Winter Territoriality of the American Redstart in Oil Palm Plantations
Abstract
1. Introduction
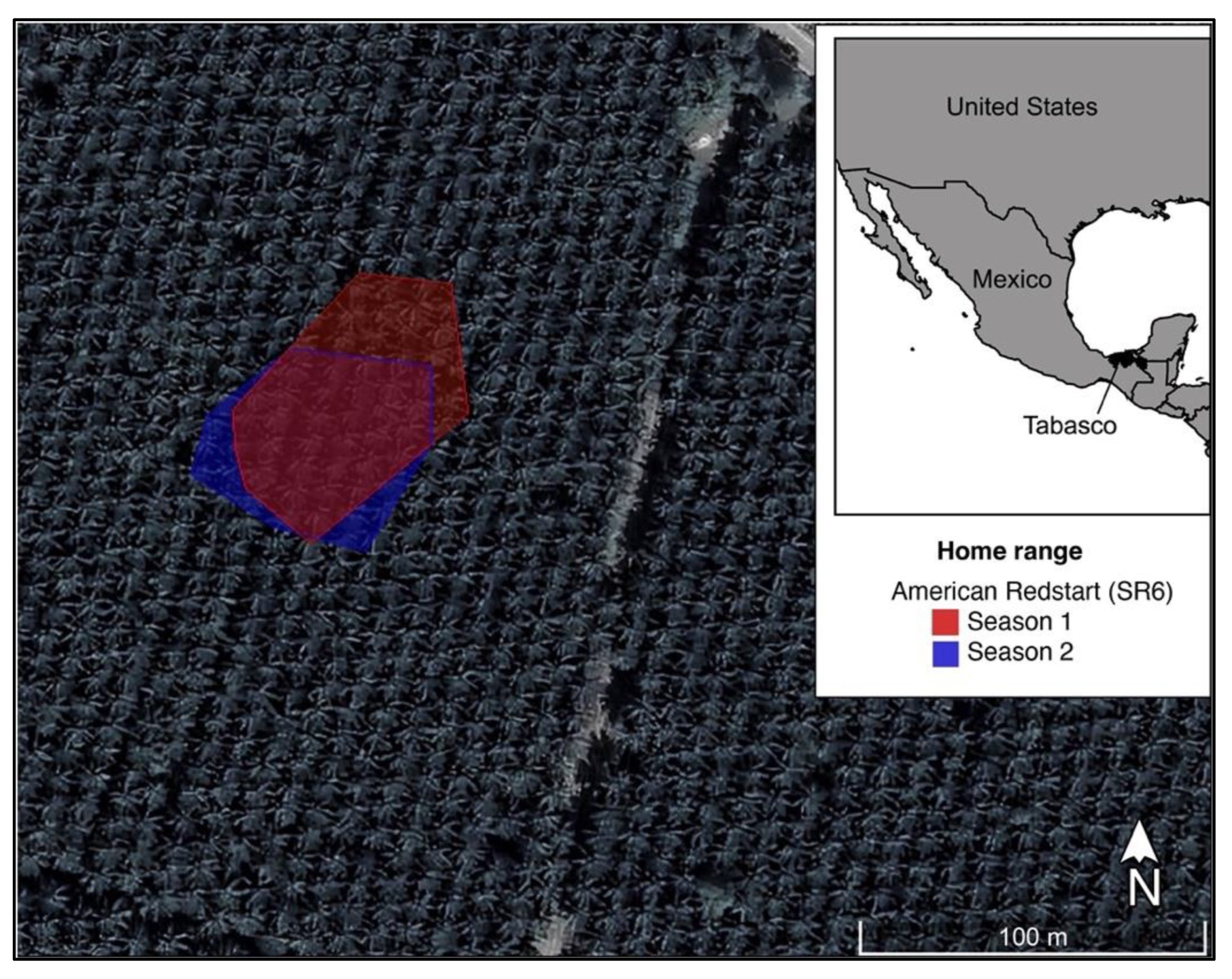
2. Materials and Methods
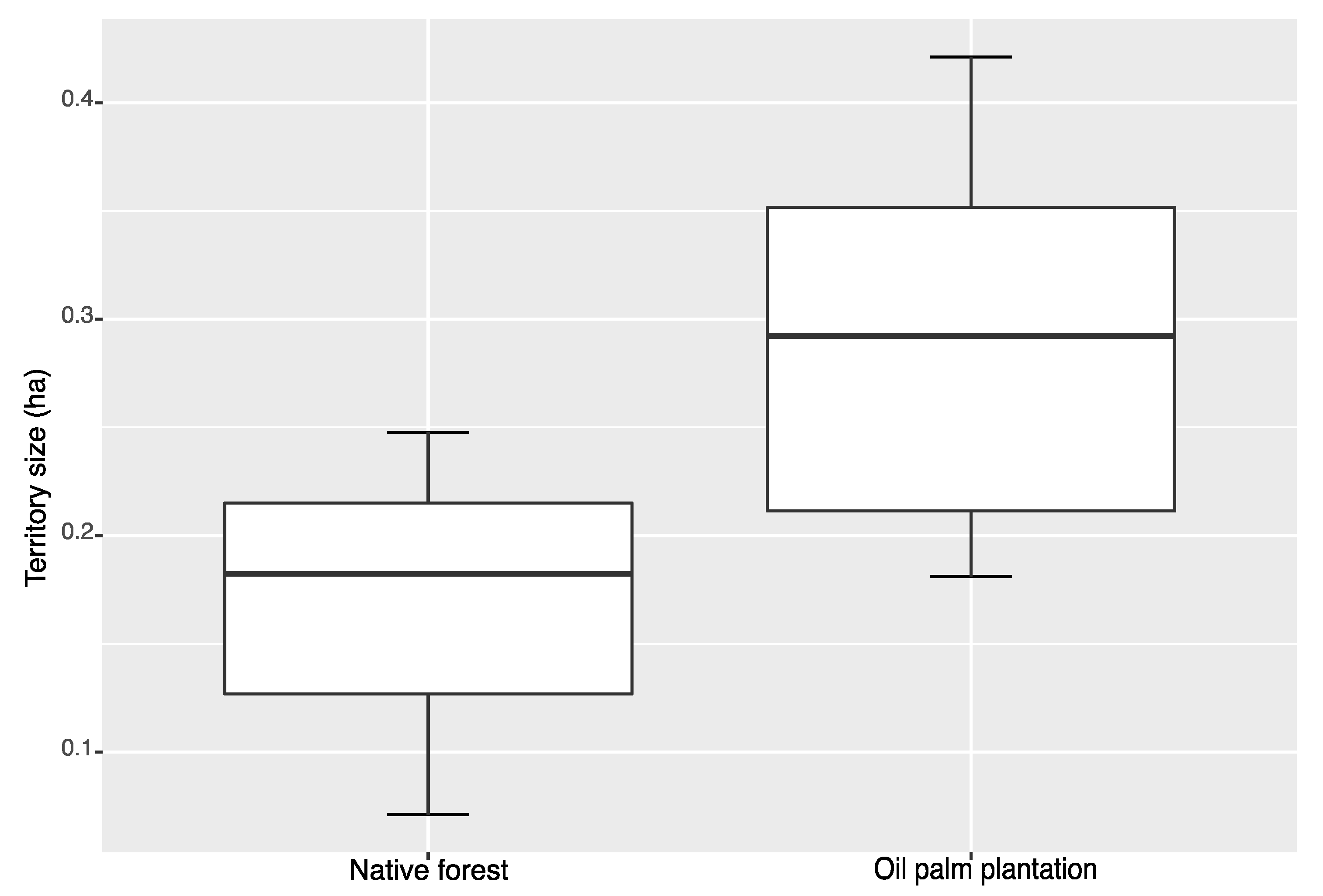
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gauthreaux, S.A.J. The Ecology and Evolution of Avian Migration Systems. In Avian Biology; Farmer, D.S., King, J.R., Eds.; Academic Press: New York, NY, USA, 1982; pp. 93–167. [Google Scholar]
- Greenwood, P.J.; Harvey, P.H. The Natal and Breeding Dispersal of Birds. Annu. Rev. Ecol. Syst. 1982, 13, 1–21. [Google Scholar] [CrossRef]
- Rappole, J.H.; Warner, D.W. Ecological Aspects of Migrant Bird Behavior in Veracruz, Mexico. In Migrant Birds in the Neotropics: Ecology, Behavior, Distribution, and Conservation; Smithsonian Institution Press: Washington, DC, USA, 1980; pp. 353–393. [Google Scholar]
- Mcneil, R. Winter Resident Repeats and Returns of Austral and Boreal Migrant Birds Banded in Venezuela. J. Field Ornithol. 1982, 53, 125–132. [Google Scholar]
- Greenberg, R. The Winter Exploitation Systems of Bay-Breasted and Chestnut-Sided Warblers in Panama; University of California Publications in Zoology: Berkeley, CA, USA, 1984; pp. 1–107. [Google Scholar]
- Albert, S.; Wolfe, J.D.; Kellerman, J.; Sherry, T.; Stutchbury, B.J.M.; Bayly, N.J.; Ruiz-Sánchez, A. Habitat Ecology of Nearctic–Neotropical Migratory Landbirds on the Nonbreeding Grounds. Condor 2020, 122, duaa055. [Google Scholar] [CrossRef]
- Marra, P.P.; Hobson, K.A.; Holmes, R.T.; Dallos, P.; Geister, C.D.; David, C. Terms and Conditions Linking Winter and Summer Events in a Migratory Bird by Using Stable-Carbon Isotopes. Science 1998, 282, 1884–1886. [Google Scholar] [CrossRef] [PubMed]
- Norris, D.R.; Marra, P.P.; Kyser, T.K.; Sherry, T.W.; Ratcliffe, L.M. Tropical Winter Habitat Limits Reproductive Success on the Temperate Breeding Grounds in a Migratory Bird. Proc. R. Soc. B Biol. Sci. 2004, 271, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Marra, P.; Sherry, T.W.; Holmes, R.T. Territorial Exclusion by a Long-Distance Migrant Warbler in Jamaica: A Removal Experiment with American Redstarts (Setophaga ruticilla). Auk 1993, 110, 565–572. [Google Scholar] [CrossRef]
- Marra, P.P. The Role of Behavioral Dominance in Structuring Patterns of Habitat Occupancy in a Migrant Bird during the Nonbreeding Season. Behav. Ecol. 2000, 11, 299–308. [Google Scholar] [CrossRef]
