.; Jombart, T.; Larocque, G.; Legendre, P.; Madi, N.; Wagner, H.H. Package “Adespatial”. R Packag. Version 2018, 82, 3–8. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing Website. Available online: https://www.r-project.org/ (accessed on 16 June 2022).
- Flores-Cárdenas, F.; Hurtado-Oliva, M.Á.; Doyle, T.W.; Nieves-Soto, M.; Díaz-Castro, S.; Manzano-Sarabia, M. Litterfall Production of Mangroves in Huizache-Caimanero Lagoon System, México. J. Coast. Res. 2017, 33, 118–124. [Google Scholar] [CrossRef]
- Muro-Torres, V.M.; Soto-Jiménez, M.F.; Green, L.; Quintero, J.; Amezcua, F. Food Web Structure of a Subtropical Coastal Lagoon. Aquat. Ecol. 2019, 53, 407–430. [Google Scholar] [CrossRef]
- Muro-Torres, V.M.; Amezcua, F.; Soto-Jiménez, M.; Balart, E.F.; Serviere-Zaragoza, E.; Green, L.; Rajnohova, J. Primary Sources and Food Web Structure of a Tropical Wetland with High Density of Mangrove Forest. Water 2020, 12, 3105. [Google Scholar] [CrossRef]
- Sheaves, M. Ecosystem Equivalence and the Ability to Generalise: Insights from Global Consistencies in Mangrove Fish Assemblages. Mar. Ecol. Prog. Ser. 2012, 461, 137–149. [Google Scholar] [CrossRef] [Green Version]
- Igulu, M.M.; Nagelkerken, I.; van der Velde, G.; Mgaya, Y.D. Mangrove Fish Production Is Largely Fuelled by External Food Sources: A Stable Isotope Analysis of Fishes at the Individual, Species, and Community Levels from Across the Globe. Ecosystems 2013, 16, 1336–1352. [Google Scholar] [CrossRef]
- Van Der Heiden, A.M.; Findley, L.T. Lista de Los Peces Marinos Del Sur de Sinaloa, México. An. Inst. Cienc. Mar Limnol. 1988, 15, 209–223. [Google Scholar]
- Mendoza, E.; Castillo-Rivera, M.; Zárate-Hernández, R.; Burgos, S. Seasonal Variations in the Diversity, Abundance, and Composition of Species in an Estuarine Fish Community in the Tropical Eastern Pacific, Mexico. Ichthyol. Res. 2009, 56, 330–339. [Google Scholar] [CrossRef]
- Díaz-Ruiz, S.; Aguirre-León, A.; Cano-Quiroga, E. Evaluación Ecológica de Las Comunidades de Peces En Dos Sistemas Lagunares Estuarinos Del Sur de Chiapas, México. Hidrobiológica 2006, 16, 197–210. [Google Scholar]
- Raz-Guzman, A.; Huidobro, L. Fish Communities in Two Environmentally Different Estuarine Systems of Mexico. J. Fish Biol. 2002, 61, 182–195. [Google Scholar] [CrossRef]
- McLusky, D.; Elliott, M. The Estuarine Ecosystem. Ecology, Threats and Management, 3rd ed.; Oxford University Press: Oxford, UK, 2004; ISBN1 0-19-853091-9 hbk. ISBN2 0-19-852508-7 pbk. [Google Scholar]
- Rashed-Un-Nabi, M.; Al-Mamun, M.A.; Ullah, M.H.; Mustafa, M.G. Temporal and Spatial Distribution of Fish and Shrimp Assemblage in the Bakkhali River Estuary of Bangladesh in Relation to Some Water Quality Parameters. Mar. Biol. Res. 2011, 7, 436–452. [Google Scholar] [CrossRef]
- Dyer, K.R. Estuaries: A Physical Introduction, 2nd ed.; Wiley: Honoken, NJ, USA, 1998; ISBN 9780471974710. [Google Scholar]
- Green-Ruiz, Y.A. Composición y Abundancia de Las Larvas de Peces Durante Un Ciclo Anual, en la Boca de Aguadulce, Laguna de Huizache Caimanero, Sin., México; Instituto Politécnico Nacional: Ciudad de México, México, 1993. [Google Scholar]
- Dantas, D.V.; Barletta, M.; Lima, A.R.A.; de Assis Almeida Ramos, J.; Da Costa, M.F.; Saint-Paul, U. Nursery Habitat Shifts in an Estuarine Ecosystem: Patterns of Use by Sympatric Catfish Species. Estuaries Coasts 2012, 35, 587–602. [Google Scholar] [CrossRef]
- Robertson, D.R.; Allen, G.R. Shorefishes of the Tropical Eastern Pacific: An Information System. Smithson. Trop. Res. Insitute 2015.