- Studds, C.E.; Marra, P.P. Nonbreeding Habitat Occupancy and Population Processes: An Upgrade Experiment with a Migratory Bird. Ecology 2005, 86, 2380–2385. [Google Scholar] [CrossRef]
- Peele, A.M.; Marra, P.M.; Sillett, T.S.; Sherry, T.W. Combining Survey Methods to Estimate Abundance and Transience of Migratory Birds among Tropical Nonbreeding Habitats. Auk 2015, 132, 926–937. [Google Scholar] [CrossRef]
- Ornat, A.L.; Greenberg, R. Sexual Segregation by Habitat in Migratory Warblers in Quintana Roo, Mexico. Auk 1990, 107, 539–543. [Google Scholar]
- Bakermans, M.H.; Vitz, A.C.; Rodewald, A.D.; Rengifo, C.G. Migratory Songbird Use of Shade Coffee in the Venezuelan Andes with Implications for Conservation of Cerulean Warbler. Biol. Conserv. 2009, 142, 2476–2483. [Google Scholar] [CrossRef]
- Oliveira, S.L.; Flaspohler, D.J.; Knowlton, J.L.; Wolfe, J.D. Do Oil Palm Plantations Provide Quality Habitat for Migratory Birds? A Case Study from Mexico. Ecol. Indic. 2022, 139, 108964. [Google Scholar] [CrossRef]
- Newton, I. The Migration Ecology of Birds; Academy Press: Oxford, UK, 2008; ISBN 978-0-12-517367-4. [Google Scholar]
- Fretwell, S.D.; Lucas, H.L., Jr. On Territorial Behavior and Other Factors Influencing Habitat Distribution in Birds. Acta Biotheor. 1969, 19, 16–36. [Google Scholar] [CrossRef]
- Van Horne, V. Density as a Misleading Indicator of Habitat Quality. J. Wildl. Manag. 1983, 47, 893. [Google Scholar] [CrossRef]
- Brown, D.R.; Long, J.A. What Is a Winter Floater? Causes, Consequences, and Implications for Habitat Selection. Condor 2007, 109, 548–565. [Google Scholar] [CrossRef]
- Wunderle, J.M.; Latta, S.C. Winter Site Fidelity of Nearctic Migrants in Shade Coffee Plantations of Different Sizes in the Dominican Republic. Auk 2000, 117, 596–614. [Google Scholar] [CrossRef]
- Peele, A.M. Population Regulation of a Long-Distance Migratory Bird Population in the Non-Breeding Season: A Test of Buffer and Crowding Effects. Ph.D. Thesis, Tulane University, New Orleans, LA, USA, 2015. [Google Scholar]
- Lefebvre, G.; Poulin, B.; McNeil, R. Spatial and Social Behaviour of Nearctic Warblers Wintering in Venezuelan Mangroves. Can. J. Zool. 1994, 72, 757–764. [Google Scholar] [CrossRef]
- Toms, J.D. Non-Breeding Competition between Migrant American Redstarts (Setophaga ruticilla) and Resident Adelaide’s Warblers (Dendroica adelaidae) in the Guanica Biosphere Reserve, Southwest Puerto Rico. Ph.D. Thesis, University of Missouri, Columbia, MO, USA, 2011. [Google Scholar]
- Toms, J.D. Linking Behavior and Community Ecology: Interspecific Aggression Provides Evidence for Competition Between a Migrant and Resident Warbler. Ethology 2013, 119, 1057–1066. [Google Scholar] [CrossRef]
- Stenger, J. Food Habits and Available Food of Ovenbirds in Relation to Territory Size. Auk 1958, 75, 335–347. [Google Scholar]
- Brown, J.L. The Evolution of Diversity in Avian Territorial Systems. Wilson Bull. 1964, 76, 160–169. [Google Scholar]
- Wilson, E.O. Sociobiology; Harvard University Press: Cambridge, MA, USA, 1975. [Google Scholar]
- Sherry, T.W.; Holmes, R.T. Winter Habitat Quality, Population Limitation, and Conservation of Neotropical Nearctic Migrant Birds. Ecology 1996, 77, 36–48. [Google Scholar] [CrossRef]
- Sherry, T.W.; Holmes, R.T.; Pyle, P.; Patten, M.A. American Redstart (Setophaga ruticilla). In Birds of the World; Billerman, S.M., Keeney, B.K., Rodewald, P.G., Schulenberg, T.S., Eds.; Cornell Lab of Ornithology: Ithaca, NJ, USA, 2020. [Google Scholar]
- Reid, J.L.; Harris, J.B.C.; Martin, L.J.; Barnett, J.R.; Zahawi, R.A. Distribution and Abundance of Nearctic–Neotropical Songbird Migrants in a Forest Restoration Site in Southern Costa Rica. J. Trop. Ecol. 2008, 24, 685–688. [Google Scholar] [CrossRef][Green Version]
- Parrish, J.D.; Sherry, T.W. Sexual Habitat Segregation by American Redstarts Wintering in Jamaica: Importance of Resource Seasonality. Auk 1994, 111, 38–49. [Google Scholar] [CrossRef]
- Greenberg, R.; Bichier, P.; Angón, A.C. The Conservation Value for Birds of Cacao Plantations with Diverse Planted Shade in Tabasco, Mexico. Anim. Conserv. 2000, 3, 105–112. [Google Scholar] [CrossRef]
- Moo-Culebro, L.Y. Avifauna Asociada a Plantaciones de Palma Africana, Acahual y Matriz Circundante de la Sierra Tabasqueña. Master’s Thesis, Universidad Juáres Autônoma de Tabasco, Villahermosa, Mexico, 2018. [Google Scholar]
- Oliveira, S.L.; Flaspohler, D.J.; Knowlton, J.L.; Webster, C.R.; Wolfe, J.D. Migratory Bird Community Structure in Oil Palm (Elaies guineensis) Plantations and Native Forest Fragments in Southern Mexico. J. Field Ornithol. 2021, 92, 1–17. [Google Scholar] [CrossRef]
- Foster, W.A.; Snaddon, J.L.; Turner, E.C.; Fayle, T.M.; Cockerill, T.D.; Farnon Ellwood, M.D.; Broad, G.R.; Chung, A.Y.C.; Eggleton, P.; Khen, C.V.; et al. Establishing the Evidence Base for Maintaining Biodiversity and Ecosystem Function in the Oil Palm Landscapes of South East Asia. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 3277–3291. [Google Scholar] [CrossRef]