- Flores-Verdugo, F.J.; Day, J.W.J.; Briseño-Dueñas, R. Structure, Litter Fall, Decomposition, and Detritus Dynamics of Mangroves in a Méxican Coastal Lagoon with an Ephemeral Inlet. Mar. Ecol. Prog. Ser. 1987, 35, 83–90. [Google Scholar]
- Serrano, D.; Valle-Levinson, A. Effects of River Discharge and the California Current on Pycnocline Depth at the Eastern Entrance to the Gulf of California. Cont. Shelf Res. 2021, 215, 104356. [Google Scholar] [CrossRef]
- Thayer, G.W.; Colby, D.R.; Hettler, W.F. Utilization of the Red Mangrove Prop Root Habitat by Fishes in South Florida. Mar. Ecol. Prog. Ser. 1987, 35, 25–38. [Google Scholar] [CrossRef]
- Vega-Cendejas, M.E.; Hernández De Santillana, M. Fish Community Structure and Dynamics in a Coastal Hypersaline Lagoon: Rio Lagartos, Yucatan, Mexico. Estuar. Coast. Shelf Sci. 2004, 60, 285–299. [Google Scholar] [CrossRef]
- Faunce, C.H.; Serafy, J.E.; Lorenz, J.J. Density-Habitat Relationships of Mangrove Creek Fishes within the Southeastern Saline Everglades (USA), with Reference to Managed Freshwater Releases. Wetl. Ecol. Manag. 2004, 12, 377–394. [Google Scholar] [CrossRef]
- Blaber, S.J.M. Feeding Selectivity of a Guild of Piscivorous Fish in Mangrove Areas of North-West Australia. Mar. Freshw. Res. 1986, 37, 329–336. [Google Scholar] [CrossRef]
- Whitfield, A.K.; Elliott, M. Fishes as Indicators of Environmental and Ecological Changes within Estuaries: A Review of Progress and Some Suggestions for the Future. J. Fish Biol. 2002, 61, 229–250. [Google Scholar] [CrossRef]
- Alongi, D.M. Present State and Future of the World’s Mangrove Forests. Environ. Conserv. 2002, 29, 331–349. [Google Scholar] [CrossRef] [Green Version]
- Barbier, E.; Cox, M. Economic and Demographic Factors Affecting Mangrove Loss in the Coastal Provinces of Thailand, 1979–1996. AMBIO A J. Hum. Environ. 2002, 31, 351–357. [Google Scholar] [CrossRef]
- Thu, P.M.; Populus, J. Status and Changes of Mangrove Forest in Mekong Delta: Case Study in Tra Vinh, Vietnam. Estuar. Coast. Shelf Sci. 2007, 71, 98–109. [Google Scholar] [CrossRef]
- Laffaille, P.; Feunteun, E.; Lefeuvre, J.-C. Composition of Fish Communities in a European Macrotidal Salt Marsh (the Mont Saint-Michel Bay, France). Estuar. Coast. Shelf Sci. 2000, 51, 429–438. [Google Scholar] [CrossRef] [Green Version]