- Hostetler, J.A.; Sillett, T.S.; Marra, P.P. Full-Annual-Cycle Population Models for Migratory Birds. Auk 2015, 132, 433–449. [Google Scholar] [CrossRef]
- Tudela, F. La Modernización Forzada del Trópico: El Caso de Tabasco; Colegio de México: Mexico City, Mexico, 1992. [Google Scholar]
- Arriaga-Weiss, S.L.; Calmé, S.; Kampichler, C. Bird Communities in Rainforest Fragments: Guild Responses to Habitat Variables in Tabasco, Mexico. Biodivers. Conserv. 2008, 17, 173–190. [Google Scholar] [CrossRef]
- INEGI–Instituto Nacional de Estadística y Geografía. Anuario Estadístico y Geográfico de Tabasco; INEGI: Aguascalientes, Mexico, 2015. [Google Scholar]
- Barg, J.J.; Jones, J.; Robertson, R.J. Describing Breeding Territories of Migratory Passerines: Suggestions for Sampling, Choice of Estimator, and Delineation of Core Areas. J. Anim. Ecol. 2005, 74, 139–149. [Google Scholar] [CrossRef]
- Cooper, N.W.; Sherry, T.W.; Marra, P.P. Experimental Reduction of Winter Food Decreases Body Condition and Delays Migration in a Long-Distance Migratory Bird. Ecology 2015, 96, 1933–1942. [Google Scholar] [CrossRef]
- Calenge, C. The Package “Adehabitat” for the R Software: A Tool for the Analysis of Space and Habitat Use by Animals. Ecol. Model. 2006, 197, 516–519. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2020. [Google Scholar]
- Hansteen, T.L.; Andreassen, H.P.; Ims, R.A. Effects of Spatiotemporal Scale on Autocorrelation and Home Range Estimators. J. Wildl. Manag. 1997, 61, 280. [Google Scholar] [CrossRef]
- Hogg, R.V.; Craig, A.T. Introduction to Mathematical Statistics, 4th ed.; Macmillan: New York, NY, USA, 1978; ISBN 978-0-02-355710-1. [Google Scholar]
- Wolfe, J.D.; Ryder, T.B.; Pyle, P. Using Molt Cycles to Categorize the Age of Tropical Birds: An Integrative New System. J. Field Ornithol. 2010, 81, 186–194. [Google Scholar] [CrossRef]
- Studds, C.E.; Marra, P.P. Linking Fluctuations in Rainfall to Nonbreeding Season Performance in a Long-Distance Migratory Bird, Setophaga ruticilla. Clim. Res. 2007, 35, 115–122. [Google Scholar] [CrossRef]
- Johnson, M.D.; Sherry, T.W.; Holmes, R.T.; Marra, P.P. Assessing Habitat Quality for a Migratory Songbird Wintering in Natural and Agricultural Habitats. Conserv. Biol. 2006, 20, 1433–1444. [Google Scholar] [CrossRef]
- Bennett, R.; Leuenberger, W.; Leja, B.B.B.; Cáceres, A.S.; Johnson, K.; Larkin, J. Conservation of Neotropical Migratory Birds in Tropical Hardwood and Oil Palm Plantations. PLoS ONE 2018, 13, e0210293. [Google Scholar] [CrossRef]