| Family | Species | aH | aT | aTo | bH | bT | bTo | TP |
|---|---|---|---|---|---|---|---|---|
| Sphyrnidae | Sphyrna lewini | 8 | 7 | 15 | 23.2 | 25.9 | 49.1 | 3.9 |
| Narcinidae | Narcine vermiculata | 0 | 5 | 5 | 0 | 0 | 0 | 4.4 |
| Rhinobatidae | Pseudobatos glaucostigma | 0 | 1 | 1 | 0 | 0 | 0 | 4 |
| Pseudobatos leucorhynchus | 1 | 0 | 1 | 3.1 | 0 | 3.1 | 3.6 | |
| Urotrygonidae | Urobatis halleri | 0 | 1 | 1 | 0 | 0 | 0 | 4.6 |
| Urotrygon chilensis | 2 | 27 | 29 | 0 | 0 | 0 | 3.6 | |
| Urotrygon munda | 19 | 0 | 19 | 0.1 | 0 | 0.1 | 4.5 | |
| Urotrygon nana | 0 | 4 | 4 | 0 | 0 | 0 | 3.6 | |
| Mobulidae | Mobula japanica | 1 | 0 | 1 | 26.9 | 0 | 26.9 | 3.4 |
| Myliobatidae | Aetobatus narinari | 0 | 2 | 2 | 0 | 134.1 | 134.1 | 3.02 |
| Rhinoptera steindachneri | 2 | 7 | 9 | 0.3 | 272.5 | 272.8 | 3.6 | |
| Gymnuridae | Gymnura marmorata | 0 | 2 | 2 | 0 | 0 | 0 | 4.2 |
| Elopidae | Elops affinis | 168 | 145 | 313 | 163.5 | 75.7 | 239.2 | 1.9 |
| Albulidae | Albula nemoptera | 1 | 7 | 8 | 0.5 | 2 | 2.5 | 3.9 |
| Albula vulpes | 1 | 70 | 71 | 1.3 | 94.9 | 96.2 | 2 | |
| Ophichthidae | Ophichthus zophochir | 1 | 0 | 1 | 0.4 | 0 | 0.4 | 3.8 |
| Myrichthys aspetocheiros | 0 | 1 | 1 | 0 | 0.2 | 0.2 | 3.5 | |
| Congridae | Ariosoma gilberti | 0 | 1 | 1 | 0 | 0 | 0 | 4.4 |
| Pristigasteridae | Opisthopterus dovii | 1 | 0 | 1 | 0 | 0 | 0 | 4.2 |
| Pliosteostoma lutipinnis | 133 | 24 | 157 | 11.9 | 0.3 | 12.2 | 3.6 | |
| Engraulidae | Anchovia macrolepidota | 41 | 19 | 60 | 3.3 | 1.3 | 4.6 | 3.5 |
| Anchoa lucida | 2 | 1 | 3 | 0.1 | 0 | 0.1 | 3.5 | |
| Anchoa mundeola | 0 | 2 | 2 | 0 | 0 | 0 | 2 | |
| Anchoa nasus | 1 | 0 | 1 | 0.1 | 0 | 0.1 | 3.5 | |
| Anchoa walkeri | 464 | 5 | 469 | 255.6 | 0.1 | 255.7 | 3.5 | |
| Cetengraulis mysticetus | 21 | 184 | 205 | 0.2 | 36.5 | 36.7 | 3.2 | |
| Clupeidae | Dorosoma smithi | 0 | 16 | 16 | 0 | 4 | 4 | 3.4 |
| Lile stolifera | 2591 | 10 | 2601 | 1852.8 | 0 | 1852.8 | 1.8 | |
| Opisthonema libertate | 15 | 214 | 229 | 7.1 | 96.5 | 103.6 | 2.5 | |
| Opisthonema medirastre | 0 | 2 | 2 | 0 | 0.7 | 0.7 | 3.3 | |
| Chanidae | Chanos chanos | 194 | 81 | 275 | 141.1 | 155.8 | 296.9 | 2 |
| Ariidae | Ariopsis guatemalensis | 34 | 25 | 59 | 24.2 | 53.3 | 77.5 | 3.6 |
| Ariopsis seemanni | 34 | 232 | 266 | 41.1 | 271.3 | 312.4 | 3.6 | |
| Bagre panamensis | 56 | 42 | 98 | 51.2 | 79.8 | 131 | 4.4 | |
| Bagre pinnimaculatus | 5 | 3 | 8 | 3.4 | 1.8 | 5.2 | 4.2 | |
| Cathorops fuerthii | 158 | 84 | 242 | 476 | 39.5 | 515.5 | 3.6 | |
| Occidentarius platypogon | 46 | 28 | 74 | 84.6 | 95.8 | 180.4 | 4.1 | |
| Synodontidae | Synodus scituliceps | 0 | 13 | 13 | 0 | 11.8 | 11.8 | 3.6 |
| Fundulidae | Fundulus xenicus | 27 | 0 | 27 | 0 | 0 | 0 | 3.2 |
| Mugilidae | Mugil cephalus | 331 | 266 | 597 | 242.8 | 127.9 | 370.7 | 3.6 |
| Mugil curema | 432 | 261 | 693 | 348.3 | 110.6 | 458.9 | 2 | |
| Hemiramphidae | Hyporhamphus unifasciatus | 2 | 0 | 2 | 2.3 | 0 | 2.3 | 3.8 |
| Belonidae | Strongylura exilis | 3 | 0 | 3 | 39.9 | 0 | 39.9 | 4.5 |
| Tylosurus crocodilus | 0 | 1 | 1 | 0 | 5.2 | 5.2 | 4.4 | |
| Triglidae | Prionotus ruscarius | 0 | 4 | 4 | 0 | 0.6 | 0.6 | 3.4 |
| Cichlidae | Oreochromis mossambicus | 1 | 0 | 1 | 0.9 | 0 | 0.9 | 1.8 |
| Oreochromis sp. | 437 | 44 | 481 | 162.8 | 22.7 | 185.5 | 1.8 | |
| Centropomidae | Centropomus armatus | 1 | 10 | 11 | 0.4 | 1.9 | 2.3 | 4.6 |
| Centropomus medius | 3 | 10 | 13 | 1.9 | 0.1 | 2 | 3.2 | |
| Centropomus nigrescens | 3 | 1 | 4 | 2.6 | 0 | 2.6 | 3.2 | |
| Centropomus robalito | 143 | 67 | 210 | 62.7 | 6.2 | 68.9 | 3.5 | |
| Centropomus viridis | 4 | 17 | 21 | 3.8 | 0.4 | 4.2 | 3.8 | |
| Serranidae | Epinephelus labriformis | 2 | 0 | 2 | 0.4 | 0 | 0.4 | 4 |
| Nematistidae | Nematistius pectoralis | 1 | 48 | 49 | 0.3 | 96.4 | 96.7 | 4 |
| Carangidae | Alectis ciliaris | 0 | 1 | 1 | 0 | 0.7 | 0.7 | 3.8 |
| Carangoides otrynter | 0 | 2 | 2 | 0 | 0.7 | 0.7 | 4.4 | |
| Caranx caballus | 25 | 45 | 70 | 14.9 | 31.6 | 46.5 | 4 | |
| Caranx caninus | 169 | 602 | 771 | 66.5 | 31.7 | 98.2 | 3.5 | |
| Caranx vinctus | 1 | 3 | 4 | 0.6 | 2.1 | 2.7 | 4 | |
| Chloroscombrus orqueta | 177 | 103 | 280 | 39.4 | 27.3 | 66.7 | 3.9 | |
| Hemicaranx leucurus | 4 | 15 | 19 | 2 | 5.5 | 7.5 | 4.4 | |
| Hemicaranx zelotes | 0 | 6 | 6 | 0 | 3.4 | 3.4 | 4.1 | |
| Oligoplites altus | 23 | 67 | 90 | 8.2 | 69.9 | 78.1 | 3.3 | |
| Oligoplites refulgens | 5 | 3 | 8 | 1.2 | 1.5 | 2.7 | 4.3 | |
| Oligoplites saurus | 5 | 2 | 7 | 4.1 | 0.5 | 4.6 | 3.4 | |