- Holmes, R.T.; Sherry, T.W.; Reitsma, L. Population Structure, Territoriality and Overwinter Survival of Two Migrant Warbler Species in Jamaica. Condor 1989, 91, 545. [Google Scholar] [CrossRef]
- Cooper, N.W.; Sherry, T.W.; Marra, P.P. Modeling Three-Dimensional Space Use and Overlap in Birds. Auk 2014, 131, 681–693. [Google Scholar] [CrossRef]
- Smith, T.M.; Shugart, H.H. Territory Size Variation in the Ovenbird: The Role of Habitat Structure. Ecology 1987, 68, 695–704. [Google Scholar] [CrossRef]
- Stanley, C.Q.; Dudash, M.R.; Ryder, T.B.; Shriver, W.G.; Marra, P.P. Variable Tropical Moisture and Food Availability Underlie Mixed Winter Space-Use Strategies in a Migratory Songbird. Proc. R. Soc. B 2021, 288, 20211220. [Google Scholar] [CrossRef]
- Ruiz-Sánchez, A.; Renton, K.; Rueda-Hernández, R. Winter Habitat Disturbance Influences Density and Territory Size of a Neotropical Migratory Warbler. J. Ornithol. 2017, 158, 63–73. [Google Scholar] [CrossRef]
- Marshall, M.R.; Cooper, R.J. Territory Size of a Migratory Songbird in Response to Caterpillar Density and Foliage Structure. Ecology 2004, 85, 432–445. [Google Scholar] [CrossRef]
- Brown, D.R.; Sherry, T.W. Alternative Strategies of Space Use and Response to Resource Change in a Wintering Migrant Songbird. Behav. Ecol. 2008, 19, 1314–1325. [Google Scholar] [CrossRef]
- Holmes, R.T.; Sherry, T.W. Site Fidelity of Migratory Warblers in Temperate Breeding and Neotropical Wintering Areas: Implications for Population Dynamics, Habitat Selection, and Conservation. In Ecology and Conservation of Neotropical Migrant Landbirds; Smithsonian Institute Press: Washington, DC, USA, 1992; pp. 563–575. [Google Scholar]
- Faaborg, J.R.; Arendt, W.J. Population Sizes and Philopatry of Winter Resident Warblers in Puerto Rico. J. Field Ornithol. 1984, 14, 151–166. [Google Scholar]
- Stamps, J.A.; Krishnan, V.V.; Reid, M.L. Search Costs and Habitat Selection by Dispersers. Ecology 2005, 86, 510–518. [Google Scholar] [CrossRef]
- Johnson, M.D.; Sherry, T.W. Effects of Food Availability on the Distribution of Migratory Warblers among Habitats in Jamaica. J. Anim. Ecol. 2001, 70, 546–560. [Google Scholar] [CrossRef]
- Latta, S.C.; Faaborg, J.R. Demographic and Population Responses of Cape May. America 2002, 83, 2502–2515. [Google Scholar]