| Selar crumenophthalmus | 7 | 1 | 8 | 2.2 | 0.6 | 2.8 | 4.1 | |
| Selene brevoortii | 1 | 9 | 10 | 0.1 | 2.4 | 2.5 | 3.6 | |
| Selene orstedii | 0 | 1 | 1 | 0 | 0.3 | 0.3 | 3 | |
| Selene peruviana | 21 | 54 | 75 | 0.9 | 9.7 | 10.6 | 3.6 | |
| Trachinotus kennedyi | 1 | 3 | 4 | 0.5 | 1.4 | 1.9 | 3 | |
| Trachinotus paitensis | 2 | 36 | 38 | 2.1 | 39.6 | 41.7 | 2.8 | |
| Trachinotus rhodopus | 0 | 5 | 5 | 0 | 7.8 | 7.8 | 4 | |
| Lutjanidae | Hoplopagrus guentherii | 0 | 1 | 1 | 0 | 1.2 | 1.2 | 3.8 |
| Lutjanus argentiventris | 20 | 17 | 37 | 1.9 | 13.9 | 15.8 | 3.7 | |
| Lutjanus guttatus | 0 | 11 | 11 | 0 | 0 | 0 | 3.8 | |
| Lutjanus novemfasciatus | 5 | 3 | 8 | 2.4 | 0.1 | 2.5 | 3.5 | |
| Gerreidae | Deckertichthys aureolus | 3 | 1 | 4 | 0.9 | 0.2 | 1.1 | 3.6 |
| Diapterus peruvianus | 151 | 503 | 654 | 26.9 | 87.9 | 114.8 | 2.2 | |
| Eucinostomus argenteus | 14 | 7 | 21 | 9.9 | 0 | 9.9 | 3.6 | |
| Eucinostomus currani | 23 | 97 | 120 | 3.7 | 51.2 | 54.9 | 3.6 | |
| Eucinostomus entomelas | 53 | 24 | 77 | 17.5 | 3.5 | 21 | 3.5 | |
| Eucinostomus gracilis | 0 | 13 | 13 | 0 | 0.1 | 0.1 | 3.8 | |
| Eugerres axillaris | 40 | 42 | 82 | 9.7 | 7.2 | 16.9 | 1.7 | |
| Gerres cinereus | 22 | 53 | 75 | 6.3 | 12.6 | 18.9 | 3.8 | |
| Haemulidae | Conodon serrifer | 16 | 0 | 16 | 5.6 | 0 | 5.6 | 3.8 |
| Pomadasys branickii | 2 | 0 | 2 | 0.8 | 0 | 0.8 | 3.9 | |
| Rhencus macracanthus | 74 | 95 | 169 | 32.9 | 101.4 | 134.3 | 3.2 | |
| Rhencus panamensis | 28 | 42 | 70 | 12.3 | 32.6 | 44.9 | 3.8 | |
| Brachygenys californiensis | 0 | 1 | 1 | 0 | 0 | 0 | 3.5 | |
| Orthopristis chalceus | 1 | 1 | 2 | 1 | 0 | 1 | 3.6 | |
| Haemulopsis axillaris | 31 | 0 | 31 | 17.4 | 0 | 17.4 | 3.8 | |
| Haemulopsis leuciscus | 7 | 5 | 12 | 4.8 | 4.2 | 9 | 3.8 | |
| Haemulopsis nitidus | 12 | 49 | 61 | 5.2 | 7.4 | 12.6 | 3.7 | |
| Polynemidae | Polydactylus approximans | 18 | 11 | 29 | 43.2 | 11.5 | 54.7 | 4.1 |
| Polydactylus opercularis | 1 | 0 | 1 | 0.3 | 0 | 0.3 | 3.3 | |
| Sciaenidae | Bairdiella icistia | 2 | 0 | 2 | 0.8 | 0 | 0.8 | 3.9 |
| Isopisthus remifer | 7 | 70 | 77 | 1.7 | 0.2 | 1.9 | 4.4 | |
| Cynoscion othonopterus | 1 | 0 | 1 | 0.9 | 0 | 0.9 | 3.9 | |
| Cynoscion reticulatus | 30 | 11 | 41 | 8.7 | 1.6 | 10.3 | 4.7 | |
| Cynoscion squamipinnis | 11 | 0 | 11 | 6.9 | 0 | 6.9 | 3.9 | |
| Cynoscion stolzmanni | 24 | 3 | 27 | 8.3 | 3.1 | 11.4 | 4.5 | |
| Cynoscion xanthulus | 192 | 8 | 200 | 138.8 | 5.1 | 143.9 | 4.3 | |
| Larimus acclivis | 2 | 0 | 2 | 0 | 0 | 0 | 4 | |
| Larimus effulgens | 132 | 13 | 145 | 9.3 | 3.3 | 12.6 | 3.7 | |
| Stellifer fuerthii | 146 | 79 | 225 | 1.2 | 0.8 | 2 | 3.7 | |
| Stellifer illecebrosus | 86 | 0 | 86 | 1.5 | 0 | 1.5 | 4.1 | |
| Stellifer sp. | 0 | 3 | 3 | 0 | 0 | 0 | ||
| Umbrina xanti | 3 | 1 | 4 | 2.1 | 3.5 | 5.6 | 3.6 | |
| Menticirrhus elongatus | 5 | 1 | 6 | 3.3 | 0 | 3.3 | 3.5 | |
| Menticirrhus nasus | 4 | 5 | 9 | 2 | 15.1 | 17.1 | 3.6 | |
| Micropogonias altipinnis | 27 | 0 | 27 | 10.4 | 0 | 10.4 | 3.7 | |
| Micropogonias ectenes | 9 | 5 | 14 | 5.7 | 2.1 | 7.8 | 3.8 | |
| Micropogonias megalops | 0 | 6 | 6 | 0 | 0 | 0 | 3.4 | |
| Paralonchurus goodei | 40 | 12 | 52 | 1.5 | 0 | 1.5 | 3.9 | |
| Mullidae | Pseudupeneus grandisquamis | 0 | 5 | 5 | 0 | 0 | 0 | 3.8 |
| Chaetodontidae | Chaetodon humeralis | 0 | 1 | 1 | 0 | 0.4 | 0.4 | 2.7 |
| Pomacentridae | Abudefduf troschelii | 0 | 1 | 1 | 0 | 0 | 0 | 3.9 |
| Labridae | Bodianus diplotaenia | 2 | 0 | 2 | 1.3 | 0 | 1.3 | 3.4 |
| Eleotridae | Gobiomorus maculatus | 33 | 12 | 45 | 4.2 | 0.9 | 5.1 | 1.7 |
| Dormitator latifrons | 112 | 120 | 232 | 34.2 | 1.7 | 35.9 | 1.9 | |
| Eleotris picta | 1 | 0 | 1 | 1.6 | 0 | 1.6 | 3.9 | |
| Gobiidae | Microgobius sp. | 1 | 1 | 2 | 0 | 0 | 0 | |
| Gobionellus microdon | 16 | 3 | 19 | 0.9 | 0.1 | 1 | 1.8 | |
| Ephippidae | Chaetodipterus zonatus | 4 | 23 | 27 | 2 | 6 | 8 | 2.5 |
| Sphyraenidae | Sphyraena ensis | 1 | 0 | 1 | 3.7 | 0 | 3.7 | 3.9 |
| Trichiuridae | Trichiurus lepturus | 2 | 0 | 2 | 0 | 0 | 0 | 3.9 |
| Scombridae | Scomber japonicus | 4 | 3 | 7 | 4.7 | 2.1 | 6.8 | 3.4 |
| Scomberomorus sierra | 18 | 1 | 19 | 19.3 | 5.5 | 24.8 | 3.8 | |
| Katsuwonus pelamis | 0 | 3 | 3 | 0 | 52.7 | 52.7 | 4.3 | |
| Stromaetidae | Peprilus medius | 20 | 10 | 30 | 7.2 | 4.6 | 11.8 | 3.9 |
| Peprilus snyderi | 10 | 24 | 34 | 3.5 | 22.3 | 25.8 | 4.3 | |
| Paralichthydae | Paralichthys woolmani | 1 | 0 | 1 | 3.4 | 0 | 3.4 | 4.5 |
| Syacium ovale | 1 | 6 | 7 | 0 | 0 | 0 | 3.5 | |
| Cyclopsetta panamensis | 5 | 7 | 12 | 0 | 2.8 | 2.8 | 3.7 | |
| Cyclopsetta querna | 8 | 5 | 13 | 1.5 | 3.8 | 5.3 | 3.7 | |