- Wunderle, J.M.; Lebow, P.K.; White, J.D.; Currie, D. Sex and Age Differences in Site Fidelity, Food Resource Tracking, and Body Condition of Wintering Kirtland’s Warblers (Setophaga kirtlandii) in the Bahamas. Ornithol. Monogr. 2014, 80, 1–62. [Google Scholar] [CrossRef]
- Rosenberg, K.V.; Kennedy, J.A.; Dettmers, R.; Ford, R.P.; Reynolds, D.; Alexander, J.D.; Beardmore, C.J.; Blancher, P.J.; Bogart, R.E.; Butcher, G.S.; et al. Partners In Flight Landbird Conservation Plan: 2016 Revision for Canada and Continental United States. Partn. Flight Sci. Comm. 2016, 119, 96–103. [Google Scholar]
- Terborgh, J. Where Have All the Birds Gone? Essays on the Biology and Conservation of Birds That Migrate to the American Tropics; Princeton University Press: Princeton, NJ, USA, 1989. [Google Scholar]
- Marra, P.; Cohen, E.B.; Loss, S.R.; Rutter, J.E.; Tonra, C.M. A Call for Full Annual Cycle Research in Animal Ecology. Biol. Lett. 2015, 11, 20150552. [Google Scholar] [CrossRef]
- Rushing, C.S.; Marra, P.P.; Dudash, M.R. Winter Habitat Quality but Not Long-Distance Dispersal—Influences Apparent Reproductive Success in a Migratory Bird. Ecology 2016, 97, 1218–1227. [Google Scholar] [CrossRef]

| Bird | Territory Size (ha) | Vegetation | Sex | Age |
|---|---|---|---|---|
| SR1 | 0.421 | Oil palm | Male | Adult |
| SR2 | 0.181 | Oil palm | Male | Young |
| SR3 | 0.071 | Native forest | Male | Adult |
| SR4 | 0.248 | Native forest | Male | Adult |
| SR5 | 0.182 | Native forest | Male | Adult |
| SR6 | 0.344 | Oil palm | Male | Young |
| SR7 | 0.360 | Oil palm | Male | Young |
| SR8 | 0.241 | Oil palm | Male | Adult |
| SR6 | 0.292 | Oil palm | Male | Adult |
| SR14 | 0.182 | Oil palm | Male | Young |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, S.L.; Flaspohler, D.J.; Wolfe, J.D. Winter Territoriality of the American Redstart in Oil Palm Plantations. Diversity 2022, 14, 1079. https://doi.org/10.3390/d14121079
Oliveira SL, Flaspohler DJ, Wolfe JD. Winter Territoriality of the American Redstart in Oil Palm Plantations. Diversity. 2022; 14(12):1079. https://doi.org/10.3390/d14121079
Chicago/Turabian StyleOliveira, Samuel L., David J. Flaspohler, and Jared D. Wolfe. 2022. "Winter Territoriality of the American Redstart in Oil Palm Plantations" Diversity 14, no. 12: 1079. https://doi.org/10.3390/d14121079
APA StyleOliveira, S. L., Flaspohler, D. J., & Wolfe, J. D. (2022). Winter Territoriality of the American Redstart in Oil Palm Plantations. Diversity, 14(12), 1079. https://doi.org/10.3390/d14121079