| Citharichthys gilberti | 2 | 0 | 2 | 0.1 | 0 | 0.1 | 4.1 | |
| Etropus crossotus | 6 | 0 | 6 | 0.2 | 0 | 0.2 | 4 | |
| Achiridae | Achirus mazatlanus | 70 | 134 | 204 | 6 | 0.3 | 6.3 | 3.7 |
| Trinectes fonsecensis | 4 | 3 | 7 | 0 | 0 | 0 | 3.8 | |
| Cynoglossidae | Symphurus elongatus | 93 | 4 | 97 | 0.1 | 0 | 0.1 | 4.1 |
| Ballistidae | Balistes polylepis | 1 | 3 | 4 | 0 | 0.1 | 0.1 | 3.4 |
| Tetraodontidae | Sphoeroides annulatus | 0 | 2 | 2 | 0 | 0.1 | 0.1 | 3.8 |
| Sphoeroides lobatus | 0 | 4 | 4 | 0 | 0 | 0 | 4 |
| Variable | SS (Trace) | Pseudo-F | p | Prop. |
|---|---|---|---|---|
| Temperature | 3815.5 | 1.07 | 0.34 | 0.02 |
| Salinity * | 10,101.0 | 2.92 | 0.00 | 0.05 |
| Mangrove height * | 10,311.0 | 2.98 | 0.00 | 0.05 |
| Mangrove area * | 7993.2 | 2.28 | 0.00 | 0.04 |
| Agriculture area | 5557.6 | 1.57 | 0.14 | 0.03 |
| Depth * | 12,166.0 | 5.03 | 0.00 | 0.16 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muro-Torres, V.; Amezcua, F.; Ramírez-Ortiz, G.; Flores-de-Santiago, F.; Amezcua-Linares, F.; Hernández, Y. Assessing the Spatiotemporal Relationship between Coastal Habitats and Fish Assemblages at Two Neotropical Estuaries of the Mexican Pacific. Diversity 2022, 14, 619. https://doi.org/10.3390/d14080619
Muro-Torres V, Amezcua F, Ramírez-Ortiz G, Flores-de-Santiago F, Amezcua-Linares F, Hernández Y. Assessing the Spatiotemporal Relationship between Coastal Habitats and Fish Assemblages at Two Neotropical Estuaries of the Mexican Pacific. Diversity. 2022; 14(8):619. https://doi.org/10.3390/d14080619
Chicago/Turabian StyleMuro-Torres, Victor, Felipe Amezcua, Georgina Ramírez-Ortiz, Francisco Flores-de-Santiago, Felipe Amezcua-Linares, and Yareli Hernández. 2022. "Assessing the Spatiotemporal Relationship between Coastal Habitats and Fish Assemblages at Two Neotropical Estuaries of the Mexican Pacific" Diversity 14, no. 8: 619. https://doi.org/10.3390/d14080619
APA StyleMuro-Torres, V., Amezcua, F., Ramírez-Ortiz, G., Flores-de-Santiago, F., Amezcua-Linares, F., & Hernández, Y. (2022). Assessing the Spatiotemporal Relationship between Coastal Habitats and Fish Assemblages at Two Neotropical Estuaries of the Mexican Pacific. Diversity, 14(8), 619. https://doi.org/10.3390/d14080619









